Abstract
Labor resembles an inflammatory response that includes secretion of cytokines/chemokines by resident and infiltrating immune cells into reproductive tissues and the maternal/fetal interface. Untimely activation of these inflammatory pathways leads to preterm labor, which can result in preterm birth. Preterm birth is a major determinant of neonatal mortality and morbidity; therefore, the elucidation of the process of labor at a cellular and molecular level is essential for understanding the pathophysiology of preterm labor. Here, we summarize the role of innate and adaptive immune cells in the physiological or pathological activation of labor. We review published literature regarding the role of innate and adaptive immune cells in the cervix, myometrium, fetal membranes, decidua and the fetus in late pregnancy and labor at term and preterm. Accumulating evidence suggests that innate immune cells (neutrophils, macrophages and mast cells) mediate the process of labor by releasing pro-inflammatory factors such as cytokines, chemokines and matrix metalloproteinases. Adaptive immune cells (T-cell subsets and B cells) participate in the maintenance of fetomaternal tolerance during pregnancy, and an alteration in their function or abundance may lead to labor at term or preterm. Also, immune cells that bridge the innate and adaptive immune systems (natural killer T (NKT) cells and dendritic cells (DCs)) seem to participate in the pathophysiology of preterm labor. In conclusion, a balance between innate and adaptive immune cells is required in order to sustain pregnancy; an alteration of this balance will lead to labor at term or preterm.
Keywords: B cells, cytotoxic T cells, dendritic cells, macrophages, mast cells, neutrophils, NKT cells, parturition, preterm delivery, regulatory T cells, T cells
Introduction
Pregnancy demonstrates the capabilities of the human immune system. The fetus, a semi-allogeneic graft, grows and develops within the mother without succumbing to immunological rejection, a process which depends on the proper establishment of fetomaternal tolerance.1 This tolerance is initiated by the presentation of the paternal–fetal antigen from semen and is facilitated by seminal plasma factors.2,3,4 Antigen is processed by dendritic cells (DCs) and then presented to T cells in the uterine draining lymph nodes.3,5 As a result, antigen-specific regulatory T cells (Tregs) proliferate in order to create peripheral tolerance towards fetal antigens and allow conceptus implantation.6,7 Treg numbers are maintained through pregnancy, creating a tolerogenic anti-inflammatory state or hyporesponsiveness towards paternal antigens until late gestation.7,8,9,10 During late pregnancy, we have proposed that circulating maternal leukocytes (innate and adaptive) are recruited into reproductive tissues (cervix and myometrium) and to the maternal/fetal interface (decidual tissues) by chemotactic processes,11,12,13,14,15,16 where a pro-inflammatory state develops and leads to labor and delivery of the baby.17,18,19 It is thought that the premature activation of this pro-inflammatory pathway can lead to a breakdown of fetomaternal tolerance and play a role in the induction of labor, which subsequently can result in preterm birth.20,21,22
Preterm birth is a major determinant of neonatal mortality and morbidity.23 In 2011, 11.7% of all births in the United States were diagnosed as preterm.23 Among problems occurring after preterm birth are chronic respiratory illnesses, neurodevelopmental disorders and long-term cognitive impairment.24,25,26 However, the mechanisms that lead to preterm birth/labor are poorly understood. The main goal of this review is to summarize the innate and adaptive immune cell components that participate in term and in preterm labor, clarifying the contributions of resident, adaptive leukocytes to the physiological or pathological activation of parturition. Achieving a deeper understanding of the innate and adaptive immune cell components involved in preterm labor might allow us to develop strategies to prolong pregnancy and thereby improve pregnancy outcomes.
Innate immune cells in term and preterm labor
Labor is an inflammatory process.27,28 A number of studies in humans and mice have reported the presence of inflammatory neutrophils and macrophages in the uterus, decidua, cervix and fetal membranes during labor.27,29,30,31,32,33,34 The spreading and homing of these granulocytes is facilitated by chemokines and cellular adhesion molecules.16 Additionally, mast cells are present in the uterus and cervix during late gestation and may contribute to the process of labor.35,36,37,38 Uterine contractions, cervical ripening and dilation, and rupture of the fetal membranes (ROM) are processes which must occur in a timely fashion for a successful delivery.39 Innate immune cells have been linked to these processes by various studies, and this section aims to discuss the possible roles for these cells in the processes of term and preterm labor.
Neutrophils
Neutrophil numbers are higher in the circulation of women who undergo labor than in those who do not undergo labor.40 Labor-related granulocytes are activated since they exhibit an increased ability to migrate.40 The presence of neutrophils in reproductive tissues at term and their ability to migrate to this region during labor have been well documented in both humans and rodents.11,27,29,41 Neutrophils participate in the process of labor by releasing pro-inflammatory cytokines and secreting matrix metalloproteinases (MMPs);42,43,44,45,46,47 however, their role in each anatomical compartment seems to be unique and will be discussed further below.
In the myometrium, mRNA levels of CXCL8, a neutrophil chemoattractant, are higher in women at term during labor than in women without labor,28 suggesting that neutrophils are more abundant in the myometrium during labor. However, it was recently demonstrated in a murine model of infection-induced preterm birth (intrauterine administration of lipopolysaccharide (LPS)) that there is no increase in the neutrophil numbers (Gr-1+ cells) in the myometrium 6 h after LPS administration.48 Preliminary studies in our laboratory put forward evidence on the role of myometrial neutrophils during LPS-induced preterm birth. We found that LPS administration in the peritoneum causes high rates of preterm birth and this is associated with increases in proportion and in absolute numbers of neutrophils in the myometrium (NGL, unpublished data).49 This discrepancy between studies may be due to the fact that we collected tissues prior to delivery (24 h after LPS injection) instead of 6 h after the intrauterine administration of LPS. Nonetheless, the potential role of myometrial neutrophils in the process of labor needs continuing exploration.
Neutrophils are proposed to play a central role in the cervical ripening process,42,43,46,50 although recent studies have indicated that this is likely not the case.51,52 In mice, neutrophil numbers in the cervix do not vary from 15 days postcoitum (dpc) to the time of cervical ripening (late 18 dpc).52 It has been consistently reported that there are no differences in the number of cervical neutrophils between women without labor with cervical ripening and women who had not undergone the ripening process.51 However, the number of neutrophils is higher in the cervixes of women who have just completed a vaginal delivery following spontaneous labor at term than in women who were in the first trimester of pregnancy.51 This finding supports the new hypothesis that neutrophil function is required shortly after parturition, in the phase of postpartum tissue repair.52,53
There have been several studies in mice implicating decidual neutrophils in the process of infection-induced preterm birth.34,48 A large influx of neutrophils into the decidua and myometrium is observed during LPS-induced preterm labor and during term labor; however, this increment was not seen in a non-infectious model of preterm birth (caused by mifepristone).41,54 Another study reported a sevenfold increase in neutrophils in the decidua after 6 h of intrauterine LPS administration.48 Despite these findings, the role of neutrophils as a causative agent of preterm labor is questioned since the depletion of these cells does not alter the timing or success of labor and does not prevent LPS-induced preterm birth.48,52 Neutrophil depletion prior to LPS administration did, however, reduce the amount of a key pro-inflammatory cytokine, IL-1β, in the uteroplacental tissues.48 This finding is relevant since systemic administration of IL-1β leads to preterm birth in mice.55 These results suggest that neutrophils are not a necessary component in infection-induced preterm birth, yet they may be required in inflammation-induced preterm birth.
In human decidual tissues, the number of neutrophils was higher in women with preterm labor associated with chorioamnionitis than in women with term gestations (with and without labor) and in women with spontaneous preterm labor/birth without chorioamnionitis.34 The maternal origin of these decidual leukocytes (e.g., neutrophils) in preterm labor/birth associated with acute chorioamnionitis was proven by FISH.56 Maternal cells could be recruited into this maternal/fetal interface by decidual-derived chemokines, such as CXCL8.11,12 Human decidual neutrophils release several inflammatory mediators and MMPs, which degrade the extracellular matrix of the fetal membranes during both term and preterm labor.44,47,57,58,59,60 Taken together, these data suggest that decidual neutrophils contribute to the physiological ROM and pathological preterm premature rupture of membranes (PPROM) during term and preterm labor.
Macrophages
Macrophages are among the primary innate immune cells that contribute to the processes of term and preterm labor, and their roles have been studied in humans, mice and rats. Macrophages are significant during late gestation primarily due to their secretory products, which include MMPs, IL-1β, IL-6, TNF-α and nitric oxide (NO).61,62,63 These versatile leukocytes are being extensively studied to deepen our understanding of the parturition process. We discuss below the possible effector actions of macrophages in term and preterm labor.
Macrophages play a relevant role in the uterus during parturition. In mice, the number of uterine macrophages at 15 dpc (4 days prior to birth) was significantly higher than in non-pregnant controls though these numbers dropped to non-pregnant levels one day prior to birth.33 This trend for macrophages to decrease immediately prior to labor correlates with another study, performed in rats, which found that NO synthesis in the uterus was elevated during pregnancy but reduced during term labor.64 NO, which can be produced by macrophages,61 has been demonstrated to inhibit myometrial contractions.65 Altogether, these results suggest that a decrease in macrophages, and the resultant reduction in NO, is required for the onset of labor.
Although the aforementioned studies indicate that macrophage numbers decrease in the uterus prior to labor, a study in rats found concentrations of CCL2, a monocyte/macrophage chemoattractant, increased in the myometrium near the time of delivery in comparison to earlier points of gestation and during RU486-induced preterm labor.66 Additionally, macrophages may exert effects on the uterus during parturition through the release of pro-inflammatory cytokines, such as TNFA, which are able to upregulate uterine activation proteins,67 allowing the uterus to prepare for labor. These findings suggest that macrophages are instead recruited into the uterus during labor.
