Abstract
The initiation of mammalian puberty is underpinned by an increase in Kisspeptin (Kiss1) signaling via its receptor (Kiss1r/GPR54) on gonadotropin-releasing hormone (GnRH) neurons. Animals and humans with loss-of-function mutations in Kiss1 or Kiss1r fail to go through puberty. The timing of puberty is dependent on environmental factors, and malleability in puberty timing suggests a mechanism that can translate environmental signals into patterns of Kiss1/Kiss1r gene expression. Epigenetics is a powerful mechanism that can control gene expression in an environment-dependent manner. We investigated whether epigenetic DNA methylation is associated with gene expression changes at puberty. We used bisulfite-PCR-pyrosequencing to define the methylation in the promoters of Kiss1 and Kiss1r before and after puberty in female rats. Both Kiss1 and Kiss1r showed highly significant puberty-specific differential promoter methylation patterns. By identifying key differentially methylated residues associated with puberty, these findings will be important for further studies investigating the control of gene expression across the pubertal transition.
Keywords: puberty, methylation, kisspeptin, Kiss1r, neuroendocrine, reproduction
Introduction
Puberty is the transitional period between childhood and adulthood in which reproductive capacity is achieved. Mammalian puberty onset is critically dependent on the activation of gonadotropin-releasing hormone (GnRH) neurons located within the rostral hypothalamus.1 GnRH peptide stimulates the secretion of the gonadotrophins, luteinizing hormone (LH), and follicle-stimulating hormone (FSH), from the anterior pituitary gland; these, in turn, stimulate gonadal maturation, including steroid production.1 Although the fertility-regulating circuitry develops during gestation and early neonatal life,2–5 puberty occurs somewhat later, and mechanisms to explain this delayed activation of the GnRH neuronal network have been lacking.
Afferent inputs to the GnRH neuronal network that modulate puberty appear to change at or before puberty. There is an increase in stimulatory drive, in the form of glutamate and Kisspeptin1 (Kiss1) signaling,6–9 and a decrease in inhibitory drive, in the form of opiate and GABA signaling.10,11 Among the stimulators, Kiss1 is the most potent activator of GnRH firing and secretion described to date and has been shown to be absolutely required (along with its receptor Kiss1r) for puberty onset.12–16 Interestingly, Kiss1 also appears to be involved in the very early development of GnRH neurons, where it controls their innervation of the median eminence.5 Not surprisingly, loss-of-function mutations in Kiss1 or Kiss1r result in failed puberty and infertility in both humans and animal models.12,17,18
Kiss1-expressing neurons that modulate puberty and reproduction reside in two main areas within the hypothalamus: the RP3V and the arcuate nucleus (ARC). Kiss1 neurons undergo a curious maturation, as many are generated prenatally, but additional cells appear around the time of puberty in the RP3V.19 Those in the RP3V do not initiate Kiss1 expression until the late prepubertal period in-step with the rise in estrogen levels in the lead-up to the onset of GnRH neuron activation.20,21 Indeed, estrogen is a strong stimulator of Kiss1 expression in the RP3V both at puberty and as a positive feedback stimulator driving the LH surge during normal female reproduction.22–24 The mechanism by which the initiation and upregulation of Kiss1 gene expression in the RP3V is time-locked to the peripubertal period is unknown.
Recently, it was reported that global pharmacologic inhibition of DNA methylation prevented Kiss1 upregulation in the mediobasal hypothalamus (MBH, the brain region containing the ARC Kiss1 neurons) and delayed puberty.25 The underlying mechanism appears to be relief of a repressive chromatin state, mediated by Polycomb group silencers, which initiates upregulation of Kiss1 expression in the MBH and onset of puberty.25 However, Kiss1 expression in neurons of the RP3V have not been investigated. Because of the controversy surrounding the relative roles of the RP3V and ARC Kiss1 neurons in puberty, we investigated RP3V. We examined whether methylation of the Kiss1 and Kiss1r promoters is altered along with puberty progression in the RP3V and POA, respectively, in female rats.
