Abstract
Introduction
Lysophosphatidic acid (LPA) is a bioactive lipid that binds to G protein–coupled receptors (LPA1–6). Recently, we reported that abrogation of LPA receptor 1 (LPA1) ameliorated murine collagen-induced arthritis, probably via inhibition of inflammatory cell migration, Th17 differentiation and osteoclastogenesis. In this study, we examined the importance of the LPA–LPA1 axis in cell proliferation, cytokine/chemokine production and lymphocyte transmigration in fibroblast-like synoviocytes (FLSs) obtained from the synovial tissues of rheumatoid arthritis (RA) patients.
Methods
FLSs were prepared from synovial tissues of RA patients. Expression of LPA1–6 was examined by quantitative real-time RT-PCR. Cell surface LPA1 expression was analyzed by flow cytometry. Cell proliferation was analyzed using a cell-counting kit. Production of interleukin 6 (IL-6), vascular endothelial growth factor (VEGF), chemokine (C-C motif) ligand 2 (CCL2), metalloproteinase 3 (MMP-3) and chemokine (C-X-C motif) ligand 12 (CXCL12) was measured by enzyme-linked immunosorbent assay. Pseudoemperipolesis was evaluated using a coculture of RA FLSs and T or B cells. Cell motility was examined by scrape motility assay. Expression of adhesion molecules was determined by flow cytometry.
Results
The expression of LPA1 mRNA and cell surface LPA1 was higher in RA FLSs than in FLSs from osteoarthritis tissue. Stimulation with LPA enhanced the proliferation of RA FLSs and the production of IL-6, VEGF, CCL2 and MMP-3 by FLSs, which were suppressed by an LPA1 inhibitor (LA-01). Ki16425, another LPA1 antagonist, also suppressed IL-6 production by LPA-stimulated RA FLSs. However, the production of CXCL12 was not altered by stimulation with LPA. LPA induced the pseudoemperipolesis of T and B cells cocultured with RA FLSs, which was suppressed by LPA1 inhibition. In addition, LPA enhanced the migration of RA FLSs and expression of vascular cell adhesion molecule and intercellular adhesion molecule on RA FLSs, which were also inhibited by an LPA1 antagonist.
Conclusions
Collectively, these results indicate that LPA–LPA1 signaling contributes to the activation of RA FLSs.
Introduction
Rheumatoid arthritis (RA) is a chronic inflammatory disease characterized by synovial hyperplasia with proliferation of fibroblast-like synoviocytes (FLSs), angiogenesis, infiltration of inflammatory cells such as lymphocytes and macrophages, and bone destruction of multiple joints [1]. FLSs are especially responsible for inflammation through cytokine and chemokine production and are also key cells of the invasive synovium, suggesting that they play a major role in the initiation and perpetuation of the destruction of inflamed joints [2].
Lysophosphatidic acid (LPA) is a bioactive lipid that binds to its specific cell surface G protein–coupled receptors (LPA1–6). LPA is generated via the hydrolysis of lysophosphatidylcholine by a secretory protein, autotaxin (ATX), which exhibits lysophospholipase D activity [3]. ATX was shown to be highly expressed in tumor cells, including neuroblastoma, breast cancer and renal cell carcinoma [4-6]. Moreover, LPA was reported to induce the production of interleukin 8 (IL-8) and vascular endothelial growth factor (VEGF) by cancer cells, angiogenesis and cancer growth [7-11].
It has previously been shown that expression of ATX by FLSs in the RA synovium and concentration of ATX in the RA synovial fluid are increased [12]. In addition, LPA1–3 mRNA has been reported to be expressed in RA FLSs, and incubation with LPA induced cell motility and cytokine expression by the FLSs, indicating that LPA may contribute to the pathogenesis of RA by stimulation of FLSs [13,14]. We recently demonstrated that treatment with an LPA receptor 1 (LPA1) antagonist, LA-01, ameliorated murine collagen-induced arthritis, probably via inhibition of inflammatory cell migration, Th17 differentiation and osteoclastogenesis [15].
In this study, we extensively analyzed the stimulatory effects of LPA for RA FLSs, as well as the effects of an LPA1 antagonist, LA-01, against this stimulation.
Methods
Specimens
Synovial tissues were obtained from RA patients (n = 10) who fulfilled American College of Rheumatology criteria [16] and from patients with osteoarthritis (OA) (n = 5). RA patients were a median (range) of 67 years old (45 to 80), and had a disease duration of 14 years (2 to 30) and C-reactive protein level of 0.68 mg/dl (0.0 to 2.85). Seven patients (70%) were positive for rheumatoid factor, and seven (70%) were positive for anticitrullinated protein antibodies. All patients provided informed consent. The experimental protocol was approved by the ethics committee of the Tokyo Medical and Dental University.
Fibroblast-like synoviocytes
Synovial tissues from RA patients were minced and incubated with 0.5 mg/ml collagenase (Sigma-Aldrich, St Louis, MO, USA) for 1 hour at 37°C, then passed through a metal screen to obtain single-cell suspensions. Harvested cells were plated in cell culture plates and incubated with Dulbecco’s modified Eagle’s medium (DMEM) (Sigma-Aldrich) supplemented with 10% fetal calf serum (FCS) (Sigma-Aldrich). Adherent cells were maintained in the medium as FLSs and were used after five passages in the following experiments [17].
