Abstract
A substantial proportion of patients with autism spectrum disorder (ASD) display hyperactivity as a comorbid symptom. Exposure to valproic acid (VPA) during pregnancy produces ASD-like core behavioral phenotypes as well as hyperactivity in offspring both in human and experimental animals, which makes it a plausible model to study ASD-related neurobiological processes. In this study, we examined the effects of two of currently available attention defecit hyperactivity disorder (ADHD) medications, methylphenidate (MPH) and atomoxetine (ATX) targeting dopamine and norepinephrine transporters (DAT and NET), respectively, on hyperactive behavior of prenatally VPA-exposed rat offspring. In the prefrontal cortex of VPA exposed rat offspring, both mRNA and protein expression of DAT was increased as compared with control. VPA function as a histone deacetylase inhibitor (HDACi) and chromatin immunoprecipitation experiments demonstrated that the acetylation of histone bound to DAT gene promoter was increased in VPA-exposed rat offspring suggesting epigenetic mechanism of DAT regulation. Similarly, the expression of NET was increased, possibly via increased histone acetylation in prefrontal cortex of VPA-exposed rat offspring. When we treated the VPA-exposed rat offspring with ATX, a NET selective inhibitor, hyperactivity was reversed to control level. In contrast, MPH that inhibits both DAT and NET, did not produce inhibitory effects against hyperactivity. The results suggest that NET abnormalities may underlie the hyperactive phenotype in VPA animal model of ASD. Profiling the pharmacological responsiveness as well as investigating underlying mechanism in multiple models of ASD and ADHD may provide more insights into the neurobiological correlates regulating the behavioral abnormalities.
Keywords: Valproic acid, Autism, Hyperactivity, Norepinephrine transporter, Atomoxetine
INTRODUCTION
ASD is a neurodevelopmental disorder, characterized by impaired social interaction and communication as well as restricted, repetitive and stereotyped behaviors (Wilkins and Matson, 2009). Recently, it has been noted that individuals with ASD often have inattention and hyperactivity as comorbid symptoms. Leyfer and colleagues assessed 109 children diagnosed with ASD and found that 30.6% children had ADHD (Leyfer et al., 2006). Also, Simonoff and coauthors examined 112 children with ASD, 28.2% of children with ASD had comorbid ADHD (Simonoff et al., 2008). Using DSM-IV based rating scales to assess a clinically referred sample of children with ASD, it was found that 40% of 3–5 year old and over 50% of 6–12 year old children met DSM-IV criteria for at least one subtype of ADHD (Gadow et al., 2005). Despite the seemingly ample line of clinical evidences suggesting the link between the comorbid ADHD-like behavioral phenotypes, especially hyperactivity in ASD patients, few studies investigated the underlying mechanism or the characteristics of pharmacological response profile.
Recently, several researchers including us reported that adolescence and adulthood rats prenatally exposed to VPA, one of the environmental factor causing ASD in human and an widely used animal model of ASD, showed increased locomotor activity and repetitive/stereotypic behavior. VPA is a medication used for the treatment of epileptic seizure and bipolar disorder (Phiel et al., 2001). In some cases, even pregnant moms may take VPA for the treatment of their condition. Children exposed in utero to VPA have developmental delay including reduced cognitive function, ADHD, and learning difficulties. Christianson and their collegues first observed association between in utero VPA exposure and ASD (Christianson et al., 1994). Affected children demonstrated developmental delay and one of these children also had ASD. Some reports appeared thereafter in the literature associating VPA exposure to ASD (Williams et al., 2001; Schneider and Przewlocki, 2005).
The dysfunction of the catecholaminergic neuronal system, particularly DA and NE system, plays a key role in the manifestation of hyperactivity phenotypes in ADHD (Shaywitz et al., 1976). Most research suggests that hyperactivity is associated with an increase in synaptic DA concentration as well as the dysfunction in NE systems that is supported by the determination of neurotransmitter release and clinical pharmacological evidences. For example, when electrically stimulated brain slices obtained from spontaneous hypertensive rats (SHR), one of the animal model of ADHD, and Wistar Kyoto rats (WKY) as a control, the release of dopamine is more pronounced in prefrontal cortex of SHR than that of WKY. Also, SHR is more sensitive to dopamine receptor antagonists, quinpirole than WKY (Russell et al., 1995). It has been also suggested that clonidine, an α1/α2 adrenergic agonist or guanfacine, an α2 adrenergic agonist, alleviated hyperactivity in some patients (Hunt et al., 1995).
DAT and NET regulate extracellular levels of DA and NE. The dysregulation of their function or expression may induce abnormal neurotransmitter homeostasis. In DAT knock-out (DAT KO) mice, extracellular levels of DA is increased and function of dopamine receptors D1 and D2 are altered. Also, DAT KO mice have behavioral abnormalities such as hyperactivity in the open field test and reduced prepulse inhibition (Barr et al., 2004, Yamashita et al., 2006). In NET knock-out (NET KO) mice, extracellular levels of NE is elevated and NE clearance is decreased (Xu et al., 2000; Vizi et al., 2004). Both MPH and ATX are the first-line drugs for the treatment of ADHD. The drugs regulate synaptic DA and NE concentration via blocking DAT and NET, respectively. MPH is a DA and NE reuptake inhibitor, which increase DA and NE availability (Challman and Lipsky, 2000). ATX binds DAT, NET, and serotonin transporter (SERT), but has high affinity and selectivity for NET and blocks NE reuptake (Bymaster et al., 2002).
In this study, we examined the expression of DAT and NET in the prefrontal cortex of prenatally VPA-exposed male rat offspring and then investigated the pharmacological effects of MPH and ATX on the hyperactive phenotype of prenatally VPA-exposed male rat offspring.
MATERIALS AND METHODS
Materials
Agarose, PIPES, Potassium hydroxide, Tween® 20, 2-mercaptoethanol, Trypsin, MPH, ATX and VPA were purchased from Sigma (St. Louis, MO, USA). ECLTM Western blotting detection reagents were obtained from Amersham Life Science (Arlington Heights, IL, USA). Trizol and SuperScriptTM II Reverse Transcriptase were purchased from Invitrogen (Carlsbad, CA, USA). DNase Ι was purchased from Roche (Mannheim, Germany). Protein G Agarose was obtained from Millipore Corporation (Billerica, MA, USA). Taq polymerase and dNTP were obtained from Takara (Shiga, Japan). Protease inhibitor cocktail was obtained from Calbiochem (La Jolla, CA, USA). Chelex 100 was obtained from BioRad (Hercules, CA, USA). Antibodies were purchased from the following companies: anti-β-actin from Sigma (St. Louis, MO, USA), Histone H3, Acetyl-histone H3 and HDAC1 antibody from Cell signaling (Boston, MA, USA), DAT, NET and peroxidase-conjugated secondary antibody from Santa Cruz biotechnology (Dallas, TX, USA).
