Abstract
Objective
The purpose was to describe the clinicopathological characteristics of pancreatic neuroendocrine tumors (pNETs) and evaluate prognosis-related factors in potentially resectable pNETs.
Methods
The clinical data of 104 patients with pNETs who underwent surgery were retrospectively analyzed.
Results
The mean (SD) age was 49.8 (14.6) years. The percentages of TNM stages I, II, III, and IV tumors were 25.0%, 44.2%, 22.1%, and 8.7%, respectively. Twenty-seven cases were functional. Nonfunctional pNETs were more common in patients with large tumors, advanced age, higher mitotic count, neural invasion, extrapancreatic organ invasion, liver metastases, and advanced staging (P < 0.05). The 5-year overall survival rate was 93%. The relapse rate was 28.6% (28/98), and the mean (SD) relapse time was 38.7 (31.7) months. Reduced survival rate was associated with older patients (>60 years) (P = 0.026), patients with a higher Ki-67 index (>2%) (P = 0.024), regional lymph node metastases (P = 0.033), liver metastases (P = 0.015), neural invasion (P = 0.017), necrosis (P = 0.042), and major vascular invasion (P = 0.023). Age of more than 60 years (P = 0.047; hazard ratio [HR], 5.2), major vascular invasion (P = 0.030; HR, 5.8), and a Ki-67 index greater than 2% (P = 0.008; HR, 10.3) were independent predictors.
Conclusions
Nonfunctional pNETs were more common with aggressive clinical presentation. Age of more than 60 years, major vascular invasion, and a Ki-67 index greater than 2% were independent predictive factors. Patients who underwent a potentially curative resection seemed to achieve long-term survival.
Key Words: neuroendocrine tumors, pancreas, survival, prognosis
Neuroendocrine tumors (NETs), once known as carcinoid tumors, consist of a heterogeneous group of neoplasms that present a significant clinical challenge. These tumors secrete peptides and neuroamines that cause specific clinical syndromes. Gastroenteropancreatic NETs originate from the pancreas or gastrointestinal tract, with the common location in the pancreas (20.5%–38%).1,2 Pancreatic NETs (pNETs), derived from precursors in the ductal epithelium, also initially thought to be derived from the islets of Langerhans, are rare. They constitute approximately 1% to 2% of all pancreatic neoplasms,3–6 leading to an overall incidence of fewer than 2 per 100,000 cases.3,4,6–14 Pancreatic NETs tend to be slow growing, although aggressive variants exist and often cause hormone hypersecretion and associated symptoms.
The Surveillance, Epidemiology, and End Results program and National Cancer Registry of Spain recently revealed that the incidence and prevalence of NETs has increased substantially during the last decade, which may partially reflect the increased number of diagnoses of benign and incidentally identified lesions because of clinical awareness and improved diagnostic techniques, such as advanced endoscopic and radiological imaging.1,15,16 There is literature available on the factors predictive of outcome for pNETs. However, because of the rarity of pNETs, there are few data available on epidemiology and survival of pNETs in China or in Asia.10,14 As a major referral center for pancreatic tumors in China, we have resected 104 cases of pNETs in the past 10 years. In this study, we report our data on the clinicopathological features, survival, and prognosis-related factors of pNETs in a single Chinese center.
MATERIAL AND METHODS
A total of 104 Chinese patients with pNETs who underwent surgery were included. The institutional review board approval was obtained for this retrospective analysis. In this study, the diagnosis was based on pathological morphology and immunohistochemical assessment through surgical specimen and intraoperative biopsy by experienced pathologists.17 The following pathological features were recorded as being present or absent in the tumors: tumor necrosis, lymph node invasion, neural invasion, extrapancreatic organ invasion, and major vascular invasion. Subsequently, patients were also classified according to European Neuroendocrine Tumor Society TNM classifications.18 In particular, the Ki-67 proliferative index was expressed as the percentage of Ki-67–positive cells in 2000 tumor cells within areas of the highest immunostaining using the MIB1 antibody (Dako, Denmark).17 Mitotic count was based on counting 50 high-power fields and in the area of highest mitotic activity and reported as the number of mitoses per 10 high-power fields. Immunohistochemistry was performed on formalin-fixed, paraffin-embedded tissues. The tissue blocks were sectioned at 4 μm. Slides were then stained using the Bond-Max Leica autostainer (Leica Biosystems, United Kingdom). Antibody detection was performed using the biotin-free Bond Polymer Refined Detection System (DS9800; Leica Microsystems, United Kingdom) according to the manufacturer’s protocol. The antibodies used were as follows: CgA (Dako, Denmark), Syn (Dako, Denmark), NSE (Dako, Denmark), and SSTR2A (Abcam, United Kingdom). Tumors with more than 5% positive cells were considered positive.
