Abstract
This study investigated whether combining melatonin and apoptotic adipose-derived mesenchymal stem cells (A-ADMSC) was superior to ADMSC alone in ameliorating sepsis-induced acute lung injury. Adult male Sprague-Dawley rats (n=50) were randomized equally into five groups: sham controls (SC), sepsis induced by cecal-ligation and puncture (CLP), CLP-melatonin, CLP-A-ADMSC, and CLP-melatonin-A-ADMSC. Circulating interleukin (IL)-6 at 6, 18, and 72 hrs, were highest in CLP and lowest in SC groups, higher in CLP-melatonin than CLP-A-ADMSC and CLP-melatonin-A-ADMSC groups, higher in CLP-A-ADMSC than CLP-melatonin-A-ADMSC groups (all p<0.001). Immune reactivity (indicated by circulating cytotoxic-, and regulatory-T cells) and WBC count at 72 h exhibited the same pattern as that of circulating IL-6 (all p<0.001). Changes in histological scoring of lung parenchyma and the number of CD68+ and CD14+ cells showed a similar pattern compared to that of IL-6 level in all groups (all p<0.001). Changes in protein expressions of inflammatory (oxidative stress, RANTES, TNF-α, NF-κB, MMP-9, MIP-1, IL-1β), apoptotic (cleaved caspase 3 and PARP, mitochondrial Bax), fibrotic (Smad3, TGF-β) markers and those of reactive-oxygen-species (NOX-1, NOX-2) displayed an identical pattern compared to that of circulating IL-6 in all groups (all p<0.001). Anti-oxidative capacities (GR+, GPx+, HO-1, NQO-1+) and angiogenesis marker (CXCR4+ cells) were lowest in SC group but highest in CLP-melatonin-A-ADMSC group, lower in CLP than CLP-melatonin and CLP-A-ADMSC groups, and lower in CLP-melatonin than CLP-A-ADMSC groups (all p<0.001). In conclusion, combined melatonin and A-ADMSC were superior to A-ADMSC alone in protecting the lung from sepsis-induced injury.
Keywords: Sepsis-induced organ injury, inflammation, oxidative stress, reactive oxygen species, adipose-derived mesenchymal stem cells, and melatonin
Introduction
Despite advanced antibiotic regimens in our daily clinical practice, sepsis-induced mortality during hospitalization remains high [1-4]. The mortality rate is especially remarkable, ranging from 20% to over 60%, in ICU patients who usually are immunocompromised, in an unstable condition with common development of septic shock [5-7]. In the United States, severe sepsis accounts for as many deaths annually as myocardial infarctions [5]. Undoubtedly, there is an urgent need for innovative, safe, and efficacious therapeutic strategies for the sepsis syndrome.
Although the etiologies of sepsis syndrome and its prognosis have been widely investigated [5-8], its underlying mechanisms remain heavily debated [8-12].
Although various causes of sepsis-induced mortality have been proposed, uncontrolled immune response, overwhelming inflammation, over-production of reactive oxygen species (ROS), and endotoxin release from infectious microorganisms into circulation have been reported to be the major contributors [8-16]. This hyper-inflammatory and overwhelming responses involve activation of the innate immune system [9-12], accumulation of neutrophil and macrophage [12], secretion of cytokines [9,14], recruitment of T and B cells [12,16], and formation of antibodies [15] in an attempt to eliminate the pathogens but this process also causes collateral damage to major organs such as the kidneys, lungs, brain, and coagulation system, thereby leading to anergy of host-defense mechanisms, rapid organ failure and potential mortality [13]. Accordingly, strategies that target the inflammatory reaction, the generation and propagation of overwhelming immune responses, ROS and oxidative stress may have therapeutic potential.
Melatonin, or N-acetyl-5-methoxytryptamine, an indole, the chief secretory product of the pineal gland, is a direct free radical scavenger and an indirect antioxidant that acts to stabilize cell membranes, thereby making them less susceptible to oxidative insult and ultimately suppressing inflammatory reaction [13,17-20]. Previous experimental studies have identified the protective ability of melatonin against ischemia-reperfusion injury in multiple organs, including kidney, heart, and intestine [21-24]. However, whether melatonin therapy might be similarly effective against acute sepsis-induced lung injury remains unclear.
Growing evidence has demonstrated that mesenchymal stem cell (MSC) therapy not only improves ischemia-related organ dysfunction pro-bably through angiogenesis, paracrine/cytokine effect, and stem cell homing [25,26], but can also attenuate inflammation and down-regulate innate and adaptive immunity [27-30] through suppressing immunogenicity [27-31]. Intriguingly, adipose tissue-derived MSC may have more potent immunomodulatory capacity than that of bone marrow-derived MSC [30]. Another interesting finding is that apoptotic MSCs (i.e., dying stem cells) possess intense anti-inflammatory and immunomodulatory properties [31]. Additionally, one noteworthy finding is that we recently demonstrated the superiority of apoptotic adipose-derived MSC (A-ADMSC) to healthy ADMSC in protecting the lung from acute ischemia-reperfusion injury [32]. Moreover, combined therapy with melatonin and A-ADMSC was superior to A-ADMSC alone in attenuating acute lung ischemia-reperfusion injury [32]. Furthermore, another recent experimental study of our group also showed that A-ADMSC was more effective than healthy ADMSC in protecting the heart from sepsis-induced damage and reducing mortality [33].
Based on these recent reports [27-33], we hypothesized that: 1) melatonin therapy would be effective in ameliorating sepsis-induced acute lung injury; 2) A-ADMSC-assisted melatonin therapy might be superior to A-ADMSC therapy alone in attenuating acute sepsis-induced acute lung injury in rodents.
Materials and methods
Ethics
All animal experimental procedures were approved by the Institute of Animal Care and Use Committee at Kaohsiung Chang Gung Memorial Hospital (Affidavit of Approval of Animal Use Protocol No. 2008121108) and performed in accordance with the Guide for the Care and Use of Laboratory Animals (NIH publication No. 85-23, National Academy Press, Washington, DC, USA, revised 1996).
Animal grouping, estimation of animal number, rationale of treatment strategy, and adipose tissue isolation for ADMSC culturing
The procedure and protocol of the present study were based on our recent reports [30,32,33]. Pathogen-free, adult male Sprague-Dawley (SD) rats weighing 350-400 g (Charles River Technology, BioLASCO Taiwan Co. Ltd., Taiwan) were randomly divided into sham controls (SC, n=10) (i.e., cecal exposure without ligation or puncture), sepsis induced by cecal ligation and puncture [CLP + saline 3.0 mL, intra-peritoneal (i.p.)] at 30 min, 6 h, and 24 h after the procedure (n=10), CLP- melatonin [melatonin, i.p. at 30 minutes (20 mg/kg), 6 h (50 mg/kg), and 24 h (50 mg/kg) after the procedure; n=10], CLP-A-ADMSC [autologous apoptotic-ADMSC (1.2 x 106) at 30 minutes, 6 h, and 24 h after the procedure; n=10], and CLP-melatonin-A-ADMSC (n=10).
Based on the results of our recent report [33], the mortality rate from CLP-induced sepsis syndrome was about 40% to 50% in rats. Since the main purpose of the present study focused on clarifying the therapeutic effects of melatonin and A-ADMSC against lung parenchymal injury rather than determining the impact on mortality, the CLP procedure was modified to reduce the mortality rate. Therefore, only 10 animals were utilized in each group in the current study. Additionally, since A-ADMSC was found to be superior to the healthy ADMSC in reducing organ injury [32,33], this study was designed to focus on the effects of A-ADMSC + melatonin rather than healthy ADMSC + melatonin against sepsis-induced lung injury.
The choice of time points of ADMSC administration at 30 min, 6 h, and 18 h after CLP was based on our recent study that demonstrated marked attenuation of acute ischemia/reperfusion-induced lung injury following ADMSC through penile venous administration at these time points [30,32,33]. Besides, these time points were initially chosen in an attempt to mimic the clinical schedule of antibiotic treatment for patients with sepsis syndrome.
Rats in A-ADMSC and melatonin-A-ADMSC groups were anesthetized with inhalational 2.0% isoflurane 14 days before CLP to harvest autologous peri-epididymal adipose tissue as we recently reported [30,32,33]. Isolated ADMSCs were cultured in a 100 mm diameter dish with 10 mL DMEM culture medium containing 10% FBS for 14 days. Flow cytometric analysis was performed for identification of cellular characteristics (i.e., stem cell surface markers) after cell labeling with appropriate antibodies on day 14 of cell cultivation prior to implantation.