RT-PCR
Total RNA was prepared from the FLSs of RA tissue (n = 10) and OA synovial tissue (n = 5), and first-strand cDNA was synthesized. Quantitative real-time RT-PCR was performed as described previously [18]. cDNA was amplified with primers for LPA1 (sense, 5′-ACC CAA TAC TCG GAG ACT GAC TGT-3′; antisense, 5′-CGT CAG GCT GGT GTC AAT GA-3′), LPA2 (sense, 5′-TCA TCA TGG GCC AGT GCT ACT-3′; antisense, 5′-GTG GGA GCT GAG CTC TTT GC-3′), LPA3 (sense, 5′-CTT GAC TGC TTC CCT CAC CAA-3′; antisense, 5′-CGC ATC CTC ATG ATT GAC ATG-3′), LPA4 (sense, 5′-TCC TCA GTG GCG GTA TTT CAG-3; antisense, 5′-AAG CAG GTG GTG GTT GCA TT-3′), LPA5 (sense, 5′-GGT GGT GAG CGT GTA CAT GTG T-3′; antisense, 5′-AGT GGT GCA GTG CGT AG TAG GA-3′), LPA6 (sense, 5′-AGA ACC AAA AGA AAT GCA AAG ATT G-3′; antisense, 5′-ACG GCG GGT GCA CTT C-3′) and 18S rRNA (sense, 5′-AAC CAG ACA AAT CGC TCC AC-3′; antisense, 5′-ACT CAA CAC GGG AAA CCT CA-3′). 18S rRNA was used as an internal control to standardize the amount of sample mRNA, and the relative expression of real-time PCR products was determined.
Cell surface expression of lysophosphatidic acid receptor 1 on fibroblast-like synoviocytes
FLSs were stained with anti-LPA1 monoclonal antibody (mAb) (1G6; LSBio, Seattle, WA, USA) as a first antibody, and phycoerythrin-conjugated anti-mouse immunoglobulin G (IgG) antibody (BioLegend, San Diego, CA, USA) as a second antibody. Mouse IgG2b (BioLegend) was used as an isotype control. Cells were then analyzed by flow cytometry (FACSCalibur; BD Biosciences, San Jose, CA, USA).
Proliferation assay
FLSs were plated at a density of 2 × 103 cells/well in 96-well flat-bottom plates. Cells were incubated with a selective LPA1 antagonist (LA-01 (0, 1 or 10 nM); provided by Ono Pharmaceutical, Osaka, Japan) [15,19] for 30 minutes and then stimulated with LPA (Cayman Chemical, Ann Arbor, MI, USA) (0, 1 or 10 μM) in FCS-free DMEM at 37°C for 72 hours. The proliferation of FLSs was measured by using a cell-counting kit with WST-8 (2-(2-methoxy-4-nitrophenyl)-3-(4-nitrophenyl)-5-(2,4-disulfophenyl)-2H-tetrazolium, monosodium salt; Dojindo, Kumamoto, Japan) according to the manufacturer’s protocol. LPA1, LPA2 and LPA3 share 50% to 57% amino acid identity in humans and comprise the endothelial cell differentiation gene (Edg) family of LPA receptors [20]. The half-maximal inhibitory concentration (IC50) of LA-01 was 0.086, 2.8 and 0.90 μmol/L for LPA1, LPA2 and LPA3, respectively, which was determined by LPA1-, LPA2- or LPA3-transfected CHO cells [15,19]. LPA4–6 receptors have been classified into the non-Edg family of LPA receptors and are structurally distant from the Edg family of LPA receptors [20]. The IC50 of LA-01 for LPA4–6 was not determined. Incubation with LA-01 did not affect viability of the FLSs (data not shown).
Enzyme-linked immunosorbent assay
RA FLSs were cultured overnight in 96-well plates (2 × 104 cells/well), then incubated with LA-01 (0, 1 or 10 nM) or Ki16425 (2 nM) (Cayman Chemical) 30 minutes before stimulation with LPA (10 μM) in FCS-free DMEM at 37°C for 24 hours. Protein levels of IL-6, chemokine (C-C motif) ligand 2 (CCL2), VEGF, matrix metalloproteinase 3 (MMP-3) and chemokine (C-X-C motif) ligand 12(CXCL12) in the culture supernatant were assessed by using ELISA kits (R&D Systems, Minneapolis, MN, USA) according to the instructions supplied by the manufacturer.
Pseudoemperipolesis
FLSs were seeded onto 96-well plates (2 × 104 cells/well) and cultured for 48 hours. CD4- and CD8-positive (CD4+ and CD8+, respectively) T cells and CD19+ B cells were purified from human peripheral blood of healthy volunteers by using MACS microbeads (>95% purity; Miltenyi Biotec, Auburn, CA, USA) and added to the FLS-cultured wells (1 × 105 cells/well). The cells were treated with LA-01 (0, 1 or 10 nM) for 30 minutes, followed by stimulation with LPA (10 μM) in FCS-free DMEM. After 12 hours, the wells were washed three times with medium. Pseudoemperipolesis was assessed by counting the number of cells beneath FLSs in three independent fields under a microscope.
Scrape motility assay
RA FLSs were plated at a density of 1 × 105 cells/ml in 12-well plates in DMEM with 10% FCS. After overnight incubation, FLSs was washed twice with FCS-free medium. The tip of a plastic pipette was drawn across the center of the well to produce a scraped area. Culture wells were washed twice with PBS, and free cells were removed. After pretreatment with LA-01 (0, 1 or 10 nM) for 30 minutes, cells were incubated with LPA (10 μM) in FCS-free DMEM. A cell-free area was measured by using ImageJ software (National Institutes of Health, Bethesda, MD, USA) at 0 and 48 hours, and the ratio was then calculated (cell-free area at 48 hours per cell-free area at 0 hours).
