Abstract
Background
Roles of immune reaction and toll-like receptor-4 (TLR-4) have widely been established in the pathogenesis of alcoholic liver disease (ALD).
Methods
We evaluated the biologic efficacy of Korean Red Ginseng (KRG), urushiol, and probiotics (Lactobacillus rhamnosus R0011 and Lactobacillus acidophilus R0052) in mouse models of ALD. Sixty C57BL/6 mice were equally divided into six feeding groups for 10 weeks: normal diet, alcohol, control, alcohol + KRG, alcohol + urushiol, and alcohol + probiotics. Alcohol was administered via a Lieber–DeCarli liquid diet containing 10% alcohol. TLR-4 expression, proinflammatory cytokines, and histology, as well as the results of liver function tests were evaluated and compared.
Results
No between-group differences were observed with regard to liver function. TLR-4 levels were significantly lower in the KRG, urushiol, and probiotics groups than in the alcohol group (0.37 ± 0.06 ng/mL, 0.39 ± 0.12 ng/mL, and 0.33 ± 0.07 ng/mL, respectively, vs. 0.88 ± 0.31 ng/mL; p < 0.05). Interleukin-1β levels in liver tissues were decreased among the probiotics and KRG groups. The tumor necrosis factor-α level of liver tissue was decreased in the KRG group.
Conclusion
The pathological findings showed that alcohol-induced steatosis was significantly reduced by KRG and urushiol. As these agents improve immunologic capacity, they may be considered in potential anti-ALD treatments.
Keywords: alcoholic liver disease, Lactobacillus, Panax ginseng, urushiol
1. Introduction
Alcoholic liver disease (ALD) is a leading cause of liver-related deaths worldwide. The chronic spectrum of alcohol-ingestion-related diseases includes steatosis, steatohepatitis, cirrhosis, and hepatocellular carcinoma [1–3]. Bacterial translocation due to disruption of the gut-barrier function by alcohol induces endotoxemia [4]. Activation of the toll-like receptor-4 (TLR-4)-mediated signaling pathway, proinflammatory cytokines, and the reactive oxygen species induced by endotoxins [lipopolysaccharide (LPS)] are important factors in the pathogenesis of ALD [5]. Although multiple medications have been proposed as potential therapeutic agents for patients with ALD, only abstinence and nutritional support have generally been employed as specific therapies [6].
Pro- and prebiotics have been proposed as potential therapeutic agents for the treatment of ALD and liver cirrhosis in animal and human studies [7,8]. Lacidofil is composed of Lactobacillus rhamnosus R0011 and Lactobacillus acidophilus R0052. According to an animal study, lactobacillus-treated mice with ALD showed improvement [9]. An in vitro study also demonstrated the anti-inflammatory effects of Lactobacillus, which downregulates cytokines [10].
Ginseng is an oriental herb that has been consumed for more than 2,000 years. Korean Red Ginseng (KRG) and its primary ginsenosides have potent protective effects with regard to alcohol-induced hepatocytic injury [11]. In addition, ginsenoside Re, a major constituent of ginseng, inhibits the expression of proinflammatory cytokines in LPS-stimulated peritoneal macrophages in mice [12]. Ginseng has extensively been reported to maintain homeostasis of the immune system, and enhance resistance to illness or microbial attacks through the regulation of immune system [13].
Urushiol is an allergic oil found in plants of the Anacardiaceae family. This oil is a major component of lacquer tree (Rhus vernicifera Stokes) sap [14]. Urushiol has been used as the traditional folk medicine in Korea and has anti-inflammatory, antimicrobial, and antioxidative effects in mice, according to in vitro studies [14,15]. However, no study has evaluated the effects of Lacidofil, KRG, or urushiol on the gut–liver axis in the context of ALD. The current study evaluated the biologic efficacy of Lacidofil, KRG, and urushiol in a mouse model of ALD.
2. Materials and methods
2.1. Chemicals
This study used and stored 20 mg of Lacidofil (a bacteria culture of L. rhamnosus R0011 and L. acidophilus R0052; Pharmbio Korea Co., Ltd, Chungbuk, Korea) at 5°C.
The Korean Society of Ginseng donated an undiluted solution of KRG. This KRG sample contained seven glycosides known as ginsenosides (mg/g): Rg1 (2.481), Rb1 (5.481), Rg3(s) (0.197), Re (2.975), Rc (2.248), Rb2 (2.175), and Rb (0.566). The extract had a moisture content of 36.68%.
