Abstract
Development of peripheral neuropathy, which can present as painful neuropathy or loss of sensation, sometimes limit the use of paclitaxel in the treatment of solid tumors such as breast cancer. Previous studies reported development of thermal hyperalgesia in mice treated with paclitaxel. In this study an automated flinch detection system for the formalin test (20 μl of 5% formalin injected subcutaneously into the paw dorsum) was used to evaluate chemical nociception in BALB/c mice treated with paclitaxel 2 mg/kg alone or coadministered with minocycline 50 mg/kg, intraperitoneally for 5 consecutive days. Reaction latency to thermal stimuli (hot-plate) was also measured. Injection of formalin resulted in biphasic paw flinches; phase 1 (1–9 minutes) and phase 2 (10–40 minutes). Treatment with paclitaxel reduced cumulative flinches in both phases 1 and 2 by 28% and 43%, respectively at day 7. However, treatment with paclitaxel also induced thermal hyperalgesia. Co-administration of paclitaxel with minocycline prevented development of both paclitaxel-induced hyposensitivity to chemical nociception and thermal hyperalgesia. In conclusion, the results indicate paclitaxel induces chemical hyposensitivity and thermal hyperalgesia in mice. Minocycline protected against paclitaxel-induced chemical hyposensitivity and thermal hyperalgesia, thus, providing further support of the usefulness of the drug in prevention of chemotherapy-induced neuropathy.
Taxanes such as paclitaxel and docetaxel are used in the treatment of various types of cancer including breast and prostate cancer. However, their use is limited by development of dose-limiting peripheral neuropathy. Paclitaxel-induced peripheral neuropathy, which is predominantly sensory neuropathy, affects the distal extremities, hands and feet, presenting in a glove-stocking pattern1. These sensory neuropathy symptoms can present as paraesthesia, numbness and/or pain, which can severely affect the patient's quality of life2,3,4.
Paclitaxel-induced peripheral neuropathy has been studied extensively using rodent models5,6,7,8,9. The behavioral changes induced by paclitaxel that have been mostly observed are thermal hyperalgesia, cold and mechanical allodynia6,7,8,10 but hypoalgesia has also been reported10,11,12. Few studies have studied the sensory changes to chemical nociception. In one study using rats they observed mechanical allodynia as well as hyperalgesia to chemical nociception induced by formalin after treatment with paclitaxel13, whilst in another study although they observed mechanical hyperalgesia they did not observe any changes in formalin-induced nociception14. No study has reported what occurs to chemical nociception induced by formalin in mice treated with paclitaxel.
The current study sought to address what happens to chemical nociception in mice with paclitaxel-induced neuropathy and whether these changes if they occur can be prevented by a neuroprotective drug. In this study minocycline, a semi-synthetic tetracycline, was used because it has been reported to protect against the development of paclitaxel-induced neuropathy. Previous studies observed that rats treated with minocycline were protected against the development of thermal hyperalgesia, cold and mechanical allodynia as well as loss of intraepidermal nerve fibers15,16,17,18. One study reported that treatment with minocycline also prevented the development of paclitaxel-induced cold hyperalgesia in mice19, whilst another showed that a chemically modified tetracycline can protect against thermal hyperalgesia20. No study as yet has reported the effects of minocycline on paclitaxel-induced changes to chemical nociception.
