Abstract
Background
The Survey of Autonomic Symptom (SAS) scale was reported as an easy instrument to assess the autonomic symptoms in patients with early diabetic neuropathy. In this study, we investigated the relationship between the SAS scale and the parameters of cardiac autonomic neuropathy (CAN) in Korean patients with diabetic peripheral neuropathy (DPN).
Methods
The SAS scale was tested in 30 healthy controls and 73 patients with DPN at Chonbuk National University Hospital, in Korea. The SAS score was compared to the parameters of the CAN test and the total symptom score (TSS) for DPN in patients with DPN.
Results
The SAS symptom score and total impact score were increased in patients with DPN compared to the control group (P=0.01), particularly in sudomotor dysfunction (P=0.01), and vasomotor dysfunction (P=0.01). The SAS score was increased in patients with CAN compared to patients without CAN (P<0.05). Among the diverse CAN parameters, the valsalva ratio and postural hypotension were associated with the SAS score (P<0.05). However, there was no association between the SAS scale and TSS for DPN, and TSS for DPN did not differ between patients with and without CAN.
Conclusion
SAS is a simple instrument that can be used to assess autonomic symptoms in patients with diabetes and can be used as a screening tool for autonomic neuropathy, particularly for CAN.
Keywords: Autonomic neuropathy, Diabetes, Survey of autonomic symptom scale
INTRODUCTION
Patients with diabetes have various chronic complications, such as retinopathy, neuropathy, nephropathy, and other cardiovascular diseases. Among them, previous studies have reported that the prevalence of autonomic neuropathy was between 7.7% and 90% on the based of autonomic testing [1], and autonomic symptoms may occur frequently in patients with diabetes. However, they could not be detected easily because of several reasons. For example, autonomic symptoms are diverse and vague, and autonomic examinations were not performed due to their diversity and complexity and because these examinations are time-consuming. Thus, there is a need for simple devices and methods for the evaluation of autonomic functions in clinical practice.
Previously, various instruments were developed to measure these autonomic symptoms [2]. One validated scale is the Autonomic Symptom Profile (ASP) and the Composite Autonomic Symptom Scale (COMPASS), which was developed to provide a score of the autonomic symptoms. The COMPASS scores correlated well with the Composite Autonomic Scoring Scale (CASS), which were obtained from autonomic testing. However, the correlation between autonomic symptoms and CASS was weak overall [3]. Furthermore, these instruments had many items to check; thus it was time-consuming to use in everyday clinical practice. Therefore, Zilliox et al. [4] developed the Survey of Autonomic Symptom (SAS) scale to easily measure autonomic symptoms in patients with early diabetic neuropathy.
Diabetic autonomic neuropathy can occur in many diverse internal organs, such as those in cardiovascular, metabolic, gastrointestinal, genitourinary, sudomotor and papillary systems [1]. Previous studies have reported that cardiac autonomic neuropathy (CAN) was associated with major mortality and morbidity in patients with diabetes [5,6,7]. Thus, CAN testing is needed to care for patients with diabetes; however, it could not be performed in everyday clinical practice for several reasons. There is also no simple assessing tool for autonomic neuropathy symptoms in Korea. Furthermore, diabetic peripheral neuropathy (DPN) can be easily assessed using the clinical score, total symptom score (TSS), monofilament test, vibration test, or quantitative sensory test (QST) in clinical practice. However, there were a few studies that have examined the relationship between the autonomic neuropathy and the DPN. Thus, we studied the relationship using the SAS instrument and a series of CAN tests in patients with DPN in Korea.
METHODS
From November 2012 to March 2013, a total of 77 patients with DPN and 30 normal participants were enrolled at Chonbuk National University Hospital in Korea. The demographic information and important medical histories of patients were reviewed by medical records, including age, sex, race, type of diabetes, duration of diabetes, height, body weight, body mass index (BMI), hypertension, hemoglobin A1c (HbA1c), fasting plasma glucose (FPG), and postprandial glucose (PPG). The participants' medical records were carefully reviewed to exclude subjects with other neurological or autonomic disorders. A diagnosis of type 2 diabetes was made based on the American Diabetes Association (ADA) guidelines. The normal group was made based on patients' medical history, FPG, PPG, and HbA1c levels. The normal group was not included in the prediabetic range. In both groups, patients who had coronary heart disease or renal dysfunction were not included in this study. Among the diabetes group, two patients were taking medication with significant anticholinergic properties, antidepressants or β-blockers, which may influence neuropathy. Thus, these patients were excluded in this study. Both the diabetes and normal groups were asked to respond to the SAS scale. In addition, patients with DPN underwent cardiac autonomic neurological examinations on the same day. Caffeine, smoking and some medications that may affect CAN, and these were discontinued for at least 12 hours prior to performing CAN. We compared the SAS scale to a series of CAN tests respectively. These studies were approved by the Institutional Review Board of Chonbuk National University Hospital for research.
