Abstract
Background
We hypothesize that the differences in lung cancer risk in Native Hawaiians (NH), whites and Japanese Americans (JA), may in part be due to variation in the metabolism of 1,3-butadiene (BD), one of the most abundant carcinogens in cigarette smoke.
Methods
We measured two biomarkers of BD exposure, monohydroxybutyl mercapturic acid (MHBMA) and dihydroxybutyl mercapturic acid (DHBMA) in overnight urine samples among 585 NH, JA and white smokers in Hawaii. These values were normalized to creatinine levels. Ethnic-specific geometric means were compared adjusting for age at urine collection, sex, body mass index and nicotine equivalents (a marker of total nicotine uptake).
Results
We found that mean urinary MHBMA differed by race/ethnicity (p=0.0002). The values were highest in whites and lowest in JA. This difference was only observed in individuals with the GSTT1-null genotype (p=0.0001). No difference across race/ethnicity was found among those with at least one copy of the GSTT1 gene (p≥0.72). Mean urinary DHBMA did not differ across racial/ethnic groups.
Conclusions
The difference in urinary MHBMA excretion levels from cigarette smoking across three ethnic groups is in part explained by the GSTT1 genotype. Mean urinary MHBMA levels are higher in whites among GSTT1-null smokers.
Impact
The overall higher excretion levels of MHBMA in whites and lower levels of MHBMA in JA are consistent with the higher lung cancer risk in the former. However, the excretion levels of MHBMA in NH are not consistent with and, thus, unlikely to explain, their high risk of lung cancer.
Keywords: 1,3 Butadiene metabolites; Race/ethnicity
Introduction
Globally, lung cancer is the most common cancer and the leading cause of cancer-related deaths(1). While smoking is recognized as a major causative agent for lung cancer, the risk of lung cancer due to cigarette smoking differs between individuals and among ethnic and racial groups(2, 3). For the same lifetime smoking exposure, compared to whites, Native Hawaiian smokers are at a greater risk of developing lung cancer, whereas Japanese American smokers are at a lower risk of developing the disease(2, 3). In Hawaii, Native Hawaiians have the highest lung cancer incidence and cancer mortality rate compared to all racial/ethnic groups(4). A constitutionally lower CYP2A6 activity has been associated with lower nicotine and carcinogen uptakes in smokers(5). While the lower CYP2A6 activity in Japanese Americans can explain in part the lower lung cancer risk in this group(5), the relatively lower CYP2A6 activity in Native Hawaiians is inconsistent with their high risk for the disease. In addition, environmental or genetic factors that may explain the higher risk of lung cancer in Native Hawaiians have yet to be identified(2, 3). It is possible that the increased lung cancer risk in Native Hawaiian smokers is in part the result of differences in metabolic activation and detoxification of carcinogens present in cigarette smoke(2).
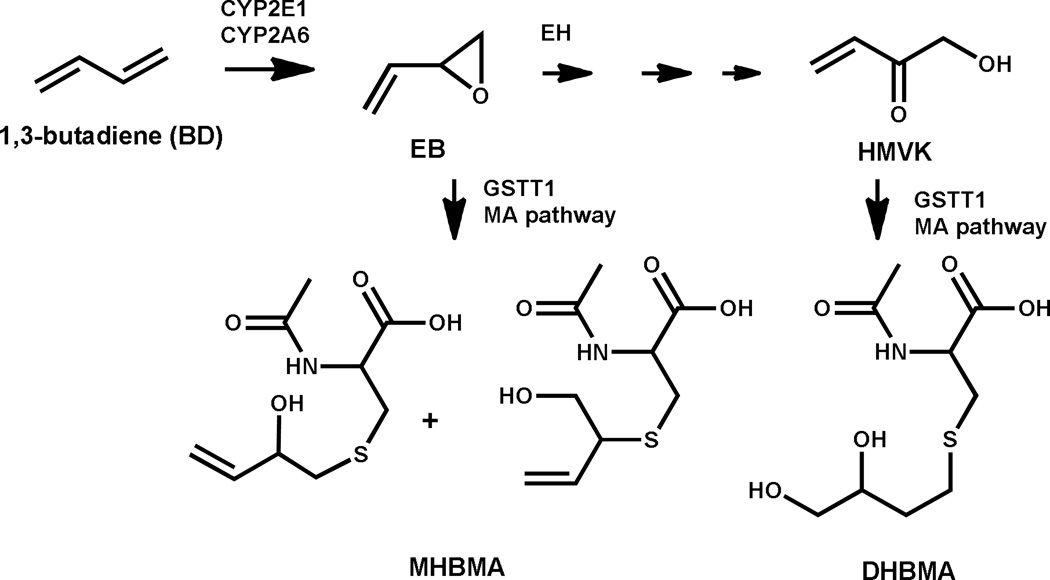
1,3-Butadiene (BD) (Figure 1) is a volatile and colorless gas present in abundant quantities in cigarette smoke(6, 7). BD is a powerful carcinogen in laboratory mice and rats, inducing tumors of the lung, hematopoietic system, heart (hemangiosarcoma), forestomach, Harderian gland, preputial gland, liver, mammary gland, ovary, and kidney in mice(8–10) and the pancreas, testis, thyroid gland, mammary gland, uterus, and Zymbal gland in rats(8, 11). Toxicology risk assessment studies of tobacco constituents found that BD has a very high cancer risk index as compared to other tobacco carcinogens(12). The International Agency for Research on Cancer considers BD to be carcinogenic to humans (Group 1 compound)(13). Human exposure to BD is typically measured by monitoring urinary excretion of two mercapturic acid metabolites, dihydroxybutylmercaturic acid (DHBMA) and monohydroxybutyl mercapturic acid (MHBMA). Urinary concentrations of both DHBMA and MHBMA are elevated in smokers compared to non-smokers(14). A study of Shanghai smokers found that MHBMA was associated with lung cancer but not after adjusting the regression model for cotinine levels, which correlated with MHBMA (Spearman's correlation=0.43)(15). Inhalation exposure to BD is associated with the development of lung tumors in laboratory mice, but not in rats(13). In epidemiologic studies, investigators found that workers occupationally exposed to BD had an increased risk of non-Hodgkin lymphoma and leukemia(13, 16).
Figure 1. Scheme of the metabolism, detoxification of 1,3-butadiene.
