Abstract
Background/Aims
This study investigated the expression of nuclear factor κB (NF-κB) and the chemokine receptor (CXCR4) in patients with diffuse large B-cell lymphoma (DLBCL) who received rituximab-based therapy.
Methods
Seventy patients with DLBCL and treated with rituximab-CHOP (R-CHOP) were included, and immunohistochemistry was performed to determine the expression of NF-κB (IκB kinase α, p50, and p100/p52) and CXCR4. To classify DLBCL cases as germinal center B-cell-like (GCB) and non-GCB, additional immunohistochemical expression of CD10, bcl-6, or MUM1 was used in this study. The expression was divided into two groups according to the intensity score (negative, 0 or 1+; positive, 2+ or 3+).
Results
The median age of the patients was 66 years (range, 17 to 87), and 58.6% were male. Twenty-seven patients (38.6%) had stage III or IV disease at diagnosis. Twenty-three patients (32.9%) were categorized as high or high-intermediate risk according to their International Prognostic Indexs (IPIs). The overall incidence of bone marrow involvement was 5.7%. Rates of positive NF-κB and CXCR4 expression were 84.2% and 88.6%, respectively. High NF-κB expression was associated with CXCR4 expression (p = 0.002), and 56 patients (80.0%) showed coexpression. However, the expression of NF-κB or CXCR4 was not associated with overall survival and EFS. On multivariate analysis that included age, gender, performance status, stage, and the IPI, no significant association between the grade of NF-κB or CXCR4 expression and survival was observed.
Conclusions
The current study suggests that the tissue expression of NF-κB and CXCR4 may not be an independent prognostic marker in DLBCL patients treated with R-CHOP.
Keywords: Lymphoma, NF-kappa B, CXCR4
INTRODUCTION
Despite the benefits of adding rituximab (a chimeric immunoglobulin G1 monoclonal antibody targeting CD20) to cyclophosphamide, vincristine, adriamycin, and prednisolone (CHOP) therapy, the relapse rate is relatively high, and survival after relapse remains low, in patients with diffuse large B-cell lymphoma (DLBCL) [1]. The prognosis of patients with DLBCL has been shown to be influenced by several established surgical-pathological features. In particular, because DLBCL is considered a heterogeneous disease based on its development, growth, and progression depending on the deregulation of various cellular pathways, it can be subdivided into several subtypes according to its immunochemical features [2,3,4]. Two representative examples are nuclear factor κB (NF-κB) and the chemokine receptor CXCR4.
The NF-κB family comprises five members that form homodimers or heterodimers, and can be activated through the classical or alternative pathway. Previous studies have demonstrated that abnormal activation of NF-κB contributes to lymphoma development and progression, as well as resistance of malignant cells to chemotherapy and radiation [5]. Therefore, NF-κB may be a predictor of prognosis in lymphoma patients or a beneficial target for lymphoma treatment, although the underlying mechanism in lymphoma remains unknown [6,7]. For example, Bavi et al. [8] reported a significant correlation between high expression of NF-κB and a poor prognosis in patients with DLBCL.
CXCR4 is an α-chemokine receptor specific for stromal derived factor-1 (SDF-1), which has potent chemotactic activity for lymphocytes. CXCR4 expression has been demonstrated in various tumors and was linked to metastasis to tissues containing a high concentration of SDF-1 [9]. In recent years, CXCR4 expression was also found in malignant lymphoma cell lines, and its inhibition by a monoclonal antibody enhanced apoptosis, decreased proliferation, and inhibited migration [10].
Interestingly, Okera et al. [11] reported that NF-κB expression was positively correlated with CXCR4 expression in prostate cancer. Furthermore, NF-κB has been suggested to regulate migration and metastasis by up-regulating the expression of CXCR4 in breast cancer and leukemia [12,13].
Accordingly, the present study analyzed the expression of these molecules and their impact on the survival of patients with DLBCL who received rituximab-based therapy.
