Abstract
Acute kidney injury (AKI) is associated with extended hospital stays, high risks of in-hospital and long-term mortality, and increased risk of incident and progressive chronic kidney disease. Patients with urological diseases are a high-risk group for AKI owing to the coexistence of obstructive uropathy, older age, and preexistent chronic kidney disease. Nonetheless, precise data on the incidence and outcomes of postoperative AKI in urological procedures are lacking. Benign prostatic hyperplasia and prostate cancer are common diagnoses in older men and are frequently treated with surgical procedures. Whereas severe AKI after prostate surgery in general appears to be unusual, AKI associated with transurethral resection of the prostate (TURP) syndrome and with rhabdomyolysis (RM) after radical prostatectomy have been frequently described. The purpose of this review is to discuss the current knowledge regarding the epidemiology, risk factors, outcomes, prevention, and treatment of AKI associated with prostatic surgery. The mechanisms of TURP syndrome and RM following prostatic surgeries will be emphasized.
Keywords: Acute kidney injury, Prostatic hyperplasia, Prostate cancer, Transurethral resection of the prostate, Prostatectomy, Rhabdomyolysis
Core tip: Postoperative acute kidney injury has a significant effect on patient outcomes and has been associated with longer hospital stays, high risks of in-hospital and long-term mortality. Urology patients are a high-risk group for acute kidney injury (AKI) because of the common occurrences of obstructive uropathy, older age, and chronic kidney disease, as well as postoperative complications. The purpose of this review is to discuss the current knowledge regarding the epidemiology, risk factors, outcomes, prevention, and treatment of AKI associated with prostatic surgery.
INTRODUCTION
Prostatic diseases are associated with morbidity and mortality in elderly men. Benign prostatic hyperplasia (BPH) is the fourth most common diagnosis in older men[1]. Meanwhile, in developing countries, prostate cancer (PCa) is the most common solid neoplasm, and it is the currently second-leading cause of cancer mortality for men. Beyond conservative medical therapy, the surgical approach remains an important step for the treatment of these diseases[2].
A number of surgical techniques have been developed over the years to treat prostate diseases (Table 1). In recent decades, new surgical methods for treating BPH and PCa have been developed, such as laser- and robot-assisted prostatectomy. Although these procedures have been associated with lower postoperative complication rates in some studies, their efficacy and long-term robustness remain to be proven. At present, the gold-standard treatments for PCa and BPH are still open radical prostatectomy (ORP) and transurethral resection of the prostate (TURP), respectively[2].
Table 1.
Surgical approach to the treatment of benign prostatic hyperplasia and prostate cancer
| Benign prostatic hyperplasia |
| Transurethral resection |
| Open simple prostatectomy |
| Electrovaporization |
| Laser prostatectomy |
| Holmium laser enucleation |
| GreenLight™ laser vaporization |
| Transurethral incision |
| Transurethral needle ablation |
| Prostate cancer |
| Radical Prostatectomy |
| Open (retropubic or perineal) |
| Minimally invasive |
| Laparoscopic |
| Robot-assisted |
In surgical patients, outcomes are strictly dependent on the occurrence of complications. Urology patients are a high-risk group for acute kidney injury (AKI) because of the common occurrences of obstructive uropathy, older age, and CKD, as well as bleeding and urinary obstruction, that sometimes follow the surgery. However, precise data on the incidence and outcomes of postoperative AKI in urological procedures are lacking[3].
Observational studies that compared different surgical approaches to treating prostatic diseases rarely monitored AKI as a relevant early postoperative complication. For instance, in a prospective multicenter analysis of the postoperative complications of 10654 patients subjected to transurethral prostatic surgery for BPH, no AKI case was reported[4]. Similarly, in a prospective observational study of 280 patients subjected to laparoscopic (LSP) or open simple prostatectomy (OSP) for BPH, three patients developed AKI (defined as a 50% rise above the patient’s baseline serum creatinine level)[5]. Moreover, Marmiroli et al[6] studied the postoperative outcomes of 100 patients ≥ 75 years old who had undergone TURP or OSP for BPH and found an incidence of 1% of AKI that required dialysis in this high-risk population. As is the case with the literature on BPH, AKI seems to be infrequently or underreported in patients undergoing ORP for PCa. Recently, one large retrospective analysis, including more than 77000 patients, examined outcomes after robot-assisted radical prostatectomy (RARP) and ORP, and AKI was not cited as a major complication[7].
Based on the available literature, episodes of severe AKI after prostate surgery appear to be unusual. Because the current recommended Kidney Disease Improving Global Outcomes (KDIGO) AKI definitions[8] (Table 2) have never been employed in this situation, acute subclinical serum creatinine (SCr) increases have never been systematically monitored across prostatic surgery outcome studies, and the exact incidence of AKI cannot be determined. However, AKI secondary to TURP syndrome has been consistently described[9,10]. Furthermore, a number of small case studies and case reports of rhabdomyolysis (RM)-associated-AKI after radical prostatectomy have also been published[11-13].
Table 2.
Kidney disease improving global outcomes acute kidney injury definitions
| AKI is defined as any of the following |
| Increase in serum creatinine by ≥ 0.3 mg/dL within 48 h; or |
| Increase in serum creatinine to ≥ 1.5 times baseline, which is known or presumed to have occurred within the prior 7 d; or |
| Urine volume < 0.5 mL/kg per hour for 6 h |
AKI: Acute kidney injury.
This review outlines AKI associated with prostatic surgery, emphasizing the mechanisms of TURP syndrome and RM following prostatic surgeries. The surveillance, prevention and treatment of these complications will also be addressed.
TURP AND AKI
TURP requires the use of an irrigating fluid to expand the operating field and to wash away debris and blood. TURP syndrome is a clinical complication caused by the systemic absorption of the irrigating fluid and is characterized by a combination of hyponatremia and fluid overload, causing potential damage to the cardiovascular, renal and nervous systems[14,15].
The incidence of TURP syndrome ranges from 1.0% to 8% of reported TURPs and appears to be decreasing in recent years[16,17]. Mortality rates are generally between 0.2% to 0.8%, but rates as high as 25% can occur if severe TURP syndrome develops[10,18].
TURP syndrome can be defined as sodium of 125 mEq/L or less after TURP with two or more circulatory and/or neurological symptoms[19]. However, no universal defining criteria have been adopted by all centers, and not all studies have used clear definitions of TURP syndrome. Of note, the definition and severity of kidney dysfunction are not always detailed in TURP syndrome, and no studies have used the definition proposed by the most recently updated Kidney Disease Improving Global Outcomes (KDIGO) AKI guidelines. Similarly, there are scant data on late prognoses because few studies have reported on outcomes more than three to six months after the event. In particular, the incidence of CKD resulting from TURP syndrome is unknown. Many aspects of TURP syndrome are still unclear, and its overall burden is not completely determined.
RISK FACTORS
Although important differences appears according to the compositions of the irrigation fluids, the most important risk factor for TURP syndrome is the amount of fluid absorbed, which can vary from < 300 to 3000 mL[20]. After the absorption of 1.0 mL of fluid, serum sodium reduction is approximately 6 to 8 mEq/L, and it can achieve 20 mEq/L after absorption of 3.0 L[21]. The rate of fluid absorption is most likely also an important risk factor, and absorption in excess of 200 to 300 mL per 10 min is more frequently related to hyponatremia[22]. The risk of the TURP syndrome is higher in the presence of bleeding, longer resection times, higher absorption of irrigating fluid and prostate size larger than 45 g[23]. Smoking is a factor known to be associated with the increased risk of TURP syndrome , but the malignancy does not seem to be associated with the increased risk[24].
FLUID ABSORPTION MECHANISM
In TURP, a resectoscope loaded with a diathermy loop is introduced into the bladder to resect prostatic tissue (Figures 1 and 2). TURP typically takes 60 min, and approximately 10 to 20 liters of irrigating fluid are generally required. During TURP, irrigation pressure is regulated to achieve 60 mmHg, a much higher threshold than that for physiological intravesical pressure, which peaks at 25 mmHg during micturition[25]. When prostate tissue is resected, veins may be severed, and irrigating fluid can be rapidly absorbed into the vascular system. Intravenous fluid absorption begins when the fluid pressure exceeds the prostate venous pressure by approximately 12.5 mmHg[26], and it rarely ceases once it begins[20]. Most of the fluid absorption takes place during the second half of the resection when the resectoscope approaches the vein plexus, reaching larger vessels, and an extended area for fluid influx is opened[20]. Herein, small amounts of fluid are always absorbed during TURP, and absorption in excess of one liter has been reported to occur in 5%-20% of procedures[27,28]. Extravasation can occur after instrumental damage of the prostatic capsule or the bladder wall during TURP, which occurs in at least 10% of surgeries (Figure 3). The fluid pressure must only exceed an intra-abdominal pressure of approximately 3.72 mmHg for extravasation to occur, and several liters of irrigating fluid will be rapidly deposited in the periprostatic, retroperitoneal or intraperitoneal spaces[29]. The fluid is absorbed by lymphatic drainage, a slower process than vascular intake, which can translate into a delayed reduction in serum sodium compared with classic intravascular absorption. Extravasation contributes to TURP syndrome development in approximately 20% of cases[30].
