Abstract
Background
The effect of methyl jasmonate (MJ) on ginsenoside production in different organs of ginseng (Panax ginseng Meyer) was evaluated after the whole plant was dipped in an MJ-containing solution. MJ can induce the production of antioxidant defense genes and secondary metabolites in plants. In ginseng, MJ treatment in adventitious root resulted in the increase of dammarenediol synthase expression but a decrease of cycloartenol synthase expression, thereby enhancing ginsenoside biosynthesis. Although a previous study focused on the application of MJ to affect ginsenoside production in adventitious roots, we conducted our research on entire plants by evaluating the effect of exogenous MJ on ginsenoside production with the aim of obtaining new approaches to study ginsenoside biosynthesis response to MJ in vivo.
Methods
Different parts of MJ-treated ginseng plants were analyzed for ginsenoside contents (fine root, root body, epidermis, rhizome, stem, and leaf) by high-performance liquid chromatography.
Results
The total ginsenoside content of the ginseng root significantly increased after 2 d of MJ treatment compared with the control not subjected to MJ. Our results revealed that MJ treatment enhances ginsenoside production not in the epidermis but in the stele of the ginseng root, implying transportation of ginsenosides from the root vasculature to the epidermis. Application of MJ enhanced protopanaxadiol (PPD)-type ginsenosides, whereas chilling treatment induced protopanaxatriol (PPT)-type ginsenosides.
Conclusion
These findings indicate that the production of PPD-type and PPT-type ginsenosides is differently affected by abiotic and biotic stresses in the ginseng plant, and they might play different defense mechanism roles.
Keywords: chilling, elicitor, ginsenoside, methyl jasmonate, Panax ginseng
1. Introduction
Panax ginseng Meyer, which is commonly known as Korean ginseng, is one of the most important traditional medicines in East Asia. Triterpene glycoside saponin, named ginsenoside, is the main bioactive ingredient in P. ginseng and is known to exhibit various pharmacological and physiological effects including anticancer [1–3], antidiabetic [4,5], immunomodulatory [1,6], neuroprotective [1], radioprotective [7], antiamnestic [1], and antistress properties [8,9]. The natural role of saponins in plants has been suggested to play a defensive role against pathogen and pest attacks [10]. The most important physiological role of ginsenosides in the ginseng plant is part of the defense mechanisms from pathogen attacks [11]. Naturally occurring ginsenosides are present to protect the ginseng from microbial and fungal infection; the bitter taste of ginsenosides makes them antifeedants [12–16].
Ginsenoside is contained in ginseng root at >4% by dry weight [17]. Ginsenosides are classified into two groups by the skeleton of aglycones, namely dammarane type and oleanane type. Dammarane-type tetracyclic structure is unique in ginseng, although other oleanane-type triterpenes are also observed in other plants. Dammarane-type ginsenosides consist mainly of two types that are classified according to their aglycone moieties, protopanaxadiol (PPD) and protopanaxatriol (PPT) ginsenoside.
Ginsenoside backbones are synthesized via the isoprenoid pathway by cyclization of 2,3-oxidosqualene mediated by dammarenediol synthase (DDS) or β-amyrin synthase (β-AS). Although many reports have been published regarding the pharmacological effects of ginsenosides, little is known about the ginsenoside biosynthesis pathway or its regulation. Complete cDNA clones for several enzymes from ginseng have been reported. The genes encoding squalene synthase (SS), squalene epoxidase (SE), β-AS, lanosterol synthase, cycloartenol synthase (CAS), and DDS have been identified. Metabolic engineering such as overexpression or gene silencing of those genes has altered ginsenoside levels. Upregulation of ginsenoside levels by elicitors is also an attractive strategy to achieve greater ginsenoside quantities [18].
The accumulation of secondary metabolites can be enhanced by exposing plant cell and tissue cultures to biotic and abiotic elicitors [19]. When plants perceive environmental changes, they generate biological responses through specific signal transduction. Methyl jasmonate (MJ) has been reported to play an important role in the production of antioxidant defense genes and secondary metabolites in plants [20–22]. It has been reported that MJ stimulates ginsenoside production in cultured ginseng cells, hairy root, and adventitious roots [23–26].
MJ also increases the production of soyasaponin in cultured Glycyrrhiza glabra cells [27] and saikosaponin in the adventitious roots of Bupleurum falcatum [28]. The stimulation of saponin production by MJ treatment may be mediated by the upregulation of the genes involved in the biosynthesis of these saponins.
Elicitation using MJ treatment has been conducted on ginseng hairy roots and adventitious roots. Treatment of in vitro cultures with MJ may increase the production of ginsenosides up to ninefold [29]. However, no elicitation studies with MJ have been done with the entire P. ginseng plant. Although ginseng root is usually used for medicinal purposes, ginsenosides are distributed in many parts of the ginseng plant, including the root, leaf, and berry. Different parts of the plant contain distinct ginsenoside profiles [2], which may exhibit different pharmacological activities. We conducted our research on whole 3-yr-old ginseng plants. The aim of the present study was to investigate which organs of the ginseng plant respond to elicitor treatment in vivo, thereby potentially enhancing ginsenoside production.
2. Materials and methods
2.1. Ginseng materials and treatment
Three-yr-old ginseng plants hydroponically cultured in perlite and peat moss at 23 ± 2°C under white fluorescent light (60–100 μmol/m2/s) in a controlled greenhouse (kindly provided by i-farm in Yeo-Ju, Korea) were used for whole plant treatment. Ginseng roots were dipped in water containing 50μM MJ and were maintained in the dark. After 2 d, fine root, root body (the inner part including xylem and pith), epidermis (the outer surface including cortex), rhizome, stem, and leaf parts were separately used for ginsenoside analysis. For chilling treatment, 1-yr-old ginseng roots were kept at 4°C for 4 wk. For ginsenoside analysis, rhizome, epidermis, upper and lower root body, and fine root parts were sampled separately.
