Abstract
Atherosclerosis is an inflammatory disease and the main cause of cardiovascular disease. Inflammation promotes plaque instability and clinical disease, such as myocardial infarction, stroke and peripheral vascular disease. Subclinical atherosclerosis begins with thickening of the arterial intimal layer, and increased intima-media thickness (IMT) in the carotid artery is a widely used measurement of subclinical atherosclerosis. Activation of CD137 (tumor necrosis factor receptor super family 9) promotes inflammation and disease development in murine atherosclerosis. CD137 is expressed in human atherosclerosis, but its role is largely unknown. This study uses a genetic approach to investigate CD137 in human atherosclerotic disease. In publicly available data on genotype and gene expression from the HapMap project, the minor T allele of rs2453021, a single nucleotide polymorphism in CD137, was significantly associated with CD137 gene expression. In the PROCARDIS and Wellcome Trust Case Control Consortium (WTCCC) cohorts of 13,029 cases and controls, no significant association was detected between the minor T allele of rs2453021 and risk for coronary artery disease or myocardial infarction. However, in the IMPROVE multicenter study of 3,418 individuals, the minor T allele of rs2453021 was associated with increased IMT of the common carotid artery (CCA), as measured by ultrasonography, with presence of plaque in CCA and with increased incidence of adverse noncardiac vascular events. Taken together, this study shows that the minor T allele of rs2453021 is associated with increased IMT in the CCA and increased risk of incident noncardiac vascular events, thus providing the first human genetic evidence for involvement of CD137 in atherosclerosis.
INTRODUCTION
Cardiovascular disease (CVD) is a leading cause of mortality and morbidity throughout the world (1–3). Most of the clinical manifestations of CVD (for example, myocardial infarction [MI], stroke and peripheral vascular disease) are caused by atherosclerosis (2). Subclinical atherosclerosis begins with subendothelial retention of lipoproteins and thickening of the arterial intima layer that may subsequently develop into atherosclerotic plaques (4,5). There is ample evidence that atherosclerosis is an inflammatory disease (1–3,6). The vessel wall is progressively infiltrated by leukocytes, lipids and debris, which contribute to plaque development and plaque inflammation, promote plaque instability and increase the risk for plaque rupture and clinical disease (1–3,6).
T cells are central orchestrators of the chronic inflammatory activity in atherosclerotic plaques. Both effector T cells (TEFF) and regulatory T cells (TREG) are involved in the control of inflammatory activity and lesion development. Low levels of TREG have been associated with increased risk for MI (7), and depletion of TREG promotes increased experimental atherosclerosis and dyslipidemia (8). Conversely, depletion of the TEFF population reduces atherosclerosis development (9,10).
T cells require two signals for activation: a signal from the T-cell receptor upon antigen recognition and an additional signal from a costimulatory receptor. The tumor necrosis factor receptor superfamily member 9 (TNFRSF9), CD137, is a costimulatory receptor of the TREG as well as TEFF subtypes of T cells (11,12). CD137 plays a key role in activation of the proinflammatory CD4+CD28null T cells, a subpopulation that is increased in patients with acute coronary syndrome (ACS) compared with patients with stable angina or controls. This result suggests a role of increased CD137 activation in ACS (13). We have previously shown that CD137 is expressed in both human and murine atherosclerotic lesions. Stimulation of CD137 in a murine model of atherosclerosis promotes atherosclerotic disease, with increased lesion size, increased local inflammatory activity and increased plaque infiltration of T cells (14). Conversely, CD137 deficiency reduces atherosclerosis in Apoe−/− and Ldlr−/− mice (15). Thus, CD137 plays a significant role in murine atherogenesis, but CD137 in human atherosclerosis is not well understood.
Efforts to develop agents that target human CD137 are underway (11,16,17). Because CD137 signaling influences experimental atherosclerosis, and knowledge on CD137 in human CVD is limited, intervention in CD137 signaling in human atherosclerosis is at present unwarranted. To gain further information about CD137 in human atherosclerosis, on the basis of our previous published experimental data and knowledge of the effects of CD137 activation on atherosclerosis (14), we hypothesized that genetic variants in CD137 influence CD137 expression and atherosclerotic disease. To study CD137 in atherosclerosis, we used a genetic approach of mRNA expression in lymphoblastoid cell lines together with population-based cohorts with a high prevalence of cardiovascular risk factors.
MATERIALS AND METHODS
Selection of Genetic Variants in CD137
Publicly available data on genotype from the HapMap CEU population (18) and gene expression from lymphoblastoid cell lines derived from the same population (19) were used to investigate association of genotype with gene expression. The 42 single nucleotide polymorphisms (SNPs) in CD137 were identified in HapMap (HapMap Data Freeze #21a, http://hapmap.ncbi.nlm.nih.gov) (18). CD137 mRNA expression was investigated by using the Affymetrix GeneChip Human Exon 1.0 ST Array data accessible through the GEO Series accession number GSE9372, as previously described (19).
Measurement of Carotid Intima-Media Thickness (IMT)
Association of genotype and subclinical atherosclerosis, measured as intimamedia thickness (IMT) in the carotid arteries and determined by ultrasonography, was studied in a cohort of 3,430 participants in the IMPROVE study (20,21). In short, the IMPROVE study is a multicenter study that enrolled 54- to 79-year-old men and women with at least three risk factors for CVD in Finland, France, Italy, the Netherlands and Sweden. Risk factors were as follows: male sex or ≥5 years after menopause for women; dyslipidemia, defined as hypercholesterolemia (mean calculated low-density lipoprotein [LDL] cholesterol levels >160 mg/dL or treatment with lipid-lowering drugs); hypertriglyceridemia (triglyceride levels >200 mg/dL or treatment with triglyceride-lowering drugs); hypoalphalipoproteinemia (high-density lipoprotein [HDL] cholesterol <40 mg/dL); hypertension (defined as diastolic blood pressure >90 mmHg and/or systolic blood pressure >140 mmHg or treatment with antihypertensive drugs); diabetes (defined as fasting glucose level >126 mg/dL [7.0 mmol/L] or treatment with insulin or oral hypoglycemic drugs); smoking; or family history of CVD. Participants were free of symptomatic CVD and any conditions that might limit longevity, for example, cancer, at the time of enrollment. All subjects were included after written consent. The carotid IMT was measured using B-mode ultrasound, and clinical and biochemical data were collected as previously described (20). In brief, the far walls of both the left and right carotid arteries were examined at two levels of the common carotid: the bifurcation and the internal carotid arteries.
