Abstract
Rose hip powder (RHP) alleviates osteoarthritis (OA) due to its anti-inflammatory and cartilage-protective properties. Substances contained in RHP might contribute to its clinical efficacy. The activity of two RHP (i.e., RH-A, from the whole fruit, RH-B, from fruits without seeds) was investigated in human peripheral blood leukocytes (PBL) and primary chondrocytes (NHAC-kn). RH-A and RH-B diminished the secretion of chemokines and cytokines in LPS/IFN-γ-activated PBL, including CCL5/RANTES, CXCL10/IP-10, interleukin- (IL-) 6, and IL-12. Most effects were transcriptional, since gene expression levels were significantly influenced by RH-A and RH-B. In IL-1β treated normal chondrocytes (NHAC-kn), both RH preparations reduced the expression of matrix metalloproteinase- (MMP-) 1, MMP-3, and MMP-13 and ADAMTS-4. These changes are associated with diminished inflammatory damage or cartilage erosion. Principal component analysis revealed that (1) RH-A and RH-B modified a large pattern of biomarkers, and (2) RH-B outperformed RH-A. Furthermore, RH-B contained more chondroprotective and anti-inflammatory constituents than RH-A. Thus, RHP contributed to restore cellular homeostasis in PBL and chondrocytes. RH preparations from fruits without seeds are thus expected to have an improved OA-preventive or OA-therapeutic profile, as subsequently shown in a related clinical trial.
1. Introduction
Osteoarthritis (OA) reflects the degradation and erosion of the extracellular matrix (ECM) and the subsequent narrowing of space in joints. The changes in ECM structure are due to the activation of enzymatic systems, that is, matrix metalloproteinase (MMPs) and aggrecanase in chondrocytes and synoviocytes [1, 2]. The proinflammatory interleukin- (IL-) 1β has a key role in inducing the OA phenotype in chondrocytes [3]. Likewise, nitric oxide (NO) production also correlates with pathophysiological changes in chondrocytes [4–8]. IL-1β-activated chondrocytes produce a variety of chemokines [9–12]; this might reflect the implication of cell recruitment during inflammatory processes in OA diseases.
Natural substances reportedly attenuate or delay the onset and progression of OA. Glucosamine and chondroitin have been the most promising substances so far identified, although their effectiveness is a matter of debate. Clinical studies have demonstrated a beneficial effect of rose hip powder (RHP) in the treatment of OA [13–15] (for reviews see [16, 17]). One of its constituents, galacto-lipid (2S)-1, 2-di-O-[(9Z, 12Z, 15Z)-octadeca-9, 12, 15-trienoyl]-3-O-β-D-galacto-pyranosyl glycerol (GLGPG), has been found to inhibit chemotaxis of neutrophils [13, 14] and thus could impair pathophysiological cell recruitment to OA lesions. Other RHP constituents such as ascorbic acid, polyphenols, flavonoids, and unsaturated fatty acids might contribute to alleviate OA mainly via their anti-inflammatory properties. Indeed, RHP extracts and unsaturated fatty acids inhibited cyclooxygenase- (COX-) 1 and COX-2 activity [18, 19] and associated PGE2 production [20]. In a previous in vitro study, the multiple effects of RHP on the production of inflammatory mediators by peripheral blood leukocyte and anabolic and catabolic processes in chondrocytes have been described [20]. The current study aimed at the identification of biological activities of different parts of the rose hip fruit and an improved use of rose hip preparations in the management of OA conditions.
2. Materials and Methods
2.1. Rose Hip Preparations and Reagents
RHP was prepared from Rosa canina and provided by Axellus, Ishøj, Denmark; RH-A consists of dried rose hip powder as described previously [20]; RH-B was prepared from dried rose hip, where the seeds had been removed before the preparation of the powder. The contents in main constituents (see Table 1) have been measured by standard procedures implemented at the Analytical Research Center, DSM Nutritional Products, Kaiseraugst (Switzerland). Briefly, betulinic acid, oleanolic acid, and ursolic acid were determined according to validated in-house methods (available on request); vitamin C and vitamin E were analyzed according to official methods EN14130 and EN12822, respectively; linoleic acid, EPA, and DHA were measured according to the official method of ISO 12966-2. RHP solutions were prepared in DMSO and added to the culture medium concomitantly with the stimulating agent. E. coli lipopolysaccharide (LPS, serotype 055:B5) and fetal bovine serum (FBS) were from Sigma/Aldrich (Saint-Louis, MO). RPMI 1640, DMEM, 2-mercaptoethanol, and MEM nonessential amino acids (NEAA) were from Invitrogen (Carlsbad, CA). Human IL-1β and recombinant interferon-γ (IFN-γ) were from PeproTech EC (London, UK).
Table 1.
Constituents of RH-A and RH-B.
| Constituent [mg/kg] | RH-A (with seeds) |
RH-B (without seeds) |
|---|---|---|
| GLGPG1 | 114 | 210 |
| MGDG2 | 71 | 120 |
| DGDG3 | 1220 | 1900 |
| Betulinic acid4 | 662 | 1078 |
| Betulinic, oleanolic, and ursolic acid | 1060 | 1711 |
| Vitamin C | 3146 | 3182 |
| Vitamin E | 129 | 212 |
| Total carotenoids | 190 | 250 |
| β-Carotene | 36 | 36 |
| Lycopene | 89 | 137 |
| Total fatty acid | 32810 | 7360 |
| Linoleic acid | 16390 | 2230 |
| EPA | <LOD5 | 80 |
| DHA | <LOD | 60 |
1Galacto-lipid (2S)-1, 2-di-O-[(9Z, 12Z, 15Z)-octadeca-9, 12, 15-trienoyl]-3-O-β-D-galactopyranosyl glycerol.
2Mono-galactosyl diglyceride.
3Di-galactosyl diglyceride.