Figure 1.
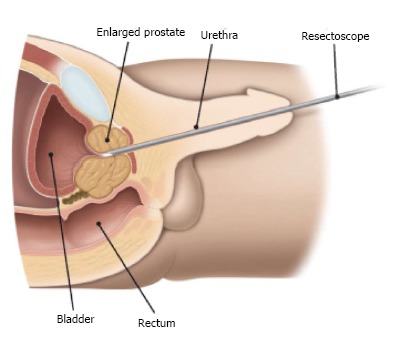
Surgery through the urethra. Courtesy of the European Association of Urology.
Figure 2.
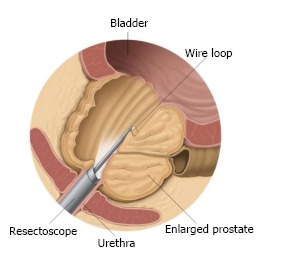
The resectoscope removes parts of the prostate tissue during transurethral resection of the prostate. Courtesy of the European Association of Urology.
Figure 3.
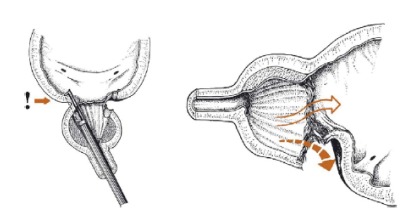
Division of the bladder neck with subsequent extraperitoneal extravasation. From Rassweiler et al[28].
IRRIGATION FLUID PHYSIOPATHOLOGY
An ideal irrigating fluid should be isotonic, nonhemolytic, electrically inert, nontoxic when absorbed, not metabolized, and transparent; should not influence blood osmolality; should be rapidly excreted; and should not cause significant expansion of extracellular fluid volume. However, no such solution exists; each irrigating fluid has particular physicochemical and pharmacokinetic characteristics and each comes with its own potential complications.
Distilled water
Distilled water (D.W.) was the first irrigating fluid used in TURP surgeries and is still often used because of its advantages: clear visual field, absence of electric conductivity, volume of distribution equal to the body’s full water content, and lower cost[31,32]. After the description of cases of hemolytic AKI[33,34], iso- or hypo-osmolar non-hemolytic solutions other than D.W. were introduced to overcome this issue. D.W. continues to be used, but it should be restricted to laser TURP or to procedures that are expected to be short and performed by very skilled surgeons with all precautions taken to avoid the risk of TURP syndrome[32].
Glycine
Glycine (Gly) is a neutral, nonessential amino acid, used as an irrigating fluid solute since 1949. A 1.5% Gly solution is commonly used in TURP because it is nonconductive, hypo-osmotic (osmolality of approximately 200 mOsm/L), and nonhemolytic and it provides good optical visibility[35]. This amino acid spreads through intracellular compartments, creating osmotic water movement into cells, which gradually increases serum sodium and minimizes the risk of severe hyponatremia. However, Gly presents some drawbacks and is commonly associated with TURP syndrome. It has cardiotoxic properties, and can cause serious visual disturbances due to retina damage[36]. Hyper-ammoniemic encephalopathy[37] and hyperoxaluria have been associated with Gly metabolites[38].
Sorbitol solutions
Solutions based in sorbitol are frequently used in the United States of America as irrigating fluids, typically in the concentration range of 2.2% to 3% and frequently in association with mannitol[39]. Sorbitol is non-electrolytic, has an osmolality of approximately 180 mOsm/L, and clears rapidly from the plasma after its transformation to fructose and glucose by liver cells. Sorbitol is considered a safe irrigating fluid and is associated with low rates of TURP syndrome.
Mannitol
Mannitol is frequently used as an irrigating fluid in concentrations of 3% or 5% solution (osmolalities of 175 and 275 mmOsm/L, respectively)[40]. Mannitol is nonconductive and nonhemolytic and gives a satisfactory visual operating field. Mannitol is excreted unchanged in the urine, promoting osmotic, electrolyte-free diuresis, which could help to increase serum sodium concentration. Mannitol is considered a suitable irrigating fluid that is associated with low rates of TURP syndrome.
Physiologic saline
Saline solution cannot be used as an irrigating fluid during the standard prostatic resection because its dissipates the electrical current of bipolar resectoscope, therefore preventing both cutting and coagulation. Normal saline (0.9%) osmolality is approximately 300 mOsm/L, which makes it the most suitable irrigation fluid for TURP. Very few cases of TURP syndrome have been described with this solution. Fluid overload is more likely during the absorption of normal saline solution due to the higher volume expansion[41]. In addition, the excessive sodium chloride infusion can cause hyperchloremic acidosis.
CLINICAL AND LABORATORY PRESENTATION
TURP syndrome occurs from 15 min to 24 h after prostate resection. The incidence and severity of TURP syndrome symptoms increase progressively as more solution is absorbed. When the threshold of 3.0 L is exceeded[10], the symptoms are severely impaired.
Some symptoms may be noticed in the intraoperative period. Vague, nonspecific symptoms may occur such as the sense of being unwell[27], transitory feeling of burning, accompanied by nausea, restlessness and headache. Neurologic events are more frequently observed when glycine solutions are used and in patients with decreases in serum sodium of 10 mEq/L or more[42]. Focal or generalized seizures and altered mental states can occur. This is generally associated with irrigant absorption levels as high as 2.0 to 3.0 L. Brain stem herniation, persistent brain injury and death have also been reported[43]. Visual disturbances, including transitory blindness have been observed, mostly related to glycine solution[36].
Patients may develop both hypervolemic and hypovolemic complications. Bradycardia and hypotension at the end of the operation, or immediately after, are often early signs suggesting TURP syndrome[44]. Shortness of breath and pulmonary edema can occur in surgeries where mild/less severe bleeding is observed[45]. Chest pain and hypertension have also been observed in 5% of the patients, particularly when more than 1 L is absorbed[46]. Small elevations in cardiac enzymes can occur, especially when Gly solution is used[47].
Most patients subjected to TURP are elderly with coexisting diseases, reduced functional heart and kidney reserves and less capacity to endure stress[48]. CKD patients are also at exacerbated risk for TURP syndrome[15]. AKI has been reported in TURP syndrome patients; it is typically oliguric and can be observed as early as the first postoperative day. Bilen et al[9] assessed a group of 439 patients who had undergone TURP using mostly distilled water as the irrigating fluid. AKI defined as an increase in postoperative SCr > 1 mg/dL occurred in 16 (3.64%) of the patients.
Severe TURP syndrome, defined by a drop in serum sodium concentration to < 120 mEq/L, is a rare but well-described event in the specialized literature, characteristically reported when more than 3.0 L of irrigating fluid are absorbed[49]. A review of 24 severe cases in which Gly 1.5% was used as the irrigating fluid demonstrated neurological complaints in 92%, cardiovascular signs in 54%, visual disturbances in 42%, and gastrointestinal symptoms in 25% of these patients, with a mortality rate of 25%. AKI is observed in more than 50% of cases of severe TURP syndrome, sometimes requiring renal replacement therapy (RRT)[50].
Hyponatremia (< 135 mEq/L), a hallmark of the syndrome, is seen in nearly all patients, and it is more frequently observed at the end of surgery (or one to two hours subsequent)[51]. Hyponatremia might be transitory and could go undetected if serum sodium is assessed more than three hours after surgery completion. Although most irrigating fluids are hypo-osmolar (approximately 200 mOsmol/L), compared with normal serum osmolality (approximately 290 mOsm/L), hypo-osmolality in TURP syndrome is less pronounced than that observed in other hyponatremia etiologies because the solute contents of irrigating fluids (Gly, sorbitol, mannitol) prevent large osmolality reductions[52]. In TURP syndrome, serum osmolar gaps reflect the concentrations of the infused/absorbed irrigants and can achieve 30 to 60 mOsm/kg[53]. Herein, serum osmolality should be measured in all TURP syndrome patients.
