Abstract
Numerous bacteria coordinate gene expression in response to small signalling molecules in many cases known as acylhomoserine lactones (AHLs), which accumulate as a function of cell density in a process known as quorum sensing. This work aimed to determine if phenotypes that are important to define microbial activity in foods such as biofilm formation, swarming motility and proteolytic activity of two Pseudomonas fluorescens strains, isolated from refrigerated raw milk, are influenced by AHL molecules. The tested P. fluorescens strains did not produce AHL molecules in none of the evaluated media. We found that biofilm formation was dependent on the culture media, but it was not influenced by AHLs. Our results indicate that biofilm formation, swarming motility and proteolytic activity of the tested P. fluorescens strains are not regulated by acyl-homoserine lactones. It is likely that AHL-dependent quorum sensing system is absent from these strains.
Keywords: milk, Pseudomonas fluorescens, biofilm formation, quorum sensing, quorum quenching
Introduction
Bacteria communicate using small diffusible signalling molecules, called autoinducers, to coordinate gene expression in response to population density in a mechanism known as quorum sensing (QS) (Fuqua and Winans, 1994; Whitehead et al., 2001). Many different types of QS signals were identified and the fatty acid derivatives (N-acylhomoserine lactones - AHLs), or autoinducer-1 (AI1), produced by Gram-negative bacteria are the best known and most studied (Fuqua et al., 1996; Eberl, 1999; Whitehead et al., 2001). Quorum sensing allows bacteria to control different functions such as surface colonization and motility, production of exopolymers and antibiotics, biofilm development, bioluminescence, cell differentiation, competence, pigment production, conjugation, sporulation, toxin production, virulence gene expression, and production of several hydrolytic enzymes (Smith et al., 2004). Many of these phenotypes including biofilm formation, motility, production of enzymes, and toxins are important to define microbial activity in foods.
Although signalling compounds are produced by bacteria in foods (Gram et al., 1999; Cloak et al., 2002; Gram et al., 2002; Christensen et al., 2003; Jay et al., 2003; Bruhn et al., 2004; Jay, 2005), the role of QS in food deterioration is unknown. Pinto and collaborators (Pinto et al., 2007) demonstrated that AHL-production is common among many psychrotrophic bacteria isolated from raw milk. Since all these microorganisms were isolated from the same source, cross-communication is relevant and raises the question of the kinds of phenotypes that are regulated when they are all growing together, and the relation of those to spoilage. The understanding of the role of QS in regulating spoilage phenotypes in bacteria is relevant and may be used to create new ways to preserve food products (Pinto et al., 2007). Pillai and Jesudhasan (2006) reinforce that understanding the mechanism of quorum sensing may hold the key to food preservation and prevention of pathogen and spoilage bacterial growth and persistence in foods.
Among Gram-negative psychrotrophic bacteria, Pseudomonas prevails in refrigerated raw milk (Wiedmann et al., 2000; Dogan and Boor, 2003; Pinto et al., 2006), since they present a well-established physiologic mechanism of growth at low temperatures (Jay et al., 2003). In the Pseudomonas genus, Pseudomonas fluorescens constitutes the major milk deteriorative species (Wiedmann et al., 2000; Dogan and Boor, 2003; Pinto et al., 2006) due to its ability to produce thermostable proteases and lipases that hydrolyze casein and lipids decreasing yield and sensory quality of dairy products (Sørhaug and Stepaniak, 1997; Wiedmann et al., 2000; Dogan and Boor, 2003).
Quorum sensing systems based on AHLs signalling molecules have been identified in several P. fluorescens strains, as it has been shown for strain NCIMB 10586 (El-Sayed et al., 2001), 2–79 (Shaw et al., 1997; Cha et al., 1998; Khan et al., 2005), F113 (Koerstgens et al., 2001), 5064 (Cui et al., 2005), 2P24 (Wei and Zhang, 2006), and 395 (Liu et al., 2007). In strain NCIMB 10586, quorum sensing regulates the synthesis of the antibiotic mupirocin through a LuxR-LuxI homologous system from Vibrio fisheri; in strain F113, AHLs are synthesized by a novel synthase; in strain 5064 the system regulates biosurfactant production but the details have not been worked out; in strain 2P42 quorum sensing regulates root colonization, biofilm formation and plant disease-suppressive ability through a regulatory system homologous to LuxR-LuxI; and finally in strain 395, AHLs had a slight effect on aprX expression through a system not understood. However, several studies have shown that other strains of P. fluorescens do not produce AHLs, as it has been the case for strain 1855.344 (Cha et al., 1998), B52 (Allison et al., 1998), and SBW25 (Bruijn and Raaijmakers, 2009). Furthermore, AHL molecules did not influence growth and proteolytic activity of a strain isolated from milk (Pinto et al., 2010) neither the production of antifungal metabolites by P. fluorescens 2P24 used as a biocontrol agent (Wei and Zhang, 2006). A better understanding of the role of QS on the spoilage potential of P. fluorescens strains from food sources is of great interest due to their importance in food deterioration.
