Abstract
Aim of the Review:
To describe the significant improvement in the diagnosis, treatment and outcome of children diagnosed with hepatoblastoma (HB) that has occurred in the past four decades. Recent findings are mainly focused on lessons learned from the experiences of the Childhood Liver Tumors Strategy Group (SIOPEL). Important milestones were the risk stratification of HB that allowed to tailor down therapy for standard-risk HB and intensify treatment for high-risk HB. The multi-institutional international cooperative SIOPEL trials are reviewed and current treatment guidelines are given. Intensified cooperation between the SIOPEL and the Children's Oncology Group (COG) and the national study groups from Germany (GPOH) and Japan (JPLT) led to the acceptance and use of one staging system (PRETEXT) and the formation of a single robust database containing data of 1605 HB patients. This will allow analysis with enough statistical power of treatment directing factors that will form one of the bases of the next-generation clinical trial that is currently designed by all four collaborating study groups.
Summary:
Successive SIOPEL trials and increasing international collaboration have improved survival rates of patients with HB through risk stratification, advances in chemotherapy and increased complete resection rates including liver transplantation as a surgical option.
KEY WORDS: Chemotherapy, hepatoblastoma, liver transplantation, PRETEXT, risk stratification, SIOPEL-studies, surgical resection
INTRODUCTION
With survival rates increasing from 20% to 80% over the past decades, the treatment of hepatoblastoma (HB) has been one of the great success stories in pediatric oncology. Because of an enormous multidisciplinary and multicenter effort throughout the years, it became possible to achieve ever-increasing complete resection rates where a complete surgical removal of this rare liver tumor is paramount for a realistic chance for cure.
Historical background
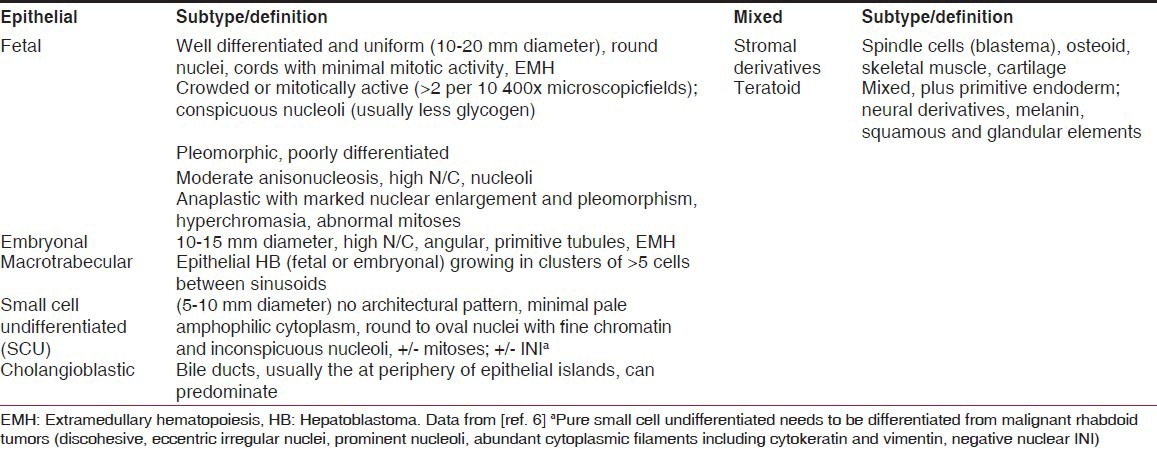
In 1898, the first case of a child with HB was published in the English literature.[1] A 6-week-old boy was described whose autopsy showed a large tumor that occupied the lower half of the right liver lobe. Because cysts and cartilaginous and bony deposits were seen, the tumor was described as a teratoma, with tissue representatives of the three embryonic germ cell layers. In 1962, the term “Hepatoblastoma” was introduced for this type of tumor by Willis, who defined it as “an embryonic tumor that contains hepatic epithelial parenchyma.”[2] At that time, HB was usually not distinguished from hepatocellular carcinoma (HCC). Through the work of Ishak and Glunz in 1967, morphologic criteria were defined for HB and HCC that were refined in the decades that followed.[3,4] HB is currently thought to originate from the hepatoblast (hepatocyte precursor cell) that often recapitulates the stages of liver development, displaying a combination of histological patterns.[5] Recently, consensus was reached regarding a new classification of pediatric liver tumors at the Los Angeles Childrens Oncology Group (COG) International Pathology Pediatric Liver Tumors Symposium [Table 1].[6,7]
Table 1.