2.2. Analysis of ginsenosides by high-performance liquid chromatography
Milled powder (0.3–1 g) of freeze-dried adventitious roots, leaves, and roots of ginseng were twice soaked in an 80% (v/v) methanol solution at 70°C for 1 h. The extract was filtered and then evaporated to remove the liquid. The residue was dissolved in distilled water followed by extraction with water-saturated n-butanol. The butanol layer was then evaporated to produce the saponin fraction. Each sample was dissolved in methanol (1 g/5 mL), filtered using a 0.45-μm filter, and then used for high-performance liquid chromatography (HPLC) analysis. The HPLC separation was carried out on an Agilent 1260 series HPLC system (Palo Alto, CA, USA). This experiment employed a C18 (250 mm × 4.6 mm, ID 5 μm) column using distilled water (Solvent A) and acetonitrile (Solvent B) mobile phases, with a flow rate of 1.6 mL/min and the following gradient: A/B ratios of 80.5:19.5 for 0–29 min, 70:30 for 29–36 min, 68:32 for 36–45 min, 66:34 for 45–47 min, 64.5:35.5 for 47–49 min, 0:100 for 49–61 min, and 80.5:19.5 for 61–66 min. The sample was detected at a wavelength of 203 nm. Quantitative analysis was performed via a one-point curve method using external standards of authentic ginsenosides.
2.3. Statistical analyses
The values of the ginsenoside contents and relative gene expression were expressed as mean ± standard deviation (SD). Statistical analyses were carried out using the GraphPad Prism software (GraphPad, San Diego, CA, USA) by one-way analysis of variance (ANOVA). Duncan's multiple range test was employed to test for significant differences between the treatments at p < 0.05 and p < 0.01.
3. Results
3.1. Response of different ginseng tissues to MJ treatment
The total ginsenoside contents in each tissue of the entire ginseng plant were analyzed. Cultivation of ginseng by hydroponics involves a shorter cultivation period in a greenhouse in which variables such as light, temperature, moisture, and carbon dioxide content can be controlled [30,31]. Therefore, we used hydroponically cultured 3-yr-old ginseng plants (Fig. 1). Fig. 2 shows that ginsenoside accumulations within the aerial parts (leaf and stem) were increased as compared with the control. Total ginsenoside contents in the leaf were higher than other tissues. In addition, total ginsenoside contents within the underground parts (rhizome, root body, epidermis, and fine root) were also increased, except in the epidermis. Total ginsenoside contents of the root body in MJ-treated plants increased by approximately twofold compared with that of the control. This result demonstrates that the increase in ginsenoside contents of the root body is the highest among all tested ginseng organs. In rhizome, total ginsenoside accumulation and its composition was significantly increased after MJ treatment. Total ginsenoside content of fine roots was increased by approximately 6 mg/g compared with the control, which is the most increased content observed in underground parts. In the epidermis, total ginsenoside content was only minimally influenced by MJ treatment.
Fig. 1.

Photographs of different organs of a 3-yr-old ginseng plant used for ginsenoside analysis. Three-yr-old ginseng plants hydroponically cultured in perlite and peat moss were used for methyl jasmonate (MJ) treatment. For ginsenoside analysis, different organs were sampled separately: the (A) leaf, stem, (B) rhizome, and fine root were sampled. The main root was divided again into the epidermis (the outer surface including the cortex) and (C) the root body (the inner part including the xylem and pith) by peeling. (D) A horizontal close-up image of the main root. All bars indicate 1 cm.
Fig. 2.
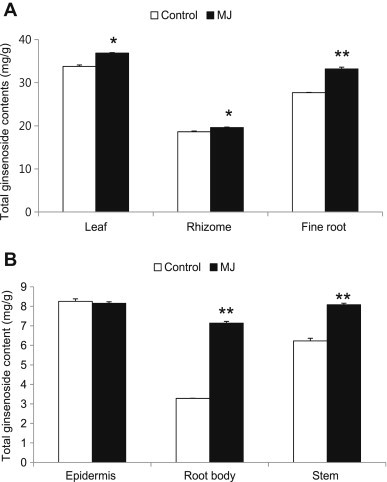
Effects of methyl jasmonate (MJ) on ginsenoside production in different ginseng plant tissues. Three-yr-old ginseng plants cultured hydroponically in perlite and peat moss were treated with 50μM of MJ by dipping the root in a foil-wrapped glass vial for 2 d. Total ginsenoside contents in (A) the leaf, rhizome, and fine root, as well as (B) the epidermis, root body, and stem, shown in Fig. 1, were analyzed. Vertical bars indicate the mean value ± standard deviation from three independent experiments. *p < 0.05 and **p < 0.01 denote significant difference from the control, respectively.
Fig. 3 shows the accumulation of individual ginsenosides in different tissues. The content of ginsenoside Re in aerial parts (leaf and stem) of the ginseng plant was the highest. In leaf, ginsenoside Re and Rd contents were mainly enhanced. The ratio between PPD-type and PPT-type ginsenosides was significantly changed in the stem. The content of ginsenoside Rd was increased more than other ginsenosides; therefore, the ratio of PPD-type ginsenoside was increased. In rhizome, the ratio of PPD-type ginsenoside was also increased due to accumulated ginsenoside Rd, although the content of ginsenoside Rg1 in the rhizome was the highest. The greatest increase of ginsenoside level was shown in the root body. All individual ginsenoside contents were increased. Levels of ginsenosides Rb1 and Rg1 were doubled as compared with the control. Although the content of ginsenoside Rg1 was the highest, ginsenoside Rd was enhanced fivefold. In addition, ginsenoside Rc and Rb2, which was not detected in the control, accumulated after MJ treatment, showing in the increased ratio of PPD-type ginsenoside. In fine root, all individual ginsenosides were also increased. Fine roots contained mostly ginsenoside Re, but the ratio of ginsenoside Rb1 was enhanced upon MJ treatment. Therefore, the ratio of PPD-type ginsenoside was increased to greater than that of the PPT-type ginsenoside. In the epidermis, all individual ginsenoside accumulation was similar to the control.