The sap (40 mL) of a lacquer tree was diluted in 1 L of distilled water, which subsequently underwent double extraction with 1 L of n-hexane. The hexane extract was concentrated under reduced pressure, yielding an oil (26.9 g). The oil was then purified via silica gel column chromatography (Merck 7734) and eluted with 20% acetone/hexane. It was further purified using the same method (Merck 9385), followed by octadecyl silica gel column chromatography (YMC GEL ODS-A) with a gradient of methanol in water to yield urushiols. The final concentration of extracted urushiol was 10 mg/mL.
2.2. Animals
Age-matched 6-week-old male C57BL/6 mice (Dooyeol Biotech, Inc., Seoul, Korea) were used in all experiments. Only male mice were used, given the hormonal changes of female mice. The mean body weights of the mice in each group are listed in Table 1. A total of 60 male C57BL/6 mice were housed individually in steel microisolator cages maintained at 22°C with a 12-hour/12-hour light/dark cycle. The mice were randomly assigned to six dietary groups (n = 10). Each group of mice received one of the following six diets for 10 weeks: (1) standard chow diet (normal feed); (2) alcohol diet (a Lieber–DeCarli liquid diet with 10% alcohol); (3) control diet (a Lieber–DeCarli liquid diet with 10% alcohol and normal feed); (4) KRG diet (200 mg/kg/day of KRG with normal feed for 4 weeks after a Lieber–DeCarli liquid diet with 10% alcohol for 6 weeks); (5) urushiol diet (0.128 mg/mL/day of urushiol with normal feed for 4 weeks after a Lieber–DeCarli liquid diet with 10% alcohol for 6 weeks); and (6) probiotics diet (1 mg/mL/day of L. rhamnosus R0011 and L. acidophilus R0052 with normal feed for 4 weeks after a Lieber–DeCarli liquid diet with 10% alcohol for 6 weeks; Fig. 1). The liquid diets were based on the Lieber–DeCarli ethanol formulation and purchased from Dooyeol Biotech, Inc. Protein, fat, and carbohydrates constitute, respectively, 18.9%, 16.5%, and 64.5% of the calories of the Lieber–DeCarli liquid diet. Lacidofil, KRG, and urushiol suspended in distilled water were orally administered using a gastric tube five times a week, for 4 weeks.
Table 1.
Mean Body Weight (g) of Each Group
| Normal | Alcohol | Control | KRG | Urushiol | Probiotics |
|---|---|---|---|---|---|
| 25.6 ± 1.56 | 24.3 ± 1.5 | 27.1 ± 2.2 | 28.2 ± 2.9* | 30.4 ± 3.0* | 29.1 ± 3.4* |
*p < 0.05 versus alcohol group
KRG, Korean Red Ginseng
Fig. 1.
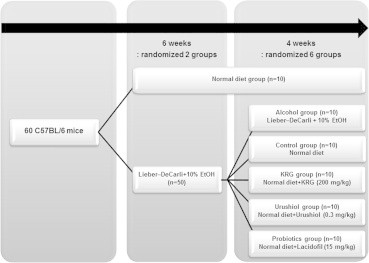
Study design. n, number; KRG, Korean Red Ginseng.
At the end of the treatment period, the animals were sacrificed via isoflurane inhalation. A midline abdominal incision was performed, and blood was collected through the orbital canal. Whole-blood (600 μL) samples were centrifuged at 1,500 × g for 15 minutes to collect the serum. The liver was rapidly excised and stored at −80°C.
2.3. Ethics statement
The animals received humane care, and all procedures were conducted in accordance with the National Institutes of Health Guidelines for the Care and Use of Laboratory Animals. The Institutional Animal Care and Use Committee of the Hallym University College of Medicine, Gangwon Do, Korea approved this study.
2.4. Liver function test
Levels of aspartate aminotransferase, alanine aminotransferase, and gamma-glutamyl transferase were analyzed with a biochemical blood analyzer (KoneLab 20, Thermo Fisher Scientific, Waltham, Finland).
