Abstract
Multiple subsets of nociceptive, parasympathetic, and sympathetic nerves innervate human nasal mucosa. These play carefully coordinated roles in regulating glandular, vascular, and other processes. These functions are vital for cleaning and humidifying ambient air before it is inhaled into the lungs. The recent identification of distinct classes of nociceptive nerves with unique patterns of transient receptor potential sensory receptor ion channel proteins may account for the polymodal, chemo- and mechanicosensitivity of many trigeminal neurons. Modulation of these families of proteins, excitatory and inhibitory autoreceptors, and combinations of neurotransmitters introduces a new level of complexity and subtlety to nasal innervation. These findings may provide a rational basis for responses to air-temperature changes, culinary and botanical odorants (“aromatherapy”), and inhaled irritants in conditions as diverse as allergic and nonallergic rhinitis, occupational rhinitis, hyposmia, and multiple chemical sensitivity.
Keywords: transient receptor potential, TRPV1, nociceptive axon response, nociception
Introduction
The nose is the contact zone that scrubs particulate and water-soluble volatile compounds from inhaled air so that only clean, humidified air reaches the lungs.1 The processes that carry out these functions are under strict physiological control that depends on local mucosal feedback systems and sensory and autonomic reflexes. The histological distributions of sensory and autonomic nerves and their neurotransmitters define the potential functions of each of these sets of nerves.2 Sensory neurons contain calcitonin gene related peptide (CGRP), gastrin releasing peptide (GRP), the tachykinins (neurokinins) substance P and neurokinin A (NKA), and possibly other peptides. CGRP receptors are most dense on arterial and arteriovenous vessels. This is consistent with the potent vasodilator action of this neuropeptide. Neurokinin receptors are widely expressed on many cell types, while GRP receptors are most dense on glands. Glutaminergic afferent neurons may also be present. Parasympathetic neurons may have two populations: larger diameter acetylcholine containing neurons and smaller diameter neurons that release vasoactive intestinal peptide (VIP) and nitric oxide. Sympathetic neurons also have at least two subpopulations that contain norepinephrine with or without neuropeptide Y (NPY). These are short- and long-acting vasoconstrictors, respectively. Adenosine triphosphate may also be a transmitter in nasal sympathetic neurons. The sensory neurons convey information about the conditions of inhaled air from the epithelium to the brain stem, and release neurotransmitters locally within the mucosa by the efferent axon response mechanism. This response is a rapidly induced defense mechanism for hypersecretion of mucus and protection of the nasal mucosa. Acetylcholine stimulates muscarinic M3 and possibly M1 receptors on glands to cause exocytosis. Sympathetic discharge constricts the vessels of the mucosa, squeezes the blood out of the erectile venous sinusoids, and so thins the mucosal lining. This process regulates the cross-section area for airflow, and so nasal patency.
Type C Neurons and Sensory Receptors
Nonmyelinated Type C neurons have specific multimodal chemoreceptors that activate the nasal mucosal trigeminal chemoreceptor system. Irritants, such as carbon dioxide, powdered mannitol, adenosine, hypertonic saline (HTS) solution, and other nociceptive agents, stimulate trigeminal neural responses that are distinct from olfactory sensations. Inhalation of odorless CO2 stimulates limited brain cortical regions, such as the cingulate gyrus.3 In contrast, hydrogen sulfide stimulates olfactory neurons that activate frontal, entorhinal, occipital, and cerebellar cortical regions. Capsaicin, the spicy essence of chili peppers, activates laryngeal and pharyngeal vagal and glossopharyngeal afferents that induce the urge to cough. Functional magnetic resonance imaging demonstrates activation of the primary and visceral insular sensory cortices, anterior midcingulate and orbitofrontal cortices responsible for planning and executive functions, and execution or repression of an active cough via the supplementary motor area and cerebellum.4