Abbreviations: EB, 3,4-epoxy-1-butene; EH, epoxide hydrolase; HMVK, hydroxyl methyl vinyl ketone; GSTT1, Glutathione S-transferase theta-1; MHBMA, monohydroxybutyl mercapturic acid; DHBMA, dihydroxybutyl mercapturic acid; CYP2A6, Cytochrome P450 2A6; CYP2E1, Cytochrome P450 2E1
BD is bioactivated to DNA-reactive epoxides by the cytochrome P450 monooxygenases CYP2E1 and CYP2A6, and epoxide hydrolase (EH) and glutathione S-transferase (GST) mediated reactions detoxify the epoxides(17–19). MHBMA is formed via GSTT1-catalyzed glutathione conjugation of the BD mono-epoxide (Figure 1); therefore, the level of this biomarker is dependent on the amount of the reactive epoxide generated by CYP2E1 and CYP2A6. Studies have found that the distribution of functional genetic variants in BD metabolizing genes, such as EH, CYP2A6, CYP2E1 and GSTT1, substantially differs by racial/ethnic groups (20–24). The GSTT1-null genotype has been reported to be associated with a frequency of BD exposure-induced chromosomal damage(24, 25). Therefore, inter-ethnic and inter-individual differences in the frequency of polymorphisms in genes influencing BD metabolism could lead to higher concentration of carcinogenic epoxides in certain individuals/groups. Although BD is not confirmed to increase lung cancer risk in smokers, in order to identify possible explanations to the lung cancer risk differences across race/ethnicity noted above, we examined the difference in BD metabolism across three racial/ethnic groups found to have disparate risk of lung cancer associated with smoking.
To our knowledge, no previous studies have examined urinary BD metabolites among Asian and Native Hawaiian smokers. One relatively large study of tobacco carcinogen biomarkers reported significantly lower levels of MHBMA and DHBMA in African American smokers compared to whites(14), suggesting that the differences BD metabolites does not explain the higher risk of lung cancer in African Americans. In order to determine whether differences in BD metabolism may contribute to racial disparities of lung cancer risk in Hawaii, we compared the levels of urinary BD-mercapturic acids MHBMA and DHBMA among Japanese American, Native Hawaiian and white smokers, three populations that have been shown to experience significantly different risks of lung cancer associated with smoking.
Materials and Methods
Study population
The details of this study have been previously published(5, 26). In brief, smokers were randomly identified from the Hawaii component of the Multiethnic Cohort study (MEC) for 87% of the subjects and, for the remaining, from the control groups of population-based case-control studies conducted in Hawaii. The MEC is a prospective study of >215,000 men and women of five racial/ethnic groups: African Americans, Japanese Americans, Native Hawaiians, Latinos, and whites, recruited from the state of Hawaii and the Southern California region, primarily Los Angeles County, between 1993 and 1996(27). For the present study, participants must have reported smoking at least 10 cigarettes per day on the baseline questionnaire, have had no previous history of cancer, and have both parents of Japanese or Caucasian ancestry or any amount of Hawaiian ancestry. The same selection criteria were used to recontact controls from various population-based case-control studies completed by some of the authors.
Data collection
For this study, all interviews were conducted in the participants’ home. Information was collected on lifetime tobacco and alcohol use and lung cancer related occupational exposures, as well as on usual diet through a quantitative food frequency questionnaire. Participants were also instructed on how to keep a food record and a diary of all medications and dietary supplements for the 3 days preceding a 12-hour overnight urine collection and a blood draw. Urine collection began between 5 and 9 pm for a period of 12 hours which included all urine passed through the night and the first morning urine. The urine was kept on ice until processing, which occurred within 4 hours of the last sample. Aliquots were subsequently stored in a −80°C freezer until analysis.
In all, a total of 595 participants were included in the study. Eleven participants were excluded because they had no valid DHBMA measurement, had extreme nicotine equivalent values (bottom and top 1%), or had missing cigarettes per day information. The final sample size for this analysis was 584.
Laboratory analysis and quality control
Urinary MHBMA and DHBMA concentrations were determined using the HPLC-ESI−-MS/MS method recently developed in the Tretyakova laboratory (S. Kotapati; personal communication). Briefly, 200 µl urine aliquots were diluted with 200 µl of double distilled water and further acidified with 20 µl of 1N HCl. Following the addition of an internal standard mix (60 ng each of 2H6-MHBMA and 2H7-DHBMA), the samples were vortexed and centrifuged and the supernatant was loaded onto an OASIS HLB (1ml/30 mg) 96 well plate and SPE was performed as described previously. SPE fractions containing MHBMA and DHBMA were dried and reconstituted in 30 µl of 0.1% formic acid. HPLC-ESI−-MS/MS analyses were performed using a Thermo TSQ Vantage mass spectrometer. The analytes were separated on an Agilent Pursuit 3 Diphenyl column (3 µm, 2.0 × 150 mm) using a gradient of 0.1% formic acid and ACN. The complete details of the HPLC-ESI−-MS/MS method for MHBMA and DHBMA are reported elsewhere (S Kotapati; personal communication). The concentrations of MHBMA and DHBMA in the smoker urine samples were determined by comparing the peak areas of the analytes with their corresponding internal standards using standard curves.
Thirty-five blind duplicates were included for quality control measures. The mean coefficients of variation (CVs) for these duplicates were 17.6% and 14.0% for MHBMA and DHBMA, respectively. For samples where the metabolite could not be detected, the value of limit of detection/2 was used (0.1 ng/mL for MHBMA [26 samples] and 2.5 ng/mL for DHBMA [2 samples]). The limit of quantitation (LOQ) for MHBMA and DHBMA are 0.5 and 10 ng/ml, respectively.
Total cotinine (nmol/mL), total nicotine (nmol/mL) and total trans-3'-hydroxycotinine (nmol/mL) were previously measured(5). In brief, total urinary nicotine, cotinine and trans-3'-hydroxycotinine were measured by gas chromatography/mass spectrometry. For total nicotine (free + nicotine N-glucuronide) and total cotinine (free + cotinine N-glucuronide) concentration, the samples were treated with base to cleave the glucuronide conjugates, and the nicotine and cotinine were quantified by gas chromatography/mass spectrometry analysis(28). For total 3-HC (3-HC + its glucuronide), the sample was first treated with h-glucuronidase and then analyzing 3-HC by gas chromatography/mass spectrometry, as described previously(29). The sum of these metabolites accounts for over 80% of nicotine and its metabolites(30) and has been used as a measure of total nicotine uptake. The phenotypic measure of CYP2A6 activity was quantified as total trans-3'-hydroxycotinine (nmol/mL)/total cotinine (nmol/mL)(31).
Genotyping
DNA was extracted from blood leukocytes using a QiaAmp DNA Blood extraction kit (Qiagen, Germantown MD). The samples were genotyped using a predesigned TaqMan GSTT1 copy number assay (Hs00010004_cn) and run on the 7900HT Fast Real-Time System (Life Technologies, Foster City, CA). Copy number counts were calculated using Life Technologies CopyCaller v2.0 software. Approximately 5% of blind duplicates were included for quality control. Test for Hardy Weinberg Equilibrium was met for all three populations (p>0.05).
