Abstract
Although vaccination campaigns have significantly reduced the global burden of rubella disease, there are still regional outbreaks and cases of congenital rubella syndrome (CRS). Rubella vaccination elicits a strong humoral, as well as cellular, response. The relationship between these two measures in response to rubella vaccine is poorly understood. We have previously reported no correlation between rubella virus-specific cytokine secretion and IgG antibody levels after rubella vaccination. In the current study, we extend our previous work to report correlations between secreted cytokines and functional neutralizing antibodies after rubella vaccination in four distinct cohorts. There was evidence of significant differences (p <0.05) in rubella virus-specific humoral and cellular responses between cohorts. When investigating relationships between rubella vaccine-specific humoral and cellular immunity, we observed a significant correlation between neutralizing antibodies and IFN-γ (rs = 0.21, p = 0.0004). We also observed correlations in subjects with extreme humoral immune phenotypes and IFN-γ levels in two of the four cohorts (rs = 0.32, p = 0.01; rs = 0.36, p = 0.01, respectively). These findings indicate that there is a high level of heterogeneity in rubella-specific immune responses between study populations. We believe that the novel correlation discovered between IFN-γ and neutralizing antibody titers will give future insight into the functional mechanisms of immunity induced by rubella virus and other live viral vaccines.
Keywords: Cytokines, Antibodies, MMR, Immunity, Humoral, Cellular
MeSH Keywords: Polymorphism, Single Nucleotide; Cytokines; Receptors, Cytokine; Antibodies; Measles-Mumps-Rubella Vaccine; Immunity; Immunity, Humoral; Immunity, Cellular
Introduction
The year 2012 witnessed the highest number of rubella cases in England and Wales since 1999 [1]. In the first half of 2013, Poland reported 21,283 rubella cases and Japan recorded 5,442 cases [2, 3]. With these few examples of the resurgence of rubella cases comes the threat of larger outbreaks and exposure to the most at-risk population—unvaccinated pregnant women. The World Health Organization has targeted rubella for elimination in Europe, the Americas, and the Western Pacific Regions [4]. Vaccine campaigns have eliminated some areas of endemic rubella. However, recent increases in sporadic nationwide epidemics and a concomitant increase in the cases of congenital rubella syndrome (CRS) emphasize the importance of these campaigns.
Neutralizing antibodies are considered the best correlate of protection against rubella [5]. However, there is evidence of false-positive results using antibody-based assays due to the presence of infectious mononucleosis, Rh factor, and parvovirus infection [6–8]. Also, individuals with sub-protective titers (≤ 15 IU) of serum IgG antibodies may still be protected against rubella, which is likely due to cellular immunity [9]. Thus, the measurement of rubella-specific humoral immunity as a correlate may not be ideal under certain conditions. Cellular immunity measures to rubella virus vaccine include a lymphoproliferative response and the production of inflammatory cytokines, such as IFN-γ, IL-6, and TNF-α [10–13]. It is not well understood what the relationship is between cellular and humoral immune responses and how they correlate with protection.
In this study, we characterized rubella virus-specific humoral and cellular immune responses in four separate cohorts that received the measles-mumps-rubella-II (MMR-II) vaccine. We also examined correlations between neutralizing antibodies and inflammatory cytokines (IL-6 and IFN-γ). The objective of this study was to identify differences and commonalities in immune responses between cohorts and to elucidate potential correlates of rubella vaccine-specific immunity.
Methods
Study participants
The following methods are similar or identical to our previous published studies [13–20] The study cohorts were comprised of healthy children, older adolescents, and adults (age at enrollment of 11 to 40 years) who resided in Rochester, MN, and San Diego, CA, with clinical and demographic characteristics previously reported [16, 21, 22] (See Table 1 for demographics).
Table 1.
Study Cohorts Demographics
| Study Demographics
| |||||
|---|---|---|---|---|---|
| Rochester 1 | Rochester 2 | Rochester 3 | San Diego | Total | |
|
| |||||
| Age at enrollment | |||||
| N* | 318 | 380 | 341 | 986 | 2025 |
| Mean (SD) | 15.6 (2.0) | 14.5 (2.3) | 15.2 (2.3) | 25.1 (4.5) | 20.0 (6.2) |
| Median | 16 | 15 | 15 | 24 | 19 |
| Q1, Q3 | 14.0, 17.0 | 13.0, 17.0 | 13.0, 17.0 | 22.0, 27.0 | 15.0, 24.0 |
| Range | (12.0–19.0) | (11.0–18.0) | (11.0–22.0) | (19.0–40.0) | (11.0–40.0) |
|
| |||||
| Age at most recent vaccination | |||||
| N* | 318 | 380 | 341 | 698 | 1737 |
| Mean (SD) | 10.9 (2.3) | 7.8 (3.4) | 6.8 (3.2) | 20.6 (3.4) | 13.3 (6.9) |
| Median | 12 | 8 | 5 | 19 | 12 |
| Q1, Q3 | 11.0, 12.0 | 5.0, 11.0 | 4.0, 10.0 | 18.0, 22.0 | 7.0, 19.0 |