Ultrasonography was performed from the anterior, lateral and posterior views and recorded on S-VHS. IMT measurements were carried out by a centralized laboratory, and repeatability was monitored as described (20). The variables measured were the mean and maximum IMT of the common carotid at the first centimeter (cm) proximal to the bifurcation (CC-first cm-IMTmean and CC-first cm-IMTmax) and in a segment excluding the first centimeter proximal to the bifurcation (CC-IMTmean and CC-IMTmax) and of the bifurcation (Bif-IMTmean and Bif-IMTmax) and of the internal carotid arteries (ICA-IMTmean and ICA-IMTmax). Composite IMT variables considering the whole carotid tree were derived from the segment-specific measurements: IMTmean, IMTmax and IMTmean-max (the average of maximal IMT recorded at the different segments). Presence of plaque in a segment was defined as IMTmax >1.5 mm. IMT determinations were repeated at 15 and 30 months of follow-up. Thirty-month progression of all IMT measurements (IMTmax progression, change over time, in mm/year) was calculated by linear regression considering all three time points (0, 15 and 30 months). Results in the present study were analyzed by three models: (a) a basic model adjusting for sex, age and genetic background (multiple dimension scaling [MDS]), (b) a model including also the identified confounders (hypercholesterolemia, hypertriglyceridemia and current smoking) and (c) a model including all established risk factors listed in Table 1, except the three variables describing family history of vascular disease, because of a high degree of missing data. Subjects were followed up with respect to incident vascular events (coronary, cerebral or peripheral) until 36 months (20).
Table 1.
Subject characteristics and risk factor prevalence according to the rs2453021 genotype in IMPROVE.
| Genotype | ||||
|---|---|---|---|---|
|
|
||||
| CC | CT | TT | P | |
| n | 1,517 | 1,510 | 391 | |
| Subject characteristics | ||||
| Male sex (%) | 48.4 | 47.9 | 48.3 | 0.888 |
| Age (years) [mean (95% CI)] | 64.4 (64.2–64.7) | 64.0 (63.8–64.3) | 64.1 (63.6–64.7) | 0.091 |
| BMI (kg/m2) [mean (95% CI)] | 27.2 (27.0–27.4) | 27.3 (27.1–27.5) | 27.1 (26.7–27.5) | 0.859 |
| CRP (mg/L) [median (IQR)] | 1.86 (0.74–3.67) | 1.86 (0.78–3.53) | 1.86 (0.80–3.58) | 0.719 |
| Risk factors | ||||
| Hypercholesterolemia (%) | 68.3 | 69.7 | 76.2 | 0.008 |
| Hypertriglyceridemia (%) | 23.7 | 28.2 | 27.6 | 0.012 |
| Hypoalphalipoproteinemia (%) | 12.5 | 13.7 | 14.1 | 0.308 |
| Hypertension (%) | 70.6 | 67.3 | 68.3 | 0.122 |
| Diabetes (%) | 27.9 | 26.1 | 24.2 | 0.114 |
| Family history of peripheral vascular disease (%) | ||||
| No | 79.5 | 77.4 | 81.3 | 0.435 |
| Yes | 11.0 | 13.2 | 11.0 | |
| Not known | 9.6 | 9.4 | 7.7 | |
| Family history of cerebrovascular disease (%) | ||||
| No | 60.3 | 61.2 | 59.8 | 0.902 |
| Yes | 35.2 | 35.3 | 36.1 | |
| Not known | 4.6 | 3.6 | 4.1 | |
| Family history of coronary heart disease (%) | ||||
| No | 32.9 | 35.1 | 34.3 | 0.389 |
| Yes | 63.2 | 61.6 | 62.4 | |
| Not known | 3.8 | 3.2 | 3.3 | |
| Current smoker (%) | 16 | 15.2 | 9.5 | 0.006 |
| Smoking pack-year category (%) | ||||
| Never smoker | 48.4 | 49.6 | 50.4 | 0.234 |
| 1st quartile | 12.3 | 12.3 | 14.9 | |
| 2nd quartile | 13.8 | 11.9 | 12.1 | |
| 3rd quartile | 11.9 | 13.4 | 10.8 | |
| 4th quartile | 13.6 | 12.9 | 11.8 | |
P values are from linear regression and χ2 tests for linear-by-linear association. Dyslipidemia was defined as hypercholesterolemia (mean calculated LDL cholesterol levels >160 mg/dL or treatment with lipid-lowering drugs), hypertriglyceridemia (triglyceride levels >200 mg/dL or treatment with triglyceride-lowering drugs) and hypoalphalipoproteinemia (HDL cholesterol <40 mg/dL). Hypertension was defined as diastolic blood pressure >90 mmHg and/or systolic blood pressure >140 mmHg or treatment with antihypertensive drugs. Diabetes was defined as fasting glucose level >126 mg/dL (7.0 mmol/L) or treatment with insulin or oral hypoglycemic drugs (20). IQR, interquartile range. Genotype: CC, homozygote major C allele; CT, heterozygote; TT, homozygote minor T allele.
rs2453021 and Incident Vascular Events
Association of rs2453021 with clinical outcome was analyzed by using prospective data collected during the 36-month follow-up of the IMPROVE cohort (20). Vascular events were analyzed by Cox regression as (a) any vascular event, (b) cardiac events only, (c) non-cardiac events (cerebral or peripheral), (d) cerebral vascular events only and (e) peripheral vascular events only. Analyses were performed using the same three models as for the IMT measurements. Genotyping of rs2453021 in IMPROVE was performed by using Illumina GoldenGate technology at the SNP&SEQ Technology Platform (Uppsala, Sweden).
rs2453021 and Coronary Artery Disease
Association of rs2453021 with coronary artery disease (CAD) was analyzed in 5,765 CAD and/or MI cases from the PROCARDIS project (22), along with 7,264 controls from PROCARDIS and the Wellcome Trust Case Control Consortium (WTCCC) (23); in total, 13,029 individuals were analyzed using imputed genotype data. Briefly, PROCARDIS is a multicenter European project that assembled a uniquely large and carefully phenotyped cohort of subjects with CAD with onset of MI or other ACS before 65 years of age. All PROCARDIS participants included in the present study had been genotyped by using either the Illumina Human 1M or 610K quad arrays and imputed to 1,000 Genomes (CEU panel August 2009 release) by using MACH software (24).
Statistics
For expression quantitative trait locus (eQTL) studies, the association was calculated according to an additive linear model. Briefly, genotypes were re-coded as 1, 2 or 3 corresponding to homozygote major allele, heterozygote or homozygote minor allele. This value was fitted to the TNFRSF9 (CD137) gene expression in a linear regression model, and the P value for the fit of the slope was calculated. All eQTL calculations were performed in R version 2.9.2.