International consensus classification of the histological subtypes of hepatoblastoma
Epidemiology
Risk factors
Thus far, no environmental risk factors for HB have been described, but HB has been associated with prematurity or a low birth weight.[8,9] Medications such as furosemide, total parenteral nutrition, oxygen therapy, radiation, plasticizers and other toxins are postulated to play a role, but no hypothesis for the exact mechanisms is currently available. Familial cases have been reported. In this respect, the occurrence of HB with familial adenomatous polyposis and Beckwith — Wiedemann syndrome is striking and suggests a role in the pathogenesis of HB for chromosomes 5 and 11, respectively.[10,11]
HB is a malignancy of the liver with a fairly constant annual incidence of 1.2-1.5 cases per million children younger than 15 years of age in Western countries,[12] with a small increase reported in the USA, Europe and Japan during the last 30 years.[13,14,15] Because prematurity and very low birth weight have been found to be associated with the later appearance of HB, increase in these patient cohorts may be partly responsible for this increase in incidence.[15]
HB comprises 1% of all pediatric malignancies and affects mostly infants and young children between 6 months and 3 years, but cases in neonates and adolescents have also been reported. After neuroblastoma and nephroblastoma, primary epithelial tumors of the liver are the third most common intraabdominal neoplasms in children.[16] HB is the most frequent liver tumor in Western countries. In Asia and Africa, HCC occurs more frequently than HB, which is probably a consequence of the higher prevalence of hepatitis B infection on those continents.
The bottom line: A survey
In 1975, Exelby published a landmark paper in which he reported the results of a 10-year questionnaire entitled the “American Academy of Pediatrics Surgical Section Survey — 1974.”[17] From 110 replies, 375 liver tumors were reported, of which 252 were malignant (HB [n = 129] and HCC [n = 98]) and 123 benign. All patients with HB underwent primary surgical exploration. A definitive partial hepatic resection was carried out in 86 patients and biopsy was carried out only in 43 patients. Of seventy-eight patients who had complete excision of the tumor, 45 (60%) survived. In two-thirds of the patients, the tumor was not excised completely and there were no survivors in this group. The overall survival rate in HB was 35%.
Excessive blood loss was the most common complication during and immediately after the operation, and cardiac arrest occurred in nine patients. There were eight deaths in the operating room and 17 deaths in the immediate postoperative period attributable to the operation.
It was apparent that no cures were obtained from irradiation and/or chemotherapy in inoperable cases. There were three cases of HB, however, in whom the tumors originally thought to be unresectable could be removed after irradiation to the liver and combined chemotherapy (vincristine, actinomycin and cyclophosphamide). These three patients were long-term survivors after subsequent hepatic lobectomy. Eleven patients were given prophylactic chemotherapy after complete surgical excision of the tumor, and eight of these patients were alive. In cases in whom incomplete excision or biopsy only was carried out, no patient survived in either group. At this time, it seemed that complete operative excision offered the only chance of cure in children with HB through which cure rates of 60% could be achieved in this subgroup of patients.
Management
As has been shown in the past and has been confirmed nowadays, surgery alone can cure very few patients with HB, mostly ones with the pure fetal HB variant.[18] More than half of patients present with unresectable primary tumors or distant metastases. Evidence that HB is a chemosensitive tumor began to accumulate in the early 1970s when responses were seen to combinations of cyclophosphamide, vincristine, 5-fluorouracil and actinomycin-D.[19] Introduction of cisplatin- and doxorubicin-containing regimens in the 1980s had a major impact on survival.[20,21,22,23] Over 30 years later, cisplatin still remains the backbone of the chemotherapy regimen. Chemotherapy may reduce tumor volume, making the tumor resectable, and may lead to the complete disappearance of lung metastases. The tumor response rate to the present cisplatin-containing chemotherapy regimens varies from 70% to 90% according to the different series.[20,21,22,23,24,25] Neoadjuvant chemotherapy not only makes the tumor “smaller,” and consequently more likely to be completely resected, but also more solid, less prone to bleeding and more demarcated from the remaining healthy liver parenchyma.[26] Also, potentially existing micrometastases in the lungs are treated early. Because of these arguments, some clinicians currently recommend starting preoperative chemotherapy after biopsy and deferring definitive surgery until after 2-3 months of therapy. This treatment philosophy was adopted by the SIOPEL Childhood Liver Tumors Strategy Group. In contrast to the SIOPEL approach, the North American Study Groups (first CCSG and POG, now COG) still recommend primary surgery whenever prudently possible as the initial treatment. Thus far, no controlled comparison has been performed between these two treatment strategies. However, the present survival rates of patients with tumors not eligible for up-front resection reported by the different study groups are comparable regardless of the first therapeutic modality used, and vary between 70% and 90%.
