Risks for illness and death caused by the Blastomyces dermatitidis fungus are affected by demographic, geographic, and behavioral factors.
Keywords: Blastomyces dermatitidis, conidia, airborne, yeast, blastomycosis, endemic mycoses, mycosis, mortality, fungi
Abstract
Blastomycosis is a potentially fatal fungal infection endemic to parts of North America. We used national multiple-cause-of-death data and census population estimates for 1990–2010 to calculate age-adjusted mortality rates and rate ratios (RRs). We modeled trends over time using Poisson regression. Death occurred more often among older persons (RR 2.11, 95% confidence limit [CL] 1.76, 2.53 for those 75–84 years of age vs. 55–64 years), men (RR 2.43, 95% CL 2.19, 2.70), Native Americans (RR 4.13, 95% CL 3.86, 4.42 vs. whites), and blacks (RR 1.86, 95% CL 1.73, 2.01 vs. whites), in notably younger persons of Asian origin (mean = 41.6 years vs. 64.2 years for whites); and in the South (RR 18.15, 95% CL 11.63, 28.34 vs. West) and Midwest (RR 23.10, 95% CL14.78, 36.12 vs. West). In regions where blastomycosis is endemic, we recommend that the diagnosis be considered in patients with pulmonary disease and that it be a reportable disease.
Blastomycosis is a systemic infection caused by the thermally dimorphic fungus Blastomyces dermatitidis that can result in severe disease and death among humans and animals. B. dermatitidis is endemic to the states bordering the Mississippi and Ohio Rivers, the Great Lakes, and southern Canada; it is found in moist, acidic, enriched soil near wooded areas and in decaying vegetation or other organic material (1). Conidia, the spores, become airborne after disruption of areas contaminated with B. dermatitidis. Infection occurs primarily through inhalation of the B. dermatitidis spores into the lungs, where they undergo transition to the invasive yeast phase. The infection can progress in the lung, where the infection may be limited, or it can disseminate and result in extrapulmonary disease, affecting other organ systems (2).
The incubation period for blastomycosis is 3–15 weeks. About 30%–50% of infections are asymptomatic. Pulmonary symptoms are the most common clinical manifestations; however, extrapulmonary disease can frequently manifest as cutaneous and skeletal disease and, less frequently, as genitourinary or central nervous system disease. Liver, spleen, pericardium, thyroid, gastrointestinal tract, or adrenal glands may also be involved (3). Misdiagnoses and delayed diagnoses are common because the signs and symptoms resemble those of other diseases, such as bacterial pneumonia, influenza, tuberculosis, other fungal infections, and some malignancies (4). Accurate diagnosis relies on a high index of suspicion with confirmation by using histologic examination, culture, antigen detection assays, or PCR tests (5).
Antifungal agents, such as itraconazole for mild or moderate disease and amphotericin B for severe disease, can provide effective therapy, especially when administered early (1,2). With appropriate treatment, blastomycosis can be successfully treated without relapse; however, case-fatality rates of 4%–22% have been observed (4,6–9). Although spontaneous recovery can occur (10,11), case-patients often require monitoring of clinical progress and administration of drugs on an inpatient basis. Previous studies estimated average hospitalization costs for adults to be $20,000; that is likely less than the current true cost (12). Some reviews of outbreaks indicate a higher distribution of infection among persons of older age, male sex (2,13), black, Asian, and Native American racial/ethnic groups (3,13), and those who have outdoor occupations (13,14). Both immunocompetent and immunocompromised hosts may experience disease and death (2,6,15–19), although B. dermatitidis disproportionately affects immunocompromised patients, who tend to have more rapid and extensive pulmonary involvement, extrapulmonary infection, complications, and higher mortality rates (25%–54%) (2,6,16–19).
Past studies have expanded the knowledge about blastomycosis through focusing on cases documented in specific immunocompromised persons and statewide occurrences or in areas in which the disease is endemic (4,6–9,16–18); however, such studies may be limited for making definitive conclusions by their scope and small sample size. Much remains unknown about the public health burden of blastomycosis-related deaths in the United States. Reports suggest an increase in the number of blastomycosis cases in recent years (13,20). Clearer identification of risk factors from national data may raise awareness of blastomycosis in the United States and support adding it to the list of reportable diseases in regions where the pathogen is endemic to improve surveillance and control. In this study, we assessed the public health burden of blastomycosis-related deaths by examining US mortality-associated data and evaluating demographic, temporal, and geographic associations as potential risk factors.