Results
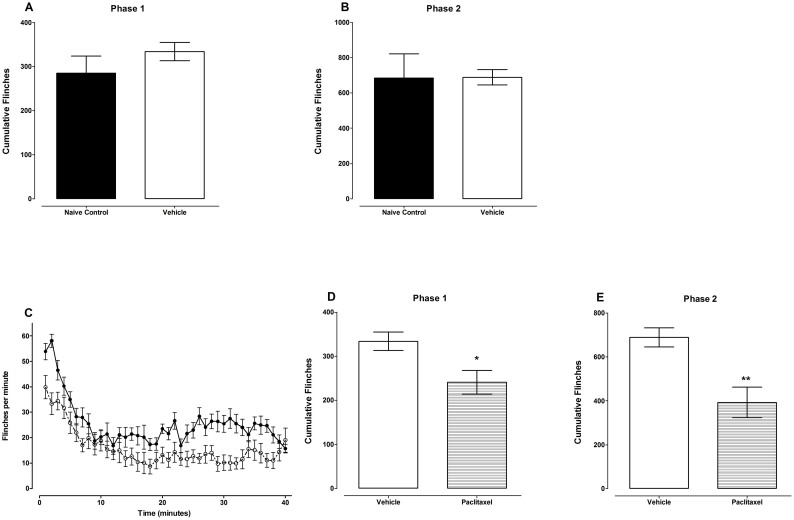
Subcutaneous injection of formalin into the mouse paw dorsum resulted in biphasic paw flinches; phase 1 was defined as from 1–9 minutes and phase 2 from 10–40 minutes. Unpaired Student's t test showed that mice that were treated with vehicle for paclitaxel (Cremophor EL and ethanol diluted in normal saline; n = 12) had cumulative flinches similar to naïve control animals (n = 8) in both phases 1 and 2, 334 ± 21 versus 285 ± 39 and 689 ± 43 versus 685 ± 137, on day 7 after first administration of vehicle (p = 0.245 and p = 0.974, respectively; Fig. 1A and B). Thus, the vehicle-treated mice were used for comparison with paclitaxel-treated mice.
Figure 1. Paclitaxel-induces hyposensitivity to chemical nociception in BALB/c mice.
(A and B) Comparison of BALB/c mice treated with vehicle for paclitaxel (Cremophor EL and ethanol diluted in normal saline on five consecutive days) and naïve mice on paw flinches induced by injection of 5% formalin s.c. on the paw dorsum on day 7 measured using an automated flinch detection system. Cumulative flinches in (A) phase1 (1–9 minutes) and (B) phase 2 (10–40) minutes (n = 8–9 per group). (C, D and E) Effect of treatment with paclitaxel (2 mg/kg on five consecutive days) on paw flinches induced by injection of 5% formalin s.c. on the paw dorsum on day 7 measured using an automated flinch detection system (A) Time course of flinches. Cumulative flinches in (B) phase1 (1–9 minutes) and (C) phase 2 (10–40) minutes (n = 12 per group). * P < 0.05 and * * P < 0.01 compared to vehicle-treated mice.
Unpaired Student's t test showed that administration of paclitaxel produced a significant reduction in the cumulative flinches in the formalin test (chemical hyposensitivity) in both phases 1 and 2 from 334 ± 21 to 241 ± 27 and from 689 ± 43 to 393 ± 70, respectively on day 7 after first drug administration compared to vehicle-treated animals (p < 0.013 and p < 0.002, respectively; n = 12 for both groups; Fig. 1D and E). The reduction in cumulative flinches caused by paclitaxel was 28% and 43%, in phases 1 and 2, respectively.
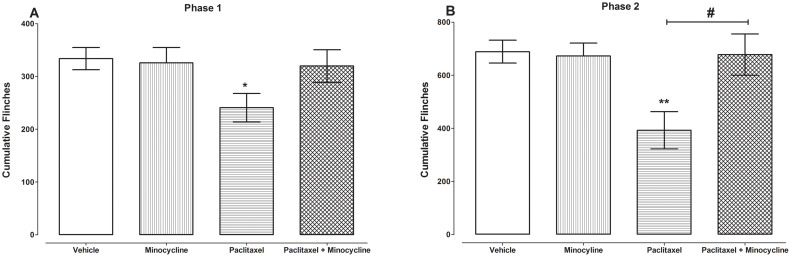
In order to evaluate whether minocycline could prevent the paclitaxel-induced chemical hyposensitivity minocycline was coadministered with paclitaxel and compared to control vehicle-only-treated and paclitaxel plus vehicle-treated mice. Unpaired Student's t test showed that mice that were treated with minocycline alone (n = 7) had cumulative flinches similar to vehicle-treated animals (n = 12) in both phases 1 and 2, 334 ± 21 versus 326 ± 29 and 689 ± 43 versus 673 ± 48, on day 7 after first administration of minocycline or vehicle (p = 0.823 and p = 0.815, respectively; Fig. 2A and B).