4Betulinic acid, oleanolic acid, and ursolic acid were determined according to a validated in-house method; vitamin C and vitamin E were analyzed according to the official methods EN14130 and EN12822, respectively; linoleic acid, EPA, and DHA were measured according to the official method of ISO 12966-2.
5Limits of detection.
2.2. Cell Culture
Human peripheral blood leukocytes (PBL) and primary chondrocytes from healthy individuals have been cultured and treated with inflammatory stimuli as described [20–22]. Human primary cell culture protocols and in vitro peripheral blood leukocyte experiments were approved by the Swiss Federal Office of Public Health (no. A050573/2) and the Ethical Commission of the Kanton Aargau, Switzerland. PBL were obtained from healthy donors. PBL (at 8 × 106 viable cells/mL) were cultured for 12–24 h in phenol-red free RPMI 1640 (containing 0.25% FBS, 0.1 mM NEAA, 50 U/mL penicillin, 50 μg/mL streptomycin, and 50 μM 2-mercaptoethanol) and stimulated with LPS (100 ng/mL) and IFN-γ (20 U/mL) with graded amounts of test substances. Normal human articular chondrocytes from knee (NHAC-kn) were seeded into 6-well plates at 0.5 × 106 cells per well and, where indicated, activated with 10 ng/mL IL-1β in supplemented CBM (Lonza, Walkersville, MD) in the presence of graded amounts of test compounds for 4 h. In all cell cultures, vehicle (i.e., DMSO) was included at 0.5% final concentration.
For molecular analysis, NHAC-kn cells and PBLs were lysed in RLT buffer (Qiagen, Hilden, Germany) after 4 and 12 h of culture, respectively, and total RNA was extracted. For the analysis of secreted mediators and proteins, PBLs were cultured for 24 h; supernatants were collected and stored at −80°C until use for analysis.
2.3. RNA Isolation, cDNA Synthesis, and RT-PCR
The isolation of total RNA, synthesis of cDNA and quantitative RT-PCR has been performed as detailed before [20].
2.4. Multiparametric Analysis of Cytokines, Chemokines, and Interleukins
Multiparametric kits were purchased from BIO-RAD Laboratories (Hercules, CA) and used in the LiquiChip Workstation IS 200 (Qiagen, Hilden, Germany) to measure the amount of secreted proteins. Data evaluation was done using the LiquiChip Analyser software (Qiagen).
2.5. Statistical Analysis
Data were evaluated by statistical tools described previously [20]. P values < 0.05 (obtained by using Student's t test or one-way ANOVA) were considered to reflect statistically significant differences. Statistical differences between treatment groups were evaluated by the Student's t-test. Principal component analysis (PCA) was performed with the package “chemometrics” [23] of the statistical software [24] using the nipals algorithm with centered and scaled input data.
3. Results
3.1. Composition of RH-A and RH-B
RH-B, prepared from rose hip fruits after removal of seeds, displayed more potent anti-inflammatory effects. The distinct biological activity of RH-A and RH-B correlated with different contents of constituents: RH-B contained higher amounts of ursolic acid, betulinic acid, GLGPG, and DHA (Table 1). Of those, ursolic acid and betulinic acid and 3-omega PUFAs, but not β-carotene, vitamin C, or vitamin E, displayed anti-inflammatory effects (our unpublished data and [25, 26]). Removal of seeds increased the contents of α-tocopherol and GLGPG; the latter had anti-inflammatory properties [20]. Other constituents (e.g., vitamin C) were homogenously distributed in rose hip fruit parts.
3.2. Effect of RH-A and RH-B on Peripheral Blood Leukocytes
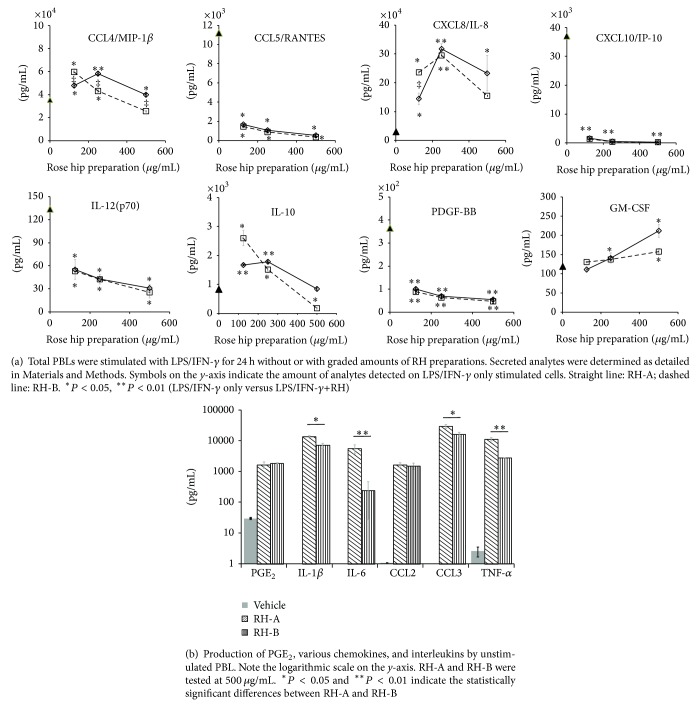
We investigated the effect of rose hip preparations on cells of the peripheral blood, which is an obligatory passage for nutritional supplements to the target tissue (i.e., cartilage). LPS/IFN-γ treatment induced the secretion of large quantities of CCL2/MCP-1, CCL3/MIP-1α, CCL4/MIP-1β, CCL5/RANTES, CXCL10/IP-10, and CXCL8/IL-8 (Table 2 and Figure 1). Rose hip preparations significantly reduced the secretion of, for example, CCL5/RANTES, and CXCL10/IP-10. Conversely, CXCL8/IL-8 and CCL3/MIP-1α secretion were increased, while other chemokines (CCL11/eotaxin, CCL2/MCP-1, and CCL4/MIP-1β) were not markedly altered. Similarly the secretion of interleukins and cytokines was influenced by RH preparations: IL-6 and IL-12(p70) were less produced in the presence of increasing RHP amounts, while IL-10, IL-1β, and TNF-α were secreted at higher levels. The secretion of CCL5/RANTES, CXCL10/IP-10, and IL-12(p70) was influenced by the lowest tested concentration, whereas changes in others (e.g., CXCL8/IL-8, CCL3/MIP-1α, and IL-6) required high RHP concentrations. RH-B had more potent effects and thus a larger impact on inflammatory mediators than its RH-A counterpart (see Table 2). It should be mentioned that secretion of PGE2, CCL2/MCP-1, CCL4/MIP-1β, and cytokines (e.g., IL-1β, IL-6 and TNF-α) was stimulated by RH-A or RH-B even in the absence of LPS/IFN-γ (Figure 1). RH-A and RH-B did not impair cell viability (data not shown; see also [20]), since some of the cellular parameters were not altered by graded amount of the substances.