| Range | (2.0–17.0) | (1.0–14.0) | (1.0–15.0) | (17.0–39.0) | (1.0–39.0) |
|
| |||||
| Time since most recent vaccination to enrollment | |||||
| N* | 318 | 380 | 341 | 698 | 1737 |
| Mean (SD) | 4.7 (2.4) | 6.7 (2.4) | 8.4 (2.7) | 3.4 (1.7) | 5.3 (3.0) |
| Median | 4.8 | 6.7 | 8.5 | 3 | 4.9 |
| Q1, Q3 | 2.8, 6.1 | 4.9, 8.2 | 6.4, 10.3 | 2.2, 4.0 | 2.9, 7.4 |
| Range | (0.6–12.3) | (0.4–15.4) | (0.6–16.8) | (0.1–8.6) | (0.1–16.8) |
|
| |||||
| Gender | |||||
| Male | 170 (53.5%) | 209 (55.0%) | 195 (57.2%) | 723 (73.3%) | 1297 (64.0%) |
| Female | 148 (46.5%) | 171 (45.0%) | 146 (42.8%) | 263 (26.7%) | 728 (36.0%) |
|
| |||||
| Self-declared Race | |||||
| American Indian or Alaska Native | 2 (0.6%) | 2 (0.5%) | 0 (0.0%) | 19 (1.9%) | 23 (1.1%) |
| Asian, Hawaiian or Pacific Islander | 7 (2.2%) | 15 (3.9%) | 5 (1.5%) | 48 (4.9%) | 75 (3.7%) |
| Black or African American | 4 (1.3%) | 4 (1.1%) | 76 (22.3%) | 162 (16.4%) | 246 (12.1%) |
| White | 299 (94.0%) | 337 (88.7%) | 247 (72.4%) | 530 (53.8%) | 1413 (69.8%) |
| Multiple | 6 (1.9%) | 14 (3.7%) | 8 (2.3%) | 82 (8.3%) | 110 (5.4%) |
| Other | 0 (0.0%) | 2 (0.5%) | 5 (1.5%) | 126 (12.8%) | 133 (6.6%) |
| Unknown | 0 (0.0%) | 6 (1.6%) | 0 (0.0%) | 19 (1.9%) | 25 (1.2%) |
The total number of subjects included in this study is determined by the availability of demographic data and immune measures
The Rochester cohorts (Rochester 1, 2, and 3) consisted of individuals from three independent cohorts of healthy schoolchildren and young adults from all socioeconomic strata in Rochester, MN. Specifically, between December 2001 and August 2002, we enrolled 346 healthy children (age 12 to 19 years, Rochester 1). A detailed description of Rochester 1 has been published elsewhere [23, 24]. Between December 2006–August 2007, we enrolled 440 healthy children (age 11 to 18 years, Rochester 2), as previously published [14, 16]. In November 2008–September 2009, we enrolled 388 healthy children, enriched with African-American youth (age 11 to 22 years, Rochester 3) [25, 26]. All participants had written records of receiving two doses of age-appropriate measles-mumps-rubella (MMR II, Merck) vaccine.
In July 2005–September 2006, we enrolled an additional 1,076 healthy older adolescents and healthy young adults (age 18 to 40 years, San Diego cohort) from armed forces personnel in San Diego, CA. Subject enrollment for this study has been previously described in detail [21, 22]. We recruited these individuals because they were active military personnel who were recently vaccinated with smallpox vaccine. As members of the U.S. military, they represent a cross section of the U.S. population. The Institutional Review Boards of the Mayo Clinic and Naval Health Research Center approved this study, and written informed consent was obtained from each subject, or from the parents of all children who participated in the study; written assent was obtained from age-appropriate participants.
Rubella virus-specific cytokine secretion
The levels of secreted cytokines following stimulation of isolated PBMCs with live rubella virus were measured, as previously described by our group [13, 27]. Briefly, 2 ×105/ml PBMCs were stimulated with the W-Therien strain of rubella virus (a gift from Dr. Teryl Frey, Georgia State University, Atlanta, GA) with optimized multiplicity of infection (MOI) and incubation times depending on the specific cytokine measured. For the measurement of IL-6 and IFN-γ, PBMCs were stimulated with an MOI of 5. The supernatants were removed post stimulation at 24 hrs for IL-6, and 48 hrs for IFN-γ. All samples were stored at −80 °C until assayed. Cytokine levels were quantified using BD OptEIA™ Human ELISA kits, and absorbance levels were measured using a Molecular Devices SpectraMax 340PC384.
Rubella-specific Neutralizing Antibodies
The levels of neutralizing antibodies against live rubella virus were measured using a soluble immunocolorimetric assay (sICA) that was adapted for high-throughput [28]. Briefly, serial dilutions of subject sera were incubated with the rubella vaccine virus strain HPV77. After incubation, the virus/sera mixture was added to Vero cells cultured in a flat bottom 96-well plate and incubated for 72 hours at 37°C, 5% CO2. Then, cells were fixed in cold methanol for 10 minutes and blocked with PBS supplemented with 5% skim milk (Difco; BD, NJ, USA) and 0.1% Tween 20 for 30 minutes, and then washed three times with PBS supplemented with 0.05% Tween 20 (PBS-T). Fixed cells were incubated with anti-E1 glycoprotein (CDC, GA, USA) for 30 minutes, and washed three times with PBS-T. The secondary goat anti-mouse HRP-conjugated antibody (Invitrogen, CA, USA) was added for 30 minutes. Plates were washed again and antibody conjugate was visualized by adding NeA-Blue TMB substrate (Clinical Science Products, MA, USA) for 10 minutes. The optical density (OD) values were read at 450 nm/630 nm on an Eon microplate spectrophotometer (BioTek, VT, USA). The Loess method of statistical interpolation was used to estimate neutralization titers (NT50) from observed values [29].