Figure 2. Coadministration of minocycline with paclitaxel protects against the development of paclitaxel-induced hyposensitivity to chemical nociception.
Effect of coadministration of paclitaxel with minocycline (50 mg/kg on five consecutive days) on paw flinches induced by injection of 5% formalin s.c. on the paw dorsum on day 7 measured using an automated flinch detection system. Cumulative flinches in (A) phase1 (1–9 minutes) and (B) phase 2 (10–40) minutes (n = 7–15 per group). * P < 0.05 and * * P < 0.01 compared to vehicle-treated mice; # P < 0.05 compared to paclitaxel plus vehicle-treated mice.
Unpaired Student's t test showed that mice that were treated with paclitaxel plus minocycline (n = 15) had cumulative flinches similar to vehicle-only-treated control animals (n = 12) in both phases 1 and 2, 334 ± 21 versus 320 ± 31 and 689 ± 43 versus 678 ± 78, on day 7 after first drug administration (p = 0.669 and p = 0.770, respectively; Fig. 2A and B). Moreover, in phase 2 mice that were treated with paclitaxel plus minocycline (n = 15) had cumulative flinches significantly higher than paclitaxel plus vehicle-treated animals (n = 12), 678 ± 78 versus 393 ± 70, on day 7 after first drug administration (p < 0.014; Fig. 2B).
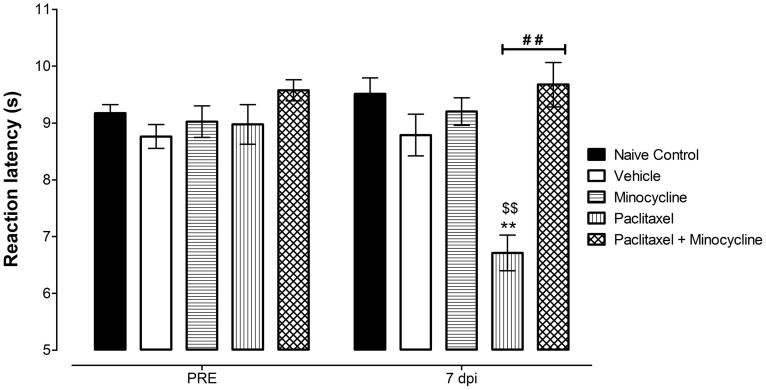
As previously described20 mice treated with paclitaxel were more sensitive to heat compared to vehicle-treated and naïve mice (Fig. 3).
Figure 3. Coadministration of minocycline with paclitaxel protects against the development of paclitaxel-induced thermal hyperalgesia.
Effect of administration of paclitaxel 2 mg/kg, minocycline 50 mg/kg or coadministration of paclitaxel with minocycline on five consecutive days on reaction latency times on baseline (PRE) and on day 7 post drug administration (7 dpi) measured using the hot plate test. (n = 8 per group). ** P < 0.01 compared to vehicle-treated mice; ## P < 0.01 compared to paclitaxel plus vehicle-treated mice; and $$ P < 0.01 compared to baseline values.
Two-way repeated measures ANOVA showed that mice that were treated with vehicle for paclitaxel (Cremophor EL and ethanol diluted in normal saline; n = 8) had reaction latency times similar to naïve control animals (n = 8) both before treatment (baseline) and at 7 days post treatment, 9.2 ± 0.1 s versus 8.8 ± 0.2 s and 9.5 ± 0.3 s versus 8.8 ± 0.4 s (p > 0.05 for both times; Fig. 3). Thus, the vehicle-treated mice were used for comparison with paclitaxel-treated mice.