Associations of genotype with baseline IMT and IMT progression were investigated by linear regression assuming an additive genetic effect, by using PLINK version 1.07 (25). Cox regression analysis of genotype associations with incident vascular events and additional statistical analyses were performed by using IBM SPSS Statistics version 20 (IBM Corporation, Armonk, NY, USA). Analyses of genotype associations with IMT and incident events were adjusted for sex, age and genetic background (MDS components 1–3 based on CardioMetabo-chip data) (21) and, where indicated, for potential confounders and additional covariates. Associations of genotype with presence of plaque were investigated by logistic regression by using the same adjustments. Because of non-normal distribution, all baseline IMT variables and C-reactive protein levels measured in IMPROVE were logarithmically transformed before statistical analysis.
Disease (CAD and MI)-SNP association was tested by applying a logistic regression model by using STATA/MP 11 (StataCorp LP, College Station, TX, USA). Analyses were adjusted for age, sex, country of origin and relatedness.
RESULTS
The Minor Allele of rs2453021 Was Associated with Decreased CD137 mRNA Expression
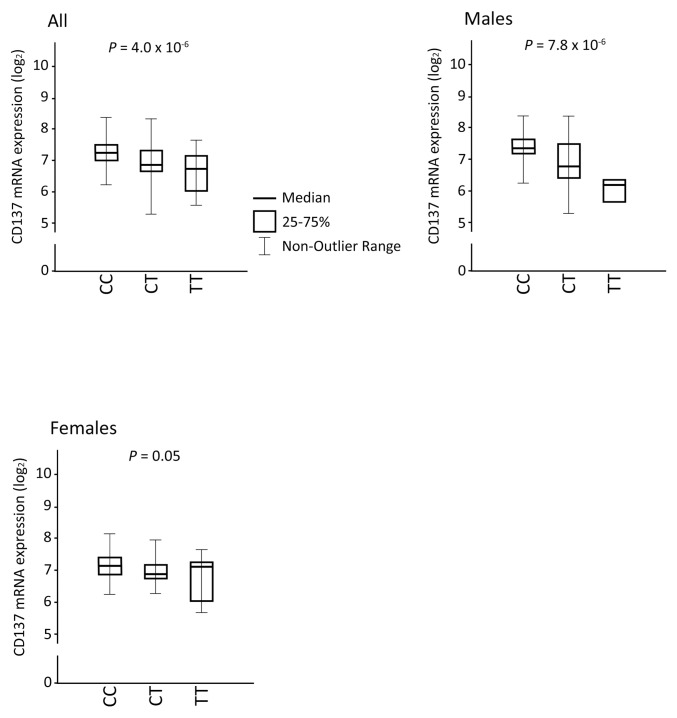
We first set out to investigate association between SNPs at the CD137 locus and CD137 mRNA expression levels. To study this, we combined two publicly available databases: the genotyping project of HapMap as well as an expression analysis performed on cell lines from the same population (18,19). Thus, we created a new data set containing both genotype and expression data. The minor T allele of rs2453021 was associated with significantly lower CD137 expression (all individuals, β = 0.797, 95% confidence interval [CI] 0.726–0.876, P = 4.0 × 10−6, n = 171; Figure 1). Although there was overlap in the distributions of CD137 mRNA levels, the linear effect of the rs2453021 genotype was highly significant, as evidenced by the low P value. There was a difference between sexes in that the minor T allele was associated with lower CD137 expression in males, but this comparison of CD137 expression only reached borderline significance in females (β = 0.689, 95% CI 0.589–0.805, P = 7.8 × 10−6, n = 87, versus β = 0.894, 95% CI 0.799–1, P = 0.050, n = 84) (Figure 1). There was a significant interaction of sex and genotype and expression level of CD137 (P = 0.007). There was no association between other investigated CD137 SNPs and CD137 mRNA expression (data not shown). Accordingly, rs2453021 was selected for further studies.
Figure 1.
The minor T allele of rs2453021 was associated with lower CD137 mRNA expression. A combined data set of genotyping data from the HapMap CEU population together with gene expression data from transcriptomic analysis of lymphoblastoid cell lines derived from the same population was created. rs2453021 genotype and CD137 mRNA expression was analyzed in a linear regression model for (a) all individuals (β = 0.797, 95% CI 0.726–0.876, P = 4.0 × 10−6), (b) in males only (β = 0.689, 95% CI 0.589–0.805, P = 7.8 × 10−6) and (c) in females only (β = 0.894, 95% CI 0.799–1, P = 0.05). Values shown are log2 CD137 mRNA expression. P values were calculated for fit of slope. For all, n = 171; males, n = 87; and females, n = 84. Genotype: CC, homozygote major C allele; CT, heterozygote; TT, homozygote minor T allele.
The Minor T Allele of rs2453021 Was Associated with Increased IMT and Presence of Plaque in the Common Carotid Artery
We then turned to the IMPROVE cohort to investigate whether rs2453021 genotype was associated with carotid IMT at baseline or at 30 months of progression of carotid IMT, as determined by high-resolution ultrasound. Basic characteristics and prevalence of established cardiovascular risk factors of the subjects in this study are presented in Table 1. Genotype data passing quality control were obtained for rs2453021 in 3,418 of 3,430 available subjects (call-rate 0.9965). We observed that the rs2453021 minor T allele (minor allele frequency 0.34) was associated with increased prevalence of hypercholesterolemia, hypertriglyceridemia and decreased prevalence of current smoking (Table 1). Therefore, IMT associations were investigated by three different models (Table 2): (a) a basic model adjusting for sex, age and genetic background (MDS), (b) a model that also included the identified confounders (hypercholesterolemia, hypertriglyceridemia and current smoking) and (c) a model that included all variables listed in Table 1, except the three variables describing family history of peripheral, cerebrovascular and coronary heart disease, as such information was missing for many subjects.
Table 2.