Fig. 3.
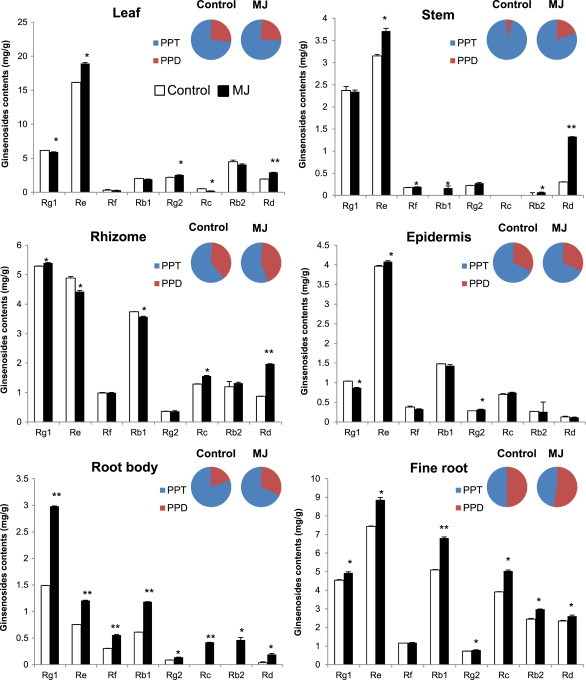
Individual ginsenoside contents by methyl jasmonate (MJ) elicitation in different ginseng plant tissues. Three-yr-old ginseng plants were treated with 50μM of MJ for 2 d. Individual ginsenoside contents in the leaf, stem, rhizome, epidermis, root body, and fine root were analyzed. Protopanaxadiol (PPD) indicates the sum of ginsenoside Rb1, Rb2, Rc, and Rd, and protopanaxatriol (PPT) indicates the sum of ginsenoside Re, Rg, Rg1, and Rg2. Vertical bars indicate the mean value ± standard deviation from three independent experiments. *p < 0.05 and **p < 0.01 denote significant difference from the control, respectively.
3.2. Response of different ginseng tissues to chilling treatment
As a representative treatment for abiotic stress, we treated chilling stress to root and analyzed ginsenoside contents. Total ginsenoside contents of chilled ginseng were analyzed in different organs from 1-yr-old root (rhizome, epidermis, upper root body, lower root body, and fine root; Fig. 4). As shown in Fig. 4, total ginsenoside levels of all tissues were increased. In particular, total ginsenoside contents of the lower root body after removing the epidermis increased approximately eightfold as compared with the control. Total ginsenoside contents of the lower root body showed the highest increased level of approximately 14 mg/g compared with the control (Fig. 4C). Total ginsenoside contents of the upper root body after removing the epidermis were increased threefold as compared with the control. The ratio of ginsenoside accumulation in the upper root body to lower root body in the control was 1:2. After chilling treatment, this ratio was changed to 1:5. In addition, the ratio of ginsenoside accumulation in the lower root body to fine root in the control was 1:13. After chilling treatment, a 1:2 ratio was noted.
Fig. 4.
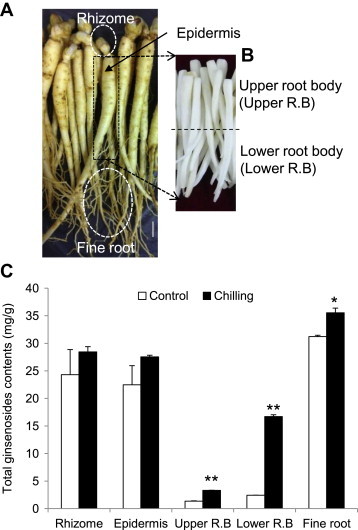
Effect of chilling on ginsenoside production in different ginseng plant tissues. (A) Photograph of different organs of a 1-yr-old ginseng root used for ginsenoside analysis. For ginsenoside analysis, different tissues were sampled separately; the rhizome and fine root were sampled. (B) After the epidermis was collected by peeling, the main root body was divided again into upper and lower halves. All bars indicate 1 cm. (C) Total ginsenoside contents in rhizome, epidermis, upper root body, lower root body, and fine root, shown in (A) and (B), were analyzed. Vertical bars indicate the mean value ± standard deviation from three independent experiments. *p < 0.05 and **p < 0.01 denote significant difference from the control, respectively.
We also analyzed the contents of individual ginsenosides in different organs upon chilling treatment (Fig. 5). In the epidermis, the contents of ginsenosides Re, Rb1, and Rg2 were significantly increased after chilling. By contrast, ginsenoside Rg1 was decreased in root rhizome and epidermis. Ginsenoside Re increased to the highest level in the epidermis as compared with the control (∼6.6 mg/g) and was not significantly increased in rhizome (Fig. 5). In fine root, ginsenoside Re content was increased, whereas ginsenoside Rg1 content was essentially unchanged, and ginsenosides Rb1, Rc, and Rb2 were reduced. Ginsenoside Re content was increased to the highest ratio and level compared with the control in fine root. The upper and lower roots of the body both showed increased ginsenoside accumulation of most ginsenosides. In the upper root, ginsenosides Rc and Rb2 were not detected but were present after chilling treatment. Ginsenoside Rd content significantly increased by approximately 14-fold. All individual ginsenosides in the lower root body highly increased after chilling treatment (Fig. 5). Ginsenosides Re, Rb2, and Rd were dramatically enhanced. The ratio of ginsenosides Re and Rb2 was increased more than 20-fold. The ratio of PPT-type ginsenosides was increased in all tissues except the rhizome, which showed a static level.