METHODS
Patients and treatment
We retrospectively reviewed the medical records of 70 patients with newly diagnosed DLBCL who received R-CHOP at Kyungpook National University Hospital (KNUH) between August 2003 and December 2009. All of the tissues investigated in the present study were obtained from excisional biopsy material. All patients were evaluated using standard laboratory tests, computed tomography (CT), and a unilateral bone marrow (BM) aspirate and biopsy at the time of diagnosis. Additional information was also recorded, including age, gender, performance status, the presence of B symptoms (fever, night sweats, weight loss), the presence of bulky disease (defined as tumor size > 10 cm), the presence of extranodal disease, the presence of BM involvement, the International Prognostic Index (IPI) [14], serum lactate dehydrogenase levels, hemoglobin levels, white blood cell counts, and platelet levels. All of the patients were staged according to the Ann Arbor Staging classification [15] using CT. All of the patients were treated with six cycles of rituximab plus CHOP (R-CHOP), while the patients with bulky disease received six cycles of R-CHOP with radiotherapy.
Immunohistochemistry and scoring of NF-κB and CXCR4 expression
Written informed consent for immunohistochemistry (IHC) was received from the patients, and the study was approved by the Institutional Review Board at KNUH. The tissue sections were deparaffinized in xylene and rehydrated using a graded series of ethanol solutions. A rabbit polyclonal anti-NF-κB p100/p52 antibody (dilution, 1:300; ab31409, Abcam, Cambridge, UK), NF-κB p50 antibody (1:150; sc-114, Santa Cruz Biotechnology, Santa Cruz, CA, USA), IκB kinase α (Ikkα) antibody (1:40; sc-7183, Santa Cruz Biotechnology), and rabbit anti-human CXCR4 polyclonal antibody (1:100; E18444, Spring Bioscience, Pleasanton, CA, USA) were used as primary antibodies. Immunohistochemical staining was carried out according to the manufacturer's protocol with the Ventana BenchMark XT autoimmunostainer (Ventana Medical Systems, Tucson, AZ, USA) using the ultraView kit. Primary antibodies were incubated at 37℃ for 32 minutes, followed by standard Ventana signal amplification, counterstaining with hematoxylin for 4 minutes and staining with a bluing reagent for 4 minutes. Slides were then removed from the immunostainer, mounted and examined by light microscopy.
Interpretation of immunohistochemical analysis of NF-κB and CXCR4 expression
Based on prior studies, breast carcinoma tissue was used as the positive control for NF-κB expression. The tumors were classified into four grades: grade 0, no expression; grade 1, less than 10% expression; grade 2, between 10% and 50% expression; grade 3, more than 50% expression. For statistical reasons, the expression of each of NF-κB p100/p52, NF-κB p50, and Ikkα was analyzed as a dichotomous covariate-namely, NF-κB p100/p52-low (grade 0 and 1) versus NF-κB p100/52-high (grade 2 and 3), NF-κB p50-low (grade 0 and 1) versus NF-κB p50-high (grade 2 and 3), and NF-κB/Ikkα-low (grade 0 and 1) versus NF-κB/Ikkα-high (grade 2 and 3). We defined positive expression for NF-κB when the expression of p100/52, p50, and Ikkα was high. Placenta was used as the positive control for CXCR4. Staining was scored as follows: grade 0, no expression; grade 1, less than 10% expression; grade 2, between 10% and 50% expression; grade 3, more than 50% expression. CXCR4 overexpression (positive) was defined as grade 2 or 3. Two pathologists who were blinded to the patients' characteristics examined all the slides. The average percentage concordance for slides was 90.0%. In discordant cases, each pathologist retested and reviewed the slides again by capturing 10 pieces within a same sample.
