Abstract
Regenerative medicine is energizing and empowering basic science and has the potential to dramatically transform health care in the future. Given the remarkable intrinsic regenerative properties of the liver, as well as widespread adoption of regenerative strategies for liver disease (eg, liver transplant, partial hepatectomy, living donor transplant), hepatology has always been at the forefront of clinical regenerative medicine. However, an expanding pool of patients awaiting liver transplant, a limited pool of donor organs, and finite applicability of the current surgical approaches have created a need for more refined and widely available regenerative medicine strategies. Although cell-based therapies have been used extensively for hematologic malignant diseases and other conditions, the potential application of cellular therapy for acute and chronic liver diseases has only more recently been explored. New understanding of the mechanisms of liver regeneration and repair, including activation of local stem/progenitor cells and contributions from circulating bone marrow–derived stem cells, provide the theoretical underpinnings for the rational use of cell-based therapies in clinical trials. In this review, we dissect the scientific rationale for various modalities of cell therapy for liver diseases being explored in animal models and review those tested in human clinical trials. We also attempt to clarify some of the important ongoing questions that need to be addressed in order to bring these powerful therapies to clinical translation. Discussions will cover transplant of hepatocytes and liver stem/progenitor cells as well as infusion or stimulation of bone marrow–derived stem cells. We also highlight tremendous scientific advances on the horizon, including the potential use of induced pluripotent stem cells and their derivatives as individualized regenerative therapy for liver disease.
We are now living in a golden age of regenerative and individualized medicine in which sweeping scientific advances are poised to fundamentally alter the way we approach health and disease, as well as the delivery of medical therapies. In this new era, there is a growing armamentarium of therapeutic options that may benefit patients with acute or chronic liver disease. For the past 30 years, many patients with end-stage liver disease (ESLD) have benefitted from liver transplant as a treatment option. As a regenerative medicine option designed to “replace” a failing liver, liver transplant has transformed the care of patients with liver disease and the practice of hepatology. However, due in part to epidemic levels of chronic hepatitis C virus infection and nonalcoholic fatty liver disease, the applicability of this lifesaving procedure has now become more limited because of a mismatch between the number of patients awaiting liver transplant and the availability of suitable donor organs. Thus, the fatality rate of patients on the waiting list for liver transplant can be as high as 20%, depending on the severity of the underlying hepatic disease and the availability of organ donors in a specific United Network for Organ Sharing region.1
The remarkable innate ability of the liver to regenerate and the advent of living donor liver transplant have partially addressed the shortage of organs for transplant. In this way, transplant hepatology has always been at the forefront of clinical regenerative medicine. However, the limited applicability of current surgical paradigms has continued to stimulate extensive research into other approaches in the realm of liver regenerative medicine,2,3 including the enticing and seemingly limitless potential of cell-based therapies.
In this review, we focus on the potential role of various modalities of cellular therapy as a means to “repair” or “regenerate” a failing liver or to augment native liver regeneration after hepatectomy or living donor liver transplant. We begin with discussions of hepatocyte and liver stem/progenitor cell (LSPC) transplant. Thereafter, we review the use of circulating or bone marrow–derived stem cell therapies for chronic liver disease, including a review of the clinical trials to date. We conclude with a discussion of the future of cell-based therapy in hepatology, including the astonishing diagnostic and therapeutic potential of induced pluripotent stem cells (iPSCs) and their derivatives in liver disease. We will not address artificial and bioartificial liver support devices, which are outside the scope of the current review and have been reviewed in detail elsewhere.4
HEPATOCYTE TRANSPLANT
Initial attempts at cellular therapy for liver disease consisted of using primary hepatocytes infused via the portal vein to patients with ESLD or certain genetic and metabolic liver disorders.5–12 Various reports have indicated a beneficial effect. However, the observed improvements in liver function are rather modest and of uncertain duration. The hepatocytes are typically harvested from livers that are not deemed to be suitable for liver transplant, but these cells are limited in number, variable in quality, and not able to be expanded in vitro. The ability of hepatocytes to effectively repopulate a diseased liver appears to be limited to a select group of disorders that allow a growth advantage to the transplanted cells (such as hereditary tyrosinemia, Wilson disease, or progressive familial intrahepatic cholestasis).13 Furthermore, these procedures continue to require immunosuppression, and there has been insufficient experience to define the amount and duration of immunosuppression needed in this setting. Lastly, how long these hepatocytes will be viable and the nature of their interaction with native hepatocytes remain unclear. All these factors have conspired against making primary hepatocyte transplant a current option for patients with ESLD or metabolic/genetic disorders. There is a growing body of literature seeking alternative sources of abundant, high-quality hepatocytes for transplant in patients with acute liver failure, chronic liver disease, and during regeneration after large hepatic resections.14,15 Several approaches are in development, including hepatocytes derived from cell lines, xenotransplant of animal-derived hepatocytes, and even in vivo expansion of human hepatocytes in fumarylacetoacetate hydrolase-deficient animal incubators.16–18 Although these approaches are promising, further basic science advances will be needed before these methods can be translated to human studies.
LIVER STEM/PROGENITOR CELLS
Liver stem/progenitor cells (also known as oval cells in rodents) are thought to represent tissue-specific, bipotential precursors to liver parenchymal cells. When hepatocyte replication is impaired or overwhelmed, the LSPCs residing in the terminal bile ductules (canals of Hering) are activated to proliferate and differentiate. Numerous studies have investigated the activation of the liver stem cell compartment in various forms of chronic liver disease,19 including chronic viral hepatitis,20–23 alcoholic liver disease,23,24 and fatty liver disease.25,26 Although they are considered to have the ability to become hepatocytes or cholangiocytes, the true ability of LSPCs to transdifferentiate into mature hepatocytes and the extent to which this process might contribute to liver regeneration and repair in various disease states are not entirely clear. Nonetheless, methods to isolate and characterize liver-specific stem cells have been developed in fetal and adult rodent models.27–31 Furthermore, bipotential mouse embryonic liver cell lines have been developed that retain the ability to undergo morphogenesis into hepatocytes or cholangiocytes in vitro.32,33 Directed differentiation techniques have also allowed generation of hepatic progenitor cells from human embryonic stem cells.34 Transplant of LSPCs from various sources has been accomplished via intrasplenic injection or infusion into a peripheral vein or the portal vein, with the idea that they may augment the impaired regeneration seen in the setting of chronic liver disease and promote reverse remodeling of fibrosis.35 Engraftment and repopulation have been observed, even in the setting of fibrosis,27,36 but generally, a regenerative stimulus such as partial hepatectomy or retrorsine injection is required for optimal engraftment. Although some studies suggest reduced fibrosis after LSPC transplant,36 there have also been descriptions of a severe fibrogenic response that is, in fact, driven by the activation of the hepatic progenitor compartment.37 Furthermore, as in hepatocyte transplant, adult LSPCs are available only in limited numbers, and there are ethical constraints on the use of human fetal LSPCs. Thus, caution and additional study will be needed to clarify the therapeutic potential of LSPCs.
