Abstract
Background
Psoriasis may predispose to cardiovascular disease and diabetes. However, the role of TNF inhibitor in mediating this risk is controversial.
Objective
To assess this relationship, we estimated change in metabolic physiologic measures before and after initiation of TNF inhibitor therapy compared with methotrexate therapy among psoriasis patients.
Methods
We conducted a retrospective cohort study, 2007–2012, using computerized clinical data for 1,274 new users of TNF inhibitor and 979 new users of methotrexate therapy to compare change in blood pressure, lipids, triglycerides, fasting plasma glucose, and body mass index before and after start of TNF inhibitors or methotrexate. The study was restricted to new users. We computed within-person change in each measure, so that each patient served as their own control. In addition, we compared TNF inhibitor patients to methotrexate patients, by computing the adjusted difference in their group means. In secondary analyses, we examined phototherapy as a comparator.
Results
Among starters of TNF inhibitor and MTX therapy, within-person change in physiologic measures at 6 months did not differ significantly. We observed no important or significant changes in any of the physiologic measures with initiation of TNF inhibitor compared with methotrexate. The same results were found in subgroup analyses focused on men, and on those with hypertension, diabetes mellitus, or obesity. The same results were observed with phototherapy, except that diastolic blood pressure declined by 0.6 mm Hg within-person during the 6 months after starting phototherapy (p<0.05).
Conclusions
The study provides no evidence for improvement of physiologic measures associated with the metabolic syndrome resulting from TNF inhibitor use for psoriasis.
Keywords: psoriasis, TNF inhibitor, methotrexate, physiologic measures, phototherapy
Introduction
Psoriasis has been associated with an increased risk of metabolic outcomes, including cardiovascular disease and diabetes, in controlled observational studies conducted in the Danish and Dutch populations,1–5 the Nurses Health Study,6 the National Health and Nutrition Examination Survey (NHANES),7 the Kaiser Permanente Southern California pediatric population,8 the General Practice Research Database and overlapping Health Improvement Network,9–15 and in an administrative claims database.16–17
This finding has led to the assertion that a proinflammatory state associated with severe psoriasis drives the development of metabolic syndrome, similar to rheumatoid arthritis.18 An implication is that therapy with TNF inhibitor could have a beneficial pleiotropic effect on both psoriasis and metabolic disease.19 However, studies of the association of TNF inhibitor with metabolic disease outcomes in psoriasis have yielded mixed results, partly because of differences across studies.20–24 Meta-analysis of more than 10,000 patients from 22 randomized controlled trials found no evidence for a benefit of TNF inhibitor compared with placebo.20 In contrast, studies of the Danish21 and Kaiser Permanente Southern California22 populations have reported a benefit of TNF inhibitor or methotrexate (MTX) compared with therapies for less severe psoriasis. Similarly, in Canadian and U.S. insurance databases, TNF inhibitor use was associated with reduced risk of diabetes.24 Some have argued that surveillance bias may be responsible for the associations among psoriasis, TNF inhibitor therapy, and metabolic disease endpoints,4, 5 while others have expressed concern about the role of the pharmaceutical industry financing in advancing this line of research.25
We sought to examine changes in risk of metabolic syndrome in relation to new use of TNF inhibitor. To minimize bias, especially surveillance bias and healthy user bias, we assessed metabolic physiologic measures using a before-and-after study in which each patient served as their own control. Past before-and-after studies of this hypothesis have been conducted with up to 100 patients selected from hospital-based settings.26–29 In contrast, the present study comprised all 2963 patients available in the well-characterized Kaiser Permanente Northern California membership. In addition, we used a control group, comprised of new users of MTX therapy, to account for important changes over time that could have affected the results. Phototherapy was used as a secondary comparator. Phototherapy was used as a secondary comparator. The results of this study will help elucidate whether or not TNF inhibitors have beneficial or adverse effects on diabetes and cardiovascular risk.
Materials and Methods
Setting
This study was conducted among 3.2 million members of Kaiser Permanente Northern California, a closed-model system that provides pre-paid, comprehensive, integrated healthcare. A key difference between Kaiser Permanente and other medical care settings is that physicians are on staff, and their compensation is independent of the patient’s utilization. The membership has been well characterized for research studies.30
Kaiser Permanente Northern California practice guidelines for psoriasis therapy state that etanercept or adalimumab should be prescribed as first-line TNF inhibitor therapy and infliximab should be prescribed as second- or third-line TNF inhibitor therapy. Infliximab may be prescribed for patients whose out-of-pocket drug costs are high, such as those on Medicare without supplemental coverage. According to the health plan’s rules, only specialists (ie dermatologists in this study) and not primary care providers can initiate biologic agents. With respect to non-biologic systemic drugs used to treat psoriasis, they are typically prescribed by dermatologists and not by primary care providers (but rheumatologists may prescribe systemic drugs for psoriatic arthritis, which may treat concomitant psoriasis). The study protocol was approved by the local Institutional Review Board at the Kaiser Foundation Research Institute.