PATHOPHYSIOLOGY
Hemodynamics
The rapid volume expansion which can reach up to 200 mL/min can cause hypertension and reflex bradycardia. Hypertension coupled with hyponatremia can trigger pulmonary edema and hypovolemia due to net water flux from the intravascular space into the pulmonary interstitium[54]. In sequence, a major hypokinetic hemodynamic phase ensues, distinguished by low cardiac output, hypovolemia and hypotension[44,55].
Natriuresis has been highlighted as a key element in promoting dilutional hyponatremic shock and explains why hypovolemic hypotension persists despite the administration of large amounts of fluid. The osmotic diuresis leads to sodium losses and occurs when the renal reabsorption mechanisms are either overwhelmed (Gly) or absent (mannitol)[56]. The capacity of the kidneys to control the urine’s composition is then undermined, and a number of small solutes, including amino acids and sodium, are ultimately lost from the body.
Other factors that contribute to the hemodynamic changeover include metabolic acidosis[57], acute hypothermia[58], release of endotoxins into the bloodstream[59], and depression of the heart conductivity system[47].
Central nervous system
Even moderate osmolality reduction could result in a fluid influx into the cerebral space, leading to brain edema[59,60]. Other factors that contribute to central nervous system impairment in TURP syndrome, such as the very low serum sodium concentration itself, are Gly toxicity and the accumulation of its metabolic derivates (ammonia, serine, and/or glyoxylate)[37,61].
Renal disease
AKI following TURP has been reported since 1947, and a variety of mechanisms have been proposed for its development[62,63]. When sterile water was used for irrigation, intravascular hemolysis was thought to be the principal insult[64]. Hemolysis takes place in the blood as well as in the bladder, where hemolyzed blood is absorbed. In both cases, hemoglobinuria develops and renal injury occurs through a number of pathways. Heme proteins have powerful oxidant effect, it can trigger renal vasoconstriction[65] and under acidic conditions, precipitate with Tamm-Horsfall proteins contributing to tubule obstruction. Hemolysis should be investigated in all AKI events after TURP[34] although these events have become rare with the observed change to other irrigating fluids, and other pathogenic mechanisms have been described.
Hyponatremia-associated RM resulting in AKI has been reported as a complication following TURP in a small number of cases[66]. After a number of hours, muscle cellular swelling induced by hyponatremia will peak because of the potassium outflux from the muscle cells into the extracellular fluid. Hyponatremia also reduces the concentration gradient for sodium entry into the muscle cells, resulting in a decreased outward flux of calcium, which leads to increased intracellular calcium[67] destroying the cell structure[68]. At this point, all patients with AKI-related TURP syndrome should be screened for RM.
It has been suggested that “hemodynamic” acute tubular necrosis is an important cause of AKI in some patients after TURP. Hypotension coupled with osmotic diuresis results in ischemic kidney episodes[69]. Another possible mechanism is sudden kidney cell swelling as a result of acute hypo-osmolality, similar to the development of central nervous system edema[70].
More recently, Kim et al[71] described three cases of AKI after laser vaporization of the prostate using distilled water as the irrigating fluid. All patients developed significant hyponatremia, and two of them required RRT[71]. Histological findings were tubular cell necrosis and Tamm-Horsfall protein stasis with regurgitation into the Bowman capsule accompanied by an amount of eosinophilic interstitial infiltrate. Special staining for hemoglobin and myoglobin had negative results, and there was no histologic evidence of ischemic damage. Hyponatremia and hemodynamic mechanisms could not be ruled out, but this scenario strongly suggested direct damage to the tubular epithelium by urinary stasis and the backflow of the irrigating fluid, hemoglobin, and prostate secretions resulting from high intravesical pressure. This report suggests transient vesicoureteral reflux as a new pathogenic mechanism of kidney injury in TURP syndrome, although this has yet to be confirmed by other studies[71].
The physiopathology of renal injury in TURP syndrome is complex, multifactorial and not completely understood. It is most likely that one or more of a number of mechanisms are implicated.
PREVENTION
Prostate gland size and operative time
There are no definitive data establishing an operative time threshold beyond which excessive fluid is absorbed, but after one hour of surgery, the risk increases significantly[72], and after that point, the patient´s overall status, the volume of fluid absorbed, and the anticipated time to completion should be reassessed[73]. For patients with large glands and expected long procedures, it is advised that bipolar TURP or other low-risk techniques be used.
Fluid bag height
Fluid is infused using the force of gravity (elevating the infusion bag to different heights) or by inflating a large blood pressure cuff around the infusion bag. Placing the irrigating fluid bag at 60 cm above the operating table has been advised to avoid fluid absorption. Nevertheless, two studies including almost 600 patients did not demonstrate conclusive benefits in higher fluid bag[74,75].
Intraprostatic vasopressin injection
Transrectal intraprostatic vasopressin (IPVP) injected at the operating site is considered to vasoconstrict intraprostatic vessels and reduce blood loss and fluid absorption during TURP[76]. IPVP appears to be effective and could be used in patients with large prostates or when fluids associated with higher incidence of TURP syndrome such as Gly and D.W. are used.
Low-pressure irrigation
Irrigating fluid absorption is less pronounced when TURP is performed under low pressure. A number of measures to maintaining low intra-bladder pressure has been used, such as suprapubic catheterization, intermittent drainage of the irrigating fluid and continuous flow resection[77].
Bipolar TURP
Bipolar resection of the prostate utilizes a specialized resectoscope loop that incorporates both the active and the return electrodes. The bipolar loop resects, coagulates, vaporizes and transects the tissue. Because the bipolar resectoscope uses a 0.9% sodium chloride solution as the irrigation fluid, the risk of TURP syndrome is eliminated, allowing for longer and safer resections[78]. Omar et al[79] recently published a systematic review and meta-analysis comparing bipolar and monopolar TURP. The study comprised 24 trials, and no case of TURP syndrome was observed in the bipolar group. Therefore, bipolar TURP is a safe procedure that is suitable for high-risk patients such as CKD patients and those with large glands.
Laser and vaporization prostatectomy vs TURP syndrome
Photoselective vaporization of the prostate (PVP) and other laser techniques are novel procedures that promote effective hemostasis with nearly bloodless removal of prostate tissue and minimal absorption of irrigating fluid[80]. PVP can use normal saline as the irrigating fluid, and laser therapies have been reported to successfully treat patients with very large prostates (> 100 g) and those with ongoing oral anticoagulation[71].
Trans-operatory monitoring of fluid absorption
A key aspect to preventing the development of TURP syndrome is monitoring the fluid absorption during the endoscopic surgery. A number of alternatives have been attempted to achieve this goal. Volumetric fluid balance, the difference between the amount of irrigating fluid used and the output volume, is the most commonly used technique to estimate fluid absorption. However, other variables such as bleeding, irrigant leakage, urinary output (diuresis), and blood dilution make this a comparatively unreliable tool[81]. Although it has limitations, volumetric fluid balance is simple, noninvasive, and inexpensive, and it should be performed in every surgery.
The gravimetric method is often used and requires that the surgery take place on a bed-scale. The method relies on the supposition that increases in the body weight are generated by fluid absorption. Bleeding and intravenous infusions must be considered in recordings, that must be carried out when the bladder is empty[82].
To minimize the risk of hyponatremia, an intraoperative approach based on the amount of absorbed fluid is suggested[27]. If more than 1.0 L of fluid is estimated to be absorbed, the surgical team should temporarily halt the procedure, fluid inflow should cease and serum sodium should be measured. If mental and cardiovascular status are maintained, surgery can be resumed for as short a period as possible. If more than 2.0 L of fluid were absorbed, hemorrhage points should be coagulated and the procedure should be terminated. Serum sodium concentration and mental status should be closely monitored.
MANAGEMENT AND TREATMENT OF TURP SYNDROME
The urologic surgeon and the anesthetist should be aware of the development of TURP syndrome. Asymptomatic and stable patients should be kept under observation. Specific treatment is not required, particularly if sodium reduction is below 5 mEq/L. In these cases, if renal function is adequate, excretion of the excess water and metabolism of the infused solute will rapidly correct the hyponatremia[83]. There is no specific treatment for the visual symptoms of Gly intoxication, and even blindness is typically resolved in 24 h without the need for specific treatment[84].