Statistical Methods
MHBMA and DHBMA levels concentrations in urine were expressed as ng/ mg creatinine (Cr). MHBMA/(MHBMA+DHBMA) metabolic ratio was also calculated as it may be indicative of metabolic processing of BD in a given individual(32). Spearman’s partial correlation coefficients, adjusting for age, sex and race/ethnicity, were computed to examine the correlation between BD metabolites (MHBMA, DHBMA and MHBMA/[MHBMA+DHBMA] ratio) and measures of smoking (cigarettes smoked per day [CPD], and urinary nicotine equivalents [NE] (nmol/mL) = total cotinine (nmol/mL) + total nicotine (nmol/mL) + total trans-3'-hydroxycotinine (nmol/mL), a measure of total nicotine uptake. Multivariable linear models regressed the urinary levels of MHBMA and DHBMA on the following predictors: age at time of urine collection (continuous), sex (when results were not stratified by sex), race (when results were not stratified by race), nicotine equivalents (natural log) and BMI (natural log). All metabolite concentrations were transformed by taking the natural log to better meet model assumptions. To examine ethnic/racial differences, covariate-adjusted geometric means were computed for each ethnic/racial group at the mean covariate vector. Stepwise regression analysis was also used to determine if there were variables that additionally predicted the urinary levels of MHBMA and DHBMA after the five predictors were forced in the model. Variables allowed to compete in the stepwise regression analysis were variables that provided additional information on smoking (e.g. smoking duration), a polymorphism involved in metabolism (i.e. GSTT1), CYP2A6 activity and dietary or lifestyle factors that are metabolized by the same enzymes as BD (e.g. alcohol is metabolized by CYP2E1). We also examined whether the geometric means of these metabolites differed by the GSTT1 polymorphism. The GSTT1 copy number polymorphism was modeled by the number of gene copies (2, 1, or 0) and adjusted for the five previously mentioned predictors. In addition, we performed analyses stratified at the median of CPD, nicotine equivalents and CYP2A6 activity. Here, CYP2A6 activity was quantified as total trans-3'-hydroxycotinine (nmol/mL)/total cotinine (nmol/mL) (31).
Results
Baseline characteristics of this study population have been previously presented (5). On average, Native Hawaiian men and women were the heaviest (median BMI=28 kg/m2), whereas white women were the leanest (median BMI=24 kg/m2). Japanese American women reported smoking the fewest cigarettes per day (median CPD=16), while white men reported smoking the most (median CPD=25). When adjusting for age, CPD and creatinine levels, white men had the highest nicotine equivalents (geometric means=45.2 nmol/mL) and Japanese American women had the lowest nicotine equivalents (geometric means=29.2 nmol/mL). The Spearman’s correlation (r) between CPD and nicotine equivalents were statistically significant in all three ethnic groups (whites: r=0.18; Native Hawaiians: r=0.19; Japanese Americans: r=0.16; p’s<0.03). However, by sex, the correlation between CPD and NE was only statistically significant in females (r=0.21; p=0.0002) and not in males (r=0.05; p=0.39).
The Spearman’s partial correlation coefficients between BD metabolites and nicotine equivalents, stratified by sex and adjusted for age and race/ethnicity are presented in Supplemental Table 1. MHBMA and the MHBMA/(MHBMA+DHBMA) ratio were positively correlated with nicotine equivalents (r=0.15; p=0.0003 and r=0.15; p=0.0002, respectively). This correlation for MHBMA was slightly stronger in females than in males (r=0.19; p=0.0009 and r=0.10; p=0.08, respectively). The correlation between MHBMA and nicotine equivalents was strongest in Native Hawaiians (r=0.21; p=0.001) and was not observed in the other racial/ethnic groups (whites: r=0.13; p=0.08 and Japanese Americans: r=0.08; p=0.26).
Table 1 presents the geometric means of each BD metabolite, stratified by race/ethnicity and sex, and adjusted for age, BMI and nicotine equivalents. Urinary MHBMA differed by race/ethnicity (p=0.0002), with whites excreting the highest MHBMA concentrations, followed by Native Hawaiians and Japanese Americans (mean=6.7, 5.3 and 4.4 ng/mg Cr, respectively). These ethnic differences were observed in each sex (males: p=0.02 and females: p=0.01). Among both sexes, only the MHBMA levels in Japanese Americans were significantly different from those of whites (p’s≤0.003). The geometric means for DHBMA did not differ by race/ethnicity, overall or in either sex (p's>0.15). The findings for the MHBMA/(MHBMA+DHBMA) metabolic ratio was similar to those for MHBMA (p=0.005).
Table 1.
Geometric means (95% confidence limits) for 1,3 butadiene metabolites by race/ethnicity and sex
| MHBMA (ng/mg Cr) | DHBMA (ng/mg Cr) | MHBMA/ (MHBMA + DHBMA) |
||||||
|---|---|---|---|---|---|---|---|---|
| N | geometric means (95% CL)a |
geometric means (95% CL)a |
geometric means (95% CL)a |
|||||
| Allb | 584 | |||||||
| Japanese Americans | 196 | 4.4 | (3.8–5.1)d | 506.8 | (461.9–556.0) | 0.009 | (0.007– 0.010)d | |
| Native Hawaiian | 193 | 5.3 | (4.6–6.2)d | 488.6 | (444.6–536.9) | 0.011 | (0.009–0.012) | |
| Whites | 195 | 6.7 | (5.8–7.8) | 553.0 | (504.4–606.3) | 0.012 | (0.010–0.014) | |
| p-valuec | 0.0002 | 0.17 | 0.005 | |||||
| Men | 284 | 5.3 | (4.7–6.0) | 485.2 | (449.8–523.5)e | 0.011 | (0.010–0.012) | |
| Japanese Americans | 96 | 4.3 | (3.5–5.3)d | 469.9 | (412.7–535.0) | 0.009 | (0.007–0.011)d | |
| Native Hawaiian | 93 | 5.2 | (4.2–6.4) | 460.7 | (402.2–527.7) | 0.011 | (0.009–0.014) | |
| Whites | 95 | 6.6 | (5.4–8.1) | 527.7 | (462.6–601.9) | 0.012 | (0.010–0.015) | |
| p-valuec | 0.02 | 0.31 | 0.12 | |||||
| Females | 300 | 5.5 | (4.9–6.2) | 547.5 | (508.5–589.4)e | 0.010 | (0.009–0.011) | |