2.5. Serum and liver tissue cytokines
After rinsing the tissue samples with a cell wash buffer once, they were cut into 3 mm × 3 mm pieces and transferred to a 2 mL tissue grinder. Subsequently, the liver tissue was homogenized with a cell lysis kit (Bio-Plex cell lysis kit; Bio-Rad Laboratories, Inc., Seoul, Korea) according to the manufacturer's recommendations. The tumor necrosis factor-α (TNF-α) and interleukin-1β (IL-1β) levels of the serum and liver tissue were analyzed with a biochemical analyzer [enzyme-linked immunosorbent assay (ELISA) kit for Bio-Plex Pro Mouse Cytokine Assay kit; Bio-Rad Laboratories, Inc., Seoul, Korea].
2.6. TLR-4 analysis (Western blot analysis)
The liver samples were homogenized in a complete Bio-Plex cell lysis kit (Bio-Rad Laboratories, Inc.). Protein was boiled in the loading buffer, both having the same concentration (50 mM Tris-HCl, pH 6.8, 2% sodium dodecyl sulfate, 12.5% glycerol, 125 mM dithiotheritol, and 0.05% bromophenol blue) for 5 minutes. The sample was then separated using sodium dodecyl sulfate-polyacrylamide gel electrophoresis, and transferred and immobilized on a nitrocellulose membrane. The membrane was blocked with 5% non-fat dry milk in a Tris-buffered saline containing 0.1% Tween 20 for 2 hours at room temperature under agitation. The membrane was washed six times in 0.1% Tween 20, followed by incubation under agitation with 1:2,000 goat polyclonal mouse serum albumin antibodies (HRP, 60R-AG002hrp; Fitzgerald Industries International, MA, USA) for 1 hour at room temperature. After the final wash, the membrane was reacted with the ECL substrate solution (Power-Opti ECL; Bionote, Inc., Gyeonggi-do, Korea) and exposed to a ChemiDoc XRS + system.
2.7. TLR-4 analysis (ELISA analysis)
After rinsing the tissue samples with the cell wash buffer once, they were cut into 3 mm × 3 mm pieces and transferred to a 2 mL tissue grinder. The liver tissue was then homogenized with a cell lysis kit (Bio-Plex cell lysis kit; Bio-Rad Laboratories, Inc.), according to the manufacturer's instructions. A biochemical analyzer assessed the TLR-4 levels in the serum and liver tissue (ELISA kit for TLR-4; USCN Life Science Inc., Wuhan, China). As per the manufacturer's instructions, absorbance (A) was detected at 450 nm (A450). The content of each sample was estimated using the standard curve.
2.8. Pathology
Specimens were fixed with 10% formalin and routinely embedded in paraffin; the tissue sections were processed with hematoxylin and eosin, Masson's trichrome, and reticulin fiber staining. Fatty liver was classified, based on the ALD clinical research network's scoring system for alcoholic fatty liver disease [16], from Grade 0 to Grade 3 (0: <5%; 1: 5–33%; 2: 34–66%; and 3: >66% of steatosis). All specimens underwent a blind analysis by the same hepatopathologist (S.H.H.).
2.9. Statistical analysis
Continuous variables were expressed as means and standard deviation. Analyses were performed with Prism 5.0 (GraphPad, San Diego, CA, USA). One-way analysis of variance, the Kruskal–Wallis test, Dunn's multiple comparison test, and Tukey's multiple comparison test were performed. A p value of <0.05 was considered significant.
3. Results
3.1. Body weight
The mean body weight of the alcohol group was significantly lower than that of the KRG, urushiol, and probiotics groups (24.3 ± 1.52 vs. 8.2 ± 2.9, 30.4 ± 3.0, and 29.1 ± 3.4, respectively; p < 0.05).
3.2. Liver function test
No statistical differences were observed, with regard to the liver function tests, between the normal, alcohol, control, KRG, urushiol, and probiotics groups (p > 0.05; Table 2). The following results were found (stated as the normal, control, probiotics, KRG, and urushiol groups vs. the alcohol group): aspartate aminotransferase (186.1 ± 60.1 U/L, 186.3 ± 79.8 U/L, 174.0 ± 45.6 U/L, 182.5 ± 55.8 U/L, and 164.3 ± 62.8 U/L, respectively, vs. 191.2 ± 57.0 U/L); alanine aminotransferase (30.7 ± 24.9 U/L, 33.1 ± 24.8 U/L, 41.1 ± 12.0 U/L, 41.2 ± 14.9 U/L, and 31.2 ± 4.8 U/L, respectively, vs. 35.3 ± 11.3 U/L); and gamma-glutamyl transferase (8.1 ± 4.1 U/L, 8.0 ± 5.9 U/L, 7.8 ± 4.6 U/L, 8.5 ± 3.0 U/L, and 9.2 ± 4.8 U/L, respectively, vs. 7.4 ± 3.9 U/L). Thus, treatment with probiotics, KRG, or urushiol did not ameliorate the results of the serum liver function test.