HTS sprayed onto the inferior turbinate generates a rapid onset, sharp burning sensation (“first pain”) that is likely mediated by fast conducting, thinly myelinated Aδ nerve fibers.5 A parasthetic, tingling, “second pain” sensation follows. Both the intensity of the first pain and duration of the second pain were related to the HTS dose.6 HTS may have stimulated Type C neurons that have nerve endings in the epithelium but also are extensively branched around submucosal glandular acini, since substance P was released during the first 3-min period. Other colocalized neurotransmitters, such as NKA and GRP, may also have been released at the same time and sites. Tachykinin NK-1 receptor mRNA and autoradiographic GRP binding sites have been localized to glands.6,7 These neuropeptides may have stimulated the HTS dose-dependent glandular secretion that occurs during the 3- to 5-min period after HTS application. This secretion is not altered by the anticholinergic drug ipratropium bromide, indicating that central parasympathetic reflexes were not recruited. There was also no change in albumin concentration in nasal lavage fluid, indicating no alteration in baseline vascular permeability. This finding was reproduced in normal, acute allergic rhinitis, and acute and chronic rhinosinusitis subjects.8
These findings suggest that HTS-induced axon responses in humans induce copious glandular exocytosis with no significant vascular component. Thus, human nasal mucosal neurogenic inflammation is a rapid-onset mucosal defense mechanism designed to stimulate secretion of submucosal gland mucins and antimicrobial proteins onto the nasal epithelial lining fluid where they will replenish gel and sol phase components to adsorb inhaled irritants and particulate material and kill or neutralize microbes and their toxins.
Histamine nasal provocation causes itch, vascular permeability, and cholinergic reflex-mediated glandular secretion. These effects are consistent with the symptoms of allergic rhinoconjunctivitis that follow mast cell histamine release. Histamine H1 receptors have been localized to a population of very narrow-diameter neurons that are localized to distinct spinal cord dorsal horn regions, ascending pathways, thalamus, and thalamocortical radiations from capsaicin-sensitive neurons.9 HistamineH3andH4 receptors on these and other neurons act as inhibitory autoreceptors that inhibit neuron depolarization.10 H2 receptors are present on epithelium and glands. Histamine-induced axon responses have not been clearly demonstrated in human airway mucosa.
“Itch” neurons may rely in part on GRP as a pruritogenic neurotransmitter. GRP receptors are restricted to lamina I of the dorsal horn of the spinal cord, which receives input from Type C neurons.11 Mice with point mutations in GRP receptors have reduced scratching behavior in response to pruritogenic stimuli, yet intact thermal, mechanical, neuropathic, inflammatory, and pain responses.
The population of itch-specific neurons may be small. Other H1 receptor-bearing neurons appear to express the capsaicin receptor that may mediate a burning, prickly heat sensation. The capsaicin receptor is a multimodal sensory receptor and ion channel that has been classified as the transient receptor potential vanilloid 1 (TRPV1) protein. Other distinct combinations of proteins, such as purinergic P2X receptors and acid sensing ion channel 3 (ASIC3), may also be present on subsets of TRPV1 neurons. These would respond to ATP, adenosine, H+, K+, and Ca2+ that may be released by cellular injury or ischemia, or during inflammation.12 The functions of these neurons may be significantly modulated by inflammatory mediators, such as leukotriene B4, nerve growth factor (NGF; TrkA receptor), brain-derived neurotrophic factor (BDNF), and neurotrophin-4 (NT-4) (TrkB receptor), and NT-3 (TrkC receptor). These can induce plasticity of neurotransmitter, sensory, and inhibitory autoreceptor expression, which alters the sensations or sensitivities of dorsal root ganglion and dorsal horn interneuron subpopulations.13
Cold dry air inhalation causes a dose-dependent obstruction of nasal airflow in humans with idiopathic nonallergic rhinitis.14 Healthy controls do not develop obstruction. In contrast, individual mediators with G protein-coupled 7-transmembrane receptors, such as bradykinin and endothelin 1, could not discriminate between these idiopathic rhinitis and control subjects.15 In contrast, both bradykinin and endothelin 1 were able to stimulate nociceptive neurons and recruit parasympathetic reflexes during severe allergic rhinitis, indicating the induction of mucosal hypersensitivity.16,17 Other trigeminal irritants, such as low-dose chlorine that does not induce any sensory perception, can induce neurogenic nasal airflow obstruction without neuropeptide release.18 A local mucosal mechanism is hypothesized.