Kiss1 exerts its effects on GnRH neurons directly through its receptor, Kiss1r, which in the rostral hypothalamus is expressed primarily by GnRH neurons. GnRH neuron cell bodies are located in a rostro-caudal continuum from the medial septum to the anterior hypothalamic area.1,26 Although not all reports are consistent, it appears that Kiss1r does not show a dramatic increase in expression in association with puberty.26,27 Thus, Kiss1r represents a target that, although critical for puberty, is unlikely to undergo changes in methylation that alter its expression. However, it is possible that methylation changes at the time of puberty may act to ensure that Kiss1r expression is retained in the face of other major changes to the HPG axis.
Given the shared importance of Kiss1r and Kiss1 expression for puberty, and the potential for these proteins to be differentially methylated in a manner that may reflect their distinct patterns of gene expression across puberty, the two were investigated. The goal of this study was to understand the changes in DNA methylation across the pubertal transition, particularly with respect to Kiss1 and Kiss1r. For Kiss1, which is upregulated at and after puberty, we hypothesized that methylation changes would support this increased expression by either increasing binding by transcriptional activators or decreasing binding of transcriptional repressors. For Kiss1r, which is expressed both before and after puberty, we predicted that methylation changes would act to ensure that expression levels did not vary across puberty either by showing no changes in factor binding or by showing changes whose overall effects on transcription would not be different before versus after puberty. We examined the methylation patterns for ~2 kb proximal promoter regions for Kiss1 and Kiss1r genes before and after puberty in female rats. We defined methylation status and patterns for both genes before and after puberty and observed differential methylation at specific nucleotides in the promoters of these two genes before and after puberty and attempted to correlate these to both gene expression at the same ages and brain regions and potential transcription factor binding sites. The findings highlight specific loci within the promoters that may be differentially modified in physiological situations in which puberty timing is altered as well as provide a resource for further investigation of the epigenetic control of reproduction.
Results
Mapping of CpG dinucleotides in the promoters of Kiss1 and Kiss1r
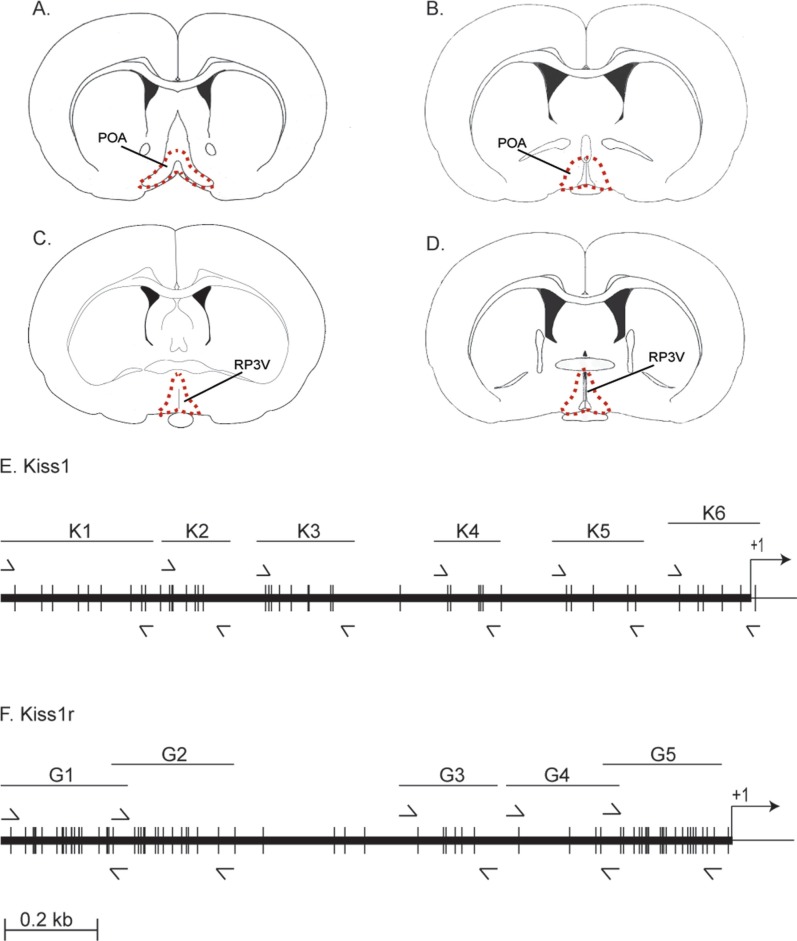
Differential methylation of CpG dinucleotides in the promoter regions of mammalian genes have been associated with environmental factors and gene expression changes.28–31 This study was conducted to examine whether Kiss1 and Kiss1r promoter methylation changes across puberty in female rats and whether this is associated with gene expression changes. We initially mapped the distribution of CpG dinucleotides within the first approximately 2 kb 5′ (upstream) of the transcriptional start site (Figs. 1E, F). Each promoter showed a unique number and pattern of CpG dinucleotides. Kiss1 has 41 CpG/2.8 kb, and does not appear to have any well-defined CpG islands, although the density of CpGs does vary across the region (Fig. 1E). In contrast, Kiss1r has 71 CpG/2.2 kb, and two well-defined CpG islands—regions with >50% GC content, and a frequency of CpG dinucleotides greater than 60%,31 one right at the transcription start site and another nearly 2 kb upstream (Fig. 1F).