Table 2.
Effects of the KRG, Urushiol, and Probiotics on Liver Function of Mice
| Normal (n = 10) | Alcohol (n = 10) | Control (n = 10) | KRG (n = 10) | Urushiol (n = 10) | Probiotics (n = 10) | |
|---|---|---|---|---|---|---|
| AST1) | 186.1 ± 60.1 | 191.2 ± 57.0 | 186.3 ± 79.8 | 174.0 ± 45.6 | 182.5 ± 55.8 | 164.3 ± 62.8 |
| ALT1) | 30.7 ± 24.9 | 35.3 ± 11.3 | 33.1 ± 24.8 | 41.1 ± 12.0 | 41.2 ± 14.9 | 31.2 ± 4.8 |
| γ-GT1) | 8.1 ± 4.1 | 7.4 ± 3.9 | 8.0 ± 5.9 | 7.8 ± 4.6 | 8.5 ± 3.0 | 9.2 ± 4.8 |
ALT, alanine aminotransferase; AST, aspartate aminotransferase; γ-GT, gamma-glutamyl transferase; KRG, Korean Red Ginseng
U/L, mean ± standard deviation. Statistical differences were not observed with regard to the liver function tests among the normal, alcohol, control, KRG, urushiol, and probiotics groups (p > 0.05).
3.3. Cytokines
Serum cytokines, TNF-α, and IL-1β level analyses revealed that the probiotics, KRG, and urushiol groups did not differ from the alcohol group (p > 0.05). Although the serum TNF-α levels in the probiotics and urushiol groups, as well as the IL-1β levels in the urushiol group, were lower than those in the alcohol group, these results were not significant.
TNF-α level of the liver tissue in the KRG group was 379.9 ± 201.5 pg/mL, which was significantly lower than that in the alcohol group (687.4 ± 110.5 pg/mL; p < 0.05). IL-1β levels of the liver tissue in the probiotics (37.33 ± 18.48 pg/mL; p < 0.05) and KRG (26.18 ± 7.17 pg/mL; p < 0.01) groups were decreased compared with those in the alcohol group (65.21 ± 3.91 pg/mL; Fig. 2). KRG reduced proinflammatory cytokines.
Fig. 2.
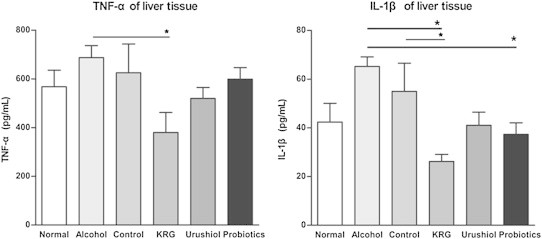
TNF-α of liver tissue in the KRG group is significantly lower than that in the alcohol group. IL-1β levels of liver tissue in the probiotics and KRG groups are decreased compared with that in the alcohol group. *p < 0.05. IL, interleukin; KRG, Korean Red Ginseng; TNF, tumor necrosis factor.
3.4. TLR-4 of liver tissue (Western blot analysis and ELISA analysis)
Western blot recognition of the protein in the liver tissue homogenate is summarized in Fig. 3. The Western blot analysis revealed positive bands of appropriate sizes for each protein studied. TLR-4 and GAPDH antibodies were detected as single bands at 95 kDa and 37 kDa, respectively.
Fig. 3.
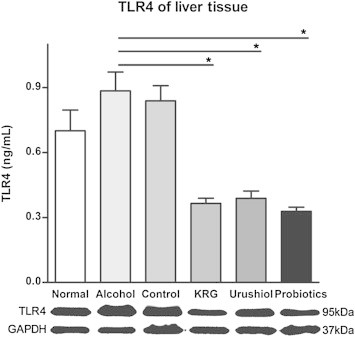
TLR-4 levels of liver tissue are lower in the probiotics, KRG, and urushiol groups, compared to those in the alcohol group. TLR-4 and GAPDH antibodies are detected as single bands at 95 kDa and 37 kDa, respectively. *p < 0.05. KRG, Korean Red Ginseng; TLR-4, toll-like receptor-4.