Statistical analysis
Descriptive statistics are reported as proportions and medians. The baseline characteristics were compared using the chi-square and Fisher exact tests. A chi-square test was also used to assess the correlation between the levels of the two proteins. Overall survival (OS) was defined as the time from diagnosis to death from any cause. Event-free survival (EFS) was defined as the time from diagnosis to relapse or death from any cause [16]. The OS and EFS were calculated using the Kaplan-Meier test and compared using the log-rank test. Cox's proportional hazard regression model was used for the survival analyses. The analyses were adjusted for age, gender, performance status, stage, and IPI. The hazard ratio and 95% confidence interval were also estimated. A cutoff p value of 0.05 was adopted for all of the statistical analyses. All of the analyses were performed using SPSS version 14 (SPSS Inc., Chicago, IL, USA).
RESULTS
Patients' characteristics
The patients' characteristics are summarized in Table 1. The total cohort included 70 patients. The median age was 60.7 years (range, 17 to 87), and 58.6% were male. Twenty-seven patients (38.6%) had stage III or IV disease at diagnosis. Twenty-three patients (32.9%) were categorized as high or high-intermediate risk according to their IPI. The overall incidence of BM involvement was 5.7%. Seven patients (10.0%) had bulky disease. The characteristics of the germinal center B-cell-like (GCB) and non-GCB groups were similar.