Figure 1.
Details of hypothalamic microdissection and distribution of promoter CpG dinucleotides. A–D. show schematic diagrams of representative brain levels, modified from Allen Mouse Brain Atlas, Allen Institute for Brain Science (http://mouse.brain-map.org/experiment/thumbnails/100048576?image_type=atlas), in accordance with their usage policy (http://www.alleninstitute.org/Media/policies/citation_policy_content.html) from which tissue was taken. Dotted line demarcates region dissected and processed for DNA extraction and bisulfite sequencing. Rostral (A) and caudal (B) POA, containing GnRH neurons were pooled for Kiss1r analysis. Similarly, rostral (C) and caudal (D) regions of the RP3V were pooled for Kiss1 analysis. POA, preoptic area; RP3V, rostral periventricular region of the third ventricle. E. Schematic drawing of the Kiss1 promoter showing positions of CpG dinucleotides (small vertical lines) and amplification primers (⇀ Forward, ↽ Reverse). +1 is the transcription start site and → indicates the direction of transcription. Note that in the ~2.8 kb 5′ to +1 that only 1 CpG is not covered by the primers. F. Schematic drawing of the Kiss1r promoter showing positions of CpG dinucleotides and amplification primers. Decorations are as in E. Note that in the ~2 kb 5′ to +1 that only 5 CpGs are not covered by the primers.
Overall level of CpG promoter methylation differs between Kiss1 and Kiss1r and is not influenced by physiological state
Within the Kiss1 and Kiss1r proximal promoters, the levels of overall methylation are strikingly different. The Kiss1 promoter is mostly methylated irrespective of physiological state. Of the six regions surveyed in the Kiss1 promoter, two had insufficient coverage for analysis and the remaining four showed that over 75% of all CpGs were methylated both before and after puberty (Fig. 2A). In contrast, the Kiss1r promoter was mostly unmethylated. Of the five regions surveyed in the Kiss1r promoter, one had insufficient coverage for analysis and of the remaining 4 most showed <20% CpG residues methylated (Fig. 2B). One exception was region 4, which showed nearly 60% methylated CpGs. This overall percentage of methylation in Kiss1r, like Kiss1, was irrespective of puberty.
Figure 2.