CIRCULATING STEM CELLS
Several publications have provided evidence suggesting that bone marrow–derived stem cells are mobilized after hepatic resection (ie, partial hepatectomy), inflammatory hepatic disease, or ischemic injury.38–41 Circulating bone marrow–derived stem cells consist of 2 major types of adult stem cells: hematopoietic stem cells (HSCs), which are CD34 and CD133 positive, and mesenchymal stem cells (MSCs) that lack a well-defined surface antigen expression pattern and can also be found in adipose tissue.42 True pluripotent stem cells present in bone marrow are estimated to be less than 0.1% of CD133+ cells. The migration of these stem cells appears to be mediated by a chemoattractant, such as stromal cell–derived factor 1.43 Subsequently, the secretion of interleukin 8, matrix metallo-proteinase 9, hepatocyte growth factor, and stem cell factors facilitates homing and engraftment of MSCs44,45 in the liver.
The underlying mechanisms of the beneficial effect observed after the infusion of HSCs and MSCs have not been well characterized. Trans-differentiation into hepatocytes, stimulation of native hepatocyte proliferation, an antifibrotic effect, immunomodulatory effects, and cell plasticity are all possible mechanisms involved in this beneficial effect. Initially, several investigators postulated that the mobilized bone marrow–derived stem cells were able to trans-differentiate into hepatocytes. Other interpretations for their beneficial effects have attributed them to fusion of adult stem cells with local hepatocytes46 or a paracrine proliferative effect on native hepatocytes.47 Another postulated mechanism of action is an ability to remodel fibrosis.48 Specifically, endogenous hepatocytes in cirrhosis have been reported to have a decreased proliferative capacity. Stem cell therapy may initially exert a beneficial effect by the expression of matrix metalloproteinase 9 to decrease fibrosis. As fibrosis diminishes, local hepatocytes in a cirrhotic liver may regain their ability to proliferate.
Houlihan and Newsome49 have recently described some potential adverse events associated with this type of cell therapy in patients with liver diseases. Hepatic stellate cells and myofibroblasts may derive from bone marrow stem cells.50 These observations raise the possibility that cell therapies may have the potential to enhance, not diminish, hepatic fibrosis. It has also been pointed out that MSCs can undergo malignant transformation.51
The approaches utilized in the preparation of bone marrow–derived stem cells for clinical use have included infusion of collected autologous stem cells and mobilization of bone marrow stem cells by the administration of granulocyte colony-stimulating factor (GC-SF).
CLINICAL TRIALS WITH MSCs
The initial 2 reported pilot studies52,53 included a total of 12 patients and suggested a beneficial effect after the infusion of an autologous MSC preparation (Table 1). The authors described improvement of the model for end-stage liver disease (MELD) score, quality of life (by 36-Item Short-Form Health Survey measurement), and serum albumin levels and decreasing prothrombin time. Subsequently, there have been 4 randomized controlled trials performed; 3 of them54–56 reported beneficial effects associated with MSCs, characterized by less ascites, decreased MELD score, increased serum albumin level, and decreased total bilirubin level. There was no observed improvement in patient survival during the time of observation. Another randomized controlled trial57 did not show any significant improvement between the treatment and control groups.
TABLE 1.
Trials of Mesenchymal Stem Cell Transplant in Patients With Chronic Liver Diseases
| Reference | Cell therapy | Dose, route | No. of patients | Type of study | Results |
|---|---|---|---|---|---|
| Amer et al,54 2011 | BM-MSC (bone marrow–derived hepatocytes) | Single dose, intrahepatic, intrasplenic | 10 Intrahepatic 10 Intrasplenic 20 Controls |
Controlled trial | Less ascites/edema, increased albumin |
| Zhang et al,56 2012 | UC-MSC | Multiple doses, peripheral vein | 30 Treatment 15 Controls |
Randomized controlled trial | Less ascites, decreased MELD |
| Mohamadnejad et al,52 2007 | BM-MSC | Single dose, peripheral vein | 4 | Uncontrolled trial | Decreased MELD in 2 of 4 patients |
| Kharaziha et al,53 2009 | BM-MSC | Single dose, portal vein | 8 | Uncontrolled trial | Decreased MELD |
| Peng et al,55 2011 | BM-MSC | Single dose, hepatic artery | 53 Treatment 105 Controls |
Randomized controlled trial | Decreased T Bil, improved INR and MELD score |
| Mohamadnejad et al,57 2013 | BM-MSC | Single dose, peripheral vein | 15 Treatment 12 Placebo |
Randomized controlled trial | No differences between the groups |
BM-MSC = bone marrow–derived mesenchymal stem cells; INR = international normalized ratio; MELD = model of end-stage liver disease; T Bil = total bilirubin; UC-MSC = umbilical cord-derived mesenchymal stem cell.
These trials utilized different doses of MSCs and different routes of administration. Amer et al54 administered an average of 2 × 107 “hepatic lineage–committed” cells in a 5-mL cell suspension that was injected intrasplenically or intrahepatically with ultrasonographic guidance. Peng et al55 did not provide the exact number of cells that were injected into the hepatic artery. Zhang et al56 administered 0.5 × 106 cells/kg intravenously every 4 weeks three times. Mohamadnejad et al57 administered one median dose of 1.95 × 108 cells intravenously. This variability of protocols precludes a comprehensive comparison among these trials and the ability to draw conclusions in terms of a preferred dose and route of administration.
Mohamadnejad et al,57 who have pioneered the use MSCs in compensated or early decompensated cirrhosis, recently reported a randomized placebo-controlled trial in decompensated cirrhosis. They studied 27 patients, 15 of whom were randomized to MSCs. At the end of the trial, they found that Child-Pugh-Turcotte (CPT) score, MELD score, serum albumin level, international normalized ratio, and serum aminotransferase level were not different between the groups. Thus, they were unable to document a beneficial effect of MSC therapy administered via a peripheral vein. They suggested that as a next step, repeated infusion of MSCs via the hepatic artery or portal vein should be evaluated in the setting of a randomized placebo-controlled trial.
CLINICAL TRIALS WITH AUTOLOGOUS BONE MARROW–DERIVED STEM CELLS
Autologous bone marrow stem cells have been evaluated in 10 studies; 6 of them (Table 2) utilized unsorted bone marrow–derived mononuclear cells.58–63 The clinical conditions treated with this approach included cirrhosis associated with hepatitis C virus or hepatitis B virus, alcoholic liver disease, primary sclerosing cholangitis, drug-induced acute liver failure, cryptogenic cirrhosis, and decompensated cirrhosis. The end points utilized to assess the efficacy of cellular therapy included CPT score, quality of life, and improvement in albumin, bilirubin, and aminotransferase levels and prothrombin time. Most of the patients had improvement in the measured parameters. One patient died of sepsis, and one patient had development of hepatorenal syndrome that led to discontinuation of the trial.64 Three of the studies were randomized controlled trials, 2 of which revealed efficacy61,62 and one that did not.63
TABLE 2.