The procedures of cecal ligation and puncture (CLP) and measurement of tail systolic blood pressure (SBP)
Rats were anesthetized with inhalational 2.0% isoflurane and placed in a supine position on a warming pad at 37°C with the abdomen shaved. Under sterile conditions, the abdominal skin and muscle were opened and the cecum exposed in all groups. In the SC group, the abdomen was then closed and the animal was allowed to recover from anesthesia. In the CLP groups, the cecum was prolene suture ligated over its distal portion (i.e., distal ligation) and the cecum distal to the ligature was punctured twice with an 18G needle to allow the cecal contents to be expressed intraperitoneally, as previously described [33,34]. The abdominal wound was closed and the animal was allowed to recover from anesthesia.
The tail SBP was measured (Kent Scientific Corporation, Model no: CODA, U.S.A.) based on our recent report [33] by a technician who was blinded to the treatment protocols prior to and at 28 h (only for the survival animals) after CLP or sham procedure. The tail cuff approach to blood pressure measurement was conducted as follows: Initially the rat was warmed in a box at 37°C for 20 minutes before being placed in a restraining apparatus which was also kept at 37°C. The tail was inserted through the cuff which contained a photoelectric pulse detector through which the systolic blood pressure was recorded when the first oscillation appeared during the gradual reduction of cuff pressure. Mean blood pressure was determined from the cuff pressure when the amplitude of the oscillation reached its maximum. The SBP was consecutively and continuously measured for 30 times in each rat. Data of SBP recorded for the first 10 times and those recorded when the animals were agitated were discarded. Finally, reliable data of SBP were averaged for each rat and expressed as mean ± SD.
Definition of apoptotic ADMSCs
Serum deprivation of cells in vitro for induction of apoptosis was performed according to a previous study [35] and our recent report [33] with some modifications. Hence, apoptotic ADMSCs were first cultured in normal culture medium followed by 12 hours of hypoxia (1% oxygen) and 12 hours of serum-free cell culture. The principal concept in the present study for the induction of A-ADMSC using short-term hypoxia and serum deprivation is to artificially create a “stress environment” that would activate intra-cellular signaling pathway for secreting cytokines/chemokines and other mediators to seek the survival pathway.
Analyses of circulating levels of inflammatory biomarkers
Blood samples were stored at -80°C until analyses of interleukin (IL)-6 was performed in batches at the end of the experiment. Serum IL-6 concentration was assessed in duplicate with a commercially available ELISA kit (R&D systems, Inc. Minneapolis, MN). Circulating white blood cell (WBC) count was measured prior to and at 6 and 72 h after CLP with standard laboratory methods.
Flow cytometric quantification of helper T cells, cytotoxic T cells, and regulatory T cells (Tregs)
The procedure and protocol were based on our recent report [34]. In details, peripheral blood mononuclear cells (PBMCs) were obtained from the tail vein using a 27# needle. PBMCs and splenocytes (1.0 x 106 cells) were triple-stained with FITC-anti-CD3 (BioLegend), PE-anti-CD8a (BD Bioscience), and PE-Cy™5 anti-CD4 (BD bioscience). To identify CD4+CD25+Foxp3+ Tregs, PBMCs and splenocytes were triple-stained with Alexa Fluor® 488-anti-CD25 (BioLegend), PE-anti-Foxp3 (BioLegend), and PE-Cy™5 anti-CD4 (BD bioscience) according to the manufacturer’s protocol of Foxp3 Fix/Perm buffer set. The numbers of CD3+CD4+ helper T cells, CD3+CD8+ cytotoxic T cells and CD4+CD25+Foxp3+ Tregs were analyzed using flow cytometry (FC500, Beckman Coulter).
Histological assessment of lung injury
To identify alveolar sac distribution in lung parenchyma, the lung specimens from all animals were fixed in 10% buffered formalin before embedding in paraffin. Tissues were sectioned at 5 µm for light microscopy. Hematoxylin and eosin (H & E) staining was performed to determine the number of alveolar sacs in a blinded fashion as we previously reported [30]. Three lung sections from each rat were analyzed and three randomly selected high-power fields (HPFs; 100 x) were examined in each section. The mean number per HPF for each animal was then determined by summation of all numbers divided by 9. The extent of crowded area, which was defined as region of thickened septa in lung parenchyma associated with partial or complete collapse of alveoli on H & E-stained sections, was also performed in a blinded fashion. The following scoring system [28,30] was adopted: 0=no detectable crowded area; 1≤15% of crowded area; 2=15-25% of crowded area; 3=25-50% of crowded area; 4=50-75% of crowded area; 5≥75%-100% of crowded area/per HPF.
Immunohistochemical (IHC) and immunofluorescent (IF) studies
The procedures and protocols for IHC and IF examinations were also based on our recent study [27,30,32,33]. Briefly, for IHC staining, rehydrated paraffin sections were first treated with 3% H2O2 for 30 minutes and incubated with Immuno-Block reagent (BioSB) for 30 minutes at room temperature. Sections were then incubated with primary antibodies specifically against glutathione peroxidase (GPx; 1:500, Abcam) glutathione reductase (GR; 1:100, Abcam), NAD(P)H quinone oxidoreductase (NQO-1) (1: 400, Abcam), and CD14 (1:300, Bio SS) at 4°C overnight. Irrelevant antibodies and mouse control IgG [Abcam]) provided controls in the current study. IF staining was performed for the examinations of, HO-1 (1:250, Abcam), CD68 (1:100, Abcam ), CXCR4 (1:100, Santa cruz), and CD31 (1:200, SeroTec) using respective primary antibody with the use of irrelevant antibodies as controls. Three sections of lung specimens were analyzed in each rat. For quantification, three randomly selected HPFs (x 200 for IHC and IF studies) were analyzed in each section. The mean number per HPF for each animal was then determined by summation of all numbers divided by 9. An IHC-based scoring system was adopted for semi-quantitative analyses of NQO 1, CD14, GR and GPx as a percentage of positive cells in blinded fashion (score of positively-stained cell for GR and GPx: 0=no stain %; 1≤15%; 2=15~25%; 3=25~50%; 4=50~75%; 5≥75%-100%/per HPF).
Western blot analysis of lung specimens
Equal amounts (10-30 μg) of protein extracts from the left lung were loaded and separated by SDS-PAGE using 8-10% acrylamide gradients. Following electrophoresis, the separated proteins were transferred electrophoretically to a polyvinylidene difluoride (PVDF) membrane (Amersham Biosciences). Nonspecific proteins were blocked by incubating the membrane in blocking buffer (5% nonfat dry milk in T-TBS containing 0.05% Tween 20) overnight. The membranes were incubated with monoclonal antibodies against, polyclonal antibodies against tumor necrotic factor (TNF)-α (1:1000, Cell Signaling), nuclear factor (NF)-κB (1:250, Abcam), interleukin (IL)-1β (1:1000, Cell Signaling), MMP-9 (1:3000, Abcam), macrophage inflammatory protein (MIP)-1α (1:2000, Abcam), NOX-1 (1:1000, Sigma), NOX-2 (1:750, Sigma), NAD(P)H quinone oxidoreductase (NQO) 1 (1: 1000, Abcam), heme oxygense (HO)-1 (1:250, Abcam), γ-H2AX (1:1000, Cell Signaling), Bax (1:1000, Abcam), caspase 3 (1:1000, Cell Signaling), poly (ADP-ribose) polymerase (PARP) (1: 1000, Cell Signaling), Bcl-2 (1:250, Abcam), Bcl-2 (1:200, Abcam), Smad 3 (1:1000, Cell Signaling), transforming growth factor (TGF)-β ( 1:500, Abcam), Smad1/5 (1:1000, Cell Signaling), and bone morphogenetic protein (BMP)-2 (1:5000, Abcam). Signals were detected with horseradish peroxidase (HRP)-conjugated goat anti-mouse, goat anti-rat, or goat anti-rabbit IgG.
The Oxyblot Oxidized Protein Detection Kit was purchased from Chemicon (S7150). The procedure of 2,4-dinitrophenylhydrazine (DNPH) derivatization was carried out on 6 μg of protein for 15 minutes according to manufacturer’s instructions. One-dimensional electrophoresis was carried out on 12% SDS/polyacrylamide gel after DNPH derivatization. Proteins were transferred to nitrocellulose membranes which were then incubated in the primary antibody solution (anti-DNP 1:150) for two hours, followed by incubation with the secondary antibody solution (1:300) for one hour at room temperature. The washing procedure was repeated eight times within 40 minutes.