Ripening and dilation of the cervix are the next steps in parturition after the initiation of uterine contractions;39,53 an inflammatory response has been associated with these processes.31,51 During pregnancy at term, but before the onset of labor, women with a ripened cervix were found to have greater numbers of cervical macrophages in comparison to women who were not undergoing cervical ripening.51 A murine model similarly found an increased proportion of macrophages in the cervix the day before birth (18 dpc) in comparison to mid/late gestation (15 dpc).31 A large number of cervical macrophages was also found in antepartum and in LPS-induced preterm labor.21,68 These data suggest the possible involvement of macrophages in cervical remodeling.
Although their presence suggests that macrophages play a role in the cervix during the process of labor, the characterization of these cells also supports this theory. Murine cervical macrophages expressing markers associated with adhesion (CD11bhigh) and migration (CD54) were lower prior to birth (18 dpc) than in mid/late gestation(15 dpc).31 However, macrophages that express markers associated with MMP activation (CD147) and cell matrix remodeling (CD169) are significantly higher at 18 dpc than at 15 dpc.31 These results suggest that cervical macrophages are probably not migrating or binding to vessels prior to birth, but are instead remodeling and degrading the extracellular matrix,31 which are important processes in ripening of the human cervix.69,70 The fact that cervical leukocytes (e.g., macrophages) secrete MMP-9 at term pregnancy71 and that macrophage depletion prevents LPS-induced preterm birth in mice,21 suggests that macrophages are a main source of MMP-9 and contribute to the process of labor at both term and preterm stages. Moreover, macrophage-derived cytokines IL-1β and TNF-α increase the levels of MMP-1, MMP-3 and MMP-9,72 which may be another pathway whereby macrophages participate in the cervical ripening process. Despite the evidence above, it is important to point out that several studies have contrarily suggested that macrophages are not necessary for cervical ripening in mice,52,68,73,74 but play a role in postpartum repair.52,53 Further research on the human cervix during labor and preterm labor must be performed in order to come to a definitive conclusion.
Macrophages could also play a role in the rupture of the fetal membranes since macrophages are recruited by these tissues11 and produce MMP-9.63 MMP-9 concentrations are significantly increased in the fetal membranes during labor, preterm labor and PPROM,58,75,76,77 which directly links this enzyme to physiological ROM and pathological PPROM. Additionally, pro-inflammatory cytokines released by macrophages during labor can regulate the further release of MMPs78 by the fetal membranes, suggesting another mechanism whereby macrophages may contribute to ROM and PPROM.
Macrophages also reside in the decidua near or during the time of labor.14,34 In human decidual tissues, macrophage proportions are higher at term than in preterm gestations without labor.14 Macrophage tissue density is even greater in decidua from women who delivered term and preterm with labor in comparison to women who delivered at term without labor.34 In mice, the proportion of decidual macrophages increases prior to birth (18 dpc) in comparison to mid/late gestation (15 dpc).41 Altogether, these results suggest that decidual macrophages have a role prior to the onset of labor.
Macrophages are also implicated in the etiology of preterm labor since CCL2 concentrations are increased in the amniotic fluid of women delivering preterm, both in the presence and absence of intra-amniotic infection, in comparison to women delivering at term.79 One of the most significant indicators of the role of macrophages in preterm labor was the demonstration that the depletion of macrophages in pregnant mice protected these animals from LPS-induced preterm birth.21 Ultimately, macrophages are potentially involved in several processes during parturition. The precise role of this cell type in labor remains disputed, yet much evidence gives credibility to their putative roles. Further studies are required to fully elucidate the roles of macrophages in the physiological process of labor and the pathological induction of preterm labor.
Currently, we are investigating the role of macrophages during preterm birth using animal models. Our preliminary data suggest that the plasticity of these cells at the maternal/fetal interface is unique, and that besides participating in the process of labor, macrophages play a central role in the maintenance of fetomaternal tolerance during late pregnancy.
Mast cells
Mast cells (MCs) are also important innate immune effector cells during late gestation and labor due to their secretion of mediators.16,80 Fast-acting MC mediators are histamine, serotonin, heparin, proteoglycans, proteases, prostaglandins and leukotrienes.81 MCs also secrete the long-term modulators IL-1β, IL-3, IL-5, IL-6 and TNF-α.36 Moreover, human MCs induce the expression of endothelial adhesion molecules,82 and express several chemokine receptors.83 This combination of MC recruitment and up-regulation of cellular adhesion molecules allows MCs to localize within the uterus and cervix, where they may play a role in the development of a pro-inflammatory environment. Due to their presence and actions in cervical tissue during late gestation, MCs and histamine have been associated with the stimulation of cervical contractility;38 however, MCs have been detected in higher proportions in postpartum than during late gestational cervix which indicates a greater role for this cell type in postpartum uterine cervical repair than during labor.84 We therefore focus the section below on discussing the role of MCs and their mediators in the uterus during term and preterm labor.
MC degranulation releases mediators which likely play major roles in the process of labor by remodeling uterine smooth muscle cells and stimulating uterine contractions.35,36,37,85,86 The release of histamine and serotonin has been linked to uterine contractility since MCs reside adjacent to smooth muscle in the myometrium.35,87 Indeed, during murine pregnancy MCs are more abundant in the myometrium than in the endometrium.88 The degranulation of MCs in vitro, utilizing compound 48/80, induces greater uterine contractility in tissue from late gestation than in non-pregnant uterine tissue.36 In addition, the tissue density of human uterine MCs is greater during pregnancy than in the non-pregnant state, which also suggests that uterine MCs modulate myometrial contractility during late pregnancy.89 A link between MCs and allergy has been suggested as a mechanism of preterm labor/birth37,90 since MCs are one of the cells effecting immediate hypersensitivity reactions and allergic disease81 and allergies play a central role in uterine contractions.37,90 Furthermore, pre-treatment of guinea pigs with a histamine H1 receptor antagonist decreased the rate of preterm birth induced by an allergic reaction.91 This finding suggests a vital role for histamine, and therefore MCs, in the processes of term and preterm labor.
A recent study contradicts the notion of MC involvement in labor. This study found that, in human myometrial tissues, the abundance of MCs was similar at mid-pregnancy and during labor.86 In MC deficient KitW-sh mice, labor still occurs and leukocyte recruitment into the myometrium is not different from wild-type controls.86 A possible explanation is that MCs are not the sole leukocyte recruiters,16 and the pro-inflammatory cascade can be upregulated by other leukocyte subpopulations even in the absence of MCs. Further research is needed in order to clarify the role of mast cells during term and preterm labor.
Adaptive immune cells in term and preterm labor
The adaptive immune system creates memory and responds to specific antigens. During pregnancy, the adaptive immune limbs of both the mother and the fetus must tolerate each other in order to maintain pregnancy until term. A breakdown of this fetomaternal tolerance may lead to labor. In term pregnancy, lack of the tolerogenic state results in physiologic labor. However, a premature retreat of this tolerogenic state might lead to preterm labor.
T cells
During pregnancy, maternal T cells recognize fetal antigens through interactions with antigen-presenting cells.92 Fetal antigen-specific T cells maintain fetomaternal immune tolerance across pregnancy.7 We previously proposed that maternal circulating T cells infiltrate into the maternal/fetal interface prior to delivery and during labor at term.11,93 Decidual T cells are activated and have both a regulatory and an effector phenotype.94,95,96,97 The next section further addresses the putative roles of specific T-cell subsets during late pregnancy and in term and preterm labor.
Effector T cells
We recently provided evidence that decidual CD4+ T cells are involved in term parturition.14 Specifically, we demonstrated that decidual CD4+ T cells are more abundant in term than in preterm gestations without labor. These T cells express CD45RO, but not CD45RA, which suggests that they are memory cells that were generated early in pregnancy when fetal–antigen presentation occurs.7,14,92 We also demonstrated that decidual CD4+ T cells express IL-1β, TNF-α and MMP-9 during spontaneous labor at term.14 The fact that decidual T cells express activation markers such as CD2598 and labor mediators implicated in both term and preterm labor17,29,55,58,75,77,99,100,101,102 suggests that the adaptive limb of the immune system participates during labor.
Additionally, we demonstrated that during term labor T cells are preferentially recruited into the rupture zone of the fetal membranes by chemotactic processes facilitated by CXCL10 and CCL5.13,14,93 However, T-cell attraction to the rupture zone was significantly diminished in premature ROM cases.13 These data suggest that T-cell recruitment into the maternal/fetal interface is required for term pregnancy, and the dysregulation of this recruitment may lead to pathological rupture of membranes.
Th17 cells (CD3+CD4+IL-17A+) also congregate in human decidua,103 and their tissue density is higher in cases of chorioamnionitis than in cases without chorioamnionitis.104 This finding further supports the idea that pro-inflammatory adaptive immune cells at the maternal/fetal interface are associated with chorioamnionitis, which can lead to preterm labor/birth. Studies in our laboratory are currently exploring the potential role for this T-cell subset in preterm labor using LPS-induced and RU486-induced preterm birth models.
Fetal T cells might also play a role during preterm labor. Memory fetal T cells (CD45RO+RA−) are present in higher proportions in cord blood from cases of preterm labor compared to term labor.105 Fetal T cells are also activated (CD25+CD69+) during preterm labor.106 Indeed, acute chorioamnionitis, a leading cause of preterm deliveries, is associated with an increase in cord blood T-cell chemokines (CXCL9, -10 and -11).107 These results suggest that fetal T cells can contribute to the pathophysiology of preterm labor.
Cytotoxic T cells (CTLs) are present at the maternal/fetal interface in term gestations in the absence of labor, where they express perforin and granzyme B.95,97,108 In placenta, CTLs are abundant in cases with villitis of unknown etiology and express T-cell chemokine receptors (CXCR3 and CCR5).107 In peripheral circulation, CD300a+ CTLs have an effector memory phenotype, and their proportion is higher in women with chronic chorioamnionitis than in women without this lesion.109 Taken together, these data suggest that CTLs may participate in pathological inflammation associated with preterm birth, but their role during spontaneous labor at term and preterm requires further exploration.
