Abstract
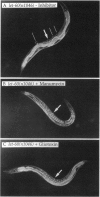
Attachment of Ras protein to the membrane, which requires farnesylation at its C terminus, is essential for its biological activity. A promising pharmacological approach of antagonizing oncogenic Ras activity is to develop inhibitors of farnesyltransferase. We use Caenorhabditis elegans vulval differentiation, which is controlled by a Ras-mediated signal transduction pathway, as a model system to test previously identified farnesyltransferase inhibitors. We show here that two farnesyltransferase inhibitors, manumycin and gliotoxin, suppress the Multivulva phenotype resulting from an activated let-60 ras mutation, but not the Multivulva phenotype resulting from mutations in the lin-1 gene or the lin-15 gene, which act downstream and upstream of let-60 ras, respectively, in the signaling pathway. These results are consistent with the idea that the suppression of the Multivulva phenotype of let-60 ras by the two inhibitors is specific for Ras protein and that the mutant Ras protein might be more sensitive than wild-type Ras to the farnesyltransferase inhibitors. This work suggests that C. elegans vulval development could be a simple and effective in vivo system for evaluation of farnesyltransferase inhibitors against Ras-activated tumors.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aroian R. V., Koga M., Mendel J. E., Ohshima Y., Sternberg P. W. The let-23 gene necessary for Caenorhabditis elegans vulval induction encodes a tyrosine kinase of the EGF receptor subfamily. Nature. 1990 Dec 20;348(6303):693–699. doi: 10.1038/348693a0. [DOI] [PubMed] [Google Scholar]
- Barbacid M. ras genes. Annu Rev Biochem. 1987;56:779–827. doi: 10.1146/annurev.bi.56.070187.004023. [DOI] [PubMed] [Google Scholar]
- Beitel G. J., Clark S. G., Horvitz H. R. Caenorhabditis elegans ras gene let-60 acts as a switch in the pathway of vulval induction. Nature. 1990 Dec 6;348(6301):503–509. doi: 10.1038/348503a0. [DOI] [PubMed] [Google Scholar]
- Bos J. L. ras oncogenes in human cancer: a review. Cancer Res. 1989 Sep 1;49(17):4682–4689. [PubMed] [Google Scholar]
- Brenner S. The genetics of Caenorhabditis elegans. Genetics. 1974 May;77(1):71–94. doi: 10.1093/genetics/77.1.71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clark S. G., Lu X., Horvitz H. R. The Caenorhabditis elegans locus lin-15, a negative regulator of a tyrosine kinase signaling pathway, encodes two different proteins. Genetics. 1994 Aug;137(4):987–997. doi: 10.1093/genetics/137.4.987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clark S. G., Stern M. J., Horvitz H. R. C. elegans cell-signalling gene sem-5 encodes a protein with SH2 and SH3 domains. Nature. 1992 Mar 26;356(6367):340–344. doi: 10.1038/356340a0. [DOI] [PubMed] [Google Scholar]
- Clarke S. Protein isoprenylation and methylation at carboxyl-terminal cysteine residues. Annu Rev Biochem. 1992;61:355–386. doi: 10.1146/annurev.bi.61.070192.002035. [DOI] [PubMed] [Google Scholar]
- Cox A. D., Garcia A. M., Westwick J. K., Kowalczyk J. J., Lewis M. D., Brenner D. A., Der C. J. The CAAX peptidomimetic compound B581 specifically blocks farnesylated, but not geranylgeranylated or myristylated, oncogenic ras signaling and transformation. J Biol Chem. 1994 Jul 29;269(30):19203–19206. [PubMed] [Google Scholar]