Association of rs2453021 with carotid IMT: baseline and 30-month progression.
| Model 1 | Model 2 | Model 3 | |||||||
|---|---|---|---|---|---|---|---|---|---|
|
|
|
|
|||||||
| β (95% CI) | n | P | β (95% CI) | n | P | β (95% CI) | n | P | |
| Baseline IMT | |||||||||
| CC-IMTmean | 0.0039 (0.0004–0.0074) | 3,415 | 0.028 | 0.0045 (0.0011–0.0080) | 3,411 | 0.010 | 0.0047 (0.0013–0.0082) | 3,292 | 0.007 |
| CC-IMTmax | 0.0063 (0.0008–0.0119) | 3,415 | 0.025 | 0.0070 (0.0015–0.0126) | 3,411 | 0.013 | 0.0075 (0.0019–0.0131) | 3,292 | 0.008 |
| IMT progression | |||||||||
| IMTmax prog | −0.0091 (−0.0177 to −0.0005) | 3,031 | 0.038 | −0.0092 (−0.0178 to −0.0006) | 3,028 | 0.037 | −0.0077 (−0.0164 to 0.0010) | 2,922 | 0.084 |
The rs2453021 was associated with increased mean intima-media thickness (IMTmean) and maximum IMT (IMTmax) in the common carotid (CC) artery in the IMPROVE cohort, independently of vascular risk factors. The rs2453021 was associated with decreased IMT progression (IMTmaxprog) by using models 1 and 2, but this association was abolished when compensating for all risk factors using model 3 (see below). Carotid artery atherosclerosis was evaluated in participants with at least three risk factors for CVD, but no clinical CVD at the time of enrollment using high-resolution ultrasound at baseline and 30 months. Results are from linear regression analysis assuming an additive genetic effect. Effect sizes (β) with 95% CIs for the rare T allele are shown. Model 1 is adjusted for sex, age and genetic background (MDS); model 2 is adjusted for sex, age, genetic background (MDS) and variables in Table 1 found to be associated with rs2453021 (hypercholesterolemia, hypertriglyceridemia, current smoking); and model 3 is adjusted for sex, age, genetic background (MDS) and all variables in Table 1 (hypercholesterolemia, hypertriglyceridemia, hypoalphalipoproteinemia, hypertension, diabetes, current smoking, smoking pack-year categories, BMI and CRP), except the three variables describing family history of vascular disease, due to a high degree of missing data. No other significant associations with baseline IMT or 30-month IMT progression were observed for any of the three models (data not shown).
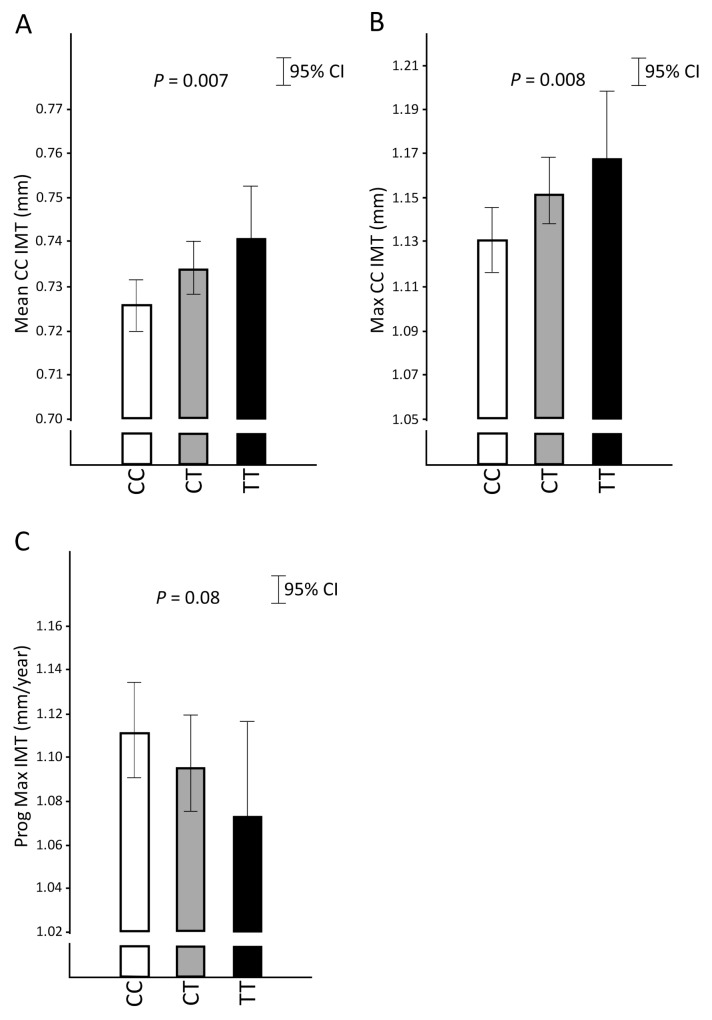
The rs2453021 minor T allele was associated with thicker CC-IMT independently of vascular risk factors; the T allele showed a significant positive association with mean and maximum IMT of the common carotid artery (CC-IMTmean and CC-IMTmax) in all three models at baseline (Table 2). The rs2453021 T allele also showed a negative association with the 30-month progression of the maximum IMT measured in the whole carotid tree (IMTmax progression) in the first two models, but this association disappeared after adjusting for all vascular risk factors in model 3, indicating that it may at least in part be due to confounding factors (Table 2 and Figure 2). Among the additional covariates in model 3, the loss of significance was attributed mainly to diabetes; addition of covariates one by one to model 2 showed that further adjustment for diabetes had the greatest impact (SNP-IMTmax progression association after addition of diabetes to model 2: β = −0.0083, 95% CI −0.0169–0.0004; P = 0.062). The prevalence of diabetes was positively associated with IMTmax progression (unadjusted β = 0.0139, 95% CI 0.0009–0.0269; P = 0.037). Taken together with the tendency toward lower diabetes prevalence for the T allele (Table 1), this result indicates that the SNP-IMTmax progression association seen in models 1 and 2, at least in part, may be due to confounding by diabetes status. No other significant association with baseline IMT or IMT progression at 30 months were observed in any of the three models in analyses of men and women together (data not shown). Analyses performed in men and women separately showed that the minor T allele of rs2453021 was associated with higher CC-IMTmean in men but not in women (model 3, P = 0.007 and P = 0.32, respectively), with slower IMTmax progression in men but not in women (model 3, P = 0.01 and P = 0.95, respectively) and with higher CC-IMTmax in women but not in men (model 3, P = 0.02 and P = 0.11, respectively).
Figure 2.