Conceptual Framework
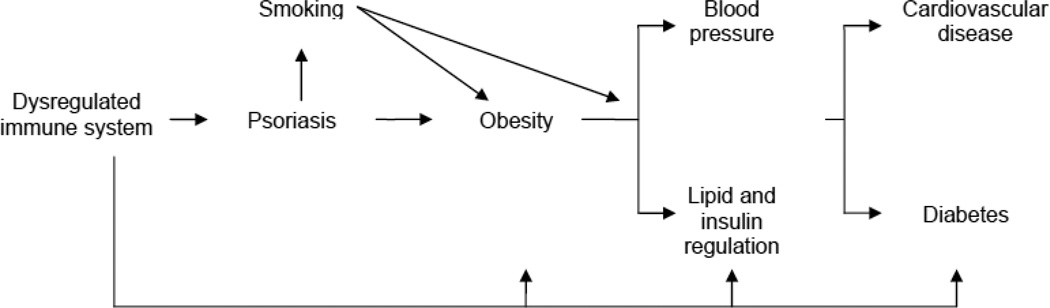
Fig. 1 presents the causal model posited for the study. The study was designed to clarify the possible mediating effects of TNF inhibition on obesity, blood pressure, lipids, and insulin regulation.
Figure 1.
Causal Model Posited for the Study
Study Design
We used a difference-in-difference design to study changes in physiologic measures before and after starting TNF inhibitor therapy or MTX, with phototherapy examined as a secondary comparator. A patient was excluded if he/she started both a TNF inhibitor and MTX at the same time. Each patient’s “after” measure was compared to the patient’s “before” measure. In addition, the group mean difference among the TNF inhibitor starters was compared to the group mean difference among the MTX and phototherapy starters.
Study Population
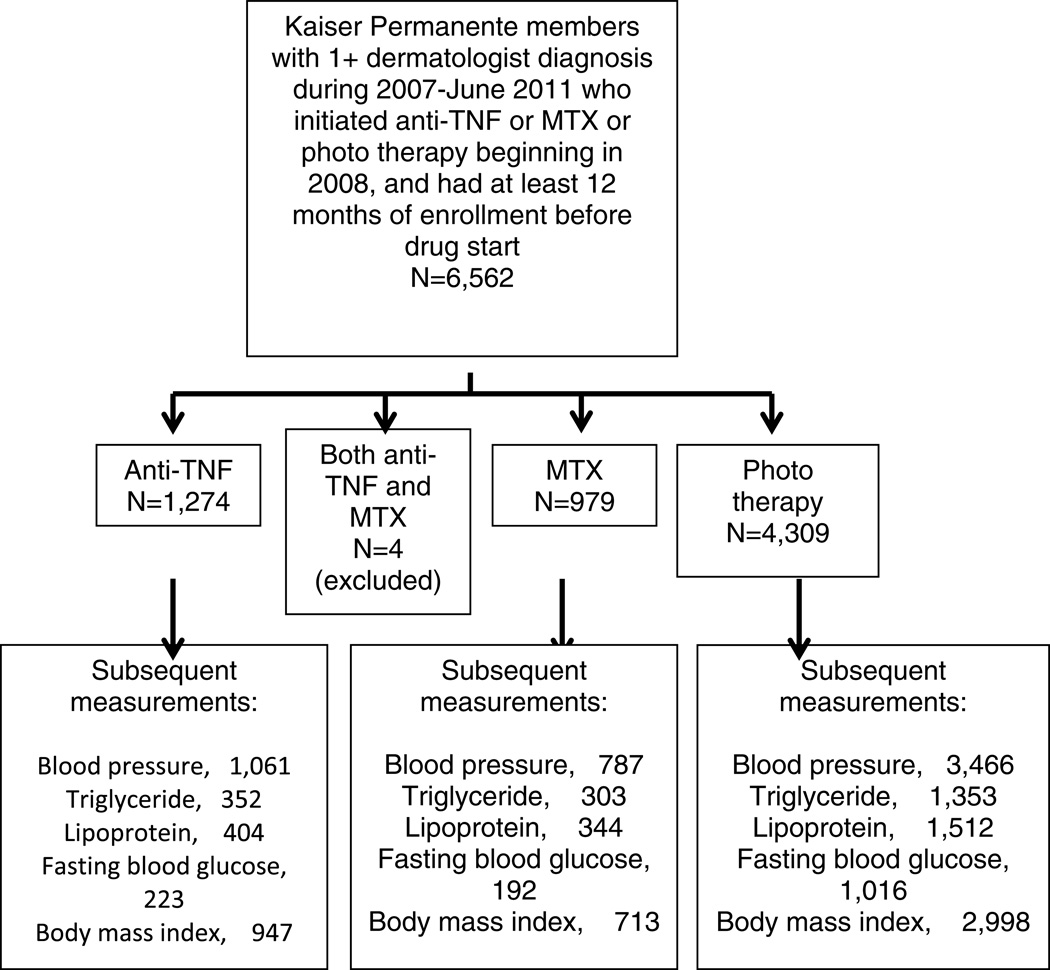
Psoriasis patients starting a TNF inhibitor were compared to a control group consisting of psoriasis patients starting MTX or phototherapy. Inclusion criteria into the study population were: aged 18 years or over at the first recorded date of PsO in the computerized data; ≥1 diagnoses of psoriasis (ICD-9 code 696.1) recorded by a dermatologist during January 1, 2007-December 31, 2012; 16 months of enrollment before starting therapy, at least one outcome measurement during the 16 months before starting therapy; and starting TNF inhibitor or MTX therapy on or after January 1, 2008 (Fig. 2). Four patients who started both TNF inhibitor or MTX on the same day were excluded from the study. The incident user design required patients to have 16 months without exposure to the drug before the index date, i.e., the date of first use of TNF inhibitor or MTX within the study timeframe. The case definition we used for psoriasis (≥1 dermatologist-rendered diagnoses) has been validated against chart review; it has a positive predictive value of 90%.31
Figure 2.