| Japanese Americans | 100 | 4.5 | (3.7–5.5)d | 546.5 | (479.7–622.6) | 0.008 | (0.007–0.010)d | |
| Native Hawaiian | 100 | 5.4 | (4.5–6.6) | 518.1 | (456.0–588.6) | 0.010 | (0.009–0.013) | |
| Whites | 100 | 6.9 | (5.6–8.4) | 579.5 | (510.0–658.4) | 0.012 | (0.010–0.014) | |
| p-valuec | 0.01 | 0.48 | 0.04 | |||||
Means are adjusted for age at urine collection, BMI (natural log), nicotine equivalents (natural log)
Also adjusted for sex
P-value is comparing the difference across the three race/ethnicities
P-value comparing the difference with whites is <0.05
P-value comparing difference across sex was significant p= 0.02
The associations of age, race, nicotine equivalents and BMI with urinary MHBMA and DHBMA concentrations are presented in Table 2. Overall, these variables explained only 5.3% of the variance in urinary MHBMA concentrations. Japanese American (p<0.001) and Native Hawaiian (p=0.02) ethnicities vs. white ethnicity were negatively associated with urinary MHBMA, while nicotine equivalents was positively associated with MHBMA concentrations (p≤0.025). We found that only 2.5% of the variance in DHBMA was explained by the above-mentioned covariates. Here, sex was the only significant variable in the model (p=0.03), with male sex being positively associated with DHBMA levels. The same covariates explained 4.9% of the variance in the MHBMA/(MHBMA+DHBMA) ratio. In order to identify other possible determinants of BD metabolites, we conducted a stepwise regression analysis including additional measures of cigarette smoking (e.g,. smoking duration), measures of dietary intake, CYP2A6 activity and GSTT1 polymorphism data. The GSTT1 copy number polymorphism was significantly associated with MHBMA levels (p<0.0001) but not with DHBMA levels (p=0.10). The inclusion of GSTT1 polymorphism data increased the variance for MHBMA explained by the model from 5.3 to 37.1%. In addition, this inclusion resulted in the Japanese ethnicity to be no longer significantly associated with MHBMA levels (p=0.22). In contrast, alcohol consumption was significantly positively associated with DHBMA and negatively associated with MHBMA/(MHBMA+DHBMA) ratio (p’s≤0.002). The addition of alcohol increased the variance for DHBMA explained by the model to 5.8%. For MHBMA/(MHBMA+DHBMA) ratio, the addition of GSTT1 polymorphism and alcohol consumption increased the variance for the ratio explained by the model to 32.6%.
Table 2.
Demographic, smoking and nutritional determinants of 1,3 butadiene metabolites
| Base Model | Including covariates from step-wise regression |
|||||||
|---|---|---|---|---|---|---|---|---|
| Independent variables | Cumulative R2 |
Regression coefficient |
p-value | Cumulative R2 |
Regression coefficient |
p-value | ||
| MHBMA (ng/mg Cr) | Age (years) | 0.00004 | 1.00 | 0.005 | 0.16 | |||
| Japanese American vs. whites | −0.429 | <.0001 | −0.106 | 0.22 | ||||
| Native Hawaiian vs. whites | −0.238 | 0.02 | −0.212 | 0.01 | ||||
| Nicotine Equivalents (nmol/mL) |
0.188 | 0.002 | 0.187 | 0.0002 | ||||
| BMI (kg/m2) | −0.050 | 0.81 | 0.128 | 0.45 | ||||
| Sex | 5.30% | 0.038 | 0.65 | 0.116 | 0.10 | |||
| + GSTT1 (1/0) vs. (1/1)a | −0.495 | <.0001 | ||||||
| (0/0) vs. (1/1) | 37.1% | −1.563 | <.0001 | |||||
| DHBMA (ng/mg Cr) | Age (years) | 0.005 | 0.08 | 0.007 | 0.02 | |||
| Japanese American vs. whites | −0.087 | 0.19 | −0.020 | 0.77 | ||||
| Native Hawaiian vs. whites | −0.124 | 0.07 | −0.072 | 0.29 | ||||
| Nicotine Equivalents (nmol/mL) |
0.001 | 0.97 | 0.238 | 0.54 | ||||
| BMI (kg/m2) | −0.057 | 0.67 | 0.025 | 0.85 | ||||
| Sex | 2.50% | 0.121 | 0.03 | 0.181 | 0.001 | |||
| + alcohol (g/1000kcal/day)a | 5.80% | 0.010 | <0.0001 | |||||
|
MHBMA/ (MHBMA + DHBMA) |
Age (years) | −0.005 | 0.29 | −0.002 | 0.68 | |||
| Japanese American vs. whites | −0.338 | 0.001 | −0.097 | 0.29 | ||||
| Native Hawaiian vs. whites | −0.117 | 0.27 | −0.154 | 0.09 | ||||
| Nicotine Equivalents (nmol/mL) |
0.186 | 0.003 | 0.151 | 0.004 | ||||
| BMI (kg/m2) | 0.012 | 0.96 | 0.076 | 0.67 | ||||
| Sex | 4.90% | −0.078 | 0.36 | −0.088 | 0.24 | |||
| + GSTT1 (1/0) vs. (1/1)a | −0.486 | <.0001 | −0.458 | <.0001 | ||||
| (0/0) vs. (1/1) | −1.424 | <.0001 | 30.5% | −1.427 | <.0001 | |||
| +alcohol (g/1000kcal/day)a | −0.0049 | <.0001 | 32.6% | −0.005 | <.0001 | |||
Additional predictor identified through stepwise regression.
Variables allowed to compete in the stepwise regression: GSTT1 copy number polymorphism, CYP2A6 activity, smoking duration (years) and intakes of cruciferous vegetable, total fruits, caffeine, green leafy vegetables, total vegetables, alcohol, processed meats and soy.
The genotype distribution for the GSTT1 copy number polymorphism by race/ethnicity can be found in Table 3. There was a greater frequency of null genotypes (0/0) among Japanese Americans (45%) and Native Hawaiians (24%) than whites (25%). The frequencies are similar to that of the previous literature(33). We found that the urinary MHBMA and MHBMA/(MHBMA+DHBMA) ratio levels differed by the GSTT1-null polymorphism (p<0.0001), where the carriers of the null genotype (0/0) had lower urinary MHBMA levels than those who have one copy of the GSTT1 gene (1/0), who in turn excreted less MHBMA than individuals with two copies of the gene (1/1) (geometric means= 11.1, 6.7, 2.3 ng/mg Cr, respectively; p-trend < 0.0001) (Table 3). This trend was consistently observed across racial/ethnic groups (p’s<0.0001). In addition, among the GSTT1-null carriers, whites had statistically significant higher urinary MHBMA levels than the other racial/ethnic groups considered here (p=0.0001). Among the GSTT1-null carriers, the racial/ethnic difference remained even after adjusting for CYP2A6 activity (p=0.0001). Urinary DHBMA levels did not appear to differ by GSTT1 copy number genotype (p≥0.05).
Table 3.