Expression of vascular cell adhesion molecule and intercellular adhesion molecule on RA fibroblast-like synoviocytes
FLSs were stimulated with LPA (10 μM) 30 minutes after adding LA-01 (0, 1 or 10 nM) in FCS-free DMEM at 37°C for 12 hours. Cells were stained with allophycocyanin-conjugated mAb against vascular cell adhesion molecule (anti-VCAM, clone STA; BioLegend) or phycoerythrin-conjugated mAb against intracellular adhesion molecule (anti-ICAM, clone HA58; eBioscience, San Diego, CA, USA). Allophycocyanin- or phycoerythrin-conjugated mouse IgG1 (BioLegend) was used as an isotype control. Cells were then analyzed by flow cytometry (Accuri C6 Flow Cytometer; BD Biosciences).
Statistical analysis
Data are expressed as mean ± standard error of the mean (SEM). The comparison of the data from the two groups was conducted by using Student’s t-test. P-values less than 0.05 were considered significant.
Results
Expression of lysophosphatidic acid receptors in RA fibroblast-like synoviocytes
The expression of LPA1–6 mRNA in FLSs from RA and OA patients was analyzed by quantitative real-time RT-PCR. The expression of LPA1 mRNA in RA FLSs was significantly higher than that in OA FLSs (Figure 1A). The expression of LPA3 and LPA4 was also significantly higher in RA FLSs than that in OA FLSs, although the ratios of LPA3 and LPA4 expression in RA FLSs to OA FLSs were smaller than those of LPA1 expression. Cell surface LPA1 expression was analyzed by flow cytometry. RA FLSs were expressed LPA1 on the cell surface, and the expression level was substantially higher than that of OA FLSs (Figure 1B).
Figure 1.
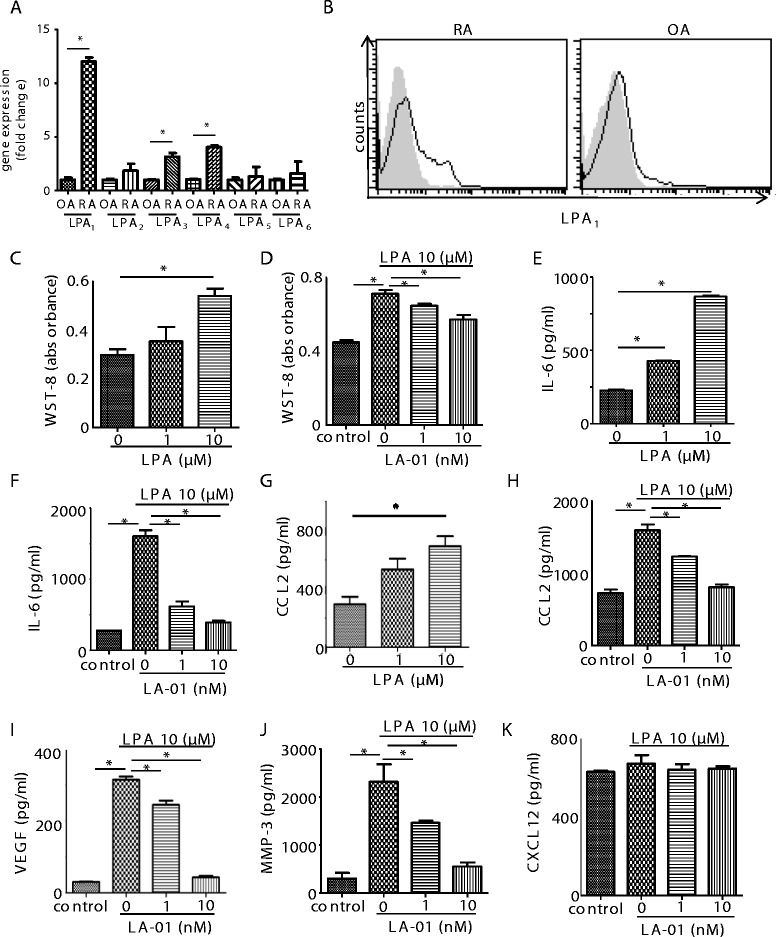
Expression of lysophosphatidic acid receptors and the effect of lysophosphatidic acid receptor 1 on proliferation and production of inflammatory mediators in rheumatoid arthritis fibroblast-like synoviocytes. The expression levels of lysophosphatidic acid receptor 1 through 6 (LPA1–6) mRNA in fibroblast-like synoviocytes (FLSs) derived from the rheumatoid arthritis (RA) synovium (n = 10) were compared to those in FLSs from osteoarthritis (OA) synovium (n = 5) by real-time RT-PCR (A). Data were derived from samples from multiple individuals. Data are presented as the mean ± SEM. *P < 0.05 for RA vs OA. Cell surface expression of LPA1 on RA (n = 5) and OA (n = 3) FLSs was analyzed by flow cytometry (B). Filled histogram (gray): isotype control; open histogram (black line): LPA1. Representative histograms are shown. RA FLSs were cultured with lysophosphatidic acid (LPA) for 72 hours (C). FLSs were preincubated with an LPA1 inhibitor, LA-01, for 30 minutes,then stimulated with 10 μM LPA for 72 hours (D). Control: no stimulation with LPA. Cell proliferation was measured by using a cell counting kit (C) and (D). RA FLSs were cultured with LPA for 24 hours. Concentrations of interleukin 6 (IL-6) and chemokine (C-C motif) ligand 2 (CCL2) in the culture supernatant were measured by enzyme-linked immunosorbent assay (ELISA) (E) and (G). FLSs were preincubated with LA-01 for 30 minutes, then stimulated with 10 μM LPA for 24 hours. Concentrations of IL-6, CCL2, vascular endothelial growth factor (VEGF), matrixmetalloproteinase (MMP-3) and CXCL12 in the culture supernatant were measured by ELISA (F), and (H) through (K). Control: no stimulation with LPA. Data are presented as the means (±SEM) of one of three independent experiments analyzed in triplicate. *P < 0.05 vs control or LA-01 0 nM (C) through (K).