Tumors were classified as functional pNETs according to clinical signs and symptoms of hormonal excess, along with the presence of increased levels of corresponding peptides and hormones from the serum. Tumors were classified as nonfunctional if they were not associated with distinct clinical manifestations or hormone alterations.
Follow-up information was collected from clinical records and the tumor registry at Zhong Shan Hospital, Fudan University. The duration of overall survival (OS) was calculated from the date of operation until tumor-specific death or the patient’s last follow-up. The relapse time was computed from the date of remission to recurrence.
The statistical analyses were performed using the SPSS statistical package version 16.0. Pearson χ2 test, Fisher exact test, Mann-Whitney U test, Pearson correlation test, and Spearman correlation test were used to evaluate the association between variables when appropriate. Survival was estimated according to the Kaplan-Meier product limit method and life tables method. Survival curves were compared by using the log-rank test. The analysis of risk factors was performed by univariate and multivariate analyses by the Cox proportional hazards method. Multivariate analyses using the Cox proportional hazards model were carried out to identify the factors independently associated with prognosis. Statistical significance was defined as P < 0.05.
RESULTS
General Data
Among 104 patients with pNETs from January 1999 to December 2011, 52 patients (50.0%) were men and 52 (50.0%) were women. The median age at diagnosis was 52 years (range, 9–79 years), and the mean (SD) age was 49.8 (14.6) years.
Clinicopathological Characteristics
The median size of the tumor (in the case of multifocality, the largest lesion was recorded) was 3.5 cm (mean [SD] size, 4.2 [3.1] cm; range, 0.8–18 cm). In 8 cases (7.7%), the tumor was multifocal. The majority of pNETs were located in the distal part of the pancreas (57.7%), followed by the head (36.5%). At diagnosis, 72 patients (69.2%) had a localized disease, 28 patients (26.9%) presented with regional spread including lymph node metastases and/or extrapancreatic organ invasions, 9 patients (8.7%) presented with synchronous liver metastases, and 13 patients (12.5%) presented with metachronous liver metastases. Considering the 22 cases with liver metastases, a single lesion was present in 1 case (4.5%), whereas multiple lesions were detected in the remaining 21 cases (95.5%). No patient presented with distant metastases at other sites without liver involvement. Regional lymph node metastases were detected in 21 patients (42.9%). The median number of lymph nodes detected in pathology reports was 3.0 (mean [SD] number, 5.4 [4.4]; range, 1.0–16.0), the median number of metastatic lymph nodes was 0 (mean [SD] number, 0.74 [1.15]; range, 0-6.0), and the positive rate was 22.0%. The percentages of G1 and G2 tumors were 56.7% and 43.4%, respectively. Regarding the ENETS TNM staging, the percentages of stage I, stage II, stage III, and stage IV were 25.0%, 44.2%, 22.1%, and 8.7%, respectively. We found that the World Health Organization (WHO) histological grading was strongly linked with ENETS TNM staging (Spearman ρ = 0.452, P < 0.001).
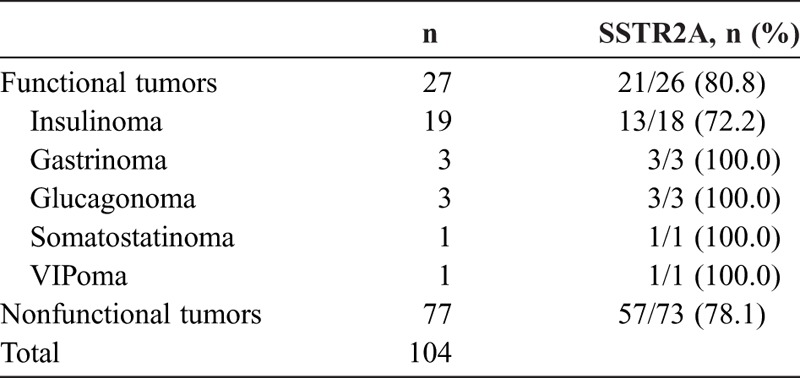
Twenty-seven patients (26.0%) had functional pNETs. The most frequent entity of functional pNETs was insulinoma (18.3%), followed by gastrinoma, glucagonoma, somatostatinoma, and VIPoma, accounting for 2.9%, 2.9%, 1.0%, and 1.0% of the whole group, respectively. The positive rates of SSTR2A for functional and nonfunctional pNETs were 80.8% (21/26) and 78.1% (57/73) (P > 0.05), respectively (Table 1). Concurrently, 4 patients with pNETs had multiple endocrine neoplasia type 1 and 2 patients had von Hippel-Lindau disease in association with familial syndromes. The highest percentage was found in gastrinoma (16.7%).