Animals
Pregnant Sprague-Dawley rats were obtained from Orient-bio Inc. (Kyunggi-do, Korea). Animals were maintained on a 12:12-h circadian cycle with lights on at 06:00, at a constant temperature (22 ± 2°C) and humidity (55 ± 5%). Animal experiments were carried out in accordance with the Principle of Laboratory Animal Care (NIH publication No. 85-23, revised 1985) and the Animal Care and Use Guidelines of Konkuk University, Korea (KU12115). All efforts were made to minimize the number of animals required for this study as well as their suffering. Behavioral experiments were performed between 10:00 and 16:00 in dedicated test room.
Subcutaneous injection of VPA to pregnant rat and treatment of MPH and ATX
The sodium salt of VPA was dissolved in 0.9% saline for a concentration of 100 mg/ml, pH 7.3. The dosage was adjusted according to the body weight of the pregnant rat on the day of injection. Pregnant rats received a single subcutaneous injection of 400 mg/kg VPA on gestational day 12. Control rats were treated with saline. In our previous publication, we reported that the number of live birth or body weight gain of rat pups was not significantly different (Kim et al., 2013). MPH (5 mg/kg) and ATX (3 mg/kg) were injected intraperitoneally in each treatment group.
Western blot analysis
Cells were washed twice with PBS and lysed with 2x SDS-PAGE sample buffer (120 mM Tris-HCl (pH 6.8), 20% glycerol, 4% SDS, 28.8 mM 2-mercaptoethanol, 0.01% bromophenol blue). Prefrontal cortices were homogenized using RIPA buffer (150 mM sodium chloride, 1% Triton X-100, 0.5% sodium deoxycholate, 0.1% SDS, 50 mM Tris, pH 8.0), and the lysates were diluted with 2X SDS-PAGE sample buffer. An aliquot containing 50 mg of total protein was separated by 10% SDS-PAGE and transferred to nitrocellulose membranes. The membranes were blocked with 0.001% polyvinylalcohol in PBS containing 0.2% tween-20 for 1 hr. The membranes were incubated with first antibody (All antibodies diluted 1:5000) overnight at 4°C and then with peroxidase-conjugated secondary antibody for 2 hrs at room temperature. Specific bands were detected using the ECL system and exposed to LAS-3000 image detection system (Fuji, Tokyo, Japan).
RT-PCR
Total RNA was isolated from cortical tissues using Trizol reagent and 1 μg of total RNA was converted to cDNA using Superscript II reverse transcriptase, according to the manufacturer’s instructions. Specific DNA bands were amplified by PCR. PCR reactions were carried out at 95°C for 20 sec, 60°C for 40 sec, and 72°C for 1 min to detect mRNA levels for SLC6A2, SLC6A3 and GAPDH. This cycle was repeated 34 times for SLC6A2, SLC6A3 and 23 times GAPDH followed by an extension cycle at 72°C for 5 min. The amplified DNA products were resolved by 2.0% agarose gel electrophoresis and visualized by staining with ethidium bromide and exposed to Bio-Rad electrophoresis image analyzer (Hercules, CA, USA). The size of product base pairs are 71 (SLC6A2), 258 (SLC6A3) and 314 (GAPDH). All primers were purchased from Bioneer (Daejeon, Korea). The primers used in this analysis are:
-
SLC6A2 (NET), 5′-ACTCTGCCTGGTGCTTCCAA-3′ (forward)
5′-ATCCATACCGTGGCCTCCTT-3′ (reverse)
-
SLC6A3 (DAT), 5′-GGCTTACAGGACCTCAGAG-3′ (forward)
5′-AGTGTAGCGTTTCGGGATCT-3′ (reverse)
-
GAPDH, 5′-TCCCTCAAGATTGTCAGCAA-3′ (forward)
5′-AGATCCACAACGGATACATT-3′ (reverse)
Chromatin immunoprecipitation
Chromatin immunoprecipitation was performed according to the reported method (Nelson et al., 2006) with minor modifications. Prefrontal cortex of control or VPA-exposed rat offspring at week 4 was homogenized using PBS and 43 μl of 37% formaldehyde was added to 1.6 ml of homogenates. After 15 min incubation at room temperature, 225 μl of 1 M glycine was added and incubated for 5 min. The homogenates were centrifuged (2,000 g for 5 min at 4°C), then washed twice with cold PBS. Pellets were resuspended and lysed with 1 ml IP buffer (150 mM sodium chloride, 50 mM Tris-HCl pH 7.5, 5 mM EDTA, 0.5% IGEPAL CA-630, 1.0% Triton X-100) by pi-petting up and down on ice. After centrifugation (12,000 g for 1 min at 4°C), 1 mL of the washed and resuspended pellet was sonicated (40% of power, during 10 sec at 5 times, resting period during 90 sec between sonication) on ice to shear the chromatin. After centrifugation (12,000 g for 10 min at 4°C), supernatants were used for immunoprecipitation. Primary antibody (histone, acetyl-histone or HDAC1 antibody) was added to 1 ml of supernatant, and the samples were incubated for 12 hrs at 4°C on a rotating platform. IgG was used as a control antibody. After incubation, mixture of 20 μl of IP buffer and 20 μl of Protein G Agarose was added to the sample, and incubated for 45 min at 4°C on a rotating platform. After incubation, samples were washed five times by centrifugation (2,000 g for 3 min at 4°C), and the supernatants were removed. 100 μl of 10% Chelex 100 was added to the washed beads for DNA isolation, and the samples were boiled for 10 min at 90°C. After centrifugation (12,000 g for 1 min at 4°C), 80 μl of supernatant was transferred to new tube, and 120 μl of DDW was added to beads. After centrifugation (12,000 g for 1 min at 4°C), 120 μl of supernatant was collected and added to the previous supernatant. Isolated DNA was used for PCR reaction. After amplification and electrophoresis, the band intensity was quantified using AlphaImager® HP system (ProteinSimple Bioscience & Technology, Santa Clara, CA, USA). We used 1 μg DNA for PCR experiment and loaded 10 μl of PCR products on 2.0 % agarose gel. The primers used in this analysis are:
-
SLC6A2 (NET), 5′-GTAGGTTAAGTGGCTTGGGTGTAT-3′ (forward)
5′-GGAATTTTTGGACACTAAGGTTGT-3′ (reverse)
-
SLC6A3 (DAT), 5′-GAAATGGGTTCTGAAAAGGTCTTA-3′ (forward)
5′-TCTTTGCAAGGTTATAGAGTCTCG-3′ (reverse)
Open-field locomotor activity
We selected two or three 4 weeks old male rats from each litter for the analysis of locomotive activity. For this behavior experiments, total 9 male pups from 4 dams for each treatment group were assigned randomly. The observation apparatus consisted of five plastic boxes (42×42 cm) with a field bordered by 42 cm high sidewalls. Rats were moved into test boxes 5 min before recording for habituation. The total distance moved (ambulatory movements) and rearing number were monitored for 10 min (Han et al., 2008) using CCD camera-assisted automated motion tracking apparatus and software (EthoVision 3.1, Noldus information Technology, the Netherlands).