Unpaired Student's t test showed that administration of paclitaxel produced a significant reduction in the reaction latency times (thermal hyperalgesia) on day 7 after first drug administration from 9.0 ± 0.3 s to 6.7 ± 0.3 s compared to baseline values (p < 0.0003; n = 8; Fig. 3). The reduction in reaction latency times caused by paclitaxel on day 7 was 25% compared to baseline values. Two-way repeated measures ANOVA showed that mice treated with paclitaxel had similar baseline values but lower reaction latency times at day 7 post administration compared to vehicle-treated animals (p > 0.05 and p < 0.001, respectively; n = 8 for both groups; Fig. 3).
In order to evaluate whether minocycline could prevent the paclitaxel-induced thermal hyperalgesia minocycline was coadministered with paclitaxel and compared to control vehicle-only-treated and paclitaxel plus vehicle-treated mice. Two-way repeated measures ANOVA showed that mice that were treated with minocycline alone (n = 8) had reaction latency times similar to vehicle-treated animals (n = 8) both on baseline and on day 7 after first administration of minocycline or vehicle 9.0 ± 0.3 s versus 9.2 ± 0.1 s and 9.2 ± 0.2 s versus 9.5 ± 0.3 s (p > 0.05 for both times; Fig. 3).
Two-way repeated measures ANOVA showed that mice that were treated with paclitaxel plus minocycline (n = 8) had reaction latency times similar to vehicle-only-treated control animals (n = 8) both on baseline and on day 7 after first administration of drugs or vehicle, 9.6 ± 0.2 s versus 9.2 ± 0.1 s and 9.7 ± 0.4 s versus 9.5 ± 0.3 s (p > 0.05 for both times; Fig. 3). Moreover, on day 7 post drug administration mice that were treated with paclitaxel plus minocycline (n = 8) had reaction latency times significantly higher than paclitaxel plus vehicle-treated animals (n = 8), 9.7 ± 0.4 s versus 6.7 ± 0.3 s (p < 0.001; Fig. 3).
Discussion
The findings of this study show that paclitaxel induces hyposensitivity to chemical nociception, whilst at the same time inducing thermal hyperalgesia, and this hyposensitivity to chemical nociception and thermal hyperalgesia can be prevented by coadministration with minocycline.
The paclitaxel treatment regimen and dose used in this study has been reported to produce painful neuropathy in mice, which manifested as thermal hyperalgesia, cold allodynia and mechanical allodynia21,22,23. Our group also previously observed thermal hyperalgesia using the same paclitaxel treatment regimen20,24. Using the same treatment schedule in the current study paclitaxel-induced hyposensitivity to chemical nociception and thermal hyperalgesia. There are few reports on the effect of chemical nociception on paclitaxel-induced neuropathy. Two recent studies on rats reported different observations, in one they observed mechanical allodynia as well as hyperalgesia to chemical nociception induced by formalin after treatment with paclitaxel13, whilst in the other study although they observed mechanical hyperalgesia they did not observe any changes in formalin-induced nociception14.
Our findings of paclitaxel-induced hyposensitivity to chemical nociception are similar to what has been found in other models of peripheral neuropathy25,26. Mice with streptozotocin-induced diabetic peripheral neuropathy were found to have reduced responses to the formalin test25. Rats with chronic-constriction injury of the sciatic nerve were found to be hypersensitive to cold (cold allodynia) but had reduced responses to the formalin test26.
Paclitaxel-induced peripheral neuropathy is associated with degeneration or loss of intraepidermal nerve fibers (IENFs) that has been associated with thermal hyperalgesia and mechanical allodynia15,18,27,28. It is possible that this loss of IENFs could also contribute to hyposensitivity to chemical stimuli. The question whether a drug which protects against the loss of IENFs can protect against the development of hyposensitivity was addressed in the current study. Minocycline has been reported to protect against the development of paclitaxel-induced loss of IENFs and also against the development of thermal hyperalgesia, cold and mechanical allodynia15,16,17,18. Treatment with minocycline prevented the development of paclitaxel-induced hyposensitivity to chemical nociception and thermal hyperalgesia. This result shows that a neuroprotective drug can inhibit the development of both paclitaxel-induced hyposensitivity to chemical nociception thermal hyperalgesia and also suggest that this hyposensitivity could be due to loss of IENFs.