Table 1.
Baseline characteristics
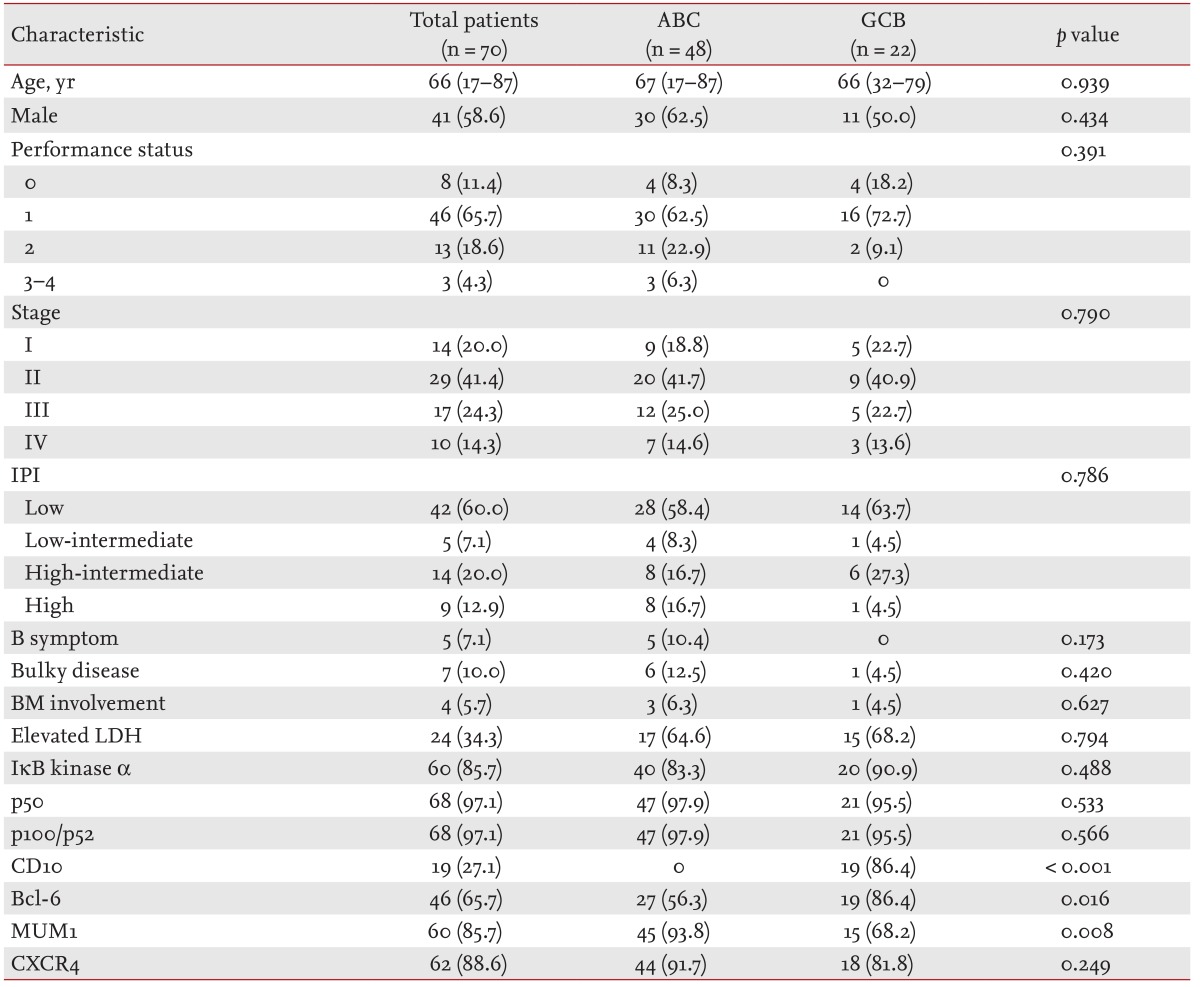
Values are presented as median (range) or number (%).
ACB, activated B-cell-like; GCB, germinal center B-cell-like; IPI, International Prognostic Index; BM, bone marrow; LDH, lactate dehydrogenase.
IHC analysis of NF-κB and CXCR4 expression
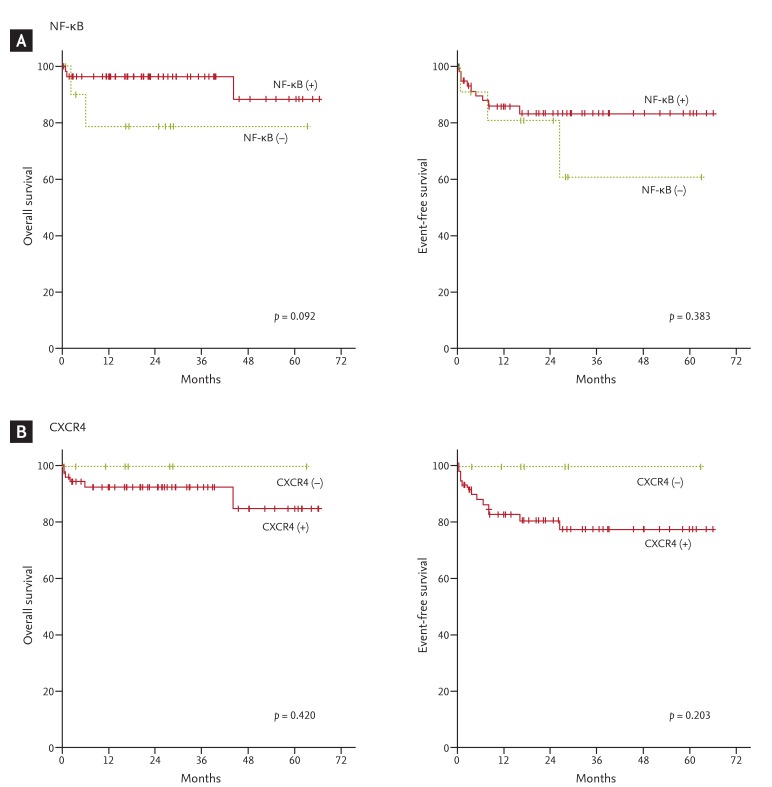
The NF-κB and CXCR4 expression results are shown in Fig. 1. Positive NF-κB expression was found in 59 patients (84.3%) and CXCR4 in 62 patients (88.6%). Regarding the NF-κB and CXCR4 statuses, no significant difference was found between the GCB and non-GCB groups. High NF-κB expression was associated with CXCR4 expression (p = 0.002), and 56 patients (80.0%) showed coexpression (Table 2). With a median follow-up duration of 24.7 months (range, 0.3 to 66.4) among the patients alive at the last follow up, the 5-year OS and EFS rates were 80.0% and 73.7%, respectively. The expression of NF-κB or CXCR4 was not associated with OS or EFS (Fig. 2). On multivariate analysis that included age, gender, performance status, stage, and IPI, no significant association was observed between the grade of NF-κB or CXCR4 expression and survival. The IPI and older age were independent prognostic factors of OS for patients with DLBCL (Table 3).
