Abstract
HIV-infected patients have a significantly greater risk of cardiovascular disease. Several markers including osteoprotegerin have been shown to be involved in the development and progression of atherosclerosis. We investigated the relationship between T-cell phenotype, osteoprotegerin, and atherosclerosis evaluated by carotid intima-media thickness (c-IMT) in 94 HIV+ patients on suppressive antiretroviral therapy with Framingham score <10%. As for the control group, 24 HIV-negative subjects were enrolled. c-IMT was assessed by ultrasound. CD4+/CD8+ T-cell activation (CD38+ HLADR+) and senescence (CD57+ CD28−) were measured by flow cytometry. IL-6 and OPG levels were measured by ELISA kit. c-IMT was higher in HIV+ than in controls. Among HIV+ patients, 44.7% had pathological c-IMT (≥0.9 mm). CD8+ T-cell activation and senescence and OPG plasma levels were higher in HIV+ patients than in controls. Subjects with pathological c-IMT exhibited higher CD8+ immune activation and immunosenescence and OPG levels than subjects with normal c-IMT. Multivariate analysis showed that age, CD8+ CD38+ HLADR+, and CD8+ CD28− CD57+ were independently associated with pathological c-IMT. Several factors have been implicated in the pathogenesis of atherosclerosis in HIV patients. Immune activation and immunosenescence of CD8+ T cell together with OPG plasma levels might be associated with the development and progression of early atherosclerosis, even in the case of viral suppression.
1. Introduction
HIV-infected patients, with the increasing life expectancy, appear to have a significantly greater risk of cardiovascular disease (CVD) than HIV-negative individuals. It now appears clear that both HIV infection itself and antiretroviral therapy (ART) are associated with a higher risk of CVD and metabolic disorders. CVD, which occurs in the general population with advancing age, seems to be correlated with a premature aging in HIV-infected patients, occurring at an earlier age in HIV-infected than in uninfected subjects [1, 2]. Moreover, recent studies showed how the risk is elevated also in HIV-positive subjects virologically suppressed and with a low Framingham score [3]. An important aspect of this premature aging is the immune activation and the consequent immunosenescence that causes a thymic involution, a reduced circulating naive T cells, an increased number of CD4+ well-differentiated CD28− T cells, and an increased level of proinflammatory cytokines [4–7]. Recent studies have introduced the hypothesis that chronic inflammation, immune activation, and immunosenescence might contribute to the endothelial activation/dysfunction with consequent atherosclerosis in the setting of HIV infection [8]. Several markers, such as VCAM-1, ICAM-1, and von Willebrand factor antigen, have been shown to reliably indicate the increased activation of endothelial cells in atherosclerosis. Tumor necrosis factor-α (TNF-α) has been implicated in myocardial dysfunction resulting from acute coronary syndrome and high levels of C-reactive protein and IL-1 and IL-6 have been associated with subclinical atherosclerosis [9–12]. Furthermore, new soluble markers including osteoprotegerin (OPG), member of the TNF superfamily, have been shown to be involved in the development and progression of atherosclerosis [13, 14]. Given the morbidity and the mortality associated with overt CVD, early identification and management of subclinical disease are desirable before complications of overt CVD develop [15]. Measurement of carotid intima-media thickness (c-IMT) has previously been correlated with the extent of coronary atherosclerosis and increases in c-IMT are predictive of future CVD events [16]. The aim of our study was to investigate the relationship of lymphocytes T-cell phenotype, IL-6, and OPG plasma levels with atherosclerosis evaluated by c-IMT in HIV-positive patients on suppressive ART.
2. Materials and Methods
2.1. Ethics Statement
The study protocol designed according to the Helsinki Declaration II was approved by the local ethics committee. All the patients gave written informed consent to participate.
2.2. Patients
We recruited 118 patients from the Department of Public Health and Infectious Diseases of “Sapienza” University of Rome. Ninety-four were HIV-infected subjects on ART since 48 weeks with undetectable viremia (<37 copies/mL) and low cardiovascular diseases risk defined by a Framingham score <10%. As for the control group, we enrolled 24 HIV-negative individuals matched for age, gender, and Framingham score. For each patient, we collected medical and family history, lifestyle, smoking status, ART, HIV-RNA zenith, and nadir CD4+ cell count. Current lymphocytes T CD4+ and CD8+ cell count was determined by flow cytometric analysis (MACSQuant Analyzer, Miltenyi Biotec, Germany) and HIV-1 RNA plasma levels were detected by a quantitative reverse polymerase chain reaction (Amplicor HIV Monitor; Roche Diagnostic System, Branchburg, NJ, version 1.5, l.o.d. 37 copies/mL). Triglycerides, total cholesterol, high density lipoprotein cholesterol (HDL), and low density lipoprotein cholesterol (LDL) were measured in blood samples. Body mass index (BMI) was calculated (kg/m2) and recorded for each individual. Due to the high influence of CMV on immunosenescence and due to immune activation and in order to eliminate the CMV as a confounding factor, only CMV positive subjects were included in both study populations. Exclusion criteria were age <18 years, previous virological failure, recent AIDS-defining illness, coinfection with hepatitis virus, and presence of other comorbidities (metabolic syndrome, diabetes mellitus, arterial hypertension, kidney disease, and hormonal dysfunction).
2.3. IL-6 and Osteoprotegerin Detection
IL-6 was measured with an ELISA kit (eBioscience Bender MedSystems, Inc., Vienna, Austria). The detection limit of assay was 3.1 pg/mL. Osteoprotegerin (OPG) levels were measured by ELISA kits (Biomedica Gruppe, Vienna, Austria). The detection limit of assay was 0.12 pmol/L.