Statistical analysis
Data were expressed as mean ± standard error of mean (S.E.M) and analyzed for statistical significance using one-way analysis of variance (ANOVA) followed by Newman-Keuls test as a post-hoc test. Two-way ANOVA was used to identify the effects of VPA exposure, drug treatment (MPH or ATX), or the interaction between the two factors. If significant effects were found in any one of the factors, post-hoc comparisons were conducted using Bonferroni’s post-tests. Differences were considered statistically significant when the P value was less than 0.05 (p<0.05). All statistical analyses were conducted using PASW Statistics (18.0; SPSS Inc, Chicago, IL, USA).
RESULTS
The expression of DAT was increased by VPA exposure
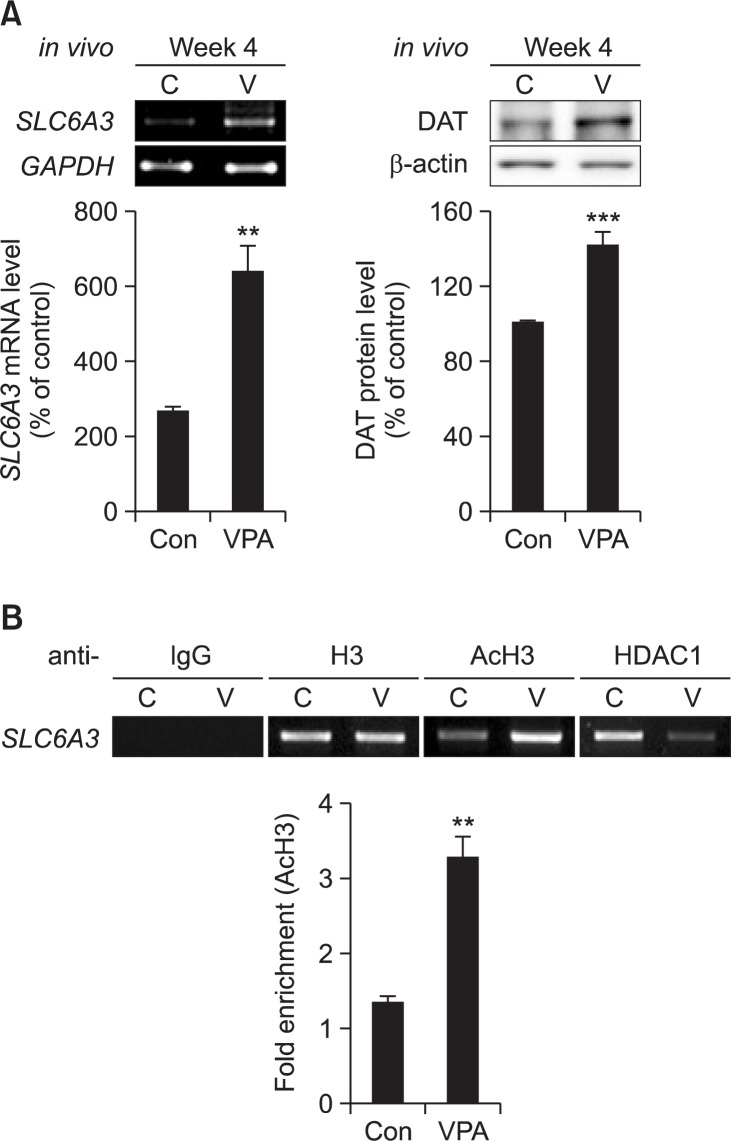
The mRNA level of SLC6A3 (DAT) was increased in the prefrontal cortex of 4 weeks old prenatally VPA-exposed male rat offspring (Fig. 1A). The protein level of DAT was also increased by VPA exposure (Fig. 1A). Chromatin IP showed that VPA exposure enhanced acetylation of SLC6A3 gene-bound histone, and decreased HDAC1 binding to SLC6A3 gene in the prefrontal cortex of 4 weeks old prenatally VPA-exposed male rat offspring (Fig. 1B). These results suggested that prenatal VPA exposure induced DAT expression in prefrontal cortex of male offspring through its HDAC inhibitory activity.
Fig. 1.
DAT expression was increased in prenatally VPA-exposed rat offspring. (A) Protein and mRNA level of DAT was examined in VPA-exposed rat offspring at week 4. Prefrontal cortex of each male rat brain was isolated and used for RT-PCR and Western blot analysis. mRNA level of GAPDH was used as a loading control. Protein level of β-actin was used as a loading control for DAT protein. (B) Chromatin IP was conducted as described in materials and methods. At week 4, prefrontal cortex of each male rat brain was used for Chromatin IP. Prenatal VPA exposure enhanced the acetylation of histone H3 bound to SLC6A3 gene, and detached HDAC1 protein from SLC6A3 gene. All data are expressed as mean ± S.E.M. **p<0.01, ***p<0.001 vs. control (n=4).
The expression of NET was increased by VPA exposure
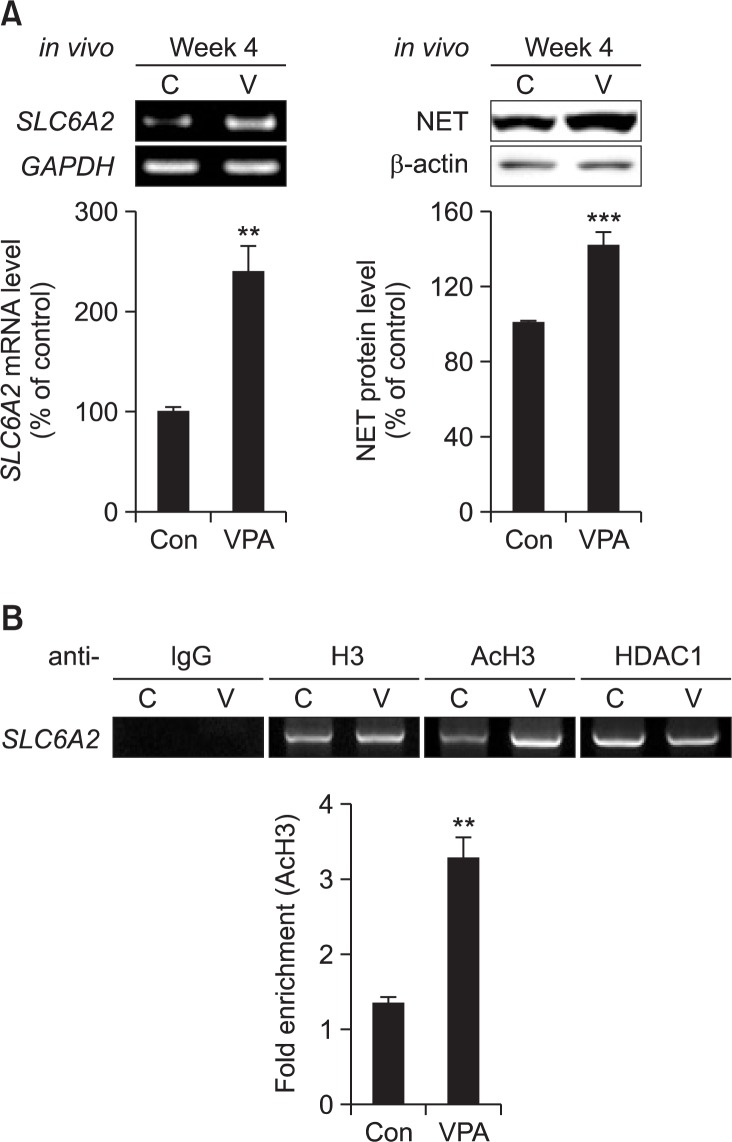
The mRNA and protein level of SLC6A2 (NET) was increased in the prefrontal cortex of prenatally VPA-exposed male rat offspring at week 4 (Fig. 2A). Chromatin IP revealed that VPA exposure enhanced acetylation of SLC6A2 gene-bound histone, and decreased HDAC1 binding from SLC6A2 gene in the prefrontal cortex of prenatally VPA-exposed male rat offspring at week 4 (Fig. 2B). These results suggested that VPA exposure also induced NET expression in prefrontal cortex of male offspring through its HDAC inhibitory activity.