Treatment with probiotics, KRG, and urushiol was associated with reduced TLR-4 levels in the liver tissue compared with those in the alcohol group. Liver tissue TLR-4 levels were 0.33 ± 0.070 ng/mL (p < 0.001) in the probiotics group, 0.37 ± 0.063 ng/mL (p < 0.01) in the KRG group, and 0.39 ± 0.12 ng/mL (p < 0.05) in the urushiol group, but 0.88 ± 0.31 ng/mL in the alcohol group (Fig. 3).
3.5. Pathological findings
In the alcohol group, four mice exhibited Grade 0 steatosis, four exhibited Grade 1 steatosis, and two exhibited Grade 2 steatosis (p < 0.01 vs. normal group). In the KRG group, eight mice exhibited Grade 0 steatosis, one exhibited Grade 1 steatosis, and one exhibited Grade 2 steatosis (p < 0.05 vs. alcohol group). In the urushiol group, eight mice exhibited Grade 0 steatosis and two exhibited Grade 1 steatosis (p < 0.05 vs. alcohol group). In the probiotics group, six mice exhibited Grade 0 steatosis, whereas four mice exhibited Grade 1 steatosis (p > 0.05 vs. alcohol group; Table 3 and Fig. 4).
Table 3.
Pathological Findings of Each Group
| Group (n) | Grade of steatosis |
|||
|---|---|---|---|---|
| Grade 0 | Grade 1 | Grade 2 | Grade 3 | |
| Normal | 10 | 0 | 0 | 0 |
| Alcohol** | 4 | 4 | 2 | 0 |
| Control | 7 | 2 | 1 | 0 |
| KRG* | 8 | 1 | 1 | 0 |
| Urushiol* | 8 | 2 | 0 | 0 |
| Probiotics | 6 | 4 | 0 | 0 |
*p < 0.05 versus alcohol group
**p < 0.01 versus normal group
KRG, Korean Red Ginseng
Fig. 4.
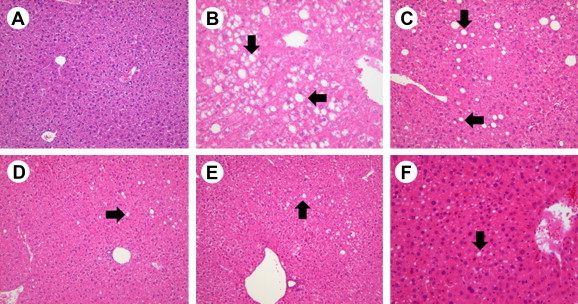
Pathological findings. (A) Normal, (B) alcohol, (C) control, (D) KRG, (E) urushiol, (F) and probiotics groups demonstrate various steatosis grades (arrow: steatosis; hematoxylin and eosin stain). Steatosis grade in the alcohol group is higher than that of the normal group (p < 0.01). Steatosis grade in the KRG and urushiol groups is lower than that of the alcohol group (p < 0.05). KRG, Korean Red Ginseng.
In the necroinflammatory finding of the liver, two mice showed hepatitis in the alcohol group, whereas no inflammation was observed in the KRG, urushiol, and probiotics groups.
4. Discussion
LPS-induced Kupffer cell activation is most likely the primary pathogenesis of ALD. LPS binds to the LPS-binding protein, and is initially transferred to CD 14 and eventually to TLR-4 and myeloid differentiation factor-2 complexes in Kupffer cells. The activation of TLR-4, which is a transmembrane protein that responds primarily to LPS, activates innate immune responses that involve various transcription factors and proinflammatory cytokines [4,5,17]. TLR-4-deficient mice had lower levels of steatosis, inflammation, and proinflammatory cytokines [17,18]. Another study showed that chronic alcohol exposure leads to the hyporesponsiveness of monocytes to LPS because of decreased negative regulators of TLR-4 activation [19].
The present study showed that KRG and probiotic diets did not improve liver function. However, these diets effectively reduced alcohol-induced TLR-4 expression of the liver tissue. These results match those of a previous study demonstrating that the hepatic TLR-4 overexpression that had been increased in LPS- and D-galactosamine-fed rats was significantly downregulated by a Lactobacillus casei Zhang treatment [20]. Another report suggested that ginsenoside Re suppresses the expression of proinflammatory cytokines and the activation of their transcription factor NF-κB by inhibiting the binding of LPS to TLR-4 on immune cells such as macrophages [12]. Together, these results suggest that probiotic and KRG diets display anti-ALD effects by suppressing TLR-4 expression.