Staging
A universally accepted staging system for childhood hepatocellular tumors did not exist at first. Through the various staging systems used, the different approaches to treatment adopted by the different study groups are reflected. The North American Co-operative Study Groups on HB favoring primary surgery use a postsurgical staging system based on the results of the initial attempt at complete resection of the tumor mass. Thus, four stages are identified: Stage 1, complete resection, no microscopic residual disease; Stage II, microscopic residual; Stage III, macroscopic residual disease; and Stage IV, distant metastases. This surgical staging system is in contrast to the one developed and used by the SIOPEL group, whose therapeutic strategy is based on primary chemotherapy and delayed surgery. The PRETEXT system (pretreatment extension of disease) is based on pretreatment imaging with ultrasound, computed tomography (CT) scans and/or magnetic resonance imaging, and describes the site and size of the tumor, invasion of the vessels and distant spread, as judged on the imaging. The system identifies four PRETEXT stages (I-IV), which reflect the number of sections of the liver that are involved by the tumor and describes the extent of the disease beyond the liver using the following letters: “V” if the tumor extends into the vena cava and/or all three hepatic veins, “P” if the main and/or both left and right branches of the portal vein are involved by the tumor, “C” if there is involvement of the caudate lobe, “E” if there is evidence of extrahepatic intraabdominal disease and “M” if there are distant metastases.[27,28] Once the prognostic power of the PRETEXT system had been shown, the SIOPEL, COG, the German Society for Pediatric Oncology-Hematology (GPOH) and the Japanese study group for Pediatric Liver Tumors (JPLT) increased their cooperation. This staging system was finally adopted by all four groups.[29] The great advantage was that now patient subgroups in the various study trials as defined by stage became comparable.
Lessons learnt from the successive SIOPEL studies
SIOPEL-1 was the first international prospective study that used the concept of neoadjuvant chemotherapy and delayed surgery conducted between 1990 and 1994. An open or closed biopsy was recommended in cases when the clinical picture was equivocal and the pathology was centrally reviewed. A total of 91 centers in 30 different countries participated in the study. A total of 193 patients were registered, of whom 153 had an HB and 115 underwent delayed surgery.
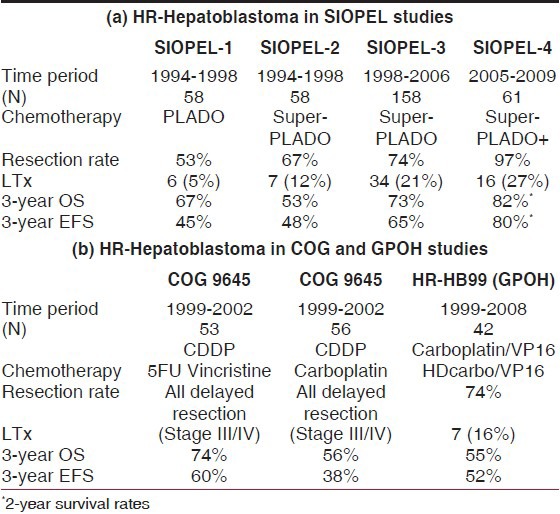
The overall 5-year event-free survival (EFS) was 66% (confidence interval [CI] 59-74%) and the 5-year overall survival (OS) was 75% (CI 68-82%).[30,31] On the basis of these results, the SIOPEL group recommended delayed surgery as the standard treatment for HB, as it seemed much less risky than “up front” surgery because tumor shrinkage achieved with preoperative chemotherapy necessitated smaller resections.[32] In this study, the resection rate was 53%, with 12 patients with unresectable tumors who underwent a liver transplant (8% of all patients) achieving comparable outcome as seen after partial liver resection. This observation could be confirmed in an overview of the world experience of liver transplantation for HB at that time.[33] Liver transplantation thus became the tool to increase the resection rate.[34]
There were two aspects that were crucial in planning the next studies. Firstly, SIOPEL-1 data showed that two “risk groups” could be distinguished [Table 2]. There were patients with resectable tumors and no evident metastases (designated as “standard risk” patients) and patients with either unresectable tumors (all four sectors involved) and/or extrahepatic tumor, usually lung metastases, or low alfa-feto protein (designated as the “high risk” patients).[31] Later, tumor rupture was added as a high-risk factor [Table 2]. Secondly, within the American trials, two intergroup regimens had suggested that cisplatin was the crucial element of the PLADO regimen. It was judged that cisplatin could be intensified as there had been little nephrotoxicity or ototoxity reported in SIOPEL-1, whereas the doxorubicin dose and dose intensity could not be readily increased for fear of the higher incidence of cardiotoxicity.
Table 2.
SIOPEL risk stratification for hepatoblastoma

SIOPEL-2 (pilot) and SIOPEL-3
Risk stratification
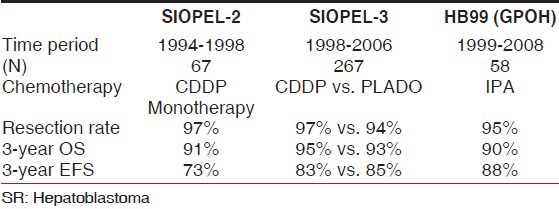
Based on these findings, the SIOPEL group decided for the subsequent SIOPEL-2 (pilot) and SIOPEL-3 trials to treat tumors according to risk stratification.[17,35,36] Standard risk (SR-HB), defined as tumor confined to the liver, involving at the most three hepatic sectors (PRETEXT I-III) and AFP >100 ng/mL was treated with the less-toxic cisplatin monotherapy. High risk (HR-HB), defined as tumor involving all four liver sectors (PRETEXT-IV) or vascular invasion (V/P+) or extrahepatic disease (E+/M+) or low alfa-fetoprotein (AFP <100 ng/mL) at diagnosis or tumor rupture, was treated with more intensified chemotherapy called SuperPLADO (cisplatin alternating with carboplatin-doxorubicin). The protocol also encouraged liver transplantation in unresectable tumors.