Biofilm formation and bacterial food spoilage due to hydrolytic enzymes generate significant problems to the food industry (Gram et al., 2002). Bacteria can also use swarming motility to colonize nutrient-rich environments, which facilitates colony spreading and accelerates biomass production (Fraser and Hughes, 1999). This work aimed to determine if phenotypes related to food spoilage such as biofilm formation, swarming motility and proteolytic activity of P. fluorescens strains isolated from refrigerated raw milk are regulated by AHL molecules.
Material and Methods
Bacterial strains and growth conditions
Two highly proteolytic P. fluorescens strains (07A and 041) previously isolated from raw milk (Martins et al., 2005) where chosen for this study due to their spoilage potential of milk and dairy products (Pinto et al., 2006). The transconjugant strains were obtained by cloning the gentamicin-3-acetyltransferase gene on broad-host-range expression vector which was transferred to the wild-type strains as described later. The strains and plasmids used in this study are listed in Table 1.
Table 1.
Bacterial strains and plasmids used in this study.
| Strain | Plasmid | Description | Reference or source |
|---|---|---|---|
| P. fluorescens 07A | Wild type | (Martins et al., 2005) | |
| P. fluorescens 07A-2 | pMLBAD-aiiA-Trmr-Gmr | Transconjugant, express the lactonase enzyme | This study |
| P. fluorescens 041 | Wild type | (Martins et al., 2005) | |
| P. fluorescens 041-3 | pMLBAD-aiiA-Trmr-Gmr | Transconjugant, express the lactonase enzyme | This study |
| E. coli MT102 | pSB403, Tcr | Monitor strain: exhibits the highest sensitivity for 3-oxo-C6-HSL. However, several other AHL molecules are detected by this sensor. | (Winson et al., 1998; Viana, 2006) |
| E. coli XL1-Blue | pMLBAD-aiiA- Trmr | Donor of pMLBAD-aiiA, Trmr that codify the lactonase enzyme | (Wopperer et al., 2006) |
| E. coli XL1-Blue | pMLBAD-aiiA- Trmr-Gmr | Donor of pMLBAD-aiiA, Gmr that codify the lactonase enzyme | This study |
| E. coli HB101 | pRK600, Cmr | Helper | Laboratory of Microbiology, University of Zürich |
The strains were grown at 25 °C in Luria-Bertani (LB), King’s B and TYEP medium. The transconjugant strains were grown in media supplemented with gentamicin and trimethroprim 20 μg mL−1. Escherichia coli MT102 pSB403 was grown at 30 °C in Luria-Bertani (LB) medium supplemented with tetracycline 50 μg mL−1 (Winson et al., 1998). Growth of liquid cultures was monitored spectrophotometrically with an Ultrospec 3100 Pro spectrophotometer (Biochrom, Ltd., Cambridge, England) by measurement of the optical density at 600 nm.
DNA manipulations
Cloning, restriction enzyme analysis, and transformation of E. coli were performed using established procedures. PCR was performed with TaKaRa Ex Taq polymerase (TaKaRa Shuzo, Shiga, Japan). Plasmid DNA was isolated using the QIAprep Spin Miniprep kit, and chromosomal DNA was purified with the DNeasy tissue kit. DNA fragments were purified from agarose gels by using the QIAquick gel extraction kit (all kits from Qiagen, Hilden, Germany).
Construction of the P. fluorescens transconjugant strains
The gentamicin-3-acetyltransferase gene (GenBank accession number U25061) of pBBR1MCS-5 was amplified by using the primer pair Gem-F (5′ ATT ATG CAT GAA CCT GAA TCG CCA GCG G 3′) and Gem-R (5′ ATT ATG CAT GTT GAA CGA ATT GTT AGG TGG C 3′). The introduced restriction site NsiI is underlined. The amplicon was digested with NsiI and ligated directionally into the broadhost-range expression vector pMLBAD-aiiA-Trmr yielding pMLBAD-aiiA-Trmr-Gmr. This plasmid containing the aiiA gene was transferred to E. coli XL1-Blue by transformation. The aiiA gene encodes the lactonase enzyme that hydrolyzes the ester bond of the homoserine lactone ring of AHLs, thus inhibiting QS communication (Dong et al., 2000).
Afterwards, plasmids were delivered to P. fluorescens strains by triparental mating as previously described (de Lorenzo and Timmis, 1994). Briefly, donor (E. coli XL1-Blue pMLBAD-aiiA-Trmr-Gmr) and recipient strains, as well as the helper strain E. coli HB101 (pRK600), were grown overnight in 5 mL of LB medium supplied with the appropriate antibiotics. After culturing to an optical density of 0.9 at 600 nm, 2 mL of cell culture were harvested, washed, and resuspended in 500 μL of LB medium. Donor and helper cells (100 μL each) were mixed and incubated for 10 min at room temperature. Then, 200 μL of the recipient cells were added and the mixture was spot inoculated onto the surfaces of pre-heated LB agar plates. After overnight incubation at 30 °C, the cells were plated on Pseudomonas Isolation Agar (PIA) (Becton Dickinson Biosciences, Sparks, MD) containing antibiotics for counter selection of the donor, helper, and untransformed recipient cells.