- Ferguson E. L., Horvitz H. R. Identification and characterization of 22 genes that affect the vulval cell lineages of the nematode Caenorhabditis elegans. Genetics. 1985 May;110(1):17–72. doi: 10.1093/genetics/110.1.17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferguson E. L., Horvitz H. R. The multivulva phenotype of certain Caenorhabditis elegans mutants results from defects in two functionally redundant pathways. Genetics. 1989 Sep;123(1):109–121. doi: 10.1093/genetics/123.1.109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Forrester K., Almoguera C., Han K., Grizzle W. E., Perucho M. Detection of high incidence of K-ras oncogenes during human colon tumorigenesis. 1987 May 28-Jun 3Nature. 327(6120):298–303. doi: 10.1038/327298a0. [DOI] [PubMed] [Google Scholar]
- Garcia A. M., Rowell C., Ackermann K., Kowalczyk J. J., Lewis M. D. Peptidomimetic inhibitors of Ras farnesylation and function in whole cells. J Biol Chem. 1993 Sep 5;268(25):18415–18418. [PubMed] [Google Scholar]
- Gibbs J. B., Schaber M. D., Schofield T. L., Scolnick E. M., Sigal I. S. Xenopus oocyte germinal-vesicle breakdown induced by [Val12]Ras is inhibited by a cytosol-localized Ras mutant. Proc Natl Acad Sci U S A. 1989 Sep;86(17):6630–6634. doi: 10.1073/pnas.86.17.6630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Han M., Aroian R. V., Sternberg P. W. The let-60 locus controls the switch between vulval and nonvulval cell fates in Caenorhabditis elegans. Genetics. 1990 Dec;126(4):899–913. doi: 10.1093/genetics/126.4.899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Han M., Golden A., Han Y., Sternberg P. W. C. elegans lin-45 raf gene participates in let-60 ras-stimulated vulval differentiation. Nature. 1993 May 13;363(6425):133–140. doi: 10.1038/363133a0. [DOI] [PubMed] [Google Scholar]
- Han M. Ras proteins in developmental pattern formation in Caenorhabditis elegans and Drosophila. Semin Cancer Biol. 1992 Aug;3(4):219–228. [PubMed] [Google Scholar]
- Han M., Sternberg P. W. Analysis of dominant-negative mutations of the Caenorhabditis elegans let-60 ras gene. Genes Dev. 1991 Dec;5(12A):2188–2198. doi: 10.1101/gad.5.12a.2188. [DOI] [PubMed] [Google Scholar]
- Han M., Sternberg P. W. let-60, a gene that specifies cell fates during C. elegans vulval induction, encodes a ras protein. Cell. 1990 Nov 30;63(5):921–931. doi: 10.1016/0092-8674(90)90495-z. [DOI] [PubMed] [Google Scholar]
- Hara M., Akasaka K., Akinaga S., Okabe M., Nakano H., Gomez R., Wood D., Uh M., Tamanoi F. Identification of Ras farnesyltransferase inhibitors by microbial screening. Proc Natl Acad Sci U S A. 1993 Mar 15;90(6):2281–2285. doi: 10.1073/pnas.90.6.2281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hill R. J., Sternberg P. W. The gene lin-3 encodes an inductive signal for vulval development in C. elegans. Nature. 1992 Aug 6;358(6386):470–476. doi: 10.1038/358470a0. [DOI] [PubMed] [Google Scholar]
- Horvitz H. R., Sternberg P. W. Multiple intercellular signalling systems control the development of the Caenorhabditis elegans vulva. Nature. 1991 Jun 13;351(6327):535–541. doi: 10.1038/351535a0. [DOI] [PubMed] [Google Scholar]