Voltage-gated Ion Channel Protein Family
TRPV1 was the first of a family of 143 human voltage-gated ion channels to be identified and its activating ligands characterized.19 This family includes other sensors: sodium channels responsible for cell depolarization; calcium channels that mediate complete depolarization that activates the functions of neuron, gland, muscle, and other electro-excitable cells; and potassium channels that repolarize and maintain the resting membrane potential of these cells.19–21 The basic motif of this family of transmembrane proteins is the presence of a single extracellular loop that dips into, but does not cross, the plasma membrane. Transmembrane alpha helices bracket this intramembrane loop. These proteins polymerize into tetramers with adjacent intramembrane loops that form the pores that regulate the flow of specific ions into or out of cells. Heterotetramers may form that have modified responses compared to homotetramers.
TRPV1 is a highly promiscuous polymodal chemoreceptor. Capsaicin, temperatures above 43°C, local anesthetics, nicotine, ethanol, endocannabinoid family of arachidonic acid metabolites, and products of 12- and 15-lipoxygenases interact with distinct portions of TRPV1 to activate a rapid influx of Na+ and Ca2+.22 Other receptors, such as the bradykinin B2 receptor, activate intracellular phosphokinases that can phosphorylate the C-terminal region of TRPV1 to further fine-tune its responses.
TRPV1 was initially thought to be present only on nociceptive nerves, but it is also present in the substantia nigra, hippocampus, and hypothalamus. In addition,TRPV1is also present on airway, skin, gastrointestinal, bladder, and rectal epithelial cells. This means that capsaicin and its many other ligands can activate a broad array of tissues without neural involvement. Expression of TRPV1 and probably other, related sensors on epithelium greatly expands the potential for complex interactions between these cells, intermediate messenger molecules, and Type C neurons. Human 293t embryonic kidney cells transfected with TRPV1 exhibited calcium ion influx indicative of depolarization when stimulated with capsaicin and cyclohexanone.23 Wild-type and transfected cells did not respond to other chemically related trigeminal and olfactory agents, such as acetic acid, R-(−)-carvone, S-(+)-carvone, amyl acetate, toluene, benzaldehyde, (−)-nicotine, or R-(+)-limonene. This implies that additional, separate TRP, olfactory, or other sensory receptor proteins that mediate the trigeminal, irritant actions of these chemicals remain to be identified.
The TRP Thermometer and Aromatherapy
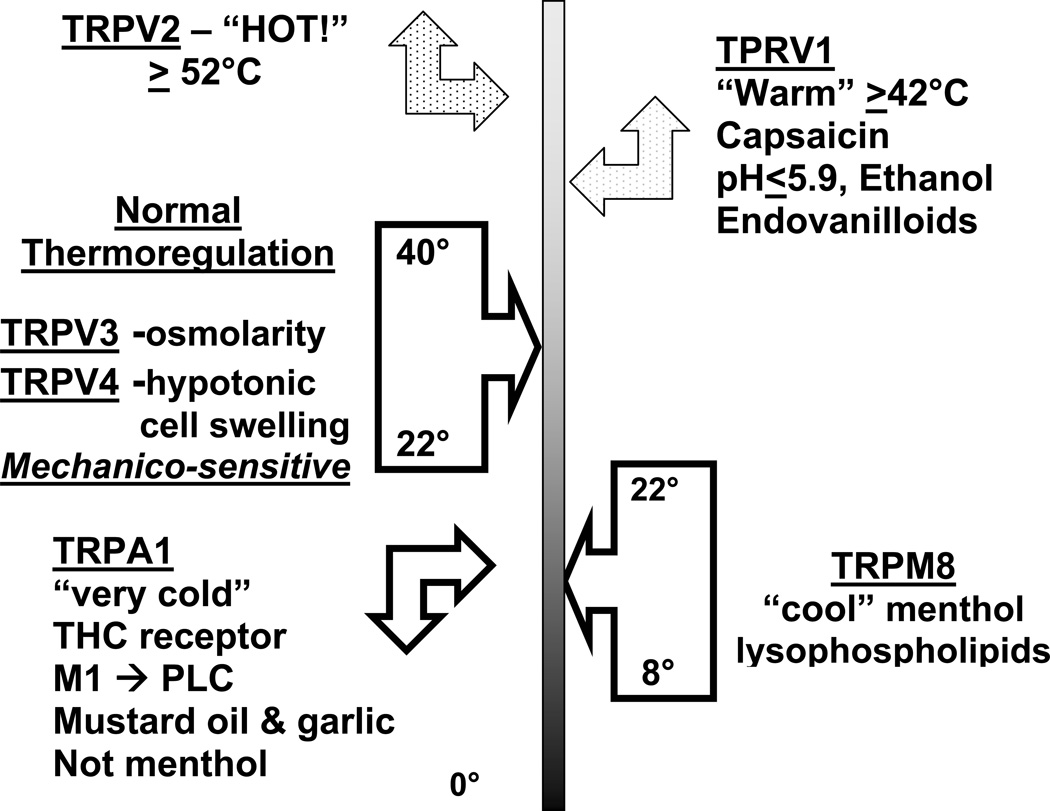