The minor T allele of rs2453021 in CD137 was associated with increased intima-media thickness (IMT) in the common carotid (CC) artery independently of vascular risk factors. IMT was measured by high-resolution ultrasonography in the IMPROVE cohort. Participants had at least three risk factors of CVD at the time of enrollment but were free of clinical disease. Measurements were made at baseline and at 15 and 30 months. Thirty-month progression of IMT (Prog CC-IMT) (change over time; in mm/year) was calculated by linear regression considering all three time points (0, 15 and 30 months). Values are mean CC-IMT (Mean CC-IMT; in mm) (A), maximum CC-IMT (Max CC-IMT; in mm) (B) and maximum IMT progression (Prog Max IMT; in mm/year) (C) according to rs2453021 genotype. Results are adjusted for sex, age, cardiovascular risk factors (hypercholesterolemia, hypertriglyceridemia, hypoalphalipoproteinemia, hypertension, diabetes, current smoking, smoking pack-year categories, body mass index [BMI] and CRP) and genetic background (MDS) (model 3). The antilog of mean levels with 95% CI of logarithmically transformed data are shown. Linear regression P values are indicated. Genotype: CC, homozygote major C allele; CT, heterozygote; TT, homozygote minor T allele.
We then proceeded to analyze association of rs2453021 with the presence of plaque at baseline in all subjects. The rs2453021 minor T allele was significantly associated with presence of plaque in the common carotid artery, using all three models (CC-IMTmax >1.5 mm; model 3 odds ratio 1.19, 95% CI 1.03–1.38, P = 0.02), but not with presence of plaque in any other carotid segment (not shown). No significant association with the presence of plaque was detected in any segment when men and women were analyzed separately using models 1–3; loss of significance with respect to common carotid plaque presence was due to loss of statistical power, since effect sizes (odds ratios) were preserved in both sexes (model 3 odds ratios 1.23 [95% CI 0.97–1.57], P = 0.091 in women, and 1.18 [0.97–1.42], P = 0.097 in men).
The Minor Allele of rs2453021 Was Associated with Increased Risk of Incident Noncardiac Vascular Events
To explore the association of the minor T allele of rs2453021 with incident clinical events, we analyzed prospective data collected during the 36-month follow-up of the IMPROVE cohort by Cox regression analysis, with adjustments according to models 1–3. Among a total of 3,418 subjects, 190 subjects suffered at least one event during a median follow-up of 36.2 months (interquartile range 35.8–37.4 months). The minor T allele of rs2453021 was significantly associated with increased risk of noncardiac events using model 3 (hazard ratio per rare T allele 1.42, 95% CI 1.02–1.98, P = 0.036; n = 74 subjects with events among 3,295 subjects with nonmissing covariates [Table 3]). There was no association between rs2453021 and “cardiac events only” or “any vascular event” or with peripheral vascular or cerebrovascular events separately, using model 3 (Table 3). In a sex-stratified analysis using model 3, the rs2453021 T allele was associated with higher risk of peripheral events in men, but not in women (P = 0.04 and P = 0.77, respectively). No significant association between the minor allele and risk for any vascular events was observed using models 1 and 2 (data not shown). Furthermore, there was no detectable association between rs243021 genotype and levels of C-reactive protein (CRP) in blood (Table 1).
Table 3.
Association of rs2453021 with incident vascular events: model 3.
| Hazard ratio (95% CI) | n | P | |
|---|---|---|---|
| Any vascular eventa | 1.085 (0.869–1.354) | 176 | 0.472 |
| Cardiac event | 0.924 (0.694–1.232) | 111 | 0.592 |
| Noncardiac eventb | 1.424 (1.024–1.981) | 74 | 0.036 |
| Cerebrovascular event | 1.403 (0.973–2.023) | 60 | 0.070 |
| Peripheral event | 1.716 (0.935–3.149) | 21 | 0.081 |
The minor T allele of rs2453021 was associated with increased risk of incident noncardiac vascular events. Data collected at the 36-month follow-up of participants of the IMPROVE cohort were analyzed to explore associations of rs2453021 and incident clinical vascular events. Cardiac events included MI, sudden cardiac death, angina pectoris and coronary revascularization; cerebrovascular events included ischemic stroke and transient ischemic attack; and peripheral events included revascularization of peripheral arteries and new diagnosis of intermittent claudication (20). Results are from Cox regression with adjustments according to model 3 (sex, age, genetic background [MDS] and risk factors for CVD [hypercholesterolemia, hypertriglyceridemia, hypoalphalipoproteinemia, hypertension, diabetes, current smoking, smoking pack-year categories, BMI and CRP]). Hazard ratios with 95% CIs per rare T allele are given. The number of subjects suffering one or more vascular events among 3,295 individuals with nonmissing covariates is shown.
The first of a cardiac, cerebrovascular or peripheral events.
The first of a cerebrovascular or peripheral events.
The Minor Allele of rs2453021 Was Not Associated with Increased Risk of CAD in PROCARDIS/WTCCC
To investigate the association between rs2453021 genotype and CAD or MI, we turned to the combined PROCARDIS/WTCCC case-control cohort (22,23). In total, 13,029 individuals, including 5,765 cases and 7,264 controls, were investigated. Basic clinical characteristics of this sample have been reported (22). There was no association between presence of the minor T allele of rs2453021 and risk for CAD or MI in this sample (Table 4).
Table 4.
Association of rs2453021 and risk for CAD or MI.
| Odds ratio (95% CI) | P | |
|---|---|---|
| All | 1.013 (0.933–1.100) | 0.757 |
| Women only | 1.019 (0.880–1.180) | 0.798 |
| Men only | 1.011 (0.920–1.113) | 0.814 |
| MI | 1.028 (0.945–1.119) | 0.521 |
| CAD only | 1.013 (0.877–1.169) | 0.862 |
The minor T allele of rs2453021 is not associated with increased risk for CAD or MI. A total of 13,029 individuals in the PROCARDIS cohort including WTCCC controls (5,765 cases and 7,264 controls) were investigated for association between rs2453021 and risk for CAD or MI. All results were adjusted for age, sex, country and relatedness. The T allele frequency was 0.394.
DISCUSSION
This preformed hypothesis-driven candidate gene study links rs2453021 to CD137 expression levels and risk of atherosclerosis-related vascular events in humans and thus provides the first genetic evidence that suggests involvement of CD137 in human atherosclerosis. The minor T allele of rs2453021 was associated with lower CD137 mRNA expression in lymphoblastoid cells. Independently of established vascular risk factors, the minor T allele was also associated with increased IMT and a higher prevalence of IMT-defined plaque in the CCA. Furthermore, the minor T allele was associated with an increased risk of clinical noncardiac (cerebrovascular or peripheral) vascular events.
We and others have previously shown that activation of CD137 has adverse effects on experimental atherosclerosis (13–15,26). However, the impact of the CD137 genotype on human atherosclerosis and clinical disease has not been investigated. This study uses genetic and gene expression databases combined with a CAD case-control study and a prospective study of subjects with high cardiovascular risk.