Statistical Analysis
The differences between the median values of the stimulated observations and the median values of the unstimulated observations were determined for IL-6 and IFN-γ and these measures were used in analysis. Distributions of outcomes and demographics were described with counts and percentages for categorical variables, with measures of central tendency (means and medians), and with variability (range, interquartile ranges, and standard deviation) for continuous variables. Wilcoxon rank sum tests were utilized to ascertain if differences in cytokine and neutralizing antibody levels existed between cohorts. Spearman correlations were employed to investigate associations between neutralizing antibody titers and levels of both IL-6 and IFN-γ, with and without controlling for: age; age at most recent recorded vaccination; years from most recent recorded vaccination to enrollment age; gender; self-declared race and ethnicity. Correlations were then repeated within a subset of 224 “extreme responders,” defined as individuals who exhibited no meaningful neutralizing antibody response (NT50 estimated as < 25; 18 subjects from Rochester 1, 34 from Rochester 2, and 30 each from Rochester 3 and San Diego), and an equal number of individuals from within each cohort with the highest NT50 values.
Results
Humoral and Cellular Responses to Rubella Vaccine across Cohorts
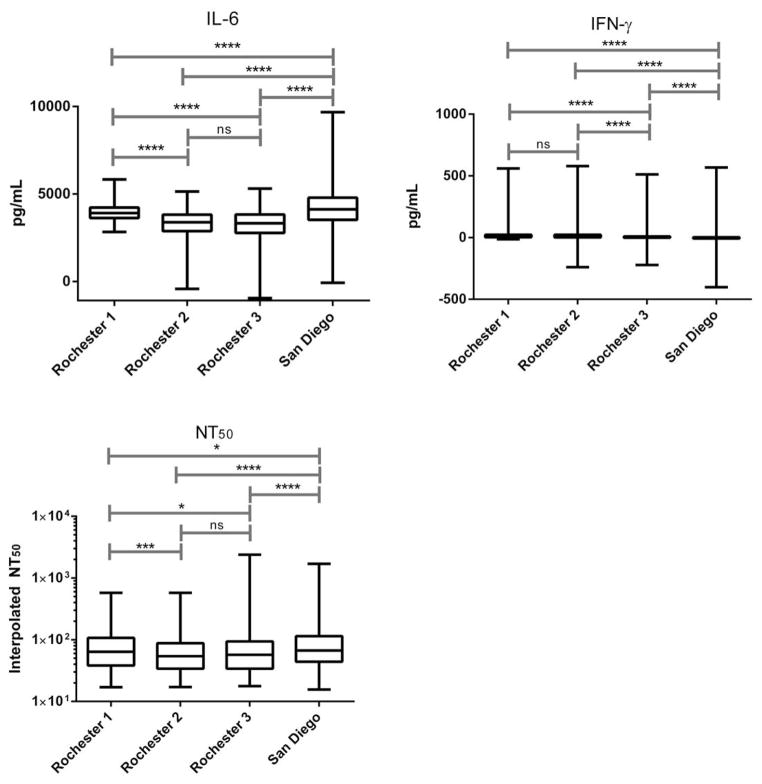
The median neutralizing antibody titer (NT50) was: 64.1 (IQR; 38.4, 107.2) for Rochester 1; 54.2 (IQR; 33.9, 88.0) for Rochester 2; 57.2 (IQR; 34.0, 93.9) for Rochester 3; and 67.0 (IQR; 44.0, 114.5) for the San Diego cohort (Figure 1). There were statistically significant (p ≤ 0.05) differences in median NT50 for all comparisons except Rochester 2 and Rochester 3 (p = 0.3). Secreted cytokine levels were calculated as the difference between the median of rubella-virus stimulated replicates minus the median of the unstimulated replicate values. The median of the differences for IL-6 production across study cohorts were: 3910.3 pg/mL (IQR; 3622.3, 4220.1) for Rochester 1; 3378.9 pg/mL (IQR; 2874.3, 3817.7) for Rochester 2; 3328.0 pg/mL (IQR; 2771.4, 3829.8) for Rochester 3; and 4117.4 pg/mL (IQR; 3521.8, 4787.9) for the San Diego cohort. The median of the differences for IFN-γ production for Rochester 1 was 7.9 pg/mL (IQR; 3.2, 23.4), Rochester 2 was 9.3 pg/mL (IQR; 2.3, 23.5), Rochester 3 was 1.9 pg/mL (IQR; −0.9, 9.3), and the San Diego cohort was −1.4 pg/mL (IQR; −6.4, 3.1). There were also statistically significant differences in rubella virus-induced secreted cytokine levels (IL-6 & IFN-γ) between study cohorts. IL-6 levels were significantly different (p = <0.0001) between all cohorts, except in the Rochester 2 and Rochester 3 cohorts (p = 0.7). IFN-γ levels were also remarkably different between all cohorts (p = < 0.0001), except for Rochester 1 and Rochester 2 (p = 0.4).
Figure 1. Distribution of Humoral and Cellular Immune Responses across Cohorts.
Box plots comparing secreted cytokine levels and NT50 across cohorts. Rubella-specific IL-6 (Rochester 1 n = 297, Rochester 2 n = 380, Rochester 3 n = 311, San Diego n = 946) and IFN-γ (Rochester 1, n = 297, Rochester 2, n = 380, Rochester 3 n = 292, San Diego n = 925) levels were measured by ELISA and reported as the difference between rubella virus-stimulated and unstimulated values. NT50 levels (Rochester 1, n = 318; Rochester 2, n = 373; Rochester 3, n = 338; San Diego, n = 985) were calculated using interpolated data from a high-throughput soluble immunochemical assay. There were significant differences in immune measures across cohorts. However, Rochester 2 and Rochester 3 displayed strikingly similar median values of NT50 and IL-6 (ns = not significant; * = p ≤ 0.05; *** = p ≤ 0.001; **** = p ≤ 0.0001).