Tregs
There are two main Treg subsets: thymic Tregs (tTregs) and extrathymic or peripheral Tregs (pTregs). During pregnancy, CD4+ pTregs have been categorized into four subsets: DRhigh+CD45RA−, DRlow+CD45RA−, DR−CD45RA− and naïve DR−CD45RA+.110 The proportion of each subset seems to be relevant in the pathophysiology of pregnancy complications such as preterm labor. Women with preterm labor have a reduced proportion of naïve DR−CD45RA+ Tregs, accompanied by higher proportions of DR−CD45RA− and DRlow+CD45RA− Tregs within their total pTreg pool.110,111 Indeed, the suppressive activity of pTregs is strongly reduced in term and preterm labor,111 which is correlated with a reduction in the expression of HLA-DR in preterm cases.112 This suggests that the lack of suppressive function during late pregnancy could trigger the onset of parturition at term and preterm gestations.113
At term pregnancy, Tregs are found at the maternal/fetal interface, have a unique phenotype (CD4+CD25brightFoxP3+CD69+HLA-DR+CTLA-4+), and exhibit suppressive function in vitro.108,114 However, the role of decidual Tregs remains undetermined. Currently, we are investigating the function and phenotypic characteristics of these cells during term and preterm labor.
Mechanistic studies have successfully demonstrated that the systemic ablation of Tregs by targeting FoxP3+ cells leads to pregnancy failure during early gestation.6,7,115 A separate study suggested that regulatory T cells are not required in late pregnancy;116 however, the targeted depletion of CD25+ cells is not specific for Tregs. Preliminary data from our laboratory demonstrates that systemic depletion of FoxP3+ cells during late gestation does lead to pregnancy complications (NGL, unpublished data).
Additional unpublished data from our laboratory demonstrate that LPS-induced preterm labor causes an expansion of CD4+ Tregs in the spleen and thymus but a reduction of uterine CD4+ Tregs (NGL, unpublished data).117 We also found that the administration of vaginal progesterone, a clinical strategy to prevent preterm birth in women with a short cervix,118 increases the proportion of decidual CD4+ Tregs (NGL, unpublished data).119 Altogether, these data suggest that preterm birth is characterized by altered proportions of CD4+ Tregs at the maternal/fetal interface and that natural progesterone can restore the number of these cells during late pregnancy, preventing preterm birth.
B cells
A few years ago, we suggested a role for B cells during term labor since the fetal membranes from laboring women who delivered at term exhibit B-cell attraction in vitro.12,16Current preliminary data demonstrate that B cells are indeed present in the decidua and cord blood at term and preterm stages (NGL, unpublished data). However, the role of B cells in the processes of term and preterm labor is still under investigation.
Several studies have linked various B cell subsets to pregnancy. B1 cells are present in lower proportions in maternal blood during pregnancy and return to non-pregnant proportions post-partum.120 However, B2 cell frequencies are unchanged by pregnancy in the peripheral blood of women.120 Regulatory B cells exist during early and late gestation and release IL-10.121,122 Regulatory B cells are potential immune players in the development of immunological tolerance, and their presence during mid-gestation may be important in sustaining pregnancy until labor. B10 cells suppress TNF-α secretion by CD4+ T cells during pregnancy,122 and this may regulate the inflammatory state prior to labor. Furthermore, B cells isolated from term placentas produce increased amounts of asymmetric IgG upon stimulation by IL-6, IL-10 and IL-4.123 Therefore, an abnormal disruption of B cell-derived cytokine and asymmetric antibody production could play a part in disturbing fetal tolerance and possibly in eliciting preterm labor.
Bridges between the innate and adaptive immune systems in term and preterm labor
Immune tolerance involves both the innate and adaptive immune systems. Therefore, fetomaternal tolerance must involve the participation of immune cells that bridge the innate and adaptive immune systems, such as DCs and natural killer T (NKT) cells. The roles of these cells during late gestation, labor and preterm labor are discussed below.
NKT cells in term and preterm labor
NKT cells are a unique lymphocyte subpopulation that express markers and characteristics of both the adaptive and innate limbs of the immune system. NKT cells recognize lipid antigens presented by the non-polymorphic CD1D molecule,124 which is expressed by trophoblast cells, placenta, and choriocarcinoma cell lines.125,126 There are two types of NKT cells, type I and type II.127 Type I NKT or invariant NKT (iNKT) cells can be activated by the marine-derived glycolipid α-galactosylceramide,124 and this molecule has been utilized to explore the role of iNKT cell activation during pregnancy in vivo.128,129,130 Therefore, this section will primarily focus on iNKT cells.
During murine pregnancy, NK1.1+TCRαβ+ NKT cells have been observed in the decidua and uterus primarily in early gestation, although they are still present near term (14–18 dpc).129,131 Murine NK1.1+CD3+ NKT cell proportions were higher in the livers of pregnant mice during late gestation (16 dpc) than in non-pregnant controls.132 iNKT cells can secrete large quantities of IL-4 (Th2) and IFN-γ (Th1) upon TCR activation,133 and their activation has been shown to have roles in activating NK cells, B cells and T cells.134 As a result of their immunological effects and their presence during gestation, iNKT cells may participate in pathological or physiological responses during late gestation.
iNKT cells have been involved in the induction of an increased cytotoxic state during human pregnancy complications such as preeclampsia.135 The proportion of iNKT cells expressing activation markers (CD69+), perforin and IFN-γ is increased in the blood of pre-eclamptic women in comparison to pregnant women without this pathology.135 Although the aforementioned study did not address preterm labor, these results indicate that pro-inflammatory iNKT cells are increased during late gestational pregnancy complications; this Th1-like environment could potentially disrupt fetomaternal tolerance and lead to preterm labor.
The role of iNKT cells in the induction of LPS-induced preterm birth has been studied in iNKT cell deficient (Jα18−/−) mice.136 The injection of LPS at 15 dpc caused preterm birth in wild-type mice but not in iNKT cell-deficient mice,136 suggesting that iNKT cells modulate the process of labor induced by microbial products. Conversely, the stimulation of iNKT cells in vivo by injection of α-galactosylceramide during late gestation (16 dpc) induced early preterm birth (17 dpc),128 which may be due to an expansion of NK1.1+TCRαβ+ NKT cells in the uterus.129 In contrast, studies conducted in our laboratory found that the activation of iNKT cells during late gestation (16 dpc) through α-galactosylceramide administration induced late preterm birth (birth at 18 dpc, 28 h post-injection), which is relevant since 70% of all preterm births in woman fall under this category (NGL, unpublished data).130 Current experiments in our laboratory are addressing the immune mechanisms whereby iNKT cell activation leads to late preterm birth.
DCs in term and preterm labor
DCs are specialized in antigen recognition and presentation. DCs exhibit properties that include induction of antigen-specific T-cell activation, T-cell suppression, Treg generation and peripheral tolerance.137,138 Lymphoid CD8α+ DCs (DCs1) induce a Th1 response whereas myeloid CD8α DCs (DCs2) elicit a Th2 response.139,140 A third type of DCs is the inflammatory DCs which initiates a Th1 response as well.141 Due to their immunomodulatory properties, these three subsets of DCs are relevant in the study of fetomaternal tolerance and inflammation during labor and preterm labor.
DCs contribute to fetomaternal tolerance during early pregnancy.142 In mice, uterine DCs have a DC2 phenotype at 15 dpc,143 which suggests that these cells contribute to the tolerogenic state by inducing a local anti-inflammatory (Th2) response during late gestation. Later in pregnancy (17.5 dpc), the predominant DC subset in the uterus is CD11c+CD8α−MHCII− (immature phenotype).144 The fact that immature DCs express the anti-inflammatory cytokine IL-10,144 a potential early biomarker of preterm birth,145 suggests that these cells may participate in the etiology of preterm labor. Moreover, in T and B cell-deficient mice (Rag1−/−) injected with LPS to induce preterm birth, uterine DC activation was observed,146 suggesting the participation of DCs in the induction of labor. Further research is needed in order to establish a role for DCs during late gestation, labor and preterm labor.
Conclusions
During late pregnancy, paternal–fetal antigen-specific memory T cells (including Tregs) participate in the maintenance of fetomaternal peripheral tolerance. Collectively, these cells create an anti-inflammatory environment which will sustain pregnancy. We suggest the following pathway could lead to labor: (1) activation of innate and adaptive immune cells increases their migratory ability; (2) reproductive tissues and the maternal/fetal interface actively recruit the activated cells through the release of chemokines such as CXCL10, CXCL8, CCL2 and CCL5; and (3) infiltrating leukocytes amplify the pro-inflammatory microenvironment at the maternal/fetal interface leading to labor. A triggered stimulus (e.g., infection/inflammation, sterile inflammation, stress, etc.) can cause the premature activation of this pathway, eliciting a shift from an anti-inflammatory to a pro-inflammatory microenvironment and consequently preterm labor (Figure 1).
Figure 1.
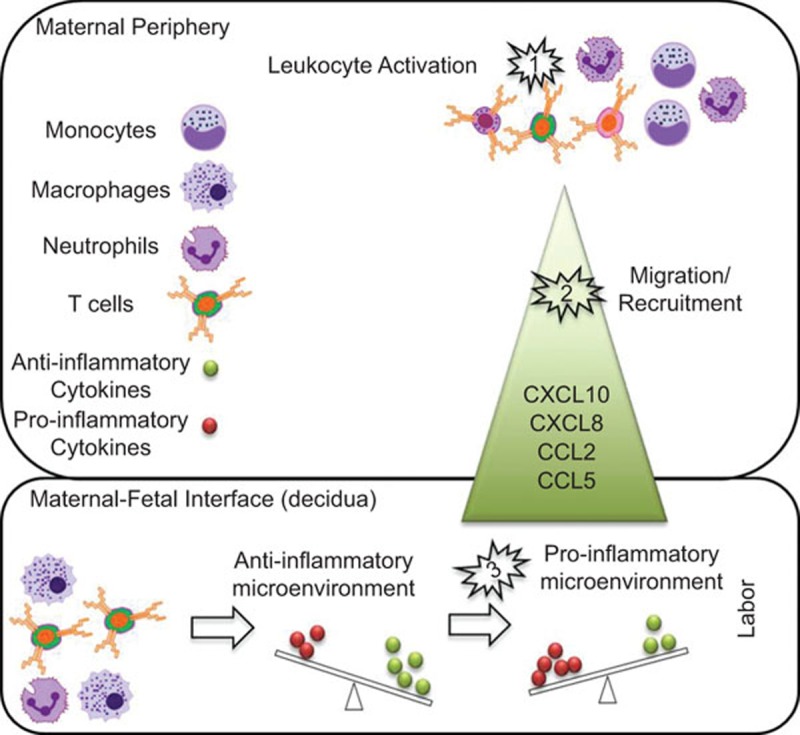
A suggested pathway that leads to term or preterm labor. The following pathway could lead to labor: (1) activation of innate and adaptive immune cells increases their migratory ability; and (2) the maternal/fetal interface actively recruits the activated cells through the release of chemokines such as CXCL10, CXCL8, CCL2 and CCL5; (3) infiltrating leukocytes amplify the pro-inflammatory microenvironment at the maternal/fetal interface leading to labor. A triggered stimulus (e.g., infection/inflammation, sterile inflammation, stress, etc.) can cause the premature activation of this pathway, eliciting a shift from an anti-inflammatory to a pro-inflammatory microenvironment and consequently preterm labor.