2.4. Immune Activation and Immunosenescence
Lymphocyte surface phenotypes were evaluated by flow cytometry using fresh peripheral blood. For the activation and the senescence analysis of CD4+ and CD8+ T cells, the following fluorochrome-labeled antibodies were used: Pacific Blue-CD3 (BioLegend, 500 uL), FITC-CD28 (BioLegend, 2 mL), PE-CD57 (BioLegend, 2 mL), PerCp/Cy5.5-HLA DR (BioLegend, 500 uL), Pe/Cy7-CD8 (BioLegend, 2 mL), CD38-APC (BioLegend, 2 mL), and APC/Cy7-CD4 (BioLegend, 2 mL). The flow cytometer was calibrated using MACSQuantTM Calibration Beads and for the automatic compensation marked beads (BDTM CompBeads anti-mouse Ig, k) and unmarked beads (BDTM CompBeads negative control, FBS) were used. For the lysis of red blood cells, FACS Lysing Solution (BD, Lysing Solution) was used. The flow cytometer used for the analysis was the MACSQuant Analyzer with 8 channels and the data were analyzed using the FlowJo V10 software. Before each acquisition, both calibration and automatic compensation were performed. Immune activation was defined as HLADR+ CD38+ whereas immunosenescence was defined as CD57+ CD28− [17, 18].
2.5. Carotid Intima-Media Thickness Measurement
c-IMT measurement was obtained for each patient using a B-mode ultrasound recording with a 7 to 14 MZ array probe (Esaote Technology). Patients must lay in supine position in a dark room with a slight hyperextension and must turn the neck to the opposite side. The common carotid, the bifurcation, and at least the first 2 cm of the internal carotid were examined on the long and short axes. In addition, 3 measurements were made at the far and near walls of each internal carotid and specifically at the carotid bifurcation and the bulb and 1 cm after the bifurcation. The mean value (expressed as mm) of the 3 measurements taken at each site of the internal carotid (left and right) was calculated for each patient and used as the final measurement of internal c-IMT. According to published population studies, we defined normal c-IMT as IMT <0.9 mm and pathological c-IMT as IMT >0.9 mm [19]. In order to avoid interoperator differences, all the measurements of c-IMT were performed by a single operator.
2.6. Statistical Analysis
Continuous data were analyzed with Student's t test, whereas the nonparametric Mann-Whitney test was applied for values not normally distributed. Pearson correlation coefficient was used for correlations. Linear regression model was tested to evaluate the association between lymphocytes T-cell phenotype, IL-6, OPG, and c-IMT. To explore the factors independently associated with c-IMT, multivariable logistic regression was performed. Data were expressed as median (range) or mean ± standard deviation (SD), as appropriate. A P value of <0.05 was considered statistically significant. Statistical analyses were performed using STATA (version 9) software (STATA Corp. LP, College Station, TX, USA).
3. Results
3.1. Characteristics of the Study Population
Out of 94 HIV-infected subjects with a low cardiovascular diseases risk (Framingham score <10%), 72 (76.5%) were males and 22 (23.5%) were females with a mean age of 47.4 ± 11.4 years. 52.1% were not smokers and mean BMI was 20.7 ± 2.4 kg/m2. Median nadir and current lymphocytes T CD4+ cells count were 195.5 cell/mmc (range 4–1318) and 643.5 cell/mmc (range 159–1705), respectively. Out of 94 HIV-infected subjects on ART, 62.7% were on PI and 37.3% were on NNRTI based regimen. All the patients were CMV positive. The mean plasma concentration of total cholesterol was 188.3 ± 47.4 mg/dL, HDL cholesterol was equal to 48 ± 14.6 mg/dL, LDL cholesterol was equal to 114 ± 46.3 mg/dL, and triglycerides were equal to 145.3 ± 80.9 mg/dL. None of the subjects were receiving lipid-lowering therapy. General characteristics of study population are summarized in Table 1.
Table 1.
Clinical characteristics of study population.
| HIV+ | HIV− | P value | |
|---|---|---|---|
| Age (M ± SD) | 47.4 ± 11.4 | 48 ± 9 | P = 0.432 |
| Sex | 72 M, 22 F | 18 M, 6 F | P = 0.581 |
| Smoke status (n, %) | |||
| No | 49 (52.1%) | 15 (62.5%) | P = 0.745 |
| Yes | 45 (47.9%) | 9 (37.5%) | P = 0.669 |
| CD4+ (mmc) (M ± SD) | 685.8 ± 395.5 | 770 ± 221 | P = 0.485 |
| CD4+ % (M ± SD) | 27.3 ± 7.6 | 36 ± 9.3 | P = 0.563 |
| CD4+ (mmc) nadir (M ± SD) | 242.3 ± 227.6 | — | n.a. |
| CD4+ % nadir (M ± SD) | 14.7 ± 9.6 | — | n.a. |
| HIV-RNA zenith (cp/mL) (M ± SD) | 297218.1 ± 114812.4 | — | n.a. |
| HIV-RNA (cp/mL) | <37 | <37 | n.a. |
| ART (n, %) | 94/94 (100%) | — | n.a. |
| PI based regimen | 59/94 (62.7%) | ||
| NNRTI based regimen | 35/94 (37.3%) | ||
| CMV serostatus (n, %) | 94/94 (100%) | 24/24 (100%) | n.a. |
| Lipid-lowering therapy | |||
| No | 94 (100%) | 24 (100%) | n.a |
| Yes | 0 | 0 | |
| SBP (mmHg) | 120.3 ± 12.5 | 116.8 ± 18.4 | P = 0.362 |
| DBP (mmHg) | 80.2 ± 10.6 | 83.5 ± 12.1 | P = 0.175 |
| Triglycerides (mg/dL) (M ± SD) | 145.3 ± 80.9 | 109.6 ± 42 | P = 0.921 |
| Cholesterol total (mg/dL) (M ± SD) | 188.3 ± 47.4 | 144.3 ± 37.4 | P = 0.875 |
| Cholesterol HDL (mg/dL) (M ± SD) | 48 ± 14.6 | 53.2 ± 16.6 | P = 0.396 |
| Cholesterol LDL (mg/dL) (M ± SD) | 114 ± 46.3 | 102.5 ± 22.4 | P = 0.581 |
| Body mass index (kg/m2) | 21 ± 2.2 | 19 ± 2.7 | P = 0.497 |
| Framingham score (%) | 7.1 ± 2.8 | 3.2 ± 2.1 | P = 0.694 |
M: mean; SD: standard deviation; ART: antiretroviral therapy; PI: protease inhibitor; NNRTI: nonnucleoside reverse-transcriptase inhibitor; CMV: cytomegalovirus; SBP: cystolic blood pressure; DBP: diastolic blood pressure; HDL: high density lipoprotein; LDL: low density lipoprotein.
