Abstract
Aim:
To investigate the effects of pyrroloquinoline quinone (PQQ), an oxidoreductase cofactor, on high glucose-induced mouse endothelial cell damage in vitro.
Methods:
Mouse brain microvascular endothelial bEND.3 cells were exposed to different glucose concentrations (5.56, 25 and 40 mmol/L) for 24 or 48 h. The cell viability was examined using MTT assay. Flow cytometry was used to analyze the apoptosis and ROS levels in the cells. MitoTracker Green staining was used to examine the mitochondria numbers in the cells. Western blot analysis was used to analyze the expression of HIF-1α and the proteins in JNK pathway.
Results:
Treatment of bEND.3 cells with high glucose significantly decreased the cell viability, while addition of PQQ (1 and 10 μmol/L) reversed the high glucose-induced cell damage in a concentration-dependent manner. Furthermore, PQQ (100 μmol/L) significantly suppressed the high glucose-induced apoptosis and ROS production in the cells. PQQ significantly reversed the high glucose-induced reduction in both the mitochondrial membrane potential and mitochondria number in the cells. The high glucose treatment significantly increased the expression of HIF-1α and JNK phosphorylation in the cells, and addition of PQQ led to a further increase of HIF-1α level and a decrease of JNK phosphorylation. Addition of JNK inhibitor SP600125 (10 μmol/L) also significantly suppressed high glucose-induced apoptosis and JNK phosphorylation in bEND.3 cells.
Conclusion:
PQQ protects mouse brain endothelial cells from high glucose damage in vitro by suppressing intracellular ROS and apoptosis via inhibiting JNK signaling pathway.
Keywords: diabetes mellitus, pyrroloquinoline quinone, oxidoreductase cofactor, high glucose, endothelial cell, ROS, HIF-1α, JNK
Introduction
Diabetes mellitus (DM) is a common metabolic endocrine disease characterized by an increase in circulating blood glucose due to the failure of the body to produce insulin and/or to efficiently use the insulin. Various acute and chronic DM complications represent a severe threat to human health1,2. The incidence of DM has displayed a gradual upward trend, and this disease has become one of the most challenging health issues of the 21st century. Worldwide, DM affects the health of 230 million people, and this number is projected to reach 350 million by the year 20253.
Vascular disease is the most common complication of DM and provides the pathogenic basis for other chronic complications4,5,6. The pathogenesis of vascular disease is related to the damage of the vascular endothelial cells, the activation of the platelets, and abnormalities in coagulation and fibrinolysis. Among these, endothelial cell damage is the earliest important manifestation of DM vascular disease, and it is also the initial factor in the development of arteriosclerosis7,8. With the occurrence of vascular disease, the damage to the endothelial cells becomes more apparent. The extent of endothelial damage can reflect the degree of DM vascular disease9.
The proper glucose concentration is an important requirement for normal cell growth. A high blood glucose state can damage vascular endothelia through several major mechanisms, one of which is oxidative stress. It has been confirmed that oxidative stress plays a central role in the occurrence of macrovascular and microvascular DM complications, and the early manifestation of oxidative damage to blood vessels results in progressive endothelial dysfunction10,11. The increase in superoxide generation mediated by the mitochondrial electron transport chain under high blood glucose conditions can activate other pathways that are involved in the pathogenesis of DM vascular diseases, such as the polyol pathway and the generation of glycosylation end products (AGEs)12,13. A long-term high blood glucose state in DM patients can induce the increased production of oxygen free radicals through the increased levels of glucose autoxidation and protein glycosylation, as well as through the activation of the polyol pathways, thus reducing the clearance ability of the antioxidant system and initiating oxidative stress14,15. A high glucose concentration induces the production of reactive oxygen species (ROS) in vascular endothelial cells through the actions of oxidants, which leads to the increased expression of inflammation-related factors. These inflammation factors can inhibit the activity of the endothelial form of nitric oxide synthase (eNOS), thus lowering the level of nitric oxide (NO) and leading to vasodilation and, consequently, damage to the vascular endothelia16. In addition to direct oxidative stress damage, AGEs can mediate the increased generation of oxygen radicals through direct or indirect binding to vascular endothelial cell receptors, which leads to decreased NO synthesis and increased synthesis of endothelin (ET-1) in vascular endothelial cells, resulting in damage to the vascular endothelia17.
Pyrroloquinoline quinone (PQQ) is the third water-soluble coenzyme of oxidoreductases that was discovered in the last century, following flavin nucleotide and nicotinamide nucleotide18. As a new oxidoreductase cofactor, PQQ is different from the other redox cofactors, such as the pyridine nucleotides (NAD, NADP) and flavins (FMN, FAD), and is present in nearly all biological tissues. Although there is still debate about whether PQQ is a new type of vitamin, it has been found that PQQ performs many important physiological functions in humans and animals19,20, including roles in nutrition, protecting the heart from ischemia/reperfusion injury, preventing the liver from alcoholic damage21, clearing radicals from the body, increasing the level of nerve growth factor in astrocytes, and inhibiting the production of melanin in melanoma cells22.
PQQ is 50-100 times more effective than ascorbic acid at clearing oxygen radicals23. However, there has been no investigation of the effect of PQQ on high-glucose-damaged endothelial cells. The current study examined the mechanism by which PQQ protects high-glucose-damaged bEND.3 endothelial cells using MTT assays, flow cytometry analysis, DCFH-DA labeling, MitoTracker Green labeling, and Western blot analysis.
Materials and methods
Reagents
The mouse brain microvascular endothelial cell line bEND.3 was purchased from the American Type Culture Collection (ATCC). Dulbecco's modified Eagle's medium (DMEM), trypsin, and fetal bovine serum (FBS) were purchased from Invitrogen (USA). Cell culture plates were purchased from Corning (USA).
MTT [3-(4,5)-dimethylthiazol-2-yl)-3,5-diphenyltetrazolium bromide], 2′,7′-dichlorofluorescein diacetate (DCFH-DA), PQQ, and 𝒟-glucose were purchased from Sigma-Aldrich (USA). Annexin V and propidium iodide (PI) were purchased from Roche. MitoTracker Green and tetramethylrhodamine methyl ester perchlorate (TMRM) were purchased from Invitrogen. The following primers were used in this study: PGC-1α (GenBank code: NM_008904.2): upstream primer P1, 5′-GTGGATGAAGACGGATTGCC-3′ and downstream primer P2, 5′-GACTGCGGTTGTGTATGGGA-3′ NRF-1 (GenBank code: NM_001164226.1): upstream primer P1, 5′-TCTCACCCTCCAAACCCAAC-3′ and downstream primer P2, 5′-CCGTAATGCCTGGGTCCATG-3′. All of the above primers were synthesized by and purchased from Invitrogen and Biotechnology Co Ltd. The PGC-1α PCR resulted in a fragment of 446 bp, whereas the NRF-1 PCR resulted in a 286 bp fragment.
The rabbit anti-human HIF-1α polyclonal antibody and the mouse anti-human actin monoclonal antibody were purchased from Abcam (UK). The rabbit anti-human PGC-1α polyclonal antibody and the mouse anti-human NRF-1 monoclonal antibody were purchased from Santa Cruz. The rabbit anti-p-JNK polyclonal antibody and the rabbit anti-t-JNK polyclonal antibody were purchased from Cell Signaling. The secondary antibodies, including IRDye 800-conjugated affinity-purified goat anti-mouse IgG and IRDye 800-conjugated affinity-purified goat anti-rabbit IgG, were purchased from Odyssey.