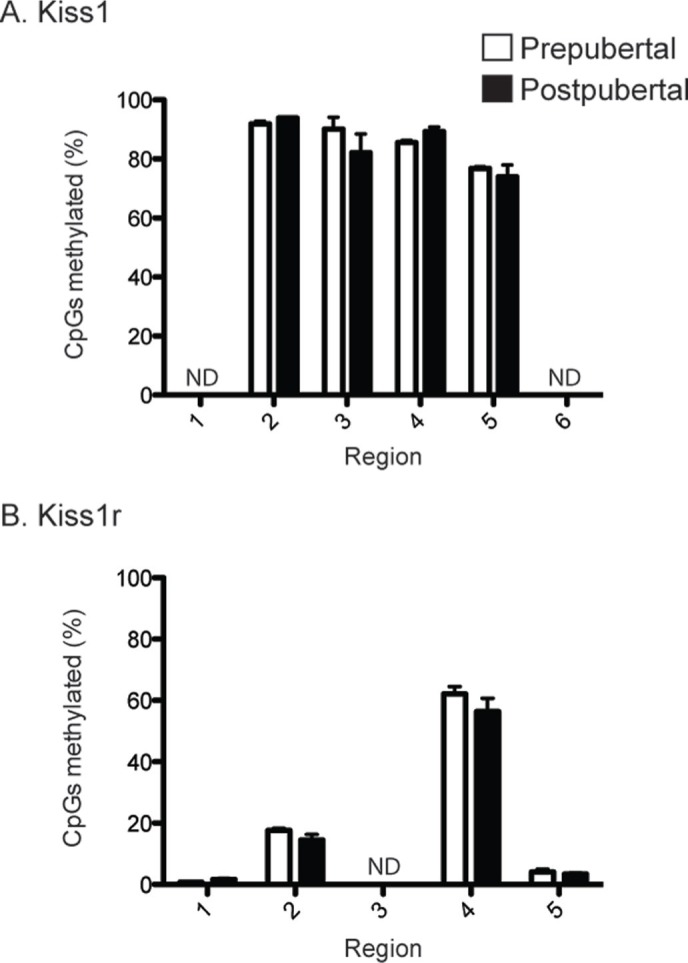
Details of methylation across entire promoter regions. A. Bar graph showing the percentage of CpG residues within each of the 6 regions across the Kiss1 promoter. Note that the promoter as a whole is mostly methylated irrespective of physiological state. B. Bar graphs showing the percentage of CpG residues within each of the 5 regions spanning the Kiss1r promoter. Note that the promoter as a whole is mostly unmethylated irrespective of physiological state. ND, no data, indicating that insufficient sequencing coverage was obtained for these three regions. This mainly because of the high poly-T content that arises as a consequence of bisulfite conversion. Error bars indicate standard error of the mean.
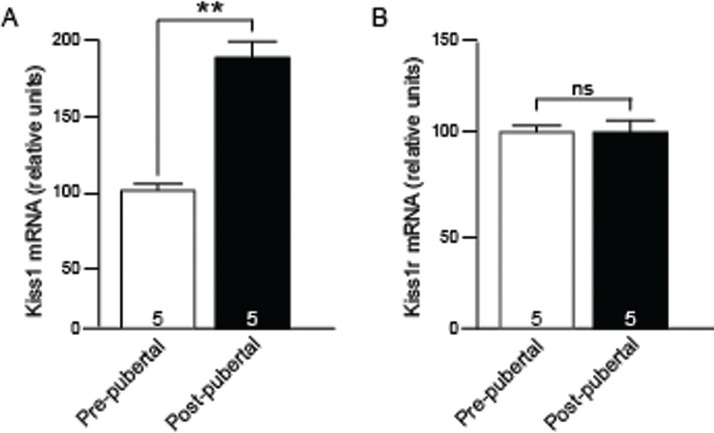
In order to determine the relationship between promoter methylation and gene transcription, quantitative real-time PCR (qPCR) was used. Importantly, much of mRNA assessed by qPCR was derived from the same tissue as was used for methylation analysis (3/5 samples pre-pubertal and 2/5 post-pubertal for Kiss1r, and 1/5 samples pre-pubertal, and 2/5 post-pubertal for Kiss1). The remainder of samples were derived from different animals, but dissections used the same landmarks as for methylation analysis. This qPCR analysis showed no significant difference in Kiss1r expression between pre- and post-pubertal ages, which is consistent with previous observations (Fig. 3A, [26]). In contrast, Kiss1 showed a significant increase in expression between pre- and post-puberty, again consistent with previous observations (Fig. 3B,).32,33 When expression was then compared to overall percent methylation in Kiss1r (~23% methylated) and Kiss1 (~80%), which remained static across puberty, there was no obvious association with gene expression level or change. Together these results suggest that gross, or overall, levels of methylation across the 2 kb proximal promoter regions of Kiss1 and Kiss1r do not correlate well to levels of expression for these two active genes at least within the RP3V and POA, respectively. The overall percentage of methylated residues across a region can be a useful predictor of transcriptional activity. However, methylation differences at individual nucleotide residues that may be bound by specific transcription factors, are also critical regulators of gene activity.