TLR-4 levels of the liver tissue were also decreased in urushiol-fed mice compared with those in alcohol-fed mice. According to a study that evaluated the biological effects of urushiol, the antibacterial effect against Helicobacter pylori and anti-inflammatory effect due to the reduction of the IL-1β levels in gastric tissue were demonstrated using a mouse model [15]. The current study is the first to statistically evaluate the effects of urushiol on TLR-4 levels of the liver tissue using an ALD mouse model. In addition to its antibacterial and anti-inflammatory effects on the stomach (as demonstrated by earlier studies), we hypothesize that urushiol also exerts anti-ALD effects by modulating cytokines.
This study also demonstrated that the TNF-α level in the liver tissue of the KRG group was significantly lower than those in the alcohol group. Previous data showed that Panax notoginseng saponins reduced significantly the TNF-α level in CCl4-treated mice with hepatic fibrosis [21]. Another study demonstrated that ginsenoside Rg1 inhibited LPS-induced TNF-α production via dendritic cells [22]. KRG saponin fraction inhibited nitric oxide production and attenuated the release of TNF-α, IL-6, and granulocyte–monocyte colony-stimulating factor [23]. These results match our findings and suggest the presence of a KRG anti-inflammatory effect in both hepatic fibrosis and the ALD inflammatory cascade.
IL-1β levels of the liver tissue in the probiotics and KRG groups decreased compared with those in the alcohol group. These results match those of earlier studies, in which Rg3, an ingredient of Panax ginseng active in neural stem cells, attenuated the upregulation of the LPS-induced IL-1β level [24]. In addition, ginsenoside Rd pretreatment attenuated the increased expression of proinflammatory cytokines (e.g., IL-1β and TNF-α) due to lead (Pb) exposure [25]. Another study demonstrated the efficacy of probiotics in lowering the heightened IL-1β level induced by Candida albicans infection in mice [26]. Assuming IL-1β to be a dangerous cytokine in the ALD inflammatory cascade, Lacidofil and KRG may be hypothesized to have potent anti-ALD effects.
We used a chronic ethanol feeding model (the NIAAA model for ALD). The 4–6-week Lieber–DeCarli diet containing ethanol has been widely used by many laboratories. However, this model induces only mild steatosis and elevates serum alanine aminotransferase slightly, with little or no liver inflammation [27]. In our study, in the liver function test, there was no significant change as shown in the NIAAA model for ALD. The pathological findings of our study showed that alcohol-induced steatosis was significantly reduced by KRG and urushiol. In addition, two mice developed Grade 2 steatosis in the alcohol group and one in the KRG group. Therefore, it is supposed that KRG and urushiol can be used in the treatment of ALD, especially steatosis of liver.
Of the agents that we evaluated, KRG was notably the most effective in reducing the molecular markers that we assessed in mice. However, because the sample size was limited, serum levels of TLR-4, IL-1β, and TNF-α were not significantly ameliorated. Furthermore, injurious cytokines such as IL-6 were not assessed in this study. Therefore, additional clinical or animal model studies are needed.
In conclusion, the current study suggests that KRG, urushiol, and probiotics have potential therapeutic effects, which (in the context of ALD) implicates immune-modulated cytokines in the TLR-4 pathway.
Conflicts of interest
All contributing authors declare no conflicts of interest.
Acknowledgments
This research was supported by a grant from the Korea Society of Ginseng (2011); the Basic Science Research Program through the National Research Foundation of Korea (NRF), funded by the Ministry of Education, Science, and Technology (NRF-2010-0021482); Cooperative Research Program for Agriculture Science and Technology Development (Project No. PJ009859), Rural Development Administration, Republic of Korea; and Hallym University Research Fund.