The SAS scale consists of 11 items in females and 12 in males (Table 1). It assessed both the presence of autonomic symptoms and the degree of severity. Each item was rated using an impact score ranging from 1 (least severe) to 5 (most severe). These questions assessed the following autonomic symptom domains: orthostatic, sudomotor, vasomotor, gastrointestinal, urinary, and sexual dysfunction (for males). It was translated into the Korean language with the consent of the developer of the SAS scale. The transcript was also retranslated by experts of the English and Korean languages.
Table 1.
Survey of autonomic symptoms
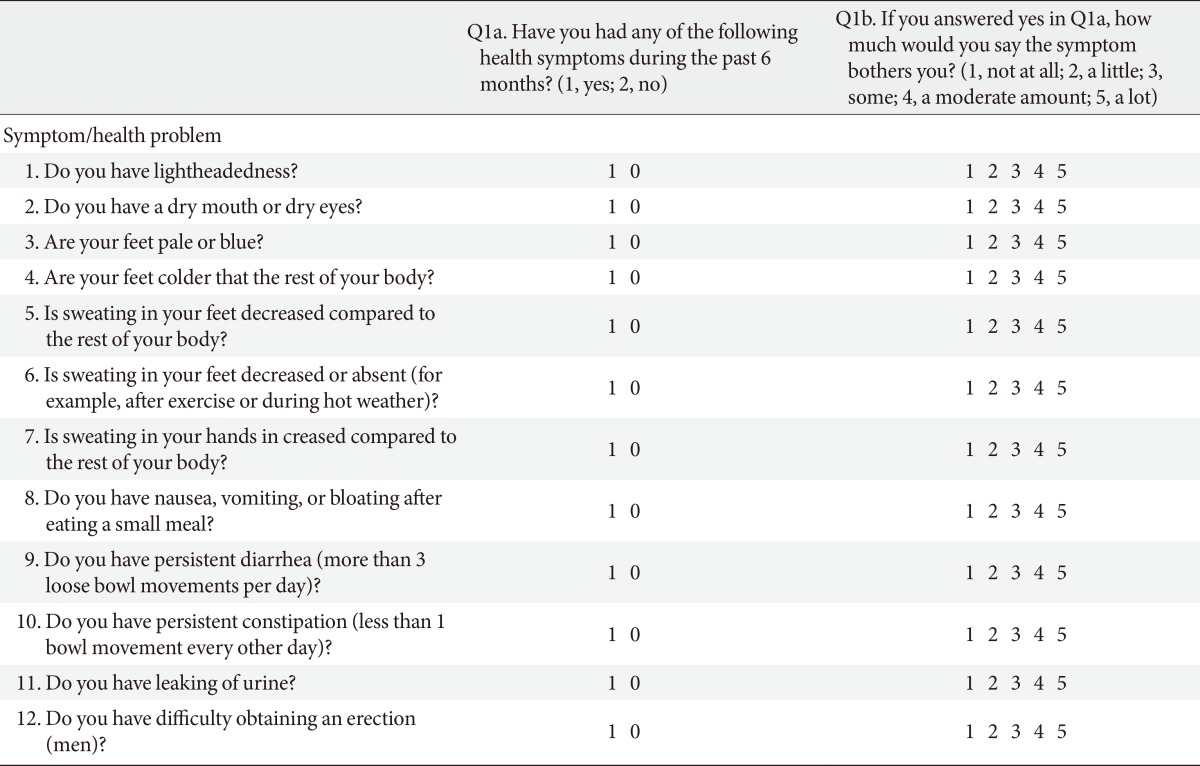
Adapted from Zilliox et al. Neurology 2011;76:1099-105, with permission from Lippincott Williams & Wilkins [4]. Number of symptoms reported: sum of column a, 0-12 for men, and 0-11 for women; total symptom impact score: sum of column b, 0-60 for men and 0-55 for women.