Immunoreactive bands were visualized by enhanced chemiluminescence (ECL; Amersham Biosciences), which was then exposed to Biomax L film (Kodak). For quantification, ECL signals were digitized using Labwork software (UVP). For oxyblot protein analysis, a standard control was loaded on each gel.
Statistical analyses
Quantitative data are expressed as mean ± SD. Statistical analysis was performed by ANOVA followed by Bonferroni multiple-comparison post hoc test. All analyses were conducted using SAS statistical software for Windows version 8.2 (SAS institute, Cary, NC). A probability value <0.05 was considered statistically significant.
Results
Apoptotic cell expression and flow cytometric analysis for circulating level of immune cells at 72 h after CLP procedure
Immunofluorescent microscopic findings (Figure 1A and 1B) showed that the incidence of cellular apoptosis was significantly higher in A-ADMSC than in healthy ADMSC (Figure 1C). Additionally, to investigate the level of acute immune response, circulating level of CD3+/CD4+ helper T cells (Figure 1D), CD3+/CD8+ cytotoxic T cells (Figure 1E), and CD4+CD25+Foxp3+ Tregs (Figure 1F) were measured at 72 h after CLP procedure. The circulating level of these three biomarkers were significantly higher in CLP and CLP-melatonin groups than in SC, CLP-A-ADMSC and CLP-melatonin-A-ADMSC animals, and significantly higher in CLP-A-ADMSC and CLP-melatonin-A-ADMSC than in SC groups, but it showed no difference between CLP and CLP-melatonin or between CLP-A-ADMSC and CLP-melatonin-A-ADMSC groups.
Figure 1.

Microscopic findings for the incidence of A-ADMSC after starvation-hypoxia stress and flow cytomtric Analysis of circulating Immune Cells (n=6). (A and B) Immunofluorescent (IF) microscopic findings (400 x) of cellular apoptosis of apoptotic adipose-derived mesenchymal stem cells (A-ADMSCs). (C) Cellular apoptosis was significantly higher in A-ADMSC than in healthy ADMSC. * vs. normal control, p<0.0001. Blue color indicates DAPI stain for nuclei. (D) Flow cytometric analysis of CD3+/CD4+ cells. * vs. other groups with different symbols (*, †, ‡), p<0.001. (E) Flow cytometric analysis of CD3+/CD8+ cells. * vs. other groups with different symbols (*, †, ‡), p<0.001. (F) Flow cytometric analysis of Treg+ cells. * vs. other groups with different symbols (*, †, ‡), p<0.001. Statistical analysis in (F) using one-way ANOVA, followed by Bonferroni multiple comparison post hoc test. Symbols (*, †, ‡) indicate significance (at 0.05 level). SC=sham control; CLP=cecal-ligation and puncture; Mel=melatonin.
Mortality rate and combined end-point at 72 h after CLP procedure
The mortality rate at 72 h after the CLP procedure did not differ among the five groups (Table 1). In addition, the presence of dark bloody ascites caused by sepsis was similar among the five groups (Table 1). However, the combined end-point (defined as mortality or ascites >1.0 mL) was significantly higher in CLP than in SC, CLP-melatonin, CLP-A-ADMSC and CLP-melatonin-A-ADMSC groups, but it showed no difference among the latter four groups (Table 1).
Table 1.
Serial Changes of BP, Inflammatory Biomarkers and Combined End-Points at 72 Hours after CLP
| Variables | Group 1 (n=10) | Group 2 (n=10) | Group 3 (n=10) | Group 4 (n=10) | Group 5 (n=10) | p-value |
|---|---|---|---|---|---|---|
| Mortality | 0% (0) | 40% (4) | 30% (3) | 10% (1) | 20% (2) | 0.181 |
| Dark bloody ascites* | 0% (0) | 40% (4) | 20% (2) | 10% (1) | 10% (1) | 0.230 |
| Combined end-point† | 0% (0)a | 60% (6)b | 30% (3)a | 10% (1)a | 20% (2)a | 0.02 |
| SBP (mmHg) at 0 h | 101 ± 8.8 | 108 ± 12.1 | 105 ± 9.6 | 108 ± 9.1 | 109 ± 8.8 | > 0.1 |
| SBP (mmHg) at 28 h | 109 ± 8.2 | 109 ± 21.5 | 106 ± 20.6 | 115 ± 16.4 | 109 ± 16.8 | >0.05 |
| WBC (x 103) at 0 h | 11.0 ± 1.7 | 11.2 ± 1.3 | 11.5 ± 1.9 | 11.0 ± 3.0 | 11.4 ± 2.3 | >0.5 |
| WBC (x 103) at 6 h | 10.0 ± 1.2a | 16.5 ±1.2b | 15.3 ± 1.1b | 13.1 ± 0.9c | 13.0 ± 1.9c | <0.001 |
| WBC (x 103) at 72 h | 7.9 ± 1.2a | 13.0 ± 1.5b | 12.5 ± 1.5b | 10.3 ± 1.7c | 10.9 ± 0.8c | <0.001 |
| IL-6 (pg/mL) at 0 h | 6.28 ± 1.49 | 7.3 ± 2.0 | 7.0 ± 1.9 | 7.1 ± 2.4 | 7.9 ± 1.8 | >0.5 |
| IL-6 (pg/mL) at 6 h | 9.2 ± 2.9a | 182 ± 103b | 177 ± 116b | 138 ± 61c | 140 ± 66c | <0.0001 |
| IL-6 (pg/mL) at 18 h | 9.4 ± 2.7a | 151 ± 146b | 129 ± 26c | 90.8 ± 26.2d | 87.6 ± 37.7d | <0.0001 |
| Il-6 (pg/mL) at 72 h | 6.0 ± 1.0a | 95.4 ± 50.8b | 70.8 ± 19.4c | 58.2 ± 9.8d | 66.3 ± 7.5d | <0.0001 |
Procedure. Data are expressed as mean ± SD or % (n).
indicate ≥1.0 cc ascites was collected.
defined as mortality or abdominal fluid presented ≥1.0 cc.
CLP=cecal-ligation and puncture; SBP=systolic blood pressure; WBC=white blood cell count; IL=interleukin. Group 1=sham control; Group 2=CLP; Group 3=CLP + melatonin; Group 4=CLP + apoptotic adipose derived mesenchymal stem cell (A-ADMSC); Group 5=CLP + melatonin + A-ADMSC.
Within a row, means without a common letters were different (P<0.05).
Within a row, means without a common letters were different (P<0.05).
Within a row, means without a common letters were different (P<0.05).
Within a row, means without a common letters were different (P<0.05).
Systolic blood pressure at 0 and 28 hours after CLP procedure and time courses of circulating levels of inflammatory biomarkers
Systolic blood pressure (SBP) did not differ among the five groups prior to or at 28 h after the CLP procedure (Table 1). However, SBP was significantly lower in expired animals than that in the survivors (115 ± 13 vs. 87 ± 7, p<0.001).
Prior to the CLP procedure, WBC count did not differ among the five groups. However, by 18 h and 72 h after the CLP procedure, WBC count was significantly higher in CLP and CLP-mela-tonin groups than in other animals, significantly higher in CLP-A-ADMSC and CLP-melatonin-A-ADMSC than in SC groups, but it exhibited no difference between CLP-ADMSC and CLP-melatonin-A-ADMSC groups (Table 1).
The circulating level of IL-6 was similar among the five groups prior to the CLP procedure (Table 1). However, by 6 h after cecal puncturing, the IL-6 level was significantly higher in CLP and CLP-melatonin than in SC, CLP-A-ADMSC and CLP-melatonin-A-ADMSC groups, and significantly higher in CLP-A-ADMSC and CLP-melatonin-A-ADMSC than in SC groups, but it showed no significant difference between CLP and CLP-melatonin or between CLP-A-ADMSC and CLP-melatonin-A-ADMSC groups (Table 1). Moreover, by 18 h and 72 h after CLP procedure, this biomarker in circulation was highest in CLP and lowest in SC, significantly higher in CLP-melatonin than in CLP-A-ADMSC and CLP-melatonin-ADMSC groups, but it showed no difference between the latter two groups (Table 1).