Trials of Unsorted Bone Marrow–Derived Mononuclear Cell Transplant in Patients With Chronic Liver Diseases
| Reference | Cell therapy | Dose, route | No. of patients | Type of study | Results |
|---|---|---|---|---|---|
| Lyran et al,58 2007 | BM-MNC | Single dose, hepatic artery | 10 | Uncontrolled trial | Decreased T Bil and INR, increased serum albumin |
| Terai et al,59 2006 | BM-MNC | Single dose, peripheral vein | 9 | Uncontrolled trial | Improved serum albumin, total protein, CP score |
| Kim et al,60 2010 | BM-MNC | Single dose, peripheral vein | 10 | Uncontrolled trial | Less ascites, improved CP scores, increased liver volume |
| Saito et al,61 2011 | BM-MNC | Single dose, peripheral vein | 5 Treatment 5 Controls |
Randomized controlled trial | Improved CP scores and INR, higher serum albumin and total protein |
| Lyra et al,62 2010 | BM-MNC | Single dose, hepatic artery | 15 Treatment 15 Controls |
Randomized controlled trial | Improved serum albumin and CP score |
| Spahr et al,63 2013 | BM-MNC + GC-SF | Single dose, hepatic artery | 28 Treatment 30 Controls |
Randomized controlled trial | No significant differences between study groups |
BM-MNC = bone marrow–derived mononuclear cells; CP = Child-Pugh; GC-SF = granulocyte colony-stimulating factor; INR = international normalized ratio; T Bil = total bilirubin.
Spahr et al63 recently reported a randomized trial in 58 patients with decompensated alcoholic liver disease. Thirty patients were randomized to standard medical care alone, and 28 patients received a combination of GC-SF injections and autologous bone marrow mono-nuclear cell transplant. Their primary end point was a decrease of 3 or more points in the MELD score. They found no significant differences between the groups; the MELD score improved in 64% of the patients who received GC-SF and autologous bone marrow mononuclear cell transplant vs 53% among those randomized to standard medical care. As possible explanations for the lack of therapeutic effect, they pointed out that they were not able to document an expansion of the hepatic progenitor cell compartment in a 4-week liver biopsy sample, and about 31% of the patients had an alcohol relapse during therapy. They attributed the lack of a response to therapy by hepatocytes to the presence of concomitant cirrhosis.
Sorted HSCs, specifically CD34+ cells, have been used in 4 pilot studies64–67 in patients with chronic liver diseases (Table 3). These uncontrolled studies showed improvement of liver test results and CP scores.
TABLE 3.
Trials of Sorted Hematopoietic Stem Cell Transplant in Patients With Chronic Liver Diseases
| Reference | Cell therapy | Dose, route | No. of patients | Type of study | Results |
|---|---|---|---|---|---|
| Gordan et al,65 2006 | CD34+ | Single dose, portal vein or hepatic artery | 5 | Uncontrolled trial | Serum albumin improved, T Bil improved |
| Mohamadnejad et al,66 2007 | CD34+ | Single dose, hepatic artery | 4 | Uncontrolled trial | Serum albumin, INR, T Bil improved |
| Pai et al,64 2008 | CD34+ | Single dose, hepatic artery | 9 | Uncontrolled trial | CP score improved, T Bil decreased |
| Levicar et al,67 2008 | CD34+ | Single dose, hepatic artery | 5 | Uncontrolled trial | Improved T Bil |
CP = Child-Pugh; INR = international normalized ratio; T Bil = total bilirubin.
CLINICAL TRIALS WITH GC-SF
There was one controlled trial with CD133+ cells in a different clinical setting in which cell therapy was used as a mechanism to increase liver volume growth before partial hepatectomy in patients with hepatic metastatic disease.68 Another approach has been to use GC-SF injection to increase the production of HSCs. There have been 7 studies utilizing GC-SF69–75 (Table 4), although one of them was a case report of a patient with drug-induced acute liver failure72 (not included in Table 4). Two of the studies74,75 were randomized controlled studies, both of which reported efficacy; however, they differed in terms of patient population studied and dose administered.
TABLE 4.
Trials of GC-SF–Mobilized Hematopoietic Stem Cell Transplant in Patients With Chronic Liver Diseases
| Reference | Cell therapy | Dose, route | No. of patients | Type of study | Results |
|---|---|---|---|---|---|
| Yannaki et al,69 2006 | GC-SF/PBMNC | Single dose, hepatic artery | 2 | Uncontrolled trial | CP score improved, MELD score improved |
| Gaia et al,70 2006 | GC-SF | Multiple doses of GC-SF | 8 | Uncontrolled trial | Feasibility, safety study |
| Yan et al,71 2007 | GC-SF/HGF | Unclear | 2 | Uncontrolled trial | PBMNCs were transformed in hepatocyte-like cells |
| Khan et al,73 2007 | GC-SF/CD34+ | Single dose, hepatic artery | 4 | Uncontrolled trial | Improved serum albumin, T Bil, ALT |
| Han et al,74 2008 | GC-SF/PBMNC | Single dose, hepatic artery | 20 GC-SF plus PBMNC infusion 20 GC-SF |
Randomized controlled trial | GC-SF plus PBMNC group had better liver test results |
| Garg et al,75 2012 | GC-SF | Multiple doses | 23 Treatment 24 Placebo |
Randomized, blinded, controlled trial | Improved MELD score, better patient survival |
ALT = alanine aminotransferase; CP = Child-Pugh; GC-SF = granulocyte colony-stimulating factor; HGF = hepatocyte growth factor; MELD = model of end-stage liver disease; PBMNC = peripheral blood mononuclear cells; T Bil = total bilirubin.
Garg et al75 evaluated GC-SF therapy in the setting of acute-on-chronic liver failure. Forty-seven consecutive patients with acute-on-chronic liver failure were randomized to receive 12 doses of GC-SF (5 μg/kg subcutaneously) or placebo. Of the 23 patients who received GC-SF therapy, 16 (69.6%) survived, compared with 7 (29.2%) of 24 patients who received placebo. Actuarial survival at 60 days was 66% vs 26% (P=.001). There was also significant improvement in CPT scores, MELD scores, and Sequential Organ Failure Assessment scores associated with GC-SF therapy. The authors also observed a lower incidence of hepatorenal syndrome, hepatic encephalopathy, or sepsis in the GC-SF therapy group. They also reported a significant increase of the CD34+ cell population in the liver associated with GC-SF therapy.
In these pilot studies, as well as randomized trials, there were no unexpected serious adverse events that could be attributed to cell therapy. Worsening hepatic fibrosis and a possible increased risk of hepatocellular carcinoma associated with stem cell therapy should be monitored for a longer period of time in future trials.
The preliminary results obtained with various modalities of adult stem cell therapy in patients with ESLD are promising. It is difficult to reach a definite conclusion on its utility in the setting of cirrhosis. Future clinical trials should standardize the cell preparations utilized. Thus far, investigators have used bone marrow–derived mononuclear cells, bone marrow MSCs, bone marrow HSCs, peripheral blood mononuclear cells from GC-SF–mobilized peripheral blood, CD34+ cells, and CD133+ cells, which makes comparison among studies quite challenging. The period of administration has also been variable; it ranges from one infusion to multiple infusions in variable periods of time.