Fig. 2.
NET expression was increased in prenatally VPA-exposed rat offspring. (A) Protein and mRNA level of NET was examined in VPA-exposed rat offspring at week 4. Prefrontal cortex of each male rat brain was isolated and used for RT-PCR and Western blot analysis. mRNA level of GAPDH was used as a loading control. Protein level of β-actin was used as a loading control for NET protein. All data are expressed as mean ± S.E.M. **p<0.01, ***p<0.001 vs. control (n=4). (B) Chromatin IP was conducted as described in materials and methods. At week 4, prefrontal cortex of each male rat brain was used for Chromatin IP. Prenatal VPA exposure enhanced the acetylation of histone H3 bound to SLC6A2 gene, and detached HDAC1 protein from SLC6A2 gene.
Hyperactivity of VPA-exposed male rat offspring was normalized by ATX but not by MPH
Prenatal VPA exposure showed increased activity in open-field locomotor test, which is consistent with a previous report (Edden et al., 2012). In this study, we investigated the effect of MPH and ATX on hyperactivity of VPA-exposed male rat offspring.
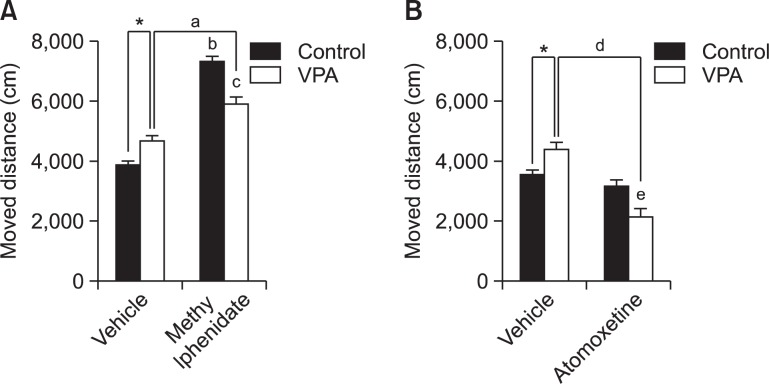
The effect of MPH on hyperactivity was shown in Fig. 3A. MPH increased the locomotive activity both in control and VPA group. Two-way ANOVA revealed that distance travelled was increased by MPH treatment [F(1,28)=158.5, p<0.0001]. There is a significant interaction between VPA exposure× MPH treatment [F(1,28)=33.51, p<0.0001]. Post-hoc comparisons showed that MPH treatment increased moving distance both in VPA-exposed rat offspring (ap<0.001) and saline-exposed rat off-spring (bp<0.001). In the MPH treated group, VPA-exposed rat offspring moved less in the open-field than saline-exposed rat offspring (cp<0.001). And VPA-exposed rat offspring moved more in the open-field than control (saline-exposed) rat off-spring (*p<0.05).
Fig. 3.
Effect of MPH and ATX on the hyperactivity of prenatally VPA-exposed rat offspring. Open-field locomotor activity test was performed with prenatally VPA-exposed male rat offspring at week 4. Moved distance was observed by EthoVision (Noldus information Technology, the Netherlands). (A) The effect of MPH on hyperactivity. All data are expressed as mean ± S.E.M. (n=8) *p<0.05 for saline-treated group vs. VPA-treated group; ap<0.001 for VPA-treated group vs. VPA and MPH-treated group; bp<0.001 for saline-treated group vs. saline and MPH-treated group; cp<0.001 for saline and MPH-treated group vs. VPA and MPH-treated group, as revealed by post-hoc Bonferroni’s comparisons following two-way ANOVA. (B) The effect of ATX on hyperactivity. All data are expressed as mean ± S.E.M. (n 8) *p<0.05 for saline-treated group vs. VPA-treated group; dp<0.001 for VPA-treated group vs. VPA and ATX-treated group; ep<0.05 for saline and ATX-treated group vs. VPA and ATX-treated group, as revealed by post-hoc Bonferroni’s comparisons following two-way ANOVA.
The effect of ATX on hyperactivity was shown in Fig. 3B. ATX decreased locomotive activity in VPA group. Two-way ANOVA revealed that moved distance was decreased by ATX treatment [F(1,28)=27.52, p<0.0001]. There is a significant interaction between VPA exposure×ATX treatment [F(1,28)=14.19, p<0.0001]. Post-hoc comparisons showed that ATX treatment decreased moving distance in VPA-exposed rat offspring (dp< 0.001) but not in control group. In the ATX treated group, VPA-exposed rat offspring moved less in the open-field compared with saline-exposed rat offspring (ep<0.05). And VPA-exposed rat offspring moved more in the open-field than control (saline-exposed) rat offspring (*p<0.05).