Here, we show that the minor T allele of rs2453021 was associated with lower CD137 mRNA expression, with increased carotid IMT and increased incidence of noncardiac vascular events. This finding is surprising, since stimulation of CD137 is known to promote atherosclerosis and increase inflammation in murine models of atherosclerosis (14,15). Previous studies have demonstrated higher CD137 expression in human atherosclerotic tissue compared with normal arteries, suggesting an association between high CD137 activity and atherosclerotic disease (14). However, because healthy normal arteries, used as controls in the previous studies, lack significant inflammatory activity, the comparison may not translate to the situation in the present study that compares genotypes in individuals with either subclinical or clinical atherosclerosis.
Observations of differences in expression of immune-regulatory molecules may be of interest for understanding the different clinical courses of atherosclerotic disease in males and females (27,28). It has been reported that genetic variants of other members of the tumor necrosis superfamily, for example, OX40 ligand and LIGHT, are associated with gender differences in risk for disease and clinical outcome (29–31). The association of rs2453021 with CD137 expression was stronger in the set of lymphoblastoid cells derived from males than from females. However, in the human cohorts, the association between rs2453021 and vascular disease was not consistently different between sexes. Nevertheless, the results of the present study, including both the results from cell lines and the cohorts, indicate that regardless of mechanism, potential sex differences may be important to consider in future studies of CD137 in atherosclerosis. The mechanisms underlying these observed sex differences are currently unknown, but genetic and epigenetic mechanisms, including hormone-induced changes, may be at play. The functional aspects of these findings need to be addressed in future mechanistic studies both in vitro and in vivo.
The present study found an association of rs2453021 with noncardiac vascular events, but not with cardiac events, suggesting that the distinct vascular sites may be affected differently. Previous studies of the TNFSF member OX40 ligand found association between genotype and MI, but not with ischemic stroke, which may suggest a similarly differential role for this TNFSF molecule at unique vascular disease locales (31,32). Although atherosclerosis underlies both CAD and carotid plaques, there are significant differences in the pathophysiology of these manifestations. The anatomical and functional properties, local rheology and specific function of the vessels differ (6,33,34). In addition, some of the established risk factors for atherosclerosis may also contribute in a differentiated way to the pathogenesis in unique vascular beds. For example, hypertension is a major risk factor for stroke, hypercholesterolemia is a major risk factor for CAD, and smoking and diabetes are major risk factors for peripheral vascular disease (34). Also, MI is largely caused by thrombosis and local lumen occlusion, whereas stroke often is a result of embolism from ruptured carotid atherosclerotic plaques (33,34). The effects of the interaction between CD137 expression, vascular bed differences and different risk factors are not known, but they may likely contribute to differences in local effects of receptor expression on atherosclerosis development. Of note, this study does not directly investigate the role of CD137 activity in the pathogenesis of human vascular disease and does not provide conclusive data on its role in plaque inflammation. Future studies are warranted to address the cellular and molecular mechanisms behind these observations.
The surprising association of the minor allele of rs2450321 with a reduction of CD137 mRNA expression in lymphoblasts and with an increase in risk for noncardiac vascular events and increased IMT, respectively, may have several explanations. For example, CD137 is known to be constitutively expressed on human and murine TREG, whereas TEFF mainly express CD137 upon activation (11,35). TREG are present at all stages of atherosclerosis development in both human and murine atherosclerosis (8,36). Depletion of TREG leads to increased atherosclerosis in murine disease models (8,37). Because CD137 is expressed on regulatory as well as effector T cells, the TREG/TEFF activity is likely to affect the result of CD137 activation (11,16). Although the net effects of CD137 stimulation in development of human atherosclerotic disease are currently unknown, it is tempting to speculate that the minor T allele of rs2453021 is associated with a more unfavorable TEFF/TREG activity balance, which would promote inflammation and aggravate atherosclerosis in the carotid arteries. Furthermore, in humans, the CD4+CD28null T-cell population, an aberrant subpopulation that lacks the key costimulatory molecule CD28, expands with age and is increased in patients with ACS compared with patients with stable angina or controls (13). CD137 may substitute for CD28 in CD4+CD28null T cells and play a role in costimulation of effector responses (13,26). It is therefore conceivable that alterations in the expression level of CD137 have effects in patients with high levels of the disease-associated CD4+CD28null T cells. Blocking of CD137 in this subset leads to an inhibition of inflammatory and cytotoxic functions, which suggests a role for CD137 in plaque rupture and ACS (1,2,6,13,38). It is possible that the decreased expression of CD137 mRNA associated with rs2453021 in the present study may play a role in reducing activation of the harmful CD4+CD28null T cells and ultimately lead to less plaque inflammation and less clinical symptoms of CAD. Importantly, the original observation of association between the minor T allele and CD137 expression level was made in lymphoblastoid cell lines and may not directly translate into expression in patients with MI, peripheral vascular disease or stroke. Bearing this limitation in mind, we report that the minor T allele of rs2453021 was found to decrease the expression of CD137, and additional disease association studies showed that this same T allele was associated with increased intima-media thickness and increased risk for noncardiac vascular events. Other considerations are that CD137 mRNA levels may not directly correlate with tissue levels of functional receptor protein, but the current study design prevented direct measurements of relevant protein levels. Potential differences in receptor efficacy, turnover time and ligand availability may also affect CD137 function. Nevertheless, the observed difference in mRNA level is interesting and warrants further studies of the functional effects of this finding.
The potential benefit of including imaging data on an individual’s atherosclerotic burden in the clinical evaluation of cardiovascular risk is under debate (39). Carotid IMT, as measured by high-resolution ultrasound, is a widely used marker of subclinical atherosclerosis. Previous studies have shown associations of genotype and IMT (40), and increased IMT has been associated with increased risk of MI and stroke (4,40,41). In the present study, we chose to use this established measurement of subclinical atherosclerosis. In the IMPROVE cohort, extensive IMT measurements were acquired according to a standardized protocol to minimize the intra- and interobserver variability inherent to this method (4,20). The use of a noninvasive evaluation of subclinical atherosclerosis in subjects free of any clinical manifestation of atherosclerosis at baseline provides an opportunity to investigate atherosclerosis in a wider population, before the onset of clinical manifestations (4,6,41). In the initial analysis, association of the rs2453021 T allele with slower IMT progression was observed, but this association did not survive adjustment for established vascular risk factors and appeared to be influenced by confounding by diabetes status. However, after adjustment for risk factors, these data do show association of rs2453021 with increased IMT.