These cell therapies have been administered to patients with different underlying etiologies and variable degrees of compensation. The mechanism of action of cell therapies may differ among different etiologies and degrees of decompensation.
iPSCs and the Future of Cell Therapies in Hepatology
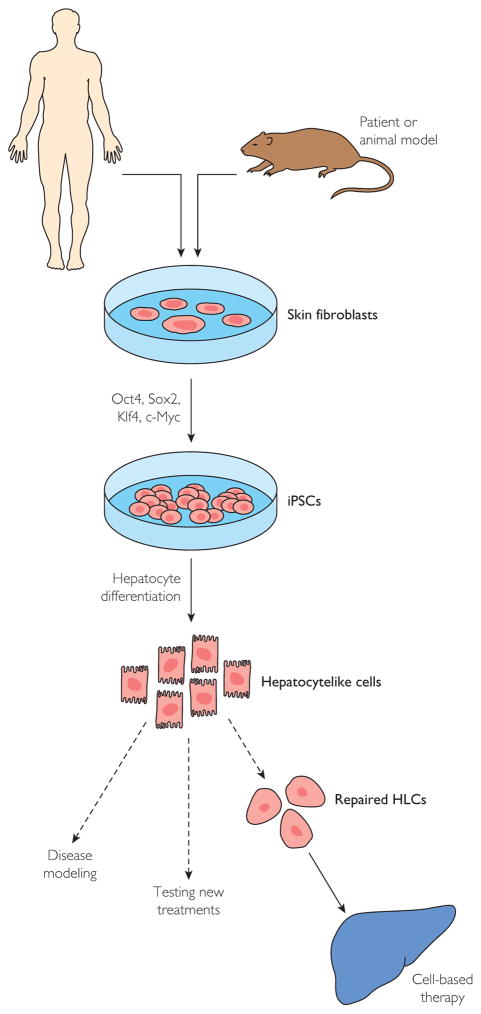
There is a growing body of evidence from experimental models of liver injury supporting the use of hepatocytes derived from embryonic stem cells.76,77 However, ethical barriers limit the widespread use of embryonic stem cell technology. Recently, however, the Nobel Prize–winning discovery of the pluripotency factors78 has revealed remarkable cellular plasticity in cells previously dogmatically considered to be “terminally differentiated.” Conceptually based on somatic cell nuclear transfer technology,79 it is now possible to generate iPSCs from virtually any tissue in the human body and to then recapitulate developmental biology in vitro to generate diverse cellular phenotypes.80 To accomplish this, cells from a patient or an experimental animal model (typically skin fibroblasts obtained via a skin biopsy) are expanded in culture and subjected to “reprogramming” by enforced expression of a limited number of pluripotency factors (eg, Oct 3/4, Sox2, Klf4, and c-Myc), which collectively revert the somatic cells back to a pluripotent state (Figure 1).
FIGURE 1.
Derivation and use of induced pluripotent stem cells (iPSCs) for liver diseases. HLCs = hepatocytelike cells.
On the basis of known developmental biology of the liver, several groups have developed methods for generating hepatocytelike cells (HLCs) from iPSCs via stepwise differentiation strategies through definitive endoderm, hepatic specification, and hepatocyte maturation.81–85 Direct differentiation from fibroblasts to either hepatocytes or bipotent hepatic cells using defined factors (without a pluripotent intermediate) have also been described.86,87 The HLCs generated in this fashion express hepatocyte proteins (eg, albumin) and share synthetic (urea) and metabolic (cytochrome P-450) features of mature hepatocytes.14 These remarkable advances are paralleled by an astounding array of genetic information from large-scale sequencing efforts as well as improved methods for seamless genetic engineering.88,89 The incredible potential of these regenerative medicine technologies spans all of modern medicine and has been embraced as a strategic priority at the national level and among many academic institutions. Multiple groups have reported the ability to generate mature hepatic cell types from adult cells, to genetically modify them in culture, and to transplant and engraft these cells within the liver in vivo.90 The stunning corollary is that mature liver cells derived from patient-specific iPSCs could potentially be a limitless source of high-quality, individualized liver cells that can be (1) studied in vitro as a patient-specific model of liver disease, (2) treated in vitro to test putative therapeutic compounds, (3) genetically modified to correct underlying disease-causing defects, and (4) transplanted (without the need for immunosuppression) as individualized, cell-based, regenerative therapies for hepatic disorders.91 Indeed, HLCs originating from iPSCs have been used successfully to model several inherited metabolic liver disorders, including α1-antitrypsin deficiency, familial hypercholesterolemia, and hereditary tyrosinemia, among others.92 The iPSC-derived HLCs have also produced beneficial effects in experimental models of both liver injury and partial hepatectomy.93 Despite these promising advances, head-to-head comparisons of mature hepatocytes and iPSC-derived HLCs do continue to yield some notable differences in gene expression (eg, α-fetoprotein) and functionality, both in vitro83 and in vivo.94 These differences indicate that additional improvements may be needed before clinical application of these cell types can be considered. Additional study will also be needed to assuage theoretical concerns about tumorigenicity, teratoma formation, epigenetic memory of reprogrammed cells, and the unknown effects of potential stray genetic changes left over from the reprogramming process.
It may be possible to generate other liver cell types using iPSC technology as well, such as cholangiocytes, endothelial cells, and stellate cells, in order to modify chronic biliary disease, liver angiogenesis, and hepatic fibrogenesis. Indeed, cholangiocytic elements derived from both embryonic stem cells95 and iPSCs96,97 have been reported. Mature and functional iPSC-derived cholangiocytelike cells would be an important advancement given the complexities of incorporating biliary elements into organ buds or bioartificial organs.94,98 The concept of cholangiocyte transplant for repopulation or repair of a diseased biliary system is conceptually appealing given the clinical access offered by endoscopic retrograde cholangiopancreatography. Such technology could be revolutionary for patients with chronic biliary disorders such as primary sclerosing cholangitis, primary biliary cirrhosis, or ischemic cholangiopathy after transplant, all of which are essentially untreatable at this time without whole-organ transplant.
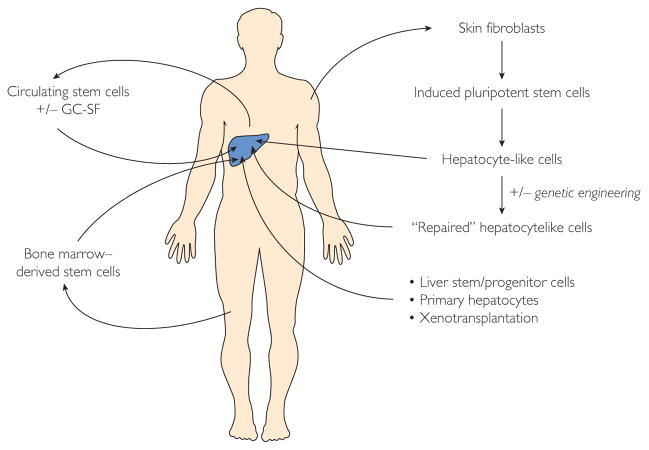
Numerous therapeutic modalities for cell-based therapy are being investigated for the treatment of liver disease (Figure 2), including cell transplant (hepatocytes, LSPCs, or HLCs), autologous transfer of circulating or bone marrow–derived stem cells, and stimulation of native stem cell compartments (ie, GC-SF).
FIGURE 2.
Various modalities of cell-based therapies for liver diseases. GC-SF= granulocyte colony-stimulating factor; +/− = with or without.
CONCLUSION
Over the past several years, there have been remarkable advancements in liver regenerative medicine including numerous promising observations at the basic science and translational levels. As a result, it appears that we are now on the cusp of new paradigms for the management of chronic liver disease including cell-based therapies. The field is ripe for ongoing basic science advancements as well as standardized and carefully designed clinical trials to bridge the final knowledge gaps and make these new therapies a reality for patients with ESLD.