CAN was diagnosed according to the ADA guidelines [8]. CAN testing was performed using a devise (DiCAN; Medicore, Seoul, Korea) by one physician. They consisted of the valsalva ratio (changes in heart rate during the valsalva maneuver), lying standing heart rate (30/15 ratio), R-R interval variation (maximum-minimum heart rate during expiration and inspiration), postural hypotension (change in systolic blood pressure after standing), and sustained handgrip (increase in diastolic blood pressure after the handgrip exercise). All of these tests and their normal values have been used according to ADA recommendations (normal valsalva ratio >1.2, normal lying standing heart rate >1.04, normal R-R interval variation >15 bpm, normal postural hypotension <10 mm Hg, and normal sustained handgrip >16 mm Hg). These results were reported as either normal or abnormal. The existence of CAN was based on the sum of the CAN test score (using Ewing methods).
DPN was diagnosed by the clinical score (Michigan Neuropathy Screening Instrument questionnaire), TSS for pain, medical record review, electrophysiology study, or QST. QST testing was performed using the device (TSH II; Medoc, Ramat Yishai, Israel). All participants of the DPN group had small or large fiber neuropathy at the QST examinations. The TSS consisted of the frequency and intensity for pain, burning, paresthesia, and numbness.
All of the statistical analyses were performed using the SPSS version 18 (IBM Co., Armonk, NY, USA). Continuous variables were described using the mean±standard error of the mean (SEM), and categorical variables were presented as percentages. The Mann-Whitney U test was used to compare between groups for variables with a skewed distribution. The Spearman correlation method was used to evaluate whether there was a significant correlation between variables. Receiver operating characteristic (ROC) curves were calculated to assess the SAS cutoff value. P values <0.05 were considered statistically significant for all calculations.