Study Population
Data Collection
Information on TNF inhibitor, MTX therapy, and phototherapy were obtained from the health plan’s pharmacy management information system, which is used to provide clinical care and provides a gold standard on prescriptions and dispensings.
Study outcomes were changes in systolic blood pressure (SBP), diastolic blood pressure (DBP), high-density lipoprotein (HDL), low-density lipoprotein (LDL), triglycerides, fasting plasma glucose, and body mass index (BMI) (Table 1). Potential modifying factors include baseline physiologic values and drugs used to control hypertension, diabetes mellitus, and dyslipidemia (Table 1), and these were obtained from the pharmacy data. In addition, we ascertained variables that could potentially confound the relationships under study. These variables included age and sex, which were obtained from membership files. In addition, we ascertained smoking history, which is recorded with each outpatient visit using the following categories: current, former, passive, or never.
Table 1.
Physiologic Measures and Drug Treatments Used for the Study
| Outcome | Source | Out of range | Relevant drug treatments |
|---|---|---|---|
| Blood pressure (systolic, diastolic), mm Hg | Vital signs | Systolic, >140 mm Hg Diastolic, >90 mm Hg |
ACE inhibitors Alpha blockers Alpha-2 receptor agonist Angiotensin II receptor blockers Beta-blockers Calcium channel blockers Central agonists Diuretics Peripheral adrenergic inhibitors Vasodilators |
| HDL, LDL, triglycerides, mg/dL | Cholesterol test | HDL < 40 mg/dL (men), 50 mg/dL (women) LDL 130–159 mg/dL (borderline) 160–189 mg/dL (high) Triglycerides 150–199 mg/dL borderline 200–499 mg/dL (high) |
Ezetimibe Fibrates Niacin Resins Statins |
| Fasting plasma glucose, mg/dL | Fasting plasma/serum glucose test | Impaired, 100–125 mg/dl Diabetes >125 mg/dl |
Alpha-glucosidase inhibitors Biguanides DPP-4 inhibitors Insulin Meglitinides Sulfonylureas Thiazolidinediones |
| Body mass index, kg/m2 | Vital signs | Obese, BMI 30 or greater | Diethylpropion Orlistat Phendimetrazine phentermine Sibutramine |
Data Analysis
For each of the seven outcomes (SBP, DBP, HDL, LDL, triglycerides, fasting plasma glucose, and BMI), we decided a priori to define the “before” measurement as the one most closely preceding the drug start date and to define the “after” measurement as the one most immediately following the drug start date. With respect to the “after” measurement, if no measurement was recorded within 6 months after starting the therapy, the patient was censored from the analysis. In addition, patients were censored on the date they switched therapies, so that those who switched before their first “after” measurement did not contribute to the analysis. In secondary analyses, we required the “after” measurement to be recorded within 3 or 12 months after starting therapy.
We constructed normal probability plots of within-person differences for visual inspection of distributions and outlying values. General linear modeling was used to assess the relationship of the therapeutic regimen with within-person and among-person differences in physiologic measures after adjusting for age, sex, smoking history, hypertension, dyslipidemia, obesity, MI, stroke, diabetes, PsA, number of PsO visits, and the baseline values of the physiologic measures recorded during the 16 months preceding the start of therapy. The natural log transformation of the triglyceride value was used in general linear modeling (Table 1). Subgroup analyses were restricted to patients whose physiologic measures were in control or out of range (Table 1). Because population subgroups may differ in their exposure pathways, we also conducted secondary analyses of the following subsets that are at increased risk for cardiovascular disease: men, and those with hypertension, diabetes mellitus, or obesity (BMI ≥30). All analyses were performed using SAS® version 9.3 (SAS Institute, Inc), PROC GLM.
Results
Characteristics of the Study Population
We identified a total of 2,253 psoriasis patients: 1,274 patients started TNF inhibitor therapy, 979 started MTX therapy, and 4,309 started phototherapy. (Fig. 2). Among the TNF inhibitor starters, MTX starters, and phototherapy starters, respectively, the mean ages were 46.7, 50.9 years (p<0.0001) and 52.0 (p<0.0001), 51.5%, 47.7%, and 52.9% were male, and the following comorbidities were present: psoriatic arthritis: 29.1%, 23.0% (p= 0.001) and 4.5% (p<0.0001); dyslipidemia: 27.1%, 33.1% (p= 0.002) and 30.6% (p=0.02); hypertension: 34.6%, 41.4% (p= 0.001), and 38.6% (p=0.009); diabetes mellitus: 16.4%, 16.7% (p=0.88), and 16.5% (p=0.94); and history of myocardial infarction: 1.7%, 2.7% (p=0.13), and 2.6% (p=0.08), respectively (Table 2).