3.2. Carotid Intima-Media Thickness
c-IMT was higher in HIV+ than in controls (mean ± SD: 0.85 ± 0.17 versus 0.28 ± 0.24 mm; median values: 0.835 versus 0.475 mm, P < 0.001). Among HIV+ patients, 52/94 (55.3%) demonstrated a normal c-IMT, whereas 42/94 (44.7%) had a pathological c-IMT.
3.3. Immune Activation
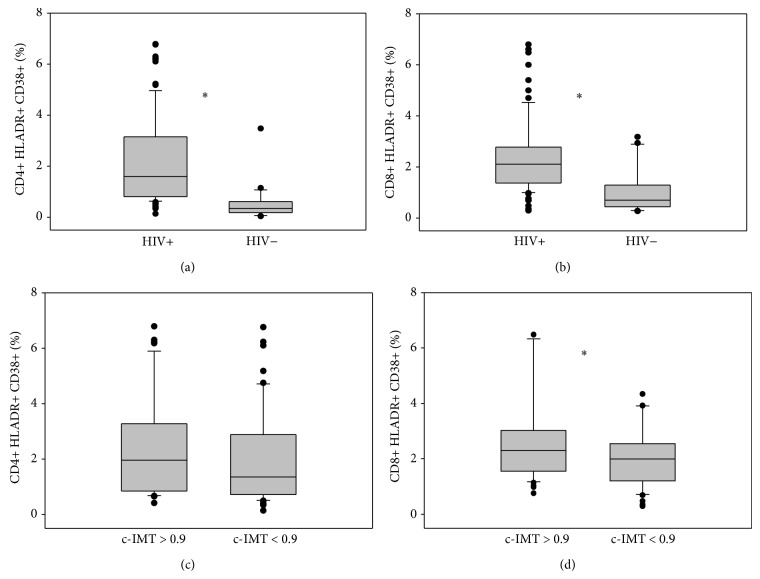
In comparison to HIV-negative patients, HIV-positive subjects exhibited higher levels of lymphocytes T CD4+ expressing HLADR+ CD38+ (median values 1.6% versus 0.34%, P < 0.001) and CD8+ HLADR+ CD38+ (median values 2.1% versus 0.69%, P < 0.001). Patients with pathological c-IMT had higher expression of lymphocytes T CD4+ and CD8+ HLADR+ CD38+ (median values 1.96% versus 1.35%, P = 0.124; median values 2.27% versus 1.99%, P = 0.048, resp.) than patients with normal c-IMT (Figure 1).
Figure 1.
CD4+ (a) and CD8+ (b) immune activation in HIV-infected subjects compared with HIV-negative controls. CD4+ (c) and CD8+ (d) immune activation in HIV-infected subjects according to c-IMT. Values are expressed as percentage. Horizontal bars represent median. Upper and lower whisker mean third quartile +1.5 (interquartile range (IQR)) and first quartile-1.5(IQR). *It represents a P value < 0.05.
3.4. Immunosenescence
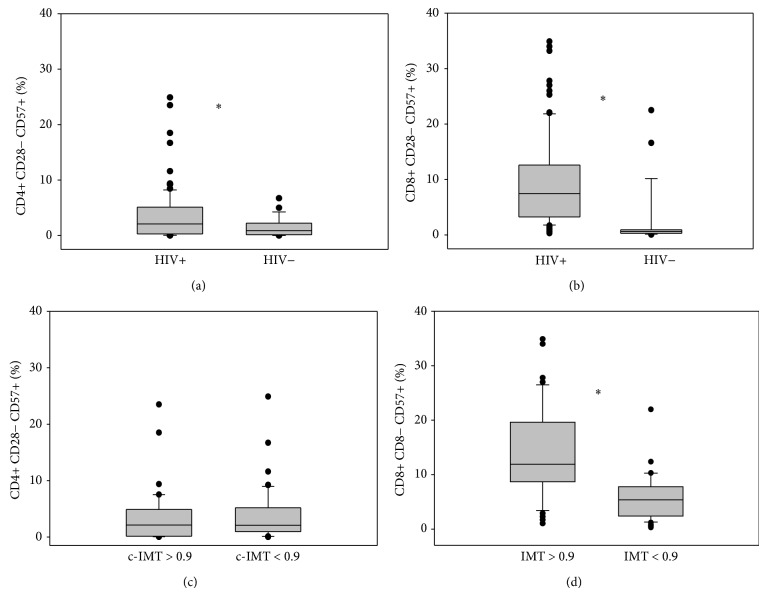
HIV-positive subjects exhibited a higher level of lymphocytes T CD4+ and CD8+ CD57+ CD28− (median values 2.08% versus 0.87%, P = 0.026; median values 7.44% versus 0.62%, P < 0.001, resp.) compared with HIV-negative patients. Patients with pathological c-IMT had higher expression of lymphocytes T CD4+ and CD8+ CD57+ CD28− (median values 2.1% versus 2%, P = 0.442; median values 11.9% versus 4.8%, P < 0.001, resp.) than patients with normal c-IMT (Figure 2). In linear regression analysis, there was a positive correlation between lymphocytes T CD8+ HLADR+ CD38+ and CD8+ CD57+ CD28− (P = 0.002). Moreover, there was a negative correlation between lymphocytes T CD4+ nadir and lymphocytes T CD4+ and T CD8+ HLADR+ CD38+ (P = 0.007 and P = 0.087, resp.). The same trend was observed for lymphocytes T CD4+ nadir and lymphocytes T CD4+ CD57+ CD28− and CD8+ CD57+ CD28−.