Lysophosphatidic acid receptor 1 inhibitor suppressed lysophosphatidic acid–induced proliferation and cytokine production in RA fibroblast-like synoviocytes
We analyzed the effects of LPA on the proliferation and production of inflammatory mediators by RA FLSs. Stimulation with LPA dose-dependently induced the proliferation of FLSs (Figure 1C). LPA stimulation also induced the production of IL-6 and CCL2 from FLSs in a dose-dependent manner (Figures 1E and 1G), which supports a previous report that LPA upregulated IL-6 mRNA expression by RA FLSs [18]. Stimulation with LPA also induced the production of VEGF and MMP-3 by RA FLSs in vitro (Figures 1I and 1J).
Next, we analyzed the effect of an LPA1 inhibitor on LPA stimulation for RA FLSs. Enhanced cell proliferation by 10 μM LPA was significantly suppressed by LA-01, the LPA1-selective antagonist (Figure 1D). The treatment with LA-01 significantly reduced the production of IL-6, CCL2, VEGF and MMP-3 by LPA-stimulated RA FLSs (Figures 1F and 1H through 1J). In contrast, the production of CXCL12 by RA FLSs was not altered by stimulation with LPA (Figure 1K). We used Ki16425, another LPA1 antagonist, to confirm the effects of LPA1 inhibition on IL-6 production from LPA-stimulated RA FLSs. Incubation with Ki16425 suppressed IL-6 production from LPA-stimulated RA FLSs as well as LA-01 (IL-6 concentrations: vehicle = 299.413 ± 28.084 pg/ml; Ki16425 = 116.785 ± 11.162 pg/ml (P < 0.05 vs vehicle); LA-01 = 145.715 ± 15.921 pg/ml (P < 0.05 vs vehicle)). These results suggest that LPA–LPA1 signaling plays important roles in proliferation and cytokine production of RA FLSs in vitro.
LPA–LPA1 signaling promoted pseudoemperipolesis
RA FLSs have been shown to promote the spontaneous migration of leukocytes beneath them, a process termed pseudoemperipolesis [21]. We examined the effect of LPA on pseudoemperipolesis. Stimulation with 10 μM LPA significantly increased the number of CD4+ and CD8+ T cells, as well as CD19+ B cells, beneath RA FLSs (Figures 2A to 2F). Moreover, incubation with LA-01 suppressed the LPA-enhanced pseudoemperipolesis of CD4+ and CD8+ T and CD19+ B cells (Figures 2A through 2F), suggesting that interaction of LPA and LPA1 promotes pseudoemperipolesis of leukocytes.
Figure 2.
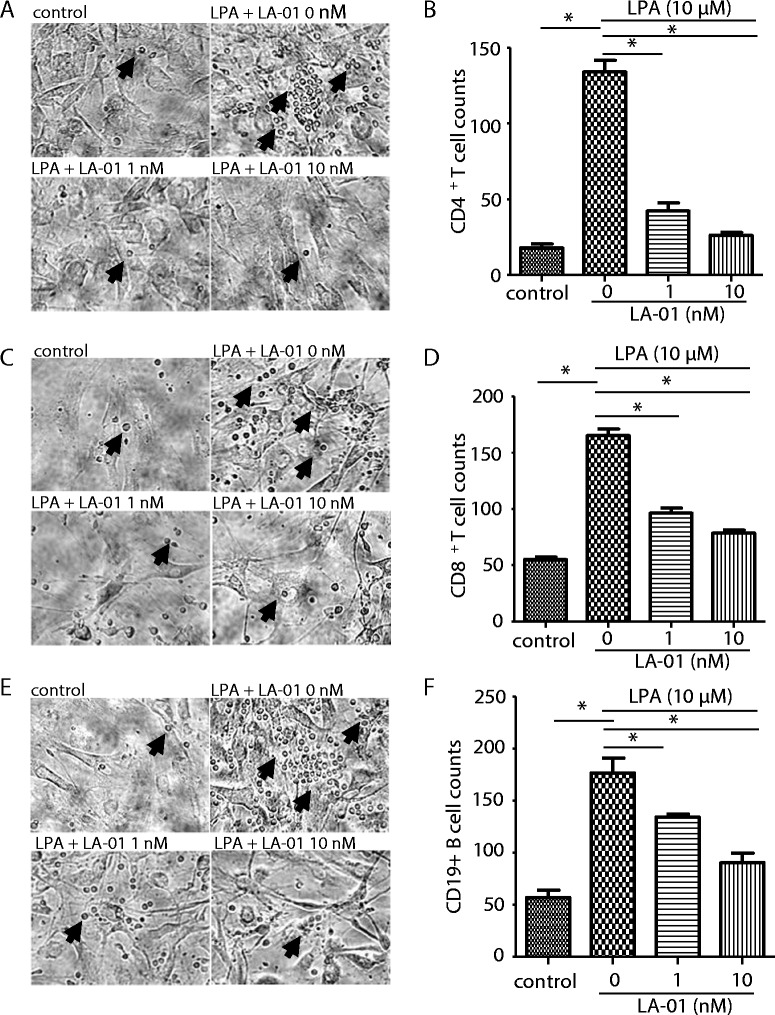
Effect of lysophosphatidic acid receptor 1 on pseudoemperipolesis and migration of rheumatoid arthritis fibroblast-like synoviocytes. After preincubation of cocultured rheumatoid arthritis (RA) fibroblast-like synoviocytes (FLSs) and CD4+ T cells (A) and (B) or CD8+ T cells (C) and (D) or CD19+ B cells (E) and (F) with a lysophosphatidic acid (LPA) receptor 1 inhibitor (LA-01; 0, 1 or 10 nM) for 30 minutes, the cells were stimulated with 10 μM LPA for 12 hours. Control: no stimulation with LPA. After the cells were washed, the number of lymphocytes beneath FLSs was counted. Representative photomicrographs of three independent experiments are shown (A, C and E). Arrows indicate the lymphocytes beneath FLSs. Original magnification, ×200. Data on the number of lymphocytes beneath FLSs are presented as one of three independent experiments analyzed in triplicate (B, D, and F). Data are presented as the mean ± SEM. *P < 0.05 vs control or LA-01 0 nM (B, D, F).