Table 2.
Baseline Characteristics of Psoriasis Patients who were Starters of TNF Inhibitor, MTX, or phototherapy, Kaiser Permanente Northern California, 2008–2012.
| Characteristics | TNF inhibitor N=1,274 |
MTX** N=979 |
P values | Photo therapy** N=4,309 |
P values |
|---|---|---|---|---|---|
| Age, years, mean (±SD) | 46.7 (13.8) | 50.9 (14.4) | <0.0001 | 52.0 (15.9) | <0.0001 |
| Age group, years, no (%) | |||||
| <30 years | 151 (11.9) | 86 (8.8) | <0.0001 | 427 (9.9) | <0.0001 |
| 30–39 years | 237 (18.6) | 133 (13.6) | 568 (13.2) | ||
| 40–49 years | 344 (27.0) | 231 (23.6) | 822 (19.1) | ||
| 50–59 years | 320 (25.1) | 241 (24.6) | 1051 (24.4) | ||
| 60–69 years | 152 (11.9) | 191 (19.5) | 837 (19.4) | ||
| ≥70 years | 70 (5.5) | 97 (9.9) | 604 (14.0) | ||
| Men, no (%) | 656 (51.5) | 467 (47.7) | 0.08 | 2280 (52.9) | 0.37 |
| No. of psoriasis visits in 1 year before index, mean (SD) | 1.8 (2.0) | 1.4 (2.1) | <0.0001 | 1.7 (4.7) | 0.38 |
| Psoriatic arthritis (ICD9 696.0), no (%) | 371 (29.1) | 225 (23.0) | 0.001 | 194 (4.5) | <0.0001 |
| Dyslipidemia (272.4), no (%) | 345 (27.1) | 324 (33.1) | 0.002 | 1319 (30.6) | 0.02 |
| Hypertension (401.xx–405.9), no (%) | 441 (34.6) | 405 (41.4) | 0.001 | 1664 (38.6) | 0.009 |
| Diabetes mellitus (250.x), no (%) | 209 (16.4) | 163 (16.7) | 0.88 | 711 (16.5) | 0.94 |
| Obesity- BMI, kg /m2, mean (±SD) | 31.3 (7.1) | 30.8 (7.1) | 0.10 | 29.7 (6.5) | <0.0001 |
| ≤20 | 18 (1.5) | 15 (1.7) | 0.04 | 101 (2.6) | <0.0001 |
| 20.1–24.9 | 188 (16.1) | 173 (19.3) | 835 (21.7) | ||
| 25–29.9 | 361 (30.8) | 298 (33.3) | 1368 (35.5) | ||
| 30.0–34.9 | 321 (27.4) | 200 (22.3) | 869 (23.7) | ||
| ≤35 | 283 (24.2) | 210 (23.46) | 679 (17.6) | ||
| Smoking status, % | |||||
| Current smoker | 171 (13.4) | 121 (12.4) | 0.56 | 429 (10.0) | 0.02 |
| Former | 304 (23.9) | 233 (23.8) | 939 (21.8) | ||
| Passive | 11 (0.9) | 5 (0.5) | 41 (1.0) | ||
| Never | 788 (61.8) | 620 (63.3) | 2900 (67.3) | ||
| History of myocardial infarction (410.xx–412.xx), no (%) | 22 (1.7) | 26 (2.7) | 0.13 | 111 (2.6) | 0.08 |
| History of stroke (430.xx–438.xx, V12.54), no (%) | 21 (1.7) | 19 (1.9) | 0.60 | 101 (2.3) | 0.14 |
Baseline information ascertained during the year before start of therapy
comparing with anti-TNF group
Within-person change in physiologic measures at 6 months among starters of TNF inhibitor and MTX therapy did not differ significantly, although, on average, the after measurement was slightly lower than the before measurement for SBP, DBP, LDL, and fasting blood glucose, while the after measurement was higher for BMI and triglyceride (Fig. 3 and Online Table A). All other metabolic measures were similar between TNF inhibitor and phototherapy except for diastolic blood pressure, which showed a slight but significant decline (0.6 mm Hg, p=0.04) (Online Table A).All other metabolic measures were similar between TNF inhibitor and phototherapy except for diastolic blood pressure, which showed a slight but significant decline (0.6 mm Hg, p=0.04) (Online Table A).
Figure 3.
Within-person Change in Physiologic Measures, Based on Measurements Made Within 6 Months of Starting TNF Inhibitor or Methotrexate Therapy.*
The difference-in-differences comparing TNF inhibitor starters with MTX starters are presented in Online Table B. For SBP, DBP, triglyceride, and LDL, the decline (indicating improved measures) was larger for starters of MTX, although none of these difference-in differences within 6 months of therapy was statistically significant.