Figure 2.
CD4+ (a) and CD8+ (b) immunosenescence in HIV-infected subjects compared with HIV-negative controls. CD4+ (c) and CD8+ (d) immunosenescence in HIV-infected subjects according to c-IMT. Values are expressed as percentage. Horizontal bars represent median. Upper and lower whisker mean third quartile +1.5 (interquartile range (IQR)) and first quartile-1.5(IQR). *It represents a P value < 0.05.
3.5. Osteoprotegerin and Interleukin-6
OPG plasma levels were significantly higher in HIV-infected patients than in healthy controls (mean ± SD: 6.59 ± 4 pmol/L versus 3.57 ± 1.6 pmol/L; median: 5.6 versus 3.5 pmol/L) (P < 0.001) (Figure 3(a)). IL-6 plasma levels were higher in HIV-positive subjects than in HIV-negative individuals (mean ± SD 49.6 ± 29.4 versus 37.4 ± 8 pg/mL; median: 44.3 versus 36.5 pg/mL) (P = 0.096). Patients with pathological c-IMT had higher OPG plasma levels (median values 6.22 pmol/L versus 4.86 pmol/L, P = 0.05) (Figure 3(b)) and IL-6 plasma levels (median values 46 pg/mL versus 40.7 pg/mL, P = 0.156) than patients with normal c-IMT. Moreover, lymphocytes T CD8+ HLADR+ CD38+ positively correlated with OPG plasma level (P = 0.008). Although not statistically significant, lymphocytes T CD4+ HLADR+ CD38+, CD4+ CD57+ CD28− and CD8+ CD57+ CD28− were associated with higher OPG and IL-6 plasma levels.
Figure 3.
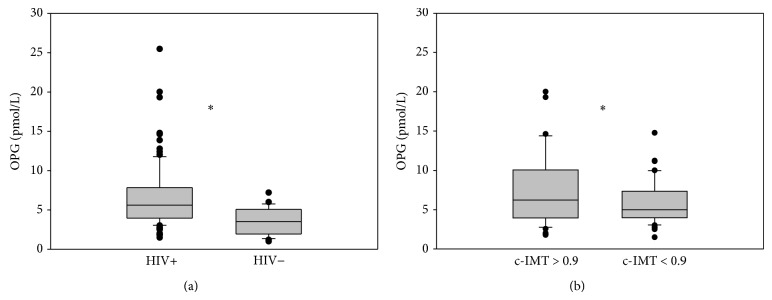
OPG plasma levels in HIV-infected subjects compared with HIV-negative controls (a) and in HIV-infected subjects according to c-IMT (b). Values are expressed as pmol/L. Horizontal bars represent median. Upper and lower whisker mean third quartile +1.5 (interquartile range (IQR)) and first quartile-1.5(IQR). *It represents a P value < 0.05.
3.6. Univariate and Multivariate Analysis
The univariate model revealed a significant association between c-IMT and age (P < 0.001), time of diagnosis (P = 0.055), lymphocytes T CD8+ CD57+ CD28− (P < 0.001), lymphocytes T CD4+ HLADR+ CD38+ (P = 0.084), and an inverse correlation between c-IMT and lymphocytes T CD4+ nadir (P = 0.008). Moreover, HIV+ subjects with pathological c-IMT had lymphocytes T CD4+ nadir lower than HIV+ subjects with normal c-IMT (P = 0.032). However, when these data were analyzed by multivariable logistic regression, only age (OR = 1.008; confidence interval CI = 1.005–1.011; P < 0.001), PI exposure (OR = 1.050; confidence interval CI = 0.992–1.111; P = 0.009), lymphocytes T CD8+ HLADR+ CD38+ (OR = 1.234; CI = 1.006–1.042; P = 0.011), and lymphocytes T CD8+ CD57+ CD28− (OR = 1.008; CI = 1.004–1.011; P < 0.001) were confirmed to be independently associated with c-IMT (Table 2).
Table 2.