Geometric means (95% CI) of BD metabolites stratified by GSTT1 CNV and race/ethnicity (adjusted for age and sex)
|
GSTT1 copy number genotype |
All | Whites | Native Hawaiians | Japanese Americans | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| N | Geometric means (95% CL)a |
N | Geometric means (95% CL)a |
N | Geometric means (95% CL)a |
N | Geometric means (95% CL)a |
p-value | |||||
| MHBMA | |||||||||||||
| 1/1 | 128 | 11.1 | (9.5–13.0) | 59 | 11.8 | (9.5–14.5) | 49 | 10.5 | (8.3–13.3) | 20 | 11.1 | (7.7–15.9) | 0.79 |
| 1/0 | 271 | 6.7 | (6.1– 7.4) | 86 | 6.6 | (5.6– 7.9) | 97 | 6.6 | (5.6– 7.8) | 88 | 6.9 | (5.8– 8.3) | 0.92 |
| 0/0 | 184 | 2.3 | (2.1– 2.6) | 49 | 3.4 | (2.7– 4.3) | 47 | 1.7 | (1.3– 2.1)b | 88 | 2.2 | (1.9– 2.7)b | 0.0001 |
| p-value | <.0001 | <.0001 | <.0001 | <.0001 | |||||||||
| DHBMA | |||||||||||||
| 1/1 | 128 | 529.5 | (467.2–600.1) | 59 | 564.5 | (478.5–665.9) | 49 | 522.1 | (434.7–627.0) | 20 | 503.7 | (379.3–668.9) | 0.73 |
| 1/0 | 271 | 535.8 | (496.1–578.7) | 86 | 536.3 | (467.8–614.9) | 97 | 509.4 | (447.0–580.5) | 88 | 563.1 | (491.1–645.5) | 0.59 |
| 0/0 | 184 | 478.2 | (433.8–527.2) | 49 | 573.0 | (477.5–687.7) | 47 | 417.9 | (346.8–503.6)b | 88 | 456.6 | (398.6–523.1) | 0.05 |
| p-value | 0.181 | 0.818 | 0.163 | 0.099 | |||||||||
| MHBMA/(MHBMA + DHBMA) ratio | |||||||||||||
| 1/1 | 128 | 0.020 | (0.017–0.024) | 59 | 0.020 | (0.016–0.025) | 49 | 0.019 | (0.015–0.025) | 20 | 0.021 | (0.015–0.032) | 0.91 |
| 1/0 | 271 | 0.012 | (0.011–0.014) | 86 | 0.012 | (0.010–0.015) | 97 | 0.013 | (0.011–0.015) | 88 | 0.012 | (0.010–0.015) | 0.90 |
| 0/0 | 184 | 0.005 | (0.004–0.006) | 49 | 0.006 | (0.005–0.008) | 47 | 0.004 | (0.003–0.005)b | 88 | 0.005 | (0.004–0.006) | 0.08 |
| p-value | <.0001 | <.0001 | <.0001 | <.0001 | |||||||||
Adjusted for age, sex, BMI (natural log) and log-nicotine equivalents (natural log)
P-value <0.05 when compared to whites
We examined whether MHBMA levels differed across race/ethnicity by strata of low (≤median) and high (>median) smoking quantity (CPD or nicotine uptake) and CYP2A6 activity (Supplemental Table 2). After adjusting for age, sex, BMI, nicotine equivalents (if applicable) and GSTT1 copy number polymorphism, the ethnic differences were more marked among heavier smokers measured by CPD (p=0.01). However, these racial/ethnic differences were not present when using the measure of nicotine equivalents (p>0.05). The MHBMA levels were statistically significantly lower among heavy smoking or low CYP2A6 activity Native Hawaiians than whites (p<0.05).
Discussion
To the best of our knowledge, this is the first study to compare the urinary concentrations of MHBMA and DHBMA in Native Hawaiian and Japanese American smokers. In this large multiethnic sample of Native Hawaiian, Japanese American, and white smokers in Hawaii, we found that urinary MHBMA differed by race/ethnicity after adjusting for smoking dose (nicotine equivalents), with higher levels in whites, intermediate levels in Native Hawaiians, and lower levels in Japanese Americans. The difference in mean MHBMA excretion between Japanese Americans and whites was explained by ethnic/racial variation in the frequency of the GSTT1-null polymorphism. However, the difference between Native Hawaiians and whites in mean MHBMA excretion remained after adjustment for GSTT1 genotype. GSTT1 genotype and nicotine equivalents were the strongest predictors of MHBMA levels. Urinary DHBMA levels differed by sex; however, they did not appear to differ by racial/ethnic group. Finally, alcohol consumption appeared to be the strongest additional predictor of DHBMA levels.
BD is a carcinogenic gas widely used in the chemical industry and also found in cigarette smoke, motor vehicle exhaust, and wood smoke. Upon inhalation, BD is metabolically activated by CYP2E1 and CYP2A6 to form 3,4-epoxy-1-butene (EB) (Figure 1)(17, 34, 35). In a small in vitro study, the investigators found that EB can be detoxified by epoxide hydrolase (EH)(36), which is the major detoxification pathway for this epoxide in rats, and likely plays a significant role in humans. EH mediated hydrolysis of EB and further biotransformation of the resulting diol by alcohol dehydrogenase (ADH) result in the formation of hydroxyl methyl vinyl ketone (HMVK)(37). Alternatively, EB and HMVK can be conjugated with glutathione and excreted in urine as the corresponding mercapturic acids, MHBMA and DHBMA(38). If not detoxified, EB acts as a direct mutagen via the formation of promutagenic DNA adducts or can be further bioactivated to an even more genotoxic diepoxide(39).
Urinary MHBMA has been widely used in the literature as a biomarker of BD exposure and as a biomarker of metabolic activation used to assess cancer risk(15, 38, 40–43). DHBMA has also been previously employed as a biomarker for BD exposure in biomonitoring studies(38, 40–43). The molar ratio MHBMA/(DHBMA+MHBMA) may be indicative of BD metabolic processing in a given individual and may represent a fraction of non-hydrolyzed BD monoepoxide potentially available for binding to biomolecules(32, 41). Urinary DHBMA appears to be a suitable biomarker of occupational exposure to BD(24). However, it remains unclear whether, in smokers, DHBMA or MHBMA/(DHBMA+MHBMA) ratio are effective measures of cigarette smoking-related BD exposure or BD metabolism, respectively. DHMBA levels do not decrease upon smoking cessation(44) and the levels in smokers are on average only 30% higher than the levels in non-smokers(14). It has been speculated that there may be other sources of DHBMA exposure that have not yet been identified(44). A study among Chinese rubber factory workers found that DHBMA levels correlated with airborne BD levels(25). In our stepwise regression analyses we did find that daily alcohol consumption (g/1000kcal/day) was positively associated with DHBMA levels. While no known mechanism exists for alcohol conversion to DHBMA, alcohol use may contribute to DHBMA levels by inducing BD-metabolizing enzymes, such as CYP2E1 (33). Alcohol use is highly correlated with tobacco use and smoking quantity(45), and ADH catalyzes the conversion of EB-diol to HMVK, the metabolic precursor of DHBMA (Figure 1)(17). In contrast, the level of MHBMA drops precipitously after smoking(44) and the average level in smokers is 12-fold higher than in non-smokers. In the present study, we observed no correlation between DHBMA and nicotine equivalents. Instead, a modest correlation between MHBMA and nicotine equivalents was observed (r=0.15) similar to the correlation of 0.21 reported by Roethig et al. in a study of more than 3,500 smokers(14). In contrast to MHBMA, our findings support prior findings that DHBMA and the MHBMA/(DHBMA+MHBMA) ratio may not be good biomarkers for smoking-related BD exposure(44).