Abbreviations and Acronyms
- CP
Child-Pugh
- ESLD
end-stage liver disease
- GC-SF
granulocyte colony-stimulating factor
- HLC
hepatocytelike cell
- HSC
hematopoietic stem cell
- iPSC
induced pluripotent stem cell
- LSPC
liver stem/progenitor cell
- MELD
model for end-stage liver disease
- MSC
mesenchymal stem cell
References
- 1.Dienstag JL, Cosimi AB. Liver transplantation—a vision realized. N Engl J Med. 2012;367(16):1483–1485. doi: 10.1056/NEJMp1210159. [DOI] [PubMed] [Google Scholar]
- 2.Terzic A, Nelson TJ. Regenerative medicine primer. Mayo Clin Proc. 2013;88(7):766–775. doi: 10.1016/j.mayocp.2013.04.017. [DOI] [PubMed] [Google Scholar]
- 3.Matteson EL, Terzic A. Introduction to the Symposium on Regenerative Medicine. Mayo Clin Proc. 2013;88(7):645–646. doi: 10.1016/j.mayocp.2013.04.013. [DOI] [PubMed] [Google Scholar]
- 4.McKenzie TJ, Lillegard JB, Nyberg SL. Artificial and bioartificial liver support. Semin Liver Dis. 2008;28(2):210–217. doi: 10.1055/s-2008-1073120. [DOI] [PubMed] [Google Scholar]
- 5.Dhawan A, Puppi J, Hughes RD, Mitry RR. Human hepatocyte transplantation: current experience and future challenges. Nat Rev Gastroenterol Hepatol. 2010;7(5):288–298. doi: 10.1038/nrgastro.2010.44. [DOI] [PubMed] [Google Scholar]
- 6.Weber A, Groyer-Picard MT, Franco D, Dagher I. Hepatocyte transplantation in animal models. Liver Transpl. 2009;15(1):7–14. doi: 10.1002/lt.21670. [DOI] [PubMed] [Google Scholar]
- 7.Ito M, Nagata H, Miyakawa S, Fox IJ. Review of hepatocyte transplantation. J Hepatobiliary Pancreat Surg. 2009;16(2):97–100. doi: 10.1007/s00534-008-0023-0. [DOI] [PubMed] [Google Scholar]
- 8.Fox IJ, Roy-Chowdhury J. Hepatocyte transplantation. J Hepatol. 2004;40(6):878–886. doi: 10.1016/j.jhep.2004.04.009. [DOI] [PubMed] [Google Scholar]
- 9.Fox IJ, Chowdhury JR. Hepatocyte transplantation. Am J Transplant. 2004;4(suppl 6):7–13. doi: 10.1111/j.1600-6135.2004.0340.x. [DOI] [PubMed] [Google Scholar]
- 10.Horslen SP, McCowan TC, Goertzen TC, et al. Isolated hepatocyte transplantation in an infant with a severe urea cycle disorder. Pediatrics. 2003;111(6 pt 1):1262–1267. doi: 10.1542/peds.111.6.1262. [DOI] [PubMed] [Google Scholar]
- 11.Strom SC, Chowdhury JR, Fox IJ. Hepatocyte transplantation for the treatment of human disease. Semin Liver Dis. 1999;19(1):39–48. doi: 10.1055/s-2007-1007096. [DOI] [PubMed] [Google Scholar]
- 12.Fox IJ, Chowdhury JR, Kaufman SS, et al. Treatment of the Crigler-Najjar syndrome type I with hepatocyte transplantation. N Engl J Med. 1998;338(20):1422–1426. doi: 10.1056/NEJM199805143382004. [DOI] [PubMed] [Google Scholar]
- 13.Rountree CB, Mishra L, Willenbring H. Stem cells in liver diseases and cancer: recent advances on the path to new therapies. Hepatology. 2012;55(1):298–306. doi: 10.1002/hep.24762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Yu Y, Fisher JE, Lillegard JB, Rodysill B, Amiot B, Nyberg SL. Cell therapies for liver diseases. Liver Transpl. 2012;18(1):9–21. doi: 10.1002/lt.22467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Duncan AW, Dorrell C, Grompe M. Stem cells and liver regeneration. Gastroenterology. 2009;137(2):466–481. doi: 10.1053/j.gastro.2009.05.044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Chistiakov DA. Liver regenerative medicine: advances and challenges. Cells Tissues Organs. 2012;196(4):291–312. doi: 10.1159/000335697. [DOI] [PubMed] [Google Scholar]
- 17.Forbes SJ, Newsome PN. New horizons for stem cell therapy in liver disease. J Hepatol. 2012;56(2):496–499. doi: 10.1016/j.jhep.2011.06.022. [DOI] [PubMed] [Google Scholar]
- 18.Fisher JE, Lillegard JB, McKenzie TJ, Rodysill BR, Wettstein PJ, Nyberg SL. In utero transplanted human hepatocytes allow postnatal engraftment of human hepatocytes in pigs. Liver Transpl. 2013;19(3):328–335. doi: 10.1002/lt.23598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Lanthier N, Rubbia-Brandt L, Spahr L. Liver progenitor cells and therapeutic potential of stem cells in human chronic liver diseases. Acta Gastroenterol Belg. 2013;76(1):3–9. [PubMed] [Google Scholar]
- 20.Libbrecht L, Desmet V, Van Damme B, Roskams T. Deep intra-lobular extension of human hepatic ‘progenitor cells’ correlates with parenchymal inflammation in chronic viral hepatitis: can ‘progenitor cells’ migrate? J Pathol. 2000;192(3):373–378. doi: 10.1002/1096-9896(2000)9999:9999<::AID-PATH700>3.0.CO;2-5. [DOI] [PubMed] [Google Scholar]
- 21.Wu N, Liu F, Ma H, et al. HBV infection involving in hepatic progenitor cells expansion in HBV-infected end-stage liver disease. Hepatogastroenterology. 2009;56(93):964–967. [PubMed] [Google Scholar]
- 22.Clouston AD, Powell EE, Walsh MJ, Richardson MM, Demetris AJ, Jonsson JR. Fibrosis correlates with a ductular reaction in hepatitis C: roles of impaired replication, progenitor cells and steatosis. Hepatology. 2005;41(4):809–818. doi: 10.1002/hep.20650. [DOI] [PubMed] [Google Scholar]
- 23.Lowes KN, Brennan BA, Yeoh GC, Olynyk JK. Oval cell numbers in human chronic liver diseases are directly related to disease severity. Am J Pathol. 1999;154(2):537–541. doi: 10.1016/S0002-9440(10)65299-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Sancho-Bru P, Altamirano J, Rodrigo-Torres D, et al. Liver progenitor cell markers correlate with liver damage and predict short-term mortality in patients with alcoholic hepatitis. Hepatology. 2012;55(6):1931–1941. doi: 10.1002/hep.25614. [DOI] [PubMed] [Google Scholar]
- 25.Richardson MM, Jonsson JR, Powell EE, et al. Progressive fibrosis in nonalcoholic steatohepatitis: association with altered regeneration and a ductular reaction. Gastroenterology. 2007;133(1):80–90. doi: 10.1053/j.gastro.2007.05.012. [DOI] [PubMed] [Google Scholar]
- 26.Chiba M, Sasaki M, Kitamura S, Ikeda H, Sato Y, Nakanuma Y. Participation of bile ductular cells in the pathological progression of non-alcoholic fatty liver disease. J Clin Pathol. 2011;64(7):564–570. doi: 10.1136/jcp.2011.090175. [DOI] [PubMed] [Google Scholar]
- 27.Oertel M. Fetal liver cell transplantation as a potential alternative to whole liver transplantation? J Gastroenterol. 2011;46(8):953–965. doi: 10.1007/s00535-011-0427-5. [DOI] [PubMed] [Google Scholar]