Multivariate analysis.
| OR | 95% CI | P value | |
|---|---|---|---|
| Age | 1.008 | 1.005–1.011 | <0.001 |
| Sex | 1.045 | 0.980–1.115 | 0.185 |
| Smoke | 1.002 | 0.947–1.060 | 0.942 |
| Time of diagnosis | 1.002 | 0.998–1.005 | 0.305 |
| PI exposure | 1.050 | 0.992–1.111 | 0.009 |
| Cholesterol total | 1.000 | 0.999–1.003 | 0.934 |
| Cholesterol HDL | 1.000 | 0.997–1.003 | 0.952 |
| Cholesterol LDL | 1.000 | 0.999–1.001 | 0.651 |
| Triglycerides | 0.999 | 0.999-1.000 | 0.172 |
| OPG | 0.998 | 0.990–1.006 | 0.632 |
| IL-6 | 1.000 | 0.999–1.001 | 0.744 |
| CD4+ nadir | 0.999 | 0.999-1.000 | 0.264 |
| CD4+ current | 1.000 | 0.998–1.001 | 0.478 |
| CD4+ HLADR+ CD38+ | 1.004 | 0.985–1.023 | 0.692 |
| CD8+ HLADR+ CD38+ | 1.234 | 1.006–1.042 | 0.011 |
| CD4+ CD28− CD57+ | 0.993 | 0.987–1.000 | 0.063 |
| CD8+ CD28− CD57+ | 1.008 | 1.004–1.011 | <0.001 |
OR: odd ratio; CI: confidence interval; PI: protease inhibitor; HDL: high density lipoprotein; LDL: low density lipoprotein OPG: osteoprotegerin; IL-6: interleukin-6.
4. Discussion
Cardiovascular disease, as already known, occurs at an earlier age in HIV-infected than in uninfected subjects [1, 20]. An important aspect of this premature aging is the immune activation and the consequent immunosenescence that causes a thymic involution, a reduced circulating naive T-cells, an increased number of CD4+ well-differentiated CD28− T cells, and an increased level of proinflammatory cytokines (IL-6, TNFα) [4, 18, 21–25]. Recent studies have introduced the hypothesis that chronic inflammation and immune activation can contribute to the initiation and progression of atherosclerosis in the setting of HIV infection [26–30]. Moreover, it has been widely described that ART is implicated in the atherosclerosis process, and, in particular PI based regimens [31]. In this study, we evaluated the relationship between lymphocytes T-cell phenotype and IL-6 and OPG plasma levels and c-IMT in HIV-positive patients on ART. We enrolled 118 patients, 94 HIV-infected subjects on ART since 48 weeks with undetectable viremia (<37 copies/mL) and low cardiovascular diseases risk defined by a Framingham score <10% and 24 HIV-negative individuals matched for age, gender, and Framingham score. We observed an increased c-IMT in HIV-positive patients than in healthy controls and, among HIV+ patients, 42/94 (44.7%) had pathological c-IMT (≥0.9 mm). Several studies showed that the measurement of c-IMT has been correlated with the extent of coronary atherosclerosis and that the increase of c-IMT is predictive of future CVD events. c-IMT is strongly associated with the traditional risk factors such as male sex, ageing, overweight, high blood cholesterol, diabetes and insulin resistance, smoking, and, in particular, elevated blood pressure, probably due to media hypertrophy [16]. New or emerging risk factors such as various lipoproteins, psychosocial status, plasma viscosity, and hyperhomocysteinemia have also been associated with c-IMT values. In the setting of HIV infection, additional factors play a role in the pathogenesis and progression of atherosclerosis where HIV itself is one of the major players. HIV determines a state of chronic inflammation with activation and progressive aging of the immune system [4, 23–25]. Recently, some authors have also suggested an association between T-cell activation/senescence and markers of subclinical carotid artery disease, even among patients on stable ART [9, 30]. In this paper, we observed a higher level of activated and senescent CD4+ and CD8+ lymphocytes T in HIV-positive subjects than in general population, although ART provides full suppression of HIV viremia. Moreover, HIV subjects with pathological c-IMT showed levels of immune activation and immunosenescence higher than HIV-subjects with normal c-IMT. The role of inflammation and endothelial activation/dysfunction in the development of atherosclerosis has been extensively studied in the general population and several markers, such as VCAM-1, ICAM-1, and von Willebrand factor antigen, have been shown to reliably indicate the increased activation of endothelial cells in atherosclerosis. TNF-α has been implicated in myocardial dysfunction resulting from acute coronary syndrome and high levels of IL-1 and IL-6 have been associated with subclinical atherosclerosis [9, 10]. Furthermore, new soluble markers including OPG have been shown to be involved in the development and progression of atherosclerosis [32–34]. The OPG/RANK/RANKL system, member of TNF superfamily and mostly implicated in bone remodelling, is involved in immune and in vascular system [35–38]. In fact, RANKL, which is expressed by osteoblast cells and their precursor, activates its receptor (RANK), expressed by osteoclast cells and their precursor, thus promoting osteoclast formation, activation, and prolonging osteoclast survival. The effects of RANKL are blocked by the secretory glycoprotein OPG, which acts as a decoy receptor for RANKL. Changes in the RANKL/OPG ratio are critical in the pathogenesis of bone disease. The relationship between bone and vascular disease is known; in this contest, OPG could be considered as a bridge from bone to vascular system. The role of OPG in cardiovascular disease is still debated. OPG might contribute to endothelial dysfunction by blocking RANKL signalling which is able to activate protective intracellular endothelial pathways such as the nitric oxide synthase pathway, to increase the adhesion and migration of inflammatory cells through the endothelium and the activity of metalloproteases [32, 39, 40]. In a previous study, we found higher OPG plasma levels in HIV-positive subjects than in healthy controls, suggesting an association between OPG plasma levels and cardiovascular disease. The increased OPG plasma concentration found in HIV-positive patients with low cardiovascular risk may suggest that OPG is implicated in the early phase of atherosclerosis development process. We showed that OPG plasma concentrations are associated with atherosclerosis in HIV-infected subjects with a low Framingham score. Therefore, OPG plasma measurement could be a useful and noninvasive tool in clinical practice in order to early discriminate subjects at risk of developing atherosclerosis [13]. Although in the multivariate analysis OPG has not been found to be an independent factor associated with c-IMT, OPG plasma levels were significantly higher in HIV-infected patients than in healthy controls (P < 0.001) and patients with pathological c-IMT had higher OPG plasma levels than HIV-positive subjects with normal c-IMT (P = 0.05). Moreover, OPG plasma levels were strictly correlated with lymphocytes T CD8+ HLADR+ CD38+, supporting the relationship with immune activation, cytokine production, and atherosclerosis. Thus, our hypothesis is that the high levels of immune activation and immunosenescence of CD8 T cells might influence the production of OPG leading to increased c-IMT in HIV-positive subjects. The measurement of OPG plasma levels together with CD8 T cells immune activation and immunosenescence might be a useful and noninvasive parameter in order to identify early atherosclerosis in HIV-positive subjects with low cardiovascular risk.