Online Table B also presents the difference-in-differences for the secondary analyses, in which the “after” measurement was recorded within 3 or 12 months after starting therapy. An association was observed for fasting blood glucose recorded within 12 months of starting therapy (p=0.01), but the difference-in-difference of 5.4 mg/dL may not be clinically significant. In addition, Online Tables E–H presents the results of subgroups analyses focusing on men and those with hypertension, diabetes, or obesity, using measures recorded within 6 months after starting therapy. None of these difference-in-difference was statistically significant. Additional results from these sensitivity and subgroups analyses are provided in Online Tables.
Discussion
In this retrospective study in which each person served as their own control, there were no clinically significant differences in physiologic measures of cardiovascular and metabolic disease using data recorded at 3, 6, or 12 months after starting TNF inhibitor compared with MTX therapy. Nor did we observe clinically significant changes in subgroups of patients including men or those with hypertension, diabetes mellitus, or obesity. Compared with MTX starters, a lower percentage of TNF inhibitor starters had hypertension, dyslipidemia, or a history of myocardial infarction at the time they started therapy. In particular, the prevalence of prior myocardial infarction among those who received a TNF inhibitor (1.7%) was half that observed in those who received MTX (2.7%). Diagnoses of dyslipidemia and hypertension were lower in TNF inhibitor starters as well (dyslipidemia, 27.1% in TNF inhibitor and 33.1% in MTX starters; hypertension 34.6% and 41.4% respectively).
These findings suggest that those in the TNF inhibitor group were healthier than those in the MTX group at the time of the drug start, so that comparisons of physiologic function and outcomes between the two groups would not be valid without careful control for baseline status. Our rationale for using a difference-in-difference design was to reduce this potential source of bias. This highly precise control of confounding may not be possible without access to vital signs and laboratory data. These types of data often are not available in large observational datasets, such as most administrative claims databases, although they are available in the Kaiser Permanente electronic medical record. Thus, comparative effectiveness research of the indirect benefits and risks of biologic therapies on cardiovascular disease, diabetes, and other co-morbid conditions may not be as valid using traditional cohort designs where these clinical factors are not measured.
Except for diastolic blood pressure, physiologic measures did not change significantly within-person during the 6 months after starting phototherapy, which declined by an average 0.6 mg Hg; this change is not clinically significant.
We analyzed the continuation of medications to treat diabetes, hypertension, and dyslipidemia before and after therapy for psoriasis. There was no change in use of these medications before and after starting the therapy for psoriasis (data not shown).
We analyzed the continuation of medications to treat psoriasis and the relation to these physiologic measures. This was done by evaluating the level of physiologic measures in relation to the number of dispensings (1–3; 4–6; 7 or more). For all physiologic measures except LDL in the MTX cohort, there were not significant differences with number of dispensings. In the MTX dispensings of 1–3; 4–6; 7 or more, the LDL mean with standard deviation were 102.1 mg/dL (37.5), 103.7 mg/dL (37.9), and 84.7 mg/dL (21.8), respectively.
The present study does not support an earlier report in which patients with psoriasis or rheumatoid arthritis treated with a TNF inhibitor had a lower adjusted risk of new-onset diabetes mellitus (0.62 with 95% CI, 0.42–0.91) compared to those treated with nonbiologic systemic therapy other than hydroxychloroquine.24 That report may have resulted from healthy user bias. The study was based on two claims-based data systems: the British Columbia provincial health care system and a commercial US health plan. Neither of the data systems contained information from vital signs or laboratory tests; only encounters, diagnoses, procedures, and immunosuppressive drugs were available.
The present findings are consistent with our previous study, funded by the Garfield Memorial Fund of Kaiser Permanente and based in our Southern California region, in which we observed that psoriasis patients using TNF inhibitor therapy had similar risk of myocardial infarction compared with patients using non-biologic therapy for severe disease (cyclosporine, acitretin, methotrexate, or phototherapy), but only half the risk of patients using topical agents alone (p<0.01).22 At baseline, users of TNF inhibitor had relatively low prevalence of dyslipidemia and hypertension (both p<0.001) compared with non-biologic users and users of topical therapy, although they had relatively high prevalence of smoking and obesity (p<0.01). Our previous study adjusted for these baseline differences to the extent they were recorded, thereby accounting to some degree for measurable differences in risk, although it did not use each patient as their own control.
The present findings are also consistent with a Danish study of psoriasis patients in which use of TNF inhibitor was compared with use of MTX or other systemic therapies (retinoids, cyclosporine, phototherapy and/or climate therapy) with respect to the risk of a composite outcome of myocardial infarction, stroke, and cardiovascular death.21 The TNF inhibitor users (hazard ratio [HR] 0.52; 95% CI, 0.17–1.53) and MTX users (HR 0.58; 95% CI 0.29–1.15) each had lower risk for cardiovascular events than users of other therapies after adjustment for age, sex, baseline use of medication, Charlson co-morbidity index, and socio-economic status. Differences in baseline risk related to the severity of psoriasis could account for users of the other therapies having lower risk. In addition, a greater proportion of other therapy users had histories of cerebrovascular disease, chronic pulmonary disease, congestive heart failure, and myocardial infarction, with the true baseline risk of cardiovascular disease being only partially captured using the Charlson comorbidity index.