- 28.Takana M, Miyajima A. Identification and isolation of adult liver stem/progenitor cells. Methods Mol Biol. 2012;826:25–32. doi: 10.1007/978-1-61779-468-1_3. [DOI] [PubMed] [Google Scholar]
- 29.Guo J, Li J, Lu Y, et al. A novel technique for hepatic progenitor cell isolation from normal adult rat livers. ASAIO J. 2012;58(1):73–78. doi: 10.1097/MAT.0b013e318239fce5. [DOI] [PubMed] [Google Scholar]
- 30.Sahin MB, Schwartz RE, Buckley SM, et al. Isolation and characterization of a novel population of progenitor cells from unmanipulated rat liver. Liver Transpl. 2008;14(3):333–345. doi: 10.1002/lt.21380. [DOI] [PubMed] [Google Scholar]
- 31.Aravalli RN, Behnan Sahin M, Cressman EN, Steer CJ. Establishment and characterization of a unique 1 microm diameter liver-derived progenitor cell line. Biochem Biophys Res Commun. 2010;391(1):56–62. doi: 10.1016/j.bbrc.2009.11.001. [DOI] [PubMed] [Google Scholar]
- 32.Strick-Marchand H, Weiss MC. Inducible differentiation and morphogenesis of bipotential liver cell lines from wild-type mouse embryos. Hepatology. 2002;36(4 pt 1):794–804. doi: 10.1053/jhep.2002.36123. [DOI] [PubMed] [Google Scholar]
- 33.Akkari L, Haouzi D, Binamé F, et al. Cell shape and TGF-beta signaling define the choice of lineage during in vitro differentiation of mouse primary hepatic precursors. J Cell Physiol. 2010;225(1):186–195. doi: 10.1002/jcp.22219. [DOI] [PubMed] [Google Scholar]
- 34.Zhao D, Chen S, Cai J, et al. Derivation and characterization of hepatic progenitor cells from human embryonic stem cells. PloS One. 2009;4(7):e6468. doi: 10.1371/journal.pone.0006468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Yovchev MI, Dabeva MD, Oertel M. Isolation, characterization, and transplantation of adult liver progenitor cells. Methods Mol Biol. 2013;976:37–51. doi: 10.1007/978-1-62703-317-6_4. [DOI] [PubMed] [Google Scholar]
- 36.Yovchev MI, Xue Y, Shafritz DA, Locker J, Oertel M. Repopulation of the fibrotic/cirrhotic rat liver by transplanted hepatic stem/progenitor cells and mature hepatocytes [published online ahead of print July 10, 2013] Hepatology. doi: 10.1002/hep.26615. http://dx.doi.org/10.1002/hep.26615. [DOI] [PMC free article] [PubMed]
- 37.Kuramitsu K, Sverdlov DY, Liu SB, et al. Failure of fibrotic liver regeneration in mice is linked to a severe fibrogenic response driven by hepatic progenitor cell activation. Am J Pathol. 2013;183(1):182–194. doi: 10.1016/j.ajpath.2013.03.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Cho KA, Woo SY, Seoh JY, Han HS, Ryu KH. Mesenchymal stem cells restore CCl4-induced liver injury by an antioxidative process. Cell Biol Int. 2012;36(12):1267–1274. doi: 10.1042/CBI20110634. [DOI] [PubMed] [Google Scholar]
- 39.Petersen BE, Bowen WC, Patrene KD, et al. Bone marrow as a potential source of hepatic oval cells. Science. 1999;284(5417):1168–1170. doi: 10.1126/science.284.5417.1168. [DOI] [PubMed] [Google Scholar]
- 40.Oh SH, Witek RP, Bae SH, et al. Bone marrow-derived hepatic oval cells differentiate into hepatocytes in 2-acetylaminofluorene/partial hepatectomy-induced liver regeneration. Gastroenterology. 2007;132(3):1077–1087. doi: 10.1053/j.gastro.2007.01.001. [DOI] [PubMed] [Google Scholar]
- 41.Thomas JA, Pope C, Wojtacha D, et al. Macrophage therapy for murine liver fibrosis recruits host effector cells improving fibrosis, regeneration, and function. Hepatology. 2011;53(6):2003–2015. doi: 10.1002/hep.24315. [DOI] [PubMed] [Google Scholar]
- 42.Seki A, Sakai Y, Komura T, et al. Adipose tissue-derived stem cells as a regenerative therapy for a mouse steatohepatitis-induced cirrhosis model. Hepatology. 2013;58(3):1133–1142. doi: 10.1002/hep.26470. [DOI] [PubMed] [Google Scholar]
- 43.Jin SZ, Meng XW, Han MZ, Sun X, Sun LY, Liu BR. Stromal cell derived factor-1 enhances bone marrow mononuclear cell migration in mice with acute liver failure. World J Gastroenterol. 2009;15(21):2657–2664. doi: 10.3748/wjg.15.2657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Li C, Kong Y, Wang H, et al. Homing of bone marrow mesenchymal stem cells mediated by sphingosine 1-phosphate contributes to liver fibrosis [published correction appears in J Hepatol. 2009;51(5):973] J Hepatol. 2009;50(6):1174–1183. doi: 10.1016/j.jhep.2009.01.028. [DOI] [PubMed] [Google Scholar]
- 45.Chen Y, Xiang LX, Shao JZ, et al. Recruitment of endogenous bone marrow mesenchymal stem cells towards injured liver. J Cell Mol Med. 2010;14(6B):1494–1508. doi: 10.1111/j.1582-4934.2009.00912.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Wang X, Willenbring H, Akkari Y, et al. Cell fusion is the principal source of bone-marrow-derived hepatocytes. Nature. 2003;422(6934):897–901. doi: 10.1038/nature01531. [DOI] [PubMed] [Google Scholar]
- 47.Alison MR, Islam S, Lim S. Stem cells in liver regeneration, fibrosis and cancer: the good, the bad and the ugly. J Pathol. 2009;217(2):282–298. doi: 10.1002/path.2453. [DOI] [PubMed] [Google Scholar]
- 48.Zhao DC, Lei JX, Chen R, et al. Bone marrow-derived mesenchymal stem cells protect against experimental liver fibrosis in rats. World J Gastroenterol. 2005;11(22):3431–3440. doi: 10.3748/wjg.v11.i22.3431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Houlihan DD, Newsome PN. Critical review of clinical trials of bone marrow stem cells in liver disease. Gastroenterology. 2008;135(2):438–450. doi: 10.1053/j.gastro.2008.05.040. [DOI] [PubMed] [Google Scholar]
- 50.Yang L, Chang N, Liu X, et al. Bone marrow-derived mesenchymal stem cells differentiate to hepatic myofibroblasts by transforming growth factor-β1 via sphingosine kinase/sphingosine 1-phosphate (S1P)/S1P receptor axis. Am J Pathol. 2012;181(1):85–97. doi: 10.1016/j.ajpath.2012.03.014. [DOI] [PubMed] [Google Scholar]
- 51.Wu XZ, Chen D. Origin of hepatocellular carcinoma: role of stem cells. J Gastroenterol Hepatol. 2006;21(7):1093–1098. doi: 10.1111/j.1440-1746.2006.04485.x. [DOI] [PubMed] [Google Scholar]
- 52.Mohamadnejad M, Alimoghaddam K, Mohyeddin-Bonab M, et al. Phase 1 trial of autologous bone marrow mesenchymal stem cell transplantation in patients with decompensated liver cirrhosis [published correction appears in Arch Iran Med. 2008;11(1):135] Arch Iran Med. 2007;10(4):459–466. [PubMed] [Google Scholar]