5. Conclusions
Several factors have been implicated in the pathogenesis of atherosclerosis in HIV patients. Age, PI exposure, and lymphocytes T CD8+ HLADR+ CD38+ and T CD8+ CD57+ CD28− were confirmed to be independently associated with c-IMT. In conclusion, the immune activation and immunosenescence of CD8+ T cells together with OPG plasma levels might be associated with the development and progression of early atherosclerosis in HIV-infected patients, even in the case of viral suppression.
Supplementary Material
The linear regression analyses showed that c-IMT positively correlated with age, time of diagnosis, immuneactivated and immunosenescent CD8+ T cells and negatively correlated with CD4+ nadir T cells. Immuneactivated CD8+ T cells positively correlated with immunosenescent CD8+ T cells and OPG plasma levels. CD4+ nadir T cells negatively correlated with immuneactivated CD4+ and CD8+ T cells.
Acknowledgment
The project described was supported by the Department of Public Health and Infectious Disease of “Sapienza” University of Rome.
Conflict of Interests
None of the authors have any conflict or potential conflict of interests.
References
- 1.Jeong S. J., Kim H. W., Ku N. S., Han S. H., Kim C. O., Choi J. Y., Song Y. G., Kim J. M. Clinical factors associated with carotid plaque and intima-medial thickness in HIV-infected patients. Yonsei Medical Journal. 2013;54(4):990–998. doi: 10.3349/ymj.2013.54.4.990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Hakeem A., Bhatti S., Cilingiroglu M. The spectrum of atherosclerotic coronary artery disease in HIV patients. Current Atherosclerosis Reports. 2010;12(2):119–124. doi: 10.1007/s11883-010-0089-4. [DOI] [PubMed] [Google Scholar]
- 3.D'Ettorre G., Francone M., Mancone M., Ceccarelli G., Ascarelli A., Vullo F., Baroncelli S., Galluzzo M. C., Catalano C., Strano S., Fedele F., Mastroianni C., Palmisano L., Vullo V. Significant coronary stenosis detected by coronary computed angiography in asymptomatic HIV infected subjects. Journal of Infection. 2012;64(1):82–88. doi: 10.1016/j.jinf.2011.09.007. [DOI] [PubMed] [Google Scholar]
- 4.Deeks S. G. HIV infection, inflammation, immunosenescence, and aging. Annual Review of Medicine. 2011;62:141–155. doi: 10.1146/annurev-med-042909-093756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Brenchley J. M., Schacker T. W., Ruff L. E., Price D. A., Taylor J. H., Beilman G. J., Nguyen P. L., Khoruts A., Larson M., Haase A. T., Douek D. C. CD4+ T cell depletion during all stages of HIV disease occurs predominantly in the gastrointestinal tract. Journal of Experimental Medicine. 2004;200(6):749–759. doi: 10.1084/jem.20040874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Brenchley J. M., Price D. A., Schacker T. W., Asher T. E., Silvestri G., Rao S., Kazzaz Z., Bornstein E., Lambotte O., Altmann D., Blazar B. R., Rodriguez B., Teixeira-Johnson L., Landay A., Martin J. N., Hecht F. M., Picker L. J., Lederman M. M., Deeks S. G., Douek D. C. Microbial translocation is a cause of systemic immune activation in chronic HIV infection. Nature Medicine. 2006;12(12):1365–1371. doi: 10.1038/nm1511. [DOI] [PubMed] [Google Scholar]
- 7.Le Saux S., Weyand C. M., Goronzy J. J. Mechanisms of immunosenescence: lessons from models of accelerated immune aging. Annals of the New York Academy of Sciences. 2012;1247(1):69–82. doi: 10.1111/j.1749-6632.2011.06297.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Coll B., Parra S., Alonso-Villaverde C., Aragonés G., Montero M., Camps J., Joven J., Masana L. The role of immunity and inflammation in the progression of atherosclerosis in patients with HIV infection. Stroke. 2007;38(9):2477–2484. doi: 10.1161/STROKEAHA.106.479030. [DOI] [PubMed] [Google Scholar]
- 9.Merlini E., Luzi K., Suardi E., Barassi A., Cerrone M., Martínez J. S., Bai F., D'Eril G. V. M., Monforte A. D., Marchetti G. T-cell phenotypes, apoptosis and inflammation in HIV+ patients on virologically effective cART with early atherosclerosis. PLoS ONE. 2012;7(9) doi: 10.1371/journal.pone.0046073.e46073 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Guzmán-Fulgencio M., Medrano J., Rallón N., Echeverria-Urabayen A., Miguel Benito J., Restrepo C., García-Álvarez M., Vispo E., San Roman J., Sánchez-Piedra C., Soriano V., Resino S. Soluble markers of inflammation are associated with Framingham scores in HIV-infected patients on suppressive antiretroviral therapy. Journal of Infection. 2011;63(5):382–390. doi: 10.1016/j.jinf.2011.08.006. [DOI] [PubMed] [Google Scholar]
- 11.Biasillo G., Leo M., della Bona R., Biasucci L. M. Inflammatory biomarkers and coronary heart disease: from bench to bedside and back. Internal and Emergency Medicine. 2010;5(3):225–233. doi: 10.1007/s11739-010-0361-1. [DOI] [PubMed] [Google Scholar]