Histopathological findings in lung parenchyma at 72 hours after CLP procedure
To determine the impact of CLP-induced sepsis on lung parenchymal damage and the therapeutic effect of melatonin-A-ADMSC treatment against sepsis-induced lung parenchymal injury, H & E-stained lung sections were examined (Figure 2). The results demonstrated that the number of alveolar sacs was lowest in CLP and highest in SC groups, significantly lower in CLP-melatonin than in CLP-A-ADMSC and CLP-melatonin-A-ADMSC groups, and significantly lower in CLP-A-ADMSC than in CLP-melatonin-A-ADMSC groups. Conversely, lung parenchymal crowding showed a reverse pattern compared to that of the number of alveolar sacs among the five groups.
Figure 2.

Histopathological findings in lung parenchyma at 72 hours after CLP procedure (n=6). (A to F) Illustrations of microscopic findings (100 x) of H & E stain in lung sections. (G) Analytic results of number of alveolar sacs, * vs. other groups with different symbols (*, †, ‡, §, ¶), p<0.001. (H) Analytic results of crowded score, * vs. other groups with different symbols (*, †, ‡, §, ¶), p<0.0001. The scale bars in right lower corner represent 100 μm. Statistical analysis in (G) and (H) using one-way ANOVA, followed by Bonferroni multiple comparison post hoc test. Symbols (*, †, ‡, §, ¶) indicate significance (at 0.05 level). HPF=high-power field; SC=sham control; CLP=cecal-ligation and puncture; Mel=melatonin; A-ADMSC=apoptotic adipose-derived mesenchymal stem cell.
Infiltrations of CD14+ and CD68+ inflammatory cells in lung parenchyma 72 hours after CLP procedure
To examine the effect of sepsis syndrome on the distributions of the inflammatory cells in lung parenchyma, IHC and IF staining for identifying CD14+ and CD68+ cells, two markers of acute inflammatory cells, were performed respectively (Figure 3). The results showed that the numbers of CD14+ and CD68+ cells were highest in CLP and lowest in SC groups, significantly higher in CLP-melatonin than in CLP-A-ADMSC and CLP-melatonin-A-ADMSC groups, and significantly higher in CLP-A-ADMSC than in CLP-melatonin-ADMSC groups.
Figure 3.

Inflammatory cells infiltrated in lung parenchyma at 72 hours after CLP procedure (n=6). (A to E) Microscopic findings (200 x) of immunohistochemical (IHC) stain for CD14+ cells (brown color). (F) Analytic results of CD14+ cells, * vs. other groups with different symbols (*, †, ‡, §, ¶), p<0.0001. The scale bars in right lower corner represent 50 μm. (G to K) Microscopic findings (200 x) of immunofluorescent (IF) stain for CD68+ cells (green color of DAPI+ cells). Abundant Dil dye stained A-ADMSCs (white arrows) were found in (J) and (K) indicated that the transplanted A-ADMSCs were trapped in lung parenchyma. (L) Analytic results of CD68+ cells, * vs. other groups with different symbols (*, †, ‡, §, ¶), p<0.0001. The scale bars in right lower corner represent 50 μm. Statistical analysis in (F) and (L) using one-way ANOVA, followed by Bonferroni multiple comparison post hoc test. Symbols (*, †, ‡, §, ¶) indicate significance (at 0.05 level). HPF=high-power field; SC=sham control; CLP=cecal-ligation and puncture; Mel=melatonin; A-ADMSC=apoptotic adipose-derived mesenchymal stem cell.
Expressions of anti-oxidant cells in lung parenchyma 72 hours after CLP procedure
To evaluate whether melatonin-A-ADMSC treatment would augment anti-oxidant expression in lung parenchyma, IHC staining for GR+ (Figure 4), GPx+ (Figure 4) and NQO 1+ cells (Figure 5), as well as IF staining for HO-1+ cells (Figure 5), four indexes of anti-oxidants, were performed. The results demonstrated that the number of cells positive for GR, GPx, NQO 1, and HO-1 were highest in CLP-melatonin-A-ADMSC and lowest in SC groups, significantly higher in CLP-A-ADMSC than in CLP-melatonin and CLP groups, and significantly higher in CLP-melatonin group than in that of the CLP group. Of particularly important finding was that some of transplanted A-ADMSC was found to be positive stain for HO-1. This finding implicates that A-ADMSCs have capacity of HO-1 secretion after transplantation. These findings suggest that acute lung injury caused by sepsis syndrome induced an elevation in intrinsic anti-oxidant levels to protect against lung parenchymal damage and that melatonin-supported A-ADMSC treatment offered the most powerful protection against sepsis-induced acute lung injury.
Figure 4.

Expressions of GR and GPx anti-oxidant cells in lung parenchyma 72 hours after CLP procedure (n=6). (A to E) Microscopic findings (200 x) of IHC stain for glutathione reductase (GR)+ cells (brown color) in lung parenchyma. (F) Analytic results of GR+ cells, * vs. other groups with different symbols (*, †, ‡, §, ¶), p<0.0001. The scale bars in right lower corner represent 50 μm. (G to K) Microscopic findings (200 x) of IHC stain for glutathione peroxidase (GPx)+ cells (brown color) in lung parenchyma. (L) Analytic results of GPx+ cells, * vs. other groups with different symbols (*, †, ‡, §, ¶), p<0.0001. The scale bars in right lower corner represent 50 μm. Statistical analysis in (F) and (L) using one-way ANOVA, followed by Bonferroni multiple comparison post hoc test. Symbols (*, †, ‡, §, ¶) indicate significance (at 0.05 level). SC=sham control; CLP=cecal-ligation and puncture; Mel=melatonin; A-ADMSC=apoptotic adipose-derived mesenchymal stem cell.
Figure 5.

Expressions of NQO 1 and HO-1 anti-oxidant cells in lung parenchyma 72 hours after CLP procedure (n=6). (A to E) Microscopic findings (200 x) of IHC stain for NAD(P)H quinone oxidoreductase (NQO) 1+ cells (brown color) in lung parenchyma. (F) Analytic results of NQO 1+ cells, * vs. other groups with different symbols (*, †, ‡, §, ¶), p<0.0001. The scale bars in right lower corner represent 50 μm. (G to K) Microscopic findings (200 x) of IF stain for heme oxygenase (HO)-1+ cells (green color of DAPI+ cells) in lung parenchyma. Abundant Dil dye stained A-ADMSCs (white arrows) were found in (J) and (K) indicated that the transplanted A-ADMSCs were trapped in lung parenchyma. Additionally, some transplanted A-ADMSCs showing positive stain for OH-1 (yellow arrows). L) Analytic results of HO-1+ cells, * vs. other groups with different symbols (*, †, ‡, §, ¶), p<0.0001. The scale bars in right lower corner represent 50 μm. Statistical analysis in (F) and (L) using one-way ANOVA, followed by Bonferroni multiple comparison post hoc test. Symbols (*, †, ‡, §, ¶) indicate significance (at 0.05 level). SC=sham control; CLP=cecal-ligation and puncture; Mel=melatonin; A-ADMSC=apoptotic adipose-derived mesenchymal stem cell.
Protein expressions of oxidative stress and reactive oxygen species in lung parenchyma 72 hours after CLP procedure
The protein expressions of NOX-1 and NOX-2, two indexes of ROS, were highest in CLP and lowest in SC groups, and significantly higher in CLP-melatonin and CLP-A-ADMSC than in CLP-melatonin-A-ADMSC groups, but NOX-1 showed no difference between CLP-melatonin and CLP-A-ADMSC-treated animals (Figure 6A and 6B). However, the protein expression of NOX-2 was significantly higher in CLP-melatonin group than in CLP-A-ADMSC group. Additionally, the expression of oxidized protein (i.e., oxidative index) was highest in CLP and lowest in SC groups, significantly higher in CLP-melatonin than in CLP-A-ADMSC and CLP-melatonin-A-ADMSC groups, and significantly higher in CLP-A-ADMSC than in CLP-melatonin-A-ADMSC-treated animals (Figure 6C).
Figure 6.

Protein expressions of oxidative stress and ROS in lung parenchyma 72 hours after CLP procedure (n=6). A. Protein expression of NOX-1 in lung parenchyma, * vs. other groups with different symbols (*, †, ‡, §), p<0.001. B. Protein expression of NOX-2 in lung parenchyma, * vs. other groups with different symbols (*, †, ‡, §, ¶), p<0.001. C. Protein expression of oxidative index (protein carbonyls) in lung parenchyma, * vs. other groups with different symbols (*, †, ‡, §, ¶), p<0.0001. (Note: Right lane and left lane shown on the upper panel represent control oxidized molecular protein standard and protein molecular weight marker, respectively). DNP=1-3 dinitrophenylhydrazone. All statistical analyses were with one-way ANOVA followed by Bonferroni multiple comparison post hoc test. Symbols (*, †, ‡, §, ¶) indicate significance (at 0.05 level). SC=sham control; CLP=cecal-ligation and puncture; Mel=melatonin; A-ADMSC=apoptotic adipose-derived mesenchymal stem cell.