TABLE 1.
The Positive Rate of SSTR2A in Functional and Nonfunctional pNETs
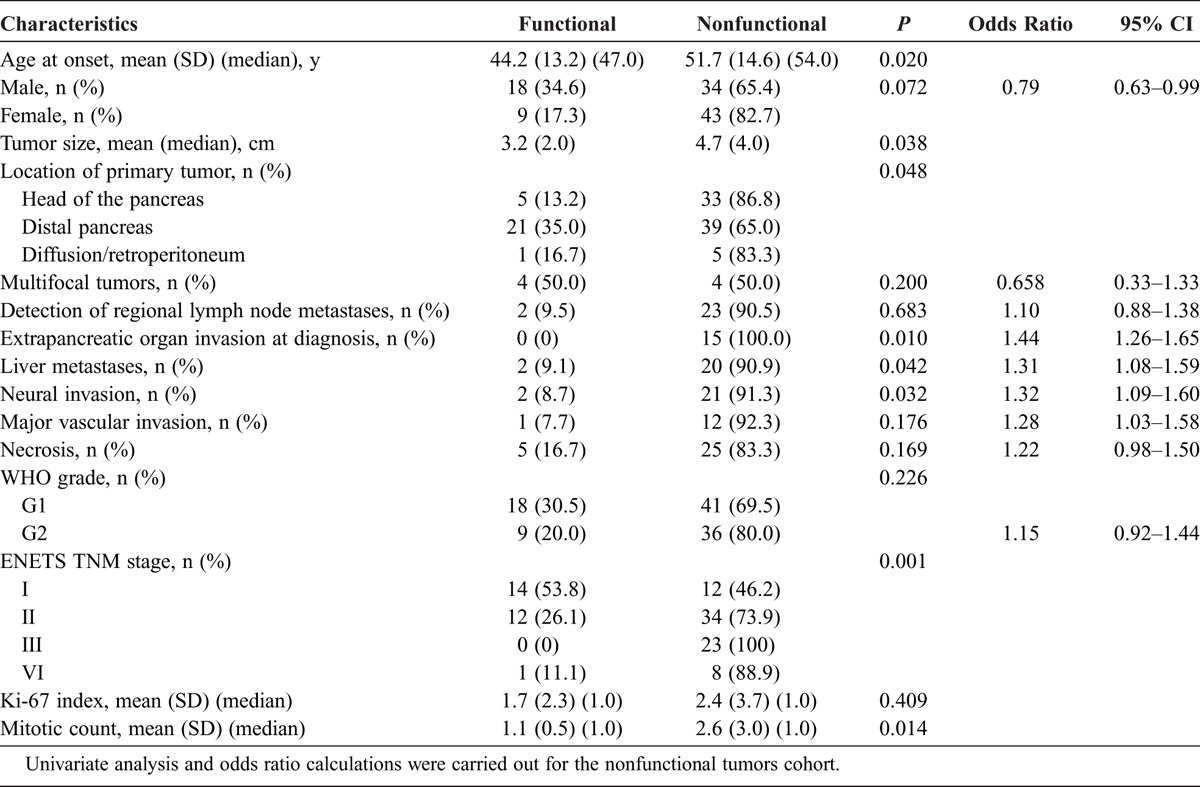
A comparison of clinicopathological characteristics between patients with functional or nonfunctional pNETs is summarized in Table 2. Among the nonfunctional group, there was a higher proportion of elder patients, larger tumor, and higher mitotic count (P < 0.05). Interestingly, functional pNETs were significantly more likely to be located in the distal part of the pancreas than nonfunctional pNETs (P < 0.05). On the other hand, aggressive clinical behaviors, such as neural invasion, extrapancreatic organ invasion at diagnosis, and liver metastases, were significantly more frequent in patients with nonfunctional pNETs than in patients with functional pNETs (P < 0.05). A strong correlation was observed between the ENETS TNM stage and clinical presentation, which means that functional pNETs were more likely to be at an early stage, as we also found that functional pNETs accounted for 53.8% (14/26) in stage I, 24.5% (12/49) in stage II, 0% (0/27) in stage III, and 10% (1/10) in stage IV (Pearson χ2 test, P = 0.001; Spearman correlation test, P = 0.001). Finally, patients with a higher Ki-67 index, regional lymph node metastases, major vascular invasion, and necrosis were associated with an increased frequency in nonfunctional pNETs, although no statistically significant difference was found (P > 0.05).