Table 2.
Secretion of proteins by activated PBL. LPS/IFN-γ-stimulated cells were cultured with 125–500 μg/mL RHP for 24 h and proteins were quantified by multiparametric analysis. Only values for (LPS/IFN-γ-stimulated) cells and (500 μg/mL RHP + LPS/IFN-γ-stimulated) cells are given. Differences between RH-A and RH-B are given in the right-side column.
| Protein | LPS/IFN-γ | LPS/IFN-γ + RH-A (500 μg/mL) | LPS/IFN-γ + RH-B (500 μg/mL) | RH-A versus RH-B | ||
|---|---|---|---|---|---|---|
| pg/mL ± stdev | pg/mL ± stdev | P valuea | pg/mL ± stdev | P value | ||
| CCL11/Eotaxin | 194 ± 7 | 150 ± 16 | 0.069 | 141 ± 15 | 0.045 | NSb |
| CCL2/MCP-1 | 1136 ± 126 | 1513 ± 145 | 0.109 | 837 ± 106 | 0.123 | 0.042 |
| CCL3/MIP-1α | 4028 ± 513 | 13050 ± 919 | 0.007 | 5455 ± 495 | 0.105 | 0.010 |
| CCL4/MIP-1β | 35500 ± 1697 | 93875 ± 1167 | 0.095 | 25800 ± 7354 | 0.211 | 0.009 |
| CXCL8/IL-8 | 30600 ± 3253 | 233250 ± 61165 | 0.042 | 155125 ± 74635 | 0.142 | NS |
| CCL5/RANTES | 11188 ± 1962 | 545 ± 91 | 0.017 | 353 ± 54 | 0.016 | NS |
| CXCL10/IP-10 | 37075 ± 11208 | 266 ± 27 | 0.043 | 228 ± 35 | 0.043 | NS |
|
| ||||||
| IL-1β | 7165 ± 1520 | 37800 ± 1980 | 0.003 | 18550 ± 1697 | 0.019 | 0.021 |
| IL-6 | 59975 ± 10006 | 47000 ± 6293 | 0.261 | 6525 ± 92 | 0.017 | 0.003 |
| IL-12 (p70) | 134 ± 17 | 31 ± 5 | 0.015 | 25 ± 7 | 0.015 | NS |
| IL-10 | 778 ± 84 | 845 ± 73 | 0.487 | 179 ± 16 | 0.010 | 0.010 |
| TNF-α | 10515 ± 474 | 27325 ± 3712 | 0.024 | 6745 ± 983 | 0.039 | 0.044 |
| GM-CSF | 113 ± 10 | 212 ± 17 | 0.018 | 158 ± 8 | 0.040 | NS |
| G-CSF | 831 ± 165 | 14075 ± 2510 | 0.018 | 1272 ± 16 | 0.064 | 0.005 |
a P value: significance value between (LPS/IFN-γ) and (LPS/IFN-γ + substance) treatment.
bNS: not significant.
Figure 1.
Effect of RH on production of PGE2 and cytokines by activated peripheral blood leukocytes.
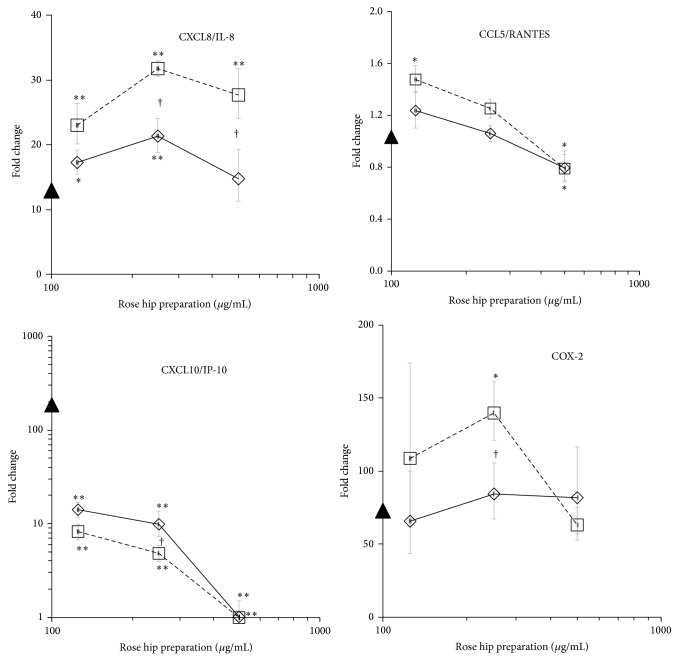
Next, we investigated whether RHP acted at the level of transcription or posttranscriptionally. Changes in gene expression levels showed a pattern which was similar to that of the respective proteins, as best illustrated for CXCL10/IP-10, IL-6, and CXCL8/IL-8 (Table 3 and Figure 2). In comparison to RH-A, the RH-B counterpart had a stronger impact on the expression of inflammatory genes. This suggests that RH-B and RH-A modulated the LPS/IFN-γ-induced changes in PBL at the level of transcription. One notable exception was CCL5/RANTES, which was released from activated PBL even though the respective gene activity was virtually unaltered. Also, IL-1β mRNA levels were decreased by RHP, whereas the cells discharged increased amounts of IL-1β (see Figure 1). RHP induced PGE2 in unstimulated PBL and increased it in LPS/IFN-γ-activated PBL (not shown; see also [20]). This might be explained by the presence of fatty acids that were converted into substrates for COX-1 in PBL.