An overview of the innate and adaptive immune cells in reproductive tissues and at the maternal/fetal interface during term and preterm labor is shown in Figure 2. Neutrophils are present in the cervix, myometrium, fetal membranes and decidua at term pregnancy; however, their density increases in the myometrium and decidua in term labor and infection-associated preterm labor. Neutrophils are present in the cervix and participate in the repair process during the postpartum period. Macrophages are present in the cervix, myometrium, fetal membranes and decidua at term pregnancy and their density increases in all these tissues, except the cervix, during term and preterm labor. Cervical macrophages also seem to participate in postpartum repair processes. Mast cells are found in cervical and myometrial tissues during late gestation; however, their roles during term and preterm labor are unclear. Effector CD4+ T cells are present in decidual tissues during term labor, and decidual Th17 cells also seem to be involved in the pathology of preterm labor. CTLs are found in term pregnancy and in placental tissues in cases with villitis of unknown etiology; however, their role during labor is unknown. The fetal membranes exhibit B-cell recruitment during term labor, and B cells are found in decidual tissues and cord blood; however, their role in preterm labor is still under investigation. Finally, in myometrial tissues, NKT cell and DC activation seem to be involved in the pathophysiology of preterm labor.
Figure 2.
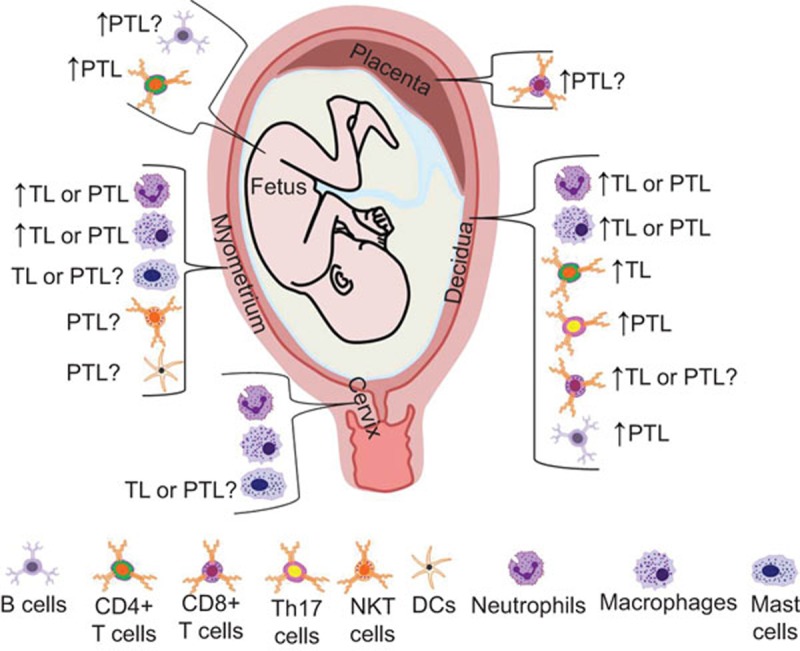
Immune cells in term and preterm labor. Schematic representation of innate and adaptive immune cells in reproductive tissues and at the maternal/fetal interface in term and preterm labor.
Overall, collaboration between the innate and adaptive limbs of the immune system is required to sustain pregnancy until term. A disruption of either limb at term may lead to physiological labor, and an untimely disruption could result in pathological preterm labor. Research targeting the immune cells involved in the process of labor might reveal new strategies to prevent preterm labor and consequently preterm birth.
Acknowledgments
The Gomez-Lopez Laboratory is funded by Wayne State University Research Initiative in Maternal, Perinatal and Child Health, Eunice Kennedy Shriver National Institute of Child Health and Human Development of the National Institutes of Health (NICHD/NIH). This laboratory is also supported, in part, by the Perinatology Research Branch, Division of Intramural Research, Eunice Kennedy Shriver NICHD/NIH.
References
- Finn R, St Hill CA, Davis JC, Hipkin LJ, Harvey M. Feto-maternal bidirectional mixed lymphocyte reaction and survival of fetal allograft. Lancet. 1977;2:1200–1202. doi: 10.1016/s0140-6736(77)90439-1. [DOI] [PubMed] [Google Scholar]
- Robertson SA, Skinner RJ, Care AS. Essential role for IL-10 in resistance to lipopolysaccharide-induced preterm labor in mice. J Immunol. 2006;177:4888–4896. doi: 10.4049/jimmunol.177.7.4888. [DOI] [PubMed] [Google Scholar]
- Robertson SA, Sharkey DJ. The role of semen in induction of maternal immune tolerance to pregnancy. Semin Immunol. 2001;13:243–254. doi: 10.1006/smim.2000.0320. [DOI] [PubMed] [Google Scholar]
- Robertson SA, Mau VJ, Tremellen KP, Seamark RF. Role of high molecular weight seminal vesicle proteins in eliciting the uterine inflammatory response to semen in mice. J Reprod Fertil. 1996;107:265–277. doi: 10.1530/jrf.0.1070265. [DOI] [PubMed] [Google Scholar]
- Beer AE, Scott JR, Billingham RE. Histoincompatibility and maternal immunological status as determinants of fetoplacental weight and litter size in rodents. J Exp Med. 1975;142:180–196. doi: 10.1084/jem.142.1.180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aluvihare VR, Kallikourdis M, Betz AG. Regulatory T cells mediate maternal tolerance to the fetus. Nat Immunol. 2004;5:266–271. doi: 10.1038/ni1037. [DOI] [PubMed] [Google Scholar]
- Rowe JH, Ertelt JM, Xin L, Way SS. Pregnancy imprints regulatory memory that sustains anergy to fetal antigen. Nature. 2012;490:102–106. doi: 10.1038/nature11462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith RN, Powell AE. The adoptive transfer of pregnancy-induced unresponsiveness to male skin grafts with thymus-dependent cells. J Exp Med. 1977;146:899–904. doi: 10.1084/jem.146.3.899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chaouat G, Voisin GA, Escalier D, Robert P. Facilitation reaction (enhancing antibodies and suppressor cells) and rejection reaction (sensitized cells) from the mother to the paternal antigens of the conceptus. Clin Exp Immunol. 1979;35:13–24. [PMC free article] [PubMed] [Google Scholar]
- Chaouat G, Petitbarat M, Dubanchet S, Rahmati M, Ledee N. Tolerance to the foetal allograft. Am J Reprod Immunol. 2010;63:624–636. doi: 10.1111/j.1600-0897.2010.00832.x. [DOI] [PubMed] [Google Scholar]
- Gomez-Lopez N, Estrada-Gutierrez G, Jimenez-Zamudio L, Vega-Sanchez R, Vadillo-Ortega F. Fetal membranes exhibit selective leukocyte chemotaxic activity during human labor. J Reprod Immunol. 2009;80:122–131. doi: 10.1016/j.jri.2009.01.002. [DOI] [PubMed] [Google Scholar]
- Gomez-Lopez N, Vadillo-Perez L, Nessim S, Olson DM, Vadillo-Ortega F. Choriodecidua and amnion exhibit selective leukocyte chemotaxis during term human labor. Am J Obstet Gynecol. 2011;204:5. doi: 10.1016/j.ajog.2010.11.010. [DOI] [PubMed] [Google Scholar]
- Gomez-Lopez N, Hernandez-Santiago S, Lobb AP, Olson DM, Vadillo-Ortega F. Normal and premature rupture of fetal membranes at term delivery differ in regional chemotactic activity and related chemokine/cytokine production. Reprod Sci. 2013;20:276–284. doi: 10.1177/1933719112452473. [DOI] [PubMed] [Google Scholar]
- Gomez-Lopez N, Vega-Sanchez R, Castillo-Castrejon M, Romero R, Cubeiro-Arreola K, Vadillo-Ortega F. Evidence for a role for the adaptive immune response in human term parturition. Am J Reprod Immunol. 2013;69:212–230. doi: 10.1111/aji.12074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gomez-Lopez N, Tanaka S, Zaeem Z, Metz GA, Olson DM. Maternal circulating leukocytes display early chemotactic responsiveness during late gestation. BMC Pregnancy Childbirth. 2013;13 Suppl 1:S8. doi: 10.1186/1471-2393-13-S1-S8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gomez-Lopez N, Guilbert LJ, Olson DM. Invasion of the leukocytes into the fetal–maternal interface during pregnancy. J Leukoc Biol. 2010;88:625–633. doi: 10.1189/jlb.1209796. [DOI] [PubMed] [Google Scholar]
- Romero R, Brody DT, Oyarzun E, Mazor M, Wu YK, Hobbins JC, et al. Infection and labor. III. Interleukin-1: a signal for the onset of parturition. Am J Obstet Gynecol. 1989;160:1117–1123. doi: 10.1016/0002-9378(89)90172-5. [DOI] [PubMed] [Google Scholar]