MTT examination of bEND.3 cell damage induced by different concentrations of glucose
bEND.3 cells were seeded in 96-well plates at a density of 2×105 cells/mL, and each well contained 100 μL of the cell suspension. After the cells attached to the bottom of the wells, the culture medium was replaced with DMEM containing 10% FBS and glucose at final concentrations of 5.56, 25, and 40 mmol/L. The plates were incubated at 37 °C with 5% CO2. Following 24, 48, or 72 h of incubation, 100 μL of MTT solution (0.5 mg/mL) was added to each well, and the cells were incubated for an additional 4 h at 37 °C with 5% CO2. Then, 100 μL of 20% SDS (cosolvent: 50% DMSO) was added to each well, and the plates were incubated for 24 h at 37 °C. A microplate reader (Bio-Tek, USA) was used to measure the OD value at 570 nm. Each experimental group contained 10 duplicate wells, and the experiments were repeated 3 times.
The effect of different concentrations of PQQ on the viability of high-glucose-damaged bEND.3 cells
bEND.3 cells in log phase were trypsinized with 0.25% trypsin, and the cell density was adjusted to 2×105 cells/mL prior to seeding in a 96-well plate, and each well contained 100 μL of the cell suspension. The following experimental groups were included in this analysis: normal control group (5.56 mmol/L glucose), high-glucose-damaged group (40 or 25 mmol/L glucose), and PQQ protection groups at different concentrations (1, 10, and 100 μmol/L PQQ+40 mmol/L glucose). The plates were incubated at 37 °C with 5% CO2. Following 48 or 72 h of incubation, 100 μL of MTT solution (0.5 mg/mL) was added to each well, and the plates were incubated for an additional 4 h at 37 °C with 5% CO2. Then, 100 μL of 20% SDS (cosolvent: 50% DMSO) was added to each well, and the plates were incubated at 37 °C for 24 h. A microplate reader was used to measure the OD value at 570 nm. Each experimental group contained 10 duplicate wells, and the experiment was repeated 3 times.
The effect of PQQ on the apoptosis of high glucose-damaged bEND.3 cells
bEND.3 cells were seeded in 6-well plates at a density of 2×105 cells/mL, and each well contained 1000 μL of the cell suspension. The following experimental groups were included in this analysis: normal control group (5.56 mmol/L glucose), high glucose-damaged group (40 mmol/L glucose), PQQ protection group (100 μmol/L PQQ+40 mmol/L glucose), JNK inhibitor group (10 μmol/L SP600125+100 μmol/L PQQ+40 mmol/L glucose) and the respective control group (10 μmol/L SP600125+40 mmol/L glucose). The plates were incubated at 37 °C in a 5% CO2 incubator. Following 48 h of incubation, the cells were washed once or twice with PBS, and the cells were trypsinized and suspended in 1× binding buffer. The cell density was adjusted to 1×106 cells/mL, and 100 μL of the cell suspension (1×105 cells) was transferred to a 5 mL centrifuge tube; 5 μL of FITC Annexin V and 5 μL of PI were added to the tube. The cell suspension was gently mixed and incubated at room temperature (25 °C) for 15 min in the dark. A 400 μL aliquot of 1× binding buffer was then added to each tube, and the samples were analyzed by flow cytometry (BD, USA) within 1 h.
The effect of PQQ on the ROS levels in high-glucose-damaged bEND.3 cells
bEND.3 cells were seeded in 6-well plates at a density of 2×105 cells/mL, and each well contained 1000 μL of the cell suspension. The following experimental groups were included in this analysis: normal control group (5.56 mmol/L glucose), high glucose-damaged group (25 or 40 mmol/L glucose), and PQQ protection group (100 μmol/L PQQ+25 or 40 mmol/L glucose). The plates were incubated at 37 °C in a 5% CO2 incubator. Following 48 or 72 h of incubation, the cells were washed twice with PBS for 5 min each. DCFH-DA (final concentration: 50 μmol/L) was added to the wells, and the cells were incubated for an additional 30 min. The fluorescent staining buffer was discarded, and the cells were washed twice with PBS and harvested. A flow cytometer was used to analyze the fluorescence density of each group of cells (excitation: 484 nm, emission: 501 nm). The experiment was repeated 3 times.
The effect of PQQ on the changes in the mitochondria levels of high-glucose-damaged bEND.3 cells
bEND.3 cells were seeded in 96-well plates at a density of 2×105 cells/mL, and each well contained 100 μL of the cell suspension. The following experimental groups were included in this analysis: normal control group (5.56 mmol/L glucose), high glucose-damaged group (25 or 40 mmol/L glucose), and PQQ protection group (100 μmol/L PQQ+25 or 40 mmol/L glucose). The plates were incubated at 37 °C with 5% CO2. Following 48 or 72 h of incubation, the cells were washed twice with pre-chilled PBS. MitoTracker Green (final concentration: 100 nmol/L) was added to the cells in the dark, and the cells were incubated for another 30 min. The cells were then washed 3 times with PBS. A microplate reader (excitation: 490 nm, emission: 516 nm) was used to measure the fluorescence of each group, and the experiment was repeated 3 times.
The effect of PQQ on the expression of HIF-1α and the JNK pathway in high-glucose-damaged bEND.3 cells
bEND.3 cells were seeded in 6-well plates at a density of 2×105 cells/mL, and each well contained 1000 μL of the cell suspension. The following experimental groups were included in this analysis: normal control group (5.56 mmol/L glucose), high glucose damage group (40 mmol/L glucose), PQQ protection group (100 μmol/L PQQ+40 mmol/L glucose), and JNK inhibitor group (100 μmol/L PQQ+40 mmol/L glucose+10 μmol/L SP600125). The plates were incubated at 37 °C in a 5% CO2 incubator. Following 48 h of incubation, the cells were washed twice with PBS.
A 1-mL aliquot of RIPA buffer [150 mmol/L NaCl, 1% NP-40, 0.5% sodium deoxycholate, 0.1% SDS, 50 mmol/L Tris (pH 7.9), 10 mmol/L NaF, PMSF and 1× protease inhibitors (Complete cocktail tablets, Roche)] were added to each well of the 6-well plate. The cell lysates were transferred to 1.5 mL centrifuge tubes and centrifuged at 16 000×g for 30 min. The supernatants were retained, and the protein concentrations were determined using the BCA method. A 5% stacking gel and a 12% resolving gel were prepared, and 50 μg of total protein was loaded into each well. The proteins were separated by electrophoresis and transferred onto a PVDF membrane (Bio-Rad, USA). The blot was blocked for 1 h at room temperature in a TBST buffer (10 mmol/L Tris-HCl, pH 7.5, 150 mmol/L NaCl, and 0.1% Tween-20) containing 5% non-fat milk. Then, a rabbit anti-human HIF-1α and PGC-1α polyclonal antibody (1:500 dilution), rabbit anti-p-JNK polyclonal antibody (1:1000 dilution), rabbit anti-t-JNK polyclonal antibody (1:1000 dilution), or mouse anti-β-actin monoclonal antibody (1:1000) dilution as added, and the blot was incubated overnight at 4 °C. Then, the respective IRDye 800-labeled secondary antibody (1:2000 dilution in PBS) was added to the blot and incubated overnight at 4 °C. The blots ere washed with TBST and scanned using the Odyssey infrared imaging system (Rockland). The relative levels of HIF-1α, PGC-1α, p-JNK, and t-JNK are represented by the ratios of the densities of HIF-1α/β-actin, PGC-1α/β-actin, and p-JNK/t-JNK, and the densities were analyzed using the Quantity One software (Bio-Rad, USA).
Statistical analysis
The statistical software Stata 7.0 was used for the statistical analysis of the experimental results, and one-way analysis of variance was used to determine statistical significance. P<0.05 was considered statistically significant.