Quantification of AHLs
Since E. coli MT102 pSB403 is able to detect low amounts of AHL, it was used for AHL detection (Winson et al., 1998). The plasmid pSB403 contains the Vibrio fischeri luxR gene with luxI promoter region as a transcriptional fusion to the bioluminescence genes luxCDABE. The V. fischeri quorum sensing system relies on 3-oxo-C6-HSL, so the sensor plasmid consequently exhibits the highest sensitivity towards this AHL molecule. However, several other AHL molecules are detected by the sensor strain, albeit with reduced sensitivity (Winson et al., 1998). A volume of 1 mL of overnight culture of E. coli MT102 pSB403 was inoculated into 5 mL LB supplemented with tetracycline and incubated at 30 °C for 1 h. Then, 100 μL of filter-sterilized culture supernatants in LB of P. fluorescens 07A and 041 were added to 100 μL of an exponential culture of the sensor strain in the wells of a microtiter dish. After incubation at 30 °C for 3 h, the expression of the bioluminescence reporter genes was measured using the program KC4 (Bio-Tek Instruments, Highland Park, Box 998, Vermont, USA). AHL concentrations were determined by comparing bioluminescence signal intensities with a defined concentration (0.1 mg mL−1) of pure 3-oxo-C6-HSL.
Extraction of putative signalling molecules from supernatants of P. fluorescens
P. fluorescens 07A and 041 wild-type and transconjugant strains (104 CFU mL−1) were inoculated in 600 mL of King’s B, LB, and TYEP. The cultures were incubated with aeration at 25 °C for 20 h or until the population reached 109 CFU mL−1. Then, the cells were harvested by centrifugation at 10,000 g for 20 min at 4 °C and 250 mL of the cell free supernatants were mixed with 100 mL of dichloromethane stabilized with ethanol in a 1,000 mL separating funnel. The mixture was shacken for 3 min with aeration every 20 s. When the two phases were separated, the dichloromethane-phase was collected (lower phase). The upper phase (aqueous phase) was mixed with 100 mL of dichloromethane and shacken again as described above. Lower dichloromethane-phase was collected and mixed with the first one. These steps were repeated until finishing the 600 mL of supernatant. Then, the remaining water was removed with water free MgSO4 and it was filtered using Whatman paper. The filtered extracts were concentrated in a rotary evaporator at 40 °C, resuspended in 250 μL ethyl acetate, and maintained at -20 °C.
Detection of putative signalling molecules in supernatant extracts of P. fluorescens
Thirty milliliter of overnight culture of E. coli MT102 pSB403 were inoculated in 150 mL of LB agar. The inoculated LB plates were allowed to solidify and then 6 μL of extracts obtained from the supernatant of King’s B, LB, and TYEP inoculated with P. fluorescens 07A were transferred as drops to the plate’s surface. Aliquots of 0.6 μL of HHL 1 mg mL−1 were used as positive controls. The plates were incubated overnight and the activation of the AHL monitor strain E. coli MT102 pSB403 was observed into a dark box that contained a highly sensitive photon-counting camera (C2400-40; Hamamatsu Photonics Herrsching, Germany) as described by Steidle et al. (2001).
Phenotypic characterization of wild type and transconjugant strains
Biofilm formation in polystyrene microtiter dishes was assayed essentially as described previously (Pratt and Kolter, 1998) with a few modifications. Cells of P. fluorescens 07A and 041 wild-type and transconjugant were grown in the wells of microtiter dishes in 100 μL of LB, minimal medium salt (MMS) or ABC medium supplemented with 10 mM citrate for 48 h at 25 °C. Thereafter, the medium was removed, and 100 μL of a 1% (wt/vol) aqueous solution of crystal violet (CV) was added. After staining at room temperature for 20 min, the dye was removed, and the wells were washed thoroughly. For quantification of attached cells, the CV was solubilized in an 800:120 (v/v) mixture of ethanol and dimethyl sulfoxide, and the absorbance was determined at 570 nm. This assay was done in two biological replicates and five independent experiments.
The ability to form a swarming colony was tested by point inoculating the strains into ABC minimal medium supplemented with 0.1% casamino acids and solidified with 0.4% agar as previously described (Eberl et al., 1996; Huber et al., 2001).
Proteolytic activity was determined in ABC, MMS, and TYEP using azocasein assay as previously described (Christensen et al., 2003). Briefly, this activity was investigated on azocasein by incubating 250 μL of 2% azocasein (w/v) with 150 μL sterile filtered culture supernatant. The mixture was incubated at 30 °C for 12 h. Subsequently, the mixture was incubated at room temperature for 15 min with 1.2 mL of 10% (w/v) trichloroacetic acid (TCA), and centrifuged for 10 min at 15,000 g. Prior to spectroscopic measurement, 600 μL supernatant were rescued and mixed with 750 μL 1 M NaOH. The proteolytic activity was quantified by the determination of the OD440 against a blank reaction mixture with 150 μL culture media or 75 μL Tris-HCl 20 mM, pH 8.0, CaCl2 5 mM instead of the enzyme solution. One unit of proteolytic activity was defined as the unit of enzyme activity per hour per μg of protein. The method of Bradford (1976), using bovine serum albumin as a standard, was used to quantify protein concentrations in supernatant of media. This assay also was done in two biological replicates and five independent experiments.