Effects of ATX on center arena movement and rearing frequency of VPA-exposed male rat offspring
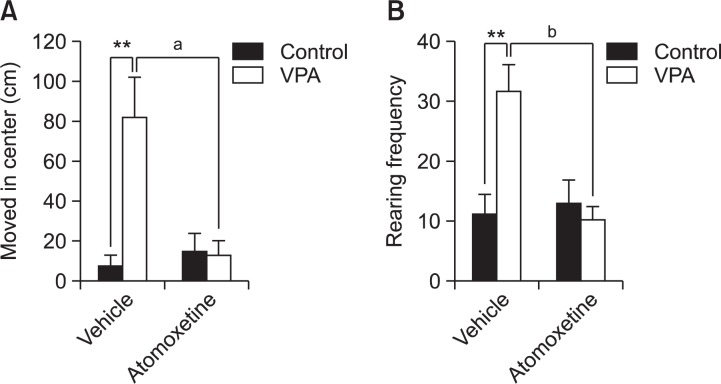
Next, we further analyzed movement in center arena as well as rearing frequency during open-field test. In VPA group, significantly increased rearing frequency and moved distance in center arena was observed in the open-field system. The analysis of data using Two-way ANOVA revealed that moved distance in center area was decreased by ATX treatment [F(1,35)=6.533, p<0.016]. There is a significant interaction between VPA exposure×ATX treatment [F(1,35)=8.789, p<0.006]. Post-hoc comparisons showed that ATX treatment decreased moving distance in the center arena in VPA-exposed rat off-spring (ap<0.001, Fig. 4A). Rearing frequency also showed a significant interaction between VPA exposure×ATX treatment [F(1,46)=8.789, p<0.01], and ATX reduced rearing number in VPA-exposed rat offspring (bp<0.001, Fig. 4B). We also checked the anxiety-like behaviors using EPM test but there was no differences in the frequency of open arm entry and open/closed arm stay ratio between VPA and VPA-ATX group (data not shown).
Fig. 4.
Effect of ATX on the movement in the center arena and rearing frequency of prenatally VPA-exposed rat offspring. The moved distance of center arena and rearing number during the open field test was checked using rat offspring at week 4. Moved distance and rearing were observed using EthoVision (Noldus information Technology, the Netherlands). (A) The moved distance in the center field. VPA group showed increased movement in center arena, which was prevented by ATX. All data are expressed as mean ± S.E.M. (n=8–9) **p<0.01 for saline-treated group vs. VPA-treated group; ap<0.001 for VPA-treated group vs. VPA and ATX-treated group, as revealed by post-hoc Bonferroni’s comparisons following two-way ANOVA. (B) The effect of ATX on rearing frequency. All data are expressed as mean ± S.E.M. (n=10–11) **p<0.01 for saline-treated group vs. VPA-treated group; bp<0.001 for VPA-treated group vs. VPA and ATX-treated group, as revealed by post-hoc Bonferroni’s comparisons following two-way ANOVA.
DISCUSSION
In this study, we found the expression of DAT and NET at early adolescent period is up-regulated in the prefrontal cortex of rat offspring prenatally exposed to VPA, which might be related to the hyperactive phenotype of VPA animal model of ASD. Increased expression and functional activity of DAT in prefrontal cortex and striatum has been observed in animal models of ADHD such as spontaneous hypertensive rat (SHR) (Watanabe et al., 1997, Pandolfo et al., 2013). Interestingly, unlike the responsiveness of both human and animal model of ADHD to MPH and ATX, hyperactivity in VPA model of ASD was reversed by ATX, but not by MPH. Instead, MPH even increased locomotor activity in VPA group although the extent was smaller compared with control group. The behavioral sensitization reminds the amphetamine-induced behavioral sensitization in control animals. The stimulant-induced hyperactivity but not by ATX suggest that neurological substrates regulating hyperactive phenotype observed in VPA animal model of ASD is different from the bona fide ADHD cases. In a retrospective analysis study using 195 patients diagnosed as pervasive developmental disorder (PDD), it has been reported that stimulants appeared ineffective and poorly tolerated with adverse reactions such as agitation, aggression and weight loss for the substantial percentage of patients suggesting the need for clinical use of other class of medications such as ATX or α2-adrenergic receptor agonist (Stigler et al., 2004). Similar conclusion was reported in a double-blind, placebo-controlled, crossover trial followed by open-label continuation analysis using 25 ASD children with ADHD (Research Units on Pediatric Psychopharmacology Autism Network, 2005).
In a recent study, chronic administration of caffeine reduced the expression of DAT in SHR to a control level. In this condition, inattention was reversed but not the hyper-locomotion suggesting that DAT expression itself can not completely explain the hyperactive phenotype observed in SHR (Pandolfo et al., 2013). Other factors including DA receptor expression, overall neurotransmission of catecholaminergic systems, func tional activity of presynaptic receptors and complex interplay with other neurotransmitter systems such as NE, 5-HT and adenosine should be considered for the comprehensive understanding of the neural correlates regulating hyperactive phenotypes in these animal models.
The results from the present study suggest that the differential responsiveness against currently available medication needs to be separately examined in other models of ASD as well. In addition to the VPA animal model of ASD, hyperactivity phenotype was also reported in several other models of ASD. For example, fragile X mental retardation 1 (Fmr1) mutant mice showed increased exploratory behavior and motor activity (Consortium, 1994). In addition, some of ASD genetic mutant animal models including neuroligin-3 (NL-3), proSAP1/shank2, and voltage-gated sodium channel type I, alpha (Scn1a) KO mice displayed hyperactivity phenotype (Banerjee et al., 2014). By determining the pharmacological responsiveness profile against currently available ADHD medication using multiple ASD models, we might obtain information whether there is general disparity on the hyperactivity phenotype and drug responsiveness between authentic ADHD and different types of ASD.
It has been reported that catecholamine neurotransmission abnormality is related with ADHD phenotype. DA plays a key role in attention, psychomotor, reinforcing and rewarding behaviors which are deficient in ADHD. Amphetamine and MPH, which have been widely used to treat ADHD, block DAT and NET and thereby enhance catecholamine neurotransmission (Gatley et al., 1996). In some ASD patients, DA system is hyperactive and a DA antagonist Risperidone rescued hyperactivity and aggression in ASD children (Munarriz et al., 2002). Young and coauthors also reported that stereotypical behavior, which is one of the typical features of ASD, could be induced by hyperactive DA function (Young et al., 1982). These results may suggest that dysregulation of DA system in brain regions such as nucleus accumbens and striatum as well as other regulators of DA function including receptors and signal transducers may account for the hyper-locomotive activity of VPA animal model of ASD.