Results
MTT examination of bEND.3 cell damage treated with different concentrations of glucose
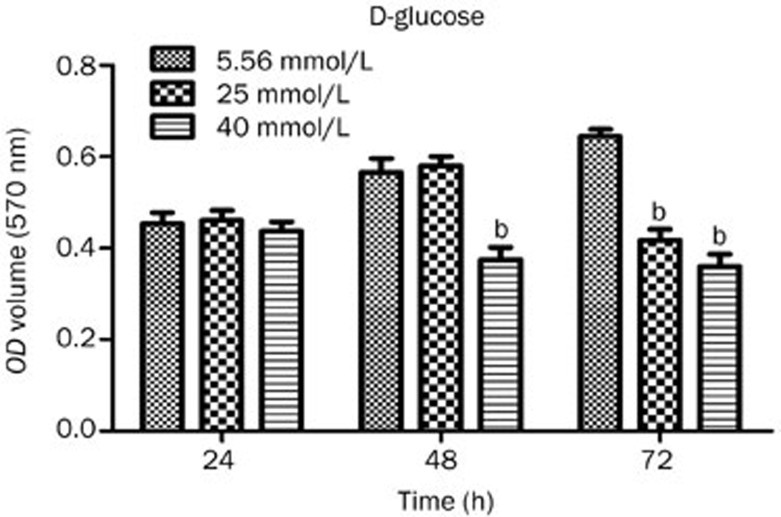
The MTT results showed that there were no significant differences (P>0.05) between the 3 groups of bEND.3 cells that were treated h with 5.56, 25, or 40 mmol/L glucose for 24 h. In contrast, the viability of bEND.3 cells that were treated with 40 mmol/L glucose for 48 h significantly decreased, and the OD value of these cells was significantly different from that of the 5.56 mmol/L glucose group (P<0.05). When bEND.3 cells were treated for 72 h with 25 or 40 mmol/L glucose, the viability of the cells significantly decreased (P<0.05). The statistical results of the MTT assay are shown in Figure 1.
Figure 1.
Statistical results of the MTT assay used to assess bEND.3 cell viability in each group treated with different concentrations of glucose for 24, 48, and 72 h. bP<0.05 compared to the 5.56 mmol/L glucose group.
The effect of different concentrations of PQQ on the viability of high-glucose-damaged bEND.3 cells
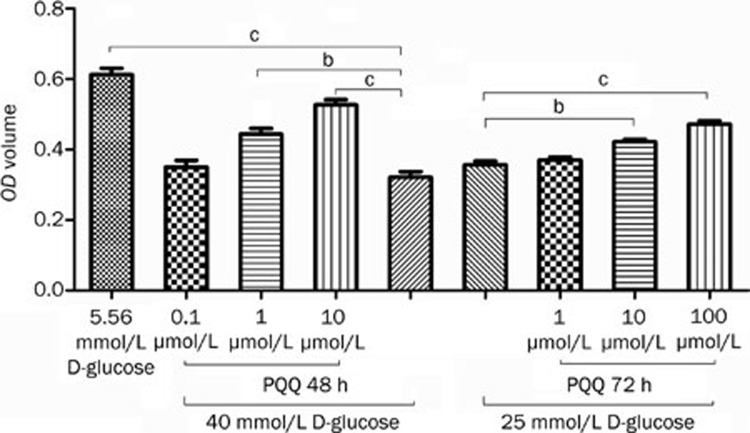
The MTT method was used to monitor the protective effect of PQQ against high glucose damage in bEND.3 cells treated with 25 or 40 mmol/L glucose. The MTT assay results showed that treatment with 10 and 100 μmol/L PQQ could effectively prevent the decrease in bEND.3 cell viability caused by 25 or 40 mmol/L glucose. Within the measured range, the protective effect increased with the increase in PQQ dosage. The detailed results are shown in Figure 2.
Figure 2.
Analysis of the protective effect of different PQQ concentrations on the viability of bEND.3 cells damaged by high glucose (25 or 40 mmol/L glucose). bP<0.05, cP<0.01.
The effect of PQQ on the apoptosis of high-glucose-damaged bEND.3 cells
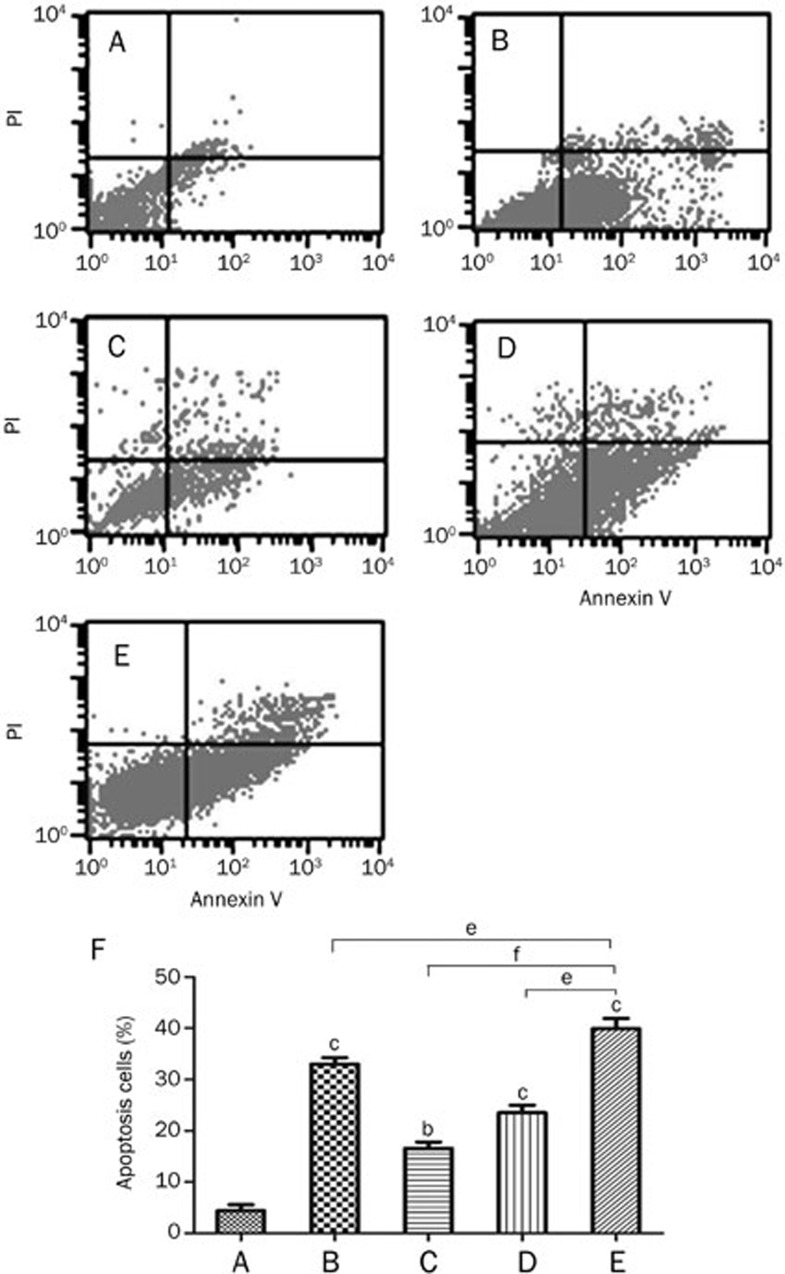
Annexin V-FITC and PI double labeling and flow cytometry were used to analyze the effect of PQQ on the rate of apoptosis in high-glucose-damaged bEND.3 cells. The analysis showed that the apoptosis rate of the normal control group (5.56 mmol/L glucose) was low (4.4%), while the apoptosis rate of the high-glucose-damaged group (40 mmol/L glucose) was high (39.94%). The early apoptosis rate (23.52%) in the PQQ protection group (100 μmol/L PQQ+40 mmol/L glucose) decreased significantly (P<0.05) compared to the high-glucose-damaged group. The proportion of apoptotic bEND.3 cells in the JNK inhibitor group (10 μmol/L SP600125+100 μmol/L PQQ+40 mmol/L glucose) (16.57%) and the respective control group (10 μmol/L SP600125+40 mmol/L glucose) (33.01%) were significantly decreased compared to the rate observed in the high glucose-treated group (P<0.01 and P<0.05). The results shown in Figure 3 demonstrate that PQQ could inhibit the high glucose-induced apoptosis by activating the JNK pathway.