Indeed, resection rates increased to 67% in SIOPEL-2 and to 74% in SIOPEL-3, with liver transplants performed in 12% and 21% of cases, respectively. The randomized SIOPEL-3 study showed that cisplatin monotherapy was as effective as PLADO in the standard risk group (n = 255) in terms of complete resection rates; these two regimens achieved comparable 3-year EFS and OS rates (CDDP monotherapy: EFS 83% [95% CI 77-90%], OS 95% [95% CI 91-99%], with a median follow-up time of 45 months) [Table 3]. As expected, cisplatin alone was less toxic than PLADO (less-acute grade 3 or 4 adverse events).[37] For HR-HB (n = 158), it was shown that compared with the historic controls of SIOPEL-1 (3 years-EFS 45% [95% CI 32-56%]; OS 67% [95% CI 55-80%]) this platinum-intensive regimen had significantly improved EFS, with OS being 65% (95% CI 57-73%) and 69% (95% CI 62-77%) at 3 years for the whole group. EFS and OS for all patients with PRETEXT-IV tumor were 68% and 69%, respectively, and 56% and 62%, respectively, for patients with metastasis. Patients with low AFP still have a very bad prognosis.[36]
Table 3.
Outcomes of standard-risk hepatoblastoma
SIOPEL-4
The regimen used in this study aimed to further intensify chemotherapy in children with high-risk HB by adopting a weekly dose dense administration of cisplatin in combination with monthly doxorubicin and delayed radical surgery. The final analysis of the trial included 62 eligible patients (39 with lung metastases). Sixty (98%, 95% CI 91-100) of 61 evaluable patients had a partial response to preoperative chemotherapy. Complete resection of all tumor lesions was achieved in 53 patients (85%) including 16 (26%) liver transplants. One child underwent primary hepatectomy for tumor rupture. At the end of therapy, 49 (79%, 95% CI 67-88) of 62 patients were in complete remission. With a median follow-up of 52 months, the 3-year EFS was 76% (95% CI 65-87) and the 3-year OS was 83% (73-93). Sixty (97%) patients had grades 3-4 hematological toxicity (anemia, neutropenia, thrombocytopenia, infections or anorexia/mucositis), four patients had toxic deaths (two from infection, one from surgical bleeding and one with tumor bleeding) and 71% patients had fever and neutropenia. Moderate-to-severe ototoxicity was documented in 50% of the patients.[38]
As a single-arm trial, SIOPEL-4 was not randomized, but outcomes were far superior to historical controls [Table 4a]. A further study will be needed before this approach can be definitively adopted as the standard of care. In fact, this study has already been planned as a high-risk arm of a new international HB trial being designed as a joint effort between COG, SIOPEL/GPOH and JPLT.
Table 4.
(a) Improved outcome over time of high-risk hepatoblastoma in the SIOPEL studies. Note the increased liver transplantation (LTx) and resection rates (RR). (b) Outcomes of COG and GPOH studies
Current treatment guidelines
As per 2014, SIOPEL recommends the following treatment strategy for HB patients not entered into a clinical trial.
Standard-risk HB
These are localized tumors with no additional adverse features [Table 2]. The recommendation is to follow the cisplatin monotherapy arm of the SIOPEL-3 study.[37] The standard treatment is four cycles of preoperative chemotherapy followed by surgical resection and two postoperative cycles of therapy.
High-risk HB
These tumors are defined as any tumor not meeting the standard risk or very high-risk criteria. The recommendation is to receive the dose intensive “superPLADO” arm of the SIOPEL-3 study.[36] Patients in this group are likely to have challenging disease from the surgical perspective and we would recommend consultation at the time of diagnosis with a specialist liver surgery/transplant service.
Very high-risk HB
These tumors are defined by the presence of metastatic disease (usually lung) or very low AFP (<100 ng/mL). Pulmonary lesions documented on the chest X-ray and/or lung CT scan will be considered to be unequivocal metastatic tumor deposits if there is one nodule larger than 10 mm or several nodules with at least one larger than 5 mm. In the other cases, the metastases will be considered as doubtful and a surgical biopsy of one of the nodules should be discussed if the general condition of the child permits it. Patients should be treated with the approach utilized in the SIOPEL-4 protocol with dose-intensive weekly cisplatin/doxorubicin induction therapy.[38] Particular attention should be paid to the potential toxicity and supportive care needs of patients on this regimen. All patients with low AFP should have an initial biopsy. The loss of INI1 expression is suggestive of rhabdoid tumor (sometimes confused with small cell undifferentiated HB) and we would recommend such patients are treated with a rhabdoid-style chemotherapy approach. It should be noted that some low-risk localized tumors with small-volume disease may also have a low AFP.