NE has also been proposed to play a key role in the pathophysiology and pharmacotherapy of ADHD (Biederman and Spencer, 1999). Used as an ADHD medication, ATX is a selective NE reuptake inhibitor in vitro and in vivo (Wong et al., 1982). Although there are a few experimental evidences suggesting the role of NET in the regulation of hyperactivity, conclusive evidence is sparse which directly connects NET in the regulation of hyperactivity in ADHD (Barr et al., 2001). Also, there are indirect pharmacological evidences suggesting the role of presynaptic α2 adrenergic receptor in the modulation of ADHD-like behaviors. Clonidine, an α1/α2 adrenergic agonist or guanfacine, an α2 adrenergic agonist, improved hyperactive tendency in some patients (Hunt et al., 1995), and it is modestly effective in the short-term treatment of irritability and hyperactivity in some children with autistic disorder (Jaselskis et al., 1992). Similarly, the infusion of presynaptic α2-receptor blocker yohimbine into prefrontal cortex induced locomotor hyperactivity in monkeys (Ma et al., 2005). Altogether, these results may suggest that NE abnormalities and reversal of NE transmission by ATX plays critical role in the regulation of hyperactive phenotype in prenatally VPA-exposed rat offspring. Whether the increased NET expression in the frontal cortex of VPA-exposed offspring has a causal or a consequential relationship to the hyperactivity phenotypes remains to be determined.
VPA is a mood stabilizer that widely prescribed as one of the medications for epilepsy and bipolar disorders (Phiel et al., 2001). The prenatal VPA exposure rat model has been suggested as an ASD-like animal model which have abnormalities including hyperactivity, social interaction deficit, and stereotyped or repetitive behaviors (Schneider and Przewlocki, 2005). Most of ASD patients have abnormalities in DA and glutamatergic system in prefrontal cortex and limbic areas, and prenatally VPA-exposed rat offspring have similar deficits in DA and glutamatergic system in the brain (Rinaldi et al., 2007, Schneider et al., 2007). As well as human ASD patients, prenatally VPA-exposed rats have increased locomotor activity in a stressful environment. This abnormal phenotype has been attributed to the increased mesolimbic-dopaminergic activities (Lipska et al., 1993, Flores et al., 2006). In addition, our results also suggest that the dysregulation of NE neurotransmission should be given further attention, at least in the VPA animal model of ASD. In addition to the increased level of DA in the frontal cortex (Narita et al., 2002), NE level were also increased in the rat hippocampus and brain stem after intraperitoneal injection with VPA (200 mg/kg) (Baf et al., 1994). In cultured cortical neurons, VPA but not valpromide, a structural analog of VPA devoid of HDACi activity, significantly increased NET gene expression through increased histone acetylation of NET gene promoter (Bayles et al., 2010), which is related to our present result (Fig. 2B). Recently, Ali and Elgoly reported that DA, NE content in different brain areas, such as cortex, hippocampus, midbrain, cerebellum and pons, are have significantly different in the VPA exposed (800 mg/kg) rat pups compared with control group (Ali and Elgoly, 2013).
In our study, MPH had no effect but ATX normalized hyperactivity in VPA animal model. In addition to the direct effects on NET, ATX also acts as a NMDA antagonist (Ludolph et al., 2010) and an allosteric modulator of GABAA receptor (Zhang et al., 1997). GABAergic system dysfunction leads to several developmental disorders, such as ADHD and ASD and some researchers reported that inhibitory neurotransmitter GABA is related with motor control and impulsivity (Sumner et al., 2010; Boy et al., 2011). Interestingly, the hyperactive Wiggling rats showed decreased levels of GABA transporter gene expression and increased GABA concentration in the frontal cortex, striatum and midbrain (Masuo et al., 2007). The down-regulation of GABA receptors might be related with hyperactivity, and chronic oral administration of ATX induced mRNA and protein expression of GABAA receptor in rat brain (Lempp et al., 2013) suggesting the possible additional therapeutic mechanism of ATX. Interestingly, we reported the decreased expression of GAD, a rate limiting enzyme in GABA biosynthetic pathway, in the brain of VPA animal model of ASD (Kim et al., 2014). Whether the alteration of GABAergic system by ATX contributes to the amelioration of behavioral abnormalities in VPA animal model, such as hyperactivity and defective social interaction, should be investigated further in the future.
In conclusion, we observed increased expression of DAT and NET in frontal cortex of prenatally VPA-exposed rat off-spring, possibly via modulation of acetylation of histone bound to the promoter regions of both genes, suggesting the catecholaminergic abnormalities of the animal model. The hyper-locomotive activity of VPA animal model was reversed by the treatment with ATX but not with MPH, which implicates dysregulation of NE rather than DA system is primarily involved in the manifestation of hyperactive phenotype of VPA animal model. Defining the molecular players and understanding their interaction underlying the hyperactive behavioral phenotypes along with investigating the pharmacological response profiles in many other animal models may provide us an opportunity to fine tune the treatment protocol against hyperacitivy in disorders like ASD and ADHD.
Acknowledgments
The authors declare no conflict of interest. This work was supported by a grant of the Korean Health Technology R&D Project, Ministry of Health & Welfare, Republic of Korea (No. A120029).
REFERENCES
- Ali EH, Elgoly AH. Combined prenatal and postnatal butyl paraben exposure produces autism-like symptoms in off-spring: Comparison with valproic acid autistic model. Pharmacol Biochem Behav. 2013;111:102–110. doi: 10.1016/j.pbb.2013.08.016. [DOI] [PubMed] [Google Scholar]
- Baf MH, Subhash MN, Lakshmana KM, Rao BS. Sodium valproate induced alterations in monoamine levels in different regions of the rat brain. Neurochem Int. 1994;24:67–72. doi: 10.1016/0197-0186(94)90130-9. [DOI] [PubMed] [Google Scholar]