Figure 3.
The effect of PQQ on the apoptosis of high-glucose-damaged bEND.3 cells. (A) normal glucose group (5.56 mmol/L glucose); (B) JNK inhibitor control group (10 μmol/L SP600125+40 mmol/L glucose); (C) JNK inhibitor+PQQ group (10 μmol/L SP600125+100 μmol/L PQQ+40 mmol/L glucose); (D) PQQ group (100 μmol/L PQQ+40 mmol/L glucose); (E) high glucose group (40 mmol/L glucose); (F) statistics of the flow cytometric analysis. bP<0.05, cP<0.01 compared to the normal control. eP<0.05, fP<0.01 compared to high glucose group.
The effect of PQQ on the ROS levels in high-glucose-damaged bEND.3 cells
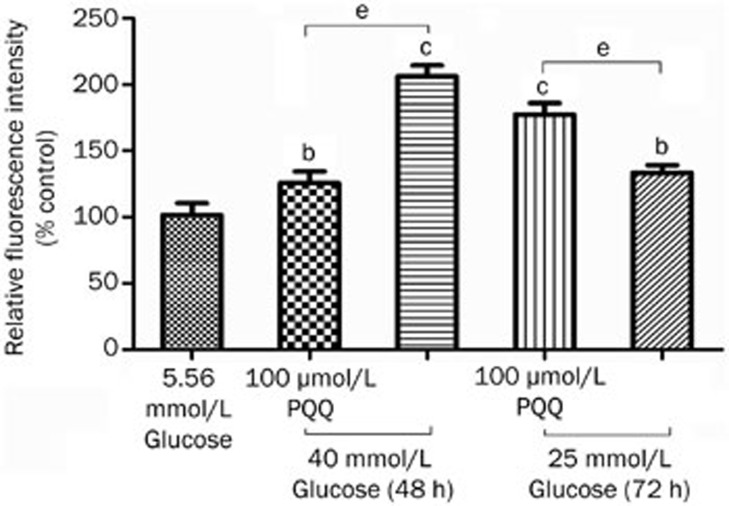
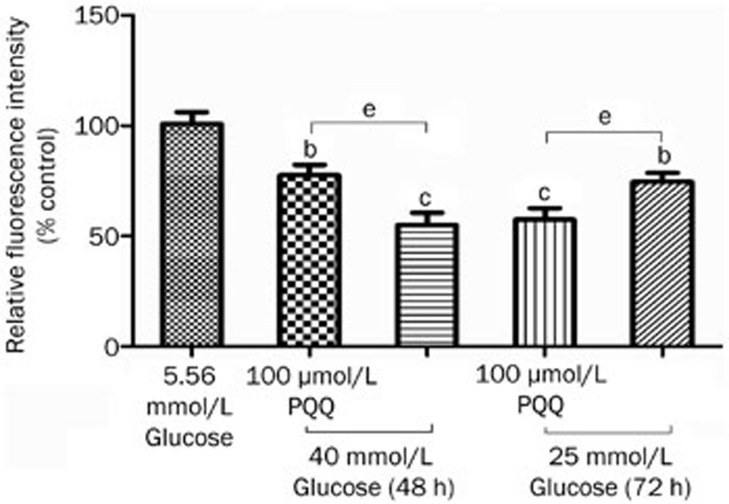
The DCFH-DA molecular probe was used to label peroxide in the bEND.3 cells. After the fat-soluble DCFH-DA enters the cell, it is hydrolyzed by lipase into DCFH. The non-fluorescent DCFH is then oxidized by intracellular ROS to become fluorescent DCF, and a flow cytometer can measure the fluorescence density of the fluorescent product DCF, thus providing a relative quantitative analysis. The results showed that, after 48 or 72 h of damage by high glucose (25 or 40 mmol/L), the intracellular fluorescence intensity significantly increased compared to that of the normal control group, and 100 μmol/L PQQ could effectively antagonize the ROS level increase induced by high glucose (25 or 40 mmol/L) damage (P<0.05). The results are shown in Figure 4.
Figure 4.
The effect of PQQ on the ROS levels in the high-glucose-damaged bEND.3 cells. bP<0.05, cP<0.01 compared to the 5.56 mmol/L glucose group. eP<0.05.
The effect of PQQ on the changes in the mitochondria levels in high-glucose-damaged bEND.3 cells
MitoTracker Green was used to stain the bEND.3 cells, and a microplate reader was used to measure the fluorescence intensity changes inside the mitochondria, which indirectly reflect the amount of cellular mitochondria. The results showed that, 48 or 72 h after high glucose (25 or 40 mmol/L) damage, the intracellular fluorescence intensity significantly decreased compared to that observed in the normal control group (P<0.01). The addition of 100 μmol/L PQQ could significantly antagonize the decrease in the mitochondrial level caused by high glucose (P<0.05). The results are shown in Figure 5.
Figure 5.
The effect of PQQ on the changes of in the mitochondria levels in high-glucose-damaged bEND.3 cells. bP<0.05, cP<0.01 compared to the normal control group. eP<0.05.