Methods
Data Source
We used publicly available multiple-cause-of-death (MCOD) data from the National Center for Health Statistics to examine blastomycosis-related deaths in the United States during 1990–2010. These data are derived from US death certificates and include information on the causes of death coded by the International Classification of Diseases, 9th and 10th Revisions (ICD-9, ICD-10), demographic variables of age, sex, and race/ethnicity, date of death, and geographic region of residence.
Case Definition
We defined a case-patient as deceased US resident listed in the MCOD dataset during 1990–2010 whose death certificate listed blastomycosis as the underlying or contributing cause of death. The ICD-9 code 116.0 (years 1990–1998) and ICD-10 codes B40.0–B40.9 (years 1999–2010) were used to identify blastomycosis-related deaths.
Analysis
To ensure more stable estimates, we aggregated data for the study period. We calculated mortality rates and rate ratios (RRs) with 95% confidence limits (CLs) by age, sex, race/ethnicity, geographic region, and year of death using a maximum likelihood analysis presuming the response variable had a Poisson distribution (21), and with bridged-race population estimates data and designated geographic boundaries from the US census. We computed age-adjusted mortality rates using adjustment weights from the year 2000 US standard population data. We assessed temporal trends in age-adjusted mortality rates using a Poisson regression model of deaths per person-years in the population, designating year and age group dummy variables as independent variables, and the population as the offset. We calculated the percentage change by year based on the estimated slope parameter and examined the Poisson regression models for overdispersion. We performed all analyses using SAS for Windows version 9.4 (SAS Institute Inc., Cary, NC, USA).
Results
We identified 1,216 blastomycosis-related deaths among 49,574,649 deaths in the United States during 1990–2010. Among those 1,216 deaths, blastomycosis was reported as the underlying cause of death for 741 (60.9%), and as a contributing cause of death for 475 (39.1%). The overall age-adjusted mortality rate for the period was 0.21 (95% CL 0.20, 0.22) per 1 million person-years. Using Poisson regression, we identified a 2.21% (95% CL −3.11, −1.29) decline in blastomycosis-related mortality rates during the period (Figure).
Figure.
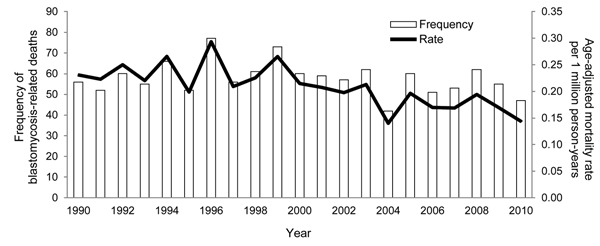
Number of blastomycosis-related deaths and age-adjusted mortality rates per 1 million person-years, by year, United States, 1990–2010.
Age
The mean age at death from blastomycosis was 60.8 years. Using 75 as the average age at death (22,23), we calculated that 19,097 years of potential life were lost. The mortality rates associated with blastomycosis increased with increasing age, peaking in the 75- to 84-year age group (Table 1). The mean age at death from blastomycosis was significantly lower among Hispanics (p<0.01), Native Americans (p<0.01), blacks (p<0.01), and Asians (p<0.01) than among whites based on the t test for difference in means.
Table 1. Blastomycosis-related deaths by sex, race/ethnicity, age group, and geographic region with corresponding age-adjusted mortality rates, United States, 1990–2010*.