TABLE 2.
Clinicopathological Characteristics of 104 Patients With Functional and Nonfunctional pNETs
Therapeutic Interventions
Among 104 patients, 102 patients (98.1%) received a potentially curative resection of the primary tumor. Two patients (1.9%) received total pancreatectomy, 38 patients (36.5%) received distal pancreatectomy with/without splenectomy, 28 patients (26.9%) received pancreatoduodenectomy, 29 patients (27.9%) received local or partial pancreatectomy, and 5 patients (4.9%) received other radical surgery such as multivisceral resection. A biopsy, in combination with bypass surgery, was performed on 2 patients (1.9%).
In 22 cases with liver metastases, the treatment was divided into liver-directed strategies (n = 16) and nonliver-directed strategies (n = 6). Liver-directed strategies included hepatic resection (22.7%), transarterial chemoembolization (54.5%), and radiofrequency ablation (13.6%). The following chemotherapeutic agents were selectively used for patients who had an arterial infusion chemotherapy: 5-fluorouracil, mitomycin C, doxorubicin, oxaliplatin, and gemcitabine. Nonliver-directed strategy referred to octreotide (Sandostatin LAR) injection (27.3%). The median interval between the remission and metastatic relapse was 29.4 months (mean [SD] time, 34.8 [29.5] months; range, 6.0–58.0 months).
Survival Analysis and Prognostic Factors
The duration of follow-up was calculated from the date of surgery to the date of recurrence, death, or last follow-up. At the last follow-up, 9 patients (8.7%) died of tumor recurrence or recurrence-related complications. An up-to-date follow-up (known death or contact within 6 months of data cutoff point) was available in 89.3% of patients. The mean follow-up time was 51.6 months. The median survival time for all 104 patients was 144.0 months (mean survival time, 133.2 months; 95% confidence interval [CI], 121.8–144.6). The 1-, 2-, and 5-year accumulative OS rates were 98%, 96%, and 93%, respectively. Twenty-eight patients had a tumor relapse after remission; the relapse rate was 28.6% (28/98). Meanwhile, the mean (SD) relapse time was 38.7 (31.7) months and the estimated median time of relapse was 37.5 months.
Numerous studies have validated the prognostic significance of the WHO 2010 classification and ENETS TNM staging. For all 104 patients, the difference in survival between the patients with G1 and G2 was statistically significant (P < 0.001); the 5-year OS rates for G1 and G2 were 100% and 80%. The ENETS TNM staging was found to be a predictor of survival (P = 0.040); the 5-year OS rates for stage I, stage II, stage III, and stage IV were 100%, 97%, 73%, and 60%.
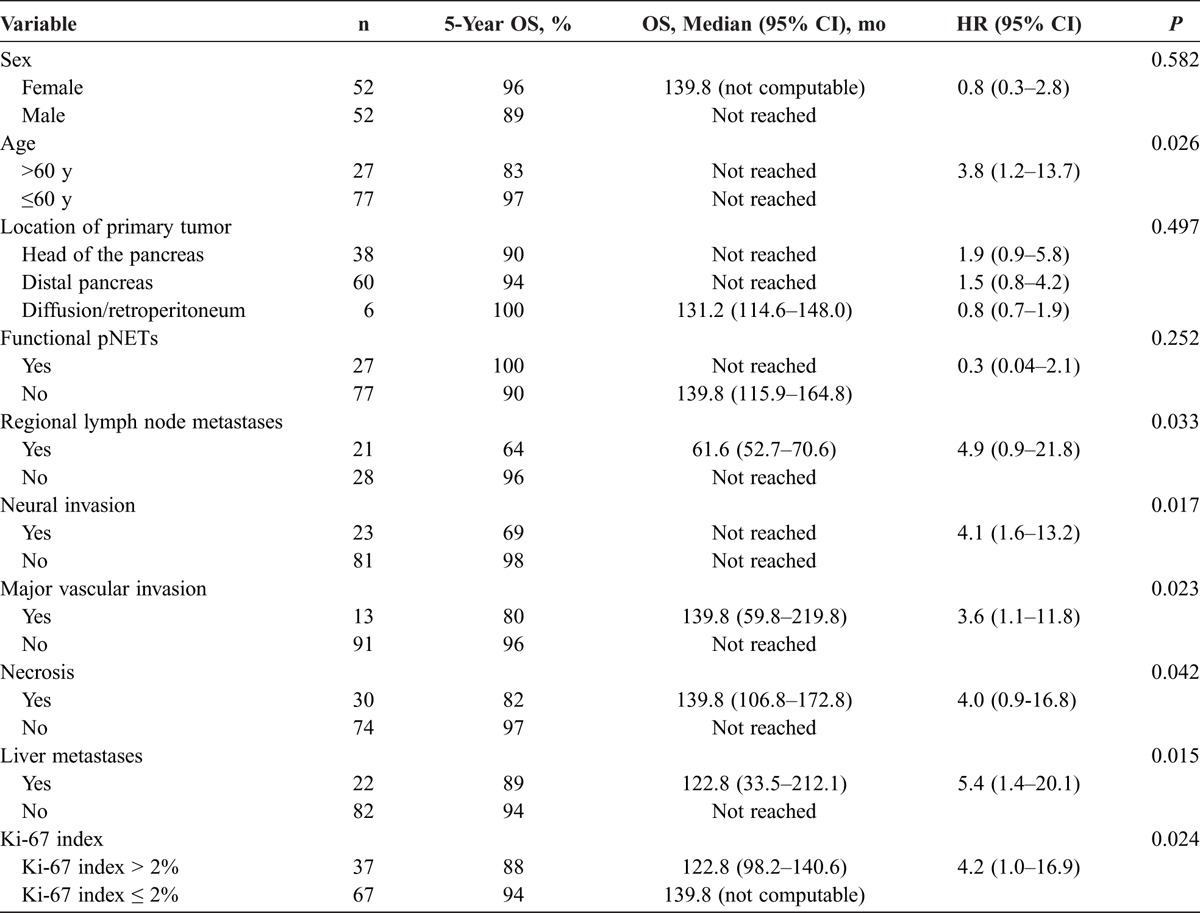
In the univariate analysis, survival rates in patients with regional lymph node metastases (P = 0.033), liver metastases (P = 0.015), neural invasion (P = 0.017), major vascular invasion (P = 0.023), and necrosis (P = 0.042) were significantly lower than those in patients without. In our study, Ki-67 was an efficient predictor when the standard value of 2% proposed by the G grading system was used as cutoff. A high Ki-67 index (Ki-67 index >2%) has been proven to portend a worse prognosis than a low Ki-67 index in survival duration (P = 0.024). The age at diagnosis was also a