Protein expressions of inflammatory biomarkers in lung parenchyma 72 hours after CLP procedure
The protein expressions of IL-1β, TNF-α, NF-κB, MMP-9, and MIP-1α, five pro-inflammatory biomarkers, were highest in CLP and lowest in SC animals, significantly higher in CLP-melatonin than CLP-A-ADMSC and CLP-melatonin-A-ADMSC groups, and significantly higher in CLP-A-ADMSC group than in CLP-melatonin-A-ADMSC group (Figure 7A-E).
Figure 7.

Protein expressions of inflammatory biomarkers in lung parenchyma 72 hours after CLP procedure. A. Protein expressions of interleukin (IL)-1 in lung parenchyma, * vs. other groups with different symbols (*, †, ‡, §, ¶), p<0.001. B. Protein expressions of tumor necrosis factor (TNF)-α in lung parenchyma, * vs. other groups with different symbols (*, †, ‡, §, ¶), p<0.001. C. Protein expressions of tumor nuclear factor (NF)-κB in lung parenchyma, * vs. other groups with different symbols (*, †, ‡, §, ¶), p<0.001. D. Protein expressions of matrix metalloproteinase (MMP)-9 in lung parenchyma, * vs. other groups with different symbols (*, †, ‡, §, ¶), p<0.005. E. Protein expressions of macrophage inflammatory protein (MIP)-1α in lung parenchyma, * vs. other groups with different symbols (*, †, ‡, §, ¶), p<0.005. All statistical analyses were with one-way ANOVA followed by Bonferroni multiple comparison post hoc test. Symbols (*, †, ‡, §, ¶) indicate significance (at 0.05 level). SC=sham control; CLP=cecal-ligation and puncture; Mel=melatonin; A-ADMSC=apoptotic adipose-derived mesenchymal stem cell.
Protein expressions of DNA damage and apoptotic biomarkers in lung parenchyma 72 hours after CLP procedure
The protein expression of γ-H2AX, an indicator of DNA damage, was highest in CLP-melatonin and lowest in SC groups, significantly higher in CLP-melatonin than in CLP-A-ADMSC and CLP-melatonin-A-ADMSC groups, and significantly higher in CLP-A-ADMSC group than in CLP-melatonin-A-ADMSC groups (Figure 8A). Consistently, the protein expressions of mitochondrial Bax (Figure 8B), cleaved (i.e., active form) caspase 3 (Figure 8C) and PARP (Figure 8D), two apoptotic markers, showed an identical pattern compared to that of γ-H2AX. Conversely, the protein expression of Bcl-2 (Figure 8E), an anti-apoptotic biomarker, exhibited an opposite pattern compared to that of γ-H2AX among the five groups.
Figure 8.

Protein expressions of DNA damage, apoptotic and anti-inflammatory biomarkers. A. Protein expression of γ-H2AX in lung parenchyma, * vs. other groups with different symbols (*, †, ‡, §, ¶), p<0.0001. B. Protein expression of mitochondrial Bax in lung parenchyma, * vs. other groups with different symbols (*, †, ‡, §, ¶), p<0.001. C. Protein expression of cleaved caspase (c-Csp) 3 I lung parenchyma, * vs. other groups with different symbols (*, †, ‡, §, ¶), p<0.0001. D. Protein expression of cleaved poly (ADP-ribose) polymerase (PARP) in lung parenchyma, * vs. other groups with different symbols (*, †, ‡, §, ¶), p<0.001. E. Protein expression of Bcl-2 in lung parenchyma, * vs. other groups with different symbols (*, †, ‡, §, ¶), p<0.008. All statistical analyses were with one-way ANOVA followed by Bonferroni multiple comparison post hoc test. Symbols (*, †, ‡, §, ¶) indicate significance (at 0.05 level). SC=sham control; CLP=cecal-ligation and puncture; Mel=melatonin; A-ADMSC=apoptotic adipose-derived mesenchymal stem cell.
Protein expressions of fibrotic, anti-fibrotic and anti-oxidant biomarkers in lung parenchyma 72 hours after CLP procedure
The protein expressions of Smad3 (Figure 9A) and TGF-β (Figure 9B), two fibrotic markers, were highest in CLP and lowest in SC groups, significantly higher in CLP-melatonin than in CLP-A-ADMSC and CLP-melatonin-A-DMSC groups, and significantly higher in CLP-A-ADMSC than in CLP-melatonin-A-ADMSC-treated animals. On the other hand, the protein expressions of Smad1/5 (Figure 9C) and BMP-2 (Figure 9D), two anti-fibrosis indexes, displayed an opposite pattern compared to that of fibrotic markers among the five groups. In addition, the protein expressions of HO-1 (Figure 9E) and NQO 1 (Figure 9F), two anti-oxidation indices, displayed an opposite pattern compared to that of fibrotic markers among the five groups.
Figure 9.

Protein expressions of Fibrotic, anti-Fibrotic and anti-oxidant biomarkers. A. Protein expression of phosphorylated (p)-Smad3 in lung parenchyma, * vs. other groups with different symbols (*, †, ‡, §, ¶), p<0.0001. B. Protein expression of transforming growth factor (TGF)-β, * vs. other groups with different symbols (*, †, ‡, §, ¶), p<0.0001. C. Protein expression of p-Smad1/5 in lung parenchyma, * vs. other groups with different symbols (*, †, ‡, §, ¶), p<0.001. D. Protein expression of Bone morphogenic protein (BMP)-2 in lung parenchyma, * vs. other groups with different symbols (*, †, ‡, §, ¶), p<0.001. E. Protein expression of HO-1 in lung parenchyma, * vs. other groups with different symbols (*, †, ‡, §, ¶), p<0.0001. F. rotein expression of NQ0 1 in lung parenchyma, * vs. other groups with different symbols (*, †, ‡, §, ¶), p<0.0001. All statistical analyses were with one-way ANOVA followed by Bonferroni multiple comparison post hoc test. Symbols (*, †, ‡, §, ¶) indicate significance (at 0.05 level). SC=sham control; CLP=cecal-ligation and puncture; Mel=melatonin; A-ADMSC=apoptotic adipose-derived mesenchymal stem cell.
Expressions of surface markers of CD31+ endothelial cells and CXCR4+ endothelial progenitor cells in lung parenchyma 72 hours after CLP procedure
To assess whether transplanted A-ADMSCs would differentiate into endothelial cells (EC) and endothelial progenitor cells (EPC), IF microscopic findings for identification of CD31+ cells (Figure 10A-E), an indicator of EC surface marker, and CXCR4+ cells (Figure 11A-E, 11G, 11H) (i.e. an indicator of EPC surface marker) were performed. As expected, some of transplanted A-ADMSC had differentiated into CD31+ cells (Figure 10D, 10E) and some of the others had differentiated into CXCR4+ cells (Figure 11D, 11E, 11G, 11H). However, majority of these transplanted A-ADMSCs remained unchanged as Dil-dye positively stained cells. Additionally, some of the transplanted A-ADMSCs had been found to engraft into the pulmonary arteries and lung parenchyma. Moreover, the positively stained cells for CD31 (Figure 10F) was highest in SC and lowest in CLP, significantly lower in CLP-melatonin than in CLP-A-ADMSC and CLP-melatonin-A-ADMSC, and significantly higher in CLP-melatonin-A-ADMSC than that of CLP-A-ADMSC. Furthermore, the positively stained cells for CXCR4 (Figure 11F) were highest in CLP-melatonin-A-ADMSC and lowest in SC, significantly higher in CLP-A-ADMSC than in CLP-melatonin and CLP, and significantly higher in CLP-melatonin than in CLP.
Figure 10.