SIOPEL RCN (resource-challenged nations)
More than 90% of cancers in childhood occur in developing countries. Despite the fact that pediatric cancer is highly treatable nowadays, 80% of children with malignancies die because they live in these so-called low-income countries (resource-challenged nations [RCN]), where access to medical care, if at all available, is often inadequate. Some RCN, notably India, have launched the cancer initiative and have chosen to concentrate treatment resources in regional cancer centers.
The SIOPEL-3 study formed the basis of the RCN HB protocol. As described earlier, it had shown that treatment of standard-risk HB with cisplatin monotherapy had equal outcome as treatment with PLADO, with cure rates now approaching those of Wilms’ tumor. Furthermore, treatment with cisplatin monotherapy is not complex, lengthy or especially expensive. Toxicity is mild to moderate with easily manageable complications; therefore, most patients are cured at “little cost.”
The aim of the RCN project was to provide simple, effective and affordable treatment to children with HB in RCN and to offer an easy data collection system to evaluate this program. A subcommittee of SIOPEL has made therapeutic guidelines for the RCN protocol that is currently running in India (Principal Investigator: Dr. S. Agarwala, Departement of Paediatric Surgery, AIIMS, New Delhi). An internet-based remote data entry system is available to register patient data in order to evaluate the results of this RCN program.[39]
The RCN program thus offers an opportunity for clinicians in RCNs to deliver appropriate care to children with HB in an affordable and evaluable way. We expect that this approach will increase the quality of care to these children.
Current challenges
Ototoxicity
Both SIOPEL and COG have put considerable effort in trying to decrease the significant ototoxicity induced by the use of cisplatin-based chemotherapy in young patients. The COG 9645 trial failed to reduce ototoxicity with the agent amifostine.[40] The ongoing SIOPEL-6 study currently investigates sodium thiosulfate (STS)[41] as an attempt to decrease the cisplatin-induced ototoxicity in standard-risk HB. There is always a fear of unwanted cancer protection by STS; however, four consecutive interim efficacy analyses have shown that trial stopping rules were not met and the Independent Data Monitoring Committee recommended that the trial be continued as planned.
Tumor recurrence
The prognosis for a patient with recurrent or progressive HB depends on many factors like the site of recurrence, prior treatment and individual patient considerations. For example, in patients with a completely resected HB and persistent lung metastases, surgical treatment of resectable pulmonary metastases, even those that developed in the course of the disease, may put the patient in complete remission with good disease-free survival.[42] If possible, isolated metastases should be resected completely in patients whose primary tumor is controlled.[32,35] In recurrent refractory disease, phase I and II clinical trials may be appropriate and should be considered.[43] Many alternative strategies and combinations of new agents are currently tested, like irinothecan, oxaliplatin, targeted agents, gene-directed therapy and immunotherapy.[7]
International developments — the CHIC project
Recent collaboration between the four major international groups studying HB-the SIOPEL, COG, JPLT, and GPOH-has led to the Children's Hepatic tumor International Collaboration (CHIC) initiative. Because it is a rare tumor, obtaining data on a large cohort of patients to define prognostic factors for directing risk-adapted therapy has been challenging. Within the CHIC project, all results of all studies conducted between 1989 and 2008 were collected in a single database and were made uniformly available for statistical analysis. This way, data from1605 patients could be brought together, numbers large enough to provide for analysis with adequate statistical power.[44]
The amount of data uniformly collected during the conduct of the eight trials that form the cohort will allow a more in-depth analysis of prognostic factors than previously performed.[45] More importantly, the combination of several factors will be analyzed and will enable an in-depth description of subgroups of patients with widely different clinical presentation. A group of clinicians and statisticians are currently involved in a unique cooperation that will formulate the basic risk stratification needed for the next-generation clinical trial with the acronym PHITT (Pediatric Hepatic Malignancy International Therapeutic Trial), to be conducted by all four collaborative groups. These developments are the starting point of a new worldwide cooperation that will be of eventual benefit for the children with liver tumors.
Footnotes
Source of Support: Nil
Conflict of Interest: None declared.