The effect of PQQ on the expression of HIF-1α in high-glucose-damaged bEND.3 cells
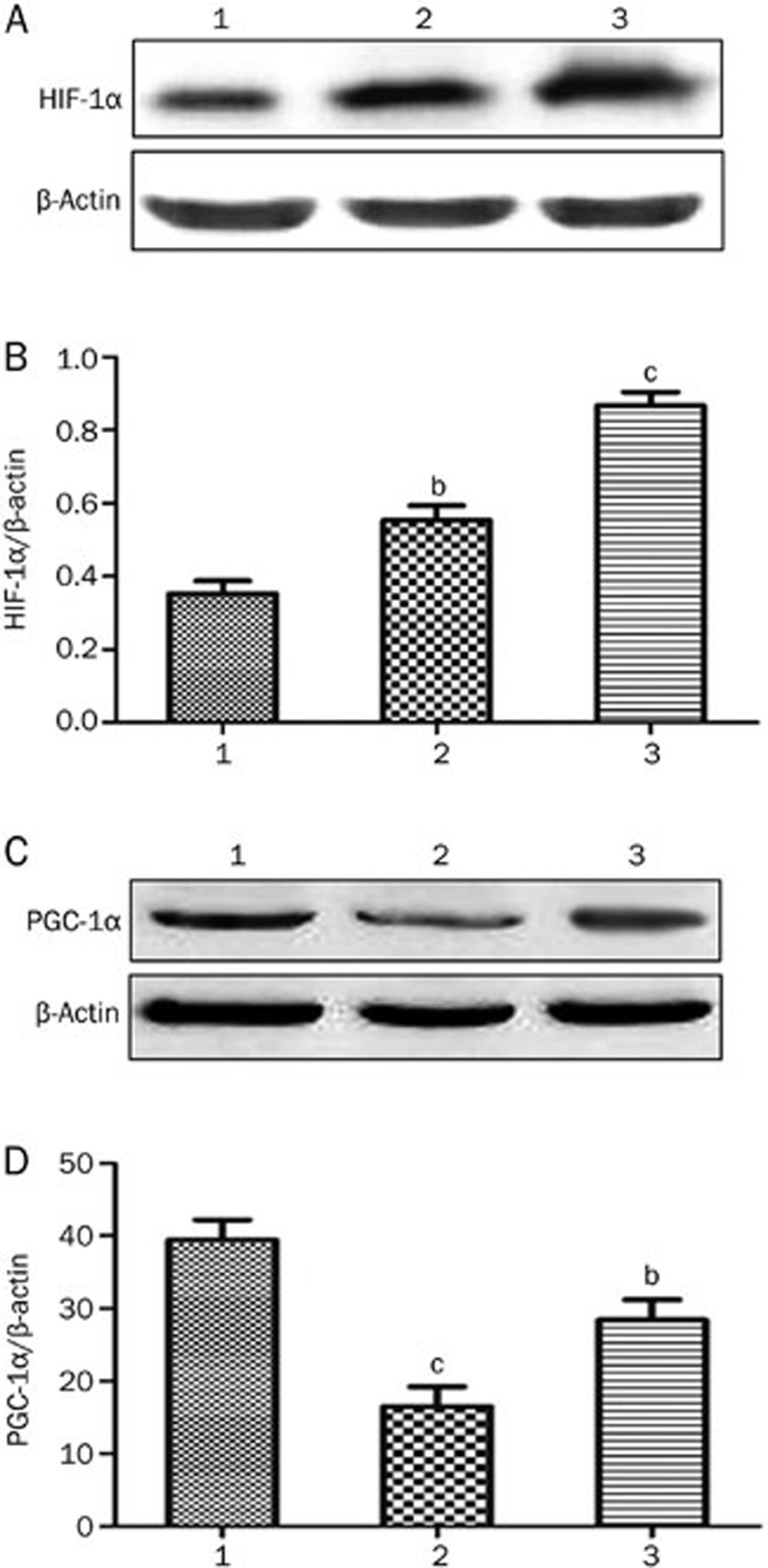
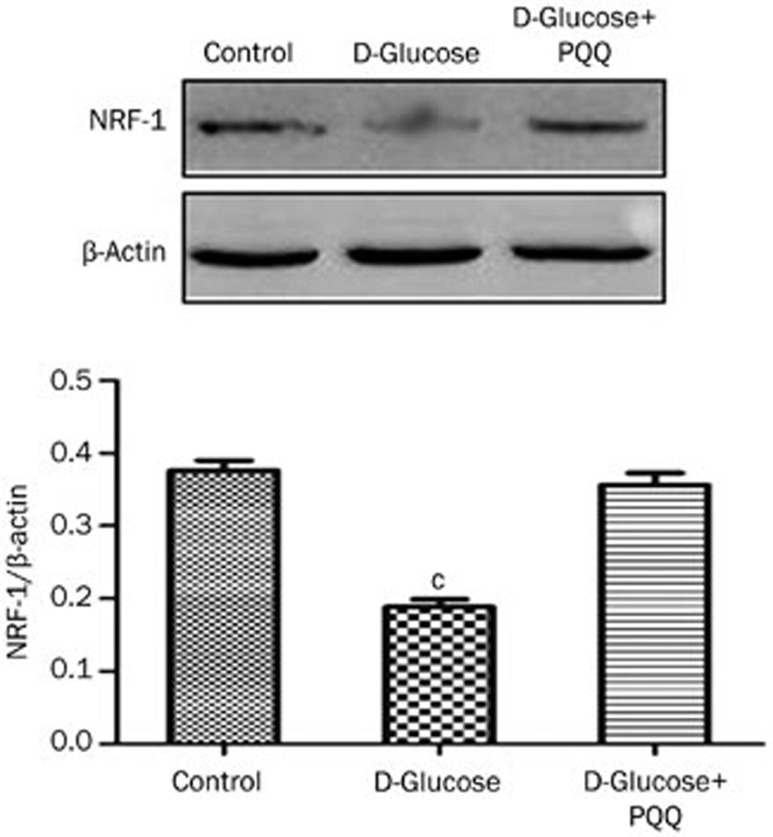
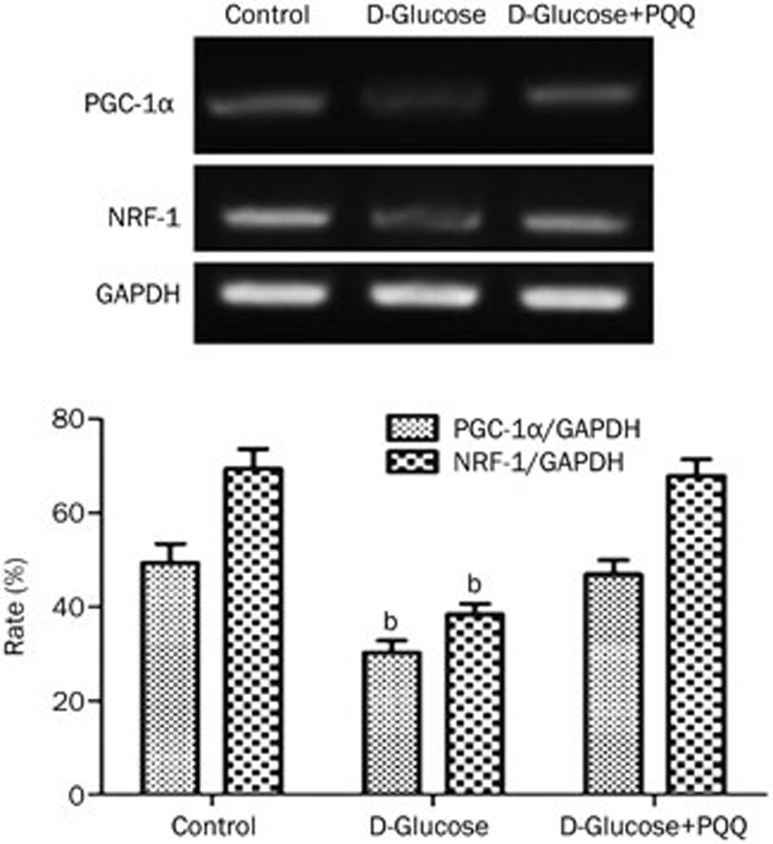
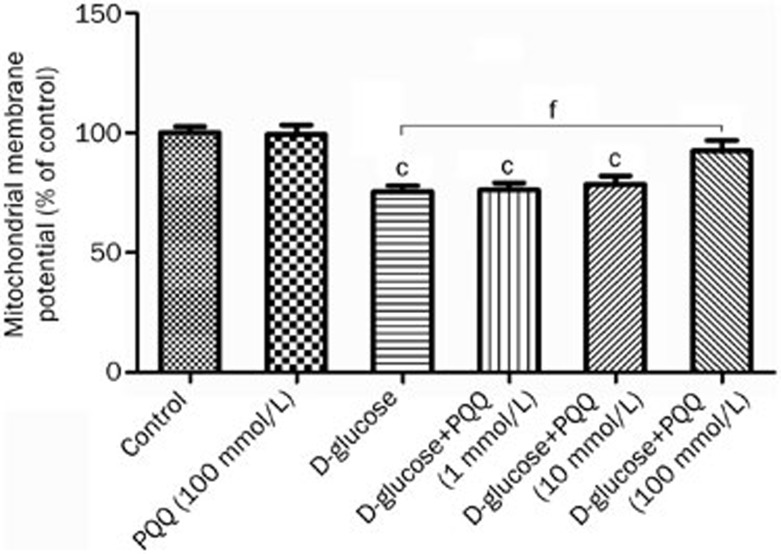
Western blot analysis was used to examine the effect of PQQ on the expression of HIF-1α and PGC-1α in the high-glucose-damaged bEND.3 cells. The results showed that high glucose damage caused an increase in the expression of HIF-1α protein in bEND.3 cells (P<0.05), and HIF-1α protein expression in the bEND.3 cells in the PQQ protection group was even higher. The differences were significant when compared to the normal control group (P<0.01). The expression of PGC-1α and NRF-1 mRNA and protein were decreased in the high-glucose-damaged bEND.3 cells (P<0.05), and the addition of PQQ could promote the expression of PGC-1α and NRF-1 mRNA and protein. The results are shown in Figures 6,7,8. TMRM were used to measure the effects of PQQ on the mitochondrial membrane potential. The results showed that PQQ could increase the mitochondrial membrane potential, and the results are shown in Figure 9.