Fig. 5.
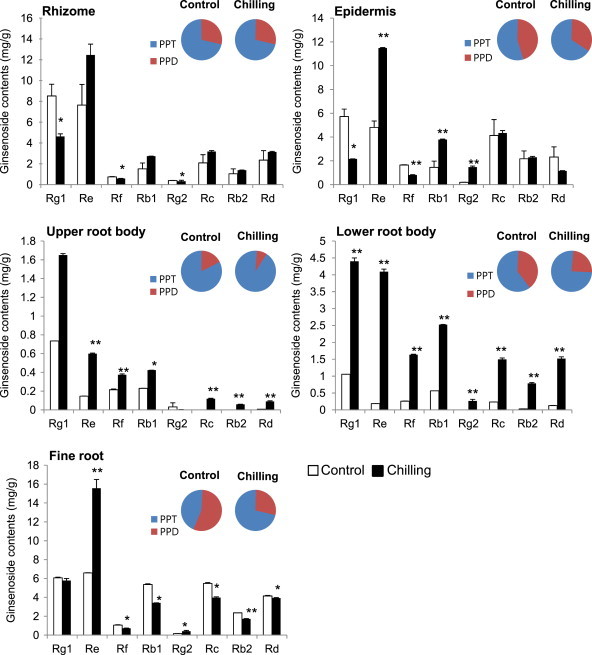
Individual ginsenoside contents upon chilling stress treatment in different ginseng plant tissues. One-yr-old ginseng plants were subjected to chilling for 4 wk. Individual ginsenoside contents in rhizome, epidermis, upper root body, lower root body, and fine root, as shown in Fig. 4, were analyzed. Protopanaxadiol (PPD) indicates the sum of ginsenoside Rb1, Rb2, Rc, and Rd, and protopanaxatriol (PPT) indicates the sum of ginsenoside Re, Rg, Rg1, and Rg2. Vertical bars indicate the mean value ± standard deviation from three independent experiments. *p < 0.05 and **p < 0.01 denote significant difference from the control, respectively.
4. Discussion
The roots of P. ginseng are used as important components of traditional oriental medicine [32], and the ginsenoside content increases with plant age [33]. Therefore, knowing where the ginsenosides localize in the ginseng root is important. Ginsenoside was reported to be at a higher content in the epidermis than in the cortex and xylem of the ginseng root [34]. Histochemical studies showed that ginsenosides are located in the periderm and outer cortex regions of the root, and root hairs contain higher ginsenoside contents than main roots [35,36]. Panax quinquefolius ginsenosides are also mostly detected in the periderm and cortex of the root [37]. Recently, multicenter matrix-assisted laser desorption/ionization mass spectrometry imaging confirmed that ginsenosides were more highly concentrated in the cortex and the periderm than that in the medulla of a lateral root, and localization of ginsenosides in the root tip is higher than that in the pith of the root [38]. In addition, a quantitative difference was detected between localizations of PPD-type ginsenosides (Rb1, Rb2, or Rc) and the PPT-type ginsenoside (Rf) in the root [38]. As saponins are known to be distributed to the root epidermis [34], we confirmed the accumulation of ginsenosides in the epidermis rather than the root body (J. Y. Oh et al, unpublished). However, our data in other work showed that ginsenoside biosynthesis genes are expressed in the root vasculature, such as phloem [18,39]. This controversial distribution and biosynthesis information led us to hypothesize that ginsenosides are produced in the root vasculature and then transported to the epidermis for a defensive role. To confirm this hypothesis, we selected MJ, known as a strong effective elicitor, to stimulate the biosynthesis of ginsenoside in vivo.
To improve the metabolite contents, some elicitors have been used to increase the expression and activities of key enzymes in the rate-limiting step of the biosynthetic pathway. MJ is a key signaling compound involved in the elicitation process, which leads to the accumulation of secondary metabolites [40]. Because ginsenosides are secondary metabolites in ginseng, the accumulation of these compounds is also controlled by the treatment of elicitors such as MJ and salicylic acid [41,42]. The total ginsenoside content increases approximately fourfold following MJ treatment in suspension cultured adventitious roots [6]. Depending on the timing of MJ application, the adventitious roots appear to show different growth effects and ginsenoside production. It was shown from previous reports that the addition of MJ at the early phase of P. ginseng growth inhibits adventitious root growth [43]. Jasmonic acid (JA) also strongly inhibited ginseng hairy root growth. [23]. To prevent a reduced biomass of adventitious roots, mostly 10μM of MJ was used in adventitious root cultures of P. ginseng 4 wk after inoculation. Higher MJ concentration and extended cultivation time also showed effects on root growth [43,44].