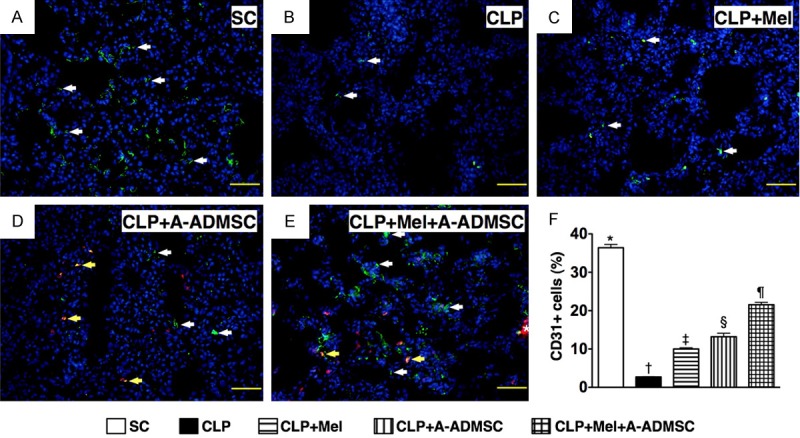
IF stain for identifying surface makers of endothelial cell (EC). (A to E) IF microscopic findings (200 x) for identification of positively-stained CD31 cells (white arrows). (D and E) Abundant transplanted A-ADMSCs with positively Dil-dye stain (red color) were found to be present in the lung parenchyma. Merge of double stain (i.e. Dil dye + CD31 staining) showing some of A-ADMSCs (D, E) exhibited EC surface marker of CD31 (yellow arrows). Some of transfused A-ADMSCs were identified to engraft into lung parenchyma (E) (asterisk marker). (F) Analytic results of CD31+ cells, * vs. other groups with different symbols (*, †, ‡, §), p<0.0001. The scale bars in right lower corner represent 50 μm. Symbols (*, †, ‡, §, ¶) indicate significance (at 0.05 level). SC=sham control; CLP=cecal-ligation and puncture; Mel=melatonin; A-ADMSC=apoptotic adipose-derived mesenchymal stem cell.
Figure 11.

IF stain for identifying surface makers of endothelial progenitor cells. (A to E & G to H) IF microscopic findings (400 x) for identification of positively-stained CXCR4 cells (white arrows). (D & E and G & H) Numerous transplanted A-ADMSCs with positively Dil-dye stain (red color) were found to be present in the lung parenchyma. (D & E) Merge of double stain (i.e. Dil dye + CXCR4 staining) showing some of A-ADMSCs exhibited EPC surface marker of CXCR4 (yellow arrows). Some of transfused A-ADMSCs were identified to engraft into the pulmonary arteries (G, H) (pink arrow) and lung parenchyma (E, G) (asterisk marker). (F) Analytic results of CXCR4+ cells, * vs. other groups with different symbols (*, †, ‡, §, ¶), p<0.0001. The scale bars in right lower corner represent 20 μm. All statistical analyses were with one-way ANOVA followed by Bonferroni multiple comparison post hoc test. Symbols (*, †, ‡, §, ¶) indicate significance (at 0.05 level). SC=sham control; CLP=cecal-ligation and puncture; Mel=melatonin; A-ADMSC=apoptotic adipose-derived mesenchymal stem cell.
Discussion
The present study, which investigated the therapeutic impact of combined melatonin-A-ADMSC treatment on acute sepsis-induced lung injury, provided several striking implications. First, the lung was one organ vulnerable to CLP-induced sepsis syndrome. Second, combined therapy with melatonin and A-ADMSC notably improved combined end-point in the setting of CLP-induced sepsis syndrome. Third, combined melatonin-A-ADMSC regimen was superior to either alone in reducing lung parenchymal injury, inflammation, generation of oxidative stress, ROS, fibrosis and apoptosis as well as augmenting the production of anti-oxidants.
Impact of the melatonin-A-ADMSC therapy on sepsis-induced lung damage
Physiologically, the lung receives not only venous return (i.e., de-oxygenated blood from right ventricle), but also both toxic and non-toxic metabolic products from blood. Besides, the lung is also exposed to the polluted air. The lung parenchyma, therefore, is often exposed to ROS as well as the bacteremia [13-15] as in sepsis. Recently, our study has demonstrated that CLP-induced sepsis syndrome caused the damage of three major organs, including the heart, kidney, and lung [33]. One important finding in the present study is remarkable lung parenchyma damage (i.e., decreased alveolar sacs and increased crowded lung score) in CLP animals. Our findings are, therefore, consistent with those of the previous studies [13,33]. The most important finding (i.e., by histopathology) in the current study is that, as compared with animals with CLP only, pulmonary damage was notably reduced in either melatonin or A-ADMSC treatment groups. Of importance is that combined melatonin-A-ADMSC therapy was superior to either regimen alone not only in attenuating lung parenchymal damage, but also in improving the combined end-point (i.e. mortality or ascites).
Mechanisms underlying improved outcomes after melatonin-A-ADMSC treatment in the setting of CLP-induced sepsis-attenuating inflammation and oxidative stress
Although the etiologies of sepsis syndrome and its prognosis have been extensively explored [5-8], the mechanistic bases are still unknown [8-12]. An important finding in the present study is that not only was the circulating level of cytokine (IL-6), but also that of immune cells (CD3+/CD4+, CD3+/CD8+ and Treg+ cells) was markedly increased in CLP animals. Additionally, expressions of inflammatory and oxidative biomarkers in lung parenchyma were remarkably augmented in CLP animals without treatment. Previous studies have demonstrated substantial upregulation of the expressions of inflammatory and oxidative biomarkers and activation of the immune system during sepsis [8-16]. In this way, the results of our study support the findings of the previous studies [8-16]. The essential finding in the current study is that, as compared with animals receiving CLP only, melatonin and especially A-ADMSC treatment significantly attenuated the circulating levels of cytokine and immune cells as well as the generation of inflammation and oxidative stress in lung parenchyma following CLP-induced sepsis. Interestingly, the finding is consistent with that of our recent study [32] that has also shown markedly suppressed expressions of inflammation and generation of ROS through melatonin and ADMSC treatment in the setting of acute lung ischemia-reperfusion injury. Of particular interest is the finding that combined melatonin-A-ADMSC treatment synergistically reduced molecular-cellular perturbations in the setting of sepsis syndrome.
Mechanisms of improving outcomes after melatonin-A-ADMSC treatment in the setting of CLP-induced sepsis-up-regulating productions of anti-oxidants and the anti-inflammatory cytokine
It is well-known that ROS production was markedly enhanced after acute ischemia-reperfusion injury and sepsis syndrome [13,32,33]. On the other hand, melatonin has been identified to be a direct free radical scavenger and an indirect antioxidant [13,36-39]. Moreover, A-ADMSC has been shown to possess potent capacity of suppressing immunogenicity and ROS generation [31-33]. This laid the basis for the utilization of melatonin-A-ADMSC regimen for the treatment of CLP-induced sepsis. As expected, the protein expressions of anti-inflammatory marker (IL-10) and anti-oxidants at cellular (GR+, GPx+, NQO 1+, HO-1+ cells) and protein (NQO 1, HO-1) levels were significantly up-regulated following either melatonin or A-ADMSC treatment, and further significantly up-regulated after combined melatonin-A-ADMSC treatment. These findings, in addition to being supported by previous studies [13,31-33,36-39], could also partially explain the effectiveness of monotherapy (i.e. melatonin or -A-ADMSC) and more effectiveness of combined regimen against sepsis-induced lung parenchymal injury.
Mechanisms underlying improved outcomes after melatonin-A-ADMSC treatment in the setting of CLP-induced sepsis-down-regulation of fibrosis and apoptosis
Enhancement of fibrosis and apoptosis frequently occurs in various disease entities, including myocardial infarction [25,26], ischemia-reperfusion injury [29,30,32] and sepsis syndrome [33]. A principal finding in the present study is that, as compared with sham controls, both fibrotic and apoptotic biomarkers in lung parenchyma were remarkably increased in CLP animals. Our findings reinforce those of previous studies [25,26,29,30,32,33]. On the other hand, these biomarkers were significantly suppressed after melatonin treatment and further reduced after A-ADMSC treatment. These findings are comparable with those of our recent report showing significant amelioration in the expressions of these biomarkers after melatonin and ADMSC treatment in the setting of acute lung ischemia-reperfusion injury [32]. Besides, the present study also demonstrated that the expressions of anti-fibrotic and anti-apoptotic biomarkers were notably up-regulated after melatonin or A-ADMSC treatment. One particularly important finding in the current study is that combined melatonin-A-ADMSC treatment was superior to either regimen alone in reducing the expressions of apoptotic and fibrotic parameters, whereas the expressions of anti-fibrotic and anti-apoptotic biomarkers showed an opposite pattern compared to that of fibrotic and apoptotic indices among the animals. These findings might once again partially explain the enhanced effectiveness of combined therapy in reducing lung parenchymal injury in the setting of CLP-induced sepsis.