Figure 3.
Kiss1 and Kiss1r mRNA expression before and after puberty. Bar graphs showing the fold-change, relative to pre-puberty levels, in gene expression for Kiss1 (A) and Kiss1r (B) before (white bars) and after (black bars) puberty. Numbers at base of bars indicate number of brains assayed. *, P < 0.05, Students t-test.
Kiss1 and Kiss1r promoters shows differential methylation across puberty
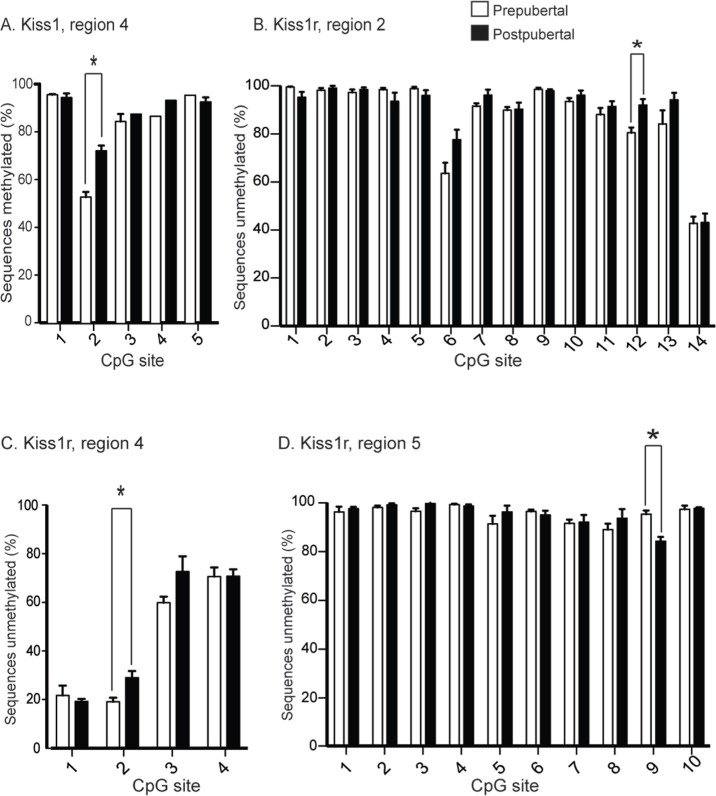
Despite a relatively static overall percent methylation over large regions, there were specific residues within each of the promoters that showed highly significant differences across puberty. The Kiss1 promoter contained one residue (region 4, CpG 2) with significantly elevated methylation after puberty (Fig. 4A). None of the residues in any of the remaining CpG-containing regions showed significantly different methylation before versus after puberty (not shown). In contrast, the Kiss1r promoter possessed several residues with differential methylation across puberty (Figs. 4B–D). Kiss1r region 2 (Fig. 4B) is somewhat distant from the transcriptional start site, but may act as a distant enhancer. In contrast, regions 4 and 5 (Figs. 4C, D) are very close to the start of transcription, and may interact with proximal promoter-binding factors to control transcription. Thus, Kiss1 and Kiss1r promoters showed differential methylation at specific residues that could affect transcriptional activity before versus after puberty.
Figure 4.
Differential methylation in the Kiss1 and Kiss1r promoters across puberty. (A) Bar graph showing the percentage of CpGs that are methylated (y-axis) within region 4 of the Kiss1 promoter before (white) and after (black) puberty. *P < 0.05, Student’s t-test. (B–D) Bar graphs showing the percentage of CpGs that are unmethylated (y-axis) within regions 2 (B), 4 (C), and 5 (D) of the Kiss1r promoter before (white) and after (black) puberty. Note that regions 2 and 4 showed a decrease in methylation after (black bars) puberty compared with before (white bars; *, P < 0.05, Student’s t-test), whereas region 5 showed and increase in methylation after puberty (*, P < 0.05, Student’s t-test). Error bars indicate standard error of the mean. Due to the large number of reads sequenced per PCR reaction per animal, power analysis indicates >90% power in this experiment.