LPA–LPA1 signaling promoted cell motility of RA fibroblast-like synoviocytes
We also analyzed the effect of LPA1 on RA FLS migration by scrape motility assay. Incubation with 10 μM LPA significantly decreased the cell-free area, indicating that LPA induced cell migration in vitro (Figures 3A and 3B), as reported previously [22]. In addition, LA-01 significantly increased the cell-free area of RA FLSs (Figures 3A and 3B), suggesting that LPA–LPA1 signaling also contributes to the promotion of RA FLS motility.
Figure 3.

The effect of lysophosphatidic acid receptor 1 on the migration of rheumatoid arthritis fibroblast-like synoviocytes. A scraped cell-free area was created on cultured RA FLSs. After preincubation with a lysophosphatidic acid (LPA) receptor 1 inhibitor (LA-01; 0, 1 or 10 nM) for 30 minutes, cells were stimulated with 10 μM LPA for 48 hours. Control: no stimulation with LPA. (A) Representative photomicrographs of three independent experiments are shown. Original magnification, ×40. (B) The cell-free area was assessed, and a ratio (cell-free area in 48 hours to cell-free area in 0 hours) was defined. Data are presented as the means (±SEM) of one of three independent experiments analyzed in triplicate. *P < 0.05 vs control or LA-01 0 nM. Upper dashed line indicates the cells are stimulated with LPA 10 uM, and lower dashed line indicates LA-01 is added with indicated concentration.
LPA–LPA1 signaling induced adhesion molecule expression on RA fibroblast-like synoviocytes
It has been reported that signaling from VCAM and ICAM in RA FLSs supports pseudoemperipolesis [21]. Therefore, we next analyzed the expression of VCAM and ICAM on RA FLSs by flow cytometry. We found that stimulation with 10 μM LPA induced the expression of VCAM and ICAM on RA FLSs (Figure 4). Moreover, LA-01 decreased the expression of VCAM and ICAM induced by LPA on RA FLSs (Figure 4). However, the expression of E-selectin on RA FLSs was not altered by LPA simulation (data not shown).
Figure 4.
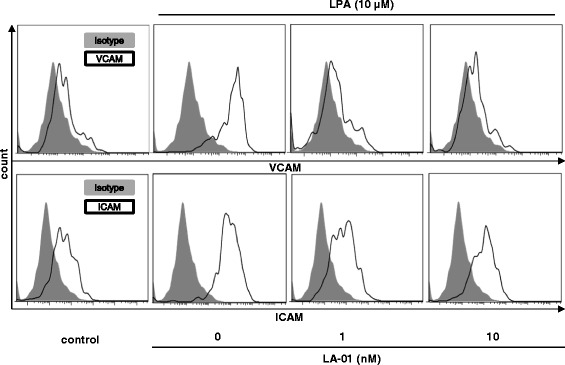
The effect of lysophosphatidic acid receptor 1 on the expression of adhesion molecules on fibroblast-like synoviocytes. Rheumatoid arthritis (RA) fibroblast-like synoviocytes (FLSs) were pretreated with a lysophosphatidic acid (LPA) receptor 1 inhibitor (LA-01; 0, 1 or 10 nM) for 30 minutes, then the cells were stimulated with 10 μM LPA for 12 hours. Cells were stained with allophycocyanin-conjugated monoclonal antibody (mAb) against vascular cell adhesion molecule (anti-VCAM) or phycoerythrin-conjugated mAb against intercellular adhesion molecule (anti-ICAM). Allophycocyanin- or phycoerythrin-conjugated mouse immunoglobulin G1 (IgG1) was used as a control. The expression of VCAM and ICAM on FLSs was analyzed by flow cytometry. Filled histogram (gray): isotype control; open histogram (black line): VCAM or ICAM.
Discussion
In this study, we found that LPA1 was highly expressed in RA FLSs. LPA stimulated RA FLSs to enhance proliferation, production of inflammatory mediators, pseudoemperipolesis, migration and the expression of adhesion molecules, which are attributable to signaling through LPA1.