Mechanisms underlying improved outcomes after A-ADMSC treatment in the setting of CLP-induced sepsis-preservation of endothelial function and repairmen of lung architecture
Growing data has demonstrated that stem cell therapy plays an important role for restoring the ischemia-related organ dysfunction through the mechanisms of angiogenesis, paracrine/chemokine effects, repairmen of injured endothelial cell function, engraftment of implanted stem cells into the native ischemic cells, and stem cell homing [25-27]. In the present study, we found that A-ADMSC and melatonin-A-ADMSC therapy markedly enhanced the expressions of EC (i.e., CD31+ cell) and EPC (i.e., CCXR4+ cells) surface markers and the engraftment of these two cells as well as transplanted A-ADMSCs in pulmonary arteries and lung parenchyma. In this way, our findings, in addition to strengthening the findings of previous studies [25-27], could also, at least in part, explain why melatonin-A-ADMSC therapy offer an augmented effect on reducing lung parenchymal injury in setting of sepsis syndrome.
Study limitations
This study has limitations. First, since this study investigated the acute rather the chronic phase of sepsis syndrome, the impact of melatonin-A-AMDSC therapy on long-term outcome after CLP-induced sepsis remains unclear. Second, although the expressions of various biomarkers had been investigated and the results of the present study were promising, the exact underlying mechanisms involved in melatonin-A-ADMSC treatment against CLP-induced sepsis are still not fully understood. Based on the findings of the present study, the direct and indirect mechanisms probably involved in the observed therapeutic effects that are illustrated in Figure 12.
Figure 12.

Proposed mechanisms underlying the effects of melatonin-A-ADMSC therapy on CLP-induced lung parenchymal injury in a rodent model based on findings of the present study. ADMSC=adipose-derived mesenchymal stem cell; BMP=bone morphogenetic protein; GPx=glutathione peroxidase; GR=glutathione reductase; HO=heme oxygense; IL-1β=interleukin (IL)-1β; MMP-9=matrix metalloproteinase-9; MIP=macrophage inflammatory protein; NQO 1=NAD(P)H quinone oxidoreductase; NOX=nicotinamide adenine dinucleotide phosphate (NADPH) oxidase; PARP=poly (ADP-ribose) polymerase; TNF=tumor necrotic factor; TGF=transforming growth factor.
In conclusion, the results of the present study highlight that combined melatonin-A-ADMSC treatment was superior to either regimen alone in ameliorating lung injury in the setting of CLP-induced sepsis in a rodent model through the down-regulation of inflammation, oxidative stress, ROS, fibrosis, and apoptosis as well as the up-regulation of anti-inflammatory, anti-fibrotic, anti-apoptotic, and anti-oxidative effects, and integrity of endothelial function.
Acknowledgements
This study was supported by a program grant from Chang Gung Memorial Hospital, Chang Gung University (Grant number: CMRPG8C0371).
Disclosure of conflict of interest
None of the authors report any conflict of interest or financial disclosure.
References
- 1.Cronshaw HL, Daniels R, Bleetman A, Joynes E, Sheils M. Impact of the Surviving Sepsis Campaign on the recognition and management of severe sepsis in the emergency department: are we failing? Emerg Med J. 2011;28:670–675. doi: 10.1136/emj.2009.089581. [DOI] [PubMed] [Google Scholar]
- 2.Dellinger RP, Levy MM, Carlet JM, Bion J, Parker MM, Jaeschke R, Reinhart K, Angus DC, Brun-Buisson C, Beale R, Calandra T, Dhainaut JF, Gerlach H, Harvey M, Marini JJ, Marshall J, Ranieri M, Ramsay G, Sevransky J, Thompson BT, Townsend S, Vender JS, Zimmerman JL, Vincent JL International Surviving Sepsis Campaign Guidelines Committee; American Association of Critical-Care Nurses; American College of Chest Physicians; American College of Emergency Physicians; Canadian Critical Care Society; European Society of Clinical Microbiology and Infectious Diseases; European Society of Intensive Care Medicine; European Respiratory Society; International Sepsis Forum; Japanese Association for Acute Medicine; Japanese Society of Intensive Care Medicine; Society of Critical Care Medicine; Society of Hospital Medicine; Surgical Infection Society; World Federation of Societies of Intensive and Critical Care Medicine. Surviving Sepsis Campaign: international guidelines for management of severe sepsis and septic shock: 2008. Crit Care Med. 2008;36:296–327. doi: 10.1097/01.CCM.0000298158.12101.41. [DOI] [PubMed] [Google Scholar]
- 3.Grozdanovski K, Milenkovic Z, Demiri I, Spasovska K. Prediction of outcome from community-acquired severe sepsis and septic shock in tertiary-care university hospital in a developing country. Crit Care Res Pract. 2012;2012:182324. doi: 10.1155/2012/182324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Tipler PS, Pamplin J, Mysliwiec V, Anderson D, Mount CA. Use of a protocolized approach to the management of sepsis can improve time to first dose of antibiotics. J Crit Care. 2013;28:148–151. doi: 10.1016/j.jcrc.2012.08.021. [DOI] [PubMed] [Google Scholar]
- 5.Angus DC, Linde-Zwirble WT, Lidicker J, Clermont G, Carcillo J, Pinsky MR. Epidemiology of severe sepsis in the United States: analysis of incidence, outcome, and associated costs of care. Crit Care Med. 2001;29:1303–1310. doi: 10.1097/00003246-200107000-00002. [DOI] [PubMed] [Google Scholar]
- 6.Brun-Buisson C, Meshaka P, Pinton P, Vallet B EPISEPSIS Study Group. EPISEPSIS: a reappraisal of the epidemiology and outcome of severe sepsis in French intensive care units. Intensive Care Med. 2004;30:580–588. doi: 10.1007/s00134-003-2121-4. [DOI] [PubMed] [Google Scholar]
- 7.Brun-Buisson C, Roudot-Thoraval F, Girou E, Grenier-Sennelier C, Durand-Zaleski I. The costs of septic syndromes in the intensive care unit and influence of hospital-acquired sepsis. Intensive Care Med. 2003;29:1464–1471. doi: 10.1007/s00134-003-1877-x. [DOI] [PubMed] [Google Scholar]
- 8.Hotchkiss RS, Karl IE. The pathophysiology and treatment of sepsis. N Engl J Med. 2003;348:138–150. doi: 10.1056/NEJMra021333. [DOI] [PubMed] [Google Scholar]
- 9.Hein F, Massin F, Cravoisy-Popovic A, Barraud D, Levy B, Bollaert PE, Gibot S. The relationship between CD4+CD25+CD127- regulatory T cells and inflammatory response and outcome during shock states. Crit Care. 2010;14:R19. doi: 10.1186/cc8876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Taylor AL, Llewelyn MJ. Superantigen-induced proliferation of human CD4+CD25- T cells is followed by a switch to a functional regulatory phenotype. J Immunol. 2010;185:6591–6598. doi: 10.4049/jimmunol.1002416. [DOI] [PubMed] [Google Scholar]
- 11.Venet F, Chung CS, Kherouf H, Geeraert A, Malcus C, Poitevin F, Bohe J, Lepape A, Ayala A, Monneret G. Increased circulating regulatory T cells (CD4(+)CD25(+)CD127(-)) contribute to lymphocyte anergy in septic shock patients. Intensive Care Med. 2009;35:678–686. doi: 10.1007/s00134-008-1337-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Venet F, Chung CS, Monneret G, Huang X, Horner B, Garber M, Ayala A. Regulatory T cell populations in sepsis and trauma. J Leukoc Biol. 2008;83:523–535. doi: 10.1189/jlb.0607371. [DOI] [PubMed] [Google Scholar]
- 13.Fry DE. Sepsis, systemic inflammatory response, and multiple organ dysfunction: the mystery continues. Am Surg. 2012;78:1–8. [PubMed] [Google Scholar]