In conclusion the results from this study show that paclitaxel induces symptoms of both hypoesthesia and hyperesthesia i.e. hyposensitivity to chemical nociception and hypersensitivity to thermal nociception. The paclitaxel-induced hyposensitivity to chemical nociception and thermal hyperalgesia can be prevented by coadministration with minocycline, suggesting it can be used as a model for investigating neuroprotection and prevention of chemotherapy-induced neuropathy.
Methods
Animals
Female BALB/c mice (8 to 12 weeks old; 20–30 g; n = 118) used in this study were kept in temperature controlled (24 ± 1°C) rooms with food and water ad libitum. The animals were supplied by the Animal Resources Center at the Health Sciences Center, Kuwait University, Kuwait. All experiments were performed during the same period of the day (8:00 AM to 4:00 PM) to exclude diurnal variations in pharmacological effects. The animals were handled in compliance with European Communities Council Directive 86/609 for the care of laboratory animals and ethical guidelines for research in experimental pain with conscious animals29. All methods were carried out in accordance with the approved guidelines and regulations of the Health Sciences Center Ethical Committee for the use of Laboratory Animals in Teaching and in Research, Kuwait University.
Drugs and drug administration
Paclitaxel (Tocris, Bristol, UK) was dissolved in a solution made up of 50% Cremophor EL and 50% absolute ethanol to a concentration of 6 mg/ml and stored at −20°C, for a maximum of 14 days, and then diluted in normal saline (NaCl 0.9%), to a final concentration of 0.2 mg/ml just before administration. The vehicle for paclitaxel was diluted at the time of injection with normal saline in the same proportion as the paclitaxel solution. Paclitaxel 2 mg/kg or its vehicle were administered to mice intraperitoneally (i.p.), in a volume of 10 μl/g body mass, once per day for 5 consecutive days. Other groups and ours have observed that this treatment regimen produce thermal hyperalgesia in mice20,21,22,23,24.
Minocycline (Sigma-Aldrich, St Louis, MO, USA) was dissolved in phosphate buffered saline and administered to mice i.p. in a volume of in a volume of 10 μl/g body mass. Minocycline (50 mg/kg) was administered alone or coadministered with paclitaxel daily for 5 days. This dose of minocycline has been shown to prevent the development of paclitaxel-induced neuropathic pain18.
Chemical nociception
An automated formalin test (automated antinociception analyzer, ANA) developed by Yaksh et al.30, was used to evaluate chemical nociception on day 7 after first injection of drugs (paclitaxel or minocycline). Small metal bands were placed around the base of mice left hind paw; these encompass about 270° of the circumference, leaving a large window on the dorsal surface. Bands were fixed in place with cyanoacrylate glue. Mice were placed in a cylindrical test chamber to become acclimated for 1 hour and formalin (5%; 20 μl) was injected subcutaneously (s.c.) into the paw dorsum. After formalin injection, mice were returned to the chamber and flinches counted for 40 min with an automated device. Briefly, the test chamber was placed above a loop antenna that generates a low-wattage electromagnetic wave. Movement of the metal band on the mouse's paw (during flinching) alters the electromagnetic field. The resulting signal was fed to a computer that uses the response amplitude and duration to separate flinches from normal locomotor activity. The phases of the formalin test were defined as early phase from 1–9 minutes and late phase from 10–40 minutes.
Thermal nociception
Reaction latencies to hot plate test were measured before (baseline latency) and on day 7 after first injection of paclitaxel. Briefly, mice were individually placed on a hot plate (Panlab SL, Barcelona, Spain) with the temperature adjusted to 55 ± 1°C. The time to the first sign of nociception, paw licking, flinching or jump response to avoid the heat was recorded and the animal immediately removed from the hot plate. A cut-off period of 20 seconds was maintained to avoid damage to the paws.