Figure 6.
(A) Western blot analysis of HIF-1α protein expression in various groups of bEND.3 cells; (B) relative changes in HIF-1α protein expression in various groups of bEND.3 cells; (C) Western blot analysis of PGC-1α protein expression in various groups of bEND.3 cells; (D) relative changes in PGC-1α protein expression in various groups of bEND.3 cells. 1) normal glucose group; 2) high glucose group; 3) PQQ group. bP<0.05, cP<0.01 compared to the normal control group.
Figure 7.
Western blot analysis of NRF-1 protein expression in various groups of bEND.3 cells. cP<0.01 compared to the normal control group.
Figure 8.
RT-PCR analysis of NRF-1 and PGC-1α mRNA levels in various groups of bEND.3 cells. bP<0.05 compared to the normal glucose group.
Figure 9.
The effect of PQQ treatment on mitochondrial membrane potential. cP<0.01 compared to the normal control group. fP<0.01.
The effect of PQQ on the JNK pathway in high-glucose-damaged bEND.3 cells
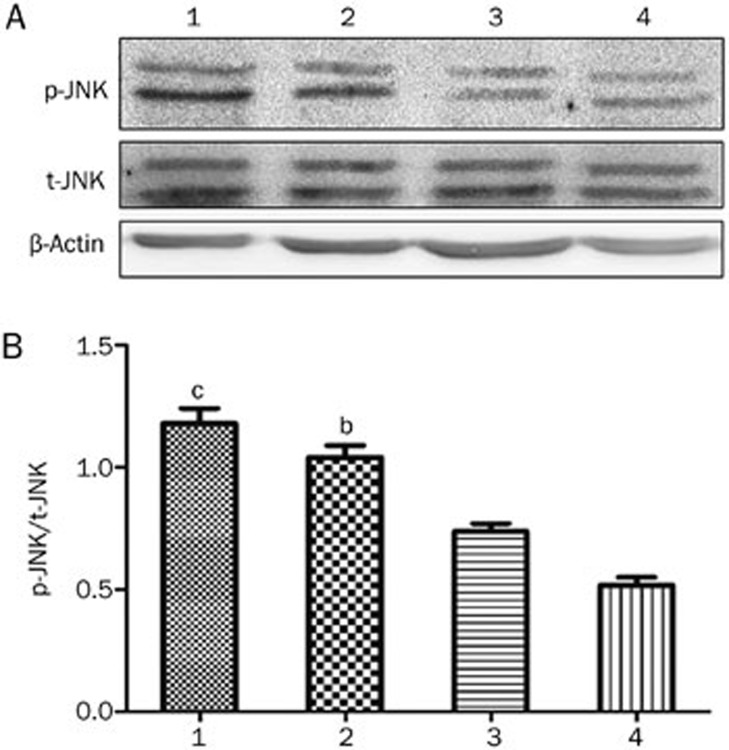
Western blot analysis was used to examine the effect of PQQ on the JNK pathway in the high-glucose-damaged bEND.3 cells. The results showed that high glucose damage did not have an obvious effect on JNK expression in bEND.3 cells (P>0.05). However, high glucose significantly promoted the expression of p-JNK in the cells (P<0.01), whereas PQQ could significantly decrease the expression of p-JNK in the high-glucose-damaged cells, as compared to the normal control group (P<0.05). The phosphorylation level of JNK was further decreased when the JNK inhibitor SP600125 was added to the PQQ protection group.
The results are shown in Figure 10.
Figure 10.
(A) Western blot analysis of p-JNK and t-JNK expression in various groups of bEND.3 cells; (B) relative changes in p-JNK protein expression in various groups of bEND.3 cells. 1, high glucose group; 2, PQQ group; 3, JNK inhibitor group; 4, normal glucose group. bP<0.05, cP<0.01 compared to the normal glucose group.
Discussion
DM is a chronic disease in which high blood glucose persistently attacks the micro- and macro-vessels throughout the body9,24. Microvascular structural changes occur in diabetic patients, including capillary permeability changes, microvascular aneurysm formation, subendothelial deposition, and perimicrovascular fibrosis25,26. The oxidative stress response plays an important role in the mechanisms underlying diabetic complications. In diabetic patients, the main sources of the oxidative stress response include glucose autoxidation, Fenton reactions catalyzed by transition metals, advanced glycosylation, the polyol pathway, mitochondrial respiratory chain deficiency, xanthine oxidase, peroxidase, nitric oxide synthase enzymes, and NADPH oxidase. The oxidative stress response plays an important role in the development and progression of diabetic complications; however, few articles have reported the application of antioxidants in the treatment of diabetes. Therefore, in this study, we used in vitro experiments to investigate the ability of PQQ to antagonize high glucose damage.
In the current study, we used bEND.3 cells to investigate the protective effect of PQQ on endothelial cells, as well as to determine its protection pathway and mechanism. In our experiments, MTT was used to measure changes in the viability of bEND.3 cells that had been treated with different concentrations of glucose for 24 and 48 h. The results indicated that obvious damage occurred in the bEND.3 cells treated with 40 mmol/L glucose for 48 h (P<0.05); therefore, this condition was established as the high-glucose-damaged endothelial cell model of DM. Then, different concentrations of PQQ (1, 10, and 100 μmol/L) were added to the high-glucose-damaged bEND.3 cells, and an MTT assay was used to measure the effects of PQQ on endothelial cell viability following high glucose damage. The results showed that 100 μmol/L PQQ effectively protected the bEND.3 cells from the damage induced by 40 mmol/L glucose (P<0.01).
Superoxide anions in the mitochondria initiate a series of reactions to generate hydrogen peroxide, ferrous ion, hydroxyl radicals, and peroxynitrite, resulting in damage to lipids, proteins, and nucleic acids27. Many functional enzymes in the mitochondria are particularly sensitive to ROS-mediated reactions, leading to changes in ATP synthesis, the disruption of intracellular calcium homeostasis, and the alteration of mitochondrial permeability. All of these changes tend to result in cell death through apoptosis28. Multiple factors can trigger programed cell death in endothelial cells via apoptosis, such as glucose, cytokines, and islet amyloid peptide/free fatty acids. Yoon and colleagues found that trimetazidine reduced the apoptosis rate of human umbilical vein endothelial cells (HUVECs) in diabetic rats29. In the present study, we found that the PQQ protection group (100 μmol/L PQQ+40 mmol/L glucose) exhibited an obvious and significant (P<0.05) decrease in the ratio of early apoptotic cells compared to the high glucose damage group. This result indicates that PQQ can effectively prevent the bEND.3 cell apoptosis caused by high glucose damage, which is consistent with previous reports.