- 12.Worm S. W., Hsue P. Role of biomarkers in predicting CVD risk in the setting of HIV infection? Current Opinion in HIV and AIDS. 2010;5(6):467–472. doi: 10.1097/COH.0b013e32833f2ea6. [DOI] [PubMed] [Google Scholar]
- 13.D'Abramo A., D'Agostino C., Oliva A., et al. Early atherosclerosis in HIV infected subjects on suppressive antiretroviral treatment: role of osteoprotegerin. ISRN AIDS. 2013;2013 doi: 10.1155/2013/737083.737083 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Hofbauer L. C., Schoppet M. Clinical implications of the osteoprotegerin/RANKL/RANK system for bone and vascular diseases. The Journal of the American Medical Association. 2004;292(4):490–495. doi: 10.1001/jama.292.4.490. [DOI] [PubMed] [Google Scholar]
- 15.Ssinabulya I., Kayima J., Longenecker C., Luwedde M., Semitala F., Kambugu A., Ameda F., Bugeza S., McComsey G., Freers J., Nakanjako D. Subclinical atherosclerosis among HIV-infected adults attending HIV/AIDS care at two large ambulatory HIV clinics in Uganda. PLoS ONE. 2014;9(2) doi: 10.1371/journal.pone.0089537.e89537 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Lee C. J., Park S. The role of carotid ultrasound for cardiovascular risk stratification beyond traditional risk factors. Yonsei Medical Journal. 2014;55(3):551–557. doi: 10.3349/ymj.2014.55.3.551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kestens L., Vanham G., Gigase P., Young G., Hannet I., Vanlangendonck F., Hulstaert F., Bach B. A. Expression of activation antigens, HLA-DR and CD38, on CD8 lymphocytes during HIV-1 infection. AIDS. 1992;6(8):793–797. doi: 10.1097/00002030-199208000-00004. [DOI] [PubMed] [Google Scholar]
- 18.Effros R. B. Loss of CD28 expression on T lymphocytes: a marker of replicative senescence. Developmental and Comparative Immunology. 1997;21(6):471–478. doi: 10.1016/S0145-305X(97)00027-X. [DOI] [PubMed] [Google Scholar]
- 19.Mancia G., Fagard R., Narkiewicz K., et al. 2013 Practice guidelines for the management of arterial hypertension of the European Society of Hypertension (ESH) and the European Society of Cardiology (ESC): ESH/ESC Task Force for the Management of Arterial Hypertension. Journal of Hypertension. 2013;31(10):1925–1938. doi: 10.1097/HJH.0b013e328364ca4c. [DOI] [PubMed] [Google Scholar]
- 20.Currier J. S. Update on cardiovascular complications in HIV infection. Topics in HIV Medicine. 2009;17(3):98–103. [PubMed] [Google Scholar]
- 21.Ferrando-Martínez S., Ruiz-Mateos E., Romero-Sánchez M. C., Muñoz-Fernández M. Á., Viciana P., Genebat M., Leal M. HIV infection-related premature immunosenescence: high rates of immune exhaustion after short time of infection. Current HIV Research. 2011;9(5):289–294. doi: 10.2174/157016211797636008. [DOI] [PubMed] [Google Scholar]
- 22.Gamadia L. E., van Leeuwen E. M. M., Remmerswaal E. B. M., Yong S.-L., Surachno S., Wertheim-Van Dillen P. M. E., Ten Berge I. J. M., Van Lier R. A. W. The size and phenotype of virus-specific T cell populations is determined by repetitive antigenic stimulation and environmental cytokines. The Journal of Immunology. 2004;172(10):6107–6114. doi: 10.4049/jimmunol.172.10.6107. [DOI] [PubMed] [Google Scholar]
- 23.Hakim F. T., Gress R. E. Immunosenescence: immune deficits in the elderly and therapeutic strategies to enhance immune competence. Expert Review of Clinical Immunology. 2005;1(3):443–458. doi: 10.1586/1744666X.1.3.443. [DOI] [PubMed] [Google Scholar]
- 24.Papagno L., Spina C. A., Marchant A., Salio M., Rufer N., Little S., Dong T., Chesney G., Waters A., Easterbrook P., Dunbar P. R., Shepherd D., Cerundolo V., Emery V., Griffiths P., Conlon C., McMichael A. J., Richman D. D., Rowland-Jones S. L., Appay V. Immune activation and CD8+ T-cell differentiation towards senescence in HIV-1 infection. PLoS Biology. 2004;2(2, article e20) doi: 10.1371/journal.pbio.0020020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Tassiopoulos K., Landay A., Collier A. C., Connick E., Deeks S. G., Hunt P., Lewis D. E., Wilson C., Bosch R. CD28-negative CD4+ and CD8+ T cells in antiretroviral therapy-naive HIV-infected adults enrolled in adult clinical trials group studies. Journal of Infectious Diseases. 2012;205(11):1730–1738. doi: 10.1093/infdis/jis260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Grunfeld C., Delaney J. A., Wanke C., Currier J. S., Scherzer R., Biggs M. L., Tien P. C., Shlipak M. G., Sidney S., Polak J. F., O'Leary D., Bacchetti P., Kronmal R. A. Preclinical atherosclerosis due to HIV infection: carotid intima-medial thickness measurements from the FRAM study. AIDS. 2009;23(14):1841–1849. doi: 10.1097/QAD.0b013e32832d3b85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Lichtner M., Cuomo M. R., Rossi R., Strano S., Massetti A. P., Mastroianni C. M., Vullo V. Increased carotid intima media thickness is associated with depletion of circulating myeloid dendritic cells in HIV-infected patients on suppressive antiretroviral treatment. Atherosclerosis. 2009;204(2):e1–e3. doi: 10.1016/j.atherosclerosis.2008.12.025. [DOI] [PubMed] [Google Scholar]