Correlation between Neutralizing Antibodies and Secreted Cytokines
Correlation Analyses revealed a correlation between serum neutralizing antibodies and IFN-γ secretion after rubella vaccination in Rochester 3 (rs=0.2084, p = 0.0004), and a trend toward significance after adjustment in Rochester 2 (rs=0.0979, p = 0.06). There was also a correlation between IL-6 and NT50 in Rochester 1 (rs=0.1045, p = 0.08) that approached significance (Table 2). The observed correlation between IFN-γ and NT50 in two cohorts suggests that this cellular immune marker may be further investigated as a co-correlate of protection. The low-level correlation between IL-6 and NT50 was only observed in one cohort, making it difficult to speculate what the biological significance is for the correlations between this inflammatory cytokine and NT50.
Table 2. Summary of Correlations between Rubella-Specific Cellular Immune Measures and Neutralizing Antibodies.
Correlations between in vitro secreted cytokine levels and serum NT50 using Spearman correlation of coefficients. Simple correlations are unadjusted for confounding factors. The Adjusted correlations account for age, age at most recent recorded vaccination, years from most recent recorded vaccination to enrollment, gender, self-declared race and ethnicity. Bolded text represents a significant correlation between IFN-γ and NT50 (p-value = 0.0004).
| Cohort | Cytokine (pg/mL) | Simple correlation (95% CI) | Adjusted correlation (95% CI) |
|---|---|---|---|
|
| |||
| Rochester 1 | |||
| IFN-γ | 0.0809 (−0.0334, 0.1928) | 0.07 (−0.0454, 0.1834) | |
| IL-6 | 0.1162 (0.0023, 0.2269) | 0.1045 (−0.0108, 0.2167) | |
| Rochester 2 | |||
| IFN-γ | 0.1148 (0.0132, 0.2137) | 0.0979 (−0.0046, 0.1982) | |
| IL-6 | 0.0181 (−0.0836, 0.1194) | 0.0187 (−0.0839, 0.1208) | |
| Rochester 3 | |||
| IFN-γ | 0.2637 (0.1525, 0.3675) | 0.2084 (0.0937, 0.317) | |
| IL-6 | −0.0918 (−0.2013, 0.0203) | −0.0629 (−0.1781, 0.0542) | |
| San Diego | |||
| IFN-γ | 0.0259 (−0.0386, 0.0903) | 0.0218 (−0.0554, 0.0988) | |
| IL-6 | −0.0516 (−0.115, 0.0122) | −0.0538 (−0.1304, 0.0234) | |
Next, we investigated correlations between the extremes of NT50 with rubella virus-specific secreted cytokine levels (Table 3). The extremes were defined as the observations for those people who had no meaningful neutralizing antibody response (NT50 < 25) and an equal number of people with the highest neutralizing antibody response in each cohort (N = 224). The median NT50 in the highest response group was 272.0, compared to 22.5 in the lowest responders (p = <0.0001). There were also significant differences in IFN-γ levels, with a median of 9.8 pg/mL in the high group and 2.3 pg/mL in the low group (p = <0.0001). When investigating correlations between IFN-γ levels and NT50 in the separate cohorts, we discovered a moderate positive association in Rochester 2 (rs=0.3197, p = 0.01), and Rochester 3 (rs=0.3606, p = 0.01).
Table 3. Correlation between IFN-γ levels and NT50 Extremes.
Correlations between in vitro rubella-specific secreted cytokine levels and serum NT50 extremes using Spearman correlation of coefficients. Simple correlations are unadjusted for confounding factors. The Adjusted correlations account for age, age at most recent recorded vaccination, years from most recent recorded vaccination to enrollment, gender, self-declared race and ethnicity. Bolded text highlights a moderate, positive relationship between NT50 extremes and IFN-γ levels (p-value < 0.0001 for Rochester 2 and Rochester 3).
| Cohort | N | Cytokine (pg/mL) | Simple correlation (95% CI) | Adjusted correlation (95% CI) | |
|---|---|---|---|---|---|
|
| |||||
| Rochester 1 | |||||
|
| |||||
| 36 | IFN-γ | 0.1666 (−0.1959, 0.485) | 0.07 (−0.2172, 0.5346) | ||
| IL-6 | 0.1954 (−0.1676, 0.5071) | 0.1136 (−0.2884, 0.4781) | |||
|
| |||||
| Rochester 2 | |||||
| 68 | IFN-γ | 0.3392 (0.1072, 0.5326) | 0.3197 (0.0733, 0.5254) | ||
| IL-6 | 0.0072 (−0.2317, 0.2452) | −0.015 (−0.2636, 0.2358) | |||
|
| |||||
| Rochester 3 | |||||
| 60 | IFN-γ | 0.4999 (0.2661, 0.673) | 0.3606 (0.0877, 0.5782) | ||
| IL-6 | −0.2452 (−0.4756, 0.0211) | −0.1512 (−0.4109, 0.1343) | |||
|
| |||||
| San Diego | |||||
| 60 | IFN-γ | 0.2192 (−0.0484, 0.4542) | 0.2207 (−0.1865, 0.5572) | ||
| IL-6 | −0.1462 (−0.3929, 0.1226) | 0.0572 (−0.3386, 0.434) | |||
Discussion
When measuring humoral and cellular responses to rubella vaccine, we observed statistically significant differences in NT50, IL-6, and IFN-γ between cohorts. However, this was not true for all comparisons. Specifically, the Rochester 2 and Rochester 3 cohorts exhibited very comparable rubella-specific NT50 and IL-6 levels; Rochester 1 and Rochester 2 cohorts displayed similar levels of IFN-γ. A significant correlation between NT50 and IFN-γ levels was observed for Rochester 3 with a trend in the same direction for Rochester 2. This association was strengthened when our focus shifted to the extreme responders in these cohorts, indicating a relationship between a strong IFN-γ response and a strong humoral response to rubella virus. The immune response to vaccination is influenced by numerous environmental and host factors [30–33]. Therefore, it is not surprising that we observed differences in immune responses across cohorts. Although all individuals received the same rubella-containing MMR-II vaccine, there were other confounding attributes that could potentially influence immunity. Three of the Rochester cohorts were recruited as residents of Olmsted County, MN, while the fourth (San Diego) represents military personnel from all over the United States (see Study Participants). There were differences in age at vaccine, age at enrollment, and time since last recorded vaccine event. We have previously reported a high level of heritability (46%) in rubella-specific antibody titers [32] and discovered numerous host genetic factors associated with differences in immunity to rubella vaccination [14–17, 34–36]. The differences and similarities observed between these cohorts highlight that other factors not associated with host genetic composition may influence immunity after rubella vaccination. In fact, a model aimed at defining all contributing factors to inter-individual differences in rubella-specific immunity will incorporate host, viral, and environmental factors. The ideal population-based study will target recruitment efforts to avoid disparities in these variables or use statistical methodology to adjust, such as those used in this study.