Table 3.
Effects of RHP on gene expression in stimulated PBL (cultured for 12 h). LPS/IFN-γ-stimulated cells were cultured with 125–500 μg/mL RHP for 12 h and gene expression was quantified by RT-PCR. Fold changes were calculated as indicated in Materials and Methods. Values for (LPS/IFN-γ-stimulated) cells and (500 μg/mL RHP + LPS/IFN-γ-stimulated) cells are given.
| Gene | LPS/IFN-γ | LPS/IFN-γ + RH-A (500 μg/mL) | LPS/IFN-γ + RH-B (500 μg/mL) | RH-A versus RH-B | ||
|---|---|---|---|---|---|---|
| Fold change | Fold change | P valuea | Fold change | P value | ||
| COX-2 | 83 | 107 | 0.061 | 81 | 0.739 | NSb |
| TNF-α | 83 | 55 | 0.006 | 42 | 0.001 | NS |
| IL-1α | 3513 | 1347 | <0.001 | 859 | <0.001 | NS |
| IL-1β | 1460 | 538 | <0.001 | 410 | <0.001 | NS |
| IL-6 | 15928 | 9261 | 0.058 | 3656 | <0.001 | 0.04 |
|
| ||||||
| CCL5/RANTES | 1 | 0.8 | 0.070 | 0.8 | 0.054 | NS |
| CXCL8/IL-8 | 13 | 15 | 0.418 | 28 | <0.001 | 0.03 |
| CXCL10/IP-10 | 187 | 1 | <0.001 | <LODc | <0.001 | Not applicable |
| CXCL2/MIP-2 | 12 | 22 | 0.012 | 25 | 0.002 | NS |
| CCL20/MIP-3α | 3634 | 5454 | 0.014 | 6738 | 0.014 | NS |
a P value: significance value between (LPS/IFN-γ) and (LPS/IFN-γ + substance) treatment.
bNS: not significant.
cBelow level of detection.
Figure 2.
Effect of RH-A (straight line) and RH-B (dashed line) on gene expression in PBL. Cells were stimulated for 12 h with the indicated amount of substances and the gene expression was quantified by RT-PCR. Symbol on y-axis indicates the expression levels in LPS/IFN-γ only stimulated PBL. * P < 0.05, ** P < 0.01 (LPS/IFN-γ only versus LPS/IFN-γ+RH). † P < 0.05 and ‡ P < 0.01 indicate the differences between RH-A and RH-B.
3.3. Rose Hip Preparations Modulate Catabolic Gene Expression in Activated Normal Human Chondrocytes
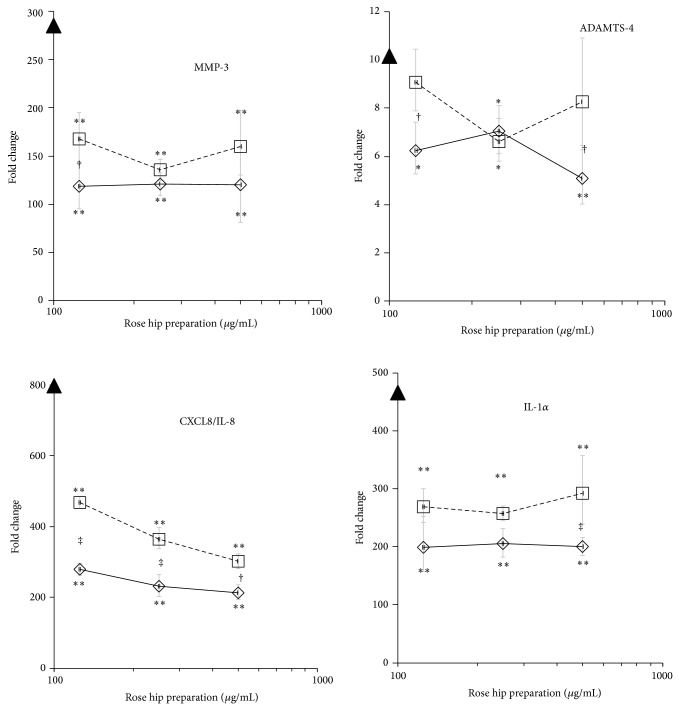
Treatment of NHAC-kn with IL-β drastically increased expression levels of catabolic and inflammatory genes which reflect the induction of OA conditions in normal chondrocytes (Table 4 and Figure 3) [12]. Within 4 h of stimulation, IL-1β significantly upregulated catabolic genes (MMP-1, MMP-3, MMP-13, and ADAMTS-4), cytokine genes (IL-1α, IL-6, and TNF-α), and chemokines (CXCL8/IL-8, CCL5/RANTES, CXCL2/MIP-2, and CXCL20/MIP-3α). RH-A concentration-dependently reduced MMP-1 and MMP-3 expression by >50%, while MMP-13 expression was diminished by 90% at the highest dose of RH-A. ADAMTS-4 mRNA levels were also reduced by ~50%. Proinflammatory cytokine genes like IL-1α and IL-6 were significantly downregulated. With regard to chemokine genes (CXCL8/IL-8, CCL5/RANTES, CXCL2/MIP-2, and CXCL20/MIP-3α), RH-A mitigated their expression levels to a similar extent at 125–500 μg/mL (up to 80% reduction). RH-B induced changes in gene expression that were comparable to those observed by RH-A, although there were quantitative differences in MMP-1, MMP-13, IL-1α, CXCL8/IL-8, and CCL-2/MIP-2: in general, RH-B exerted weaker effects than RH-A (see “fold change” in Table 4). This is in contrast to the observations made in PBL (Table 3). Notably, CCL5/RANTES gene expression was dramatically induced in IL-1β treated chondrocytes, but it was not upregulated in PBL. This observation points to cell- and tissue-specific modes of action of RHP and to distinct effects of RH-A and RH-B, respectively, in PBL and chondrocytes.