Footnotes
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
References
- 1.Pares A., Caballeria J., Bruguera M., Torres M., Rodes J. Histological course of alcoholic hepatitis. Influence of abstinence, sex and extent of hepatic damage. J Hepatol. 1986;2:33–42. doi: 10.1016/s0168-8278(86)80006-x. [DOI] [PubMed] [Google Scholar]
- 2.Korean Association for the Study of the Liver KASL clinical practice guidelines: management of alcoholic liver disease. Clin Mol Hepatol. 2013;19:216–254. doi: 10.3350/cmh.2013.19.3.216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Trabut J.B., Thepot V., Terris B., Sogni P., Nalpas B., Pol S. Prognosis assessment of alcoholic liver disease: how and why? Presse Med. 2014;43:124–134. doi: 10.1016/j.lpm.2013.04.016. [DOI] [PubMed] [Google Scholar]
- 4.Roh Y.S., Seki E. Toll-like receptors in alcoholic liver disease, non-alcoholic steatohepatitis and carcinogenesis. J Gastroenterol Hepatol. 2013;28(Suppl. 1):38–42. doi: 10.1111/jgh.12019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Petrasek J., Mandrekar P., Szabo G. Toll-like receptors in the pathogenesis of alcoholic liver disease. Gastroenterol Res Pract. 2010;2010 doi: 10.1155/2010/710381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Frazier T.H., Stocker A.M., Kershner N.A., Marsano L.S., McClain C.J. Treatment of alcoholic liver disease. Therap Adv Gastroenterol. 2011;4:63–81. doi: 10.1177/1756283X10378925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Liu Q., Duan Z.P., Ha D.K., Bengmark S., Kurtovic J., Riordan S.M. Synbiotic modulation of gut flora: effect on minimal hepatic encephalopathy in patients with cirrhosis. Hepatology. 2004;39:1441–1449. doi: 10.1002/hep.20194. [DOI] [PubMed] [Google Scholar]
- 8.Nanji A.A., Khettry U., Sadrzadeh S.M. Lactobacillus feeding reduces endotoxemia and severity of experimental alcoholic liver (disease) Proc Soc Exp Biol Med. 1994;205:243–247. doi: 10.3181/00379727-205-43703. [DOI] [PubMed] [Google Scholar]
- 9.Kirpich I.A., Solovieva N.V., Leikhter S.N., Shidakova N.A., Lebedeva O.V., Sidorov P.I., Bazhukova T.A., Soloviev A.G., Barve S.S., McClain C.J. Probiotics restore bowel flora and improve liver enzymes in human alcohol-induced liver injury: a pilot study. Alcohol. 2008;42:675–682. doi: 10.1016/j.alcohol.2008.08.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Foster L.M., Tompkins T.A., Dahl W.J. A comprehensive post-market review of studies on a probiotic product containing Lactobacillus helveticus R0052 and Lactobacillus rhamnosus R0011. Benef Microbes. 2011;2:319–334. doi: 10.3920/BM2011.0032. [DOI] [PubMed] [Google Scholar]
- 11.Park H.M., Kim S.J., Mun A.R., Go H.K., Kim G.B., Kim S.Z., Jang S.I., Lee S.J., Kim J.S., Kang H.S. Korean Red Ginseng and its primary ginsenosides inhibit ethanol-induced oxidative injury by suppression of the MAPK pathway in TIB-73 cells. J Ethnopharmacol. 2012;141:1071–1076. doi: 10.1016/j.jep.2012.03.038. [DOI] [PubMed] [Google Scholar]
- 12.Lee I.A., Hyam S.R., Jang S.E., Han M.J., Kim D.H. Ginsenoside Re ameliorates inflammation by inhibiting the binding of lipopolysaccharide to TLR4 on macrophages. J Agric Food Chem. 2012;60:9595–9602. doi: 10.1021/jf301372g. [DOI] [PubMed] [Google Scholar]
- 13.Kang S., Min H. Ginseng, the immunity boost': the effects of Panax ginseng on immune system. J Ginseng Res. 2012;36:354–368. doi: 10.5142/jgr.2012.36.4.354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Suk K.T., Kim H.S., Kim M.Y., Kim J.W., Uh Y., Jang I.H., Kim S.K., Choi E.H., Kim M.J., Joo J.S. In vitro antibacterial and morphological effects of the urushiol component of the sap of the Korean lacquer tree (Rhus vernicifera Stokes) on Helicobacter pylori. J Korean Med Sci. 2010;25:399–404. doi: 10.3346/jkms.2010.25.3.399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Suk K.T., Baik S.K., Kim H.S., Park S.M., Paeng K.J., Uh Y., Jang I.H., Cho M.Y., Choi E.H., Kim M.J. Antibacterial effects of the urushiol component in the sap of the lacquer tree (Rhus verniciflua Stokes) on Helicobacter pylori. Helicobacter. 2011;16:434–443. doi: 10.1111/j.1523-5378.2011.00864.x. [DOI] [PubMed] [Google Scholar]