- 14.Hartemink KJ, Groeneveld AB. The hemodynamics of human septic shock relate to circulating innate immunity factors. Immunol Invest. 2010;39:849–862. doi: 10.3109/08820139.2010.502949. [DOI] [PubMed] [Google Scholar]
- 15.Maury E, Blanchard HS, Chauvin P, Guglielminotti J, Alzieu M, Guidet B, Offenstadt G. Circulating endotoxin and antiendotoxin antibodies during severe sepsis and septic shock. J Crit Care. 2003;18:115–120. doi: 10.1053/jcrc.2003.50004. [DOI] [PubMed] [Google Scholar]
- 16.Monserrat J, de Pablo R, Reyes E, Diaz D, Barcenilla H, Zapata MR, De la Hera A, Prieto A, Alvarez-Mon M. Clinical relevance of the severe abnormalities of the T cell compartment in septic shock patients. Crit Care. 2009;13:R26. doi: 10.1186/cc7731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Galano A. On the direct scavenging activity of melatonin towards hydroxyl and a series of peroxyl radicals. Phys Chem Chem Phys. 2011;13:7178–7188. doi: 10.1039/c0cp02801k. [DOI] [PubMed] [Google Scholar]
- 18.Galano A, Tan DX, Reiter RJ. Melatonin as a natural ally against oxidative stress: a physicochemical examination. J Pineal Res. 2011;51:1–16. doi: 10.1111/j.1600-079X.2011.00916.x. [DOI] [PubMed] [Google Scholar]
- 19.Mauriz JL, Collado PS, Veneroso C, Reiter RJ, Gonzalez-Gallego J. A review of the molecular aspects of melatonin’s anti-inflammatory actions: recent insights and new perspectives. J Pineal Res. 2013;54:1–14. doi: 10.1111/j.1600-079X.2012.01014.x. [DOI] [PubMed] [Google Scholar]
- 20.Zhang H, Liu D, Wang X, Chen X, Long Y, Chai W, Zhou X, Rui X, Zhang Q, Wang H, Yang Q. Melatonin improved rat cardiac mitochondria and survival rate in septic heart injury. J Pineal Res. 2013;55:1–6. doi: 10.1111/jpi.12033. [DOI] [PubMed] [Google Scholar]
- 21.Kesik V, Guven A, Vurucu S, Tunc T, Uysal B, Gundogdu G, Oztas E, Korkmaz A. Melatonin and 1400 W ameliorate both intestinal and remote organ injury following mesenteric ischemia/reperfusion. J Surg Res. 2009;157:e97–e105. doi: 10.1016/j.jss.2008.12.024. [DOI] [PubMed] [Google Scholar]
- 22.Patschan D, Hildebrandt A, Rinneburger J, Wessels JT, Patschan S, Becker JU, Henze E, Kruger A, Muller GA. The hormone melatonin stimulates renoprotective effects of “early outgrowth” endothelial progenitor cells in acute ischemic kidney injury. Am J Physiol Renal Physiol. 2012;302:F1305–1312. doi: 10.1152/ajprenal.00445.2011. [DOI] [PubMed] [Google Scholar]
- 23.Sahna E, Parlakpinar H, Turkoz Y, Acet A. Protective effects of melatonin on myocardial ischemia/reperfusion induced infarct size and oxidative changes. Physiol Res. 2005;54:491–495. [PubMed] [Google Scholar]
- 24.Sener G, Sehirli AO, Keyer-Uysal M, Arbak S, Ersoy Y, Yegen BC. The protective effect of melatonin on renal ischemia-reperfusion injury in the rat. J Pineal Res. 2002;32:120–126. doi: 10.1034/j.1600-079x.2002.1848.x. [DOI] [PubMed] [Google Scholar]
- 25.Leu S, Sun CK, Sheu JJ, Chang LT, Yuen CM, Yen CH, Chiang CH, Ko SF, Pei SN, Chua S, Youssef AA, Wu CJ, Yip HK. Autologous bone marrow cell implantation attenuates left ventricular remodeling and improves heart function in porcine myocardial infarction: an echocardiographic, six-month angiographic, and molecular-cellular study. Int J Cardiol. 2011;150:156–168. doi: 10.1016/j.ijcard.2010.03.007. [DOI] [PubMed] [Google Scholar]
- 26.Yip HK, Chang LT, Wu CJ, Sheu JJ, Youssef AA, Pei SN, Lee FY, Sun CK. Autologous bone marrow-derived mononuclear cell therapy prevents the damage of viable myocardium and improves rat heart function following acute anterior myocardial infarction. Circ J. 2008;72:1336–1345. doi: 10.1253/circj.72.1336. [DOI] [PubMed] [Google Scholar]
- 27.Chen YT, Sun CK, Lin YC, Chang LT, Chen YL, Tsai TH, Chung SY, Chua S, Kao YH, Yen CH, Shao PL, Chang KC, Leu S, Yip HK. Adipose-derived mesenchymal stem cell protects kidneys against ischemia reperfusion injury through suppressing oxidative stress and inflammatory reaction. J Transl Med. 2011;9:51. doi: 10.1186/1479-5876-9-51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Le Blanc K, Tammik L, Sundberg B, Haynesworth SE, Ringden O. Mesenchymal stem cells inhibit and stimulate mixed lymphocyte cultures and mitogenic responses independently of the major histocompatibility complex. Scand J Immunol. 2003;57:11–20. doi: 10.1046/j.1365-3083.2003.01176.x. [DOI] [PubMed] [Google Scholar]
- 29.Maumus M, Guerit D, Toupet K, Jorgensen C, Noel D. Mesenchymal stem cell-based therapies in regenerative medicine: applications in rheumatology. Stem Cell Res Ther. 2011;2:14. doi: 10.1186/scrt55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Sun CK, Yen CH, Lin YC, Tsai TH, Chang LT, Kao YH, Chua S, Fu M, Ko SF, Leu S, Yip HK. Autologous transplantation of adipose-derived mesenchymal stem cells markedly reduced acute ischemia-reperfusion lung injury in a rodent model. J Transl Med. 2011;9:118. doi: 10.1186/1479-5876-9-118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Thum T, Bauersachs J, Poole-Wilson PA, Volk HD, Anker SD. The dying stem cell hypothesis: immune modulation as a novel mechanism for progenitor cell therapy in cardiac muscle. J Am Coll Cardiol. 2005;46:1799–1802. doi: 10.1016/j.jacc.2005.07.053. [DOI] [PubMed] [Google Scholar]
- 32.Yip HK, Chang YC, Wallace CG, Chang LT, Tsai TH, Chen YL, Chang HW, Leu S, Zhen YY, Tsai CY, Yeh KH, Sun CK, Yen CH. Melatonin treatment improves adipose-derived mesenchymal stem cell therapy for acute lung ischemia-reperfusion injury. J Pineal Res. 2013;54:207–221. doi: 10.1111/jpi.12020. [DOI] [PubMed] [Google Scholar]
- 33.Chang CL, Leu S, Sung HC, Zhen YY, Cho CL, Chen A, Tsai TH, Chung SY, Chai HT, Sun CK, Yen CH, Yip HK. Impact of apoptotic adipose-derived mesenchymal stem cells on attenuating organ damage and reducing mortality in rat sepsis syndrome induced by cecal puncture and ligation. J Transl Med. 2012;10:244. doi: 10.1186/1479-5876-10-244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Rittirsch D, Huber-Lang MS, Flierl MA, Ward PA. Immunodesign of experimental sepsis by cecal ligation and puncture. Nat Protoc. 2009;4:31–36. doi: 10.1038/nprot.2008.214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Zhu W, Chen J, Cong X, Hu S, Chen X. Hypoxia and serum deprivation-induced apoptosis in mesenchymal stem cells. Stem Cells. 2006;24:416–425. doi: 10.1634/stemcells.2005-0121. [DOI] [PubMed] [Google Scholar]
- 36.Reiter R, Tang L, Garcia JJ, Munoz-Hoyos A. Pharmacological actions of melatonin in oxygen radical pathophysiology. Life Sci. 1997;60:2255–2271. doi: 10.1016/s0024-3205(97)00030-1. [DOI] [PubMed] [Google Scholar]
- 37.Reiter RJ, Melchiorri D, Sewerynek E, Poeggeler B, Barlow-Walden L, Chuang J, Ortiz GG, Acuna-Castroviejo D. A review of the evidence supporting melatonin’s role as an antioxidant. J Pineal Res. 1995;18:1–11. doi: 10.1111/j.1600-079x.1995.tb00133.x. [DOI] [PubMed] [Google Scholar]
- 38.Reiter RJ, Tan DX, Osuna C, Gitto E. Actions of melatonin in the reduction of oxidative stress. A review. J Biomed Sci. 2000;7:444–458. doi: 10.1007/BF02253360. [DOI] [PubMed] [Google Scholar]
- 39.Reiter RJ, Tan DX, Poeggeler B, Menendez-Pelaez A, Chen LD, Saarela S. Melatonin as a free radical scavenger: implications for aging and age-related diseases. Ann N Y Acad Sci. 1994;719:1–12. doi: 10.1111/j.1749-6632.1994.tb56817.x. [DOI] [PubMed] [Google Scholar]