Table 4.
Effects of RHP on catabolic gene expression in human primary chondrocytes. IL-1β-stimulated cells were cultured with 125–500 μg/mL RHP for 4 h and gene expression was quantified by RT-PCR. Fold changes were calculated as indicated in Materials and Methods. Values for (IL-1β stimulated) cells and (500 μg/mL RHP + IL-1β) stimulated cells are given.
| Gene | IL-1β | IL-β + RH-A (500 μg/mL) | IL-1β + RH-B (500 μg/mL) |
P value RH-A versus RH-B |
||
|---|---|---|---|---|---|---|
| Fold change | Fold change | P valuea | Fold change | P value | ||
| MMP-1 | 28 | 16 | 0.001 | 26 | 0.620 | 0.032 |
| MMP-3 | 286 | 120 | 0.001 | 160 | 0.003 | NSb |
| MMP-13 | 1.7 | 0.2 | <0.001 | 0.5 | <0.001 | 0.040 |
| ADAMTS-4 | 10 | 5 | 0.006 | 8 | 0.295 | NS |
| CXCL2/MIP-2 | 467 | 122 | <0.001 | 180 | <0.001 | 0.091 |
| CXCL20/MIP-3α | 642 | 253 | 0.001 | 331 | 0.010 | NS |
| CCL5/RANTES | 176 | 127 | 0.008 | 151 | 0.241 | NS |
| CXCL8/IL-8 | 797 | 214 | <0.001 | 301 | <0.001 | 0.087 |
| IL-1α | 466 | 200 | 0.001 | 292 | 0.032 | NS |
| IL-6 | 1454 | 897 | 0.022 | 746 | 0.006 | NS |
| TNF-α | 1120 | 1081 | 0.727 | 1704 | 0.005 | NS |
a P values: significance value between (IL-1β) and (IL-1β + substance) treatment.
bNot significant.
Figure 3.
Effect of RH preparations on expression levels in human primary chondrocytes. NHAC-kn cells were stimulated with IL-1β for 4 h and the expression levels of indicated genes were quantified by RT-PCR. Triangles on y-axis indicate the expression levels in IL-1β only treated NHAC-kn. Straight line: RH-A; dashed line RH-B. * P < 0.05, ** P < 0.01 (IL-β only versus IL-1β + RHP). † P < 0.05 and ‡ P < 0.01 indicate the differences between RH-A and RH-B.
3.4. Comparison of Rose Hip Induced Changes of Variables in Different Cell Types
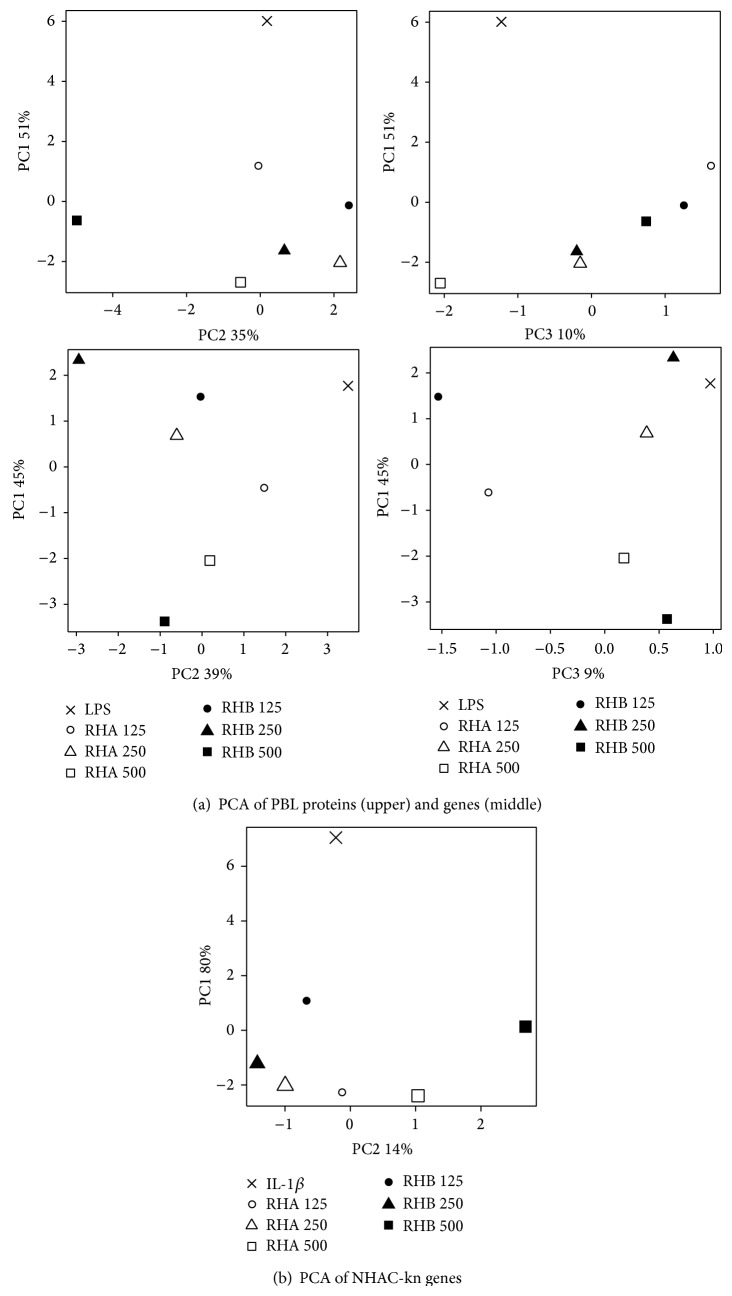
In order to comprehensively assess the rose hip induced changes in OA biomarkers, we performed principal component analyses (PCA). PCA is a dimension reduction method that allows transforming a complex highly correlated dataset (e.g., all OA biomarkers) into a much smaller set of uncorrelated variables, that is, principal components (PCs). In our experimental data set, the PCs were taken as “meta-biomarker” and derived in decreasing order of importance, the first PC explaining most of the variation (and thus representing most of the information) contained in the original dataset. Plotting the OA biomarkers in PCA space allows to visualize and interpret these changes, which is very difficult in the high-dimensional setting of before. Three different PCAs were run, illustrating the global effects of RH-A and RH-B on PBLs at the protein and gene level and on NHAC-kn gene level. The PCA were based on the expression of 17 PBL proteins, 10 PBL genes, and 14 NHAC-kn genes, respectively. At the protein level, LPS/IFN-γ-activated PBL were clearly separated from the LPS/IFN-γ-activated/RHP-treated cells showing a treatment effect on PC1. Additionally a concentration-dependent effect for RH-A could be observed for PC1 as well. Concentration-dependent effects for RH-B were mainly visualized by principal component 2 (Figure 4). At the gene level, LPS/IFN-γ-activated PBL were separated from higher dose LPS/IFN-γ-activated/RHP-treated cells within the first principal component. A similar clustering of treatments indicating a treatment effect was also observed within PC2. Additionally, RH-B exerted a more discriminating effect than RH-A within PC2. The cumulative explained variability (CEV) (explained variation of PC1 + PC2 + PC3) for PBL at the protein and gene level was 96% and 94%, respectively, with PC1 explaining 51% and 45% of the variability. For NHAC-kn, CEV was 94% for variations observed at the gene level (PC1 + PC2, PC1 by itself explaining 80%). An effect of treatment was observable and mostly reflected by PC1, whereas the effect of substance concentrations (especially contrasting the high dose from the other doses) was mainly distinguished by principal component 2. A stronger effect of RH-B as compared to RH-A could also be observed in PC1. Taken together, the data indicate a distinguishable and beneficial effect of RH on OA biomarkers with RH-B being a stronger discriminator than RH-A.