Hypertonic saline is indicated to replace the excreted sodium in symptomatic patients with marked hyponatremia, particularly those who have substantially reduced serum osmolality or a cerebral edema[85]. Hypertonic saline in a 3% solution can be given as a 100 mL bolus at 10-min intervals or continuously infused (approximately 1.0 L in 12 h). Rapid correction of hyponatremia is most likely safe following TURP because of the extremely short duration of hyponatremia and the restricted time for cerebral adaptations. A reasonable and safe strategy is to increase the serum sodium concentration to up to 12 mEq/L in the first 24 h. Furosemide may be used to reverse the fluid overload, although furosemide increases natriuresis and hyponatremia, further reducing plasma volume and increasing the cellular edema[86,87]. Furosemide should not be routinely given in TURP patients in the absence of fluid overload.
Hemodialysis will rapidly correct hyponatremia, osmotic derangements, and volume expansion and remove the non-electrolyte solute and its toxic metabolites (Gly, sorbitol, mannitol). It has been used in symptomatic patients with severe renal disease and in patients with severe neurologic symptoms and marked hyponatremia[53].
In the case of important fluid extravasation and large fluid collections, it might be necessary to carry out a open surgical drainage by percutaneous drainage[88].
ANESTHESIA AND TURP SURGERY
For years, spinal anesthesia was considered the anesthetic technique of choice for TURP. Spinal anesthesia is considered to reduce the risk of pulmonary edema, to decrease bleeding risk and to allow a prompt diagnosis of neurologic symptoms[89]. However, spinal anesthesia reduces central venous pressure, affecting prostate venous pressure, which could result in greater absorption of the irrigating fluid[89]. During general anesthesia, the detection of TURP syndrome may be more difficult, based on afterward changes in blood pressure and electrocardiographic abnormalities[89]. In fact, the best anesthetic technique for TURP procedures has not yet determined.
RADICAL PROSTATECTOMY AND RHABDOMYOLYSIS
Radical prostatectomy is the main surgical treatment for PCa. Radical retropubic prostatectomy (RRP) is performed with patients in the supine position, while in radical perineal prostatectomy (RPP), patients are placed in an exaggerated lithotomy position[90]. More recently LSP and RARP have been developed, and these minimally invasive procedures are replacing open radical prostatectomy in some countries[2].
The first description of position-induced RM with subsequent AKI after a knee-chest position was in 1953 by Gordon[91]. Renal failure following radical prostatectomy is uncommon, and the incidence of subclinical RM following this procedure is currently unknown[90]. In a recent retrospective study of 175699 patients subjected to robotic or non-robotic radical prostatectomy, the incidence of RM was 0.08%[92]. In a prospective study of 60 patients undergoing RARP and lymph node dissection with prolonged positioning in a steep Trendelenburg position, Mattei et al[93] demonstrated that ten patients developed RM (serum creatine kinase > 5000 IU/L). Although AKI following radical prostatectomy appears to be rare, there are a number of case reports that suggest RM secondary to an exaggerated lithotomy position as a cause of AKI in this setting[90,94-96].
AKI associated with myoglobinuria is an important complication of RM[97]. After glomerular filtration, myoglobin is uptake by the tubule epithelial cell through endocytic pathways and is metabolized. The precise mechanisms leading to the glomerular filtration rate impairment are unclear, however some evidence suggests that hypovolemia, vasoconstriction, intraluminal cast formation, oxidative stress and direct heme-induced cytotoxicity are all responsible for kidney injury.
RM after the exaggerated lithotomy position during surgery is usually due the intraoperative development of lower extremity compartment syndrome or from muscle breakdown in the back and gluteal regions[90]. Improperly positioned or inadequately padded patients are prone to ischemia-reperfusion injury from excessive compression. Compartment syndrome was reported in 93% of 46 patients with position-related RM associated with AKI[96]. The lower extremities were most often involved (50%), with muscle swelling and ache being the presenting symptoms[96]. Lithotomy and chest-knee position were the most frequent postures in these patients[96]. Fortunately, compartment syndrome is a rare complication of radical prostatectomy surgery[92].
Rhabdomyolysis associated with prostatectomy prevention and management
To prevent RM associated with surgical position, all pressure points should be protected, including paying special attention to the shoulders, back, and sacrum[90]. The vascular status of the patient’s lower extremity should be evaluated with a preoperative vascular examination, and repositioning the lower extremities every two hours could improve perfusion and avoid the occurrence of injury[90]. Some investigators have suggested noninvasive or invasive intraoperative monitoring to assess for impending limb compartment syndrome[94]. Others have recommended obtaining preoperative CPK levels, levels every two hours intraoperatively and levels six to eighteen hours postoperatively if the procedure is expected to be prolonged[11], especially in high-risk groups[93].
An approach to the extensive management of RM was published elsewhere[97]. The approach to RM after prostatectomy is not different from that in other settings. Early and aggressive intravascular volume expansion with crystalloids to restore kidney blood flow and increase urine flow is the cornerstone intervention for preventing and treating AKI. Intravenous fluids should be initiated ideally within the first 6 h after muscle injury, at a rate that maintains a urine output of 300 mL/h or more in adults, for at least the first 24 h[98]. There is insufficient evidence to support the routine use of bicarbonate-containing fluids, mannitol, or loop diuretics. Hyperkalemia and compartment syndrome are other complications that should be closely surveyed, early diagnosed, and effectively treated.
NEPHROLOGY CONSULTATION
Preoperative nephrology consultation might identify clinical risk factors such as CKD or other comorbidities. To accurate diagnosis and stage CKD we suggest the CKD Epidemiology Collaboration (CKD-EPI) equation. The CKD-EPI equation was published in 2009 and intended to be more generalizable across various clinical settings[99].
TURP syndrome and RM after prostatectomy are generally diagnosed and treated by anesthesiologists and urologists. Nephrologists are typically called only in the most severe cases, which require RRT. Nephrologists should be aware of the risk factors, physiopathology, clinical picture and treatment strategies of TURP syndrome and RM after prostatectomy. An active role of nephrologists in the whole procedure could improve the care of those patients. Nephrologists would be an important add for early identification and treatment of AKI, electrolytes abnormalities, fluid overload, and previous chronic kidney dysfunction.
Although the limitations in the quality of published evidence preclude firm recommendations in this field, some suggestions on preventive and management strategies are depicted in Tables 3 and 4.
Table 3.
Suggestions for the prevention and management of transurethral resection of prostate syndrome
| Preoperative |
| Estimate GFR using the CKD-EPI equation |
| Identify patient risk factors: large prostate gland (> 45 g), heart disease, CKD, and smoking |
| Advise bipolar TURP or laser techniques for high-risk patients |
| Intraoperative |
| Avoid D.W. and glycine as irrigating fluids. Sorbitol and mannitol are good options. Physiologic saline is a safe choice when feasible |
| Maintain low-pressure irrigation |
| Consider the use of intra-prostatic vasopressin injection in high-risk patients |
| Alert surgical team when surgery exceeds one hour |
| Monitor the volume of absorbed fluid. Consider aborting the procedure if the absorbed volume exceeds 1.0 L and suspend surgery if absorbed volume exceeds 2000 mL |
| Both spinal and general anesthesia are adequate |
| Avoid hypotension and central venous pressure reduction and closely monitor the vital signs |
| Post-operative |
| Assess serum sodium and serum creatinine in all patients in the immediate postoperative period |
| Apply KDIGO AKI definitions to AKI diagnosis |
| If TURP syndrome is diagnosed, initiate medical treatment: |
| Assess serum osmolality |
| Maintain asymptomatic and mildly symptomatic patients under close observation |
| Initiate hypertonic saline 3% infusion in symptomatic patients with marked hyponatremia, reduced osmolality and cerebral edema |
| Restrict diuretic use to treat fluid overload |
| If AKI occurs, test for hemolysis and rhabdomyolysis |
| Consider hemodialysis in symptomatic patients with severe renal disease |
| Patients that developed AKI should be followed and eGFR equations must be used to identify CKD |
AKI: Acute kidney injury; CKD: Chronic kidney disease; CKD-EPI: Chronic Kidney Disease Epidemiology Collaboration; D.W.: Distilled water; GFR: Glomerular filtration rate; KDIGO: Kidney disease improving global outcomes; TURP: Transurethral resection of prostate.
Table 4.