We have previously shown expression of CD137 in carotid lesions (14), and this study builds on and expands that finding. The association between the minor T allele of rs2453021 and increased IMT in the CCA suggests a role in atherosclerosis build-up in these vessels, possibly through the relative TREG/TEFF activity. The current findings suggest a preferential role of rs2453021, or perhaps CD137 in general, in noncoronary rather than coronary atherothrombotic events. We can only speculate that this relates to differences in etiology of atherothrombosis in different vascular beds. However, the role of CD137 in coronary plaque rupture and the presence of CD137 in coronary artery lesions need further investigation, as does the influence of the CD137-expression-level–associated rs2453021 in ischemic stroke.
CONCLUSION
The minor T allele of rs2453021 was associated with decreased CD137 mRNA expression and increased IMT in the CCA. There was also a significant association between the minor T allele of rs2453021 and increased risk of incident clinical noncardiac vascular events. In contrast to the noncardiac vascular events, no link was found between rs2453021 alleles and CAD. Our results indicate a possible role of CD137 in human CVD and that CD137 genotypes may have clinical impact. Further studies are warranted to determine the value of these new insights into CD137 biology.
ACKNOWLEDGMENTS
This study was supported by the Swedish Heart-Lung Foundation, the Wenner-Gren Foundation, Karolinska Institutet, Stiftelsen för Gamla Tjänarinnor, the Swedish Research Council (projects 0593, 6816 and 8691 and Linnaeus support 8703), the Swedish Foundation for Strategic Research, the Cardiovascular and Diabetes Programmes of the Karolinska Institutet and Stockholm County Council, the Knut and Alice Wallenberg Foundation and the European Commission (AtheroRemo, PROCARDIS and IMPROVE collaborative projects), the Academy of Finland, the Ministry of Education and Culture of Finland, the City of Kuopio, the British Heart Foundation and the Italian Ministry of Health (Ricerca Corrente). SE Humphries is supported by the British Heart Foundation (PG08/008).
Footnotes
Online address: http://www.molmed.org
DISCLOSURE
The authors declare that they have no competing interests as defined by Molecular Medicine, or other interests that might be perceived to influence the results and discussion reported in this paper.
REFERENCES
- 1.Hansson GK. Inflammation, atherosclerosis, and coronary artery disease. N Engl J Med. 2005;352:1685–95. doi: 10.1056/NEJMra043430. [DOI] [PubMed] [Google Scholar]
- 2.Hansson GK, Hermansson A. The immune system in atherosclerosis. Nat Immunol. 2011;12:204–12. doi: 10.1038/ni.2001. [DOI] [PubMed] [Google Scholar]
- 3.Legein B, Temmerman L, Biessen EA, Lutgens E. Inflammation and immune system interactions in atherosclerosis. Cell Mol Life Sci. 2013;70:3847–69. doi: 10.1007/s00018-013-1289-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.van den Oord SC, et al. Carotid intima-media thickness for cardiovascular risk assessment: systematic review and meta-analysis. Atherosclerosis. 2013;228:1–11. doi: 10.1016/j.atherosclerosis.2013.01.025. [DOI] [PubMed] [Google Scholar]
- 5.Tabas I, Williams KJ, Boren J. Subendothelial lipoprotein retention as the initiating process in atherosclerosis: update and therapeutic implications. Circulation. 2007;116:1832–44. doi: 10.1161/CIRCULATIONAHA.106.676890. [DOI] [PubMed] [Google Scholar]
- 6.Libby P, Ridker PM, Hansson GK. Progress and challenges in translating the biology of atherosclerosis. Nature. 2011;473:317–25. doi: 10.1038/nature10146. [DOI] [PubMed] [Google Scholar]
- 7.Wigren M, et al. Low levels of circulating CD4+FoxP3+ T cells are associated with an increased risk for development of myocardial infarction but not for stroke. Arterioscler Thromb Vasc Biol. 2012;32:2000–4. doi: 10.1161/ATVBAHA.112.251579. [DOI] [PubMed] [Google Scholar]
- 8.Klingenberg R, et al. Depletion of FOXP3+ regulatory T cells promotes hypercholesterolemia and atherosclerosis. J Clin Invest. 2013;123:1323–34. doi: 10.1172/JCI63891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Reardon CA, et al. Effect of immune deficiency on lipoproteins and atherosclerosis in male apolipoprotein E-deficient mice. Arterioscler Thromb Vasc Biol. 2001;21:1011–6. doi: 10.1161/01.atv.21.6.1011. [DOI] [PubMed] [Google Scholar]
- 10.Zhou X, Nicoletti A, Elhage R, Hansson GK. Transfer of CD4(+) T cells aggravates atherosclerosis in immunodeficient apolipoprotein E knockout mice. Circulation. 2000;102:2919–22. doi: 10.1161/01.cir.102.24.2919. [DOI] [PubMed] [Google Scholar]
- 11.Wang C, Lin GH, McPherson AJ, Watts TH. Immune regulation by 4–1BB and 4–1BBL: complexities and challenges. Immunol Rev. 2009;229:192–215. doi: 10.1111/j.1600-065X.2009.00765.x. [DOI] [PubMed] [Google Scholar]
- 12.Watts TH. TNF/TNFR family members in costimulation of T cell responses. Annu Rev Immunol. 2005;23:23–68. doi: 10.1146/annurev.immunol.23.021704.115839. [DOI] [PubMed] [Google Scholar]
- 13.Dumitriu IE, et al. High levels of costimulatory receptors OX40 and 4-1BB characterize CD4+CD28null T cells in patients with acute coronary syndrome. Circ Res. 2012;110:857–69. doi: 10.1161/CIRCRESAHA.111.261933. [DOI] [PubMed] [Google Scholar]
- 14.Olofsson PS, et al. CD137 is expressed in human atherosclerosis and promotes development of plaque inflammation in hypercholesterolemic mice. Circulation. 2008;117:1292–301. doi: 10.1161/CIRCULATIONAHA.107.699173. [DOI] [PubMed] [Google Scholar]
- 15.Jeon HJ, et al. CD137 (4-1BB) deficiency reduces atherosclerosis in hyperlipidemic mice. Circulation. 2010;121:1124–33. doi: 10.1161/CIRCULATIONAHA.109.882704. [DOI] [PubMed] [Google Scholar]