| Characteristic | No. (%) deaths | Mean age at death, y | Age-adjusted mortality rate/1 million person-years (95% CL)† | Age-adjusted mortality rate ratio (95% CL) |
|---|---|---|---|---|
| Sex | ||||
| F | 409 (33.6) | 62.3 | 0.14 (0.13, 0.16) | 1 |
| M |
807 (66.4) |
60.1 |
0.35 (0.32, 0.37) |
2.43 (2.19, 2.70) |
| Race/ethnicity | ||||
| White | 918 (75.5) | 64.2 | 0.22 (0.21, 0.23) | 1 |
| Hispanic | 25 (2.1) | 53.0 | 0.06 (0.03, 0.08) | 0.25 (0.19, 0.33) |
| Black | 223 (18.3) | 50.6 | 0.41 (0.35, 0.46) | 1.86 (1.73, 2.01) |
| Asian | 20 (1.6) | 41.6 | 0.11 (0.06, 0.15) | 0.47 (0.41, 0.55) |
| Native American |
30 (2.5) |
52.9 |
0.91 (0.57, 1.25) |
4.13 (3.86, 4.42) |
| Age, y‡ | ||||
| <1 | 1 (0.1) | NA | 0.01 (0.00, 0.04) | 0.03 (0.00, 0.19) |
| 1–4 | 1 (0.1) | NA | 0.00 (0.00, 0.01) | 0.01 (0.00, 0.05) |
| 5–14 | 6 (0.5) | 9.2 | 0.01 (0.00, 0.01) | 0.02 (0.01, 0.04) |
| 15–24 | 33 (2.7) | 19.3 | 0.04 (0.03, 0.05) | 0.09 (0.06, 0.13) |
| 25–34 | 59 (4.9) | 30.3 | 0.07 (0.05, 0.09) | 0.16 (0.12, 0.21) |
| 35–44 | 122 (10.0) | 39.7 | 0.14 (0.11, 0.16) | 0.31 (0.25, 0.39) |
| 45–54 | 189 (15.5) | 49.7 | 0.25 (0.21, 0.28) | 0.56 (0.46, 0.68) |
| 55–64 | 240 (19.7) | 59.7 | 0.44 (0.38. 0.49) | 1 |
| 65–74 | 257 (21.1) | 69.9 | 0.65 (0.57, 0.73) | 1.48 (1.24, 1.76) |
| 75–84 | 235 (19.3) | 78.9 | 0.92 (0.80, 1.04) | 2.11 (1.76, 2.53) |
| ≥85 |
73 (6.0) |
88.1 |
0.81 (0.62, 0.99) |
1.84 (1.42, 2.40) |
| Geographic region‡ | ||||
| West | 20 (1.7) | 67.1 | 0.02 (0.01, 0.02) | 1 |
| South | 643 (52.9) | 60.5 | 0.31 (0.28, 0.33) | 18.15 (11.63, 28.34) |
| Midwest | 533 (43.8) | 61.1 | 0.39 (0.36, 0.42) | 23.10 (14.78, 36.12) |
| Northeast |
20 (1.7) |
61.6 |
0.02 (0.01, 0.02) |
1.00 (0.54, 1.86) |
| Total | 1,216 (100) | 60.8 | 0.21 (0.20, 0.22) |
*CL, confidence limit; NA, not applicable. States in western region: AK, AZ, CA, CO, HI, ID, MT, NM, NV, OR, UT, WA, WY. States in southern region: AL, AR, DC, DE, FL, GA, KY, LA, MS, NC, OK, SC, TN, TX, VA, WV. States in midwestern region: IA, IL, IN, KS, MI, MN, MO, MS, NE, ND, OH, SD, WI. States in northeastern region: CT, MA, ME, NH, NJ, NY, PA, RI, VT. †Standard reference population is US standard population during year 2000. ‡Age-specific rates.
Sex
Death related to blastomycosis was significantly more likely in men than in women (p<0.05). The average age at death was significantly lower for men than for women (p = 0.02) (Table 1). The annual mortality rate over the period obtained from using Poisson regression declined for both men and women (Table 2).
Table 2. Age-adjusted time trends in blastomycosis-related mortality rate for sex, race/ethnicity, and geographic region, United States, 1990–2010*.
| Variable | Annual percent change† in age-adjusted mortality rates (95% CL) | p value |
|---|---|---|
| Sex | ||
| F | −2.28 (−3.84, −0.70) | <0.01 |
| M |
−2.32 (−3.43, −1.20) |
<0.01 |
| Race/ethnicity | ||
| White | −1.57 (−2.62, −0.51) | <0.01 |
| Hispanic | ‡ | |
| Black | −5.12 (−7.19, −3.01) | <0.01 |
| Asian | ‡ | |
| Native American |
‡ |
|
| Geographic region | ||
| Northeast | ‡ | |
| South | −5.06 (−6.28, −3.28) | <0.01 |
| Midwest | 1.70 (0.27, 3.15) | 0.02 |
| West | ‡ | |
| Total | −2.21 (−3.11, −1.29) | <0.01 |
*CL, confidence limit. †Annual percent change based on the Poisson model for death, with year and age group dummy variables as independent variables, and the base population as the offset. No other covariates were adjusted in the model. ‡Few deaths occurred in these categories, and thus, were not included in Poisson regression analyses to avoid inappropriate use of small numbers in Poisson regression modeling.
Race/Ethnicity
Native Americans and blacks were significantly more likely to die from blastomycosis-related complications than whites; overall, Asians and Hispanics were significantly less likely to die of blastomycosis than other groups (Table 1). The annual mortality rate over the period declined among blacks and whites (Table 2).