Figure 4.
Principal component analysis showing the impact of different treatments and concentrations of RH-A and RH-B in peripheral blood leukocytes (upper panels) and normal human chondrocytes (bottom panel). PBLs were treated with LPS/IFN-γ alone or LPS/IFN-γ and RHP at the indicated concentrations (in μg/mL). NHAC-kn were activated with IL-1β alone or IL-1β and RHP at the indicated concentrations.
4. Discussion
In this study, a panel of biological activities of rose hip powder prepared from the whole fruit or from shells has been compared. It provides evidence that cellular features related to inflammatory responses and cartilage destruction were modulated by the complex mixture of substances contained in RHP (see Table 1). These have been measured in murine and human cellular models and are fully consistent with those previously reported [20]. In LPS-activated murine macrophages RHP exhibited mainly anti-inflammatory properties including reduced production of nitric oxide, COX-2 dependent PGE2, various chemokines, and proinflammatory cytokines [20]. In PBL, the expression of interleukins and cytokines were mostly mitigated by RHP, while growth factor production like GM-CSF and G-CSF were increased. We observed a dichotomy with regard to chemokine expression: CCL5/RANTES, CCL11/eotaxin, and CXCL10/IP-10 drastically decreased, whereas others augmented in the presence of RHP (e.g., CXL8/IL-8, CCL3/MIP-1α, and CCL4/MIP-1β). With regard to CCL5/RANTES and CXCL8/IL-8, their cellular release was modulated by RHP, while gene expression was not altered. Therefore, RHP could contribute to improve the amplitude and length of the acute inflammatory response through a more vigorous mobilization of lymphocytes and macrophages, while it dampens an excessive cell mobilization in chronic inflammation processes like in OA. This needs to be investigated in further experiments, where RHP should be tested on distinct subpopulations of PBL such as neutrophils, monocytes, and lymphocytes.
The pattern of observed in vitro activities was more definite in the target tissue cell equivalent, that is, chondrocytes, where gene expression of CCL5/RANTES, CXCL10/IP-10, CXCL2/MIP-2, and CCL20/MIP-3α was reduced by RHP. Concomitantly, the expression of genes that degrade ECM was diminished and thus reflected a chondroprotective effect of RHP on cartilage tissue. Previous studies highlighted the importance of chemokines in OA [27], with CCL5/RANTES and CXCL8/IL-8 being activated in chondrocytes [9–11, 28]. Likewise, IL-1β activation robustly modified chemokine and chemokine receptor expression in chondrocytes [12]. The results of the present study corroborate previous data obtained with another source of RHP [20].
It is premature to unambiguously define the bioactive constituents of RH-A and RH-B, whose composition considerably differed (Table 1). Removing seeds from rose hip preparations resulted in higher contents of anti-inflammatory substances like betulinic acid, ursolic acid, galactolipids, EPA, and DHA. On the other side, seeds contained the bulk of fatty acids which could serve as substrate for inflammatory mediators such as PGE2. The multiple measured effects of RHP in different cells have been compared by PCA. These analyses demonstrated that RH-A and RH-B robustly modified a pattern or cluster of biological responses, as anticipated for beneficial effects of nutritional supplements (reviewed by, e.g., [29]). More importantly, two of the three PCAs performed (at the level of PBL and NHAC-kn genes) showed that RH-B exhibited an overall stronger effect on OA biomarkers than RH-A. Given the demonstrated health benefit of rose hip in clinical studies (for review see, e.g., [17]), we hypothesize that in clinical studies RH-B is more efficacious or can be used in lower doses than RH-A in the treatment of OA conditions. Indeed, a recently completed randomized active-controlled trial provided evidence that, taken at half the original dose, the novel enhanced rosehip powder (RH-B) had similar effects in patients with symptomatic OA as the original rosehip product (RH-A) [30].