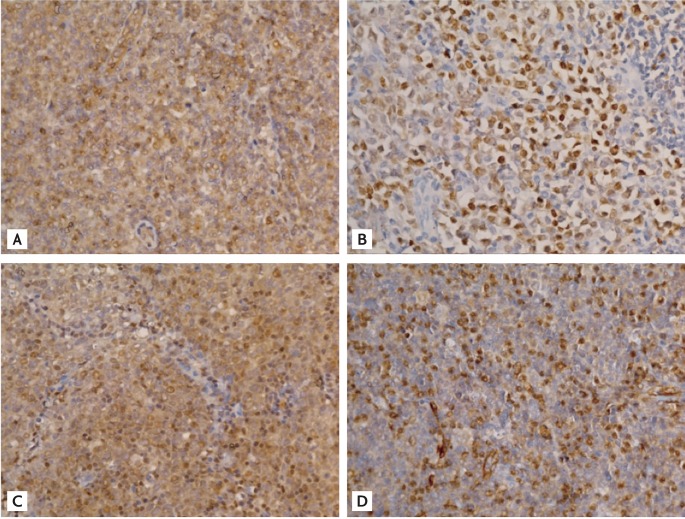
Figure 1.
Representative examples of immunohistochemical staining with (A) anti-nuclear factor κB (anti-NF-κB) p100/p52, (B) NF-κB p50, (C) IκB kinase α, and (D) CXCR4 antibodies (×400).
Table 2.
Correlation between NF-κB and CXCR4 expression
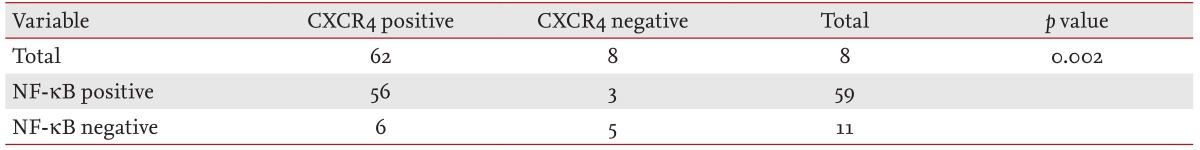
NF-κB, nuclear factor κB.
Figure 2.
Survival curves according to (A) nuclear factor κB (NF-κB) and (B) CXCR4 expression levels.
Table 3.
Univariate and multivariate analysis for factors affecting EFS and OS
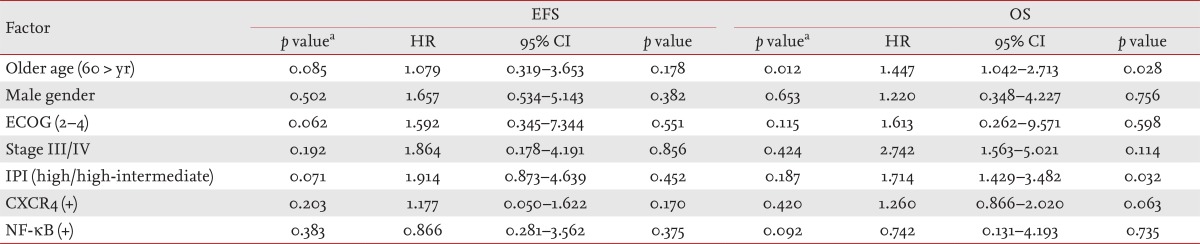
EFS, event-free survival; OS, overall survival; HR, hazard ratio; CI, confidence interval; ECOG, Eastern Cooperation Oncology Group; IPI, International Prognostic Index; NF-κB, nuclear factor κB.
aLog-rank test for univariate analysis.