Results and Discussion
Detection of bioluminescence induced by P. fluorescens
Potential signalling molecules present in supernatants obtained from P. fluorescens 07A and 041 did not induce E. coli MT102 pSB403 (Table 2). Therefore, this result suggests that P. fluorescens 07A and 041 isolated from cooled raw milk did not produce AHLs able to induce the high sensitive biosensor E. coli MT102 pSB403. Pinto and collaborators (2010) used another highly sensitive bioassay strain (Agrobacterium tumefaciens KYC55) and did not detect AHL molecules from P. fluorescens 07A, confirming the present results. Other biosensor strains have also been used and were not able to detect AHLs in this strain (Pinto et al., 2007). According to Winson et al. (1998), there is a significant advantage of using lux sensors since the sensitivity to AHL is in picomol to nanomol concentrations over a large linear range.
Table 2.
Values of bioluminescence produced by E. coli MT102 pSB403 at 175 nm, after growth in LB broth supplemented with supernatant of P. fluorescens strains and supplemented with 3-oxo-C6-HSL. Data represent average of triplicate experiments.
| Dilution rate | P. fluorescens 07A | P. fluorescens 041 | LB Negative control | 3-oxo-C6-HSL Positive control |
|---|---|---|---|---|
| 1/2 | 13532 | 15823 | 14820 | Nd1 |
| 1/4 | 15261 | 13831 | 15003 | Nd |
| 1/8 | 13967 | 15360 | 13230 | Nd |
| 1/16 | 14401 | 16580 | 15340 | Nd |
| 1/32 | 16977 | 16036 | 16720 | 51802 |
| 1/64 | 15862 | 14159 | 14579 | 28723 |
| 1/128 | 17092 | 17619 | 16220 | 21817 |
| 1/256 | 15716 | 13420 | 14943 | 20942 |
Nd - not determined. The intensity of the signal was higher than the detection limit of the equipment.
Supplementation of LB inoculated with E. coli MT102 pSB403 with extracts obtained from different media inoculated with P. fluorescens
As no activity derived from signalling molecules was found in the supernatant of LB medium inoculated with P. fluorescens 07A and 041 on the microtiter dish assay (Table 2), a correlation between the growth media and the production of signalling compounds was tested, but no AHL was found in the supernatant extracts obtained from King’s B, LB, and TYEP media inoculated with P. fluorescens. These data confirm that these strains of P. fluorescens do not produce AHLs, contrasting with studies that found other strains of P. fluorescens as AHL producers (Shaw et al., 1997; Cha et al., 1998; Laue et al., 2000; El-Sayed et al., 2001; Mcphee, 2001; Khan et al., 2005).
Mcphee (2001) found that synthesis of AHLs by Pseudomonas was influenced by the composition of the growth medium and environmental factors. According to Mcphee (2001), P. fluorescens was found to up-regulate enzyme synthesis when it grew in a spent culture supernatant, presumably having already high levels of synthesized AHL.
Phenotypic characteristics of wild type and transconjugant P. fluorescens strains
Biofilm
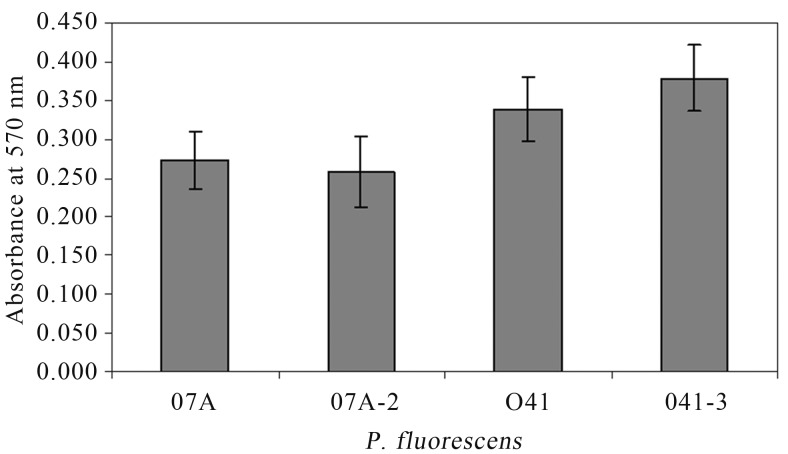
After 48 h of incubation, it was observed that P. fluorescens 07A and 041 produced less biofilm in LB and MMS than in ABC medium. The strain 041 was able to bind better than 07A in polystyrene microtiter dishes (Figure 1). Viana (2006) also observed that different strains of P. fluorescens isolated from raw milk had different abilities to bind to polystyrene and that minimal medium enhanced attachment. The ABC minimal medium is rich in divalent ions such as Ca+2, Mg+2, and Fe+2 and that could explain this phenotype. According to Fletcher et al. (1988), divalent ions as Ca+2 and Mg+2 can directly influence biofilm formation due to electrostatic interactions, and indirectly as enzyme cofactors that influence the adhesion dependent on the microorganism physiology. Additionally, the presence of ions such as Ca+2 improve cross-binding between cells and between cells and surfaces (Koerstgens et al., 2001).
Figure 1.
Biofilm formation by P. fluorescens wild type (07A and 041) and transconjugant (07A-2 and 041-3) strains in ABC medium after incubation for 48 h at 25 °C, in polystyrene microtiter plates using crystal violet staining, and quantified based on the difference between the absorbance at 570 nm. Values are means ± standard errors (n = 5).