prognostic factor for survival. We used 60 years as the cutoff between old and young patients; elderly patients (>60 years) had a poorer prognosis than their younger counterparts (P = 0.026) (Table 3). Moreover, we performed a multivariate survival analysis using the Cox proportional hazards model. It demonstrated that age (>60 years) (P = 0.047; HR, 5.2; 95% CI, 1.0–26.7), major vascular invasion (P = 0.030; HR, 5.8; 95% CI, 1.5–28.3), and a Ki-67 index greater than 2% (P = 0.008; HR, 10.3; 95% CI, 2.0–60.5) were independent prognostic factors for survival. Finally, patients who underwent liver-directed strategies for the treatment of liver metastases showed a tendency to prolong OS compared with patients who had nonliver-directed strategies (P = 0.090; median OS, 122.8 vs 61.6 months; 5-year OS, 93% vs 58%) and patients who underwent liver-directed strategies might have an increased metastatic relapse time compared with their other counterparts (P = 0.128; mean [SD], 45.3 [31.8] vs 23.1 [23.4] months).
TABLE 3.
Risk Factors for OS at the Univariate Analysis (n = 104 Patients)
DISCUSSION
During the past decades, the literature points to a trend toward increasing incidental diagnoses of pNETs and shows functional tumors presenting as a distinct minority among pNETs.19 The following changes were evident in our study: most pNETs were nonfunctional (75.9%) and that diagnosis was incidentally made in most of them. The high rate of multifocality (7.7%) indicated the importance of a careful intraoperative search for multifocality by wide exposure and by systematic use of intraoperative ultrasound.
In the present study, no significant differences in positive rates of SSTR2A, functional or nonfunctional, were seen among different subgroups (P > 0.05); these results sustain the rationale that SSTR2A is considered of particular therapeutic relevance and the use of somatostatin scintigraphy to follow disease20 because of its abundance in pNETs regardless of functional status. Notably, the tumor size was greater and patients were more elderly at diagnosis in nonfunctional pNETs compared with those in the functional pNETs group; nonfunctional pNETs were more prone to have an extrapancreatic organ invasion and synchronous or metachronous distant metastases and were more often in the advanced WHO grading and ENETS TNM staging. Other studies showed the same findings between functional pNETs and nonfunctional pNETs.14,21 The reason why nonfunctional pNETs are often diagnosed late in the course of the disease may be that nonfunctional pNETs usually become clinically apparent when they reach a size that causes compression or invasion of extrapancreatic organs or when they metastasize,19 whereas functional tumors are often discovered earlier because of a hormonal syndrome. Admittedly, it is possible that the aggressive behaviors of nonfunctional pNETs might be associated with their size and stage, compared with the predominantly benign insulinomas in functional pNETs. When we removed insulinomas from the functional cohort, we found that there was no significant association between functional status and clinical behaviors (P > 0.05), which means that a small number of relatively malignant gastrinomas and rare functioning tumors might contribute to these results.