According to previous studies, this elicitation effect of ginsenosides is attributable to an MJ-induced expression of ginsenoside biosynthetic genes [6,29]. In our experiment using ginseng adventitious root, the expression of PgHMGR1, which plays a role in the production of mevalonic acid (MVA), was increased threefold (J. Y. Oh et al, unpublished). The expression of PgDDS, which is involved in the dammarenediol backbone for ginsenoside, was strongly upregulated, whereas PgCAS was decreased (data not shown). This expression pattern is similar to a previous report wherein MJ induced changes of triterpene saponins in ginseng hairy root [29]. Exposure to MJ at 100μM in hairy roots of P. ginseng induced the expression of genes involved in ginsenoside biosynthesis, such as PgSS, PgSE, and PgDDS, with a slight decrease of PgCAS [29,45], suggesting that MJ as a signal transducer may stimulate ginsenoside production by activation of the enzymes in the MVA pathway to dammarenediol and may also inactivate enzymes for sterol production.
Our present and previous results confirmed MJ as an effective elicitor of ginsenoside synthesis in ginseng adventitious roots. However, until now, it was not clear if MJ had the same effect on the whole ginseng plant. In this study, we tested the effect of MJ as an elicitor of ginsenoside accumulation in whole ginseng plants. When we analyzed the ginsenoside contents of the whole ginseng plant after exposing the ginseng root to MJ for 2 d, a pronounced increase of the ginsenosides was observed in the leaf, stem, root body, and fine root, with the greatest increase noted in the root body. An interesting observation was that most accumulation was observed in the root body, not the epidermis, which is known to have a high ginsenoside content. Rather, the epidermis did not show any alteration, indicating that ginsenoside biosynthesis actively occurs in the root body. After production in the root cortex, ginsenosides may be transported to the epidermis to play a defensive role. Ginsenosides can be synthesized in vasculature tissue such as phloem [46] and then be transported for storage or play a defensive role. Saponin glycosides can be stored in vacuoles through the fusion of endoplasmic reticulum-derived vesicles [47] or transported by the ATP-binding cassette transporter [48] or multidrug and toxic compound extrusion transporters [49,50]. Further studies on the ginsenoside transporter will provide more detail regarding ginsenoside transport.
Upon exposure of hairy roots or roots of P. ginseng to MJ, both tissues showed increased PPD-type ginsenoside content, whereas PPT-type ginsenoside content changed only slightly compared with controls [6,29]. JA also improved the accumulation of PPD ginsenosides much more than PPT ginsenosides [23], indicating that JA and its MJ elicitor might have triggered the synthesis of PPD-type ginsenosides. Similarly, different tissues in our experiment showed more accumulation of PPD-type ginsenosides, especially in the stem, root body, and fine root (Fig. 3). The recent discovery of protopanaxadiol synthase (PPDS), a cytochrome P450 (P450) for production of PPD, confirmed its induction by MJ treatment [51]. However another P450 which is involved in PPT production from PPD, named protopanaxatriol synthase (PPTS), does not respond to MJ treatment [52]. This suggests that MJ more highly stimulates the pathway leading to PPD ginsenoside synthesis than PPT ginsenoside synthesis. Although the biosynthetic pathway of dammarenediol into different ginsenosides has yet to be determined, further studies for identification of glycosyl transferases, enzymes in biosynthetic steps from PPD or PPT to different individual ginsenosides, will elucidate the different synthesis mechanisms of individual ginsenosides.
Overall, PPT-type ginsenoside accumulation was enhanced by chilling treatment (Fig. 5). When we performed chilling treatment as an abiotic stress, PPT-type ginsenosides were increased in the epidermis, upper and lower root body, and fine root, differently than with MJ treatment. This implies that ginseng roots are capable of differentially activating distinct defense pathways. The production of various secondary metabolites, including ginsenosides, is usually associated with defense responses to stresses [53]. JA and its methyl ester MJ are signaling compounds that modulate various physiological processes in plants, such as root growth, senescence, and the defense response against pathogens and insect attack [54]. In addition, MJ induces or increases the biosynthesis of many secondary metabolites that play important roles in the adaptation of plants to particular environments [21,55]. MJ treatment actually mimics a pathogen or herbivorous attack [56]. Saponins also have potent antifungal activities, as has been shown for avenacins, which are oleanane-type triterpene saponins found in oats [24]. However, although there is less known about the physiological role of ginsenoside, it is also considered to have a defensive role [12]. Different elicitation effects of MJ and chilling on individual ginsenosides might be explained by the differentiated defense strategy of ginsenosides (Fig. 6), correlated with the different biological activities of different ginsenosides.
Fig. 6.
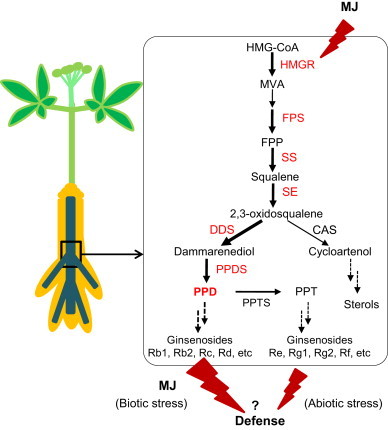
Summary of ginsenoside biosynthesis in ginseng root by elicitation. Ginsenoside production is stimulated via mevalonate (MVA) pathway genes, which are upregulated by MJ signaling. MJ elicitation enhanced PPD-type ginsenosides, whereas chilling stress increased PPT-type ginsenosides. CAS, cycloartenol synthase; DDS, dammarenediol synthase; FPP, farnesyl diphosphate; FPS, farnesyl diphosphate synthase; HMGR, 3-hydroxy-3-methylglutaryl-CoA reductase; MJ, methyl jasmonate; MVA, mevalonic acid; PPD, protopanaxadiol; PPDS, PPD synthase (cytochrome P450); PPT, protopanaxatriol; PPTS, PPT synthase (cytochrome P450); SE, squalene epoxidase; SS, squalene synthase.