Measuring humoral immunity is considered the gold standard as a correlate of protection against rubella virus, and protective humoral immunity is defined as any measurable antibody titer ≥ 10 IU/ml [37]. Vaccination with two doses of the RA27/3 strain of rubella virus induces a protective level of immunity that lasts >20 years [38] [39]. The estimated half-life for rubella vaccine-specific antibodies is ~114 years [40]. However, there is evidence of waning immunity over time. Rubella-specific neutralizing antibody titers 12-years post-vaccination can be one-half the original values [41]. Our current study revealed lower median values for NT50 in Rochester 2 (NT50 54.2) and Rochester 3 (NT50 57.2). The demographic data is strikingly similar between these two cohorts (Table 1). The median age at enrollment was identical between the two cohorts (15.0 years), with similar median age ranges at most recent vaccination event, and time elapsed since last recorded vaccination event. Although waning immunity may contribute to the lower NT50 in these cohorts, it is difficult to assess the exact influence in this study because: 1) there are no complete data available representing antibody titers shortly after vaccination in our cohorts; 2) unlike other rubella-specific antibody assays, our interpolated NT50 values were not calibrated against a standardized sample, such as the WHO 2nd International Standard Preparation for Anti-Rubella Serum. To address the second concern, we are currently testing for correlations between NT50 and those values obtained with Beckman Coulter’s Access® Rubella IgG Assay [42]. This commercial assay is calibrated against a standardized serum sample. Initial analyses reveal a strong correlation between total IgG and NT50 titers (unpublished data). It is uncertain what the mechanism is behind the lower levels of secreted IL-6 in the Rochester 2 and 3 cohorts. This may, in part, be due to a combination of the multiple similarities noted between these two cohorts and not necessarily attributable to waning cellular immunity.
To our knowledge, this is the first study to investigate correlations between functional neutralizing antibodies against rubella virus and in vitro secreted cytokine levels. We previously observed no correlation between rubella-specific IgG EIA and secreted cytokine levels in vaccinated subjects [43]. The difference between this study and previous findings is we found positive correlations between rubella-specific IFN-γ and NT50 in one cohort, which was then amplified when analyzing antibody response extremes. We conclude that this correlation may be lost when investigating across a large cohort, but the relationship is revealed when the study focus is directed toward those subjects that were characterized as having an extreme humoral response to rubella vaccination.
It is difficult to interpret the statistically significant findings into a more biologically relevant context. We have previously characterized the cellular immune response to rubella as “skewed” toward a proinflammatory response with high levels of IL-6, GM-CSF, and TNF-α [13] and moderate levels of IL-2 and IFN-γ. Intuitively, a robust Th1/proinflammatory response may lead to attenuation of the humoral response through such mechanisms as suppression of IL-4 production [44]. However, studies have revealed a synergistic relationship between IFN-γ and humoral immunity. In mice, IFN-γ enhances the secretion of IgG2A in vitro and in vivo [45, 46]. There is also evidence that IFN-γ can induce a viral-specific memory T cell to adopt a Th1/Th2 combined phenotype that could potentially contribute to a robust humoral and cellular response [47]. The contributing role of IFN-γ in humans on antibody production remains unclear. The importance of IFN-γ production in response to live viral vaccination is highlighted by the discovery that measles infection in infants with a lack of humoral response after vaccination is attenuated, and this is due to IFN-γ production by CD4+ T cells. The measurement of IFN-γ is considered a correlate of protection in tuberculosis and malaria vaccine models [48–50] [51]. We propose that IFN-γ may be used, alongside humoral measures, as a co-correlate of protection in rubella vaccine studies. Moreover, the amplification of the correlation between IFN-γ and NT50 when analyzing extreme immune outcomes suggests that vaccination in high responders induces a robust humoral and cellular response.
In summary, we detected significant differences in humoral and cellular immune response between cohorts, but a correlation between IFN-γ and neutralizing antibodies. This is the first step toward validating that IFN-γ may be used as a co-correlate of protection against rubella virus. These data may give insight into future mechanisms behind inter-individual differences in response to vaccination against rubella and other live viral vaccines. The next generation of rubella vaccine candidates may be constructed to elicit a strong IFN-γ and antibody response regardless of genetic and other environmental influences.
Acknowledgments
The authors would like to thank Caroline Vitse for assisting in the preparation and submission of the article. Research reported in this publication was supported by the National Institute of Allergy And Infectious Diseases of the National Institutes of Health under Award Number R37 AI048793-11 (which recently received a MERIT award).