REFERENCES
- 1.Misick OS. A case of teratoma hepatis. J Pathol Bacteriol. 1898;5:128–37. [Google Scholar]
- 2.Willis RA. Some uncommon and recently identified tumours. In: Cameron R, Payling Wright G, editors. The pathology of the Tumors of Children. Springfield: Charles C. Thoma; 1962. pp. 57–61. [Google Scholar]
- 3.Ishak KG, Glunz PR. Hepatoblastoma and hepatocarcinoma in infancy and childhood: Report of 47 cases. Cancer. 1967;20:396–422. doi: 10.1002/1097-0142(1967)20:3<396::aid-cncr2820200308>3.0.co;2-o. [DOI] [PubMed] [Google Scholar]
- 4.Zimmermann A. Hepatoblastoma with cholangioblastic features (‘cholangioblastic hepatoblastoma’) and other liver tumors with bimodal differentiation in young patients. Med Pediatr Oncol. 2002;39:487–91. doi: 10.1002/mpo.10173. [DOI] [PubMed] [Google Scholar]
- 5.Finegold MJ, Lopez-Terrada DH, Bowen J, Washington MK, Qualman SJ College of American Pathologists. Protocol for the examination of specimens from pediatric patients with hepatoblastoma. Arch Pathol Lab Med. 2007;131:520–9. doi: 10.5858/2007-131-520-PFTEOS. [DOI] [PubMed] [Google Scholar]
- 6.López-Terrada D, Alaggio R, de Dávila MT, Czauderna P, Hiyama E, Katzenstein H, et al. Towards an international pediatric liver tumor consensus classification: Proceedings of the Los Angeles COG liver tumors symposium. Mod Pathol. 2014;27:472–91. doi: 10.1038/modpathol.2013.80. [DOI] [PubMed] [Google Scholar]
- 7.Czauderna P, Lopez-Terrada D, Hiyama E, Häberle B, Malogolowkin MH, Meyers RL. Hepatoblastoma state of the art: Pathology, genetics, risk stratification, and chemotherapy. Curr Opin Pediatr. 2014;26:19–28. doi: 10.1097/MOP.0000000000000046. [DOI] [PubMed] [Google Scholar]
- 8.Ikeda H, Matsuyama S, Tanimura M. Association between hepatoblastoma and very low birth weight: A trend or a chance. J Pediatr. 1997;130:557–60. doi: 10.1016/s0022-3476(97)70239-7. [DOI] [PubMed] [Google Scholar]
- 9.Feusner J, Plaschkes J. Hepatoblastoma and low birth weight: A trend or chance observation? Med Pediatr Oncol. 2002;39:508–9. doi: 10.1002/mpo.10176. [DOI] [PubMed] [Google Scholar]
- 10.Haas OA, Zoubek A, Grümayer ER, Gadner H. Constitutional interstitial deletion of 11p11 and pericentric inversion of chromosome 9 in a patient with Wiedemann-Beckwith syndrome and hepatoblastoma. Cancer Genet Cytogenet. 1986;23:95–104. doi: 10.1016/0165-4608(86)90409-7. [DOI] [PubMed] [Google Scholar]
- 11.Steenman M, Westerveld A, Mannens M. Genetics of Beckwith-Wiedemann syndrome-associated tumors: Common genetic pathways. Genes Chromosomes Cancer. 2000;28:1–13. doi: 10.1002/(sici)1098-2264(200005)28:1<1::aid-gcc1>3.0.co;2-#. [DOI] [PubMed] [Google Scholar]
- 12.Mann JR, Kasthuri N, Raafat F, Pincott JR, Parkes SE, Muir KR, et al. Malignant hepatic tumors in children: Incidence, clinical features and aetiology. Paediatr Perinat Epidemiol. 1990;4:276–89. doi: 10.1111/j.1365-3016.1990.tb00651.x. [DOI] [PubMed] [Google Scholar]
- 13.Buckley JD, Sather H, Ruccione K, Rogers PC, Haas JE, Henderson BE, et al. A case-control study of risk factors for hepatoblastoma. A report from the Childrens Cancer Study Group. Cancer. 1989;64:1169–76. doi: 10.1002/1097-0142(19890901)64:5<1169::aid-cncr2820640534>3.0.co;2-i. [DOI] [PubMed] [Google Scholar]
- 14.Surveillance Epidemiology and End Results Program. [Cancer Statistics Review 1975-2009]. Available from: http://www.seer.cancer.gov .
- 15.Spector LG, Birch J. The epidemiology of hepatoblastoma. Pediatr Blood Cancer. 2012;59:776–9. doi: 10.1002/pbc.24215. [DOI] [PubMed] [Google Scholar]
- 16.Clatworthy HW, Jr, Schiller M, Grosfeld JL. Primary liver tumors in infancy and childhood.41 cases variously treated. Arch Surg. 1974;109:143–7. doi: 10.1001/archsurg.1974.01360020005002. [DOI] [PubMed] [Google Scholar]
- 17.Exelby PR, Filler RM, Grosfeld JL. Liver tumors in children in the particular reference to hepatoblastoma and hepatocellular carcinoma: American Academy of Pediatrics Surgical Section Survey-1974. J Pediatr Surg. 1975;10:329–37. doi: 10.1016/0022-3468(75)90095-0. [DOI] [PubMed] [Google Scholar]