- Banerjee S, Riordan M, Bhat M. Genetic aspects of autism spectrum disorders: insights from animal models. Front Cell Neurosci. 2014;8:58. doi: 10.3389/fncel.2014.00058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barr AM, Lehmann-Masten V, Paulus M, Gainetdinov RR, Caron MG, Geyer MA. The selective serotonin-2A receptor antagonist M100907 reverses behavioral deficits in dopamine transporter knockout mice. Neuropsychopharmacology. 2004;29:221–228. doi: 10.1038/sj.npp.1300343. [DOI] [PubMed] [Google Scholar]
- Barr CL, Wigg K, Zai G, Roberts W, Malone M, Schachar R, Tannock R, Kennedy JL. Attention-deficit hyperactivity disorder and the adrenergic receptors alpha 1C and alpha 2C. Mol. Psychiatry. 2001;6:334–337. doi: 10.1038/sj.mp.4000863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bayles R, Baker E, Eikelis N, El-Osta A, Lambert G. Histone modifications regulate the norepinephrine transporter gene. Cell Cycle. 2010;9:4600–4601. doi: 10.4161/cc.9.22.13888. [DOI] [PubMed] [Google Scholar]
- Biederman J, Spencer T. Attention-deficit/hyperactivity disorder (ADHD) as a noradrenergic disorder. Biol. Psychiatry. 1999;46:1234–1242. doi: 10.1016/s0006-3223(99)00192-4. [DOI] [PubMed] [Google Scholar]
- Boy F, Evans CJ, Edden RA, Lawrence AD, Singh KD, Hu sain M, Sumner P. Dorsolateral prefrontal gamma-aminobutyric acid in men predicts individual differences in rash impulsivity. Biol. Psychiatry. 2011;70:866–872. doi: 10.1016/j.biopsych.2011.05.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bymaster FP, Katner JS, Nelson DL, Hemrick-Luecke SK, Threlkeld PG, Heiligenstein JH, Morin SM, Gehlert DR, Perry KW. Atomoxetine increases extracellular levels of norepinephrine and dopamine in prefrontal cortex of rat: a potential mechanism for efficacy in attention deficit/hyperactivity disorder. Neuropsychopharmacology. 2002;27:699–711. doi: 10.1016/S0893-133X(02)00346-9. [DOI] [PubMed] [Google Scholar]
- Challman TD, Lipsky JJ. Methylphenidate: its pharmacology and uses. Mayo Clin Proc. 2000;75:711–721. doi: 10.4065/75.7.711. [DOI] [PubMed] [Google Scholar]
- Christianson AL, Chesler N, Kromberg JG. Fetal valproate syndrome: clinical and neuro-developmental features in two sibling pairs. Dev Med Child Neurol. 1994;36:361–369. doi: 10.1111/j.1469-8749.1994.tb11858.x. [DOI] [PubMed] [Google Scholar]
- Consortium Fmr1 knockout mice: a model to study fragile X mental retardation. Cell. 1994;78:23–33. [PubMed] [Google Scholar]
- Edden RA, Crocetti D, Zhu H, Gilbert DL, Mostofsky SH. Reduced GABA concentration in attention-deficit/hyperactivity disorder. Arch Gen Psychiatry. 2012;69:750–753. doi: 10.1001/archgenpsychiatry.2011.2280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flores JA, Galan-Rodriguez B, Ramiro-Fuentes S, Fernandez-Espejo E. Role for dopamine neurons of the rostral linear nucleus and periaqueductal gray in the rewarding and sensitizing properties of heroin. Neuropsychopharmacology. 2006;31:1475–1488. doi: 10.1038/sj.npp.1300946. [DOI] [PubMed] [Google Scholar]
- Gadow KD, Devincent CJ, Pomeroy J, Azizian A. Comparison of DSM-IV symptoms in elementary school-age children with PDD versus clinic and community samples. Autism. 2005;9:392–415. doi: 10.1177/1362361305056079. [DOI] [PubMed] [Google Scholar]
- Gatley SJ, Pan D, Chen R, Chaturvedi G, Ding YS. Affinities of methylphenidate derivatives for dopamine, norepinephrine and serotonin transporters. Life Sci. 1996;58:231–239. doi: 10.1016/0024-3205(96)00052-5. [DOI] [PubMed] [Google Scholar]
- Han H, Ma Y, Eun JS, Hong JT, Oh KW. Anxiolytic-like effects of cyclopeptide fraction alkaloids of Zizyphi Spinosi semen: possible involvement of GABAA receptors. Biomol Ther. 2008;16:261–269. [Google Scholar]
- Hunt RD, Arnsten AF, Asbell MD. An open trial of guanfacine in the treatment of attention-deficit hyperactivity disorder. J Am Acad Child Adolesc Psychiatry. 1995;34:50–54. doi: 10.1097/00004583-199501000-00013. [DOI] [PubMed] [Google Scholar]
- Jaselskis CA, Cook EH, JR, Fletcher KE, Leventhal BL. Clonidine treatment of hyperactive and impulsive children with autistic disorder. J Clin Psychopharmacol. 1992;12:322–327. [PubMed] [Google Scholar]
- Kim KC, Kim P, Go HS, Choi CS, Park JH, Kim HJ, Jeon SJ, Dela Pena IC, Han SH, Cheong JH, Ryu JH, Shin CY. Male-specific alteration in excitatory post-synaptic development and social interaction in pre-natal valproic acid exposure model of autism spectrum disorder. J Neurochem. 2013;124:832–843. doi: 10.1111/jnc.12147. [DOI] [PubMed] [Google Scholar]
- Kim KC, Lee DK, Go HS, Kim P, Choi CS, Kim JW, Jeon SJ, Song MR, Shin CY. Pax6-dependent cortical glutamatergic neuronal differentiation regulates autism-like behavior in prenatally valproic acid-exposed rat offspring. Mol Neurobiol. 2014;49:512–528. doi: 10.1007/s12035-013-8535-2. [DOI] [PubMed] [Google Scholar]
- Lempp T, Toennes SW, Wunder C, Russe OQ, Moser CV, Kynast KL, Freitag CM, Niederberger E. Altered gene expression in the prefrontal cortex of young rats induced by the ADHD drug atomoxetine. Prog. Neuropsychopharmacol. Biol. Psychiatry. 2013;40:221–228. doi: 10.1016/j.pnpbp.2012.08.012. [DOI] [PubMed] [Google Scholar]
- Leyfer OT, Folstein SE, Bacalman S, Davis NO, Dinh E, Morgan J, Tager-Flusberg H, Lainhart JE. Comorbid psychiatric disorders in children with autism: interview development and rates of disorders. J Autism Dev Disord. 2006;36:849–861. doi: 10.1007/s10803-006-0123-0. [DOI] [PubMed] [Google Scholar]
- Lipska BK, Jaskiw GE, Weinberger DR. Postpubertal emergence of hyperresponsiveness to stress and to amphetamine after neonatal excitotoxic hippocampal damage: a potential animal model of schizophrenia. Neuropsychopharmacology. 1993;9:67–75. doi: 10.1038/npp.1993.44. [DOI] [PubMed] [Google Scholar]
- Ludolph AG, Udvardi PT, Schaz U, Henes C, Adolph O, Weigt HU, Fegert JM, Boeckers TM, Fohr KJ. Atomoxetine acts as an NMDA receptor blocker in clinically relevant concentrations. Br J Pharmacol. 2010;160:283–291. doi: 10.1111/j.1476-5381.2010.00707.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ma CL, Arnsten AF, Li BM. Locomotor hyperactivity induced by blockade of prefrontal cortical alpha2-adrenoceptors in monkeys. Biol. Psychiatry. 2005;57:192–195. doi: 10.1016/j.biopsych.2004.11.004. [DOI] [PubMed] [Google Scholar]