TRPV1 is the only capsaicin-sensitive channel. However, other culinary spices have played major roles in the discovery of TRP protein families.24 Many of these proteins are multimodal and respond to specific temperature and osmolality ranges. A “TRP thermometer” can be constructed to illustrate this diversity (Fig. 1). TRPV3 and TRPV4 respond to ambient temperatures. They are also mechanicosensitive and are activated by changes in cell swelling caused by hyper- and hypoosmolar conditions. Temperatures ≥42°C activate TRPV1, while TRPV2 responds to dangerously high, tissue damaging temperatures ≥52°C. TRPV2 are likely present on rapidly responding Aδ neurons. TRP melanostatin 8 (TRPM8) menthol receptors respond to temperatures between 8°C and 22°C. TRPM8 proteins also respond to lysophospholipids and may be sensors of membrane fluidity. Temperature-dependent changes in fluidity may alter the shape of TRPM8 leading to pore opening, calcium influx, and cellular depolarization that lead to differences in the frequencies of depolarization rates in order to convey different temperatures. The TRP melanostatin 4 (TRPM4) responds to mint and the odorless, topical coolant icilin found in many cutaneous ointments, but does not respond to menthol or cool temperatures. “Very cold” temperatures, mustard oil and garlic isocyanate compounds, and tetrahydrocannabinol activate theTRPA1 (ankryn) receptor and “cold pain.”
Figure 1.
TRP thermometer and aromatherapy: the trigeminal chemosensory nervous system. Used with permission of the copyright holder.
The TRP thermometer may provide insights into the specific airway obstruction caused by cold dry air in idiopathic, nonallergic, noninfectious, noneosinophilic rhinitis.1 The cold temperature may directly activate TRPM8 or TRPA1 ion channels on Type C or Aδ neurons.21 Activation of cold receptors in the nose regulates brain stem motor afferents that modify the breath-to-breath work of breathing. Activation of TRPM8 by menthol and cold air has long been known to produce a transient sensation of airway patency and reduced inspiratory muscle effort. Dry air must become fully hydrated in the nose in order to prevent desiccation of laryngeal, tracheobronchial, and alveolar airways. The evaporation of water from the epithelial lining fluid into the inhaled air has an additional cooling effect due to the enthalpy change. Evaporation increases the tonicity of the epithelial lining fluid and so may act in the same fashion as the HTS provocation model. This further supports a potential role for osmolarity changes and cellular deformation that may induce torsion of putative mechanicosensors on sensory nerves. Osmosensors, such as TRPV3 and TRPV4, may be instrumental in transducing these events by depolarizing sensory neurons. These conjectures may prove to be premature, but investigation of these mechanisms may lead to the development of novel drugs that reduce dyspnea that is currently untreatable but very bothersome in chronic obstructive pulmonary disease, and to reduce the sensation of nasal congestion or fullness that occurs in all virtually all forms of rhinosinusitis.