DISCUSSION
We investigated the relationship between the immunohistochemical expression of NF-κB and CXCR4 with the clinical outcome in patients with DLBCL. The results indicate that NF-κB and CXCR4 are abnormally expressed in DLBCL and coexpressed in 80% of these patients. Although the expression of NF-κB or CXCR4 was not associated with survival for DLBCL patients, it is important to understand the biological and molecular processes of DLBCL.
Because NF-κB is known to play major roles in the growth and metastasis of malignant tumors, many studies have focused on the clinical significance of its expression in tumors to predict the prognosis of solid tumors, including lymphoma [17]. The expression of NF-κB has been reported to be a significant marker of tumor recurrence or reduced survival, and this expression was independent of the conventional clinicopathological factors for DLBCL [8,18]. Furthermore, the activated B-cell subtype of DLBCL, characterized by its pattern of gene expression, shows constitutive activity of Ikk with accumulation of NF-κB proteins in the nucleus [19]. However, the relationship between NF-κB expression and prognosis remains unclear. For example, NF-κB activation in DLBCL is caused by genetic lesions affecting multiple other genes [20]. Additionally, treatment might affect the NF-κB signaling pathway in lymphoma, leading to altered survival in these patients [6]. Saito et al. [21] also reported that rituximab plus chemotherapy improved the survival of nongerminal-center-type untreated DLBCL, leading to increased sensitivity to chemotherapy by inhibiting the NF-κB signaling pathway. Thus, when taken together, the results remain inconsistent, although the findings of the present study differ in that all of the patients received rituximab-based chemotherapy.
The chemokine receptor CXCR4 is thought to be a metastatic indicator and poor prognostic factor in several hematologic and nonhematologic malignancies. Ahn et al. [22] reported that high CXCR4 expression is associated with a poor prognosis in patients with acute myeloid leukemia. In addition, increased CXCR4 levels have been found in several solid tumor types and may portend a worse prognosis [23,24,25]. In particular, CXCR4 has been implicated in regulating the metastasis of breast and prostate cancer and is under the control of NF-κB.
A recent in vitro study demonstrated that NF-κB regulates the motility of breast cancer cells by directly up-regulating the expression of CXCR4. Okera et al. [11] also suggested that CXCR4 expression is associated with the activities of interleukin 8, matrix metalloproteinase 9, and vascular endothelial growth factor, and these factors are often regulated by NF-κB. In the present study, DLBCL patients showed increased levels of NF-κB and CXCR4 coexpression, a finding that is consistent with a previous report. This finding also suggests that clinical studies of DLBCL with coexpression of these molecules should be considered separately from other lymphomas. In the present study, although positive expression of NF-κB seemed to be associated with slightly better survival than negative expression of NF-κB, the overall prognostic impact was not statistically significant. There are several possible explanations for this result. First, the sample size of 70 patients was too small to draw statistically significant conclusions. Second, although IHC is considered a good method for NF-κB testing, various ranges of positivity rates have been reported among studies. In fact, there is no consensus regarding the interpretation of NF-κB expression. Finally, the effect of chemotherapy may also have contributed to the survival outcomes, although no data regarding the relationship between NF-κB expression and the use of chemotherapy have been reported.
In conclusion, NF-κB and CXCR4 were abnormally expressed in DLBCL and were coexpressed in 80% of these patients. In the present study, both NF-κB and CXCR4 were found to be associated with the outcomes of Korean patients with DLBCL. However, further, larger-scale studies to clarify the role of NF-κB and CXCR4 as biomarkers for these patients are warranted.
KEY MESSAGE
Diffuse large B-cell lymphoma (DLBCL) can be subdivided into several subtypes according to the immunochemical features.
Nuclear factor κB (NF-κB) and CXCR4 were coexpressed in 80% of the patients.