RESULTS
Baseline characteristics of the participants
The demographics of the participants are listed in Table 2. The mean age of 73 patients with DPN was 58.4±12.9 years. The subjects consisted of 42 males (57.5%) and 31 females (42.5%). The race of all participants was Asian. The type of diabetes of all patients was type 2 diabetes. The mean HbA1c was 62.8±4.0 mmol/mol (7.9%±1.8%). The normal group (n=30) consisted of four males (13.3%) and 26 females (86.7%). Despite of the female predominance and lower body weight in the normal group, there was no difference between the age, blood pressure, height, or BMI in either the normal controls or patients with DPN.
Table 2.
Baseline characteristic of the participants
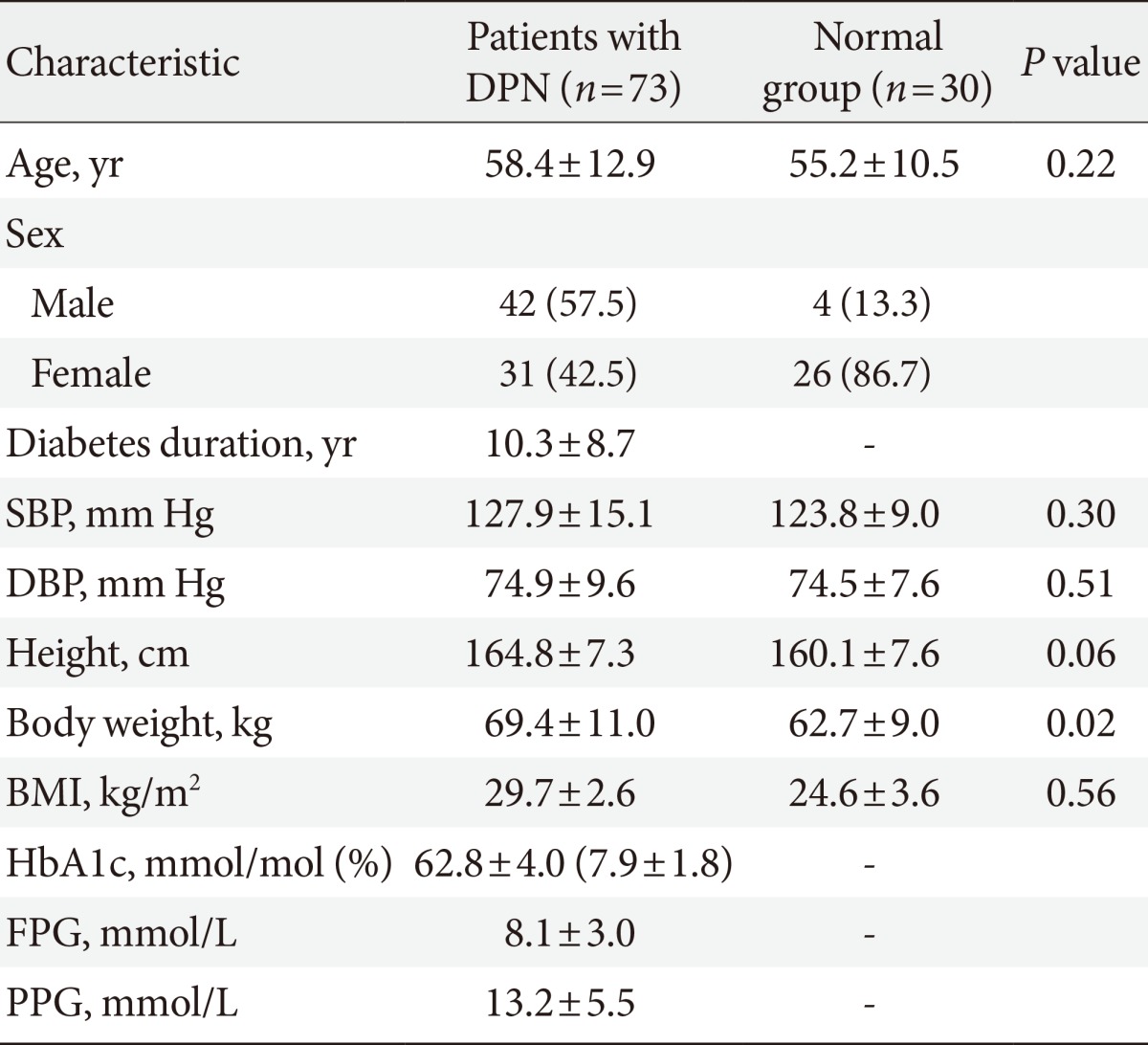
Values are presented as mean±standard deviation or number (%).
DPN, diabetic peripheral neuropathy; SBP, systolic blood pressure; DBP, diastolic blood pressure; BMI, body mass index; HbA1c, hemoglobin A1c; FPG, fasting plasma glucose; PPG, postprandial glucose.
Difference in the SAS scale between patients with DPN and the normal group
We compared the SAS scale between patients with DPN and the normal group (Table 3). There was a difference in both the SAS symptom score (4.3±2.2 in patients with DPN, 2.0±1.1 in normal group, and P=0.01) and the SAS total symptom impact score (TIS; 12.9±8.3 in patients with DPN, 5.4±2.5 in the normal group, and P=0.01). Among the SAS items, the scores of orthostatic, sudomotor, vasomotor, and sexual dysfunction items were increased in patients with DPN compared to the normal group. Among them, there was a significant difference in the sudomotor dysfunction (P=0.01) and vasomotor dysfunction (P=0.01) items in each groups.