Geographic Region
Most (96.7%) of the blastomycosis-related deaths occurred in the southern and midwestern regions, and a small proportion of deaths occurred in the northeastern and western regions. The midwestern region had the highest mortality rate, followed by the southern, northeastern, and western regions (Table 1). Percentage changes in mortality rates per year over the period, calculated by using Poisson regression, showed an increase in mortality rates in the midwestern region, and a decline in the southern region (Table 2).
Table 3 shows the results of a subanalysis of the demographic characteristics of populations in the southern and midwestern regions. In the southern region, the mean age at death from blastomycosis was significantly lower among Native Americans (p = 0.03), blacks (p<0.01), and Hispanics (p = 0.02) than among whites based on a t test for difference in means. In the midwestern region, the mean age at death from blastomycosis was significantly lower among Native Americans (p = 0.02), Asians (p<0.01), blacks (p<0.01), and Hispanics (p<0.01) than among whites. Furthermore, the mean age at death from blastomycosis in the midwestern region was significantly lower among Asians than among Native Americans (p<0.01), blacks (p<0.01), and Hispanics (p = 0.04).
Table 3. Demographic distribution and age-adjusted mortality rates for the Midwestern and Southern regions in the United States, 1990–2010.
| Characteristic | Midwest |
South |
|||||
|---|---|---|---|---|---|---|---|
| No. (%) deaths | Mean age at death, y | Age-adjusted mortality rate per 1 million person-years (95% CL) | No. (%) deaths | Mean age at death, y | Age-adjusted mortality rate per 1 million person-years (95% CL) | ||
| Sex | |||||||
| F | 186 (34.9) | 62.7 | 0.25 (0.21, 0.28) | 212 (33.0) | 61.7 | 0.18 (0.16, 0.21) | |
| M |
347 (65.1) |
60.2 |
0.57 (0.51, 0.63) |
|
431 (67.0) |
59.9 |
0.46 (0.42, 0.50) |
| Race/ethnicity | |||||||
| White | 418 (78.4) | 64.5 | 0.34 (0.31, 0.38) | 470 (73.1) | 64.0 | 0.29 (0.27, 0.32) | |
| Hispanic | 17 (3.2) | 47.2 | 0.45 (0.20, 0.70) | 4 (0.6) | 50.3 | 0.02 (0.00, 0.04) | |
| Black | 63 (11.8) | 50.7 | 0.55 (0.41, 0.69) | 157 (24.4) | 50.6 | 0.46 (0.39, 0.54) | |
| Asian | 16 (3.0) | 37.3 | 0.66 (0.29, 1.02) | 3 (0.5) | 56.7 | 0.09 (0.00, 0.20) | |
| Native American | 19 (3.6) | 52.7 | 0.34 (0.31, 0.38) | 9 (1.4) | 51.1 | 0.71 (0.23, 1.20) | |
| Total | 533 (100.0) | 61.1 | 0.39 (0.36, 0.42) | 643 (100.0) | 60.5 | 0.31 (0.28, 0.33) | |
Discussion
Our findings indicate that blastomycosis is a noteworthy cause of preventable death in the United States. These findings confirm the demographic risk factors of blastomycosis indicated in previous case reports and extend these to mortality rates. Blastomycosis death occurred more often among older persons than among younger persons (24), and more often among men than women (2,24). The age association found likely represents waning age-related immune function and higher prevalence of immunocompromising conditions. The observed sex differences in blastomycosis mortality may be attributable to differences in occupational or recreational exposures that increase risk for infection (14). For example, those who work outdoors involving construction, excavation, or forestry, or participate in outdoor recreational activities such as hunting (7,11), may more likely be exposed than those who principally work indoors.
The disproportionate burden of blastomycosis deaths sustained by persons of Native American or black race is also consistent with previous reports (3,24). Increased exposure and prevalence of infection, reduced access to health care, and genetic differences may play a role in the observed race-specific disparities in blastomycosis mortality rates (25). A finding of the current study is that even though persons of Asian descent are at lower risk for dying from blastomycosis than whites, those who died from blastomycosis did so at a much younger age (22.6 years younger). This disparity is even greater in the midwestern region, where Asians died at an age 27.2 years younger than did whites.
Consistent with the recognized geographic distribution of B. dermatitidis (1,2), we found that death related to blastomycosis occurred more often among persons who resided in the midwestern or southern regions than among those in the western and northeastern regions. During the study period, the southern region showed decreases in mortality rates, and the midwestern region, which had the highest mortality rate, showed an increase in rate.