This report is consistent with past studies of physiologic measures in psoriasis patients.26–29 In psoriasis patients treated with etanercept, adalimumab, or infliximab, with each patient serving as their own control, no significant change in total cholesterol, LDL, triglycerides, SBP or DBP was observed by week 16.13 However, etanercept was associated with a significant increase in triglycerides. In two retrospective studies (6 months and 24 weeks long) comparing psoriasis patients newly treated with TNF inhibitor or methotrexate, BMI increased significantly in TNF inhibitor users, but not methotrexate users.27, 28 The 6-month study observed no association of etanercept or infliximab with changes in fasting glucose, triglycerides, or total cholesterol.27 The 24-week study observed no significant changes in fasting plasma glucose, total cholesterol, LDL, HDL, or triglycerides in any of the groups.28
Strengths of the present study include the difference-in-difference design, access to physiologic measures, and large sample size. The level of missing data was somewhat high for all outcomes except blood pressure, and this represents a limitation of the study. To be included in the study, patients needed to have the necessary relevant laboratory testing both before and after initiation of medications for psoriasis. Thus, we may have over-represented persons with elevated risk of cardiovascular and metabolic disease. However, we do not believe this would create any systemic difference between drugs we studied that would result in bias. Similar to past studies, we did not have information on disease severity. However, the study design, comparing TNF inhibitor incident users to MTX incident users, and to phototherapy incident users, likely minimized the effect of confounding by severity of psoriasis. It is possible that that healthier people are also given MTX; a patient with important comorbidities such as anemia or liver disease would not likely to receive MTX. This study didn’t assess inflammatory markers which may drive the risk of cardiovascular disease. In the subgroup analyses, it is possible that someone who had increases in the measure in question started a medication for it and thus attenuated the effects seen.
With respect to the generalizability of the study, the Kaiser Permanente adult membership is very similar to the non-Kaiser population with a health insurance plan other than Medicaid with regards to education and health, differing by having fewer White non-Hispanics and fewer members with very low and very high household incomes. The Kaiser Permanente membership is similar to the general population (including uninsured and Medicaid/Medi-Cal) with regards to race/ethnicity, but appears to be better educated, report better health, and have lower rates of smoking. Thus, the results of this study may be generalizable to the medically insured U.S. population.30
In conclusion, we found no significant differences in physiologic and metabolic measures inbetween users of TNF inhibitor and methotrexate or phototherapy for psoriasis. The key study design strengths in the present study were the careful control for baseline risk factors and the review of actual changes in numerical values rather than new-onset ICD9 codes. This was accomplished by comparing physiologic and metabolic measures within each patient before and after starting TNF inhibitor, or MTX, and phototherapy. The study does not support the use of TNF inhibitor to improve physiologic measures related to cardiovascular disease in psoriasis patients.
Supplementary Material
Acknowledgements
This research was funded by a grant from the National Institute for Allergy and Infectious Diseases, National Institutes of Health (1RC1AI086107-01). Dr. Curtis receives support from the National Institutes of Health (AR053351) and AHRQ (R01HS018517). Dr. Harrold was supported by a grant AR053856 from the National Institute for Arthritis and Musculoskeletal and Skin Diseases.
Dr. Wu received research grants from Abbott Laboratories, AbbVie, Amgen, and Pfizer, which were not directly related to this study. Dr Curtis has received grant/research support from Roche/Genentech, UCB, Centocor, CORRONA, Amgen, Pfizer, Bristol-Myers Squibb, Crescendo, and Abbott; and has been a consultant for Roche/Genentech, UCB, Centocor, CORRONA, Amgen, Pfizer, Bristol-Myers Squibb, Crescendo, and Abbott. Dr. Harrold has a research contract with the Consortium of Rheumatology Researchers of North American (CORRONA). In the last two years Abbvie, Amgen, AstraZeneca, Janssen, Genentech, Lilly, Novartis, Pfizer, Vertex and UCB have supported CORRONA through contracted subscriptions to the database. Dr. Herrinton has research contracts with Centocor, Genentech, Proctor and Gamble, Medimmune, and Pfizer. Dr. Asgari has research contracts with Genentech, Valeant, and Pfizer.
Abbreviations
- BMI
body mass index
- CI
confidence interval
- DBP
diastolic blood pressure
- HDL
high-density lipoprotein
- LDL
low-density lipoprotein
- MTX
methotrexate
- RR
relative risk
- SBP
systolic blood pressure
- TNF
tumor necrosis factor
Footnotes
Potential conflicts of interest
The other co-authors do not have any disclosures.