Table 3.
Survey of autonomic symptoms scales between patients with DPN and the normal group
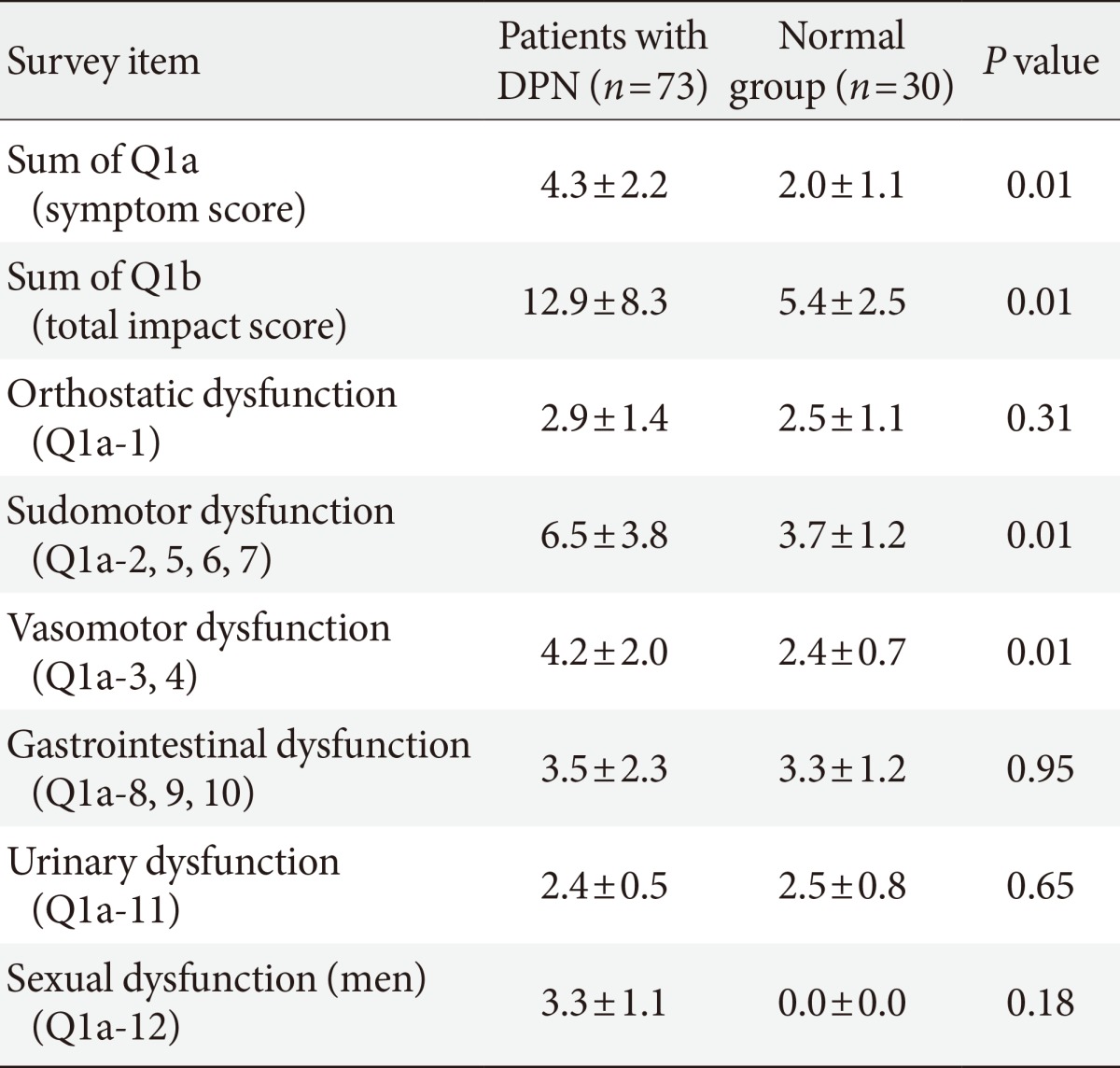
Values are presented as mean±standard deviation.
DPN, diabetic peripheral neuropathy.
Correlation between the SAS scale and CAN testing in patients with DPN
In a total of 73 patients with DPN, 58 patients (79.5%) had one or more CAN test abnormality. In patients with CAN (n=58), 38 patients (65.5%) had autonomic neuropathy according to the SAS symptom score and 43 patients (74.1%) had autonomic neuropathy according to the SAS TIS. We assessed the correlation degree between the SAS scale and CAN test in patients with DPN (Table 4). There was a significant correlation between the SAS scale (symptom score, TIS) and CAN test (valsalva ratio, postural hypotension). At subanalysis, the sudomotor dysfunction item (Q1a-2, 5, 6, 7) was significantly correlated with the CAN item (valsalva ratio, postural hypotension). In addition, the orthostatic dysfunction item (Q1a-1) was also significantly associated with the valsalva ratio and postural hypotension (P<0.05). Among the CAN test, postural hypotension and changes in diastolic blood pressure during sustained handgrip exercise represent an abnormality of the sympathetic nerve. The sudomotor dysfunction item significantly correlated with postural hypotension (P<0.05).
Table 4.
Correlations between the SAS scale, cardiac autonomic neuropathy test, and total symptom score in patients with DPN
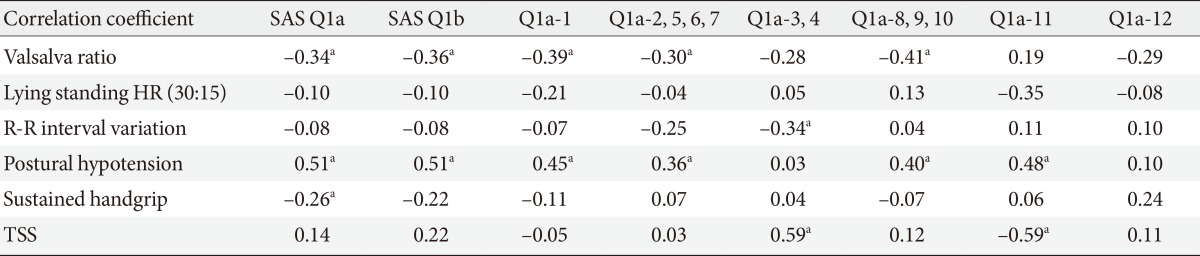
SAS, survey of autonomic symptom; DPN, diabetic peripheral neuropathy; HR, heart rate; TSS, total symptom score (for DPN).
aP<0.05.