The expression of NF-κB and CXCR4 may not be an independent prognostic marker for DLBCL patients treated with rituximab-CHOP.
Acknowledgments
This research was supported by the Kyungpook National University Research Fund, 2010.
Footnotes
No potential conflict of interest relevant to this article was reported.
References
- 1.Coiffier B, Lepage E, Briere J, et al. CHOP chemotherapy plus rituximab compared with CHOP alone in elderly patients with diffuse large-B-cell lymphoma. N Engl J Med. 2002;346:235–242. doi: 10.1056/NEJMoa011795. [DOI] [PubMed] [Google Scholar]
- 2.Hans CP, Weisenburger DD, Greiner TC, et al. Confirmation of the molecular classification of diffuse large B-cell lymphoma by immunohistochemistry using a tissue microarray. Blood. 2004;103:275–282. doi: 10.1182/blood-2003-05-1545. [DOI] [PubMed] [Google Scholar]
- 3.Kwak JY. Treatment of diffuse large B cell lymphoma. Korean J Intern Med. 2012;27:369–377. doi: 10.3904/kjim.2012.27.4.369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Pavan A, Spina M, Canzonieri V, Sansonno S, Toffoli G, De Re V. Recent prognostic factors in diffuse large B-cell lymphoma indicate NF-kappaB pathway as a target for new therapeutic strategies. Leuk Lymphoma. 2008;49:2048–2058. doi: 10.1080/10428190802444176. [DOI] [PubMed] [Google Scholar]
- 5.Johnson PW. New targets for lymphoma treatment. Ann Oncol. 2008;19(Suppl 4):iv56–iv59. doi: 10.1093/annonc/mdn198. [DOI] [PubMed] [Google Scholar]
- 6.Jazirehi AR, Huerta-Yepez S, Cheng G, Bonavida B. Rituximab (chimeric anti-CD20 monoclonal antibody) inhibits the constitutive nuclear factor-{kappa}B signaling pathway in non-Hodgkin's lymphoma B-cell lines: role in sensitization to chemotherapeutic drug-induced apoptosis. Cancer Res. 2005;65:264–276. [PubMed] [Google Scholar]
- 7.Jost PJ, Ruland J. Aberrant NF-kappaB signaling in lymphoma: mechanisms, consequences, and therapeutic implications. Blood. 2007;109:2700–2707. doi: 10.1182/blood-2006-07-025809. [DOI] [PubMed] [Google Scholar]
- 8.Bavi P, Uddin S, Bu R, et al. The biological and clinical impact of inhibition of NF-kappaB-initiated apoptosis in diffuse large B cell lymphoma (DLBCL) J Pathol. 2011;224:355–366. doi: 10.1002/path.2864. [DOI] [PubMed] [Google Scholar]
- 9.Calandra G, Bridger G, Fricker S. CXCR4 in clinical hematology. Curr Top Microbiol Immunol. 2010;341:173–191. doi: 10.1007/82_2010_26. [DOI] [PubMed] [Google Scholar]
- 10.Bertolini F, Dell'Agnola C, Mancuso P, et al. CXCR4 neutralization, a novel therapeutic approach for non-Hodgkin's lymphoma. Cancer Res. 2002;62:3106–3112. [PubMed] [Google Scholar]
- 11.Okera M, Bae K, Bernstein E, et al. Evaluation of nuclear factor kappaB and chemokine receptor CXCR4 co-expression in patients with prostate cancer in the Radiation Therapy Oncology Group (RTOG) 8610. BJU Int. 2011;108(2 Pt 2):E51–E58. doi: 10.1111/j.1464-410X.2010.09884.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Helbig G, Christopherson KW, 2nd, Bhat-Nakshatri P, et al. NF-kappaB promotes breast cancer cell migration and metastasis by inducing the expression of the chemokine receptor CXCR4. J Biol Chem. 2003;278:21631–21638. doi: 10.1074/jbc.M300609200. [DOI] [PubMed] [Google Scholar]