Footnotes
The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Competing Interests
Dr. Poland is the chair of a Safety Evaluation Committee for novel investigational vaccine trials being conducted by Merck Research Laboratories. Dr. Poland offers consultative advice on vaccine development to Merck & Co. Inc., CSL Biotherapies, Avianax, Sanofi Pasteur, Dynavax, Novartis Vaccines and Therapeutics, PAXVAX Inc, and Emergent Biosolutions. Drs. Poland and Ovsyannikova hold two patents related to vaccinia peptide research. These activities have been reviewed by the Mayo Clinic Conflict of Interest Review Board and are conducted in compliance with Mayo Clinic Conflict of Interest policies. This research has been reviewed by the Mayo Clinic Conflict of Interest Review Board and was conducted in compliance with Mayo Clinic Conflict of Interest policies.
References
- 1.Wise J. Small rise in rubella cases triggers warning. Bmj. 2013;346:f2935. doi: 10.1136/bmj.f2935. [DOI] [PubMed] [Google Scholar]
- 2.Paradowska-Stankiewicz I, Czarkowski MP, Derrough T, Stefanoff P. Ongoing outbreak of rubella among young male adults in Poland: increased risk of congenital rubella infections. Euro Surveill. 2013;18(21) [PubMed] [Google Scholar]
- 3.Nationwide rubella epidemic in Japan. Releve epidemiologique hebdomadaire/Section d’hygiene du Secretariat de la Societe des Nations = Weekly epidemiological record/Health Section of the Secretariat of the League of Nations. 2013 Jun 14;88(24):251–6. [PubMed] [Google Scholar]
- 4.Organization WH. Global Measles & Rubella Strategic Plan 2012–2020. Geneva: 2012. [Google Scholar]
- 5.Plotkin SA. Immunologic correlates of protection induced by vaccination. PediatrInfect Dis J. 2001;20(1):63–75. doi: 10.1097/00006454-200101000-00013. [DOI] [PubMed] [Google Scholar]
- 6.Watson JC, Hadler SC, Dykewicz CA, Reef S, Phillips L. Measles, mumps, and rubella--vaccine use and strategies for elimination of measles, rubella, and congenital rubella syndrome and control of mumps: recommendations of the Advisory Committee on Immunization Practices (ACIP) MMWR Recomm Rep. 1998;47(RR-8):1–57. [PubMed] [Google Scholar]
- 7.Prevention CfDCa. Epidemiology and Prevention of Vaccine-Preventable Diseases. 12. Washington, D.C: Public Health Foundation; 2012. second printing ed. [Google Scholar]
- 8.Remington JS, Klein JO, Wilson CB, Nizet V, Maldonado Y. Infectious Diseases of the Fetus and Newborn. 7. Philadelphia: Elsevier Saunders; 2010. [Google Scholar]
- 9.O’Shea S, Best JM, Banatvala JE. Viremia, virus excretion, and antibody responses after challenge in volunteers with low levels of antibody to rubella virus. The Journal of Infectious Diseases. 1983 Oct;148(4):639–47. doi: 10.1093/infdis/148.4.639. [DOI] [PubMed] [Google Scholar]
- 10.Honeyman MC, Forrest JM, Dorman DC. Cell-mediated immune response following natural rubella and rubella vaccination. Clinical and Experimental Immunology. 1974;17(4):665–71. [PMC free article] [PubMed] [Google Scholar]
- 11.Allmendinger J, Paradies F, Kamprad M, Richter T, Pustowoit B, Liebert UG. Determination of rubella virus-specific cell-mediated immunity using IFN gamma-ELISpot. J Med Virol. 2010;82(2):335–40. doi: 10.1002/jmv.21621. [DOI] [PubMed] [Google Scholar]
- 12.Pukhalsky AL, Shmarina GV, Bliacher MS, Fedorova IM, Toptygina AP, Fisenko JJ, et al. Cytokine profile after rubella vaccine inoculation: evidence of the immunosuppressive effect of vaccination. Mediators Inflamm. 2003;12(4):203–7. doi: 10.1080/09629350310001599639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Dhiman N, Haralambieva IH, Vierkant RA, Pankratz VS, Ryan E, Jacobson RM, et al. Predominant inflammatory cytokine secretion pattern in response to two doses of live rubella vaccine in health vaccinees. Cytokine. 2010;50:24–9. doi: 10.1016/j.cyto.2009.12.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Haralambieva IH, Dhiman N, Ovsyannikova IG, Vierkant RA, Pankratz VS, Jacobson RM, et al. 2′-5′-Oligoadenylate synthetase single-nucleotide polymorphisms and haplotypes are associated with variations in immune responses to rubella vaccine. Hum Immunol. 2010;71(4):383–91. doi: 10.1016/j.humimm.2010.01.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ovsyannikova IG, Haralambieva IH, Dhiman N, O’Byrne MM, Pankratz VS, Jacobson RM, et al. Polymorphisms in the vitamin A receptor and innate immunity genes influence the antibody response to rubella vaccination. J Infect Dis. 2010;201(2):207–13. doi: 10.1086/649588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ovsyannikova IG, Dhiman N, Haralambieva IH, Vierkant RA, O’Byrne MM, Jacobson RM, et al. Rubella vaccine-induced cellular immunity: evidence of associations with polymorphisms in the Toll-like, vitamin A and D receptors, and innate immune response genes. Human Genetics. 