Suggestions for the prevention and management of surgical position-related rhabdomyolysis
| Preoperative |
| Identify patient risk factors: obesity, hypovolemia, diabetes mellitus, hypertension, chronic kidney disease, peripheral vascular disease, expected surgery time longer than 5 h |
| The vascular status of the patient’s lower extremity should be carefully assessed with a well-documented preoperative vascular examination |
| The patient´s volume status should be evaluated |
| Intraoperative |
| Ensure correct patient positioning and protect all pressure points |
| Monitor lower extremities and vascular status |
| Reposition lower extremities every two hours |
| Adequate fluid reposition, avoiding hypovolemia |
| Monitor serum potassium levels |
| Appropriate operative time, completing the procedure as quickly as possible |
| Post-operative |
| Assess serum-CK and SCr 6 h and 18 h postoperatively in high-risk patients |
| Closely check serum creatinine, potassium levels, and acid-base disorders |
| Apply KDIGO AKI definitions to AKI diagnosis |
| Monitor signs of compartmental syndrome and consider fasciotomy if present |
| If RM syndrome is diagnosed, initiate medical treatment: |
| Initiate aggressive early fluid repletion; |
| Treat acid-base and electrolyte abnormalities; |
| Consider early RRT |
CK: Creatine kinase; KDIGO: Kidney disease improving global outcomes; RM: Rhabdomyolysis; RRT: Renal replacement therapy; SCr: Serum creatinine. AKI: Acute kidney injury.
CONCLUSION
Severe AKI appears to be a rare event after prostate surgery. However, it is a hazardous surgical complication that increases the risk of permanent kidney damage or death. Because mild SCr elevations were not systematically monitored across the majority of the available studies, the exact incidence of AKI is underdetermined. Studies using the current definitions of AKI are very necessary for providing a better understanding of AKI risk factors and the influence of AKI on patient outcomes after prostate surgeries. Preoperative nephrology consultation might be helpful to better assess kidney function and the presence of other risk factors for AKI, allowing for adequately planning the surgical technique and reinforcing preventive strategies. Affected patients should be followed to assess long-term prognosis and CKD development.
In the last years, several studies about urinary and serum biomarkers for the diagnosis and prognostication of AKI have been published[100,101]. The question that arises is which biomarker is a reliable differential diagnostic tool under which circumstances. As hematuria and need of bladder irrigation are common after prostatic surgeries, the urinary biomarkers might be less suitable in this setting. Further research in this field is warranted before biomarkers can be introduced in the clinical practice.
The available data suggest TURP syndrome as the main mechanism for AKI following prostatic surgery. The absorption of 1-2 L of irrigating fluid occurs in 5%-10% of patients and results in easily overlooked mild TURP syndrome. Fortunately, TURP syndrome incidence appears to be declining because of the use of laser surgery techniques and bipolar circuitry, together with the systematic institution of routine precautions to minimize the risk of TURP syndrome development (e.g., low-pressure irrigation, monitoring the extent of absorption and surgery length). TURP syndrome pathophysiology is complex, multifactorial and not completely understood. The pathogenic mechanisms postulated for AKI development include acute hemolysis, renal interstitial edema, ischemic tubular injury, RM and reflux nephropathy resulting from the absorption of irrigating fluid, dilutional hyponatremia and high intra-bladder pressure. A variety of different irrigating fluids are available, but studies in animals, volunteers and patients show that glycine solution should be avoided. Treatment of symptomatic hyponatremia should be based on the administration of hypertonic saline rather than of diuretics. RRT might be necessary in severe AKI.
RM-induced AKI after radical prostatectomy has also been described in a small number of case reports. These reports identified a number of risk factors, such as exaggerated lithotomy position, preexisting CKD, obesity, and surgery longer than 5 h. In patients at high risk for AKI, every effort should be made to ensure correct positioning during surgery. Early diagnosis of RM and aggressive volemic expansion are the keys to the patient’s successful recovery.
It is important for nephrologists to know the main aspects of the physiopathology, clinical presentation, treatment and particular characteristics of AKI in the context of prostate surgery. Close collaboration with the urologist and anesthesiology staff is extremely important to allow for the adoption of preventive measures and to detect any earlier, elusive clinical presentations of AKI.
Footnotes
P- Reviewer: Prakash J, Passadakis PS, Wang F S- Editor: Ji FF L- Editor: A E- Editor: Lu YJ
References
- 1.Sausville J, Naslund M. Benign prostatic hyperplasia and prostate cancer: an overview for primary care physicians. Int J Clin Pract. 2010;64:1740–1745. doi: 10.1111/j.1742-1241.2010.02534.x. [DOI] [PubMed] [Google Scholar]
- 2.Heidenreich A, Bastian PJ, Bellmunt J, Bolla M, Joniau S, van der Kwast T, Mason M, Matveev V, Wiegel T, Zattoni F, et al. EAU guidelines on prostate cancer. part 1: screening, diagnosis, and local treatment with curative intent-update 2013. Eur Urol. 2014;65:124–137. doi: 10.1016/j.eururo.2013.09.046. [DOI] [PubMed] [Google Scholar]
- 3.Caddeo G, Williams ST, McIntyre CW, Selby NM. Acute kidney injury in urology patients: incidence, causes and outcomes. Nephrourol Mon. 2013;5:955–961. doi: 10.5812/numonthly.12721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Reich O, Gratzke C, Bachmann A, Seitz M, Schlenker B, Hermanek P, Lack N, Stief CG. Morbidity, mortality and early outcome of transurethral resection of the prostate: a prospective multicenter evaluation of 10,654 patients. J Urol. 2008;180:246–249. doi: 10.1016/j.juro.2008.03.058. [DOI] [PubMed] [Google Scholar]
- 5.McCullough TC, Heldwein FL, Soon SJ, Galiano M, Barret E, Cathelineau X, Prapotnich D, Vallancien G, Rozet F. Laparoscopic versus open simple prostatectomy: an evaluation of morbidity. J Endourol. 2009;23:129–133. doi: 10.1089/end.2008.0401. [DOI] [PubMed] [Google Scholar]
- 6.Marmiroli R, Antunes AA, Reis ST, Nakano E, Srougi M. Standard surgical treatment for benign prostatic hyperplasia is safe for patients over 75 years: analysis of 100 cases from a high-volume urologic center. Clinics (Sao Paulo) 2012;67:1415–1418. doi: 10.6061/clinics/2012(12)11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Sammon JD, Karakiewicz PI, Sun M, Sukumar S, Ravi P, Ghani KR, Bianchi M, Peabody JO, Shariat SF, Perrotte P, et al. Robot-assisted versus open radical prostatectomy: the differential effect of regionalization, procedure volume and operative approach. J Urol. 2013;189:1289–1294. doi: 10.1016/j.juro.2012.10.028. [DOI] [PubMed] [Google Scholar]