We compared the autonomic dysfunction symptom frequency, SAS scales, and TSS in patients with DPN, according to the presence of CAN (Table 5). Patients with CAN mostly reported that they had sudomotor dysfunction (86.2%). Both the SAS symptom score and TIS significantly increased in the patients with CAN compared to patients without CAN (respectively P=0.01, and P=0.02). The sudomotor dysfunction item also significantly differed in each group (P=0.03).
Table 5.
Autonomic dysfunction symptom frequency, SAS scales, and TSS in patients with diabetes according to the CAN existence
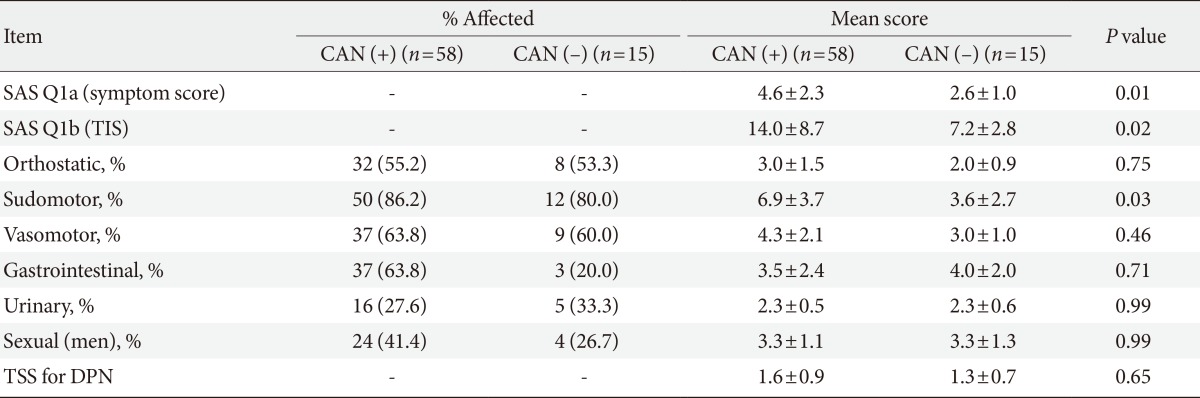
Values are presented as mean±standard deviation or number (%).
SAS, survey of autonomic symptom; TSS, total symptom score; CAN, cardiac autonomic neuropathy; TIS, total impact score; DPN, diabetic peripheral neuropathy.
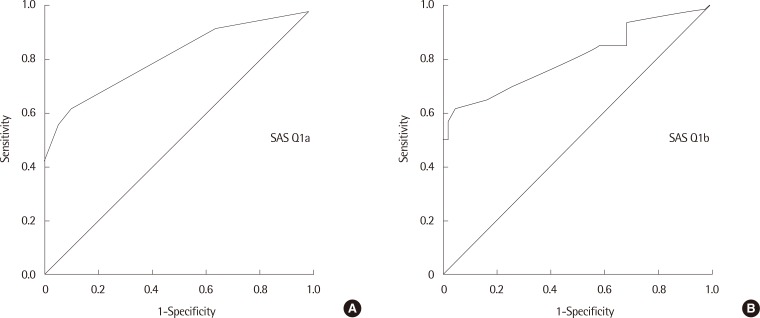
Furthermore, we studied the SAS cutoff value using ROC curves (Fig. 1). The area under the curve was as follows: the SAS symptom score was 0.813 (SEM, 0.042; 95% confidence interval [CI], 0.731 to 0.894) and SAS TIS was 0.802 (SEM, 0.043; 95% CI, 0.718 to 0.896). Based on the SAS symptom score, a cutoff value to determine CAN existence was 1.5, which would provide greater than 91.7% sensitivity and 63.4% specificity. On the basis of SAS TIS, the cutoff value to determine CAN existence was 3.5. This point would provide greater than 93.3% sensitivity and 68.3% specificity.
Fig. 1.