No significant difference (p > 0.01) was found when wild type and transconjugant strains were compared for their ability to produce biofilm (Figure 1). This result shows that the quorum quenching mechanism provided by the lactonase enzyme AiiA, which cleaves AHL molecules (Dong et al., 2000), did not influence biofilm formation in these particular strains of P. fluorescens, presumably because they are unable to produce detectable levels of AHLs (Table 2). Allison et al. (1998) suggested that QS was involved in promoting cell attachment and biofilm formation in P. fluorescens B52, but short chain AHLs were not involved. A mutant of P. fluorescens incapable to produce QS signals was significantly defective in biofilm formation (Wei and Zhang, 2006).
Swarming motility
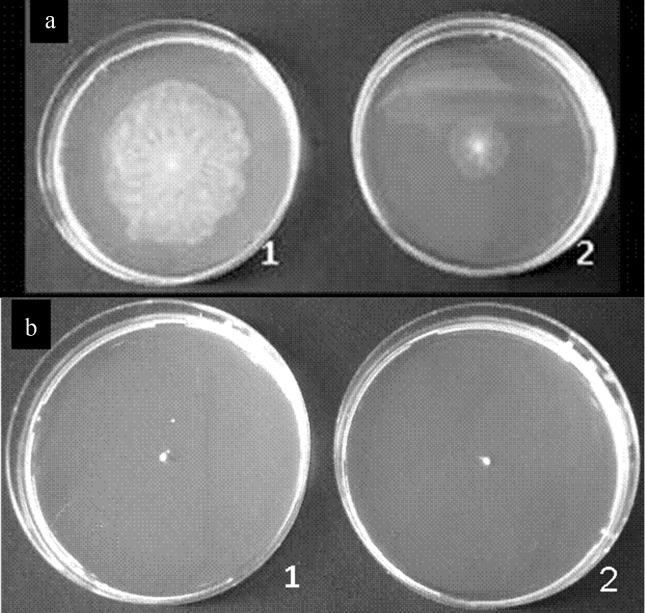
To test swarming motility, the strains 07A and 041 were point inoculated into medium containing 0.4% agar. Only P. fluorescens 07A was capable of swarming (Figure 2). When AiiA was expressed in P. fluorescens 07A, swarming motility was reduced. Since no other evaluated phenotype had been influenced by the presence of the plasmid expressing AiiA in these strains, we believe that this assay was somehow compromised due to the sensitivity of the P. fluorescens 07A transconjugant strain to the presence of gentamicin on the medium where the strain was previously grown. Additionally, an unknown factor required for swarming motility may have been compromised in this strain due to unexpected reasons.
Figure 2.
Ability to form a swarming colony on ABC medium after incubation for 18 h at 25 °C. (a) 1, P. fluorescens 07A wild type; 2, P. fluorescens 07A-2 transconjugant. (b) 1, P. fluorescens 041 wild type; 2, P. fluorescens 041-3 transconjugant.
Extracellular protease
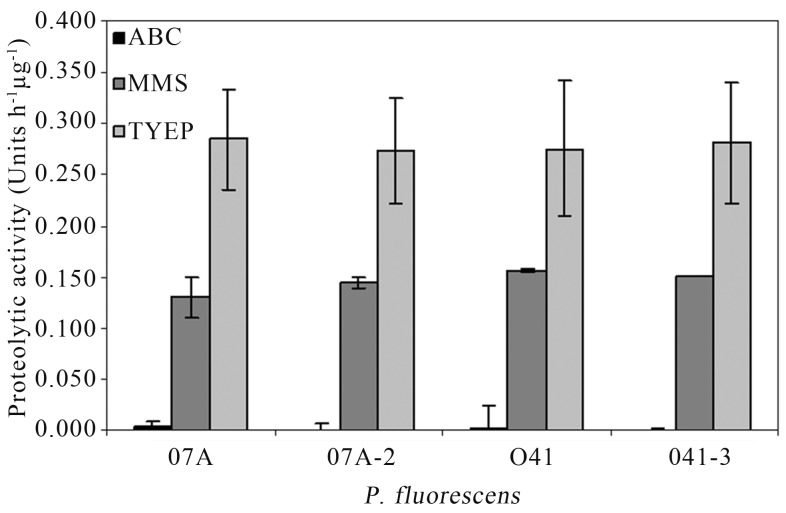
AHL-dependent QS systems control the production of extracellular proteolytic activity in many Gram-negative bacteria (Whitehead et al., 2001; Kastbjerg et al., 2007). However, quorum quenching provided by AiiA did not influence proteolytic activity in P. fluorescens 07A and 041 when they were grown in different broth media (Figure 3), indicating that the AHL-dependent regulation of this phenotype is not conserved in P. fluorescens strains. Pinto et al. (2010) also observed that synthetic AHLs or bacterial cell extracts obtained from P. fluorescens 07A added to the medium did not influence growth or proteolytic activity suggesting that QS does not regulate protease production in that strain. The present study further confirms the results with strain 07A, using a totally different approach and expands the knowledge to another importantly spoilage strain (Martins et al., 2005; Pinto et al., 2006). Given that P. fluorescens are quite diverse, it is not surprising that some strains produce AHLs (Shaw et al., 1997; Cha et al., 1998; Laue et al., 2000; El-Sayed et al., 2001; Cui et al., 2005; Khan et al., 2005; Wei and Zhang, 2006; Liu et al., 2007) and some others do not (Allison et al., 1998; Cha et al., 1998; Bruijn and Raaijmakers, 2009; Pinto et al., 2010).