- 13.Scupoli MT, Donadelli M, Cioffi F, et al. Bone marrow stromal cells and the upregulation of interleukin-8 production in human T-cell acute lymphoblastic leukemia through the CXCL12/CXCR4 axis and the NF-kappaB and JNK/AP-1 pathways. Haematologica. 2008;93:524–532. doi: 10.3324/haematol.12098. [DOI] [PubMed] [Google Scholar]
- 14.The International Non-Hodgkin's Lymphoma Prognostic Factors Project. A predictive model for aggressive non-Hodgkin's lymphoma. N Engl J Med. 1993;329:987–994. doi: 10.1056/NEJM199309303291402. [DOI] [PubMed] [Google Scholar]
- 15.Armitage JO. Staging non-Hodgkin lymphoma. CA Cancer J Clin. 2005;55:368–376. doi: 10.3322/canjclin.55.6.368. [DOI] [PubMed] [Google Scholar]
- 16.Cheson BD, Pfistner B, Juweid ME, et al. Revised response criteria for malignant lymphoma. J Clin Oncol. 2007;25:579–586. doi: 10.1200/JCO.2006.09.2403. [DOI] [PubMed] [Google Scholar]
- 17.Nogai H, Dorken B, Lenz G. Pathogenesis of non-Hodgkin's lymphoma. J Clin Oncol. 2011;29:1803–1811. doi: 10.1200/JCO.2010.33.3252. [DOI] [PubMed] [Google Scholar]
- 18.Espinosa I, Briones J, Bordes R, et al. Activation of the NF-kappaB signalling pathway in diffuse large B-cell lymphoma: clinical implications. Histopathology. 2008;53:441–449. doi: 10.1111/j.1365-2559.2008.03139.x. [DOI] [PubMed] [Google Scholar]
- 19.Davis RE, Brown KD, Siebenlist U, Staudt LM. Constitutive nuclear factor kappaB activity is required for survival of activated B cell-like diffuse large B cell lymphoma cells. J Exp Med. 2001;194:1861–1874. doi: 10.1084/jem.194.12.1861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Compagno M, Lim WK, Grunn A, et al. Mutations of multiple genes cause deregulation of NF-kappaB in diffuse large B-cell lymphoma. Nature. 2009;459:717–721. doi: 10.1038/nature07968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Saito B, Shiozawa E, Usui T, et al. Rituximab with chemotherapy improves survival of non-germinal center type untreated diffuse large B-cell lymphoma. Leukemia. 2007;21:2563–2566. doi: 10.1038/sj.leu.2404844. [DOI] [PubMed] [Google Scholar]
- 22.Ahn JY, Seo K, Weinberg OK, Arber DA. The prognostic value of CXCR4 in acute myeloid leukemia. Appl Immunohistochem Mol Morphol. 2013;21:79–84. doi: 10.1097/PAI.0b013e3182606f4d. [DOI] [PubMed] [Google Scholar]
- 23.Yao X, Zhou L, Han S, Chen Y. High expression of CXCR4 and CXCR7 predicts poor survival in gallbladder cancer. J Int Med Res. 2011;39:1253–1264. doi: 10.1177/147323001103900413. [DOI] [PubMed] [Google Scholar]
- 24.Sekiya R, Kajiyama H, Sakai K, et al. Expression of CXCR4 indicates poor prognosis in patients with clear cell carcinoma of the ovary. Hum Pathol. 2012;43:904–910. doi: 10.1016/j.humpath.2011.08.002. [DOI] [PubMed] [Google Scholar]
- 25.Jung SJ, Kim CI, Park CH, et al. Correlation between chemokine receptor CXCR4 expression and prognostic factors in patients with prostate cancer. Korean J Urol. 2011;52:607–611. doi: 10.4111/kju.2011.52.9.607. [DOI] [PMC free article] [PubMed] [Google Scholar]