Most glucose that enters the cell is broken down by the oxidative phosphorylation pathway in the mitochondrial respiratory chain. The ROS produced by malfunctioning or damaged mitochondria mainly consist of superoxide anions, which are regarded as the initial source of diabetic complications. Therefore, a reduction of the ROS produced by the mitochondria is considered one method to relieve the effects of DM30. The human body has multiple antioxidant mechanisms to remove the ROS produced during respiration and metabolic processes. The first mechanism involves superoxide dismutase (SOD). The overexpression of copper-zinc superoxide dismutase (CuZnSOD) can protect the experimental animals from renal damage caused by type II diabetes. The second mechanism involves the glutathione peroxidase (GXH-Px) and catalase (CAT) system. These two enzymes catalyze the reduction of hydrogen peroxide into water. In the animal model of type II diabetic renal damage, the overexpression of CAT has protective effects on the experimental animals31. In the current study, we used the molecular probe DCFH-DA to label the peroxides inside of the bEND.3 cells. Flow cytometry was then used to measure the intensity of the DCF fluorescent product, resulting in a relative quantitative analysis. The bEND.3 cells were stained with MitoTracker Green, and a microplate reader was used to measure the changes in fluorescence intensity inside the mitochondria, which indirectly reflected the amount of cellular mitochondria. The results showed that the cellular fluorescence intensity was significantly increased 48 h after high glucose damage, as compared to the normal control group. Moreover, 100 μmol/L PQQ significantly antagonized the increase in the ROS level (P<0.05) and the reduction in the mitochondrial levels caused by high glucose damage (P<0.05). These results suggest that PQQ protects endothelial cells from high glucose damage. It is likely that PQQ protects the cells by decreasing the cellular ROS production and by antagonizing the decrease in the number of mitochondria caused by high glucose damage.
When oxygen supplies are not sufficient, a hypoxic signal is rapidly sent to the nucleus to initiate the expression of certain genes involved in the maintenance of oxygen homeostasis and energy metabolism inside the cells and body. Studies in recent years have shown that HIF-1α is the core regulator that maintains oxygen homeostasis and regulates the expression of a series of genes pertinent to hypoxia; additionally, HIF-1α plays an important role in sensing hypoxia and transmitting hypoxic signals32. Studies by Yan et al found that high glucose can induce the expression of the HIF-1α protein in endothelial cells33. Studies by Zhang et al found that salidroside can protect brain cortex neurons and antagonize CoCl2-induced apoptosis by promoting the expression of the HIF-1α protein34. The results of the present study showed that high glucose damage resulted in the increased expression of the HIF-1α protein in bEND.3 cells (P<0.05), and the expression of HIF-1α protein in the bEND.3 cells of the PQQ protection group was even higher (P<0.01). This result indicates that PQQ antagonizes the apoptosis induced by high glucose damage by promoting HIF-1α expression, which is consistent with previous reports.
JNK is a member of the MAPK superfamily and belongs to the evolutionarily conserved serine/threonine protein kinase family. The JNK pathway is an important signal transduction pathway for cell growth, proliferation, differentiation, and apoptosis. Activated JNK can interact with the N-terminus of c-Jun and phosphorylate the active regions of transcription factors, thereby promoting gene expression and new protein synthesis and regulating cell death, proliferation, differentiation, and other functions36. Studies by Ho et al found that high concentrations of glucose can cause a dramatic increase in the oxidative stress response, JNK activity, and apoptosis in HUVECs37. The current study showed that high glucose damage did not have an obvious effect on JNK expression in bEND.3 cells (P>0.05). However, high glucose levels significantly promoted the expression of p-JNK inside the cell (P<0.01). PQQ could significantly decrease the level of p-JNK expression in the high-glucose-damaged cells compared to the normal control group (P<0.05). When the JNK inhibitor SP600125 was added to the cells of the PQQ protection group, the phosphorylation level was further decreased. The results indicate that the protection of high-glucose-damaged endothelial cells by PQQ might be related to the JNK pathway.
In summary, PQQ can protect bEND.3 cells from high glucose damage in a dose-dependent fashion by reducing apoptosis, clearing intracellular ROS, and antagonizing the reduction of mitochondria. The protective effect of PQQ might be related to the promotion of intracellular HIF-1α expression and the JNK pathway.
References
- Salvado L, Serrano-Marco L, Barroso E, Palomer X, Vazquez-Carrera M. Targeting PPARbeta/delta for the treatment of type 2 diabetes mellitus. Expert Opin Ther Targets. 2012;16:209–23. doi: 10.1517/14728222.2012.658370. [DOI] [PubMed] [Google Scholar]
- Lui MM, Sau-Man M. OSA and atherosclerosis. J Thorac Dis. 2012;4:164–72. doi: 10.3978/j.issn.2072-1439.2012.01.06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garabedian T, Alam S. High residual platelet reactivity on clopidogrel: its significance and therapeutic challenges overcoming clopidogrel resistance. Cardiovasc Diagn Ther. 2013;3:23–37. doi: 10.3978/j.issn.2223-3652.2013.02.06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kishida K, Funahashi T, Shimomura I. Molecular mechanisms of diabetes and atherosclerosis: Role of adiponectin. Endocr Metab Immune Disord Drug Targets. 2012;12:118–31. doi: 10.2174/187153012800493468. [DOI] [PubMed] [Google Scholar]
- Pandey A, Tripathi P, Pandey R, Srivatava R, Goswami S. Alternative therapies useful in the management of diabetes: A systematic review. J Pharm Bioallied Sci. 2011;3:504–12. doi: 10.4103/0975-7406.90103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakao YM, Teramukai S, Tanaka S, Yasuno S, Fujimoto A, Kasahara M, et al. Effects of renin-angiotensin system blockades on cardiovascular outcomes in patients with diabetes mellitus: A systematic review and meta-analysis. Diabetes Res Clin Pract. 2011;96:68–75. doi: 10.1016/j.diabres.2011.11.025. [DOI] [PubMed] [Google Scholar]
- Cubbon RM, Ali N, Sengupta A, Kearney MT. Insulin- and growth factor-resistance impairs vascular regeneration in diabetes mellitus. Curr Vasc Pharmacol. 2012;10:271–84. doi: 10.2174/157016112799959305. [DOI] [PubMed] [Google Scholar]
- Yoshikawa Y, Yasui H. Zinc complexes developed as metallopharmaceutics for treating diabetes mellitus based on the bio-medicinal inorganic chemistry. Curr Top Med Chem. 2012;12:210–8. doi: 10.2174/156802612799078874. [DOI] [PubMed] [Google Scholar]