- 28.Lorenz M. W., Stephan C., Harmjanz A., Staszewski S., Buehler A., Bickel M., von Kegler S., Ruhkamp D., Steinmetz H., Sitzer M. Both long-term HIV infection and highly active antiretroviral therapy are independent risk factors for early carotid atherosclerosis. Atherosclerosis. 2008;196(2):720–726. doi: 10.1016/j.atherosclerosis.2006.12.022. [DOI] [PubMed] [Google Scholar]
- 29.Melander O., Newton-Cheh C., Almgren P., Hedblad B., Berglund G., Engström G., Persson M., Smith J. G., Magnusson M., Christensson A., Struck J., Morgenthaler N. G., Bergmann A., Pencina M. J., Wang T. J. Novel and conventional biomarkers for prediction of incident cardiovascular events in the community. The Journal of the American Medical Association. 2009;302(1):49–57. doi: 10.1001/jama.2009.943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Ross A. C., Rizk N., O'Riordan M. A., Dogra V., El-Bejjani D., Storer N., Harrill D., Tungsiripat M., Adell J., McComsey G. A. Relationship between inflammatory markers, endothelial activation markers, and carotid intima-media thickness in HIV-infected patients receiving antiretroviral therapy. Clinical Infectious Diseases. 2009;49(7):1119–1127. doi: 10.1086/605578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Metkus T., Jr., Brown T. T., Post W. S. Cardiovascular disease associated with the human immunodeficiency virus: an update. Current Treatment Options in Cardiovascular Medicine. 2014;16(11):346. doi: 10.1007/s11936-014-0346-x. [DOI] [PubMed] [Google Scholar]
- 32.van Campenhout A., Golledge J. Osteoprotegerin, vascular calcification and atherosclerosis. Atherosclerosis. 2009;204(2):321–329. doi: 10.1016/j.atherosclerosis.2008.09.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Hwang J. J., Wei J., Abbara S., Grinspoon S. K., Lo J. Receptor activator of nuclear factor-κB ligand (RANKL) and its relationship to coronary atherosclerosis in HIV patients. Journal of Acquired Immune Deficiency Syndromes. 2012;61(3):359–363. doi: 10.1097/QAI.0b013e31826a6c16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Kiechl S., Werner P., Knoflach M., Furtner M., Willeit J., Schett G. The osteoprotegerin/RANK/RANKL system: a bone key to vascular disease. Expert Review of Cardiovascular Therapy. 2006;4(6):801–811. doi: 10.1586/14779072.4.6.801. [DOI] [PubMed] [Google Scholar]
- 35.Boyce B. F., Xing L. Functions of RANKL/RANK/OPG in bone modeling and remodeling. Archives of Biochemistry and Biophysics. 2008;473(2):139–146. doi: 10.1016/j.abb.2008.03.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Reid P., Holen I. Pathophysiological roles of osteoprotegerin (OPG) European Journal of Cell Biology. 2009;88(1):1–17. doi: 10.1016/j.ejcb.2008.06.004. [DOI] [PubMed] [Google Scholar]
- 37.Stejskal D., Bartek J., Pastorková R., Růžička V., Oral I., Horalík D. Osteoprotegerin, RANK, RANKL. Biomedical Papers. 2001;145(2):61–64. doi: 10.5507/bp.2001.013. [DOI] [PubMed] [Google Scholar]
- 38.Wright H. L., McCarthy H. S., Middleton J., Marshall M. J. RANK, RANKL and osteoprotegerin in bone biology and disease. Current Reviews in Musculoskeletal Medicine. 2009;2(1):56–64. doi: 10.1007/s12178-009-9046-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Schoppet M., Sattler A. M., Schaefer J. R., Herzum M., Maisch B., Hofbauer L. Increased osteoprotegerin serum levels in men with coronary artery disease. The Journal of Clinical Endocrinology & Metabolism. 2003;88(3):1024–1028. doi: 10.1210/jc.2002-020775. [DOI] [PubMed] [Google Scholar]
- 40.Vik A., Mathiesen E. B., Brox J., Wilsgaard T., Njølstad I., Jørgensen L., Hansen J.-B. Serum osteoprotegerin is a predictor for incident cardiovascular disease and mortality in a general population: the Tromsø study. Journal of Thrombosis and Haemostasis. 2011;9(4):638–644. doi: 10.1111/j.1538-7836.2011.04222.x. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
The linear regression analyses showed that c-IMT positively correlated with age, time of diagnosis, immuneactivated and immunosenescent CD8+ T cells and negatively correlated with CD4+ nadir T cells. Immuneactivated CD8+ T cells positively correlated with immunosenescent CD8+ T cells and OPG plasma levels. CD4+ nadir T cells negatively correlated with immuneactivated CD4+ and CD8+ T cells.