Receiver operating characteristic curves for the survey of autonomic symptom (SAS) scales. (A) SAS Q1a (symptom score). (B) SAS Q1b (total impact score).
Correlation between SAS scale and the TSS for DPN
We also examined the relationship between the TSS for DPN and SAS scales (Table 4). However, there was no significant correlation between the TSS for DPN and SAS scale (symptom score, TIS). Among the SAS scales, vasomotor dysfunction and urinary dysfunction were associated with the TSS for DPN. There was also no significant difference between the TSS for DPN in patients with CAN compared to patients without CAN (Table 5).
DISCUSSION
In this study, the SAS scale significantly differed in patients with DPN compared to the normal group. Furthermore, there was an association between the SAS scale and CAN tests in patients with DPN. Diabetic autonomic neuropathy is one of the most insidious chronic complications particularly in patients with a long-standing and poorly controlled glycemic status. Much attention has been given to the cardiovascular aspect of diabetic autonomic neuropathy, which has been associated with increased mortality. However, the diagnostic tests of autonomic neuropathy are more complicated and time-consuming compared with other complication assessment tools. Furthermore, currently available autonomic neuropathy questionnaires are lengthy, complex, and require training and considerable time for scoring. There is a need for simple instruments and methods for the evaluation of autonomic functions. Among the previous devices, the SAS scale is an easily administered instrument to measure autonomic symptoms in early diabetic neuropathy [4]. Thus, we studied the relationship using this instrument and a series of CAN tests in patients with DPN.
In our results, there was a significant difference in both the SAS symptom score and TIS between the normal group and patients with DPN. This finding indicated that the SAS scale has the power to distinguish autonomic dysfunction between the normal group and patients with DPN. A previous study reported that a cutoff value of greater than three for the SAS symptom score would provide greater than 90% specificity and 65% sensitivity in determining autonomic dysfunction in early diabetic neuropathy patients [4]. In addition, a cutoff value greater than seven for SAS TIS would provide greater than 90% sensitivity and 60% sensitivity. Our results revealed that approximately 52% to 58% patients with DPN responded that they had autonomic neuropathy, which was consistent with these criteria. A previous study also described that the SAS scale demonstrated a strong association with the ASP total score in patients with early diabetic neuropathy [4]. Our results also showed that patients who had a long duration of diabetes showed similar results. Thus, the SAS scale, which is simple to complete, may potentially help to detect and diagnose autonomic neuropathy in patients with DPN.
Among diverse SAS items, sudomotor and vasomotor dysfunctions significantly differed between patients with DPN and the normal group. The sudomotor dysfunction item score also significantly differed in patients with CAN compared to patients without CAN. Patients with CAN mostly responded that they suffered from sudomotor dysfunction. Similar results have also been previously reported. Sympathetic C-fibers innervating sweat glands can be impaired early in patients with diabetes. Yajnik et al. [9] proposed that an examination for sudomotor dysfunction can be used for early screening of CAN in clinical practice prior to performing the specific CAN test. Our results revealed that patients with CAN had a higher sudomotor dysfunction score compared to patients without CAN. However, there were no differences in orthostatic dysfunction, gastrointestinal dysfunction, urinary dysfunction, and sexual dysfunction. This finding may indicate that the SAS scale was not a sufficient questionnaire to assess these autonomic dysfunctions. Moreover, gastrointestinal, urinary symptoms and various features in patients with autonomic neuropathy were nonspecific. Thus, further studies examining these aspects are required.
CAN is associated with major mortality and morbidity of patients with diabetes [5,6,7]. However, a simple assessment tool for autonomic neuropathy is not used in everyday clinical practice. Thus, we examine the relationship with CAN using the Ewing methods and the SAS scale in patients with DPN. Patients with CAN had a higher SAS symptom score and TIS compared to patients without CAN. However, there was no association between the SAS scale and TSS for DPN. In addition, there was no difference in the SAS scale and TSS for DPN between patients with CAN and without CAN. DPN consisted of painful neuropathy and painless neuropathy. Regrettably, we did not assess the DPN according to these features because it would influence the result. In our study, the SAS scale was superior to TSS for DPN assessment of CAN existence in patients with DPN. Considering that CAN is associated with the mortality of diabetic patients, simple questionnaire scores, such as SAS, may be useful to assess diabetic autonomic neuropathy, particularly CAN compared with TSS for DPN in clinical practice.