- Romero R, Espinoza J, Goncalves LF, Kusanovic JP, Friel LA, Nien JK. Inflammation in preterm and term labour and delivery. Semin Fetal Neonatal Med. 2006;11:317–326. doi: 10.1016/j.siny.2006.05.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Romero R, Espinoza J, Kusanovic JP, Gotsch F, Hassan S, Erez O, et al. The preterm parturition syndrome. BJOG. 2006;113 Suppl 3:17–42. doi: 10.1111/j.1471-0528.2006.01120.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tornblom SA, Klimaviciute A, Bystrom B, Chromek M, Brauner A, Ekman-Ordeberg G. Non-infected preterm parturition is related to increased concentrations of IL-6, IL-8 and MCP-1 in human cervix. Reprod Biol Endocrinol. 2005;3:39. doi: 10.1186/1477-7827-3-39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gonzalez JM, Franzke CW, Yang F, Romero R, Girardi G. Complement activation triggers metalloproteinases release inducing cervical remodeling and preterm birth in mice. Am J Pathol. 2011;179:838–849. doi: 10.1016/j.ajpath.2011.04.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee J, Romero R, Xu Y, Kim JS, Topping V, Yoo W, et al. A signature of maternal anti-fetal rejection in spontaneous preterm birth: chronic chorioamnionitis, anti-human leukocyte antigen antibodies, and C4d. PLoS ONE. 2011;6:e16806. doi: 10.1371/journal.pone.0016806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin JA, Hamilton BE, Ventura SJ, Osterman MJ, Kirmeyer S, Mathews TJ, et al. Births: final data for 2009. Natl Vital Stat Rep. 2011;60:1–70. [PubMed] [Google Scholar]
- Goldenberg RL, Hauth JC, Andrews WW. Intrauterine infection and preterm delivery. N Engl J Med. 2000;342:1500–1507. doi: 10.1056/NEJM200005183422007. [DOI] [PubMed] [Google Scholar]
- Hack M, Fanaroff AA. Outcomes of extremely immature infants—a perinatal dilemma. N Engl J Med. 1993;329:1649–1650. doi: 10.1056/NEJM199311253292210. [DOI] [PubMed] [Google Scholar]
- McCormick MC. The contribution of low birth weight to infant mortality and childhood morbidity. N Engl J Med. 1985;312:82–90. doi: 10.1056/NEJM198501103120204. [DOI] [PubMed] [Google Scholar]
- Thomson AJ, Telfer JF, Young A, Campbell S, Stewart CJ, Cameron IT, et al. Leukocytes infiltrate the myometrium during human parturition: further evidence that labour is an inflammatory process. Hum Reprod. 1999;14:229–236. [PubMed] [Google Scholar]
- Bollapragada S, Youssef R, Jordan F, Greer I, Norman J, Nelson S. Term labor is associated with a core inflammatory response in human fetal membranes, myometrium, and cervix. Am J Obstet Gynecol. 2009;200:104.e101–104.111. doi: 10.1016/j.ajog.2008.08.032. [DOI] [PubMed] [Google Scholar]
- Osman I, Young A, Ledingham MA, Thomson AJ, Jordan F, Greer IA, et al. Leukocyte density and pro-inflammatory cytokine expression in human fetal membranes, decidua, cervix and myometrium before and during labour at term. Mol Hum Reprod. 2003;9:41–45. doi: 10.1093/molehr/gag001. [DOI] [PubMed] [Google Scholar]
- Bokstrom H, Brannstrom M, Alexandersson M, Norstrom A. Leukocyte subpopulations in the human uterine cervical stroma at early and term pregnancy. Hum Reprod. 1997;12:586–590. doi: 10.1093/humrep/12.3.586. [DOI] [PubMed] [Google Scholar]
- Payne KJ, Clyde LA, Weldon AJ, Milford TA, Yellon SM. Residency and activation of myeloid cells during remodeling of the prepartum murine cervix. Biol Reprod. 2012;87:106. doi: 10.1095/biolreprod.112.101840. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Young A, Thomson AJ, Ledingham M, Jordan F, Greer IA, Norman JE. Immunolocalization of proinflammatory cytokines in myometrium, cervix, and fetal membranes during human parturition at term. Biol Reprod. 2002;66:445–449. doi: 10.1095/biolreprod66.2.445. [DOI] [PubMed] [Google Scholar]
- Mackler AM, Iezza G, Akin MR, McMillan P, Yellon SM. Macrophage trafficking in the uterus and cervix precedes parturition in the mouse. Biol Reprod. 1999;61:879–883. doi: 10.1095/biolreprod61.4.879. [DOI] [PubMed] [Google Scholar]
- Hamilton S, Oomomian Y, Stephen G, Shynlova O, Tower CL, Garrod A, et al. Macrophages infiltrate the human and rat decidua during term and preterm labor: evidence that decidual inflammation precedes labor. Biol Reprod. 2012;86:39. doi: 10.1095/biolreprod.111.095505. [DOI] [PubMed] [Google Scholar]
- Rudolph MI, Bardisa L, Cruz MA, Reinicke K. Mast cells mediators evoke contractility and potentiate each other in mouse uterine horns. Gen Pharmacol. 1992;23:833–836. doi: 10.1016/0306-3623(92)90233-a. [DOI] [PubMed] [Google Scholar]
- Garfield RE, Bytautiene E, Vedernikov YP, Marshall JS, Romero R. Modulation of rat uterine contractility by mast cells and their mediators. Am J Obstet Gynecol. 2000;183:118–125. doi: 10.1067/mob.2000.105741. [DOI] [PubMed] [Google Scholar]
- Bytautiene E, Vedernikov YP, Saade GR, Romero R, Garfield RE. IgE-independent mast cell activation augments contractility of nonpregnant and pregnant guinea pig myometrium. Int Arch Allergy Immunol. 2008;147:140–146. doi: 10.1159/000135701. [DOI] [PubMed] [Google Scholar]
- Bytautiene E, Vedernikov YP, Saade GR, Romero R, Garfield RE. Endogenous mast cell degranulation modulates cervical contractility in the guinea pig. Am J Obstet Gynecol. 2002;186:438–445. doi: 10.1067/mob.2002.120488. [DOI] [PubMed] [Google Scholar]
- Romero R, Mazor M, Munoz H, Gomez R, Galasso M, Sherer DM. The preterm labor syndrome. Ann NY Acad Sci. 1994;734:414–429. doi: 10.1111/j.1749-6632.1994.tb21771.x. [DOI] [PubMed] [Google Scholar]
- Yuan M, Jordan F, McInnes IB, Harnett MM, Norman JE. Leukocytes are primed in peripheral blood for activation during term and preterm labour. Mol Hum Reprod. 2009;15:713–724. doi: 10.1093/molehr/gap054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shynlova O, Nedd-Roderique T, Li Y, Dorogin A, Nguyen T, Lye SJ. Infiltration of myeloid cells into decidua is a critical early event in the labour cascade and post-partum uterine remodelling. J Cell Mol Med. 2013;17:311–324. doi: 10.1111/jcmm.12012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Junqueira LC, Zugaib M, Montes GS, Toledo OM, Krisztan RM, Shigihara KM. Morphologic and histochemical evidence for the occurrence of collagenolysis and for the role of neutrophilic polymorphonuclear leukocytes during cervical dilation. Am J Obstet Gynecol. 1980;138:273–281. doi: 10.1016/0002-9378(80)90248-3. [DOI] [PubMed] [Google Scholar]
- Osmers R, Rath W, Adelmann-Grill BC, Fittkow C, Kuloczik M, Szeverenyi M, et al. Origin of cervical collagenase during parturition. Am J Obstet Gynecol. 1992;166:1455–1460. doi: 10.1016/0002-9378(92)91619-l. [DOI] [PubMed] [Google Scholar]
- Romero R, Ceska M, Avila C, Mazor M, Behnke E, Lindley I. Neutrophil attractant/activating peptide-1/interleukin-8 in term and preterm parturition. Am J Obstet Gynecol. 1991;165:813–820. doi: 10.1016/0002-9378(91)90422-n. [DOI] [PubMed] [Google Scholar]
- Osmers RG, Blaser J, Kuhn W, Tschesche H. Interleukin-8 synthesis and the onset of labor. Obstet Gynecol. 1995;86:223–229. doi: 10.1016/0029-7844(95)93704-4. [DOI] [PubMed] [Google Scholar]
- Winkler M, Fischer DC, Ruck P, Marx T, Kaiserling E, Oberpichler A, et al. Parturition at term: parallel increases in interleukin-8 and proteinase concentrations and neutrophil count in the lower uterine segment. Hum Reprod. 1999;14:1096–1100. doi: 10.1093/humrep/14.4.1096. [DOI] [PubMed] [Google Scholar]
- Helmig BR, Romero R, Espinoza J, Chaiworapongsa T, Bujold E, Gomez R, et al. Neutrophil elastase and secretory leukocyte protease inhibitor in prelabor rupture of membranes, parturition and intra-amniotic infection. J Matern Fetal Neonatal Med. 2002;12:237–246. doi: 10.1080/jmf.12.4.237.246. [DOI] [PubMed] [Google Scholar]
- Rinaldi SF, Catalano RD, Wade J, Rossi AG, Norman JE. Decidual neutrophil infiltration is not required for preterm birth in a mouse model of infection-induced preterm labor. J Immunol. 2014;192:2315–2325. doi: 10.4049/jimmunol.1302891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gomez-Lopez N, Mial T, Robertson SA.Infection-induced preterm delivery does not involve a shift in macrophage polarization from the M2→M1 phenotype but implicates a maternal pro-inflammatory state. Am J Reprod Immunol 2014. in press.