The use of population-based data and large numbers can provide insight, though some limitations associated with using MCOD data should be considered. First, potential underdiagnosis and underreporting of death related to blastomycosis may lead to underestimates of mortality rates and the true public health burden of blastomycosis in the United States. Low physician awareness of blastomycosis may be a contributor. Second, it was not possible to verify accuracy of recorded data or access supplemental data. For example, there may be reporting errors regarding correct race/ethnicity identification on death certificates and in population census reports. Third, we could not adjust for other possible confounders (i.e., smoking, socioeconomic factors, activity, lifestyle, occupation) because these data are not recorded on death certificates. These limitations must be considered along with our findings.
This study sheds light on the scope of the incidence of blastomycosis in the United States, though the true incidence may be greater than that reported here. Although B. dermatitidis infection may be difficult to prevent because of its widespread distribution in areas where blastomycosis is endemic, deaths resulting from blastomycosis can be prevented with early recognition and treatment of patients with symptomatic infection. The continued incidence of blastomycosis in the United States, as indicated by the observed modest decrease in the mortality rates over the 21-year study period, calls for improvement in provider and community awareness, which may lead to including blastomycosis as a diagnostic consideration in patients with pulmonary disease refractory to treatment. Our findings, recent reports of disproportionately high infection rates among Asians (26), and the lack of decline in the mortality rates in the midwestern region support further investigation. We also encourage improvements in blastomycosis surveillance that involve examining trends in incident cases, hospitalization (including length of stay), timely diagnosis, and treatment to further elucidate the burden of blastomycosis in the United States.
Acknowledgments
We thank Matthew Redelings for his contributions to the analysis of data in this study.
Ms Khuu is a doctoral student in epidemiology at the University of California, Los Angeles, School of Public Health. Her research interests include the epidemiology and control of infectious diseases.
Footnotes
Suggested citation for this article: Khuu D, Shafir S, Bristow B, Sorvillo F. Blastomycosis mortality rates, United States, 1990–2010. Emerg Infect Dis. 2014 Nov [date cited]. http://dx.doi.org/10.3201/eid2011.131175
References
- 1.Chapman SW, Sullivan DC. Blastomycosis. In: Infectious disease: diagnosis and treatment of human mycoses. Totowa (NJ): Humana Press. 2008. p. 279–293. [Google Scholar]
- 2.Saccente M, Woods GL. Clinical and laboratory update on blastomycosis. Clin Microbiol Rev. 2010;23:367–81 . 10.1128/CMR.00056-09 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Lemos LB, Guo M, Baliga M. Blastomycosis: organ involvement and etiologic diagnosis. A review of 123 patients from Mississippi. Ann Diagn Pathol. 2000;4:391–406. 10.1053/adpa.2000.20755 [DOI] [PubMed] [Google Scholar]
- 4.Chapman SW, Lin AC, Hendricks KA, Nolan RL, Currier MM, Morris KR, et al. Endemic blastomycosis in Mississippi: epidemiological and clinical studies. Semin Respir Infect. 1997;12:219–28 . [PubMed] [Google Scholar]
- 5.Hage CA, Knox KS, Wheat LJ. Endemic mycoses: overlooked causes of community acquired pneumonia. Respir Med. 2012;106:769–76. 10.1016/j.rmed.2012.02.004 [DOI] [PubMed] [Google Scholar]
- 6.Pappas PG, Threlkeld MG, Bedsole GD, Cleveland KO, Gelfand MS, Dismukes WE. Blastomycosis in immunocompromised patients. Medicine. 1993;72:311–25 . 10.1097/00005792-199309000-00003 [DOI] [PubMed] [Google Scholar]
- 7.Centers for Disease Control and Prevention. Blastomycosis—Wisconsin, 1986–1995. MMWR Morb Mortal Wkly Rep. 1996;45:601–3 . [PubMed] [Google Scholar]
- 8.Pappas PG, Pottage JC, Powderly WG, Fraser VJ, Stratton CW, McKenzie S, et al. Blastomycosis in patients with the acquired immunodeficiency syndrome. Ann Intern Med. 1992;116:847–53 and. 10.7326/0003-4819-116-10-847 [DOI] [PubMed] [Google Scholar]
- 9.Crampton TL, Light RB, Berg GM, Meyers MP, Schroeder GC, Hershfield ES, et al. Epidemiology and clinical spectrum of blastomycosis diagnosed at Manitoba hospitals. Clin Infect Dis. 2002;34:1310–6. 10.1086/340049 [DOI] [PubMed] [Google Scholar]
- 10.Klein BS, Vergeront JM, Davis JP. Epidemiologic aspects of blastomycosis, the enigmatic systemic mycosis. Semin Respir Infect. 1986;1:29–39 . [PubMed] [Google Scholar]
- 11.Bradsher RW. Blastomycosis. Clin Infect Dis. 1992;14:S82–90. 10.1093/clinids/14.Supplement_1.S82 [DOI] [PubMed] [Google Scholar]
- 12.Chu JH, Feudtner C, Heydon K, Walsh TJ, Zaoutis TE. Hospitalizations for endemic mycoses: a population-based national study. Clin Infect Dis. 2006;42:822–5. 10.1086/500405 [DOI] [PubMed] [Google Scholar]
- 13.Benedict K, Roy M, Chiller T, Davis JP. Epidemiologic and ecologic features of blastomycosis: a review. Current Fungal Infection Reports. 2012;6:327–35.