We found that white smokers had the highest urinary MHBMA levels, while Japanese American smokers had the lowest levels (Table 2). This relationship was present even after adjusting for nicotine equivalents, suggesting that whites are exposed to a greater quantity of BD from cigarette smoking or metabolize BD to MHBMA more efficiently than Japanese Americans. In contrast, Native Hawaiians, who are at higher risk of lung cancer than whites for the same quantity of cigarette smoked(3), did not have higher MHBMA levels compared to whites (Table 2). Instead, we found that, when compared to whites, their MHBMA levels were significantly lower in this group. Our findings suggest that BD exposure does not explain the higher lung cancer risk in Native Hawaiians. This is similar to a study conducted among African Americans and whites, where African Americans, who have been shown to have higher risk of the disease, were found to have lower levels of MHBMA(14).
We found that MHBMA and MHBMA/(MHBMA+DHBMA) ratio levels were influenced by GSTT1 copy number polymorphism, with null carriers excreting lower MHBMA levels than those with one or two copies of this gene. Our observation that MHBMA levels were affected by GSTT1 copy number polymorphsim (Table 3) is consistent with the requirement for GST in the formation of MHBMA. Our findings show that the GSTT1-null carriers would have decreased enzymatic function, leading to less conjugation of EB and the reduced urinary excretion of MHBMA (38). In a study of workers exposed to BD, the investigators found lower MHBMA/(MHBMA+DHBMA) ratio values among those who were GSTT1-null as opposed to those who were GSTT1 positive(43). In contrast, we did not find a difference in DHBMA levels across the GSTT1 genotypes. Among the GSTT1-null carriers, urinary MHBMA was highest in whites, followed by Japanese Americans and Native Hawaiians. This difference across race/ethnicity does not appear to be a result of CYP2A6 activity, as the significant associations remained even after adjustment for this phenotype. However, among the GSTT1-null carriers, racial/ethnic differences may be due to the difference of CYP2E1 enzyme activity, reflecting greater metabolism of BD to the EB(38).
To date, this is the largest study examining BD urinary metabolites across three different racial/ethnic groups, specifically whites, Japanese Americans and Native Hawaiians. This study had a number of strengths. The large sample size and the multiethnic population with well-characterized epidemiologic data enabled us to efficiently adjust for multiple confounders. One possible concern may be residual confounding by smoking dose. To address this concern we have adjusted for nicotine equivalents, a measure of total nicotine uptake, which has been found to be a better measure of smoking quantity than cigarettes per day(46). Also, due to the racial admixture among the Native Hawaiian population, we may have had lower power to detect differences between this group and whites. The CVs of 17.6 and 14.0% in the blind duplicate samples may be of some concern. This lack of reproducibility would likely result in non-differential misclassification and loss of statistical power as the laboratory technician was unaware of the sample sex, race/ethnicity and smoking dose.
In conclusion, the BD metabolite, MHBMA, was found to be lower in Japanese American and Native Hawaiian smokers as compared to whites. The overall lower levels of MHBMA in Japanese Americans is partially explained by the relatively high prevalence of GSTT1-null genotype in this ethnic group. However, among GSTT1-null individuals, MHBMA levels were found to be higher in whites than in Japanese Americans, and the higher exposure of whites to genotoxic BD metabolites may contribute to the higher lung cancer risk in this group, relative to Japanese Americans. The relatively low MHBMA levels in Native Hawaiians do not support a role for BD exposure and its metabolic activation as an explanation for the higher risk of lung cancer in Native Hawaiians. Although these findings need to be confirmed with a study examining the association of this metabolite and lung cancer risk, alternative explanations for Native Hawaiians’ high lung cancer risk should also be investigated.
Supplementary Material
Acknowledgements
The authors thank Dr. Stephen Hecht for contributing MHBMA and DHBMA standards that were used in the initial HPLC-ESI-MS method development. The authors gratefully acknowledge the time and efforts of all the study participants.
Financial Support This study was funded by NIH grants 5P01CA138338 (P.I.: S. Hecht; Project Leaders: L. Le Marchand, S.E. Murphy, N. Tretyakova; Co-Investigators: S.L. Park, S. Kotapati, L.R Wilkens, M. Tiirikainen) and R01 CA85997 (PI: L. Le Marchand; Co-Investigator: S.E. Murphy, L.R. Wilkens). This work is also supported in part by NIH grants P30 CA014089 to the USC Norris Comprehensive Cancer Center (S.L. Park) and P30 CA071789-13 to the UH Cancer Center Genomics Shared Resource (M. Tiirikainen). The MEC study is supported by UM1 CA164973 (L. Le Marchand and L.R Wilkens).