- 8.Kellum JA, Lameire N. Diagnosis, evaluation, and management of acute kidney injury: a KDIGO summary (Part 1) Crit Care. 2013;17:204. doi: 10.1186/cc11454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Bilen CY, Sahin A, Ozen H, Aki FT, Oge O, Kendi S. Nonoliguric renal failure after transurethral resection of prostate. J Endourol. 1999;13:751–754. doi: 10.1089/end.1999.13.751. [DOI] [PubMed] [Google Scholar]
- 10.Hahn RG. Irrigating fluids in endoscopic surgery. Br J Urol. 1997;79:669–680. doi: 10.1046/j.1464-410x.1997.00150.x. [DOI] [PubMed] [Google Scholar]
- 11.Bruce RG, Kim FH, McRoberts W. Rhabdomyolysis and acute renal failure following radical perineal prostatectomy. Urology. 1996;47:427–430. doi: 10.1016/s0090-4295(99)80467-1. [DOI] [PubMed] [Google Scholar]
- 12.Orihuela E, Nazemi T, Shu T. Acute renal failure due to rhabdomyolysis associated with radical perineal prostatectomy. Eur Urol. 2001;39:606–609. doi: 10.1159/000052512. [DOI] [PubMed] [Google Scholar]
- 13.Yang RH, Chu YK, Huang CW. Compartment syndrome following robotic-assisted prostatectomy: rhabdomyolysis in bone scintigraphy. Clin Nucl Med. 2013;38:365–366. doi: 10.1097/RLU.0b013e318286808e. [DOI] [PubMed] [Google Scholar]
- 14.Hahn RG. Fluid absorption in endoscopic surgery. Br J Anaesth. 2006;96:8–20. doi: 10.1093/bja/aei279. [DOI] [PubMed] [Google Scholar]
- 15.Hawary A, Mukhtar K, Sinclair A, Pearce I. Transurethral resection of the prostate syndrome: almost gone but not forgotten. J Endourol. 2009;23:2013–2020. doi: 10.1089/end.2009.0129. [DOI] [PubMed] [Google Scholar]
- 16.Neal DE. The National Prostatectomy Audit. Br J Urol. 1997;79 Suppl 2:69–75. doi: 10.1111/j.1464-410x.1997.tb16924.x. [DOI] [PubMed] [Google Scholar]
- 17.Mebust WK, Holtgrewe HL, Cockett AT, Peters PC. Transurethral prostatectomy: immediate and postoperative complications. a cooperative study of 13 participating institutions evaluating 3,885 patients. 1989. J Urol. 2002;167:999–1003; discussion 1004. [PubMed] [Google Scholar]
- 18.Estey EP, Mador DR, McPhee MS. A review of 1486 transurethral resections of the prostate in a teaching hospital. Can J Surg. 1993;36:37–40. [PubMed] [Google Scholar]
- 19.Michielsen DP, Debacker T, De Boe V, Van Lersberghe C, Kaufman L, Braeckman JG, Amy JJ, Keuppens FI. Bipolar transurethral resection in saline--an alternative surgical treatment for bladder outlet obstruction? J Urol. 2007;178:2035–2039; discussion 2039. doi: 10.1016/j.juro.2007.07.038. [DOI] [PubMed] [Google Scholar]
- 20.Hahn RG, Ekengren JC. Patterns of irrigating fluid absorption during transurethral resection of the prostate as indicated by ethanol. J Urol. 1993;149:502–506. doi: 10.1016/s0022-5347(17)36129-3. [DOI] [PubMed] [Google Scholar]
- 21.Istre O, Bjoennes J, Naess R, Hornbaek K, Forman A. Postoperative cerebral oedema after transcervical endometrial resection and uterine irrigation with 1.5% glycine. Lancet. 1994;344:1187–1189. doi: 10.1016/s0140-6736(94)90507-x. [DOI] [PubMed] [Google Scholar]
- 22.Hahn RG. Relations between irrigant absorption rate and hyponatraemia during transurethral resection of the prostate. Acta Anaesthesiol Scand. 1988;32:53–60. doi: 10.1111/j.1399-6576.1988.tb02688.x. [DOI] [PubMed] [Google Scholar]
- 23.Mebust WK, Holtgrewe HL, Cockett AT, Peters PC. Transurethral prostatectomy: immediate and postoperative complications. A cooperative study of 13 participating institutions evaluating 3,885 patients. J Urol. 1989;141:243–247. doi: 10.1016/s0022-5347(17)40731-2. [DOI] [PubMed] [Google Scholar]
- 24.Hahn RG. Smoking increases the risk of large scale fluid absorption during transurethral prostatic resection. J Urol. 2001;166:162–165. [PubMed] [Google Scholar]
- 25.Jr AB. Vesicoureteral reflux and reflux nephropathy. 4th ed. Primer on Kidney Disease. Philadelphia: Elsevier's Rights Department; 2005. pp. 399–403. [Google Scholar]
- 26.Hulten J, Bengtsson M, Engberg A, Hjertberg H, Svedberg J. The pressure in the prostatic fossa and fluid absorption. Scand J Urol Nephrol. 1984;82:33–43. [Google Scholar]
- 27.Olsson J, Nilsson A, Hahn RG. Symptoms of the transurethral resection syndrome using glycine as the irrigant. J Urol. 1995;154:123–128. [PubMed] [Google Scholar]
- 28.Rassweiler J, Teber D, Kuntz R, Hofmann R. Complications of transurethral resection of the prostate (TURP)--incidence, management, and prevention. Eur Urol. 2006;50:969–979; discussion 980. doi: 10.1016/j.eururo.2005.12.042. [DOI] [PubMed] [Google Scholar]
- 29.Hultén JO, Sundström GS. Extravascular absorption of irrigating fluid during TURP. The role of transmural bladder pressure as the driving pressure gradient. Br J Urol. 1990;65:39–42. doi: 10.1111/j.1464-410x.1990.tb14658.x. [DOI] [PubMed] [Google Scholar]
- 30.Hahn RG, Sandfeldt L, Nyman CR. Double-blind randomized study of symptoms associated with absorption of glycine 1.5% or mannitol 3% during transurethral resection of the prostate. J Urol. 1998;160:397–401. [PubMed] [Google Scholar]
- 31.Norlén H, Dimberg M, Vinnars E, Allgén LG, Brandt R. Water and electrolytes in muscle tissue and free amino acids in muscle and plasma in connection with transurethral resection of the prostate. I. Distilled water as an irrigating fluid. Scand J Urol Nephrol. 1990;24:21–26. doi: 10.3109/00365599009180354. [DOI] [PubMed] [Google Scholar]
- 32.Moskovitz B, Ross M, Bolkier M, Rosenberg B, Levin DR. The use of distilled water as an irrigating fluid in patients undergoing transurethral resection of the prostate. Eur Urol. 1989;16:267–270. doi: 10.1159/000471589. [DOI] [PubMed] [Google Scholar]
- 33.Norlén H, Allgén LG, Vinnars E, Wiklund A. Plasma haemoglobin concentrations and other influx variables in blood in connection with transurethral resection of the prostate using distilled water as an irrigating fluid. Scand J Urol Nephrol. 1987;21:161–168. doi: 10.3109/00365598709180317. [DOI] [PubMed] [Google Scholar]
- 34.Hung CL, Wu CJ, Yang S, Chen HH, Lin JS. Acute renal failure directly caused by hemolysis associated with transurethral resection of the prostate. Urology. 2002;59:137. doi: 10.1016/s0090-4295(01)01477-7. [DOI] [PubMed] [Google Scholar]
- 35.Sellevold O, Breivik H, Tveter K. Changes in oncotic pressure, osmolality and electrolytes following transurethral resection of the prostate using glycine as irrigating solution. Scand J Urol Nephrol. 1983;17:31–36. doi: 10.3109/00365598309179777. [DOI] [PubMed] [Google Scholar]
- 36.Hahn RG, Andersson T, Sikk M. Eye symptoms, visual evoked potentials and EEG during intravenous infusion of glycine. Acta Anaesthesiol Scand. 1995;39:214–219. doi: 10.1111/j.1399-6576.1995.tb04046.x. [DOI] [PubMed] [Google Scholar]
- 37.Hahn RG, Sandfeldt L. Blood ammonia levels after intravenous infusion of glycine solution with and without ethanol. Scand J Urol Nephrol. 1999;33:222–227. doi: 10.1080/003655999750015817. [DOI] [PubMed] [Google Scholar]
- 38.Hahn RG. Glycine irrigation and urinary oxalate excretion. Br J Urol. 1989;64:287–289. doi: 10.1111/j.1464-410x.1989.tb06015.x. [DOI] [PubMed] [Google Scholar]
- 39.Dimberg M, Allgén LG, Norlén H, Kolmer T. Experience with hypotonic 2.5% sorbitol solution as an irrigating fluid in transurethral resection of the prostate. Scand J Urol Nephrol. 1987;21:169–176. doi: 10.3109/00365598709180318. [DOI] [PubMed] [Google Scholar]