5. Conclusions
A new form of rosehip powder has enhanced in vitro anti-inflammatory and chondroprotective properties in human peripheral blood leukocytes and primary chondrocytes.
Acknowledgments
The authors thank J. Schierle and K. Schäfer for providing data on the rosehip composition and S. Johansen for valuable comments on the paper.
Abbreviations
- IL:
Interleukin
- LPS:
Lipopolysaccharide
- NO:
Nitric oxide
- MMP:
Matrix metalloproteinase
- OA:
Osteoarthritis
- PBL:
Peripheral blood leukocytes
- PGE2:
Prostaglandin E2
- RH:
Rose hip
- RHP:
Rose hip powder.
Conflict of Interests
This research was funded by Axellus Inc. and DSM Nutritional Products, where all authors are currently employed.
Authors’ Contribution
Joseph Schwager and Nathalie Richard contributed equally to this work.
References
- 1.Goldring S. R., Goldring M. B. The role of cytokines in cartilage matrix degeneration in osteoarthritis. Clinical Orthopaedics and Related Research. 2004;(427):S27–S36. doi: 10.1097/01.blo.0000144854.66565.8f. [DOI] [PubMed] [Google Scholar]
- 2.Martel-Pelletier J., Boileau C., Pelletier J.-P., Roughley P. J. Cartilage in normal and osteoarthritis conditions. Best Practice and Research: Clinical Rheumatology. 2008;22(2):351–384. doi: 10.1016/j.berh.2008.02.001. [DOI] [PubMed] [Google Scholar]
- 3.Kapoor M., Martel-Pelletier J., Lajeunesse D., Pelletier J.-P., Fahmi H. Role of proinflammatory cytokines in the pathophysiology of osteoarthritis. Nature Reviews Rheumatology. 2011;7(1):33–42. doi: 10.1038/nrrheum.2010.196. [DOI] [PubMed] [Google Scholar]
- 4.Abramson S. B., Attur M., Amin A. R., Clancy R. Nitric oxide and inflammatory mediators in the perpetuation of osteoarthritis. Current Rheumatology Reports. 2001;3(6):535–541. doi: 10.1007/s11926-001-0069-3. [DOI] [PubMed] [Google Scholar]
- 5.Amin A. R., Di Cesare P. E., Vyas P., Attur M., Tzeng E., Billiar T. R., Stuchin S. A., Abramson S. B. The expression and regulation of nitric oxide synthase in human osteoarthritis-affected chondrocytes: evidence for up-regulated neuronal nitric oxide synthase. Journal of Experimental Medicine. 1995;182(6):2097–2102. doi: 10.1084/jem.182.6.2097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Clancy R. M., Gomez P. F., Abramson S. B. Nitric oxide sustains nuclear factor kappaB activation in cytokine-stimulated chondrocytes. Osteoarthritis and Cartilage. 2004;12(7):552–558. doi: 10.1016/j.joca.2004.04.003. [DOI] [PubMed] [Google Scholar]
- 7.Goldring M. B., Berenbaum F. The regulation of chondrocyte function by proinflammatory mediators: prostaglandins and nitric oxide. Clinical Orthopaedics and Related Research. 2004;(427):S37–S46. doi: 10.1097/01.blo.0000144484.69656.e4. [DOI] [PubMed] [Google Scholar]
- 8.Vuolteenaho K., Moilanen T., Knowles R. G., Moilanen E. The role of nitric oxide in osteoarthritis. Scandinavian Journal of Rheumatology. 2007;36(4):247–258. doi: 10.1080/03009740701483014. [DOI] [PubMed] [Google Scholar]
- 9.Alaaeddine N., Di Battista J. A., Pelletier J.-P., Kiansa K., Cloutier J.-M., Martel-Pelletier J. Differential effects of IL-8, LIF (pro-inflammatory) and IL-11 (anti-inflammatory) on TNF-α-induced PGE2 release and on signalling pathways in human OA synovial fibroblasts. Cytokine. 1999;11(12):1020–1030. doi: 10.1006/cyto.1999.0505. [DOI] [PubMed] [Google Scholar]
- 10.Alaaeddine N., Olee T., Hashimoto S., Creighton-Achermann L., Lotz M. Production of the chemokine RANTES by articular chondrocytes and role in cartilage degradation. Arthritis & Rheumatology. 2001;44(7):1633–1643. doi: 10.1002/1529-0131(200107)44:7<1633::AID-ART286>3.0.CO;2-Z. [DOI] [PubMed] [Google Scholar]
- 11.Pulai J. I., Chen H., Im H.-J., Kumar S., Hanning C., Hegde P. S., Loeser R. F. NF-κB mediates the stimulation of cytokine and chemokine expression by human articular chondrocytes in response to fibronectin fragments. Journal of Immunology. 2005;174(9):5781–5788. doi: 10.4049/jimmunol.174.9.5781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Sandell L. J., Xing X., Franz C., Davies S., Chang L.-W., Patra D. Exuberant expression of chemokine genes by adult human articular chondrocytes in response to IL-1β . Osteoarthritis and Cartilage. 2008;16(12):1560–1571. doi: 10.1016/j.joca.2008.04.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kharazmi A., Winther K. Rose hip inhibits chemotaxis and chemiluminescence of human peripheral blood neutrophils in vitro and reduces certain inflammatory parameters in vivo. Inflammopharmacology. 1999;7(4):377–386. doi: 10.1007/s10787-999-0031-y. [DOI] [PubMed] [Google Scholar]
- 14.Larsen E., Kharazmi A., Christensen L. P., Christensen S. B. An antiinflammatory galactolipid from rose hip (Rosa canina) that inhibits chemotaxis of human peripheral blood neutrophils in vitro. Journal of Natural Products. 2003;66(7):994–995. doi: 10.1021/np0300636. [DOI] [PubMed] [Google Scholar]