Previous WHO classification systems discriminated between low- and high-grade malignant NETs but did not differentiate further prognosis of low to intermediate malignant pNETs. We found that regional lymph node metastases, liver metastases, Ki-67 index (>2%), age (>60 years), neural invasion, necrosis, and major vascular invasion were predictors for bad outcomes in this relatively large and homogeneous series of resectable pNETs. These predictive factors should be considered for risk stratification, on which the therapeutic strategy for postoperative pNETs depends. Elderly patients, major vascular invasion, and a higher Ki-67 index remained the independent predictors, which underscores the need for improved systemic therapies in elderly patients and those at advanced stage. Panzuto et al22 suggested that a possible improvement in the prognostic capability of the G grading system may be achieved by increasing the Ki-67 cutoff value between G1 and G2 from 2% to 5%, but our data verified that a cutoff of 2% is effective in predicting the prognosis of resectable pNETs and patients with a Ki-67 index greater than 5% seemed to have a reduced survival compared with patients with a Ki-67 index 5% or lower (P = 0.059; HR, 5.17; 95% CI, 1.1–26.8). The clinical behaviors and prognosis of patients with pNETs showing discordance between WHO grading and histology differentiation are still unclear. The North American Neuroendocrine Tumor Society recommended histology differentiation in pathology, which will be helpful for solving these problems in the future.20 Current studies showed that patients with functional tumors have a more favorable prognosis.1,15,23 Similarly, there was a tendency for nonfunctional pNETs to have a negative effect on prognosis in our study, although no statistical significance was reached, which may partly be explained by the favorable prognosis of the dominating insulinomas.
It is appropriate on the basis of our data to assume that a potentially radical resection correlates with a relatively long-term cure, with evidence that a 5-year OS of 93% was observed. However, the role of surgical debulking in asymptomatic patients with metastatic liver predominant NETs and in patients where an R0/R1 resection cannot be achieved remain therapeutic dilemmas.19,20 In metastatic nonfunctional pNETs, an advantage in survival after primary tumor resection has not been clearly shown.19,20 In our study, we recommended radical surgery with lymph node dissection to achieve primary R0/R1 resection for local pNETs. As for metastatic nonfunctional pNETs, a potentially curative resection should be considered when a resection of complete or more than 90% of metastatic lesions along with the primary site could be achieved. Furthermore, a palliative surgery could be performed in selected functional pNETs because it may reduce tumor burden to alleviate hormonal syndrome and delay the subsequent need for therapy. In addition, bypass surgery, in combination with biopsy, should also be performed on specific patients to minimize tumor-related complications and identify histological features. Besides, prophylactic surgery for multiple endocrine neoplasia type 1 and von Hippel-Lindau disease–associated pNETs was useful to remove multifocal lesions before malignancy develops and these patients all had good long-term survival (5-year OS, 100%). A recent report found that the 5-year OS rates did not vary significantly by ENETS stage in nonmetastatic, surgically resected pNETs. They were estimated to be 100% for patients with stage I, 90% for stage II, and 88% for stage III.24 In general, resection is recommended for a local regional disease and should still be considered for patients with an advanced disease.20