Figure 3.
Proteolytic activity on supernatant of ABC minimal medium, MMS minimal medium and TYEP medium inoculated with P. fluorescens wild type (07A and 041) and transconjugants (07A-2 and 041-3) after 24 h of incubation at 25 °C. Values are means ± standard errors (n = 5).
Our findings further highlight the diversity and complexity of P. fluorescens isolates and reiterate the importance of studies of this kind to improve our knowledge about this group of microorganisms. It would interesting to analyze a wide collection of P. fluorescens strains isolated from raw milk in order to determine if AHL production is a common trait or an exception in strains predominating in this particular environment.
Conclusions
The production of AHLs by P. fluorescens 07A and 041 was not detected under any of the conditions used in this study. Both strains produce less biofilm in LB and MMS than in ABC minimal medium, and the strain 041 is better able to bind to polystyrene microtiter dishes than 07A. It was also verified that biofilm formation, swarming motility and proteolytic activity of P. fluorescens strains isolated from refrigerated raw milk are not regulated by AHLs. It is likely that the AHL-dependent quorum sensing system is absent from these strains.
Acknowledgments
Maurilio L. Martins was supported by the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) Brasília, Brazil. We would like to thank the CNPq and the Fundação de Amparo à Pesquisa do Estado de Minas Gerais (FAPEMIG) for supporting this research. We are thankful to Leo Eberl from the University of Zurich - Switzerland for his assistance with this work.
References
- Allison DG, Ruiz B, Sanjose C, Jaspe A, Gilbert P. Extracellular products as mediators of the formation and detachment of Pseudomonas fluorescens biofilms. FEMS Microbiol Lett. 1998;167:179–184. doi: 10.1111/j.1574-6968.1998.tb13225.x. [DOI] [PubMed] [Google Scholar]
- Bradford MM. A rapid and sensitive method for the quantification of microgram quantities of proteins utilizing the principle of protein dye binding. Anal Biochem. 1976;72:248–274. doi: 10.1006/abio.1976.9999. [DOI] [PubMed] [Google Scholar]
- Bruhn JB, Christensen AB, Flodgaard LR, Nielsen KF, Larsen T, Givskov M, Gram L. Presence of acylated homoserine lactones (AHLs) and AHL-producing bacteria in meat and potential role of AHL in spoilage of meat. App Environ Microbiol. 2004;70:4293–4302. doi: 10.1128/AEM.70.7.4293-4302.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bruijn I, Raaijmakers JM. Diversity and Functional Analysis of LuxR Type Transcriptional Regulators of Cyclic Lipopeptide Biosynthesis in Pseudomonas fluorescens. App Environ Microbiol. 2009;75(14):4753–4761. doi: 10.1128/AEM.00575-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cha C, Gao P, Chen YC, Shaw PD, Farrand SK. Production of acyl-homoserine lactone quorum-sensing signals by Gram-negative plant-associated bacteria. Mol Plant Microbe Interact. 1998;11:1119–1129. doi: 10.1094/MPMI.1998.11.11.1119. [DOI] [PubMed] [Google Scholar]
- Christensen AB, Riedel K, Eberl L, Flodgaard LR, Molin S, Gram L, Givskov M. Quorum-sensing-directed protein expression in Serratia proteamaculans B5a. Microbiol. 2003;149:471–483. doi: 10.1099/mic.0.25575-0. [DOI] [PubMed] [Google Scholar]
- Cloak OM, Slow BT, Briggs C, Chen CY, Fratamico PM. Quorum sensing and production of autoinducer-2 in Campylobacter spp., Escherichia coli O157:H7, and Salmonella enterica serovar Typhimurium in foods. Appl Environ Microbiol. 2002;68:4666–4671. doi: 10.1128/AEM.68.9.4666-4671.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cui X, Harling R, Mutch P, Darling D. Identification of N-3-hydroxyoctanoyl-homoserine lactone production in Pseudomonas fluorescens 5064, pathogenic to broccoli, and controlling biosurfactant production by quorum sensing. EJPP. 2005;111:297–308. [Google Scholar]
- de Lorenzo V, Timmis KN. Analysis and construction of stable phenotypes in Gram-negative bacteria with Tn5 and Tn10-derived mini-transposons. Methods Enzymol. 1994;235:386–405. doi: 10.1016/0076-6879(94)35157-0. [DOI] [PubMed] [Google Scholar]