A potential weakness of the study is that it was performed at a single center. There was a difference in the sex and obesity of the normal group. Regretfully, our study enrolled more females and light body weight participants for normal participants compared to patients with DPN. Obesity has been previously proposed to be associated with impaired autonomic function [10]. Second, diabetic autonomic neuropathy was detected in 52% to 58% of patients with DPN according to the SAS scale. However, CAN was detected in 80% of patients with DPN according to the Ewing method. This difference may be the limitation of the SAS scale to assess the autonomic neuropathy using only the subjective symptoms. Third, we could not adjust the effect of the glycemic control status, such as HbA1c. This is a common influencing factor for neuropathy. Thus, further studies to supplement these aspects, which include more diverse participants and multicenter data, are needed.
In conclusion, the SAS scale is an easily administered instrument that can be used to measure autonomic symptoms and can be useful as a screening tool to assess autonomic neuropathic symptoms in patients with DPN. Furthermore, the SAS scale is also helpful for assessing CAN in patients with diabetes. In the future, additional studies using a large population as well as multicenter oriented studies are needed for the clinical adaptation of the SAS scale in the assessment of patients with diabetes.
Footnotes
No potential conflict of interest relevant to this article was reported.
References
- 1.Vinik AI, Maser RE, Mitchell BD, Freeman R. Diabetic autonomic neuropathy. Diabetes Care. 2003;26:1553–1579. doi: 10.2337/diacare.26.5.1553. [DOI] [PubMed] [Google Scholar]
- 2.Suarez GA, Opfer-Gehrking TL, Offord KP, Atkinson EJ, O'Brien PC, Low PA. The autonomic symptom profile: a new instrument to assess autonomic symptoms. Neurology. 1999;52:523–528. doi: 10.1212/wnl.52.3.523. [DOI] [PubMed] [Google Scholar]
- 3.Low PA, Benrud-Larson LM, Sletten DM, Opfer-Gehrking TL, Weigand SD, O'Brien PC, Suarez GA, Dyck PJ. Autonomic symptoms and diabetic neuropathy: a population-based study. Diabetes Care. 2004;27:2942–2947. doi: 10.2337/diacare.27.12.2942. [DOI] [PubMed] [Google Scholar]
- 4.Zilliox L, Peltier AC, Wren PA, Anderson A, Smith AG, Singleton JR, Feldman EL, Alexander NB, Russell JW. Assessing autonomic dysfunction in early diabetic neuropathy: the Survey of Autonomic Symptoms. Neurology. 2011;76:1099–1105. doi: 10.1212/WNL.0b013e3182120147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ewing DJ, Campbell IW, Clarke BF. The natural history of diabetic autonomic neuropathy. Q J Med. 1980;49:95–108. [PubMed] [Google Scholar]
- 6.O'Brien IA, McFadden JP, Corrall RJ. The influence of autonomic neuropathy on mortality in insulin-dependent diabetes. Q J Med. 1991;79:495–502. [PubMed] [Google Scholar]
- 7.Rathmann W, Ziegler D, Jahnke M, Haastert B, Gries FA. Mortality in diabetic patients with cardiovascular autonomic neuropathy. Diabet Med. 1993;10:820–824. doi: 10.1111/j.1464-5491.1993.tb00173.x. [DOI] [PubMed] [Google Scholar]
- 8.Boulton AJ, Vinik AI, Arezzo JC, Bril V, Feldman EL, Freeman R, Malik RA, Maser RE, Sosenko JM, Ziegler D American Diabetes Association. Diabetic neuropathies: a statement by the American Diabetes Association. Diabetes Care. 2005;28:956–962. doi: 10.2337/diacare.28.4.956. [DOI] [PubMed] [Google Scholar]
- 9.Yajnik CS, Kantikar V, Pande A, Deslypere JP, Dupin J, Calvet JH, Bauduceau B. Screening of cardiovascular autonomic neuropathy in patients with diabetes using non-invasive quick and simple assessment of sudomotor function. Diabetes Metab. 2013;39:126–131. doi: 10.1016/j.diabet.2012.09.004. [DOI] [PubMed] [Google Scholar]
- 10.Andreazzi AE, Grassiolli S, Marangon PB, Martins AG, de Oliveira JC, Torrezan R, Gravena C, Garcia RM, Mathias PC. Impaired sympathoadrenal axis function contributes to enhanced insulin secretion in prediabetic obese rats. Exp Diabetes Res. 2011;2011:947917. doi: 10.1155/2011/947917. [DOI] [PMC free article] [PubMed] [Google Scholar]