- Masuo Y, Ishido M, Morita M, Sawa H, Nagashima K, Niki E. Behavioural characteristics and gene expression in the hyperactive wiggling (Wig) rat. Eur J Neurosci. 2007;25:3659–3666. doi: 10.1111/j.1460-9568.2007.05613.x. [DOI] [PubMed] [Google Scholar]
- Munarriz R, Bennett L, Goldstein I. Risperidone in children with autism and serious behavioral problems. N Engl J Med. 2002;347:1890–1891. author reply 1890–1891. [PubMed] [Google Scholar]
- Narita N, Kato M, Tazoe M, Miyazaki K, Narita M, Okado N. Increased monoamine concentration in the brain and blood of fetal thalidomide- and valproic acid-exposed rat: putative animal models for autism. Pediatr Res. 2002;52:576–579. doi: 10.1203/00006450-200210000-00018. [DOI] [PubMed] [Google Scholar]
- Nelson JD, Denisenko O, Bomsztyk K. Protocol for the fast chromatin immunoprecipitation (ChIP) method. Nat Protoc. 2006;1:179–185. doi: 10.1038/nprot.2006.27. [DOI] [PubMed] [Google Scholar]
- Pandolfo P, Machado NJ, Kofalvi A, Takahashi RN, Cunha RA. Caffeine regulates frontocorticostriatal dopamine transporter density and improves attention and cognitive deficits in an animal model of attention deficit hyperactivity disorder. Eur Neuropsychopharmacol. 2013;23:317–328. doi: 10.1016/j.euroneuro.2012.04.011. [DOI] [PubMed] [Google Scholar]
- Phiel CJ, Zhang F, Huang EY, Guenther MG, Lazar MA, Klein PS. Histone deacetylase is a direct target of valproic acid, a potent anticonvulsant, mood stabilizer, and teratogen. J Biol Chem. 2001;276:36734–36741. doi: 10.1074/jbc.M101287200. [DOI] [PubMed] [Google Scholar]
- Research Units on Pediatric Psychopharmacology Autism Network Randomized, controlled, crossover trial of methylphenidate in pervasive developmental disorders with hyperactivity. Arch. Gen. Psychiatry. 2005;62:1266–1274. doi: 10.1001/archpsyc.62.11.1266. [DOI] [PubMed] [Google Scholar]
- Rinaldi T, Kulangara K, Antoniello K, Markram H. Elevated NMDA receptor levels and enhanced postsynaptic long-term potentiation induced by prenatal exposure to valproic acid. Proc Natl Acad Sci USA. 2007;104:13501–13506. doi: 10.1073/pnas.0704391104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Russell V, De Villiers A, Sagvolden T, Lamm M, Taljaard J. Altered dopaminergic function in the prefrontal cortex, nucleus accumbens and caudate-putamen of an animal model of attention-deficit hyperactivity disorder--the spontaneously hypertensive rat. Brain Res. 1995;676:343–351. doi: 10.1016/0006-8993(95)00135-d. [DOI] [PubMed] [Google Scholar]
- Schneider T, Przewlocki R. Behavioral alterations in rats prenatally exposed to valproic acid: animal model of autism. Neuropsychopharmacology. 2005;30:80–89. doi: 10.1038/sj.npp.1300518. [DOI] [PubMed] [Google Scholar]
- Schneider T, Ziolkowska B, Gieryk A, Tyminska A, Przewlocki R. Prenatal exposure to valproic acid disturbs the enkephalinergic system functioning, basal hedonic tone, and emotional responses in an animal model of autism. Psychopharmacology (Berl) 2007;193:547–555. doi: 10.1007/s00213-007-0795-y. [DOI] [PubMed] [Google Scholar]
- Shaywitz BA, Yager RD, Klopper JH. Selective brain dopamine depletion in developing rats: an experimental model of minimal brain dysfunction. Science. 1976;191:305–308. doi: 10.1126/science.942800. [DOI] [PubMed] [Google Scholar]
- Simonoff E, Pickles A, Charman T, Chandler S, Loucas T, Baird G. Psychiatric disorders in children with autism spectrum disorders: prevalence, comorbidity, and associated factors in a population-derived sample. J. Am. Acad. Child Adolesc. Psychiatry. 2008;47:921–929. doi: 10.1097/CHI.0b013e318179964f. [DOI] [PubMed] [Google Scholar]
- Stigler KA, Desmond LA, Posey DJ, Wiegand RE, Mcdougle CJ. A naturalistic retrospective analysis of psychostimulants in pervasive developmental disorders. J Child Adolesc Psychopharmacol. 2004;14:49–56. doi: 10.1089/104454604773840481. [DOI] [PubMed] [Google Scholar]
- Sumner P, Edden RA, Bompas A, Evans CJ, Singh KD. More GABA, less distraction: a neurochemical predictor of motor decision speed. Nat Neurosci. 2010;13:825–827. doi: 10.1038/nn.2559. [DOI] [PubMed] [Google Scholar]
- Vizi ES, Zsilla G, Caron MG, Kiss JP. Uptake and release of norepinephrine by serotonergic terminals in norepinephrine transporter knock-out mice: implications for the action of selective serotonin reuptake inhibitors. J Neurosci. 2004;24:7888–7894. doi: 10.1523/JNEUROSCI.1506-04.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Watanabe Y, Fujita M, Ito Y, Okada T, Kusuoka H, Nishimura T. Brain dopamine transporter in spontaneously hypertensive rats. J Nucl Med. 1997;38:470–474. [PubMed] [Google Scholar]
- Wilkins J, Matson JL. A comparison of social skills profiles in intellectually disabled adults with and without ASD. Behav Modif. 2009;33:143–155. doi: 10.1177/0145445508321880. [DOI] [PubMed] [Google Scholar]
- Williams G, King J, Cunningham M, Stephan M, Kerr B, Hersh JH. Fetal valproate syndrome and autism: additional evidence of an association. Dev Med Child Neurol. 2001;43:202–206. [PubMed] [Google Scholar]
- Wong DT, Threlkeld PG, Best KL, Bymaster FP. A new inhibitor of norepinephrine uptake devoid of affinity for receptors in rat brain. J Pharmacol Exp Ther. 1982;222:61–65. [PubMed] [Google Scholar]
- Xu F, Gainetdinov RR, Wetsel WC, Jones SR, Bohn LM, Miller GW, Wang YM, Caron MG. Mice lacking the norepinephrine transporter are supersensitive to psychostimulants. Nat Neurosci. 2000;3:465–471. doi: 10.1038/74839. [DOI] [PubMed] [Google Scholar]
- Yamashita M, Fukushima S, Shen HW, Hall FS, Uhl GR, Numachi Y, Kobayashi H, Sora I. Norepinephrine transporter blockade can normalize the prepulse inhibition deficits found in dopamine transporter knockout mice. Neuropsychopharmacology. 2006;31:2132–2139. doi: 10.1038/sj.npp.1301009. [DOI] [PubMed] [Google Scholar]
- Young JG, Kavanagh ME, Anderson GM, Shaywitz BA, Cohen DJ. Clinical neurochemistry of autism and associated disorders. J Autism Dev Disord. 1982;12:147–165. doi: 10.1007/BF01531305. [DOI] [PubMed] [Google Scholar]
- Zhang SJ, Huguenard JR, Prince DA. GABAA receptor-mediated Cl- currents in rat thalamic reticular and relay neurons. J Neurophysiol. 1997;78:2280–2286. doi: 10.1152/jn.1997.78.5.2280. [DOI] [PubMed] [Google Scholar]