References
- 1.Ahlehoff O, Gislason GH, Charlot M, Jørgensen CH, Lindhardsen J, Olesen JB, Abildstrøm SZ, Skov L, Torp-Pedersen C, Hansen PR. Psoriasis is associated with clinically significant cardiovascular risk: a Danish nationwide cohort study. J Intern Med. 2011 Aug;270(2):147–157. doi: 10.1111/j.1365-2796.2010.02310.x. [DOI] [PubMed] [Google Scholar]
- 2.Ahlehoff O, Gislason GH, Jørgensen CH, Lindhardsen J, Charlot M, Olesen JB, Abildstrøm SZ, Skov L, Torp-Pedersen C, Hansen PR. Psoriasis and risk of atrial fibrillation and ischaemic stroke: a Danish Nationwide Cohort Study. Eur Heart J. 2012 Aug;33(16):2054–2064. doi: 10.1093/eurheartj/ehr285. [DOI] [PubMed] [Google Scholar]
- 3.Ahlehoff O, Gislason GH, Lindhardsen J, Charlot MG, Jørgensen CH, Olesen JB, Bretler DM, Skov L, Torp-Pedersen C, Hansen PR. Psoriasis carries an increased risk of venous thromboembolism: a Danish nationwide cohort study. PLoS One. 2011 Mar 25;6(3):e18125. doi: 10.1371/journal.pone.0018125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Wakkee M, Meijer W, Neumann HA, Herings RM, Nijsten T. Psoriasis may not be an independent predictor for the use of cardiovascular and anti-diabetic drugs: a 5-year prevalence study. Acta Derm Venereol. 2009;89(5):476–483. doi: 10.2340/00015555-0688. [DOI] [PubMed] [Google Scholar]
- 5.Wakkee M, Herings RM, Nijsten T. Psoriasis may not be an independent risk factor for acute ischemic heart disease hospitalizations: results of a large population-based Dutch cohort. J Invest Dermatol. 2010 Apr;130(4):962–967. doi: 10.1038/jid.2009.321. [DOI] [PubMed] [Google Scholar]
- 6.Qureshi AA, Choi HK, Setty AR, Curhan GC. Psoriasis and the risk of diabetes and hypertension: a prospective study of US female nurses. Arch Dermatol. 2009;145(4):379–382. doi: 10.1001/archdermatol.2009.48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Love TJ, Qureshi AA, Karlson EW, Gelfand JM, Choi HK. Prevalence of the metabolic syndrome in psoriasis: results from the National Health and Nutrition Examination Survey, 2003–2006. Arch Dermatol. 2011 Apr;147(4):419–424. doi: 10.1001/archdermatol.2010.370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Koebnick C, Black MH, Smith N, et al. The association of psoriasis and elevated blood lipids in overweight and obese children. J Pediatr. 2011;159(4):577–583. doi: 10.1016/j.jpeds.2011.03.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Gelfand JM, Dommasch ED, Shin DB, et al. The risk of stroke in patients with psoriasis. J Invest Dermatol. 2009;129(10):2411–2418. doi: 10.1038/jid.2009.112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Gelfand JM, Neimann AL, Shin DB, Wang X, Margolis DJ, Troxel AB. Risk of myocardial infarction in patients with psoriasis. JAMA. 2006;296(14):1735–1741. doi: 10.1001/jama.296.14.1735. [DOI] [PubMed] [Google Scholar]
- 11.Mehta NN, Yu Y, Pinnelas R, Krishnamoorthy P, Shin DB, Troxel AB, Gelfand JM. Attributable risk estimate of severe psoriasis on major cardiovascular events. Am J Med. 2011 Aug;124(8):775.e1–775.e6. doi: 10.1016/j.amjmed.2011.03.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Mehta NN, Azfar RS, Shin DB, Neimann AL, Troxel AB, Gelfand JM. Patients with severe psoriasis are at increased risk of cardiovascular mortality: cohort study using the General Practice Research Database. Eur Heart J. 2010 Apr;31(8):1000–1006. doi: 10.1093/eurheartj/ehp567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Neimann AL, Shin DB, Wang X, Margolis DJ, Troxel AB, Gelfand JM. Prevalence of cardiovascular risk factors in patients with psoriasis. J Am Acad Dermatol. 2006 Nov;55(5):829–835. doi: 10.1016/j.jaad.2006.08.040. [DOI] [PubMed] [Google Scholar]
- 14.Abuarbara K, Azfar RS, Shin DB, Neimann AL, Troxel AB, Gelfand JM. Cause-specific mortality in patients with severe psoriasis: a population-based cohort study in the U.K. Br J Dermatol. 2010 Sep;163(3):586–592. doi: 10.1111/j.1365-2133.2010.09941.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Langan SM, Seminara NM, Shin DB, Troxel AB, Kimmel SE, Mehta NN, Margolis DJ, Gelfand JM. Prevalence of metabolic syndrome in patients with psoriasis: a population-based study in the United Kingdom. J Invest Dermatol. 2012 Mar;132(3 Pt 1):556–562. doi: 10.1038/jid.2011.365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Cohen AD, Sherf M, Vidavsky L, Vardy DA, Shapiro J, Meyerovitch J. Association between psoriasis and the metabolic syndrome. A cross-sectional study. Dermatology. 2008;216(2):152–155. doi: 10.1159/000111512. [DOI] [PubMed] [Google Scholar]