Conclusions
The regulation of nasal mucosal function by distinct classes of neurons is a new and exciting development. The exchange of neurotransmitters and cellular signaling molecules may generate bilateral trophic effects that regulate epithelial cell, superficial lamina propria vascular, submucosal gland, and deep venous sinusoid activities. Axon response release of neurotransmitters in the mucosa may influence innate and acquired immune responses given the high density of neuropeptide receptors on antimicrobial protein-rich serous cells and lymphocytes, respectively. The HTS provocation model indicates that nociceptive nerve axon responses and “neurogenic inflammation” lead to glandular exocytosis with no vascular leak in human airway mucosa. This is in stark contrast to effects in rat trachea and other rodents, where vascular permeability is induced. Irritant responses may be initiated by activation of TRP, acid-sensing ion channel, purinergic, and other sensory receptors on epithelial cells as well as Type C neurons. These polymodal responses may work in concert with specific mediator receptors to transduce a wide variety of temperature, osmolality, irritant chemical exposure, and other information about the conditions of inhaled air from the nasalmucosa to the central nervous system. A better understanding of the dynamic interactions of the many potential combinations of excitatory sensory and regulatory voltage-gated ion channels, inhibitory autoreceptors, and plasticity of peptidergic and nonpeptidergic neurotransmitters expressed in distinct trigeminal neuron subpopulations will provide for more sophisticated insights into the roles of nociceptive and other nerve subpopulations in regulating nasal mucosal functions.
Acknowledgments
The figure was used with permission of the copyright holder. Supported by Public Health Service Awards 1 RO1 ES015382 and P50 DC006760, and Department of Defense Grant A-14542.0.
Footnotes
Conflicts of Interest
The authors declare no conflicts of interest.
References
- 1.Naclerio RM, et al. Observations on the ability of the nose to warm and humidify inspired air. Rhinology. 2007;45:102–111. [PubMed] [Google Scholar]
- 2.Tai C-F, Baraniuk JN. Upper airway neurogenic mechanisms. Curr. Opin. Allergy Clin. Immunol. 2002;2:11–19. doi: 10.1097/00130832-200202000-00003. [DOI] [PubMed] [Google Scholar]
- 3.Bensafi M, et al. Neural coding of stimulus concentration in the human olfactory and intranasal trigeminal systems. Neuroscience. 2008;154:832–838. doi: 10.1016/j.neuroscience.2008.03.079. [DOI] [PubMed] [Google Scholar]
- 4.Mazzone SB, et al. Representation of capsaicin-evoked urge-to-cough in the human brain using functional magnetic resonance imaging. Am. J. Respir. Crit. Care Med. 2007;176:327–332. doi: 10.1164/rccm.200612-1856OC. [DOI] [PubMed] [Google Scholar]
- 5.Dray A, Urban L, Dickenson A. Pharmacology of chronic pain. TiPS. 1994;15:190–197. doi: 10.1016/0165-6147(94)90147-3. [DOI] [PubMed] [Google Scholar]
- 6.Baraniuk JN, et al. Hypertonic saline nasal provocation stimulates nociceptive nerves, substance P release, and glandular mucous exocytosis in normal humans. Am. J. Respir. Crit. Care Med. 1999;160:655–662. doi: 10.1164/ajrccm.160.2.9805081. [DOI] [PubMed] [Google Scholar]
- 7.Baraniuk JN, et al. Gastrin-releasing peptide in human nasal mucosa. J. Clin. Invest. 1990;85:998–1005. doi: 10.1172/JCI114577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Baraniuk JN, et al. Neuropathology in rhinosinusitis. Am. J. Respir. Crit. Care Med. 2005;171:5–11. doi: 10.1164/rccm.200403-357OC. [DOI] [PubMed] [Google Scholar]