- 18.Malogolowkin MH, Katzenstein HM, Meyers RL, Krailo MD, Rowland JM, Haas J, et al. Complete surgical resection is curative for children with hepatoblastoma with pure fetal histology: A report from the Children's Oncology Group. J Clin Oncol. 2011;29:3301–6. doi: 10.1200/JCO.2010.29.3837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Evans AE, Land VJ, Newton WA, Randolph JG, Sather HN, Tefft M. Combination chemotherapy (vincristine, adriamycin, cyclophosphamide, and 5-fluorouracil) in the treatment of children with malignant hepatoma. Cancer. 1982;50:821–6. doi: 10.1002/1097-0142(19820901)50:5<821::aid-cncr2820500502>3.0.co;2-k. [DOI] [PubMed] [Google Scholar]
- 20.Ninane J, Perilongo G, Stalens JP, Guglielmi M, Otte JB, Mancini A. Effectiveness and toxicity of cisplatin and doxorubicin (PLADO) in childhood hepatoblastoma and hepatocellular carcinoma: A SIOP pilot study. Med Pediatr Oncol. 1991;19:199–203. doi: 10.1002/mpo.2950190310. [DOI] [PubMed] [Google Scholar]
- 21.Ortega JA, Kralio MD, Haas JE, King DR, Ablin AR, Quinn JJ, et al. Effective treatment of unresectable or metastatic hepatoblastoma with cisplatin and continuous infusion doxorubicin: A report from the Children's Cancer Study Group. J Clin Oncol. 1991;9:2167–76. doi: 10.1200/JCO.1991.9.12.2167. [DOI] [PubMed] [Google Scholar]
- 22.Douglass EC, Reynolds M, Finegold M, Cantor AB, Glicksman A. Cisplatin, vincristine, and fluorouracil therapy for hepatoblastoma: A pediatric oncology group study. J Clin Oncol. 1993;11:96–9. doi: 10.1200/JCO.1993.11.1.96. [DOI] [PubMed] [Google Scholar]
- 23.Weinblatt ME, Siegel SE, Siegel MM, Stanley P, Weitzman JJ. Preoperative chemotherapy for unresectable primary hepatic malignancies in children. Cancer. 1982;50:1061–4. doi: 10.1002/1097-0142(19820915)50:6<1061::aid-cncr2820500607>3.0.co;2-q. [DOI] [PubMed] [Google Scholar]
- 24.Filler RM, Ehrlich PF, Greenberg ML, Babyn PS. Preoperative chemotherapy in hepatoblastoma. Surgery. 1991;110:591–7. [PubMed] [Google Scholar]
- 25.von Schweinitz D, Byrd DJ, Hecker H, Weinel P, Bode U, Bürger D, et al. Effciency and toxicity of ifosfamide, cisplatin, and doxorubicin in the treatment of childhood hepatoblastoma. Study committee of the Cooperative Paediatric Liver Tumor Study HB89 of the German Society of Paediatric Oncology and Haematology. Eur J Cancer. 1997;33:1243–9. doi: 10.1016/s0959-8049(97)00095-6. [DOI] [PubMed] [Google Scholar]
- 26.Kalifa C, Lemerle J, Caillaud JM. Resectability of childhood hepatoblastoma is improved by primary chemotherapy. Proc Am Soc Clin Oncol. 1984:c308. [Google Scholar]
- 27.Aronson DC, Schnater JM, Staalman CR, Weverling GJ, Plaschkes J, Perilongo G, et al. Predictive value of the pretreatment extent of disease system in hepatoblastoma: Results from the international society of Pediatric Oncology Liver Tumor Study Group SIOPEL-1 study. J Clin Oncol. 2005;23:1245–52. doi: 10.1200/JCO.2005.07.145. [DOI] [PubMed] [Google Scholar]
- 28.Roebuck DJ, Aronson D, Clapuyt P, Czauderna P, de Ville de Goyet J, Gauthier F, et al. International Childrhood Liver Tumor Strategy Group 2005 PRETEXT: A revised staging system for primary malignant liver tumors of childhood developed by the SIOPEL group. Pediatr Radiol. 2007;37:123–32. doi: 10.1007/s00247-006-0361-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Meyers RL, Tiao G, de Ville de Goyet J, Superina R, Aronson DC. Hepatoblastoma state of the art: Pre-treatment extent of disease, surgical resection guidelines and the role of liver transplantation. Curr Opin Pediatr. 2014;26:29–36. doi: 10.1097/MOP.0000000000000042. [DOI] [PubMed] [Google Scholar]
- 30.Pritchard J, Brown J, Shafford E, Perilongo G, Brock P, Dicks-Mireaux C, et al. Cisplatin, doxorubicin, and delayed surgery for childhood hepatoblastoma: A successful approach — results of the first prospective study of the International Society of Pediatric Oncology. J Clin Oncol. 2000;18:3819–28. doi: 10.1200/JCO.2000.18.22.3819. [DOI] [PubMed] [Google Scholar]
- 31.Brown J, Perilongo G, Shafford E, Keeling J, Pritchard J, Brock P, et al. Pretreatment prognostic factors for children with Hepatoblastoma — results from the International Society of Paediatric Oncology (SIOP) Study SIOPEL 1. Eur J Cancer. 2000;36:1418–25. doi: 10.1016/s0959-8049(00)00074-5. [DOI] [PubMed] [Google Scholar]