RA FLSs express inflammatory cytokines, chemokines and matrix-degrading enzymes, which contribute to the pathogenesis of RA. LPA has been reported to induce IL-6 mRNA expression on RA FLSs, as well as cell motility [13]. However, the corresponding LPA receptor on RA FLSs has not been identified. We show that LPA augmented IL-6, CCL2, VEGF and MMP-3 production by RA FLSs. Moreover, the LPA-induced production of the inflammatory mediators was inhibited by a LPA1-selective inhibitor. Therefore, the LPA–LPA1 cascade plays an important role in cytokine, chemokine and matrix-degrading enzyme production by RA FLSs. Although IC50 of LA-01 was 86 nM, which was determined by using LPA1-transfected CHO cells, 10 nM LA-01 significantly inhibited stimulation of LPA in RA FLSs. The IC50 may be dependent on cell type or on the expression level of LPA1.
Pseudoemperipolesis contributes to the chronic inflammation induced by lymphocyte recruitment in the inflamed joints and protects lymphocytes from apoptosis [21,23,24]. We show that LPA enhanced the pseudoemperipolesis of T and B cells, which is also attributable to LPA1. It has been reported that stimulation with CXCL12 and signaling from VCAM and ICAM in RA FLSs support pseudoemperipolesis [21]. Our results indicate that LPA upregulated the expression of VCAM and ICAM on RA FLSs, which was blocked by the LPA1 antagonist. Thus, LPA may enhance pseudoemperipolesis via the upregulation of VCAM and ICAM expression on RA FLSs through LPA1. Interestingly, CXCL12 production by RA FLSs was not altered by LPA simulation. Stimulation of lymphocytes by LPA via LPA1 may also contribute to the enhanced pseudoemperipolesis. In this regard, it has been reported that LPA induced chemokinesis in T cells [25] and lymphocyte transmigration through high endothelial venules [26,27]. Further studies are needed to clarify the effects of LPA–LPA1 signaling for the lymphocytes on pseudoemperipolesis.
The hyperplastic rheumatoid pannus is characterized by an overabundance of FLSs [2]. This cellular excess stems largely from an imbalance between the proliferation and apoptosis of FLSs [2]. The migration of RA FLSs may also contribute to pannus formation [2]. Our results show that LPA induced the proliferation and migration of FLSs, which was inhibited by the LPA1 antagonist. Moreover, in a recent study, researchers reported that LPA suppressed tumor necrosis factor–induced apoptosis on RA FLSs via LPA1 [28]. Therefore, it is suggested that the LPA–LPA1 signaling also contributed to the cellular excess and migration of FLSs in the RA synovium.
In this study, we show that there are important roles of LPA–LPA1 signaling on RA FLS stimulation. However, the effects of LPA signals via LPA2–6 remain unclear, although RA FLSs also expressed LPA2–6. Further studies are warranted to elucidate the roles of LPA2–6 in LPA stimulation of FLSs by using each of the LPA receptor–specific antagonists or FLSs from each LPA receptor–deficient mouse.
It was shown that conditional genetic ablation of ATX, which generates LPA via hydrolysis of lysophosphatidylcholine, in mesenchymal cells resulted in disease attenuation in animal models of arthritis [12]. We have also found that LPA1 is essential for the development of arthritis in collagen-induced arthritis [15]. The ATX–LPA–LPA1 axis may play an important role in the development of arthritis.
Conclusion
Our study suggests that LPA–LPA1 signaling in FLSs may contribute to the pathogenesis of RA by inducing proliferation, production of inflammatory mediators, pseudoemperipolesis and migration on RA FLSs. Thus, LPA1 could be a promising therapeutic target for RA.
Acknowledgements
We thank Prof. Timothy J Wright (Otsuma Women’s University, Tokyo, Japan) for his helpful advice and for improving the readability of this manuscript. This work was supported in part by the Japanese Ministry of Education, Global Center of Excellence (GCOE) Program, International Research Center for Molecular Science in Tooth and Bone Diseases, and Takeda Science Foundation.
Abbreviations
- ATX
Autotaxin
- CCL2
Chemokine (C-C motif) ligand 2
- CXCL12
Chemokine (C-X-C motif) ligand 12
- DMEM
Dulbecco’s modified Eagle’s medium
- Edg
Endothelial cell differentiation gene
- ELISA
Enzyme-linked immunosorbent assay
- FCS
Fetal calf serum
- FLS
Fibroblast-like synoviocyte
- ICAM
Intercellular adhesion molecule
- IL
Interleukin
- LPA
Lysophosphatidic acid
- mAb
Monoclonal antibody
- MMP-3
Metalloproteinase 3
- OA
Osteoarthritis
- RA
Rheumatoid arthritis
- SEM
Standard error of the mean
- VCAM
Vascular cell adhesion molecule
- VEGF
Vascular endothelial growth factor
Footnotes
Competing interests
The authors declare that they have no competing interests.
Authors’ contributions
YM participated in the design of the study, carried out the experiments and statistical analysis and drafted the manuscript. CM and CS assisted in carrying out the experiments and with manuscript preparation. YI assisted in data interpretation and manuscript preparation. WY and KS collected the clinical materials and assisted in data interpretation and manuscript preparation. MH, MM, NM and TN conceived of the study, participated in its design and coordination, and helped to draft the manuscript. All authors read and approved the final manuscript.
Contributor Information
Yoshishige Miyabe, Email: miyabe.rheu@tmd.ac.jp.
Chie Miyabe, Email: cmiyabe@partners.org.