- 16.Snell LM, Lin GH, McPherson AJ, Moraes TJ, Watts TH. T-cell intrinsic effects of GITR and 4-1BB during viral infection and cancer immunotherapy. Immunol Rev. 2011;244:197–217. doi: 10.1111/j.1600-065X.2011.01063.x. [DOI] [PubMed] [Google Scholar]
- 17.Ascierto PA, Simeone E, Sznol M, Fu YX, Melero I. Clinical experiences with anti-CD137 and anti-PD1 therapeutic antibodies. Semin Oncol. 2010;37:508–16. doi: 10.1053/j.seminoncol.2010.09.008. [DOI] [PubMed] [Google Scholar]
- 18.International HapMap Consortium. The International HapMap Project. Nature. 2003;426:789–96. doi: 10.1038/nature02168. [DOI] [PubMed] [Google Scholar]
- 19.Kwan T, et al. Genome-wide analysis of transcript isoform variation in humans. Nat Genet. 2008;40:225–31. doi: 10.1038/ng.2007.57. [DOI] [PubMed] [Google Scholar]
- 20.Baldassarre D, et al. Measurements of carotid intima-media thickness and of interadventitia common carotid diameter improve prediction of cardiovascular events: results of the IMPROVE (Carotid Intima Media Thickness [IMT] and IMT-Progression as Predictors of Vascular Events in a High Risk European Population) study. J Am Coll Cardiol. 2012;60:1489–99. doi: 10.1016/j.jacc.2012.06.034. [DOI] [PubMed] [Google Scholar]
- 21.Gertow K, et al. Identification of the BCAR1-CFDP1-TMEM170A locus as a determinant of carotid intima-media thickness and coronary artery disease risk. Circ Cardiovasc Genet. 2012;5:656–65. doi: 10.1161/CIRCGENETICS.112.963660. [DOI] [PubMed] [Google Scholar]
- 22.Farrall M, et al. Genome-wide mapping of susceptibility to coronary artery disease identifies a novel replicated locus on chromosome 17. PLoS. Genet. 2006;2:e72. doi: 10.1371/journal.pgen.0020072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Newton-Cheh C, et al. Genome-wide association study identifies eight loci associated with blood pressure. Nat Genet. 2009;41:666–76. doi: 10.1038/ng.361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Li Y, Willer CJ, Ding J, Scheet P, Abecasis GR. MaCH: using sequence and genotype data to estimate haplotypes and unobserved genotypes. Genet Epidemiol. 2010;34:816–34. doi: 10.1002/gepi.20533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Purcell S, et al. PLINK: a tool set for whole-genome association and population-based linkage analyses. Am J Hum Genet. 2007;81:559–75. doi: 10.1086/519795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Olofsson PS. Targeting T cell costimulation to prevent atherothrombosis. Circ Res. 2012;110:800–1. doi: 10.1161/CIRCRESAHA.112.265108. [DOI] [PubMed] [Google Scholar]
- 27.Haast RA, Gustafson DR, Kiliaan AJ. Sex differences in stroke. J Cereb Blood Flow Metab. 2012;32:2100–7. doi: 10.1038/jcbfm.2012.141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Lawton JS. Sex and gender differences in coronary artery disease. Semin Thorac Cardiovasc Surg. 2011;23:126–30. doi: 10.1053/j.semtcvs.2011.07.006. [DOI] [PubMed] [Google Scholar]
- 29.Malarstig A, et al. Genetic variants of tumor necrosis factor superfamily, member 4 (TNFSF4), and risk of incident atherothrombosis and venous thromboembolism. Clin Chem. 2008;54:833–40. doi: 10.1373/clinchem.2007.096479. [DOI] [PubMed] [Google Scholar]
- 30.Kong M, Kim Y, Lee C. Promoter sequence variants of LIGHT are associated with female vascular dementia. J Biomed Sci. 2008;15:545–52. doi: 10.1007/s11373-008-9242-2. [DOI] [PubMed] [Google Scholar]
- 31.Wang X, et al. Positional identification of TNFSF4, encoding OX40 ligand, as a gene that influences atherosclerosis susceptibility. Nat Genet. 2005;37:365–72. doi: 10.1038/ng1524. [DOI] [PubMed] [Google Scholar]
- 32.Olofsson PS, et al. Genetic variants of TNFSF4 and risk for carotid artery disease and stroke. J Mol Med (Berl) 2009;87:337–46. doi: 10.1007/s00109-008-0412-5. [DOI] [PubMed] [Google Scholar]
- 33.VanderLaan PA, Reardon CA, Getz GS. Site specificity of atherosclerosis: site-selective responses to atherosclerotic modulators. Arterioscler Thromb Vasc Biol. 2004;24:12–22. doi: 10.1161/01.ATV.0000105054.43931.f0. [DOI] [PubMed] [Google Scholar]
- 34.Jashari F, et al. Coronary and carotid atherosclerosis: Similarities and differences. Atherosclerosis. 2013;227:193–200. doi: 10.1016/j.atherosclerosis.2012.11.008. [DOI] [PubMed] [Google Scholar]
- 35.So T, Lee SW, Croft M. Immune regulation and control of regulatory T cells by OX40 and 4-1BB. Cytokine Growth Factor Rev. 2008;19:253–62. doi: 10.1016/j.cytogfr.2008.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.de Boer OJ, van der Meer JJ, Teeling P, van der Loos CM, van der Wal AC. Low numbers of FOXP3 positive regulatory T cells are present in all developmental stages of human atherosclerotic lesions. PLoS One. 2007;2:e779. doi: 10.1371/journal.pone.0000779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Ait-Oufella H, et al. Natural regulatory T cells control the development of atherosclerosis in mice. Nat Med. 2006;12:178–80. doi: 10.1038/nm1343. [DOI] [PubMed] [Google Scholar]
- 38.Hansson GK, Libby P. The immune response in atherosclerosis: a double-edged sword. Nat Rev Immunol. 2006;6:508–19. doi: 10.1038/nri1882. [DOI] [PubMed] [Google Scholar]
- 39.Bots ML, Sutton-Tyrrell K. Lessons from the past and promises for the future for carotid intima-media thickness. J Am Coll Cardiol. 2012;60:1599–604. doi: 10.1016/j.jacc.2011.12.061. [DOI] [PubMed] [Google Scholar]
- 40.Predazzi IM, et al. Association between OLR1 K167N SNP and intima media thickness of the common carotid artery in the general population. PLoS One. 2012;7:e31086. doi: 10.1371/journal.pone.0031086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Shah PK. Screening asymptomatic subjects for subclinical atherosclerosis: can we, does it matter, and should we? J Am Coll Cardiol. 2010;56:98–105. doi: 10.1016/j.jacc.2009.09.081. [DOI] [PubMed] [Google Scholar]