- Yellon SM, Ebner CA, Elovitz MA. Medroxyprogesterone acetate modulates remodeling, immune cell census, and nerve fibers in the cervix of a mouse model for inflammation-induced preterm birth. Reprod Sci. 2009;16:257–264. doi: 10.1177/1933719108325757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sakamoto Y, Moran P, Bulmer JN, Searle RF, Robson SC. Macrophages and not granulocytes are involved in cervical ripening. J Reprod Immunol. 2005;66:161–173. doi: 10.1016/j.jri.2005.04.005. [DOI] [PubMed] [Google Scholar]
- Timmons BC, Mahendroo MS. Timing of neutrophil activation and expression of proinflammatory markers do not support a role for neutrophils in cervical ripening in the mouse. Biol Reprod. 2006;74:236–245. doi: 10.1095/biolreprod.105.044891. [DOI] [PubMed] [Google Scholar]
- Timmons B, Akins M, Mahendroo M. Cervical remodeling during pregnancy and parturition. Trends Endocrinol Metab. 2010;21:353–361. doi: 10.1016/j.tem.2010.01.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shynlova O, Nedd-Roderique T, Li Y, Dorogin A, Lye SJ. Myometrial immune cells contribute to term parturition, preterm labour and post-partum involution in mice. J Cell Mol Med. 2013;17:90–102. doi: 10.1111/j.1582-4934.2012.01650.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Romero R, Mazor M, Tartakovsky B. Systemic administration of interleukin-1 induces preterm parturition in mice. Am J Obstet Gynecol. 1991;165:969–971. doi: 10.1016/0002-9378(91)90450-6. [DOI] [PubMed] [Google Scholar]
- Steel JH, O'Donoghue K, Kennea NL, Sullivan MH, Edwards AD. Maternal origin of inflammatory leukocytes in preterm fetal membranes, shown by fluorescence in situ hybridisation. Placenta. 2005;26:672–677. doi: 10.1016/j.placenta.2004.10.003. [DOI] [PubMed] [Google Scholar]
- Dudley DJ, Trautman MS, Mitchell MD. Inflammatory mediators regulate interleukin-8 production by cultured gestational tissues: evidence for a cytokine network at the chorio-decidual interface. J Clin Endocrinol Metab. 1993;76:404–410. doi: 10.1210/jcem.76.2.8432783. [DOI] [PubMed] [Google Scholar]
- Vadillo OF, Gonzalez AG, Furth EE, Lei H, Muschel RJ, Stetler-Stevenson WG, et al. 92-kd type IV collagenase (matrix metalloproteinase-9) activity in human amniochorion increases with labor. Am J Pathol. 1995;146:148–156. [PMC free article] [PubMed] [Google Scholar]
- Athayde N, Romero R, Gomez R, Maymon E, Pacora P, Mazor M, et al. Matrix metalloproteinases-9 in preterm and term human parturition. J Matern Fetal Med. 1999;8:213–219. doi: 10.1002/(SICI)1520-6661(199909/10)8:5<213::AID-MFM3>3.0.CO;2-R. [DOI] [PubMed] [Google Scholar]
- Maymon E, Romero R, Pacora P, Gomez R, Athayde N, Edwin S, et al. Human neutrophil collagenase (matrix metalloproteinase 8) in parturition, premature rupture of the membranes, and intrauterine infection. Am J Obstet Gynecol. 2000;183:94–99. doi: 10.1067/mob.2000.105344. [DOI] [PubMed] [Google Scholar]
- Hibbs JB, Jr, Taintor RR, Vavrin Z, Rachlin EM. Nitric oxide: a cytotoxic activated macrophage effector molecule. Biochem Biophys Res Commun. 1988;157:87–94. doi: 10.1016/s0006-291x(88)80015-9. [DOI] [PubMed] [Google Scholar]
- Pavlov O, Pavlova O, Ailamazyan E, Selkov S. Characterization of cytokine production by human term placenta macrophages in vitro. Am J Reprod Immunol. 2008;60:556–567. doi: 10.1111/j.1600-0897.2008.00657.x. [DOI] [PubMed] [Google Scholar]
- Huang WC, Sala-Newby GB, Susana A, Johnson JL, Newby AC. Classical macrophage activation up-regulates several matrix metalloproteinases through mitogen activated protein kinases and nuclear factor-kappaB. PLoS ONE. 2012;7:e42507. doi: 10.1371/journal.pone.0042507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buhimschi I, Ali M, Jain V, Chwalisz K, Garfield RE. Differential regulation of nitric oxide in the rat uterus and cervix during pregnancy and labour. Hum Reprod. 1996;11:1755–1766. doi: 10.1093/oxfordjournals.humrep.a019481. [DOI] [PubMed] [Google Scholar]
- Izumi H, Yallampalli C, Garfield RE. Gestational changes in L-arginine-induced relaxation of pregnant rat and human myometrial smooth muscle. Am J Obstet Gynecol. 1993;169:1327–1337. doi: 10.1016/0002-9378(93)90301-x. [DOI] [PubMed] [Google Scholar]
- Shynlova O, Tsui P, Dorogin A, Lye SJ. Monocyte chemoattractant protein-1 (CCL-2) integrates mechanical and endocrine signals that mediate term and preterm labor. J Immunol. 2008;181:1470–1479. doi: 10.4049/jimmunol.181.2.1470. [DOI] [PubMed] [Google Scholar]
- Fang X, Wong S, Mitchell BF. Effects of LPS and IL-6 on oxytocin receptor in non-pregnant and pregnant rat uterus. Am J Reprod Immunol. 2000;44:65–72. doi: 10.1111/j.8755-8920.2000.440201.x. [DOI] [PubMed] [Google Scholar]
- Gonzalez JM, Dong Z, Romero R, Girardi G. Cervical remodeling/ripening at term and preterm delivery: the same mechanism initiated by different mediators and different effector cells. PLoS ONE. 2011;6:e26877. doi: 10.1371/journal.pone.0026877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hassan SS, Romero R, Tarca AL, Draghici S, Pineles B, Bugrim A, et al. Signature pathways identified from gene expression profiles in the human uterine cervix before and after spontaneous term parturition. Am J Obstet Gynecol. 2007;197:250.e251–250.257. doi: 10.1016/j.ajog.2007.07.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hassan SS, Romero R, Tarca AL, Nhan-Chang CL, Vaisbuch E, Erez O, et al. The transcriptome of cervical ripening in human pregnancy before the onset of labor at term: identification of novel molecular functions involved in this process. J Matern Fetal Neonatal Med. 2009;22:1183–1193. doi: 10.3109/14767050903353216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stygar D, Wang H, Vladic YS, Ekman G, Eriksson H, Sahlin L. Increased level of matrix metalloproteinases 2 and 9 in the ripening process of the human cervix. Biol Reprod. 2002;67:889–894. doi: 10.1095/biolreprod.102.005116. [DOI] [PubMed] [Google Scholar]
- Watari M, Watari H, DiSanto ME, Chacko S, Shi GP, Strauss JF., 3rd Pro-inflammatory cytokines induce expression of matrix-metabolizing enzymes in human cervical smooth muscle cells. Am J Pathol. 1999;154:1755–1762. doi: 10.1016/S0002-9440(10)65431-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Word RA, Landrum CP, Timmons BC, Young SG, Mahendroo MS. Transgene insertion on mouse chromosome 6 impairs function of the uterine cervix and causes failure of parturition. Biol Reprod. 2005;73:1046–1056. doi: 10.1095/biolreprod.105.042663. [DOI] [PubMed] [Google Scholar]
- Gonzalez JM, Xu H, Chai J, Ofori E, Elovitz MA. Preterm and term cervical ripening in CD1 Mice (Mus musculus): similar or divergent molecular mechanisms. Biol Reprod. 2009;81:1226–1232. doi: 10.1095/biolreprod.108.075309. [DOI] [PubMed] [Google Scholar]
- Vadillo-Ortega F, Hernandez A, Gonzalez-Avila G, Bermejo L, Iwata K, Strauss JF 3 rd. Increased matrix metalloproteinase activity and reduced tissue inhibitor of metalloproteinases-1 levels in amniotic fluids from pregnancies complicated by premature rupture of membranes. Am J Obstet Gynecol. 1996;174:1371–1376. doi: 10.1016/s0002-9378(96)70687-7. [DOI] [PubMed] [Google Scholar]
- Athayde N, Edwin SS, Romero R, Gomez R, Maymon E, Pacora P, et al. A role for matrix metalloproteinase-9 in spontaneous rupture of the fetal membranes. Am J Obstet Gynecol. 1998;179:1248–1253. doi: 10.1016/s0002-9378(98)70141-3. [DOI] [PubMed] [Google Scholar]
- Xu P, Alfaidy N, Challis JR. Expression of matrix metalloproteinase (MMP)-2 and MMP-9 in human placenta and fetal membranes in relation to preterm and term labor. J Clin Endocrinol Metab. 2002;87:1353–1361. doi: 10.1210/jcem.87.3.8320. [DOI] [PubMed] [Google Scholar]
- Braundmeier AG, Nowak RA. Cytokines regulate matrix metalloproteinases in human uterine endometrial fibroblast cells through a mechanism that does not involve increases in extracellular matrix metalloproteinase inducer. Am J Reprod Immunol. 2006;56:201–214. doi: 10.1111/j.1600-0897.2006.00418.x. [DOI] [PubMed] [Google Scholar]
- Esplin MS, Romero R, Chaiworapongsa T, Kim YM, Edwin S, Gomez R, et al. Monocyte chemotactic protein-1 is increased in the amniotic fluid of women who deliver preterm in the presence or absence of intra-amniotic infection. J Matern Fetal Neonatal Med. 2005;17:365–373. doi: 10.1080/14767050500141329. [DOI] [PubMed] [Google Scholar]
- Menzies FM, Shepherd MC, Nibbs RJ, Nelson SM. The role of mast cells and their mediators in reproduction, pregnancy and labour. Hum Reprod Update. 2011;17:383–396. doi: 10.1093/humupd/dmq053. [DOI] [PubMed] [Google Scholar]