In silico analysis of transcription factor binding sites and differential methylation
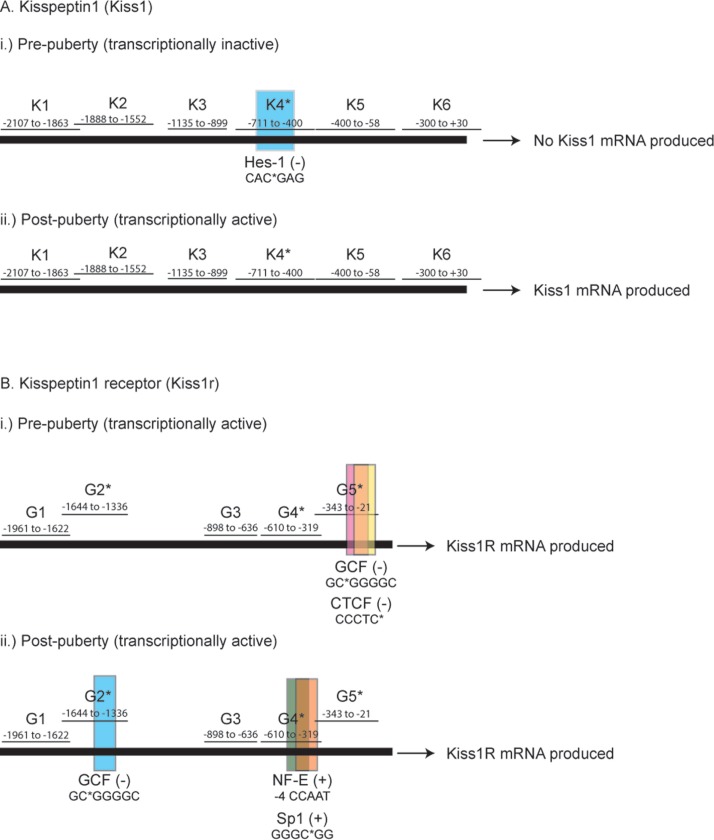
One approach to functionally dissect promoter regions with differential methylation is conduct in silico mapping of the positions at which transcriptional modulators may bind. Thus, in order to determine the functional significance of differential methylation patterns, in silico analysis of transcription factor binding sites was undertaken using the online Transcription Element Search System (TESS) resource (http://www.cbil.upenn.edu/cgi-bin/tess/tess).34 Although the Kiss1 promoter possessed only a single differentially methylated residue, this region appears likely to be bound by the transcriptional repressor Hes-1 (Fig. 5A).35 This suggests that at puberty, the Kiss1 promoter undergoes a switch from unmethylated Hes-1-binding, and thus transcriptionally inactive, to a methylated form that is not bound by Hes-1, and therefore is de-repressed and transcriptionally active. A similar conclusion was reached in a recent paper examining RP3V Kiss1 promoter methylation in sexual dimorphism in mice.36
Figure 5.
Predicted binding sites for transcription factors relative to sites of differential methylation for Kiss1 (A) and Kiss1r (B) promoters. Nucleotide numbers relative to the start of transcription are shown for each region examined. Each transcription factor predicted from the TESS analysis to bind to sites of differential methylation is shown. Sequences beneath transcription factor names denote core nucleotide binding sequence. Methylated residues within the core motif are shown with *. For NF-E, −4 indicates that the methylated C lies 4 nucleotides upstream of the 5′-most core sequence nucleotide. Factors are shown bound (present) when the promoter region is unmethylated and absent when the promoter regions is methylated. For each gene a the pre-puberty state (i) and the post-puberty state (ii) is shown. Dark horizontal bar represents promoter region sequenced, 2.8 kb for Kiss1 and 2.2 kb for Kiss1r.