A surgical approach aiming to remove the lesions before malignancy develops for small nonfunctional tumors and whether surveillance is a reasonable alternative to surgery in patients with 2 cm or smaller, asymptomatic, pathologically benign pNETs, especially if surgical risks are high, are still being debated upon.19,20 Currently, most patients presenting with an early-stage pNET are referred for surgical resection, even if they are asymptomatic.25 Most neoplasms 2 cm or smaller are likely benign or intermediate-risk lesions, and 6% of nonfunctional pNETs 2 cm or smaller are malignant when incidentally discovered.26 Although patients who underwent a radical operation seemed to achieve an acceptable long-term prognostic benefit, the relapse rate was 28.6% (28/98) and the mean (SD) relapse time was 38.7 (31.7) months in our series, which means that a continued follow-up directed at the detection of metachronous distant metastases and primary recurrence should be indicated after surgery. A recent article reported that the recurrence rates of nonmetastatic, surgically resected pNETs peaked at approximately 2 years after surgery.24 We should be aware of the long-term risk for relapse on account of the indolent nature of most pNETs. The postoperative surveillance recommendations should be formatted as concretely described in The North American Neuroendocrine Tumor Society guidelines.20
Today, the selection of therapies for advanced pNETs, especially nonfunctional pNETs, continues to be debated.19,20 The management of pNETs with liver metastases is too difficult to define exactly using a simple algorithmic approach. Although liver metastases was a bad prognostic factor, our data reflect that a long-term survival can still be achieved with active therapeutic interventions, the 5-year OS being up to 89%. Some studies27–29 have found that liver-directed therapies that include repeat surgery or radioembolization for hepatic metastasis NETs prolong survival after progression after initial surgery. Our similar results show that liver-directed strategies for pNETs with liver metastases seem to be associated with a prolonged OS and metastatic relapse time and patients with nonfunctional hepatic metastasis derive the greatest survival benefit. We think that liver-directed strategies may significantly change stage-specific treatment recommendations with appropriate combinations; however, the roles of transarterial chemoembolization and radiofrequency ablation remain controversial.20
There was a notably high proportion of early stage in our study. The inhomogeneous distribution was mainly due to referral bias, which means that all patients were treated as resectable pNETs by surgeons in a single center. In addition, the impact of a surgical procedure on pNETs with G3 and advanced stage raises the need for further research.
Footnotes
This study was supported by the National Natural Science Foundation of China, grant ID 81071740.
Xu Han and Xuefeng Xu contributed equally to this work.
The authors declare no conflict of interest.
REFERENCES
- 1. Garcia-Carbonero R, Capdevila J, Crespo-Herrero G, et al. Incidence, patterns of care and prognostic factors for outcome of gastroenteropancreatic neuroendocrine tumors (GEP-NETs): results from the National Cancer Registry of Spain (RGETNE). Ann Oncol. 2010; 21: 1794– 1803 [DOI] [PubMed] [Google Scholar]
- 2. Lepage C, Bouvier AM, Phelip JM, et al. Incidence and management of malignant digestive endocrine tumours in a well defined French population. Gut. 2004; 53: 549– 553 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Oberg K, Eriksson B. Endocrine tumours of the pancreas. Best Pract Res Clin Gastroenterol. 2005; 19: 753– 781 [DOI] [PubMed] [Google Scholar]
- 4. Plockinger U, Rindi G, Arnold R, et al. Guidelines for the diagnosis and treatment of neuroendocrine gastrointestinal tumours. A consensus statement on behalf of the European Neuroendocrine Tumour Society (ENETS). Neuroendocrinology. 2004; 80: 394– 424 [DOI] [PubMed] [Google Scholar]
- 5. Vortmeyer AO, Huang S, Lubensky I, et al. Non-islet origin of pancreatic islet cell tumors. J Clin Endocrinol Metab. 2004; 89: 1934– 1938 [DOI] [PubMed] [Google Scholar]
- 6. Moldow RE, Connelly RR. Epidemiology of pancreatic cancer in Connecticut. Gastroenterology. 1968; 55: 677– 686 [PubMed] [Google Scholar]
- 7. Oberg K. Pancreatic endocrine tumors. Semin Oncol. 2010; 37: 594– 618 [DOI] [PubMed] [Google Scholar]
- 8. Halfdanarson TR, Rubin J, Farnell MB, et al. Pancreatic endocrine neoplasms: epidemiology and prognosis of pancreatic endocrine tumors. Endocr Relat Cancer. 2008; 15: 409– 427 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Grimelius L, Hultquist GT, Stenkvist B. Cytological differentiation of asymptomatic pancreatic islet cell tumours in autopsy material. Virchows Arch A Pathol Anat Histol. 1975; 365: 275– 288 [DOI] [PubMed] [Google Scholar]