To the best of our knowledge, this study is the first to report on the evaluation of individual ginsenoside levels in separate epidermis and root body. Moreover, this is the first study to provide a detailed profile of ginsenoside composition in different organs of whole ginseng plants following elicitor treatment, although further studies of gene expression in different tissues of ginseng will be required.
Conflict of interest
All contributing authors declare no conflicts of interest.
Acknowledgments
This research was supported by iPET (312064-03-1-HD040), Korea Institute of Planning and Evaluation for Technology in Food, Agriculture, Forestry, and Fisheries, Republic of Korea.
Footnotes
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
References
- 1.Attele A.S., Wu J.A., Yuan C.S. Ginseng pharmacology: multiple constituents and multiple actions. Biochem Pharmacol. 1999;58:1685–1693. doi: 10.1016/s0006-2952(99)00212-9. [DOI] [PubMed] [Google Scholar]
- 2.Im K.S., Chung H.Y., Park S.H., Je N.K. Anticancer effect of the hydrolyzed monogluco-ginsenoside of total saponin from ginseng leaf. J Ginseng Sci. 1995;19:291–294. [Google Scholar]
- 3.Li X., Wan H.G., Lu D.Q., Wei P. Advance of research on antitumour activity of ginsenosides. Chin J Bioproc Eng. 2003;1:13–17. [Google Scholar]
- 4.Xie J.T., Mehendale S.R., Li X., Quigg R., Wang X., Wang C.Z., Wu J.A., Aung H.H., Rue P.A., Bell G.I. Anti-diabetic effect of ginsenoside Re in ob/ob mice. Biochem Biophys Acta. 2005;1740:319–325. doi: 10.1016/j.bbadis.2004.10.010. [DOI] [PubMed] [Google Scholar]
- 5.Shang W., Yang Y., Zhou L., Jiang B., Jin H., Chen M. Ginsenoside Rb1 stimulates glucose uptake through insulin-like signaling pathway in 3T3-L1 adipocytes. J Endocrinol. 2008;198:561–569. doi: 10.1677/JOE-08-0104. [DOI] [PubMed] [Google Scholar]
- 6.Wang J., Gao W.Y., Zhang J., Zuo B.M., Zhang L.M., Huang L.Z. Advances in study of ginsenoside biosynthesis pathway in Panax ginseng C. A. Meyer. Acta Physiol Plant. 2012;34:397–403. [Google Scholar]
- 7.Lee T.K., Johnke R.M., Allison R.R., O'Brien K.F., Dobbs L.J. Radio protective potential of ginseng. Mutagenesis. 2005;20:237–243. doi: 10.1093/mutage/gei041. [DOI] [PubMed] [Google Scholar]
- 8.Saito H., Yoshida Y., Takagi K. Effect of Panax ginseng root on exhaustive exercise in mice. Jpn J Pharmacol. 1974;24:119–127. doi: 10.1254/jjp.24.119. [DOI] [PubMed] [Google Scholar]
- 9.Gillis C.N. Panax ginseng pharmacology: a nitric oxide link? Biochem Pharmacol. 1997;54:1–8. doi: 10.1016/s0006-2952(97)00193-7. [DOI] [PubMed] [Google Scholar]
- 10.Osbourn A.E. Preformed antimicrobial compounds and plant defense against fungal attack. Plant Cell. 1996;8:1821–1831. doi: 10.1105/tpc.8.10.1821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Leung K.W., Wong A.S. Pharmacology of ginsenosides: a literature review. Chin Med. 2010;5 doi: 10.1186/1749-8546-5-20. article 20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Nicol R.W., Traquair J.A., Bernards M.A. Ginsenosides as host resistance factors in American ginseng (Panax quinquefolius) Can J Bot. 2002;80:557–562. [Google Scholar]
- 13.Katerere D.R., Gray A.I., Nash R.J., Waigh R.D. Antimicrobial activity of pentacyclic triterpenes isolated from African Combretaceae. Phytochemistry. 2003;63:81–88. doi: 10.1016/s0031-9422(02)00726-4. [DOI] [PubMed] [Google Scholar]
- 14.Mallvadhani U.V., Mahapatra A., Raja S.S., Manjula C. Antifeedant activity of some pentacyclic triterpene acids and their fatty acid ester analogues. J Agric Food Chem. 2003;51:1952–1955. doi: 10.1021/jf020691d. [DOI] [PubMed] [Google Scholar]
- 15.Bernards M.A., Yousef L.F., Nicol R.W. The allelopathic potential of ginsenosides. In: Inderjit, Mukerji K.G., editors. Allelochemicals: biological control of plant pathogens and diseases. Springer; Dordrecht: 2006. pp. 157–175. [Google Scholar]
- 16.Sung W.S., Lee D.G. In vitro candidacidal action of Korean red ginseng saponins against Candida albicans. Biol Pharm Bull. 2008;31:139–143. doi: 10.1248/bpb.31.139. [DOI] [PubMed] [Google Scholar]
- 17.Shibata S. Chemistry and cancer preventing activities of ginseng saponins and some related triterpenoid compounds. Kor J Med Crop Sci. 2001;16:28–37. doi: 10.3346/jkms.2001.16.S.S28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Han J.Y., In J.K., Kwon Y.S., Choi Y.E. Regulation of ginsenoside and phytosterol biosynthesis by RNA interferences of squalene epoxidase gene in Panax ginseng. Phytochemistry. 2010;71:36–46. doi: 10.1016/j.phytochem.2009.09.031. [DOI] [PubMed] [Google Scholar]