- 53.Kharaziha P, Hellström PM, Noorinayer B, et al. Improvement of liver function in liver cirrhosis patients after autologous mesenchymal stem cell injection: a phase I–II clinical trial. Eur J Gastroenterol Hepatol. 2009;21(10):1199–1205. doi: 10.1097/MEG.0b013e32832a1f6c. [DOI] [PubMed] [Google Scholar]
- 54.Amer ME, El-Sayed SZ, El-Kheir WA, et al. Clinical and laboratory evaluation of patients with end-stage liver cell failure injected with bone marrow-derived hepatocyte-like cells. Eur J Gastroenterol Hepatol. 2011;23(10):936–941. doi: 10.1097/MEG.0b013e3283488b00. [DOI] [PubMed] [Google Scholar]
- 55.Peng L, Xie DY, Lin BL, et al. Autologous bone marrow mesenchymal stem cell transplantation in liver failure patients caused by hepatitis B: short-term and long-term outcomes. Hepatology. 2011;54(3):820–828. doi: 10.1002/hep.24434. [DOI] [PubMed] [Google Scholar]
- 56.Zhang Z, Lin H, Shi M, et al. Human umbilical cord mesenchymal stem cells improve liver function and ascites in decompensated liver cirrhosis patients. J Gastroenterol Hepatol. 2012;27(suppl 2):112–120. doi: 10.1111/j.1440-1746.2011.07024.x. [DOI] [PubMed] [Google Scholar]
- 57.Mohamadnejad M, Alimoghaddam K, Bagheri M, et al. Randomized placebo-controlled trial of mesenchymal stem cell transplantation in decompensated cirrhosis. Liver Int. 2013;33(10):1490–1496. doi: 10.1111/liv.12228. [DOI] [PubMed] [Google Scholar]
- 58.Lyra AC, Soares MB, da Silva LF, et al. Feasibility and safety of autologous bone marrow mononuclear cell transplantation in patients with advanced chronic liver disease. World J Gastroenterol. 2007;13(7):1067–1073. doi: 10.3748/wjg.v13.i7.1067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Terai S, Ishikawa T, Omori K, et al. Improved liver function in patients with liver cirrhosis after autologous bone marrow cell infusion therapy. Stem Cells. 2006;24(10):2292–2298. doi: 10.1634/stemcells.2005-0542. [DOI] [PubMed] [Google Scholar]
- 60.Kim JK, Park YN, Kim JS, et al. Autologous bone marrow infusion activates the progenitor cell compartment in patients with advanced liver cirrhosis. Cell Transplant. 2010;19(10):1237–1246. doi: 10.3727/096368910X506863. [DOI] [PubMed] [Google Scholar]
- 61.Saito T, Okumoto K, Haga H, et al. Potential therapeutic application of intravenous autologous bone marrow infusion in patients with alcoholic liver cirrhosis. Stem Cells Dev. 2011;20(9):1503–1510. doi: 10.1089/scd.2011.0074. [DOI] [PubMed] [Google Scholar]
- 62.Lyra AC, Soares MB, da Silva LF, et al. Infusion of autologous bone marrow mononuclear cells through hepatic artery results in a short-term improvement of liver function in patients with chronic liver disease: a pilot randomized controlled study. Eur J Gastroenterol Hepatol. 2010;22(1):33–42. doi: 10.1097/MEG.0b013e32832eb69a. [DOI] [PubMed] [Google Scholar]
- 63.Spahr L, Chalandon Y, Terraz S, et al. Autologous bone marrow mononuclear cell transplantation in patients with decompensated alcoholic liver disease: a randomized controlled trial. PloS One. 2013;8(1):e53719. doi: 10.1371/journal.pone.0053719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Pai M, Zacharoulis D, Milicevic MN, et al. Autologous infusion of expanded mobilized adult bone marrow-derived CD34+ cells into patients with alcoholic liver cirrhosis. Am J Gastroenterol. 2008;103(8):1952–1958. doi: 10.1111/j.1572-0241.2008.01993.x. [DOI] [PubMed] [Google Scholar]
- 65.Gordon MY, Levicar N, Pai M, et al. Characterization and clinical application of human CD34+ stem/progenitor cell populations mobilized into the blood by granulocyte colony-stimulating factor. Stem Cells. 2006;24(7):1822–1830. doi: 10.1634/stemcells.2005-0629. [DOI] [PubMed] [Google Scholar]
- 66.Mohamadnejad M, Namiri M, Bagheri M, et al. Phase 1 human trial of autologous bone marrow-hematopoietic stem cell transplantation in patients with decompensated cirrhosis. World J Gastroenterol. 2007;13(24):3359–3363. doi: 10.3748/wjg.v13.i24.3359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Levicar N, Pai M, Habib NA, et al. Long-term clinical results of autologous infusion of mobilized adult bone marrow derived CD34+ cells in patients with chronic liver disease. Cell Prolif. 2008;41(suppl 1):115–125. doi: 10.1111/j.1365-2184.2008.00491.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Fürst G, Schulte am Esch J, Poll LW, et al. Portal vein embolization and autologous CD133+ bone marrow stem cells for liver regeneration: initial experience. Radiology. 2007;243(1):171–179. doi: 10.1148/radiol.2431060625. [DOI] [PubMed] [Google Scholar]
- 69.Yannaki E, Anagnostopoulos A, Kapetanos D, et al. Lasting amelioration in the clinical course of decompensated alcoholic cirrhosis with boost infusions of mobilized peripheral blood stem cells. Exp Hematol. 2006;34(11):1583–1587. doi: 10.1016/j.exphem.2006.06.012. [DOI] [PubMed] [Google Scholar]
- 70.Gaia S, Smedile A, Omedè P, et al. Feasibility and safety of G-CSF administration to induce bone marrow-derived cells mobilization in patients with end stage liver disease. J Hepatol. 2006;45(1):13–19. doi: 10.1016/j.jhep.2006.02.018. [DOI] [PubMed] [Google Scholar]
- 71.Yan L, Han Y, Wang J, Liu J, Hong L, Fan D. Peripheral blood monocytes from patients with HBV related decompensated liver cirrhosis can differentiate into functional hepatocytes. Am J Hematol. 2007;82(11):949–954. doi: 10.1002/ajh.21030. [DOI] [PubMed] [Google Scholar]
- 72.Gasbarrini A, Rapaccini GL, Rutella S, et al. Rescue therapy by portal infusion of autologous stem cells in a case of drug-induced hepatitis. Dig Liver Dis. 2007;39(9):878–882. doi: 10.1016/j.dld.2006.06.037. [DOI] [PubMed] [Google Scholar]
- 73.Khan AA, Parveen N, Mahaboob VS, et al. Safety and efficacy of autologous bone marrow stem cell transplantation through hepatic artery for the treatment of chronic liver failure: a preliminary study. Transplant Proc. 2008;40(4):1140–1144. doi: 10.1016/j.transproceed.2008.03.111. [DOI] [PubMed] [Google Scholar]
- 74.Han Y, Yan L, Han G, et al. Controlled trials in hepatitis B virus-related decompensate liver cirrhosis: peripheral blood monocyte transplant versus granulocyte-colony-stimulating factor mobilization therapy. Cytotherapy. 2008;10(4):390–396. doi: 10.1080/14653240802129901. [DOI] [PubMed] [Google Scholar]