Data and statistical analyses
The software GraphPad Prism version 5.00 (GraphPad Software Inc., USA) was used for plotting graphs, data and statistical analyses. Statistical analyses were performed using unpaired Student's t test or two-way repeated measures analysis of variance (ANOVA) followed by Bonferroni posttests. The differences were considered significant at p < 0.05. The results in the text and figures are expressed as the means ± S.E.M.
Author Contributions
W.M. designed and conducted the experiments and wrote the manuscript.
Acknowledgments
This study was supported by grant PT01/09 from Kuwait University Research Sector. I am grateful to Dr Subramanian S Parvathy for her technical assistance and to the staff from the Animal Resources Centre, HSC, Kuwait University for their support.
References
- Dougherty P. M., Cata J. P., Cordella J. V., Burton A. & Weng H. R. Taxol-induced sensory disturbance is characterized by preferential impairment of myelinated fiber function in cancer patients. Pain 109, 132–142 (2004). [DOI] [PubMed] [Google Scholar]
- Argyriou A. A., Koltzenburg M., Polychronopoulos P., Papapetropoulos S. & Kalofonos H. P. Peripheral nerve damage associated with administration of taxanes in patients with cancer. Crit Rev Oncol Hematol 66, 218–228 (2008). [DOI] [PubMed] [Google Scholar]
- Kuroi K. & Shimozuma K. Neurotoxicity of taxanes: symptoms and quality of life assessment. Breast Cancer 11, 92–99 (2004). [DOI] [PubMed] [Google Scholar]
- Wolf S., Barton D., Kottschade L., Grothey A. & Loprinzi C. Chemotherapy-induced peripheral neuropathy: prevention and treatment strategies. Eur J Cancer 44, 1507–1515 (2008). [DOI] [PubMed] [Google Scholar]
- Cavaletti G. et al. Peripheral neurotoxicity of taxol in patients previously treated with cisplatin. Cancer 75, 1141–1150 (1995). [DOI] [PubMed] [Google Scholar]
- Polomano R. C. & Bennett G. J. Chemotherapy-evoked painful peripheral neuropathy. Pain Med 2, 8–14 (2001). [DOI] [PubMed] [Google Scholar]
- Ruiz-Medina J., Baulies A., Bura S. A. & Valverde O. Paclitaxel-induced neuropathic pain is age dependent and devolves on glial response. Eur J Pain 17, 75–85 (2013). [DOI] [PubMed] [Google Scholar]
- Smith S. B., Crager S. E. & Mogil J. S. Paclitaxel-induced neuropathic hypersensitivity in mice: responses in 10 inbred mouse strains. Life Sci 74, 2593–2604 (2004). [DOI] [PubMed] [Google Scholar]
- Yamashita Y. et al. Comparison of peripheral neuropathy induced by standard and nanoparticle albumin-bound paclitaxel in rats. J Pharmacol Sci 117, 116–120 (2011). [DOI] [PubMed] [Google Scholar]
- Authier N., Gillet J. P., Fialip J., Eschalier A. & Coudore F. Description of a short-term Taxol-induced nociceptive neuropathy in rats. Brain Res 887, 239–249 (2000). [DOI] [PubMed] [Google Scholar]
- Campana W. M. et al. Prosaptide prevents paclitaxel neurotoxicity. Neurotoxicology 19, 237–244 (1998). [PubMed] [Google Scholar]
- Cavaletti G. et al. Effect on the peripheral nervous system of the short-term intravenous administration of paclitaxel in the rat. Neurotoxicology 18, 137–145 (1997). [PubMed] [Google Scholar]
- Lee-Kubli C. A. & Calcutt N. A. Altered rate-dependent depression of the spinal H-reflex as an indicator of spinal disinhibition in models of neuropathic pain. Pain 155, 250–260 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Noda K., Akita H., Ogata M. & Saji M. Paclitaxel-induced hyperalgesia modulates negative affective component of pain and NR1 receptor expression in the frontal cortex in rats. Neurosci Res 80, 32–37 (2014). [DOI] [PubMed] [Google Scholar]