- 40.Allgén LG, Norlén H, Kolmert T, Berg K. Absorption and elimination of mannitol solution when used as an isotonic irrigating agent in connection with transurethral resection of the prostate. Scand J Urol Nephrol. 1987;21:177–184. doi: 10.3109/00365598709180319. [DOI] [PubMed] [Google Scholar]
- 41.Grove JJ, Shinaman RC, Drover DR. Noncardiogenic pulmonary edema and venous air embolus as complications of operative hysteroscopy. J Clin Anesth. 2004;16:48–50. doi: 10.1016/j.jclinane.2003.03.010. [DOI] [PubMed] [Google Scholar]
- 42.Henderson DJ, Middleton RG. Coma from hyponatremia following transurethral resection of prostate. Urology. 1980;15:267–271. doi: 10.1016/0090-4295(80)90441-0. [DOI] [PubMed] [Google Scholar]
- 43.Ellis SJ. Severe hyponatraemia: complications and treatment. QJM. 1995;88:905–909. [PubMed] [Google Scholar]
- 44.Singer M, Patel M, Webb AR, Bullen C. Management of the transurethral prostate resection syndrome: time for reappraisal? Crit Care Med. 1990;18:1479–1480. doi: 10.1097/00003246-199012000-00031. [DOI] [PubMed] [Google Scholar]
- 45.Hahn RG. Fluid and electrolyte dynamics during development of the TURP syndrome. Br J Urol. 1990;66:79–84. doi: 10.1111/j.1464-410x.1990.tb14869.x. [DOI] [PubMed] [Google Scholar]
- 46.Périer C, Mahul P, Molliex S, Auboyer C, Frey J. Progressive changes in glycine and glycine derivatives in plasma and cerebrospinal fluid after transurethral prostatic resection. Clin Chem. 1990;36:2152–2153. [PubMed] [Google Scholar]
- 47.Hahn RG, Essén P. ECG and cardiac enzymes after glycine absorption in transurethral prostatic resection. Acta Anaesthesiol Scand. 1994;38:550–556. doi: 10.1111/j.1399-6576.1994.tb03950.x. [DOI] [PubMed] [Google Scholar]
- 48.Jin F, Chung F. Minimizing perioperative adverse events in the elderly. Br J Anaesth. 2001;87:608–624. doi: 10.1093/bja/87.4.608. [DOI] [PubMed] [Google Scholar]
- 49.Hatch PD. Surgical and anaesthetic considerations in transurethral resection of the prostate. Anaesth Intensive Care. 1987;15:203–211. doi: 10.1177/0310057X8701500215. [DOI] [PubMed] [Google Scholar]
- 50.Radal M, Bera AP, Leisner C, Haillot O, Autret-Leca E. [Adverse effects of glycolic irrigation solutions] Therapie. 1999;54:233–236. [PubMed] [Google Scholar]
- 51.Hahn RG. Transurethral resection syndrome from extravascular absorption of irrigating fluid. Scand J Urol Nephrol. 1993;27:387–394. doi: 10.3109/00365599309180451. [DOI] [PubMed] [Google Scholar]
- 52.Ghanem AN, Ward JP. Osmotic and metabolic sequelae of volumetric overload in relation to the TUR syndrome. Br J Urol. 1990;66:71–78. doi: 10.1111/j.1464-410x.1990.tb14868.x. [DOI] [PubMed] [Google Scholar]
- 53.Campbell HT, Fincher ME, Sklar AH. Severe hyponatremia without severe hypoosmolality following transurethral resection of the prostate (TURP) in end-stage renal disease. Am J Kidney Dis. 1988;12:152–155. doi: 10.1016/s0272-6386(88)80011-8. [DOI] [PubMed] [Google Scholar]
- 54.Ceccarelli FE, SMITH PC. Studies on fluid and electrolyte alterations during transurethral prostatectomy. II. J Urol. 1961;86:434–441. doi: 10.1016/S0022-5347(17)65195-4. [DOI] [PubMed] [Google Scholar]
- 55.Sandfeldt L, Riddez L, Rajs J, Ewaldsson C, Piros D, Hahn RG. High-dose intravenous infusion of irrigating fluids containing glycine and mannitol in the pig. J Surg Res. 2001;95:114–125. doi: 10.1006/jsre.2000.6028. [DOI] [PubMed] [Google Scholar]
- 56.Hahn RG, Nilsson A, Ståhle L. Distribution and elimination of the solute and water components of urological irrigating fluids. Scand J Urol Nephrol. 1999;33:35–41. doi: 10.1080/003655999750016258. [DOI] [PubMed] [Google Scholar]
- 57.Hahn RG. Serum amino acid patterns and toxicity symptoms following the absorption of irrigant containing glycine in transurethral prostatic surgery. Acta Anaesthesiol Scand. 1988;32:493–501. doi: 10.1111/j.1399-6576.1988.tb02773.x. [DOI] [PubMed] [Google Scholar]
- 58.Evans JW, Singer M, Coppinger SW, Macartney N, Walker JM, Milroy EJ. Cardiovascular performance and core temperature during transurethral prostatectomy. J Urol. 1994;152:2025–2029. doi: 10.1016/s0022-5347(17)32297-8. [DOI] [PubMed] [Google Scholar]
- 59.Sohn MH, Vogt C, Heinen G, Erkens M, Nordmeyer N, Jakse G. Fluid absorption and circulating endotoxins during transurethral resection of the prostate. Br J Urol. 1993;72:605–610. doi: 10.1111/j.1464-410x.1993.tb16218.x. [DOI] [PubMed] [Google Scholar]
- 60.Spasovski G, Vanholder R, Allolio B, Annane D, Ball S, Bichet D, Decaux G, Fenske W, Hoorn EJ, Ichai C, et al. Clinical practice guideline on diagnosis and treatment of hyponatraemia. Nephrol Dial Transplant. 2014;29 Suppl 2:i1–i39. doi: 10.1093/ndt/gfu040. [DOI] [PubMed] [Google Scholar]
- 61.Hahn RG. Blood ammonia concentrations resulting from absorption of irrigating fluid containing glycine and ethanol during transurethral resection of the prostate. Scand J Urol Nephrol. 1991;25:115–119. doi: 10.3109/00365599109024544. [DOI] [PubMed] [Google Scholar]
- 62.Creevy CD, Webb EA. A fatal hemolytic reaction following transurethral resection of the prostate gland; a discussion of its prevention and treatment. Surgery. 1947;21:56–66. [PubMed] [Google Scholar]
- 63.Creevy CD. Hemolytic reactions during transurethral prostatic resection. J Urol. 1947;58:125–131. doi: 10.1016/S0022-5347(17)69530-2. [DOI] [PubMed] [Google Scholar]
- 64.Orłowska-Kowalik G, Janicka L, Ksiazek A. [Acute non-inflammatory renal failure after transurethral electroresection of the prostate combined with irrigation of the bladder with distilled water] Wiad Lek. 1989;42:608–611. [PubMed] [Google Scholar]
- 65.Zager RA, Gamelin LM. Pathogenetic mechanisms in experimental hemoglobinuric acute renal failure. Am J Physiol. 1989;256:F446–F455. doi: 10.1152/ajprenal.1989.256.3.F446. [DOI] [PubMed] [Google Scholar]
- 66.Farag E, Baccala AA, Doutt RF, Ulchaker J, O’Hara J. Laser bladder perforation from photoselective vaporization of prostate resulting in rhabdomyolysis induced acute renal failure. Minerva Anestesiol. 2008;74:277–280. [PubMed] [Google Scholar]
- 67.Adler S. Hyponatremia and rhabdomyolysis: a possible relationship. South Med J. 1980;73:511–513. doi: 10.1097/00007611-198004000-00029. [DOI] [PubMed] [Google Scholar]
- 68.Korzets A, Ori Y, Floro S, Ish-Tov E, Chagnac A, Weinstein T, Zevin D, Gruzman C. Case report: severe hyponatremia after water intoxication: a potential cause of rhabdomyolysis. Am J Med Sci. 1996;312:92–94. doi: 10.1097/00000441-199608000-00007. [DOI] [PubMed] [Google Scholar]
- 69.Gravenstein D. Transurethral resection of the prostate (TURP) syndrome: a review of the pathophysiology and management. Anesth Analg. 1997;84:438–446. doi: 10.1097/00000539-199702000-00037. [DOI] [PubMed] [Google Scholar]
- 70.Wakim KG. The pathophysiologic basis for the clinical manifestations and complications of transurethral prostatic resection. J Urol. 1971;106:719–728. doi: 10.1016/s0022-5347(17)61384-3. [DOI] [PubMed] [Google Scholar]
- 71.Kim MJ, Bachmann A, Mihatsch MJ, Ruszat R, Sulser T, Mayr M. Acute renal failure after continuous flow irrigation in patients treated with potassium-titanyl-phosphate laser vaporization of prostate. Am J Kidney Dis. 2008;51:e19–e24. doi: 10.1053/j.ajkd.2007.11.031. [DOI] [PubMed] [Google Scholar]