Footnotes
Conflict of Interests: none to disclose
References
- 1.Ferlay J, Shin H-R, Bray F, Forman D, Mathers C, Parkin DM. Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. International Journal of Cancer. 2010;127:2893–2917. doi: 10.1002/ijc.25516. [DOI] [PubMed] [Google Scholar]
- 2.Le Marchand L, Wilkens LR, Kolonel LN. Ethnic differences in the lung cancer risk associated with smoking. Cancer EpidemiolBiomarkers Prev. 1992;1:103–107. [PubMed] [Google Scholar]
- 3.Haiman CA, Stram DO, Wilkens LR, Pike MC, Kolonel LN, Henderson BE, et al. Ethnic and Racial Differences in the Smoking-Related Risk of Lung Cancer. New England Journal of Medicine. 2006;354:333–342. doi: 10.1056/NEJMoa033250. [DOI] [PubMed] [Google Scholar]
- 4.Green M, editor. Hawaii Cancer Facts and Figures 2010. University of Hawaii Cancer Center; 2010. [PMC free article] [PubMed] [Google Scholar]
- 5.Derby KS, Cuthrell K, Caberto C, Carmella SG, Franke AA, Hecht SS, et al. Nicotine Metabolism in Three Ethnic/Racial Groups with Different Risks of Lung Cancer. Cancer Epidemiology Biomarkers & Prevention. 2008;17:3526–3535. doi: 10.1158/1055-9965.EPI-08-0424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Himmelstein MW, Acquavella JF, Recio L, Medinsky MA, Bond JA. Toxicology and epidemiology of 1,3-butadiene. Crit RevToxicol. 1997;27:1–108. doi: 10.3109/10408449709037482. [DOI] [PubMed] [Google Scholar]
- 7.Brunnemann KD, Kagan MR, Cox JE, Hoffmann D. Analysis of 1,3-butadiene and other selected gas-phase components in cigarette mainstream and sidestream smoke by gas chromatography-mass selective detection. Carcinogenesis. 1990;11:1863–1868. doi: 10.1093/carcin/11.10.1863. [DOI] [PubMed] [Google Scholar]
- 8.Melnick RL, Huff J, Chou BJ, Miller RA. Carcinogenicity of 1,3-butadiene in C57BL/6 × C3H F1 mice at low exposure concentrations. Cancer Res. 1990;50:6592–6599. [PubMed] [Google Scholar]
- 9.Huff JE, Melnick RL, Solleveld HA, Haseman JK, Powers M, Miller RA. Multiple organ carcinogenicity of 1,3-butadiene in B6C3F1 mice after 60 weeks of inhalation exposure. Science. 1985;227:548–549. doi: 10.1126/science.3966163. [DOI] [PubMed] [Google Scholar]
- 10.National Toxicology P. NTP Toxicology and Carcinogenesis Studies of 1,3-Butadiene (CAS No. 106-99-0) in B6C3F1 Mice (Inhalation Studies) National Toxicology Program technical report series. 1984;288:1–111. [PubMed] [Google Scholar]
- 11.Owen PE, Glaister JR, Gaunt IF, Pullinger DH. Inhalation toxicity studies with 1,3-butadiene. 3. Two year toxicity/carcinogenicity study in rats. AmIndHygAssocJ. 1987;48:407–413. doi: 10.1080/15298668791384959. [DOI] [PubMed] [Google Scholar]
- 12.Fowles J, Dybing E. Application of toxicological risk assessment principles to the chemical constituents of cigarette smoke. TobControl. 2003;12:424–430. doi: 10.1136/tc.12.4.424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.IARC monographs on the evaluation of carcinogenic risks to humans. Volume 97. 1,3-butadiene, ethylene oxide and vinyl halides (vinyl fluoride, vinyl chloride and vinyl bromide) 2008 2008/01/01 ed. [PMC free article] [PubMed] [Google Scholar]
- 14.Roethig HJ, Munjal S, Feng S, Liang Q, Sarkar M, Walk R-A, et al. Population estimates for biomarkers of exposure to cigarette smoke in adult U.S. cigarette smokers. Nicotine & Tobacco Research. 2009;11:1216–1225. doi: 10.1093/ntr/ntp126. [DOI] [PubMed] [Google Scholar]
- 15.Yuan J-M, Gao Y-T, Wang R, Chen M, Carmella SG, Hecht SS. Urinary levels of volatile organic carcinogen and toxicant biomarkers in relation to lung cancer development in smokers. Carcinogenesis. 2012;33:804–809. doi: 10.1093/carcin/bgs026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Cheng H, Sathiakumar N, Graff J, Matthews R, Delzell E. 1,3-Butadiene and leukemia among synthetic rubber industry workers: Exposure-response relationships. Chemico-Biological Interactions. 2007;166:15–24. doi: 10.1016/j.cbi.2006.10.004. [DOI] [PubMed] [Google Scholar]
- 17.Duescher RJ, Elfarra AA. Human Liver Microsomes Are Efficient Catalysts of 1,3-Butadiene Oxidation: Evidence for Major Roles by Cytochromes P450 2A6 and 2E1. Archives of Biochemistry and Biophysics. 1994;311:342–349. doi: 10.1006/abbi.1994.1246. [DOI] [PubMed] [Google Scholar]
- 18.Fustinoni S, Soleo L, Warholm M, Begemann P, Rannug A, Neumann HG, et al. Influence of metabolic genotypes on biomarkers of exposure to 1,3-butadiene in humans. Cancer EpidemiolBiomarkers Prev. 2002;11:1082–1090. [PubMed] [Google Scholar]
- 19.Wiencke JK, Pemble S, Ketterer B, Kelsey KT. Gene deletion of glutathione S-transferase theta: correlation with induced genetic damage and potential role in endogenous mutagenesis. Cancer Epidemiology Biomarkers & Prevention. 1995;4:253–259. [PubMed] [Google Scholar]
- 20.Gsur A, Zidek T, Schnattinger K, Feik E, Haidinger G, Hollaus P, et al. Association of microsomal epoxide hydrolase polymorphisms and lung cancer risk. BrJ Cancer. 2003;89:702–706. doi: 10.1038/sj.bjc.6601142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Yoshikawa M, Hiyama K, Ishioka S, Maeda H, Maeda A, Yamakido M. Microsomal epoxide hydrolase genotypes and chronic obstructive pulmonary disease in Japanese. IntJ MolMed. 2000;5:49–53. doi: 10.3892/ijmm.5.1.49. [DOI] [PubMed] [Google Scholar]
- 22.London SJ, Smart J, Daly AK. Lung cancer risk in relation to genetic polymorphisms of microsomal epoxide hydrolase among African-Americans and Caucasians in Los Angeles County. Lung Cancer. 2000;28:147–155. doi: 10.1016/s0169-5002(99)00130-0. [DOI] [PubMed] [Google Scholar]
- 23.Nakajima M, Fukami T, Yamanaka H, Higashi E, Sakai H, Yoshida R, et al. Comprehensive evaluation of variability in nicotine metabolism and CYP2A6 polymorphic alleles in four ethnic populations. Clin Pharmacol Ther. 2006;80:282–297. doi: 10.1016/j.clpt.2006.05.012. [DOI] [PubMed] [Google Scholar]
- 24.Borgie M, Garat A, Cazier F, Delbende A, Allorge D, Ledoux F, et al. Traffic-related air pollution. A pilot exposure assessment in Beirut, Lebanon. Chemosphere. 2014;96:122–128. doi: 10.1016/j.chemosphere.2013.09.034. [DOI] [PubMed] [Google Scholar]