- Mitic T, Emanueli C. Diabetes-induced epigenetic signature in vascular cells. Endocr Metab Immune Disord Drug Targets. 2012;12:107–17. doi: 10.2174/187153012800493495. [DOI] [PubMed] [Google Scholar]
- Tousoulis D, Briasoulis A, Papageorgiou N, Tsioufis C, Tsiamis E, Toutouzas K, et al. Oxidative stress and endothelial function: therapeutic interventions. Recent Pat Cardiovasc Drug Discov. 2011;6:103–14. doi: 10.2174/157489011795933819. [DOI] [PubMed] [Google Scholar]
- Zhang Y, Du Y, Le W, Wang K, Kieffer N, Zhang J. Redox control of the survival of healthy and diseased cells. Antioxid Redox Signal. 2011;15:2867–908. doi: 10.1089/ars.2010.3685. [DOI] [PubMed] [Google Scholar]
- Thomas MC. Advanced glycation end products. Contrib Nephrol. 2011;170:66–74. doi: 10.1159/000324945. [DOI] [PubMed] [Google Scholar]
- Zong H, Ward M, Stitt AW. AGEs, RAGE, and diabetic retinopathy. Curr Diab Rep. 2011;11:244–52. doi: 10.1007/s11892-011-0198-7. [DOI] [PubMed] [Google Scholar]
- Vassort G, Turan B. Protective role of antioxidants in diabetes-induced cardiac dysfunction. Cardiovasc Toxicol. 2010;10:73–86. doi: 10.1007/s12012-010-9064-0. [DOI] [PubMed] [Google Scholar]
- Shen GX. Oxidative stress and diabetic cardiovascular disorders: roles of mitochondria and NADPH oxidase. Can J Physiol Pharmacol. 2010;88:241–8. doi: 10.1139/Y10-018. [DOI] [PubMed] [Google Scholar]
- Toda N, Nakanishi-Toda M. Nitric oxide: ocular blood flow, glaucoma, and diabetic retinopathy. Prog Retin Eye Res. 2007;26:205–38. doi: 10.1016/j.preteyeres.2007.01.004. [DOI] [PubMed] [Google Scholar]
- Potenza MA, Gagliardi S, Nacci C, Carratu MR, Montagnani M. Endothelial dysfunction in diabetes: from mechanisms to therapeutic targets. Curr Med Chem. 2009;16:94–112. doi: 10.2174/092986709787002853. [DOI] [PubMed] [Google Scholar]
- Rucker R, Chowanadisai W, Nakano M. Potential physiological importance of pyrroloquinoline quinone. Altern Med Rev. 2009;14:268–77. [PubMed] [Google Scholar]
- Holscher T, Schleyer U, Merfort M, Bringer-Meyer S, Gorisch H, Sahm H. Glucose oxidation and PQQ-dependent dehydrogenases in Gluconobacter oxydans. J Mol Microbiol Biotechnol. 2009;16:6–13. doi: 10.1159/000142890. [DOI] [PubMed] [Google Scholar]
- Yamada M, Elias MD, Matsushita K, Migita CT, Adachi O. Escherichia coli PQQ-containing quinoprotein glucose dehydrogenase: its structure comparison with other quinoproteins. Biochim Biophys Acta. 2003;1647:185–92. doi: 10.1016/s1570-9639(03)00100-6. [DOI] [PubMed] [Google Scholar]
- Yakushi T, Matsushita K. Alcohol dehydrogenase of acetic acid bacteria: structure, mode of action, and applications in biotechnology. Appl Microbiol Biotechnol. 2010;86:1257–65. doi: 10.1007/s00253-010-2529-z. [DOI] [PubMed] [Google Scholar]
- Gomez-Manzo S, Contreras-Zentella M, Gonzalez-Valdez A, Sosa-Torres M, Arreguin-Espinoza R, Escamilla-Marvan E. The PQQ-alcohol dehydrogenase of Gluconacetobacter diazotrophicus. Int J Food Microbiol. 2008;125:71–8. doi: 10.1016/j.ijfoodmicro.2007.10.015. [DOI] [PubMed] [Google Scholar]
- Misra HS, Khairnar NP, Barik A, Indira Priyadarsini K, Mohan H, Apte SK. Pyrroloquinoline-quinone: a reactive oxygen species scavenger in bacteria. FEBS Lett. 2004;578:26–30. doi: 10.1016/j.febslet.2004.10.061. [DOI] [PubMed] [Google Scholar]
- Szabo C. Roles of hydrogen sulfide in the pathogenesis of diabetes mellitus and its complications. Antioxid Redox Signal. 2012;17:68–80. doi: 10.1089/ars.2011.4451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tousoulis D, Kampoli AM, Tentolouris C, Papageorgiou N, Stefanadis C. The role of nitric oxide on endothelial function. Curr Vasc Pharmacol. 2011;10:4–18. doi: 10.2174/157016112798829760. [DOI] [PubMed] [Google Scholar]
- Watanabe K, Thandavarayan RA, Harima M, Sari FR, Gurusamy N, Veeraveedu PT, et al. Role of differential signaling pathways and oxidative stress in diabetic cardiomyopathy. Curr Cardiol Rev. 2011;6:280–90. doi: 10.2174/157340310793566145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Onat D, Brillon D, Colombo PC, Schmidt AM. Human vascular endothelial cells: a model system for studying vascular inflammation in diabetes and atherosclerosis. Curr Diab Rep. 2011;11:193–202. doi: 10.1007/s11892-011-0182-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van den Oever IA, Raterman HG, Nurmohamed MT, Simsek S. Endothelial dysfunction, inflammation, and apoptosis in diabetes mellitus. Mediators Inflamm. 2010;2010:792393. doi: 10.1155/2010/792393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoon JW, Cho BJ, Park HS, Kang SM, Choi SH, Jang HC, et al. Differential effects of trimetazidine on vascular smooth muscle cell and endothelial cell in response to carotid artery balloon injury in diabetic rats. Int J Cardiol. 2012;167:126–33. doi: 10.1016/j.ijcard.2011.12.061. [DOI] [PubMed] [Google Scholar]
- Xie Y, You SJ, Zhang YL, Han Q, Cao YJ, Xu XS, et al. Protective role of autophagy in AGE-induced early injury of human vascular endothelial cells. Mol Med Report. 2011;4:459–64. doi: 10.3892/mmr.2011.460. [DOI] [PubMed] [Google Scholar]
- Zhang Z, Liew CW, Handy DE, Zhang Y, Leopold JA, Hu J, et al. High glucose inhibits glucose-6-phosphate dehydrogenase, leading to increased oxidative stress and beta-cell apoptosis. FASEB J. 2010;24:1497–505. doi: 10.1096/fj.09-136572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bento CF, Pereira P. Regulation of hypoxia-inducible factor 1 and the loss of the cellular response to hypoxia in diabetes. Diabetologia. 2011;54:1946–56. doi: 10.1007/s00125-011-2191-8. [DOI] [PubMed] [Google Scholar]
- Yan J, Zhang Z, Shi H. HIF-1 is involved in high glucose-induced paracellular permeability of brain endothelial cells. Cell Mol Life Sci. 2012;69:115–28. doi: 10.1007/s00018-011-0731-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang S, Chen X, Yang Y, Zhou X, Liu J, Ding F. Neuroprotection against cobalt chloride-induced cell apoptosis of primary cultured cortical neurons by salidroside. Mol Cell Biochem. 2011;354:161–70. doi: 10.1007/s11010-011-0815-4. [DOI] [PubMed] [Google Scholar]
- Zhang L, Pang S, Deng B, Qian L, Chen J, Zou J, et al. High glucose induces renal mesangial cell proliferation and fibronectin expression through JNK/NF-kappaB/NADPH oxidase/ROS pathway, which is inhibited by resveratrol. Int J Biochem Cell Biol. 2012;44:629–38. doi: 10.1016/j.biocel.2012.01.001. [DOI] [PubMed] [Google Scholar]
- Nishikawa T, Araki E. Impact of mitochondrial ROS production in the pathogenesis of diabetes mellitus and its complications. Antioxid Redox Signal. 2007;9:343–53. doi: 10.1089/ars.2006.1458. [DOI] [PubMed] [Google Scholar]
- Ho SY, Wu WJ, Chiu HW, Chen YA, Ho YS, Guo HR, et al. Arsenic trioxide and radiation enhance apoptotic effects in HL-60 cells through increased ROS generation and regulation of JNK and p38 MAPK signaling pathways. Chem Biol Interact. 2011;193:162–71. doi: 10.1016/j.cbi.2011.06.007. [DOI] [PubMed] [Google Scholar]