2010;127:207–21. doi: 10.1007/s00439-009-0763-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Dhiman N, Haralambieva IH, Kennedy RB, Vierkant RA, O’Byrne MM, Ovsyannikova IG, et al. SNP/haplotype associations in cytokine and cytokine receptor genes and immunity to rubella vaccine. Immunogenetics. 2010;62(4):197–210. doi: 10.1007/s00251-010-0423-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kennedy RB, Ovsyannikova IG, Pankratz VS, Haralambieva IH, Vierkant RA, Jacobson RM, et al. Genome-wide genetic associations with IFNgamma response to smallpox vaccine. Human Genetics. 2012;131(9):1433–51. doi: 10.1007/s00439-012-1179-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Haralambieva IH, Ovsyannikova IG, Dhiman N, Kennedy RB, O’Byrne M, Pankrantz VS, et al. Common SNPs/Haplotypes in IL18R1 and IL18 Genes are Associated with Variations in Humoral Immunity to Smallpox Vaccination in Caucasians and African-Americans. Journal of Infectious Diseases. 2011;204(3):433–41. doi: 10.1093/infdis/jir268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ovsyannikova IG, Haralambieva IH, Kennedy RB, Pankratz VS, Vierkant RA, Jacobson RM, et al. Impact of cytokine and cytokine receptor gene polymorphisms on cellular immunity after smallpox vaccination. Gene. 2012;510:59–65. doi: 10.1016/j.gene.2012.08.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Kennedy RB, Ovsyannikova IG, Pankratz VS, Vierkant RA, Jacobson RM, Ryan MA, et al. Gender effects on humoral immune responses to smallpox vaccine. Vaccine. 2009;27(25–26):3319–23. doi: 10.1016/j.vaccine.2009.01.086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ovsyannikova IG, Vierkant RA, Pankratz VS, Jacobson RM, Poland GA. Human leukocyte antigen genotypes in the genetic control of adaptive immune responses to smallpox vaccine. J Infect Dis. 2011;203(11):1546–55. doi: 10.1093/infdis/jir167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ovsyannikova IG, Jacobson RM, Vierkant RA, Jacobsen SJ, Pankratz VS, Poland GA. The contribution of HLA class I antigens in immune status following two doses of rubella vaccination. Human Immunology. 2004;65:1506–15. doi: 10.1016/j.humimm.2004.07.001. [DOI] [PubMed] [Google Scholar]
- 24.Ovsyannikova IG, Jacobson RM, Vierkant RA, Jacobsen SJ, Pankratz VS, Poland GA. Human leukocyte antigen class II alleles and rubella-specific humoral and cell-mediated immunity following measles-mumps-rubella-II vaccination. Journal of Infectious Diseases. 2005;191(4):515–9. doi: 10.1086/427558. [DOI] [PubMed] [Google Scholar]
- 25.Ovsyannikova IG, Haralambieva IH, Vierkant RA, O’Byrne MM, Jacobson RM, Poland GA. The Association of CD46, SLAM, and CD209 Cellular Receptor Gene SNPs with Variations in Measles Vaccine-Induced Immune Responses--A Replication Study and Examination of Novel Polymorphisms. Human Heredity. 2011;72(3):206–23. doi: 10.1159/000331585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ovsyannikova IG, Pankratz VS, Vierkant RA, Jacobson RM, Poland GA. Consistency of HLA associations between two independent measles vaccine cohorts: a replication study. Vaccine. 2012;30(12):2146–52. doi: 10.1016/j.vaccine.2012.01.038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Ovsyannikova IG, Ryan JE, Vierkant RA, O’Byrne MM, Jacobson RM, Poland GA. Influence of host genetic variation on rubella-specific T cell cytokine responses following rubella vaccination. Vaccine. 2009;27:3359–66. doi: 10.1016/j.vaccine.2009.01.079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Chen MH, Zhu Z, Zhang Y, Favors S, Xu WB, Featherstone DA, et al. An indirect immunocolorimetric assay to detect rubella virus infected cells. Journal of Virological Methods. 2007;146(1–2):414–8. doi: 10.1016/j.jviromet.2007.08.021. [DOI] [PubMed] [Google Scholar]
- 29.Cleveland WS, Gross E, Shyu WM. Local regression models. In: Chambers JM, Hastie TJ, editors. Statistical Models. Wadsworth & Brooks/Cole; 1992. [Google Scholar]
- 30.Klein SL, Jedlicka A, Pekosz A. The Xs and Y of immune responses to viral vaccines. Lancet Infect Dis. 2010;10(5):338–49. doi: 10.1016/S1473-3099(10)70049-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Newport MJ, Goetghebuer T, Weiss HA, Whittle H, Siegrist CA, et al. The MRCGTSG. Genetic regulation of immune responses to vaccines in early life. Genes Immun. 2004;5(2):122–9. doi: 10.1038/sj.gene.6364051. [DOI] [PubMed] [Google Scholar]
- 32.Tan PL, Jacobson RM, Poland GA, Jacobsen SJ, Pankratz SV. Twin studies of immunogenicity - determining the genetic contribution to vaccine failure. Vaccine. 2001;19:2434–9. doi: 10.1016/s0264-410x(00)00468-0. [DOI] [PubMed] [Google Scholar]
- 33.Poland GA, Ovsyannikova IG, Jacobson RM, Smith DI. Heterogeneity in vaccine immune response: the role of immunogenetics and the emerging field of vaccinomics. Clin Pharmacol Ther. 2007;82(6):653–64. doi: 10.1038/sj.clpt.6100415. [DOI] [PubMed] [Google Scholar]