- 10. Ito T, Tanaka M, Sasano H, et al. Preliminary results of a Japanese nationwide survey of neuroendocrine gastrointestinal tumors. J Gastroenterol. 2007; 42: 497– 500 [DOI] [PubMed] [Google Scholar]
- 11. Modlin IM, Lye KD, Kidd M. A 5-decade analysis of 13,715 carcinoid tumors. Cancer. 2003; 97: 934– 959 [DOI] [PubMed] [Google Scholar]
- 12. Carriaga MT, Henson DE. Liver, gallbladder, extrahepatic bile ducts, and pancreas. Cancer. 1995; 75: 171– 190 [DOI] [PubMed] [Google Scholar]
- 13. Metz DC, Jensen RT. Gastrointestinal neuroendocrine tumors: pancreatic endocrine tumors. Gastroenterology. 2008; 135: 1469– 1492 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Ito T, Sasano H, Tanaka M, et al. Epidemiological study of gastroenteropancreatic neuroendocrine tumors in Japan. J Gastroenterol. 2010; 45: 234– 243 [DOI] [PubMed] [Google Scholar]
- 15. Yao JC, Hassan M, Phan A, et al. One hundred years after “carcinoid”: epidemiology of and prognostic factors for neuroendocrine tumors in 35,825 cases in the United States. J Clin Oncol. 2008; 26: 3063– 3072 [DOI] [PubMed] [Google Scholar]
- 16. Modlin IM, Oberg K, Chung DC, et al. Gastroenteropancreatic neuroendocrine tumours. Lancet Oncol. 2008; 9: 61– 72 [DOI] [PubMed] [Google Scholar]
- 17. Rindi G, Arnold R, Bosman FT. Nomenclature and classification of neuroendocrine neoplasms of the digestive system. In: Bosman TF, Carneiro F, Hruban RH, et al. eds. WHO Classification of Tumours of the Digestive System. 4th ed. Lyon, France: International Agency for Research on Cancer (IARC); 2010: 13 [Google Scholar]
- 18. Rindi G, Klöppel G, Alhman H, et al. TNM staging of foregut (neuro)endocrine tumors: a consensus proposal including a grading system. Virchows Arch. 2006; 449: 395– 401 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Falconi M, Bartsch DK, Eriksson B, et al. ENETS Consensus Guidelines for the management of patients with digestive neuroendocrine neoplasms of the digestive system: well-differentiated pancreatic non-functioning tumors. Neuroendocrinology. 2012; 95: 120– 134 [DOI] [PubMed] [Google Scholar]
- 20. Kunz PL, Reidy-Lagunes D, Anthony LB, et al. Consensus guidelines for the management and treatment of neuroendocrine tumors. Pancreas. 2013; 42: 557– 577 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Zerbi A, Falconi M, Rindi G, et al. Clinicopathological features of pancreatic endocrine tumors: a prospective multicenter study in Italy of 297 sporadic cases. Am J Gastroenterol. 2010; 105: 1421– 1429 [DOI] [PubMed] [Google Scholar]
- 22. Panzuto F, Boninsegna L, Fazio N, et al. Metastatic and locally advanced pancreatic endocrine carcinomas: analysis of factors associated with disease progression. J Clin Oncol. 2011; 29: 2372– 2377 [DOI] [PubMed] [Google Scholar]
- 23. Bilimoria KY, Talamonti MS, Tomlinson JS, et al. Prognostic score predicting survival after resection of pancreatic neuroendocrine tumors: analysis of 3851 patients. Ann Surg. 2008; 247: 490– 500 [DOI] [PubMed] [Google Scholar]
- 24. Strosberg JR, Cheema A, Weber JM, et al. Relapse-free survival in patients with nonmetastatic, surgically resected pancreatic neuroendocrine tumors: an analysis of the AJCC and ENETS staging classifications. Ann Surg. 2012; 256: 321– 325 [DOI] [PubMed] [Google Scholar]
- 25. Zerbi A, Capitanio V, Boninsegna L, et al. Surgical treatment of pancreatic endocrine tumours in Italy: results of a prospective multicentre study of 262 cases. Langenbecks Arch Surg. 2011; 396: 313– 321 [DOI] [PubMed] [Google Scholar]
- 26. Bettini R, Partelli S, Boninsegna L, et al. Tumor size correlates with malignancy in nonfunctioning pancreatic endocrine tumor. Surgery. 2011; 150: 75– 82 [DOI] [PubMed] [Google Scholar]
- 27. Saxena A, Chua TC, Zhao J, et al. Liver-directed therapy for neuroendocrine neoplasm hepatic metastasis prolongs survival following progression after initial surgery. J Surg Oncol. 2012; 105: 342– 350 [DOI] [PubMed] [Google Scholar]
- 28. Mayo SC, de Jong MC, Pulitano C, et al. Surgical management of hepatic neuroendocrine tumor metastasis: results from an international multi-institutional analysis. Ann Surg Oncol. 2010; 17: 3129– 3136 [DOI] [PubMed] [Google Scholar]
- 29. Ho AS, Picus J, Darcy MD, et al. Long-term outcome after chemoembolization and embolization of hepatic metastatic lesions from neuroendocrine tumors. AJR Am J Roentgenol. 2007; 188: 1201– 1207 [DOI] [PubMed] [Google Scholar]