- 19.Gundlach H., Muller M.J., Kutchan T.M., Zenk M.H. Jasmonic acid is a signal transducer in elicitor-induced plant cell cultures. Proc Natl Acad Sci USA. 1992;89:2389–2393. doi: 10.1073/pnas.89.6.2389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Hampel D., Mosandl A., Wust M. Induction of de novo volatile terpene biosynthesis via cytosolic and plastidial pathways by methyl jasmonate in foliage of Vitis vinifera L. J Agric Food Chem. 2005;53:2652–2657. doi: 10.1021/jf040421q. [DOI] [PubMed] [Google Scholar]
- 21.Martin D.M., Gershenzon J., Bohlmann J. Induction of volatile terpene biosynthesis and diurnal emission by methyl jasmonate in foliage of Norway spruce. Plant Physiol. 2003;132:1586–1599. doi: 10.1104/pp.103.021196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Farmer E.E., Ryan C.A. Interplant communication: airborne methyl jasmonate induces synthesis of proteinase inhibitors in plant leaves. Proc Natl Acad Sci USA. 1990;87:7713–7716. doi: 10.1073/pnas.87.19.7713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Yu K.W., Gao W.Y., Son S.H., Paek K.Y. Improvement of ginsenoside production by jasmonic acid and some other elicitors in hairy root culture of ginseng (Panax ginseng C. A. Meyer) In Vitro Cell Dev Biol. 2000;36:424–428. [Google Scholar]
- 24.Hu W.W., Yao H., Zhong J.J. Improvement of Panax notoginseng cells for production of ginseng saponin and polysaccharide by high cell density cultivation in pneumatically agitated bioreactors. Biotechnol Prog. 2001;17:838–846. doi: 10.1021/bp010085n. [DOI] [PubMed] [Google Scholar]
- 25.Han J., Zhong J.J. High density cell culture of Panax notoginseng for production of ginseng saponin and polysaccharide in an airlift bioreactor. Biotechnol Lett. 2002;24:1927–1930. [Google Scholar]
- 26.Wang W., Zhang Z.Y., Zhong J.J. Enhancement of ginsenoside biosynthesis in high-density cultivation of Panax notoginseng cells by various strategies of methyl jasmonate elicitation. Appl Microbiol Biotechnol. 2005;67:752–758. doi: 10.1007/s00253-004-1831-z. [DOI] [PubMed] [Google Scholar]
- 27.Hayashi H., Hung P., Inoue K. Up-regulation of soyasaponin biosynthesis by methyl jasmonate in cultured cells of Glycyrrhiza glabra. Plant Cell Physiol. 2003;44:404–411. doi: 10.1093/pcp/pcg054. [DOI] [PubMed] [Google Scholar]
- 28.Aoyagi H., Kobayashi Y., Yamada K., Yokoyama M., Kusakari K., Tanaka H. Efficient production of saikosaponins in Bupleurum falcatum root fragments combined with signal transducers. Appl Microbiol Biotechnol. 2001;57:482–488. doi: 10.1007/s002530100819. [DOI] [PubMed] [Google Scholar]
- 29.Kim O.T., Bang K.H., Kim Y.C., Hyun D.Y., Kim M.Y., Cha S.W. Upregulation of ginsenoside and gene expression related to triterpene biosynthesis in ginseng hairy root cultures elicited by methyl jasmonate. Plant Cell Tiss Organ Cult. 2009;98:25–33. [Google Scholar]
- 30.Kim G.S., Lee S.E., Noh H.J., Kwon H., Lee S.W., Kim S.Y., Kim Y.B. Effects of natural bioactive products on the growth and ginsenoside contents of Panax ginseng cultured in an aeroponic system. J Ginseng Res. 2012;36:430–441. doi: 10.5142/jgr.2012.36.4.430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Lee G.A., Chang Y.K., Park S.Y., Kim G.A., Kim S.H., Park K.C., Kim Y.B., Cha S.W., Song B.H. Comparative analysis on concentration and uptake amount of mineral nutrients in different growth stages and temperatures of Panax ginseng C. A. Meyer grown with hydroponic culture. Kor J Med Crop Sci. 2012;20:251–258. [Google Scholar]
- 32.Lee O.R., Sathiyara G., Kim Y.J., In J.G., Kwon W.S., Kim J.H., Yang D.C. Defense genes induced by pathogens and abiotic stresses in Panax ginseng C. A. Meyer. J Ginseng Res. 2011;35:1–11. [Google Scholar]
- 33.Shi W., Wang Y., Li J., Zhang H., Ding L. Investigation of ginsenosides in different parts and ages of Panax ginseng. Food Chem. 2007;102:664–668. [Google Scholar]
- 34.Kim M.W., Ko S.R., Choi K.J., Kim S.C. Distribution of saponin in various sections of Panax ginseng root and changes of its contents according to root age. J Ginseng Res. 1987;11:10–16. [Google Scholar]
- 35.Tani T., Michinori K., Tadahisa K., Masayuki H., Teruaki H., Shigaru A., Histochemistry I.I. Ginsenoside in ginseng (Panax ginseng) root. J Nat Prod. 1981;44:401–407. [Google Scholar]
- 36.Christensen L.P., Jensen M., Kidmose U. Simultaneous determination of ginsenosides and polyacetylenes in American ginseng root (Panax quinquefolium L.) by high performance liquid chromatography. J Agric Food Chem. 2006;54:8995–9003. doi: 10.1021/jf062068p. [DOI] [PubMed] [Google Scholar]
- 37.Smith R.G., Caswell D., Carriere A., Zielke B. Variation in the ginsenoside content of American ginseng, Panax quinquefolius L., roots. Can J Bot. 1995;74:1616–1620. [Google Scholar]