- Dogan B, Boor KJ. Genetic diversity and spoilage potentials among Pseudomonas spp. isolated from fluid milk products and dairy processing plants. Appl Environ Microbiol. 2003;69:130–138. doi: 10.1128/AEM.69.1.130-138.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dong YH, Xu JL, Li XC, Zhang LH. AiiA, a novel enzyme inactivates acyl homoserine-lactone quorum-sensing signal and attenuates the virulence of Erwinia carotovora. Proc Natl Acad Sci USA. 2000;97:3526–3531. doi: 10.1073/pnas.060023897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eberl L, Winson MK, Sternberg C, Stewart GSAB, Christiansen G, Chhabra SR, Bycroft B, Williams P, Molin S, Givskov M. Involvement of N-acyl-L-homoserine lactone autoinducers in controlling the multicellular behavior of Serratia liquefaciens. Mol Microbiol. 1996;20:127–136. doi: 10.1111/j.1365-2958.1996.tb02495.x. [DOI] [PubMed] [Google Scholar]
- Eberl L. N-Acyl homoserinelactone-mediated gene regulation in Gram-negative bacteria. Syst Appl Microbiol. 1999;22:493–506. doi: 10.1016/S0723-2020(99)80001-0. [DOI] [PubMed] [Google Scholar]
- El-Sayed AK, Hothersall J, Thomas CM. Quorum sensing-dependent regulation of biosynthesis of the polyketide antibiotic mupirocin in Pseudomonas fluorescens NCIMB 10586. Microbiol. 2001;147:127–2139. doi: 10.1099/00221287-147-8-2127. [DOI] [PubMed] [Google Scholar]
- Fletcher M. Attachment of Pseudomonas fluorescens to glass and influence of electrolytes on bacterium-substratum separation distance. J Bacteriol. 1988;170:2027–2030. doi: 10.1128/jb.170.5.2027-2030.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fraser GM, Hughes C. Swarming motility. Curr Opin Microbiol. 1999;2:630–635. doi: 10.1016/s1369-5274(99)00033-8. [DOI] [PubMed] [Google Scholar]
- Fuqua C, Winans SC. A luxR-LuxI type regulatory system activates Agrobacterium Ti plasmid conjugal transfer in the presence of a plant tumor metabolite. J Bacteriol. 1994;176:2796–2806. doi: 10.1128/jb.176.10.2796-2806.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fuqua C, Winans SC, Greenberg EP. Census and consensus in bacterial ecosystems: the luxR-LuxI family of quorum-sensing transcriptional regulators. Annu Rev Microbiol. 1996;50:727–751. doi: 10.1146/annurev.micro.50.1.727. [DOI] [PubMed] [Google Scholar]
- Gram L, Christensen AB, Ravn L, Molin S, Givskov M. Production of acylated homoserine lactones by psychrotrophic members of the Enterobacteriaceae isolated from foods. Appl Environ Microbiol. 1999;65:3458–3463. doi: 10.1128/aem.65.8.3458-3463.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gram L, Ravn L, Rasch M, Bruhn JB, Christensen AB, Givskov M. Food spoilage-interactions between food spoilage bacteria. Int J Food Microbiol. 2002;78:79–97. doi: 10.1016/s0168-1605(02)00233-7. [DOI] [PubMed] [Google Scholar]
- Huber B, Riedel K, Hentzer M, Heydorn A, Gotschlich A, Givskov M, Molin S, Eberl L. The cep quorum-sensing system of Burkholderia cepacia H111 controls biofilm formation and swarming motility. Microbiol. 2001;147:2517–2528. doi: 10.1099/00221287-147-9-2517. [DOI] [PubMed] [Google Scholar]
- Jay JM, Loessner MJ, Golden DA. Modern Food Microbiology. 7 ed. Chapman & Hall; New York: 2005. p. 790. [Google Scholar]
- Jay JM, Vilai JP, Hughes ME. Profile and activity of the bacterial biota of ground beef held from freshness to spoilage at 5–7 °C. Int J Food Microbiol. 2003;81:105–111. doi: 10.1016/s0168-1605(02)00189-7. [DOI] [PubMed] [Google Scholar]
- Kastbjerg VG, Nielsen KF, Dalsgaard I, Rasch M, Bruhn JB, Givskov M, Gram L. Profiling acylated homoserine lactones in Yersinia ruckeri and influence of exogenous acyl homoserine lactones and known quorum-sensing inhibitors on protease production. J Appl Microbiol. 2007;102:363–374. doi: 10.1111/j.1365-2672.2006.03109.x. [DOI] [PubMed] [Google Scholar]
- Khan SR, Mavrodi DV, Jog GJ, Suga H, Thomashow LS, Farrand SK. Activation of the phz operon of Pseudomonas fluorescens 2–79 requires the LuxR homolog PhzR, N-(3-OH-Hexanoyl)-L-Homoserine lactone produced by the LuxI homolog PhzI, and a cis-Acting phz Box. J Bacteriol. 2005;187:6517–6527. doi: 10.1128/JB.187.18.6517-6527.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koerstgens V, Flemming HC, Wingender J, Borchard W. Influence of calcium ions on the mechanical properties of a model biofilm of mucoid Pseudomonas aeruginosa. Water Sci Technol. 2001;43:49–57. [PubMed] [Google Scholar]
- Laue BE, Jiang Y, Chhabra SR, Jacob S, Stewart GSAB, Hardman A, Downie JA, O’Gaara F, Williams P. The biocontrol strain Pseudomonas fluorescens F113 produces the Rhizobium small bacteriocin, N-(hydroxy-7-cis-tetradecenoyl) homoserine lactone, via HdtS, a putative novel N-acylhomoserine lactone synthase. Microbiol. 2000;146:2469–2480. doi: 10.1099/00221287-146-10-2469. [DOI] [PubMed] [Google Scholar]