- 14.Choptiany M, Wiebe L, Limerick B, Sarsfield P, Cheang M, Light B, et al. Risk factors for acquisition of endemic blastomycosis. Can J Infect Dis Med Microbiol. 2009;20:117–21 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Recht LD, Davies SF, Eckman MR, Sarosi GA. Blastomycosis in immunosuppressed patients. Am Rev Respir Dis. 1982;125:359–62 . [DOI] [PubMed] [Google Scholar]
- 16.Lemos LB, Baliga M, Guo M. Blastomycosis: The great pretender can also be an opportunist. Initial clinical diagnosis and underlying diseases in 123 patients. Ann Diagn Pathol. 2002;6:194–203. 10.1053/adpa.2002.34575 [DOI] [PubMed] [Google Scholar]
- 17.Grim SA, Proia L, Miller R, Alhyraba M, Costas-Chavarri A, Oberholzer J, et al. A multicenter study of histoplasmosis and blastomycosis after solid organ transplantation. Transpl Infect Dis. 2012;14:17–23. 10.1111/j.1399-3062.2011.00658.x [DOI] [PubMed] [Google Scholar]
- 18.Witzig RS, Hoadley DJ, Greer DL, Abriola KP, Hernandez RL. Blastomycosis and human immunodeficiency virus: three new cases and review. South Med J. 1994;87:715–9. 10.1097/00007611-199407000-00008 [DOI] [PubMed] [Google Scholar]
- 19.Vasquez JE, Mehta JB, Agrawal R, Sarubbi FA. Blastomycosis in northeast Tennessee. Chest. 1998;114:436–43. 10.1378/chest.114.2.436 [DOI] [PubMed] [Google Scholar]
- 20.Carlos WG, Rose AS, Wheat LJ, Norris S, Sarosi GA, Knox KS, et al. Blastomycosis in Indiana: digging up more cases. Chest. 2010;138:1377–82. 10.1378/chest.10-0627 [DOI] [PubMed] [Google Scholar]
- 21.Rothman KJ, Lash TL, Greenland S. Modern epidemiology. 3rd ed. Philadelphia: Wolters Kluwer Health; 2012. [Google Scholar]
- 22.Gardner JW, Sanborn JS. Years of potential life lost (YPLL)—what does it measure? Epidemiology. 1990;1:322–9. 10.1097/00001648-199007000-00012 [DOI] [PubMed] [Google Scholar]
- 23.Centers for Disease Control and Prevention. Health, United States, 2012: with special feature on emergency care. 2013 May [cited 2013 Jul 30]. http://www.cdc.gov/nchs/data/hus/hus12.pdf
- 24.Dworkin MS, Duckro AN, Proia L, Semel JD, Huhn G. The epidemiology of blastomycosis in Illinois and factors associated with death. Clin Infect Dis. 2005;41:e107–11. 10.1086/498152 [DOI] [PubMed] [Google Scholar]
- 25.Klein BS. Molecular basis of pathogenicity in Blastomyces dermatitidis: the importance of adhesion. Curr Opin Microbiol. 2000;3:339–43. 10.1016/S1369-5274(00)00100-4 [DOI] [PubMed] [Google Scholar]
- 26.Roy M, Benedict K, Deak E, Kirby MA, McNiel JT, Sickler CJ, et al. A large community outbreak of blastomycosis in Wisconsin with geographic and ethnic clustering. Clin Infect Dis. 2013;57:655–62. 10.1093/cid/cit366 [DOI] [PMC free article] [PubMed] [Google Scholar]