- Boyette-Davis J., Xin W., Zhang H. & Dougherty P. M. Intraepidermal nerve fiber loss corresponds to the development of taxol-induced hyperalgesia and can be prevented by treatment with minocycline. Pain 152, 308–313 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burgos E. et al. Cannabinoid agonist WIN 55,212-2 prevents the development of paclitaxel-induced peripheral neuropathy in rats. Possible involvement of spinal glial cells. Eur J Pharmacol. 682, 62–72 (2012). [DOI] [PubMed] [Google Scholar]
- Cata J. P., Weng H. R. & Dougherty P. M. The effects of thalidomide and minocycline on taxol-induced hyperalgesia in rats. Brain Res 1229, 100–110 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu C. C. et al. Prevention of paclitaxel-induced allodynia by minocycline: Effect on loss of peripheral nerve fibers and infiltration of macrophages in rats. Mol Pain 6, 76 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pevida M., Lastra A., Hidalgo A., Baamonde A. & Menendez L. Spinal CCL2 and microglial activation are involved in paclitaxel-evoked cold hyperalgesia. Brain Res Bull 95, 21–27 (2013). [DOI] [PubMed] [Google Scholar]
- Parvathy S. S. & Masocha W. Matrix metalloproteinase inhibitor COL-3 prevents the development of paclitaxel-induced hyperalgesia in mice. Med Princ Pract 22, 35–41 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Katsuyama S. et al. Intraplantar injection of linalool reduces paclitaxel-induced acute pain in mice. Biomed Res 33, 175–181 (2012). [DOI] [PubMed] [Google Scholar]
- Nieto F. R. et al. Role of sigma-1 receptors in paclitaxel-induced neuropathic pain in mice. J Pain 13, 1107–1121 (2012). [DOI] [PubMed] [Google Scholar]
- Nieto F. R. et al. Tetrodotoxin inhibits the development and expression of neuropathic pain induced by paclitaxel in mice. Pain 137, 520–531 (2008). [DOI] [PubMed] [Google Scholar]
- Thangamani D., Edafiogho I. O. & Masocha W. The anticonvulsant enaminone E139 attenuates paclitaxel-induced neuropathic pain in rodents. ScientificWorldJournal 2013, 240508 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson M. S., Ryals J. M. & Wright D. E. Diabetes-induced chemogenic hypoalgesia is paralleled by attenuated stimulus-induced fos expression in the spinal cord of diabetic mice. J Pain 8, 637–649 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vissers K., Adriaensen H., De Coster R., De Deyne C. & Meert T. F. A chronic-constriction injury of the sciatic nerve reduces bilaterally the responsiveness to formalin in rats: a behavioral and hormonal evaluation. Anesth Analg 97, 520–525, table of contents (2003). [DOI] [PubMed] [Google Scholar]
- Siau C., Xiao W. & Bennett G. J. Paclitaxel- and vincristine-evoked painful peripheral neuropathies: loss of epidermal innervation and activation of Langerhans cells. Exp Neurol 201, 507–514 (2006). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jin H. W., Flatters S. J., Xiao W. H., Mulhern H. L. & Bennett G. J. Prevention of paclitaxel-evoked painful peripheral neuropathy by acetyl-L-carnitine: effects on axonal mitochondria, sensory nerve fiber terminal arbors, and cutaneous Langerhans cells. Exp Neurol 210, 229–237 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zimmermann M. Ethical guidelines for investigations of experimental pain in conscious animals. Pain 16, 109–110 (1983). [DOI] [PubMed] [Google Scholar]
- Yaksh T. L. et al. An automated flinch detecting system for use in the formalin nociceptive bioassay. J Appl Physiol (1985) 90, 2386–2402 (2001). [DOI] [PubMed] [Google Scholar]