- 16.Tiniakos D.G. Nonalcoholic fatty liver disease/nonalcoholic steatohepatitis: histological diagnostic criteria and scoring systems. Eur J Gastroenterol Hepatol. 2010;22:643–650. doi: 10.1097/MEG.0b013e32832ca0cb. [DOI] [PubMed] [Google Scholar]
- 17.Seo Y.S., Shah V.H. The role of gut–liver axis in the pathogenesis of liver cirrhosis and portal hypertension. Clin Mol Hepatol. 2012;18:337–346. doi: 10.3350/cmh.2012.18.4.337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Hritz I., Mandrekar P., Velayudham A., Catalano D., Dolganiuc A., Kodys K., Kurt-Jones E., Szabo G. The critical role of toll-like receptor (TLR) 4 in alcoholic liver disease is independent of the common TLR adapter MyD88. Hepatology. 2008;48:1224–1231. doi: 10.1002/hep.22470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Mandrekar P., Bala S., Catalano D., Kodys K., Szabo G. The opposite effects of acute and chronic alcohol on lipopolysaccharide-induced inflammation are linked to IRAK-M in human monocytes. J Immunol. 2009;183:1320–1327. doi: 10.4049/jimmunol.0803206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Wang Y., Li Y., Xie J., Zhang Y., Wang J., Sun X., Zhang H. Protective effects of probiotic Lactobacillus casei Zhang against endotoxin- and d-galactosamine-induced liver injury in rats via anti-oxidative and anti-inflammatory capacities. Int Immunopharmacol. 2013;15:30–37. doi: 10.1016/j.intimp.2012.10.026. [DOI] [PubMed] [Google Scholar]
- 21.Peng X.D., Dai L.L., Huang C.Q., He C.M., Yang B., Chen L.J. Relationship between anti-fibrotic effect of Panax notoginseng saponins and serum cytokines in rat hepatic fibrosis. Biochem Biophys Res Commun. 2009;388:31–34. doi: 10.1016/j.bbrc.2009.07.099. [DOI] [PubMed] [Google Scholar]
- 22.Rhule A., Rase B., Smith J.R., Shepherd D.M. Toll-like receptor ligand-induced activation of murine DC2.4 cells is attenuated by Panax notoginseng. J Ethnopharmacol. 2008;116:179–186. doi: 10.1016/j.jep.2007.11.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Yayeh T., Jung K.H., Jeong H.Y., Park J.H., Song Y.B., Kwak Y.S., Kang H.S., Cho J.Y., Oh J.W., Kim S.K. Korean Red Ginseng saponin fraction downregulates proinflammatory mediators in LPS stimulated RAW264.7 cells and protects mice against endotoxic shock. J Ginseng Res. 2012;36:263–269. doi: 10.5142/jgr.2012.36.3.263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Park S.M., Choi M.S., Sohn N.W., Shin J.W. Ginsenoside Rg3 attenuates microglia activation following systemic lipopolysaccharide treatment in mice. Biol Pharm Bull. 2012;35:1546–1552. doi: 10.1248/bpb.b12-00393. [DOI] [PubMed] [Google Scholar]
- 25.Wang B., Feng G., Tang C., Wang L., Cheng H., Zhang Y., Ma J., Shi M., Zhao G. Ginsenoside Rd maintains adult neural stem cell proliferation during lead-impaired neurogenesis. Neurol Sci. 2013;34:1181–1188. doi: 10.1007/s10072-012-1215-6. [DOI] [PubMed] [Google Scholar]
- 26.Zwolinska-Wcislo M., Brzozowski T., Budak A., Kwiecien S., Sliwowski Z., Drozdowicz D., Trojanowska D., Rudnicka-Sosin L., Mach T., Konturek S.J. Effect of Candida colonization on human ulcerative colitis and the healing of inflammatory changes of the colon in the experimental model of colitis ulcerosa. J Physiol Pharmacol. 2009;60:107–118. [PubMed] [Google Scholar]
- 27.Bertola A., Mathews S., Ki S.H., Wang H., Gao B. Mouse model of chronic and binge ethanol feeding (the NIAAA model) Nat Protoc. 2013;8:627–637. doi: 10.1038/nprot.2013.032. [DOI] [PMC free article] [PubMed] [Google Scholar]