- 32.Schnater JM, Aronson DC, Plaschkes J, Perilongo G, Brown J, Otte JB, et al. Surgical view of the treatment of patients with hepatoblastoma: Results from the first prospective trial of the International Society of Pediatric Oncology Liver Tumor Study Group. Cancer. 2002;94:1111–20. [PubMed] [Google Scholar]
- 33.Otte JB, Pritchard J, Aronson DC, Brown J, Czauderna P, Maibach R, et al. Liver Transplantation for Hepatoblastoma: Results From the International Society of Pediatric Oncology (SIOP) Study SIOPEL-1 and Review of the world experience. Pediatr Blood Cancer. 2004;42:74–83. doi: 10.1002/pbc.10376. [DOI] [PubMed] [Google Scholar]
- 34.Otte JB, deVille de Goyet J, Reding R. Liver transplantation for hepatoblastoma: Indications and contraindications in the modern era. Pediatr Transplant. 2005;9:557–65. doi: 10.1111/j.1399-3046.2005.00354.x. [DOI] [PubMed] [Google Scholar]
- 35.Perilongo G, Shafford E, Maibach R, Aronson D, Brugières L, Brock P, et al. International Society of Paediatric Oncology-SIOPEL 2. Risk-adapted treatment for childhood hepatoblastoma. Final report of the second study of the Internal Society of Pediatric Oncology-SIOPEL 2. Eur J Cancer. 2004;40:411–21. doi: 10.1016/j.ejca.2003.06.003. [DOI] [PubMed] [Google Scholar]
- 36.Zsíros J, Maibach R, Shafford E, Brugieres L, Brock P, Czauderna P, et al. Successful treatment of childhood high-risk hepatoblastoma with dose-intensive multiagent chemotherapy and surgery: Final results of the SIOPEL-3HR study. J Clin Oncol. 2010;28:2584–90. doi: 10.1200/JCO.2009.22.4857. [DOI] [PubMed] [Google Scholar]
- 37.Perilongo G, Maibach R, Shafford E, Brugieres L, Brock P, Morland B, et al. Cisplatin versus cisplatin plus doxorubicin for standard-risk hepatoblastoma. N Engl J Med. 2009;361:1662–70. doi: 10.1056/NEJMoa0810613. [DOI] [PubMed] [Google Scholar]
- 38.Zsiros J, Brugieres L, Brock P, Roebuck D, Maibach R, Zimmermann A, et al. International Childhood Liver Tumors Strategy Group (SIOPEL) Dose-dense cisplatin-based chemotherapy and surgery for children with high-risk hepatoblastoma (SIOPEL-4): A prospective, single-arm, feasibility study. Lancet Oncol. 2013;14:834–42. doi: 10.1016/S1470-2045(13)70272-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Agarwala S, Ronghe MD, Aronson DC, Czauderna P, Brock P, Roebuck D, et al. Pilot of treatment guidelines for hepatoblastoma (HB) in resource challenged nations (RCN) PBC. 2009;53:717. [Google Scholar]
- 40.Malogolowkin MH, Katzenstein HM, Krailo MD, Chen Z, Bowman L, Reynolds M, et al. Intensified platinum therapy is an ineffective strategy for improving outcome in pediatric patients with advanced hepatoblastoma. J Clin Oncol. 2006;24:2879–84. doi: 10.1200/JCO.2005.02.6013. [DOI] [PubMed] [Google Scholar]
- 41.Muldoon LL, Pagel MA, Kroll RA, Brummett RE, Doolittle ND, Zuhowski EG, et al. Delayed administration of sodium thiosulfate in animal models reduces platinum ototoxicity without reduction of antitumor activity. Clin Cancer Res. 2000;6:309–15. [PubMed] [Google Scholar]
- 42.Feusner JH, Krailo MD, Haas JE, Campbell JR, Lloyd DA, Ablin AR. Treatment of pulmonary metastases of initial stage I hepatoblastoma in childhood: Report from the Childrens Cancer Group. Cancer. 1993;71:859–64. doi: 10.1002/1097-0142(19930201)71:3<859::aid-cncr2820710333>3.0.co;2-t. [DOI] [PubMed] [Google Scholar]
- 43.Zsíros J, Brugières L, Brock P, Roebuck D, Maibach R, Child M, et al. Efficacy of irinotecan single drug treatment in children with refractory or recurrent hepatoblastoma — a phase II trial of the childhood liver tumor strategy group (SIOPEL) Eur J Cancer. 2012;48:3456–64. doi: 10.1016/j.ejca.2012.06.023. [DOI] [PubMed] [Google Scholar]
- 44.Meyers R R, Haeberle B, Hiyama E, Maibach R, Krailo M, Feng Y, et al. Children's Hepatic Tumor International Collaboration (CHIC): New hepatoblastoma International Risk Groups (HB-IRG) Abstract SIOP, Hong Kong. Pediatr Blood Cancer. 2013:15. [Google Scholar]
- 45.Maibach R, Roebuck D, Brugieres L, Capra M, Brock P, Dall’Igna P, et al. Prognostic stratification for children with hepatoblastoma: The SIOPEL experience. Eur J Cancer. 2012;48:1543–9. doi: 10.1016/j.ejca.2011.12.011. [DOI] [PubMed] [Google Scholar]