- Horvitz H. R., Sulston J. E. Isolation and genetic characterization of cell-lineage mutants of the nematode Caenorhabditis elegans. Genetics. 1980 Oct;96(2):435–454. doi: 10.1093/genetics/96.2.435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang L. S., Tzou P., Sternberg P. W. The lin-15 locus encodes two negative regulators of Caenorhabditis elegans vulval development. Mol Biol Cell. 1994 Apr;5(4):395–411. doi: 10.1091/mbc.5.4.395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- James G. L., Brown M. S., Cobb M. H., Goldstein J. L. Benzodiazepine peptidomimetic BZA-5B interrupts the MAP kinase activation pathway in H-Ras-transformed Rat-1 cells, but not in untransformed cells. J Biol Chem. 1994 Nov 4;269(44):27705–27714. [PubMed] [Google Scholar]
- James G. L., Goldstein J. L., Brown M. S., Rawson T. E., Somers T. C., McDowell R. S., Crowley C. W., Lucas B. K., Levinson A. D., Marsters J. C., Jr Benzodiazepine peptidomimetics: potent inhibitors of Ras farnesylation in animal cells. Science. 1993 Jun 25;260(5116):1937–1942. doi: 10.1126/science.8316834. [DOI] [PubMed] [Google Scholar]
- Kohl N. E., Mosser S. D., deSolms S. J., Giuliani E. A., Pompliano D. L., Graham S. L., Smith R. L., Scolnick E. M., Oliff A., Gibbs J. B. Selective inhibition of ras-dependent transformation by a farnesyltransferase inhibitor. Science. 1993 Jun 25;260(5116):1934–1937. doi: 10.1126/science.8316833. [DOI] [PubMed] [Google Scholar]
- Lackner M. R., Kornfeld K., Miller L. M., Horvitz H. R., Kim S. K. A MAP kinase homolog, mpk-1, is involved in ras-mediated induction of vulval cell fates in Caenorhabditis elegans. Genes Dev. 1994 Jan;8(2):160–173. doi: 10.1101/gad.8.2.160. [DOI] [PubMed] [Google Scholar]
- Leevers S. J., Paterson H. F., Marshall C. J. Requirement for Ras in Raf activation is overcome by targeting Raf to the plasma membrane. Nature. 1994 Jun 2;369(6479):411–414. doi: 10.1038/369411a0. [DOI] [PubMed] [Google Scholar]
- Prendergast G. C., Davide J. P., deSolms S. J., Giuliani E. A., Graham S. L., Gibbs J. B., Oliff A., Kohl N. E. Farnesyltransferase inhibition causes morphological reversion of ras-transformed cells by a complex mechanism that involves regulation of the actin cytoskeleton. Mol Cell Biol. 1994 Jun;14(6):4193–4202. doi: 10.1128/mcb.14.6.4193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schafer W. R., Rine J. Protein prenylation: genes, enzymes, targets, and functions. Annu Rev Genet. 1992;26:209–237. doi: 10.1146/annurev.ge.26.120192.001233. [DOI] [PubMed] [Google Scholar]
- Stokoe D., Macdonald S. G., Cadwallader K., Symons M., Hancock J. F. Activation of Raf as a result of recruitment to the plasma membrane. Science. 1994 Jun 3;264(5164):1463–1467. doi: 10.1126/science.7811320. [DOI] [PubMed] [Google Scholar]
- Trueblood C. E., Ohya Y., Rine J. Genetic evidence for in vivo cross-specificity of the CaaX-box protein prenyltransferases farnesyltransferase and geranylgeranyltransferase-I in Saccharomyces cerevisiae. Mol Cell Biol. 1993 Jul;13(7):4260–4275. doi: 10.1128/mcb.13.7.4260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van der Pyl D., Inokoshi J., Shiomi K., Yang H., Takeshima H., Omura S. Inhibition of farnesyl-protein transferase by gliotoxin and acetylgliotoxin. J Antibiot (Tokyo) 1992 Nov;45(11):1802–1805. doi: 10.7164/antibiotics.45.1802. [DOI] [PubMed] [Google Scholar]
- Wu Y., Han M. Suppression of activated Let-60 ras protein defines a role of Caenorhabditis elegans Sur-1 MAP kinase in vulval differentiation. Genes Dev. 1994 Jan;8(2):147–159. doi: 10.1101/gad.8.2.147. [DOI] [PubMed] [Google Scholar]