- Abbas AK, Lichtman AH, Pillai S. Cellular and Molecular Immunology. 7th ed. Philadelphia, PA: Elsevier/Saunders; 2012. [Google Scholar]
- Walsh LJ, Trinchieri G, Waldorf HA, Whitaker D, Murphy GF. Human dermal mast cells contain and release tumor necrosis factor alpha, which induces endothelial leukocyte adhesion molecule 1. Proc Natl Acad Sci USA. 1991;88:4220–4224. doi: 10.1073/pnas.88.10.4220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Juremalm M, Nilsson G. Chemokine receptor expression by mast cells. Chem Immunol Allergy. 2005;87:130–144. doi: 10.1159/000087640. [DOI] [PubMed] [Google Scholar]
- Bosquiazzo VL, Durando M, Varayoud J, Ramos JG, Rodriguez HA, Munoz-de-Toro M, et al. Macrophage density in the pregnant rat uterine cervix is modulated by mast cell degranulation. J Reprod Immunol. 2005;65:147–158. doi: 10.1016/j.jri.2005.01.002. [DOI] [PubMed] [Google Scholar]
- Bytautiene E, Vedernikov YP, Saade GR, Romero R, Garfield RE. Degranulation of uterine mast cell modifies contractility of isolated myometrium from pregnant women. Am J Obstet Gynecol. 2004;191:1705–1710. doi: 10.1016/j.ajog.2004.04.008. [DOI] [PubMed] [Google Scholar]
- Menzies FM, Higgins CA, Shepherd MC, Nibbs RJ, Nelson SM. Mast cells reside in myometrium and cervix, but are dispensable in mice for successful pregnancy and labor. Immunol Cell Biol. 2012;90:321–329. doi: 10.1038/icb.2011.40. [DOI] [PubMed] [Google Scholar]
- Rudolph MI, Reinicke K, Cruz MA, Gallardo V, Gonzalez C, Bardisa L. Distribution of mast cells and the effect of their mediators on contractility in human myometrium. Br J Obstet Gynaecol. 1993;100:1125–1130. doi: 10.1111/j.1471-0528.1993.tb15178.x. [DOI] [PubMed] [Google Scholar]
- Padilla L, Reinicke K, Montesino H, Villena F, Asencio H, Cruz M, et al. Histamine content and mast cells distribution in mouse uterus: the effect of sexual hormones, gestation and labor. Cell Mol Biol. 1990;36:93–100. [PubMed] [Google Scholar]
- Garfield RE, Irani AM, Schwartz LB, Bytautiene E, Romero R. Structural and functional comparison of mast cells in the pregnant versus nonpregnant human uterus. Am J Obstet Gynecol. 2006;194:261–267. doi: 10.1016/j.ajog.2005.05.011. [DOI] [PubMed] [Google Scholar]
- Bytautiene E, Vedernikov YP, Maner WL, Saade GR, Romero R, Garfield RE. Challenge with ovalbumin antigen increases uterine and cervical contractile activity in sensitized guinea pigs. Am J Obstet Gynecol. 2008;199:658.e651–658.656. doi: 10.1016/j.ajog.2008.06.036. [DOI] [PubMed] [Google Scholar]
- Bytautiene E, Romero R, Vedernikov YP, El-Zeky F, Saade GR, Garfield RE. Induction of premature labor and delivery by allergic reaction and prevention by histamine H1 receptor antagonist. Am J Obstet Gynecol. 2004;191:1356–1361. doi: 10.1016/j.ajog.2004.06.092. [DOI] [PubMed] [Google Scholar]
- Erlebacher A, Vencato D, Price KA, Zhang D, Glimcher LH. Constraints in antigen presentation severely restrict T cell recognition of the allogeneic fetus. J Clin Invest. 2007;117:1399–1411. doi: 10.1172/JCI28214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gomez-Lopez N, Vadillo-Perez L, Hernandez-Carbajal A, Godines-Enriquez M, Olson DM, Vadillo-Ortega F. Specific inflammatory microenvironments in the zones of the fetal membranes at term delivery. Am J Obstet Gynecol. 2011;205:16. doi: 10.1016/j.ajog.2011.04.019. [DOI] [PubMed] [Google Scholar]
- Sindram-Trujillo A, Scherjon S, Kanhai H, Roelen D, Claas F. Increased T-cell activation in decidua parietalis compared to decidua basalis in uncomplicated human term pregnancy. Am J Reprod Immunol. 2003;49:261–268. doi: 10.1034/j.1600-0897.2003.00041.x. [DOI] [PubMed] [Google Scholar]
- Tilburgs T, Scherjon SA, Roelen DL, Claas FH. Decidual CD8+CD28− T cells express CD103 but not perforin. Hum Immunol. 2009;70:96–100. doi: 10.1016/j.humimm.2008.12.006. [DOI] [PubMed] [Google Scholar]
- Tilburgs T, Scherjon SA, van der Mast BJ, Haasnoot GW, Versteeg VDV-MM, Roelen DL, et al. Fetal–maternal HLA-C mismatch is associated with decidual T cell activation and induction of functional T regulatory cells. J Reprod Immunol. 2009;82:148–157. doi: 10.1016/j.jri.2009.05.003. [DOI] [PubMed] [Google Scholar]
- Tilburgs T, Schonkeren D, Eikmans M, Nagtzaam NM, Datema G, Swings GM, et al. Human decidual tissue contains differentiated CD8+ effector-memory T cells with unique properties. J Immunol. 2010;185:4470–4477. doi: 10.4049/jimmunol.0903597. [DOI] [PubMed] [Google Scholar]
- Sindram-Trujillo AP, Scherjon SA, van Hulst-van Miert PP, Kanhai HH, Roelen DL, Claas FH. Comparison of decidual leukocytes following spontaneous vaginal delivery and elective cesarean section in uncomplicated human term pregnancy. J Reprod Immunol. 2004;62:125–137. doi: 10.1016/j.jri.2003.11.007. [DOI] [PubMed] [Google Scholar]
- Romero R, Parvizi ST, Oyarzun E, Mazor M, Wu YK, Avila C, et al. Amniotic fluid interleukin-1 in spontaneous labor at term. J Reprod Med. 1990;35:235–238. [PubMed] [Google Scholar]
- Romero R, Mazor M, Brandt F, Sepulveda W, Avila C, Cotton DB, et al. Interleukin-1 alpha and interleukin-1 beta in preterm and term human parturition. Am J Reprod Immunol. 1992;27:117–123. doi: 10.1111/j.1600-0897.1992.tb00737.x. [DOI] [PubMed] [Google Scholar]
- Romero R, Mazor M, Sepulveda W, Avila C, Copeland D, Williams J. Tumor necrosis factor in preterm and term labor. Am J Obstet Gynecol. 1992;166:1576–1587. doi: 10.1016/0002-9378(92)91636-o. [DOI] [PubMed] [Google Scholar]
- Elliott CL, Loudon JA, Brown N, Slater DM, Bennett PR, Sullivan MH. IL-1beta and IL-8 in human fetal membranes: changes with gestational age, labor, and culture conditions. Am J Reprod Immunol. 2001;46:260–267. doi: 10.1034/j.1600-0897.2001.d01-11.x. [DOI] [PubMed] [Google Scholar]
- Nakashima A, Ito M, Yoneda S, Shiozaki A, Hidaka T, Saito S. Circulating and decidual Th17 cell levels in healthy pregnancy. Am J Reprod Immunol. 2010;63:104–109. doi: 10.1111/j.1600-0897.2009.00771.x. [DOI] [PubMed] [Google Scholar]
- Ito M, Nakashima A, Hidaka T, Okabe M, Bac ND, Ina S, et al. A role for IL-17 in induction of an inflammation at the fetomaternal interface in preterm labour. J Reprod Immunol. 2010;84:75–85. doi: 10.1016/j.jri.2009.09.005. [DOI] [PubMed] [Google Scholar]
- Byrne JA, Stankovic AK, Cooper MD. A novel subpopulation of primed T cells in the human fetus. J Immunol. 1994;152:3098–3106. [PubMed] [Google Scholar]
- Luciano AA, Yu H, Jackson LW, Wolfe LA, Bernstein HB. Preterm labor and chorioamnionitis are associated with neonatal T cell activation. PLoS ONE. 2011;6:0016698. doi: 10.1371/journal.pone.0016698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim MJ, Romero R, Kim CJ, Tarca AL, Chhauy S, LaJeunesse C, et al. Villitis of unknown etiology is associated with a distinct pattern of chemokine up-regulation in the feto-maternal and placental compartments: implications for conjoint maternal allograft rejection and maternal anti-fetal graft-versus-host disease. J Immunol. 2009;182:3919–3927. doi: 10.4049/jimmunol.0803834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tilburgs T, Roelen DL, van der Mast BJ, van Schip JJ, Kleijburg C, de Groot-Swings GM, et al. Differential distribution of CD4+CD25bright and CD8+CD28− T-cells in decidua and maternal blood during human pregnancy. Placenta. 2006;27:25. doi: 10.1016/j.placenta.2005.11.008. [DOI] [PubMed] [Google Scholar]
- Xu Y, Tarquini F, Romero R, Kim CJ, Tarca AL, Bhatti G, et al. Peripheral CD300a+CD8+ T lymphocytes with a distinct cytotoxic molecular signature increase in pregnant women with chronic chorioamnionitis. Am J Reprod Immunol. 2012;67:184–197. doi: 10.1111/j.1600-0897.2011.01088.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steinborn A, Schmitt E, Kisielewicz A, Rechenberg S, Seissler N, Mahnke K, et al. Pregnancy-associated diseases are characterized by the composition of the systemic regulatory T cell (Treg) pool with distinct subsets of Tregs. Clin Exp Immunol. 2012;167:84–98. doi: 10.1111/j.1365-2249.2011.04493.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schober L, Radnai D, Schmitt E, Mahnke K, Sohn C, Steinborn A. Term and preterm labor: decreased suppressive activity and changes in composition of the regulatory T-cell pool. Immunol Cell Biol. 2012;90:935–944. doi: 10.1038/icb.2012.33. [DOI] [PubMed] [Google Scholar]
- Kisielewicz A, Schaier M, Schmitt E, Hug F, Haensch GM, Meuer S, et al. A distinct subset of HLA-DR+-regulatory T cells is involved in the induction of preterm labor during pregnancy and in the induction of organ rejection after transplantation. Clin Immunol. 2010;137:209–220. doi: 10.1016/j.clim.2010.07.008. [DOI] [PubMed] [Google Scholar]