- 38.Taira S., Ikeda R., Yokota N., Osaka I., Sakamoto M., Kato M., Sahashi Y. Mass spectrometric imaging of ginsenosides localization in Panax ginseng root. Am J Chin Med. 2010;38:485–493. doi: 10.1142/S0192415X10008007. [DOI] [PubMed] [Google Scholar]
- 39.Kim T.D., Han J.Y., Huh G.H., Choi Y.E. Expression and functional characterization of three squalene synthase genes associated with saponin biosynthesis in Panax ginseng. Plant Cell Physiol. 2011;52:125–137. doi: 10.1093/pcp/pcq179. [DOI] [PubMed] [Google Scholar]
- 40.Hu X., Neill S., Cai W., Tang Z. Hydrogen peroxide and jasmonic acid mediate oligogalacturonic acid-induced saponin accumulation in suspension-cultured cells of Panax ginseng. Physiol Plant. 2003;118:414–421. [Google Scholar]
- 41.Ali M.B., Yu K.W., Hahn E.J., Paek K.Y. Methyl jasmonate and salicylic acid elicitation induces ginsenosides accumulation, enzymatic and non-enzymatic antioxidant in suspension culture Panax ginseng roots in bioreactors. Plant Cell Rep. 2006;25:613–620. doi: 10.1007/s00299-005-0065-6. [DOI] [PubMed] [Google Scholar]
- 42.Wang J., Gao W., Zuo B., Zhang L., Huang L. Effect of methyl jasmonate on the ginsenoside content of Panax ginseng adventitious root cultures and on the genes involved in triterpene biosynthesis. Res Chem Interned. 2012;39:1973–1980. [Google Scholar]
- 43.Kim Y.S., Hahn E.J., Murthy H.N., Paek K.Y. Adventitious root growth and ginsenoside accumulation in Panax ginseng cultures as affected by methyl jasmonate. Biotechnol Lett. 2004;26:1619–1622. doi: 10.1007/s10529-004-3183-2. [DOI] [PubMed] [Google Scholar]
- 44.Yu K.W., Gao W., Han E.J., Paek K.Y. Jasmonic acid improves ginsenoside accumulation in adventitious root culture of Panax ginseng C.A. Meyer. Biochem Eng. 2002;11:211–215. [Google Scholar]
- 45.Lee M.H., Jeong J.H., Seo J.W., Shim C.G., Kim Y.S., In J.G., Yang D.C., Yi J.S., Choi Y.E. Enhanced triterpene and phytosterol biosynthesis in Panax ginseng overexpressing squalene synthase gene. Plant Cell Physiol. 2004;45:976–984. doi: 10.1093/pcp/pch126. [DOI] [PubMed] [Google Scholar]
- 46.Fukuda N., Shan S., Tanaka H., Shoyama Y. New staining methodology: Eastern blotting for glycosides in the field of Kampo medicines. J Nat Med. 2006;60:21–27. [Google Scholar]
- 47.Poustka F., Irani N.G., Feller A., Lu Y., Pourcel L., Frame K., Grotewalk E.A. Trafficking pathway for anthocyanins overlaps with the endoplasmic reticulum-to-vacuole protein sorting route in Arabidopsis and contributes to the formation of vacuolar inclusions. Plant Physiol. 2007;145:1323–1335. doi: 10.1104/pp.107.105064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Jasiński M., Stukkens Y., Degand H., Purnelle B., Marchand-Brynaert J., Boutry M. A plant plasma membrane ATP binding cassette-type transporter is involved in antifungal terpenoid secretion. Plant Cell. 2001;13:1095–1107. [PMC free article] [PubMed] [Google Scholar]
- 49.Goodman C.D., Casati P., Walbot Y.A. Multidrug resistance-associated protein involved in anthocyanin transport in Zea mays. Plant Cell. 2004;16:1812–1826. doi: 10.1105/tpc.022574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Yazaki K. Transporters of secondary metabolites. Curr Opin Plant Biol. 2005;8:301–307. doi: 10.1016/j.pbi.2005.03.011. [DOI] [PubMed] [Google Scholar]
- 51.Han J.Y., Kim H.J., Kwon Y.S., Choi Y.E. The cyt P450 enzyme CYP716A47 catalyzes the formation of protopanaxadiol from dammarenediol-II during ginsenoside biosynthesis in Panax ginseng. Plant Cell Physiol. 2011;52:2062–2073. doi: 10.1093/pcp/pcr150. [DOI] [PubMed] [Google Scholar]
- 52.Han J.Y., Hwang H.S., Choi S.W., Kim H.J., Choi Y.E. Cytochrome P450 CYP716A53v2 catalyzes the formation of protopanaxatriol from protopanaxadiol during ginsenoside biosynthesis in Panax ginseng. Plant Cell Physiol. 2012;53:1535–1545. doi: 10.1093/pcp/pcs106. [DOI] [PubMed] [Google Scholar]
- 53.Zhao J., Davis L.C., Verpoorte R. Elicitor signal transduction leading to production of plant secondary metabolites. Biotechnol Adv. 2005;23:283–333. doi: 10.1016/j.biotechadv.2005.01.003. [DOI] [PubMed] [Google Scholar]
- 54.Wasternack C., Parthier B. Jasmonate-signaled plant gene expression. Trends Plant Sci. 1997;2:302–307. [Google Scholar]
- 55.Yukimine Y., Tabata H., Higashi Y., Hara Y. Methyl jasmonate-induced overproduction of paclitaxel and baccation III in Taxus cell suspension cultures. Nat Biotechnol. 1996;14:1129–1132. doi: 10.1038/nbt0996-1129. [DOI] [PubMed] [Google Scholar]
- 56.Textor S., Gershenzon J. Herbivore induction of the glucosinolate-myrosinase defense system: major trends, biochemical bases and ecological significance. Phytochem Rev. 2009;8:149–170. [Google Scholar]