Yoshiko Iwai, Email: yiwai@med.uoeh-u.ac.jp.
Waka Yokoyama, Email: yokoyama.rheu@tmd.ac.jp.
Chiyoko Sekine, Email: csekine@med.teikyo-u.ac.jp.
Kazutaka Sugimoto, Email: sugimoto@sonodakai.or.jp.
Masayoshi Harigai, Email: mharigai.mpha@tmd.ac.jp.
Masayuki Miyasaka, Email: mmiyasak@orgctl.med.osaka-u.ac.jp.
Nobuyuki Miyasaka, Email: miya.rheu@tmd.ac.jp.
Toshihiro Nanki, Email: nanki@med.teikyo-u.ac.jp.
References
- 1.Kinne RW, Bräuer R, Stuhlmüller B, Palombo-Kinne E, Burmester GR. Macrophages in rheumatoid arthritis. Arthritis Res. 2000;2:189–202. doi: 10.1186/ar86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Bottini N, Firestein GS. Duality of fibroblast-like synoviocytes in RA: passive responders and imprinted aggressors. Nat Rev Rheumatol. 2013;9:24–33. doi: 10.1038/nrrheum.2012.190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Aoki J. Mechanisms of lysophosphatidic acid production. Semin Cell Dev Biol. 2004;15:477–489. doi: 10.1016/j.semcdb.2004.05.001. [DOI] [PubMed] [Google Scholar]
- 4.Kawagoe H, Stracke ML, Nakamura H, Sano K. Expression and transcriptional regulation of the PD-Iα/autotaxin gene in neuroblastoma. Cancer Res. 1997;57:2516–2521. [PubMed] [Google Scholar]
- 5.Yang SY, Lee J, Park CG, Kim S, Hong S, Chung HC, Min SK, Han JW, Lee HW, Lee HY. Expression of autotaxin (NPP-2) is closely linked to invasiveness of breast cancer cells. Clin Exp Metastasis. 2002;19:603–608. doi: 10.1023/A:1020950420196. [DOI] [PubMed] [Google Scholar]
- 6.Stassar MJ, Devitt G, Brosius M, Rinnab L, Prang J, Schradin T, Simon J, Petersen S, Kopp-Schneider A, Zöller M. Identification of human renal cell carcinoma associated genes by suppression subtractive hybridization. Br J Cancer. 2001;85:1372–1382. doi: 10.1054/bjoc.2001.2074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Umezu-Goto M, Kishi Y, Taira A, Hama K, Dohmae N, Takio K, Yamori T, Mills GB, Inoue K, Aoki J, Arai H. Autotaxin has lysophospholipase D activity leading to tumor cell growth and motility by lysophosphatidic acid production. J Cell Biol. 2002;158:227–233. doi: 10.1083/jcb.200204026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Liu S, Umezu-Goto M, Murph M, Lu Y, Liu W, Zhang F, Yu S, Stephens LC, Cui X, Murrow G, Coombes K, Muller W, Hung MC, Perou CM, Lee AV, Fang X, Milles GB. Expression of autotaxin and lysophosphatidic acid receptors increases mammary tumorigenesis, invasion, and metastases. Cancer Cell. 2009;15:539–550. doi: 10.1016/j.ccr.2009.03.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Masuda A, Nakamura K, Izutsu K, Igarashi K, Ohkawa R, Jona M, Higashi K, Yokota H, Okudaira S, Kishimoto T, Watanabe T, Koike Y, Ikeda H, Kozai Y, Kurokawa M, Aoki J, Yatomi Y. Serum autotaxin measurement in haematological malignancies: a promising marker for follicular lymphoma. Br J Haematol. 2008;143:60–70. doi: 10.1111/j.1365-2141.2008.07325.x. [DOI] [PubMed] [Google Scholar]
- 10.Schleicher SM, Thotala DK, Linkous AG, Hu R, Leahy KM, Yazlovitskaya EM, Hallahan DE. Autotaxin and LPA receptors represent potential molecular targets for the radiosensitization of murine glioma through effects on tumor vasculature. PLoS One. 2011;6:e22182. doi: 10.1371/journal.pone.0022182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Houben AJ, Moolenaar WH. Autotaxin and LPA receptor signaling in cancer. Cancer Metastasis Rev. 2011;30:557–565. doi: 10.1007/s10555-011-9319-7. [DOI] [PubMed] [Google Scholar]
- 12.Nikitopoulou I, Oikonomou N, Karouzakis E, Sevastou I, Nikolaidou-Katsaridou N, Zhao Z, Mersinias V, Armaka M, Xu Y, Masu M, Milles GB, Gay S, Kollias G, Aidinis V. Autotaxin expression from synovial fibroblasts is essential for the pathogenesis of modeled arthritis. J Exp Med. 2012;209:925–933. doi: 10.1084/jem.20112012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kehlen A, Lauterbach R, Santos AN, Thiele K, Kabisch U, Weber E, Riemann D, Langner J. IL-1β- and IL-4-induced down-regulation of autotaxin mRNA and PC-1 in fibroblast-like synoviocytes of patients with rheumatoid arthritis (RA) Clin Exp Immunol. 2001;123:147–154. doi: 10.1046/j.1365-2249.2001.01432.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Bourgoin SG, Zhao C. Autotaxin and lysophospholipids in rheumatoid arthritis. Curr Opin Investig Drugs. 2010;11:515–526. [PubMed] [Google Scholar]