- 17.Cohen AD, Gilutz H, Henkin Y, Zahger D, Shapiro J, Bonneh DY, Vardy DA. Psoriasis and the metabolic syndrome. Acta Derm Venereol. 2007;87(6):506–509. doi: 10.2340/00015555-0297. [DOI] [PubMed] [Google Scholar]
- 18.Gelfand JM, Yeung H. Metabolic syndrome in patients with psoriatic disease. J Rheumatol Suppl. 2012 Jul;89:24–28. doi: 10.3899/jrheum.120237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Bongartz T, Kudva Y. Can treatment of chronic inflammatory diseases reduce the risk of diabetes mellitus? JAMA. 2011 Jun 22;305(24):2573–2574. doi: 10.1001/jama.2011.884. [DOI] [PubMed] [Google Scholar]
- 20.Ryan C, Leonardi CL, Krueger JG, Kimball AB, Strober BE, Gordon KB, Langley RG, de Lemos JA, Daoud Y, Blankenship D, Kazi S, Kaplan DH, Friedewald VE, Menter A. Association between biologic therapies for chronic plaque psoriasis and cardiovascular events: a meta-analysis of randomized controlled trials. JAMA. 2011 Aug 24;306(8):864–871. doi: 10.1001/jama.2011.1211. [DOI] [PubMed] [Google Scholar]
- 21.Ahlehoff O, Skov L, Gislason G, Lindhardsen J, Kristensen SL, Iversen L, Lasthein S, Gniadecki R, Dam TN, Torp-Pedersen C, Hansen PR. Cardiovascular disease event rates in patients with severe psoriasis treated with systemic anti-inflammatory drugs: a Danish real-world cohort study. J Intern Med. 2013 Feb;273(2):197–204. doi: 10.1111/j.1365-2796.2012.02593.x. [DOI] [PubMed] [Google Scholar]
- 22.Wu JJ, Poon KT, Channual JC, Shen AY. Association between tumor necrosis factor inhibitor therapy and myocardial infarction risk in patients with psoriasis. Arch Dermatol. 2012 Nov 1;148(11):1244–1250. doi: 10.1001/archdermatol.2012.2502. [DOI] [PubMed] [Google Scholar]
- 23.Abuabara K, Lee H, Kimball AB. The effect of systemic psoriasis therapies on the incidence of myocardial infarction: a cohort study. Br J Dermatol. 2011 Nov;165(5):1066–1073. doi: 10.1111/j.1365-2133.2011.10525.x. [DOI] [PubMed] [Google Scholar]
- 24.Solomon DH, Massarotti E, Garg R, Liu J, Canning C, Schneeweiss S. Association between disease-modifying antirheumatic drugs and diabetes risk in patients with rheumatoid arthritis and psoriasis. JAMA. 2011;305(24):2525–2531. doi: 10.1001/jama.2011.878. [DOI] [PubMed] [Google Scholar]
- 25.Stern RS. Psoriasis is not a useful independent risk factor for cardiovascular disease. J Invest Dermatol. 2010 Apr;130(4):917–919. doi: 10.1038/jid.2009.446. [DOI] [PubMed] [Google Scholar]
- 26.Esposite M, Mazzotta A, Saraceno R, Schipani C, Chimenti S. Influence and variation of the body mass index in patients treated with etanercept for plaque-type psoriasis. Int J Immunopathol Pharmacol. 2009 Jan-Mar;22(1):219–225. doi: 10.1177/039463200902200124. [DOI] [PubMed] [Google Scholar]
- 27.Gisondi P, Cotena C, Tessari G, Girolomoni G. Anti-tumour necrosis factor-alpha therapy increases body weight in patients with chronic plaque psoriasis: a retrospective cohort study. J Eur Acad Dermatol Venereol. 2008;22(3):341–344. doi: 10.1111/j.1468-3083.2007.02429.x. [DOI] [PubMed] [Google Scholar]
- 28.Saraceno R, Schipani C, Mazzotta A, et al. Effect of anti-tumor necrosis factor-alpha therapies on body mass index in patients with psoriasis. Pharmacol Res. 2008;57(4):290–295. doi: 10.1016/j.phrs.2008.02.006. [DOI] [PubMed] [Google Scholar]
- 29.Renzo LD, Saraceno R, Schipani C, Rizzo M, Bianchi A, Noce A, Esposito M, Tiberti S, Chimenti S, DE Lorenzo A. Prospective assessment of body weight and body composition changes in patients with psoriasis receiving anti-TNF-α treatment. Dermatol Ther. 2011 Jul-Aug;24(4):446–451. doi: 10.1111/j.1529-8019.2011.01439.x. [DOI] [PubMed] [Google Scholar]
- 30.Gordon NP. How Does the Adult Kaiser Permanente Membership in Northern California Compare with the Larger Community? Oakland, CA: Kaiser Permanente Division of Research; 2006. Jun, Available from: http://www.dor.kaiser.org/dor/mhsnet/public/kpnc_community.htm. [Google Scholar]
- 31.Asgari MM, Wu JJ, Gelfand JM, Salman C, Curtis JR, Harrold LR, et al. Validity of diagnostic codes and prevalence of psoriasis and psoriatic arthritis in a managed care population, 1996–2009. Pharmacoepidemiol Drug Saf. 2013 May;2 doi: 10.1002/pds.3447. [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.