- 34.Haralambieva IH, Oberg AL, Ovsyannikova IG, Kennedy RB, Grill DE, Middha S, et al. Genome-wide characterization of transcriptional patterns in high and low antibody responders to rubella vaccination. PLos ONE. 2013;8(5):e62149. doi: 10.1371/journal.pone.0062149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Pankratz VS, Vierkant RA, O’Byrne MM, Ovsyannikova IG, Poland GA. Associations between SNPs in candidate immune-relevant genes and rubella antibody levels: a multigenic assessment. BMC Immunol. 2010;11(1):48. doi: 10.1186/1471-2172-11-48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Ovsyannikova IG, Jacobson RM, Vierkant RA, O’Byrne MM, Poland GA. Replication of rubella vaccine population genetic studies: validation of HLA genotype and humoral response associations. Vaccine. 2009;27(49):6926–31. doi: 10.1016/j.vaccine.2009.08.109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Skendzel LP. Rubella immunity. Defining the level of protective antibody. Am J Clin Pathol. 1996;106(2):170–4. doi: 10.1093/ajcp/106.2.170. [DOI] [PubMed] [Google Scholar]
- 38.Davidkin I, Jokinen S, Broman M, Leinikki P, Peltola H. Persistence of measles, mumps, and rubella antibodies in an MMR-vaccinated cohort: a 20-year follow-up. J Infect Dis. 2008;197(7):950–6. doi: 10.1086/528993. [DOI] [PubMed] [Google Scholar]
- 39.O’Shea S, Best JM, Banatvala JE, Marshall WC, Dudgeon JA. Rubella vaccination: persistence of antibodies for up to 16 years. Br J Med. 1982;285(July):253–5. doi: 10.1136/bmj.285.6337.253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Amanna IJ, Carlson NE, Slifka MK. Duration of humoral immunity to common viral and vaccine antigens. N Engl J Med. 2007;357(19):1903–15. doi: 10.1056/NEJMoa066092. [DOI] [PubMed] [Google Scholar]
- 41.LeBaron CW, Forghani B, Matter L, Reef SE, Beck C, Bi D, et al. Persistence of rubella antibodies after 2 doses of measles-mumps-rubella vaccine. J Infect Dis. 2009;200(6):888–99. doi: 10.1086/605410. [DOI] [PubMed] [Google Scholar]
- 42.Greenwood NP, Ovsyannikova IG, Vierkant RA, O’Byrne MM, Poland GA. A qualitative and quantitative comparison of two rubella virus-specific IgG antibody immunoassays. Viral Immunol. 2010;23(4):353–7. doi: 10.1089/vim.2010.0026. [DOI] [PubMed] [Google Scholar]
- 43.Tosh PK, Kennedy RB, Vierkant RA, Jacobson RM, Poland GA. Correlation between rubella antibody levels and cytokine measures of cell-mediated immunity. Viral Immunol. 2009;22(6):451–6. doi: 10.1089/vim.2009.0068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Elser B, Lohoff M, Kock S, Giaisi M, Kirchhoff S, Krammer PH, et al. IFN-gamma represses IL-4 expression via IRF-1 and IRF-2. Immunity. 2002 Dec;17(6):703–12. doi: 10.1016/s1074-7613(02)00471-5. [DOI] [PubMed] [Google Scholar]
- 45.Snapper CM, Paul WE. Interferon-gamma and B cell stimulatory factor-1 reciprocally regulate Ig isotype production. Science. 1987;236(4804):944–7. doi: 10.1126/science.3107127. [DOI] [PubMed] [Google Scholar]
- 46.Finkelman FD, Katona IM, Mosmann TR, Coffman RL. IFN-gamma regulates the isotypes of Ig secreted during in vivo humoral immune responses. Journal of Immunology. 1988 Feb 15;140(4):1022–7. [PubMed] [Google Scholar]
- 47.Hegazy AN, Peine M, Helmstetter C, Panse I, Frohlich A, Bergthaler A, et al. Interferons direct Th2 cell reprogramming to generate a stable GATA-3(+)T-bet(+) cell subset with combined Th2 and Th1 cell functions. Immunity. 2010 Jan 29;32(1):116–28. doi: 10.1016/j.immuni.2009.12.004. [DOI] [PubMed] [Google Scholar]
- 48.Fletcher HA. Correlates of immune protection from tuberculosis. Curr Mol Med. 2007 May;7(3):319–25. doi: 10.2174/156652407780598520. [DOI] [PubMed] [Google Scholar]
- 49.Vuola JM, Keating S, Webster DP, Berthoud T, Dunachie S, Gilbert SC, et al. Differential immunogenicity of various heterologous prime-boost vaccine regimens using DNA and viral vectors in healthy volunteers. Journal of Immunology. 2005 Jan 1;174(1):449–55. doi: 10.4049/jimmunol.174.1.449. [DOI] [PubMed] [Google Scholar]
- 50.Bejon P, Mwacharo J, Kai OK, Todryk S, Keating S, Lang T, et al. Immunogenicity of the candidate malaria vaccines FP9 and modified vaccinia virus Ankara encoding the pre-erythrocytic antigen ME-TRAP in 1–6 year old children in a malaria endemic area. Vaccine. 2006 May 29;24(22):4709–15. doi: 10.1016/j.vaccine.2006.03.029. [DOI] [PubMed] [Google Scholar]
- 51.Dunachie SJ, Walther M, Epstein JE, Keating S, Berthoud T, Andrews L, et al. A DNA prime-modified vaccinia virus ankara boost vaccine encoding thrombospondin-related adhesion protein but not circumsporozoite protein partially protects healthy malaria-naive adults against Plasmodium falciparum sporozoite challenge. Infection and Immunity. 2006 Oct;74(10):5933–42. doi: 10.1128/IAI.00590-06. [DOI] [PMC free article] [PubMed] [Google Scholar]