- Gomez-Lopez N, Laresgoiti-Servitje E. T regulatory cells: regulating both term and preterm labor. Immunol Cell Biol. 2012;90:919–920. doi: 10.1038/icb.2012.48. [DOI] [PubMed] [Google Scholar]
- Tilburgs T, Roelen DL, van der Mast BJ, de Groot-Swings GM, Kleijburg C, Scherjon SA, et al. Evidence for a selective migration of fetus-specific CD4+CD25bright regulatory T cells from the peripheral blood to the decidua in human pregnancy. J Immunol. 2008;180:5737–5745. doi: 10.4049/jimmunol.180.8.5737. [DOI] [PubMed] [Google Scholar]
- Samstein RM, Josefowicz SZ, Arvey A, Treuting PM, Rudensky AY. Extrathymic generation of regulatory T cells in placental mammals mitigates maternal-fetal conflict. Cell. 2012;150:29–38. doi: 10.1016/j.cell.2012.05.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shima T, Sasaki Y, Itoh M, Nakashima A, Ishii N, Sugamura K, et al. Regulatory T cells are necessary for implantation and maintenance of early pregnancy but not late pregnancy in allogeneic mice. J Reprod Immunol. 2010;85:121–129. doi: 10.1016/j.jri.2010.02.006. [DOI] [PubMed] [Google Scholar]
- Arenas-Hernandez M, StLouis D, Romero R, Hassan S, Gomez-Lopez N. Endotoxin expands the pool of regulatory T cells in pregnancy but not in the non-pregnant state. Reprod Sci. 2014;21:130A. [Google Scholar]
- Hassan SS, Romero R, Vidyadhari D, Fusey S, Baxter JK, Khandelwal M, et al. Vaginal progesterone reduces the rate of preterm birth in women with a sonographic short cervix: a multicenter, randomized, double-blind, placebo-controlled trial. Ultrasound Obstet Gynecol. 2011;38:18–31. doi: 10.1002/uog.9017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Naik A, Rutledge N, Romero R, Hassan S, Gomez-Lopez N. Vaginal Progesterone, but not 17OHP-C, induces changes in decidual macrophages and regulatory T cells. Reprod Sci. 2014;23:332A. [Google Scholar]
- Bhat NM, Mithal A, Bieber MM, Herzenberg LA, Teng NN. Human CD5+ B lymphocytes (B-1 cells) decrease in peripheral blood during pregnancy. J Reprod Immunol. 1995;28:53–60. doi: 10.1016/0165-0378(94)00907-o. [DOI] [PubMed] [Google Scholar]
- Jensen F, Muzzio D, Soldati R, Fest S, Zenclussen AC. Regulatory B10 cells restore pregnancy tolerance in a mouse model. Biol Reprod. 2013;89:90. doi: 10.1095/biolreprod.113.110791. [DOI] [PubMed] [Google Scholar]
- Rolle L, Memarzadeh Tehran M, Morell-Garcia A, Raeva Y, Schumacher A, Hartig R, et al. Cutting edge: IL-10-producing regulatory B cells in early human pregnancy. Am J Reprod Immunol. 2013;70:448–453. doi: 10.1111/aji.12157. [DOI] [PubMed] [Google Scholar]
- Canellada A, Farber A, Zenclussen AC, Gentile T, Dokmetjian J, Keil A, et al. Interleukin regulation of asymmetric antibody synthesized by isolated placental B cells. Am J Reprod Immunol. 2002;48:275–282. doi: 10.1034/j.1600-0897.2002.01125.x. [DOI] [PubMed] [Google Scholar]
- Kawano T, Cui J, Koezuka Y, Toura I, Kaneko Y, Motoki K, et al. CD1d-restricted and TCR-mediated activation of valpha14 NKT cells by glycosylceramides. Science. 1997;278:1626–1629. doi: 10.1126/science.278.5343.1626. [DOI] [PubMed] [Google Scholar]
- Jenkinson HJ, Wainwright SD, Simpson KL, Perry AC, Fotiadou P, Holmes CH. Expression of CD1D mRNA transcripts in human choriocarcinoma cell lines and placentally derived trophoblast cells. Immunology. 1999;96:649–655. doi: 10.1046/j.1365-2567.1999.00726.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boyson JE, Rybalov B, Koopman LA, Exley M, Balk SP, Racke FK, et al. CD1d and invariant NKT cells at the human maternal–fetal interface. Proc Natl Acad Sci USA. 2002;99:13741–13746. doi: 10.1073/pnas.162491699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kumar V, Delovitch TL. Different subsets of natural killer T cells may vary in their roles in health and disease. Immunology. 2014;15:12247. doi: 10.1111/imm.12247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boyson JE, Nagarkatti N, Nizam L, Exley MA, Strominger JL. Gestation stage-dependent mechanisms of invariant natural killer T cell-mediated pregnancy loss. Proc Natl Acad Sci USA. 2006;103:4580–4585. doi: 10.1073/pnas.0511025103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang S, Li C, Kawamura H, Watanabe H, Abo T. Unique sensitivity to alpha-galactosylceramide of NKT cells in the uterus. Cell Immunol. 2002;215:98–105. doi: 10.1016/s0008-8749(02)00009-6. [DOI] [PubMed] [Google Scholar]
- Cantarella R, StLouis D, Milovic T, Romero R, Gomez-Lopez N.NKT cell activation leads to late preterm birth Reprod Sci 201421230A23793470 [Google Scholar]
- Dang Y, Beckers J, Wang CR, Heyborne KD. Natural killer 1.1+ alpha beta T cells in the periimplantation uterus. Immunology. 2000;101:484–491. doi: 10.1046/j.1365-2567.2000.t01-1-00129.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mrakovcic-Sutic I, Simin M, Radic D, Rukavina D, Radosevic-Stasic B. Syngeneic pregnancy induces overexpression of natural killer T cells in maternal liver. Scand J Immunol. 2003;58:358–366. doi: 10.1046/j.1365-3083.2003.01311.x. [DOI] [PubMed] [Google Scholar]
- Singh N, Hong S, Scherer DC, Serizawa I, Burdin N, Kronenberg M, et al. Cutting edge: activation of NK T cells by CD1d and alpha-galactosylceramide directs conventional T cells to the acquisition of a Th2 phenotype. J Immunol. 1999;163:2373–2377. [PubMed] [Google Scholar]
- Carnaud C, Lee D, Donnars O, Park SH, Beavis A, Koezuka Y, et al. Cutting edge: cross-talk between cells of the innate immune system: NKT cells rapidly activate NK cells. J Immunol. 1999;163:4647–4650. [PubMed] [Google Scholar]
- Miko E, Szereday L, Barakonyi A, Jarkovich A, Varga P, Szekeres-Bartho J. The role of invariant NKT cells in pre-eclampsia. Am J Reprod Immunol. 2008;60:118–126. doi: 10.1111/j.1600-0897.2008.00603.x. [DOI] [PubMed] [Google Scholar]
- Li LP, Fang YC, Dong GF, Lin Y, Saito S. Depletion of invariant NKT cells reduces inflammation-induced preterm delivery in mice. J Immunol. 2012;188:4681–4689. doi: 10.4049/jimmunol.1102628. [DOI] [PubMed] [Google Scholar]
- Banchereau J, Steinman RM. Dendritic cells and the control of immunity. Nature. 1998;392:245–252. doi: 10.1038/32588. [DOI] [PubMed] [Google Scholar]
- Steinman RM, Turley S, Mellman I, Inaba K. The induction of tolerance by dendritic cells that have captured apoptotic cells. J Exp Med. 2000;191:411–416. doi: 10.1084/jem.191.3.411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maldonado-Lopez R, de Smedt T, Michel P, Godfroid J, Pajak B, Heirman C, et al. CD8alpha+ and CD8alpha− subclasses of dendritic cells direct the development of distinct T helper cells in vivo. J Exp Med. 1999;189:587–592. doi: 10.1084/jem.189.3.587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pulendran B, Smith JL, Caspary G, Brasel K, Pettit D, Maraskovsky E, et al. Distinct dendritic cell subsets differentially regulate the class of immune response in vivo. Proc Natl Acad Sci USA. 1999;96:1036–1041. doi: 10.1073/pnas.96.3.1036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schlitzer A, Ginhoux F. Organization of the mouse and human DC network. Curr Opin Immunol. 2014;26:90–99. doi: 10.1016/j.coi.2013.11.002. [DOI] [PubMed] [Google Scholar]
- Plaks V, Birnberg T, Berkutzki T, Sela S, BenYashar A, Kalchenko V, et al. Uterine DCs are crucial for decidua formation during embryo implantation in mice. J Clin Invest. 2008;118:3954–3965. doi: 10.1172/JCI36682. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bizargity P, Bonney EA. Dendritic cells: a family portrait at mid-gestation. Immunology. 2009;126:565–578. doi: 10.1111/j.1365-2567.2008.02918.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blois SM, Alba Soto CD, Tometten M, Klapp BF, Margni RA, Arck PC. Lineage, maturity, and phenotype of uterine murine dendritic cells throughout gestation indicate a protective role in maintaining pregnancy. Biol Reprod. 2004;70:1018–1023. doi: 10.1095/biolreprod.103.022640. [DOI] [PubMed] [Google Scholar]
- Ruiz RJ, Jallo N, Murphey C, Marti CN, Godbold E, Pickler RH. Second trimester maternal plasma levels of cytokines IL-1Ra, Il-6 and IL-10 and preterm birth. J Perinatol. 2012;32:483–490. doi: 10.1038/jp.2011.193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bizargity P, Del Rio R, Phillippe M, Teuscher C, Bonney EA. Resistance to lipopolysaccharide-induced preterm delivery mediated by regulatory T cell function in mice. Biol Reprod. 2009;80:874–881. doi: 10.1095/biolreprod.108.074294. [DOI] [PMC free article] [PubMed] [Google Scholar]