- 15.Miyabe Y, Miyabe C, Iwai Y, Takayasu A, Fukuda S, Yokoyama W, Nagai J, Jona M, Tokuhara Y, Ohkawa R, Albers HM, Ovaa H, Aoki J, Chun J, Yatomi Y, Ueda H, Miyasaka M, Miyasaka N, Nanki T. Necessity of lysophosphatidic acid receptor 1 for development of arthritis. Arthritis Rheum. 2013;65:2037–2047. doi: 10.1002/art.37991. [DOI] [PubMed] [Google Scholar]
- 16.Cader MZ, Filer A, Hazlehurst J, de Pablo P, Buckley CD, Raza K. Performance of the 2010 ACR/EULAR criteria for rheumatoid arthritis: comparison with 1987 ACR criteria in a very early synovitis cohort. Ann Rheum Dis. 2011;70:949–955. doi: 10.1136/ard.2010.143560. [DOI] [PubMed] [Google Scholar]
- 17.Kaneko K, Miyabe Y, Takayasu A, Fukuda S, Miyabe C, Ebisawa M, Yokoyama W, Watanabe K, Imai T, Muramoto K, Terashima Y, Sugihara T, Matsushima K, Miyasaka N, Nanki T. Chemerin activates fibroblast-like synoviocytes in patients with rheumatoid arthritis. Arthritis Res Ther. 2011;13:R158. doi: 10.1186/ar3475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Watanabe K, Penfold ME, Matsuda A, Ohyanagi N, Kaneko K, Miyabe Y, Matsumoto K, Schall TJ, Miyasaka N, Nanki T. Pathogenic role of CXCR7 in rheumatoid arthritis. Arthritis Rheum. 2010;62:3211–3220. doi: 10.1002/art.27650. [DOI] [PubMed] [Google Scholar]
- 19.Tanaka M, Nakaide S, Takaoka Y: Compounds having lysophosphatidic acid receptor antagonism and uses thereof. Patent Version Number: WO/2005/058790 (2005-06-30) [http://patentscope.wipo.int/search/en/WO2005058790] (accessed 9 October 2014).
- 20.Ishii S, Noguchi K, Yanagida K. Non-Edg family lysophosphatidic acid (LPA) receptors. Prostaglandins Other Lipid Mediat. 2009;89:57–65. doi: 10.1016/j.prostaglandins.2009.06.001. [DOI] [PubMed] [Google Scholar]
- 21.Bradfield PF, Amft N, Vernon-Wilson E, Exley AE, Parsonage G, Rainger GE, Nash GB, Thomas AM, Simmons DL, Salmon M, Buckley CD. Rheumatoid fibroblast-like synoviocytes overexpress the chemokine stromal cell-derived factor 1 (CXCL12), which supports distinct patterns and rates of CD4+ and CD8+ T cell migration within synovial tissue. Arthritis Rheum. 2003;48:2472–2482. doi: 10.1002/art.11219. [DOI] [PubMed] [Google Scholar]
- 22.Zhao C, Fernandes MJ, Prestwich GD, Turgeon M, Di Battista J, Clair T, Poubelle PE, Bourgoin SG. Regulation of lysophosphatidic acid receptor expression and function in human synoviocytes: implications for rheumatoid arthritis? Mol Pharmacol. 2008;73:587–600. doi: 10.1124/mol.107.038216. [DOI] [PubMed] [Google Scholar]
- 23.Burger JA, Tsukada N, Burger M, Zvaifler NJ, Dell’Aquila M, Kipps TJ. Blood-derived nurse-like cells protect chronic lymphocytic leukemia B cells from spontaneous apoptosis through stromal cell-derived factor-1. Blood. 2000;96:2655–2663. [PubMed] [Google Scholar]
- 24.Burger JA, Zvaifler NJ, Tsukada N, Firestein GS, Kipps TJ. Fibroblast-like synoviocytes support B-cell pseudoemperipolesis via a stromal cell–derived factor-1– and CD106 (VCAM-1) –dependent mechanism. J Clin Invest. 2001;107:305–315. doi: 10.1172/JCI11092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kanda H, Newton R, Klein R, Morita Y, Gunn MD, Rosen SD. Autotaxin, an ectoenzyme that produces lysophosphatidic acid, promotes the entry of lymphocytes into secondary lymphoid organs. Nat Immunol. 2008;9:415–423. doi: 10.1038/ni1573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Zhang Y, Chen YC, Krummel MF, Rosen SD. Autotaxin through lysophosphatidic acid stimulates polarization, motility, and transendothelial migration of naive T cells. J Immunol. 2012;189:3914–3924. doi: 10.4049/jimmunol.1201604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Bai Z, Cai L, Umemoto E, Takeda A, Tohya K, Komai Y, Veeraveedu PT, Hata E, Sugiura Y, Kubo A, Suematsu M, Hayasaka H, Okudaira S, Aoki J, Tanaka T, Albers HM, Ovaa H, Miyasaka M. Constitutive lymphocyte transmigration across the basal lamina of high endothelial venules is regulated by the autotaxin/lysophosphatidic acid axis. J Immunol. 2013;190:2036–2048. doi: 10.4049/jimmunol.1202025. [DOI] [PubMed] [Google Scholar]
- 28.Orosa B, González A, Mera A, Gómez-Reino JJ, Conde C. Lysophosphatidic acid receptor 1 suppression sensitizes rheumatoid fibroblast-like synoviocytes to tumor necrosis factor-induced apoptosis. Arthritis Rheum. 2012;64:2460–2470. doi: 10.1002/art.34443. [DOI] [PubMed] [Google Scholar]