- 9.Andrew D, Craig AD. Spinothalamic lamina 1 neurons selectively sensitive to histamine: a central neural pathway for itch. Nat. Neurosci. 2001;4:72–77. doi: 10.1038/82924. [DOI] [PubMed] [Google Scholar]
- 10.Nakaya M, Takeuchi N, Kondo K. Immunohistochemical localization of histamine receptor subtypes in human inferior turbinates. Ann. Otol. Rhinol. Laryngol. 2004;113:552–557. doi: 10.1177/000348940411300707. [DOI] [PubMed] [Google Scholar]
- 11.Swain MG. Gastrin-releasing peptide and pruritus: more than just scratching the surface. J. Hepatol. 2008;48:681–683. doi: 10.1016/j.jhep.2008.01.007. [DOI] [PubMed] [Google Scholar]
- 12.Vaughan RP, et al. Adenosine sensory transduction pathways contribute to activation of the sensory irritation response to inspirited irritant vapors. Toxicol. Sci. 2006;93:411–421. doi: 10.1093/toxsci/kfl061. [DOI] [PubMed] [Google Scholar]
- 13.Spedding M, Gressens P. Neurotrophins and cytokines in neuronal plasticity. Novartis Found Symp. 2008;289:222–233. doi: 10.1002/9780470751251.ch18. [DOI] [PubMed] [Google Scholar]
- 14.Braat JP, et al. Intranasal cold dry air is superior to histamine challenge in determining the presence and degree of nasal hyperreactivity in nonallergic noninfectious perennial rhinitis. Am. J. Respir. Crit. Care Med. 1998;157:1748–1755. doi: 10.1164/ajrccm.157.6.9701016. [DOI] [PubMed] [Google Scholar]
- 15.Sheahan P, et al. Subjects with non-allergic non-infectious perennial rhinitis do not show nasal hyper-responsiveness to bradykinin. Eur. Arch. Otorhinolaryngol. 2007;264:33–37. doi: 10.1007/s00405-006-0161-4. [DOI] [PubMed] [Google Scholar]
- 16.Riccio MM, et al. Effects of intranasal administration of endothelin-1 to allergic and nonallergic individuals. Am. J. Respir. Crit. Care Med. 1995;152:1757–1764. doi: 10.1164/ajrccm.152.6.8520734. [DOI] [PubMed] [Google Scholar]
- 17.Baraniuk JN, et al. Ibuprofen augments bradykinin-induced glycoconjugate secretion by human nasalmucosa in vivo. J. Allergy Clin. Immunol. 1992;89:1032–1039. doi: 10.1016/0091-6749(92)90226-r. [DOI] [PubMed] [Google Scholar]
- 18.Shusterman D, et al. Chlorine inhalation procedure produces nasal airflow limitation in allergic rhinitis subjects without evidence of neuropeptide release. Neuropeptides. 2004;38:351–358. doi: 10.1016/j.npep.2004.08.002. [DOI] [PubMed] [Google Scholar]
- 19.Caterina MJ, et al. The capsaicin receptor: a heat-activated ion channel in the pain pathway. Nature. 1997;389:816–824. doi: 10.1038/39807. [DOI] [PubMed] [Google Scholar]
- 20.Yu FH, Catterall WA. The VGL-Chanome: a protein superfamily specialized for electrical signaling and ionic homeostasis. Science STKE. 2004:re15. doi: 10.1126/stke.2532004re15. www.stke.org/cgi/content/full/sigtrans;2004/253/re15. [DOI] [PubMed] [Google Scholar]
- 21.Belmone C, Viana F. Molecular and cellular limits to somatosensory specificity. Mol. Pain. 2008;4:14. doi: 10.1186/1744-8069-4-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Van Der Stelt M, Di Marzo V. Endovanilloids. Putative endogenous ligands of transient receptor potential vanilloid 1 channels. Eur. J. Biochem. 2004;271:1827–1834. doi: 10.1111/j.1432-1033.2004.04081.x. [DOI] [PubMed] [Google Scholar]
- 23.Silver WL, et al. TRPV1 receptors and nasal trigeminal chemesthesis. Chem. Senses. 2006;31:807–812. doi: 10.1093/chemse/bjl022. [DOI] [PubMed] [Google Scholar]
- 24.Moran MM, Xu H, Clapham DE. TRP ion channels in the nervous system. Curr. Opin. Neurobiol. 2004;14:362–369. doi: 10.1016/j.conb.2004.05.003. [DOI] [PubMed] [Google Scholar]