- Liu M, Wang H, Griffiths MW. Regulation of alkaline metalloprotease promoter by N-acylhomoserine lactone quorum sensing in Pseudomonas fluorescens. J Appl Microbiol. 2007;103:2174–2184. doi: 10.1111/j.1365-2672.2007.03488.x. [DOI] [PubMed] [Google Scholar]
- Martins ML, Araújo EF, Mantovani HC, Moraes CA, Vanetti MCD. Detection of the apr gene in proteolytic psychrotrophic bacteria isolated from refrigerated raw milk. Int J Food Microbiol. 2005;102:203–21. doi: 10.1016/j.ijfoodmicro.2004.12.016. [DOI] [PubMed] [Google Scholar]
- Mcphee JD. M.Sc. Dissertation. University of Guelph; Canada: 2001. The role of quorum sensing in the regulation of extracellular enzymes by Pseudomonas fluorescens; p. 135. [Google Scholar]
- Pillai SD, Jesudhasan PR. Quorum sensing: How bacteria communicate. Food Technol. 2006;60:42–50. [Google Scholar]
- Pinto CLO, Martins ML, Vanetti MCD. Qualidade microbiológica de leite refrigerado e isolamento de bactérias psicrotróficas proteolíticas. Cienc Tecnol Aliment. 2006;26:1–7. [Google Scholar]
- Pinto UM, Costa ED, Mantovani HC, Vanetti MCD. The proteolytic activity of Pseudomonas fluorescens 07A isolated from Milk is not regulated by quorum sensing signals. Braz J Microbiol. 2010;41:91–96. doi: 10.1590/S1517-838220100001000015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pinto UM, Viana ES, Martins ML, Vanetti MCD. Detection of acylated homoserine lactones in Gram-negative proteolytic psychrotrophic bacteria isolated from cooled raw milk. Food Cont. 2007;18:1322–1327. [Google Scholar]
- Pratt LA, Kolter R. Genetic analysis of Escherichia coli biofilm formation: roles of flagella, motility, chemotaxis and type I pili. Mol Microbiol. 1998;30:285–293. doi: 10.1046/j.1365-2958.1998.01061.x. [DOI] [PubMed] [Google Scholar]
- Shaw PD, Ping G, Daly SL, Cha C, Cronan JEJ, Rinehart KL, Farrand SK. Detecting and characterizing N-acyl homoserine lactone signal molecules by thin -layer chromatography. Proc Natl Acad Sci USA. 1997;94:6036–6041. doi: 10.1073/pnas.94.12.6036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith JL, Fratamico PM, Novak JS. Quorum sensing: A primer for food microbiologists. J Food Protec. 2004;67:1053–1070. doi: 10.4315/0362-028x-67.5.1053. [DOI] [PubMed] [Google Scholar]
- Sørhaug T, Stepaniak L. Psychrotrophs and their enzymes in milk and dairy products: Quality aspects. Trend in Food Sci Technol. 1997;8:35–40. [Google Scholar]
- Steidle A, Sigl K, Schuhegger R, Ihring A, Schmid M, Gantner S, Stoffels M, Riedel K, Givskov M, Hartmann A, Langebartels C, Eberl L. Visualization of N-acylhomoserine lactone-mediated cell-cell communication between bacteria colonizing the tomato rhizosphere. Appl Environ Microbiol. 2001;67:5761–5770. doi: 10.1128/AEM.67.12.5761-5770.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Viana ES. Ph.D. Thesis. Universidade Federal de Viçosa; Viçosa: 2006. Moléculas sinalizadoras de quorum sensing em biofilmes formados por bactérias psicrotróficas isoladas de leite; p. 159. [Google Scholar]
- Whitehead NA, Barnard AML, Slater H, Simpsom NJL, Salmond GPC. Quorum-sensing in Gram-negative bacteria. FEMS Microbiol Rev. 2001;25:365–404. doi: 10.1111/j.1574-6976.2001.tb00583.x. [DOI] [PubMed] [Google Scholar]
- Wei HL, Zhang LQ. Quorum-sensing system influences root colonization and biological control ability in Pseudomonas fluorescens 2P24. Antonie van Leeuwenhoek. 2006;89:267–280. doi: 10.1007/s10482-005-9028-8. [DOI] [PubMed] [Google Scholar]
- Wiedmann M, Weilmeier D, Dineen SS, Ralyea R, Boor K. Molecular and phenotypic characterization of Pseudomonas spp. isolated from milk. Appl Environ Microbiol. 2000;66:2085–2095. doi: 10.1128/aem.66.5.2085-2095.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Winson MK, Swift S, Fish L, Throup JP, Jørgensen F, Chabra SR, Bycroft BW, Williams P, Stewart GSAB. Construction and analysis of luxCDABE-based plasmid sensors for investigating N-acyl homoserine lactone-mediated quorum sensing. FEMS Microbiol Lett. 1998;163:185–192. doi: 10.1111/j.1574-6968.1998.tb13044.x. [DOI] [PubMed] [Google Scholar]
- Wopperer J, Cardona ST, Huber B, Jacobi CA, Valvano MA, Eberl L. A quorum-quenching approach to investigate the conservation of quorum-sensing-regulated functions within the Burkholderia cepacia complex. Appl Environ Microb. 2006;72:1579–1587. doi: 10.1128/AEM.72.2.1579-1587.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]