- 72.Desmond J. Serum osmolality and plasma electrolytes in patients who develop dilutional hyponatremia during transurethral resection. Can J Surg. 1970;13:116–121. [PubMed] [Google Scholar]
- 73.Paschopoulos M, Polyzos NP, Lavasidis LG, Vrekoussis T, Dalkalitsis N, Paraskevaidis E. Safety issues of hysteroscopic surgery. Ann N Y Acad Sci. 2006;1092:229–234. doi: 10.1196/annals.1365.019. [DOI] [PubMed] [Google Scholar]
- 74.Hahn RG, Ekengren J. Absorption of irrigating fluid and height of fluid bag during transurethral resection of the prostate. Br J Urol. 1993;72:80–83. doi: 10.1111/j.1464-410x.1993.tb06463.x. [DOI] [PubMed] [Google Scholar]
- 75.van Renen RG, Reymann U. Comparison of the effect of two heights of glycine irrigation solution on serum sodium and osmolality during transurethral resection of the prostate. Aust N Z J Surg. 1997;67:874–877. doi: 10.1111/j.1445-2197.1997.tb07617.x. [DOI] [PubMed] [Google Scholar]
- 76.Sharma DP, Harvey AB. Does intraprostatic vasopressin prevent the transurethral resection syndrome? BJU Int. 2000;86:223–226. doi: 10.1046/j.1464-410x.2000.00782.x. [DOI] [PubMed] [Google Scholar]
- 77.Hahn RG. Intravesical pressure during irrigating fluid absorption in transurethral resection of the prostate. Scand J Urol Nephrol. 2000;34:102–108. doi: 10.1080/003655900750016706. [DOI] [PubMed] [Google Scholar]
- 78.Issa MM. Technological advances in transurethral resection of the prostate: bipolar versus monopolar TURP. J Endourol. 2008;22:1587–1595. doi: 10.1089/end.2008.0192. [DOI] [PubMed] [Google Scholar]
- 79.Omar MI, Lam TB, Alexander CE, Graham J, Mamoulakis C, Imamura M, Maclennan S, Stewart F, N’dow J. Systematic review and meta-analysis of the clinical effectiveness of bipolar compared with monopolar transurethral resection of the prostate (TURP) BJU Int. 2014;113:24–35. doi: 10.1111/bju.12281. [DOI] [PubMed] [Google Scholar]
- 80.Malek RS, Kuntzman RS, Barrett DM. High power potassium-titanyl-phosphate laser vaporization prostatectomy. J Urol. 2000;163:1730–1733. [PubMed] [Google Scholar]
- 81.Olsson J, Rentzhog L, Hjertberg H, Hahn RG. Reliability of clinical assessment of fluid absorption in transurethral prostatic resection. Eur Urol. 1993;24:262–266. doi: 10.1159/000474306. [DOI] [PubMed] [Google Scholar]
- 82.Coppinger SW, Lewis CA, Milroy EJ. A method of measuring fluid balance during transurethral resection of the prostate. Br J Urol. 1995;76:66–72. doi: 10.1111/j.1464-410x.1995.tb07834.x. [DOI] [PubMed] [Google Scholar]
- 83.Agarwal R, Emmett M. The post-transurethral resection of prostate syndrome: therapeutic proposals. Am J Kidney Dis. 1994;24:108–111. doi: 10.1016/s0272-6386(12)80169-7. [DOI] [PubMed] [Google Scholar]
- 84.Hahn RG. Hallucination and visual disturbances in transurethral prostatic resection. Intensive Care Med. 1988;14:668–671. doi: 10.1007/BF00256777. [DOI] [PubMed] [Google Scholar]
- 85.Hahn RG, Gebäck T. Fluid volume kinetics of dilutional hyponatremia; a shock syndrome revisited. Clinics (Sao Paulo) 2014;69:120–127. doi: 10.6061/clinics/2014(02)08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Crowley K, Clarkson K, Hannon V, McShane A, Kelly DG. Diuretics after transurethral prostatectomy: a double-blind controlled trial comparing frusemide and mannitol. Br J Anaesth. 1990;65:337–341. doi: 10.1093/bja/65.3.337. [DOI] [PubMed] [Google Scholar]
- 87.Donatucci CF, Deshon GE, Wade CE, Hunt M. Furosemide-induced disturbances of renal function in patients undergoing TURP. Urology. 1990;35:295–300. doi: 10.1016/0090-4295(90)80148-g. [DOI] [PubMed] [Google Scholar]
- 88.Montesinos Baillo A, Banús Gassol JM, Palou Redorta J, Nogueron Castro M, Macias Giménez N. Physiopathology and surgical treatment of extravasated peritoneal fluid after transurethral resection. Eur Urol. 1984;10:183–186. doi: 10.1159/000463784. [DOI] [PubMed] [Google Scholar]
- 89.Gehring H, Nahm W, Baerwald J, Fornara P, Schneeweiss A, Roth-Isigkeit A, Schmucker P. Irrigation fluid absorption during transurethral resection of the prostate: spinal vs. general anaesthesia. Acta Anaesthesiol Scand. 1999;43:458–463. doi: 10.1034/j.1399-6576.1999.430415.x. [DOI] [PubMed] [Google Scholar]
- 90.Kikuno N, Urakami S, Shigeno K, Kishi H, Shiina H, Igawa M. Traumatic rhabdomyolysis resulting from continuous compression in the exaggerated lithotomy position for radical perineal prostatectomy. Int J Urol. 2002;9:521–524. doi: 10.1046/j.1442-2042.2002.00505.x. [DOI] [PubMed] [Google Scholar]
- 91.Gordon BS, Newman W. Lower nephron syndrome following prolonged knee-chest position. J Bone Joint Surg Am. 1953;35-A:764–768. [PubMed] [Google Scholar]
- 92.Wen T, Deibert CM, Siringo FS, Spencer BA. Positioning-related complications of minimally invasive radical prostatectomies. J Endourol. 2014;28:660–667. doi: 10.1089/end.2013.0623. [DOI] [PubMed] [Google Scholar]
- 93.Mattei A, Di Pierro GB, Rafeld V, Konrad C, Beutler J, Danuser H. Positioning injury, rhabdomyolysis, and serum creatine kinase-concentration course in patients undergoing robot-assisted radical prostatectomy and extended pelvic lymph node dissection. J Endourol. 2013;27:45–51. doi: 10.1089/end.2012.0169. [DOI] [PubMed] [Google Scholar]
- 94.Guzzi LM, Mills LM, Greenman P. Rhabdomyolysis, acute renal failure, and the exaggerated lithotomy position. Anesth Analg. 1993;77:635–637. doi: 10.1213/00000539-199309000-00036. [DOI] [PubMed] [Google Scholar]
- 95.Roth JV. Bilateral sciatic and femoral neuropathies, rhabdomyolysis, and acute renal failure caused by positioning during radical retropubic prostatectomy. Anesth Analg. 2007;105:1747–1748, table of contents. doi: 10.1213/01.ane.0000286232.69415.fa. [DOI] [PubMed] [Google Scholar]
- 96.Szewczyk D, Ovadia P, Abdullah F, Rabinovici R. Pressure-induced rhabdomyolysis and acute renal failure. J Trauma. 1998;44:384–388. doi: 10.1097/00005373-199802000-00028. [DOI] [PubMed] [Google Scholar]
- 97.Bosch X, Poch E, Grau JM. Rhabdomyolysis and acute kidney injury. N Engl J Med. 2009;361:62–72. doi: 10.1056/NEJMra0801327. [DOI] [PubMed] [Google Scholar]
- 98.Scharman EJ, Troutman WG. Prevention of kidney injury following rhabdomyolysis: a systematic review. Ann Pharmacother. 2013;47:90–105. doi: 10.1345/aph.1R215. [DOI] [PubMed] [Google Scholar]
- 99.Levey AS, Stevens LA, Schmid CH, Zhang YL, Castro AF, Feldman HI, Kusek JW, Eggers P, Van Lente F, Greene T, et al. A new equation to estimate glomerular filtration rate. Ann Intern Med. 2009;150:604–612. doi: 10.7326/0003-4819-150-9-200905050-00006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Urbschat A, Gauer S, Paulus P, Reissig M, Weipert C, Ramos-Lopez E, Hofmann R, Hadji P, Geiger H, Obermüller N. Serum and urinary NGAL but not KIM-1 raises in human postrenal AKI. Eur J Clin Invest. 2014;44:652–659. doi: 10.1111/eci.12283. [DOI] [PubMed] [Google Scholar]
- 101.Parikh CR, Thiessen-Philbrook H, Garg AX, Kadiyala D, Shlipak MG, Koyner JL, Edelstein CL, Devarajan P, Patel UD, Zappitelli M, et al. Performance of kidney injury molecule-1 and liver fatty acid-binding protein and combined biomarkers of AKI after cardiac surgery. Clin J Am Soc Nephrol. 2013;8:1079–1088. doi: 10.2215/CJN.10971012. [DOI] [PMC free article] [PubMed] [Google Scholar]