- 25.Cheng X, Zhang T, Zhao J, Zhou J, Shao H, Zhou Z, et al. The association between genetic damage in peripheral blood lymphocytes and polymorphisms of three glutathione S-transferases in Chinese workers exposed to 1,3-butadiene. Mutation Research/Genetic Toxicology and Environmental Mutagenesis. 2013;750:139–146. doi: 10.1016/j.mrgentox.2012.10.008. [DOI] [PubMed] [Google Scholar]
- 26.Derby KS, Cuthrell K, Caberto C, Carmella S, Murphy SE, Hecht SS, et al. Exposure to the carcinogen 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone (NNK) in smokers from 3 populations with different risks of lung cancer. International Journal of Cancer. 2009;125:2418–2424. doi: 10.1002/ijc.24585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kolonel LN, Henderson BE, Hankin JH, Nomura AM, Wilkens LR, Pike MC, et al. A multiethnic cohort in Hawaii and Los Angeles: baseline characteristics. Am J Epidemiol. 2000;151:346–357. doi: 10.1093/oxfordjournals.aje.a010213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Hecht SS, Carmella SG, Chen M, Koch JFD, Miller AT, Murphy SE, et al. Quantitation of Urinary Metabolites of a Tobacco-specific Lung Carcinogen after Smoking Cessation. Cancer Research. 1999;59:590–596. [PubMed] [Google Scholar]
- 29.Hecht SS, Carmella SG, Murphy SE. Effects of Watercress Consumption on Urinary Metabolites of Nicotine in Smokers. Cancer Epidemiology Biomarkers & Prevention. 1999;8:907–913. [PubMed] [Google Scholar]
- 30.Tricker AR. Nicotine metabolism, human drug metabolism polymorphisms, and smoking behaviour. Toxicology. 2003;183:151–173. doi: 10.1016/s0300-483x(02)00513-9. [DOI] [PubMed] [Google Scholar]
- 31.Malaiyandi V, Goodz SD, Sellers EM, Tyndale RF. CYP2A6 Genotype, Phenotype, and the Use of Nicotine Metabolites as Biomarkers during Ad libitum Smoking. Cancer Epidemiology Biomarkers & Prevention. 2006;15:1812–1819. doi: 10.1158/1055-9965.EPI-05-0723. [DOI] [PubMed] [Google Scholar]
- 32.Urban M, Gilch G, Schepers G, van Miert E, Scherer G. Determination of the major mercapturic acids of 1,3-butadiene in human and rat urine using liquid chromatography with tandem mass spectrometry. J ChromatogrB AnalytTechnolBiomedLife Sci. 2003;796:131–140. doi: 10.1016/j.jchromb.2003.08.009. [DOI] [PubMed] [Google Scholar]
- 33.Parl FF. Glutathione S-transferase genotypes and cancer risk. Cancer Letters. 2005;221:123–129. doi: 10.1016/j.canlet.2004.06.016. [DOI] [PubMed] [Google Scholar]
- 34.Csanady GA, Guengerich FP, Bond JA. Comparison of the biotransformation of 1,3-butadiene and its metabolite, butadiene monoepoxide, by hepatic and pulmonary tissues from humans, rats and mice. Carcinogenesis. 1992;13:1143–1153. doi: 10.1093/carcin/13.7.1143. [DOI] [PubMed] [Google Scholar]
- 35.Duescher RJ, Elfarra AA. Human liver microsomes are efficient catalysts of 1,3-butadiene oxidation: evidence for major roles by cytochromes P450 2A6 and 2E1. ArchBiochemBiophys. 1994;311:342–349. doi: 10.1006/abbi.1994.1246. [DOI] [PubMed] [Google Scholar]
- 36.Krause RJ, Sharer JE, Elfarra AA. Epoxide hydrolase-dependent metabolism of butadiene monoxide to 3-butene-1,2-diol in mouse, rat, and human liver. Drug Metab Dispos. 1997;25:1013–1015. [PubMed] [Google Scholar]
- 37.Sprague CL, Elfarra AA. Mercapturic acid urinary metabolites of 3-butene-1,2-diol as in vivo evidence for the formation of hydroxymethylvinyl ketone in mice and rats. ChemResToxicol. 2004;17:819–826. doi: 10.1021/tx049949f. [DOI] [PubMed] [Google Scholar]
- 38.van Sittert NJ, Megens HJ, Watson WP, Boogaard PJ. Biomarkers of exposure to 1,3-butadiene as a basis for cancer risk assessment. ToxicolSci. 2000;56:189–202. doi: 10.1093/toxsci/56.1.189. [DOI] [PubMed] [Google Scholar]
- 39.Swenberg JA, Bordeerat NK, Boysen G, Carro S, Georgieva NI, Nakamura J, et al. 1,3-Butadiene: Biomarkers and application to risk assessment. ChemBiolInteract. 2011;192:150–154. doi: 10.1016/j.cbi.2010.10.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Boogaard PJ, van Sittert NJ, Megens HJ. Urinary metabolites and haemoglobin adducts as biomarkers of exposure to 1,3-butadiene: a basis for 1,3-butadiene cancer risk assessment. ChemBiolInteract. 2001;135–136:695–701. doi: 10.1016/s0009-2797(01)00205-8. [DOI] [PubMed] [Google Scholar]
- 41.Albertini RJ, Sram RJ, Vacek PM, Lynch J, Rossner P, Nicklas JA, et al. Molecular epidemiological studies in 1,3-butadiene exposed Czech workers: female-male comparisons. ChemBiolInteract. 2007;166:63–77. doi: 10.1016/j.cbi.2006.07.004. [DOI] [PubMed] [Google Scholar]
- 42.Sapkota A, Halden RU, Dominici F, Groopman JD, Buckley TJ. Urinary biomarkers of 1,3-butadiene in environmental settings using liquid chromatography isotope dilution tandem mass spectrometry. ChemBiolInteract. 2006;160:70–79. doi: 10.1016/j.cbi.2005.12.006. [DOI] [PubMed] [Google Scholar]
- 43.Albertini RJ, Sram RJ, Vacek PM, Lynch J, Wright M, Nicklas JA, et al. Biomarkers for assessing occupational exposures to 1,3-butadiene. ChemBiolInteract. 2001;135–136:429–453. doi: 10.1016/s0009-2797(01)00181-8. [DOI] [PubMed] [Google Scholar]
- 44.Carmella SG, Chen M, Han S, Briggs A, Jensen J, Hatsukami DK, et al. Effects of smoking cessation on eight urinary tobacco carcinogen and toxicant biomarkers. ChemResToxicol. 2009;22:734–741. doi: 10.1021/tx800479s. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.McKee SA, Falba T, O’Malley SS, Sindelar J, O’Connor PG. SMoking status as a clinical indicator for alcohol misuse in us adults. Archives of Internal Medicine. 2007;167:716–721. doi: 10.1001/archinte.167.7.716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Benowitz NL, Dains KM, Dempsey D, Wilson M, Jacob P. Racial Differences in the Relationship Between Number of Cigarettes Smoked and Nicotine and Carcinogen Exposure. Nicotine & Tobacco Research. 2011;13:772–783. doi: 10.1093/ntr/ntr072. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.