- 75.Garg V, Garg H, Khan A, et al. Granulocyte colony-stimulating factor mobilizes CD34(+) cells and improves survival of patients with acute-on-chronic liver failure. Gastroenterology. 2012;142(3):505–512. e1. doi: 10.1053/j.gastro.2011.11.027. [DOI] [PubMed] [Google Scholar]
- 76.Shafritz DA, Oertel M. Model systems and experimental conditions that lead to effective repopulation of the liver by transplanted cells. Int J Biochem Cell Biol. 2011;43(2):198–213. doi: 10.1016/j.biocel.2010.01.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Woo DH, Kim SK, Lim HJ, et al. Direct and indirect contribution of human embryonic stem cell-derived hepatocyte-like cells to liver repair in mice. Gastroenterology. 2012;142(3):602–611. doi: 10.1053/j.gastro.2011.11.030. [DOI] [PubMed] [Google Scholar]
- 78.Takahashi K, Yamanaka S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell. 2006;126(4):663–676. doi: 10.1016/j.cell.2006.07.024. [DOI] [PubMed] [Google Scholar]
- 79.Gurdon JB, Elsdale TR, Fischberg M. Sexually mature individuals of Xenopus laevis from the transplantation of single somatic nuclei. Nature. 1958;182(4627):64–65. doi: 10.1038/182064a0. [DOI] [PubMed] [Google Scholar]
- 80.Rashid ST, Alexander GJ. Induced pluripotent stem cells: from Nobel Prizes to clinical applications. J Hepatol. 2013;58(3):625–629. doi: 10.1016/j.jhep.2012.10.026. [DOI] [PubMed] [Google Scholar]
- 81.Si-Tayeb K, Noto FK, Nagaoka M, et al. Highly efficient generation of human hepatocyte-like cells from induced pluripotent stem cells [published correction appears in Hepatology. 2010; 51(3):1094] Hepatology. 2010;51(1):297–305. doi: 10.1002/hep.23354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Sancho-Bru P, Roelandt P, Narain N, et al. Directed differentiation of murine-induced pluripotent stem cells to functional hepatocyte-like cells. J Hepatol. 2011;54(1):98–107. doi: 10.1016/j.jhep.2010.06.014. [DOI] [PubMed] [Google Scholar]
- 83.Yu Y, Liu H, Ikeda Y, et al. Hepatocyte-like cells differentiated from human induced pluripotent stem cells: relevance to cellular therapies. Stem Cell Res. 2012;9(3):196–207. doi: 10.1016/j.scr.2012.06.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Chen YF, Tseng CY, Wang HW, Kuo HC, Yang VW, Lee OK. Rapid generation of mature hepatocyte-like cells from human induced pluripotent stem cells by an efficient three-step protocol. Hepatology. 2012;55(4):1193–1203. doi: 10.1002/hep.24790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Aravalli RN, Cressman EN, Steer CJ. Hepatic differentiation of porcine induced pluripotent stem cells in vitro. Vet J. 2012;194(3):369–374. doi: 10.1016/j.tvjl.2012.05.013. [DOI] [PubMed] [Google Scholar]
- 86.Yu B, He ZY, You P, et al. Reprogramming fibroblasts into bipotential hepatic stem cells by defined factors. Cell Stem Cell. 2013;13(3):328–340. doi: 10.1016/j.stem.2013.06.017. [DOI] [PubMed] [Google Scholar]
- 87.Huang P, He Z, Ji S, et al. Induction of functional hepatocyte-like cells from mouse fibroblasts by defined factors. Nature. 2011;475(7356):386–389. doi: 10.1038/nature10116. [DOI] [PubMed] [Google Scholar]
- 88.Liu JZ, Hov JR, Folseraas T, et al. UK-PSCSC Consortium; International IBD Genetics Consortium; International PSC Study Group. Dense genotyping of immune-related disease regions identifies nine new risk loci for primary sclerosing cholangitis. Nat Genet. 2013;45(6):670–675. doi: 10.1038/ng.2616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Campbell JM, Hartjes KA, Nelson TJ, Xu X, Ekker SC. New and TALENted genome engineering toolbox. Circ Res. 2013;113(5):571–587. doi: 10.1161/CIRCRESAHA.113.301765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Choi SM, Kim Y, Liu H, Chaudhari P, Ye Z, Jang YY. Liver engraftment potential of hepatic cells derived from patient-specific induced pluripotent stem cells. Cell Cycle. 2011;10(15):2423–2427. doi: 10.4161/cc.10.15.16869. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Subba Rao M, Sasikala M, Nageshwar Reddy D. Thinking outside the liver: induced pluripotent stem cells for hepatic applications. World J Gastroenterol. 2013;19(22):3385–3396. doi: 10.3748/wjg.v19.i22.3385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Rashid ST, Corbineau S, Hannan N, et al. Modeling inherited metabolic disorders of the liver using human induced pluripotent stem cells. J Clin Invest. 2010;120(9):3127–3136. doi: 10.1172/JCI43122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Espejel S, Roll GR, McLaughlin KJ, et al. Induced pluripotent stem cell-derived hepatocytes have the functional and proliferative capabilities needed for liver regeneration in mice. J Clin Invest. 2010;120(9):3120–3126. doi: 10.1172/JCI43267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Takebe T, Sekine K, Enomura M, et al. Vascularized and functional human liver from an iPSC-derived organ bud transplant. Nature. 2013;499(7459):481–484. doi: 10.1038/nature12271. [DOI] [PubMed] [Google Scholar]
- 95.Jin LF, Ji SH, Yang JF, Ji WZ. Notch signalling dependent differentiation of cholangiocyte-like cells from rhesus monkey embryonic stem cells. Dongwuxue Yanjiu. 2011;32(4):391–395. doi: 10.3724/SP.J.1141.2011.04391. [DOI] [PubMed] [Google Scholar]
- 96.Nakamura N, Saeki K, Mitsumoto M, et al. Feeder-free and serum-free production of hepatocytes, cholangiocytes, and their proliferating progenitors from human pluripotent stem cells: application to liver-specific functional and cytotoxic assays. Cell Reprogram. 2012;14(2):171–185. doi: 10.1089/cell.2011.0064. [DOI] [PubMed] [Google Scholar]
- 97.Yanagida A, Ito K, Chikada H, Nakauchi H, Kamiya A. An in vitro expansion system for generation of human iPS cell-derived hepatic progenitor-like cells exhibiting a bipotent differentiation potential. PloS One. 2013;8(7):e67541. doi: 10.1371/journal.pone.0067541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Sanal MG. Future of liver transplantation: non-human primates for patient-specific organs from induced pluripotent stem cells. World J Gastroenterol. 2011;17(32):3684–3690. doi: 10.3748/wjg.v17.i32.3684. [DOI] [PMC free article] [PubMed] [Google Scholar]