- 15.Winther K., Apel K., Thamsborg G. A powder made from seeds and shells of a rose-hip subspecies (Rosa canina) reduces symptoms of knee and hip osteoarthritis: a randomized, double-blind, placebo-controlled clinical trial. Scandinavian Journal of Rheumatology. 2005;34(4):302–308. doi: 10.1080/03009740510018624. [DOI] [PubMed] [Google Scholar]
- 16.Chrubasik J. E., Roufogalis B. D., Chrabasik S. Evidence of effectiveness of herbal antiinflammatory drugs in the treatment of painful osteoarthritis and chronic low back pain. Phytotherapy Research. 2007;21(7):675–683. doi: 10.1002/ptr.2142. [DOI] [PubMed] [Google Scholar]
- 17.Chrubasik C., Roufogalis B. D., Müller-Ladner U., Chrubasik S. A systematic review on the Rosa canina effect and efficacy profiles. Phytotherapy Research. 2008;22(6):725–733. doi: 10.1002/ptr.2400. [DOI] [PubMed] [Google Scholar]
- 18.Jäger A. K., Eldeen I. M. S., Van Staden J. COX-1 and -2 activity of rose hip. Phytotherapy Research. 2007;21(12):1251–1252. doi: 10.1002/ptr.2236. [DOI] [PubMed] [Google Scholar]
- 19.Jäger A. K., Petersen K. N., Thomasen G., Christensen S. B. Isolation of linoleic and α-linolenic acids as COX-1 and -2 inhibitors in rose hip. Phytotherapy Research. 2008;22(7):982–984. doi: 10.1002/ptr.2446. [DOI] [PubMed] [Google Scholar]
- 20.Schwager J., Hoeller U., Wolfram S., Richard N. Rose hip and its constituent galactolipids confer cartilage protection by modulating cytokine, and chemokine expression. BMC Complementary and Alternative Medicine. 2011;11, article 105 doi: 10.1186/1472-6882-11-105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Richard N., Arnold S., Hoeller U., Kilpert C., Wertz K., Schwager J. Hydroxytyrosol is the major anti-inflammatory compound in aqueous olive extracts and impairs cytokine and chemokine production in macrophages. Planta Medica. 2011;77(17):1890–1897. doi: 10.1055/s-0031-1280022. [DOI] [PubMed] [Google Scholar]
- 22.Richard N., Porath D., Radspieler A., Schwager J. Effects of resveratrol, piceatannol, triacetoxystilbene, and genistein on the inflammatory response of human peripheral blood leukocytes. Molecular Nutrition and Food Research. 2005;49(5):431–442. doi: 10.1002/mnfr.200400099. [DOI] [PubMed] [Google Scholar]
- 23.Filzmoser P. P., Varmuza K. Chemometrics: Multivariate Statistical Analysis in Chemometrics. 2012, http://cran.r-project.org/web/packages/chemometrics/index.html.
- 24.A. R Core Team Vienna . Team Vienna, R: A language and environment for statistical computing. Vienna, Austria: R Foundation for Statistical Computing; 2012. [Google Scholar]
- 25.Chakravarti B., Maurya R., Siddiqui J. A., Kumar Bid H., Rajendran S. M., Yadav P. P., Konwar R. In vitro anti-breast cancer activity of ethanolic extract of Wrightia tomentosa: role of pro-apoptotic effects of oleanolic acid and urosolic acid. Journal of Ethnopharmacology. 2012;142(1):72–79. doi: 10.1016/j.jep.2012.04.015. [DOI] [PubMed] [Google Scholar]
- 26.Viji V., Helen A., Luxmi V. R. Betulinic acid inhibits endotoxin-stimulated phosphorylation cascade and pro-inflammatory prostaglandin E2 production in human peripheral blood mononuclear cells. British Journal of Pharmacology. 2011;162(6):1291–1303. doi: 10.1111/j.1476-5381.2010.01112.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Yuan G. H., Masuko-Hongo K., Sakata M., et al. The role of C-C chemokines and their receptors in osteoarthritis. Arthritis & Rheumatology. 2001;44(5):1056–1070. doi: 10.1002/1529-0131(200105)44:5<1056::AID-ANR186>3.0.CO;2-U. [DOI] [PubMed] [Google Scholar]
- 28.Cecil D. L., Johnson K., Rediske J., Lotz M., Schmidt A. M., Terkeltaub R. Inflammation-induced chondrocyte hypertrophy is driven by receptor for advanced glycation end products. Journal of Immunology. 2005;175(12):8296–8302. doi: 10.4049/jimmunol.175.12.8296. [DOI] [PubMed] [Google Scholar]
- 29.Calder P. C., Ahluwalia N., Albers R., Bosco N., Bourdet-Sicard R., Haller D., Holgate S. T., Jönsson L. S., Latulippe M. E., Marcos A., Moreines J., Mrini C., Müller M., Pawelec G., Van Neerven R. J. J., Watzl B., Zhao J. A consideration of biomarkers to be used for evaluation of inflammation in human nutritional studies. British Journal of Nutrition. 2013;109(supplement 1):S1–S34. doi: 10.1017/S0007114512005119. [DOI] [PubMed] [Google Scholar]
- 30.Christensen R., Tarp S., Altman R. Comparing different preparations and doses of rosehip powder in patients with osteoarthritis of the knee: an exploratory randomized active-controlled trial. International Journal of Clinical Rheumatology. 2014;21:S24. doi: 10.2217/ijr.14.13. [DOI] [Google Scholar]