The Kiss1r promoter showed a number of differentially methylated sites that could be bound by transcription factors (Fig. 5B). Within region 2 and overlapping with CpG 12, there is a predicted binding site for GC-binding factor (GCF), a transcriptional repressor.37 Within region 4 and overlapping with CpG 2, which loses methylation across puberty, there appear to be overlapping binding sites for NF-E and Sp1. Finally, region 5 contains CpG 9, which becomes increasingly methylated across puberty. This region is predicted to be bound by GCF and CCCTC binding factor (CTCF). CTCF, like GCF, is a well-described transcriptional repressor, and its binding has been shown to be regulated by differential methylation in cancer cells,38 where hypermethylation leads to loss of CTCF binding and activation of transcription. Thus, both Kiss1 and Kiss1r promoters show intriguing patterns of methylation changes across puberty, which may modulate the binding of specific transcription factors.
Discussion
This is the first comprehensive examination of methylation in the Kiss1 and Kiss1r promoters from cells in the RP3V and POA, respectively, during puberty. Although recent studies have examined methylation in these promoters,25,36,39 only very small regions of the promoters were examined. The data presented here show puberty-specific DNA methylation patterns spread across >2 kb in the promoters of both genes examined. In the case of Kiss1r, methylation differences did not appear to be associated with gross changes in gene expression, although they may indicate different modes of transcriptional control before and after puberty. In contrast, the Kiss1 promoter exhibited differential DNA methylation in association with elevated expression coincident with puberty, but it is unclear whether the changes in methylation were causal. In support of a causal role, a recent report that examined the Kiss1 promoter in the mouse RP3V also found increased methylation, suggesting a blockage of repressor binding to activate gene expression.36 In mice and humans, specific regions within Kiss1 or Kiss1r promoters that are important for transcriptional activity have been identified.40,41 Differential methylation or other epigenetic signatures in transcriptionally important promoter regions support that differential methylation is important for transcriptional control. Studies in which either the Kiss1 or Kiss1r promoters were functionally dissected in rats, unfortunately, have not been performed. Moreover, the sequences of the promoter regions of rat Kiss1 and Kiss1r differ considerably from those in mice, so a nucleotide-by-nucleotide methylation comparison is not possible. Thus, a functional comparison between methylation and transcriptional control awaits further study for these genes in the rat.
One feature of these data is that the changes are small. However, if a methylation change occurs even at a single nucleotide that is part of a transcription factor binding site, it could significantly influence whether the transcription factor binds. This is consistent with methylation changes in other genes, which may be small/subtle, but are nonetheless able to alter gene expression (see for example reference 30).
Interestingly, in terms of the overall amount of methylation at the >2 kb proximal promoter, irrespective of physiological state, the Kiss1 promoter was mostly methylated compared to the Kiss1r promoter, which was mostly unmethylated. Overall levels of DNA methylation within promoters have been grossly correlated to gene activity.31,42 Because methylated regions are more likely to form heterochromatin and thus less likely to be accessible to transcriptional machinery, they tend to be thought of as less likely to be expressed. Currently, however, a wide variety of methylation patterns have been observed within active promoters,43,44 consistent with the gene-specific patterns we observed in this study; thus, the dogma of more methylation equals less expression is being revised. It is also possible that differences in methylation across the promoters of different genes may represent differences in the molecular manner in which gene expression is modulated; that is, a gene whose transcriptional regulatory regions are mostly methylated may require a fundamentally different mechanism for transcriptional activation by comparison with a gene whose promoter is mostly unmethylated. In the current situation, for example, Kiss1 expression is under tight control to ensure that levels vary appropriately across the estrous cycle. In contrast, Kiss1r appears to be rather evenly expressed both before and after puberty, a relaxed level of control that may be mirrored in the mostly unmethylated promoter.
One technical caveat to the interpretation of these data is that they are derived from a mixed cell population, so it is possible that the level and pattern of cell-type specific methylation is underrepresented. Although global gene-specific patterns of methylation that correspond well to gene expression at puberty have been observed,27 the ability to isolate genomic DNA from single GnRH and Kiss1 neurons both before and after puberty would be a strategy for further validation.
In silico analysis revealed a number of interesting possibilities regarding the binding of transcriptional modulators. For Kiss1, only a single factor, the transcriptional repressor Hes-1, was identified to potentially bind at the differentially methylated region. This is suggests a model in which puberty-specific methylation reduces Hes-1 binding in order to de-repress (ie, activate) the Kiss1 promoter at the time of puberty, which is consistent with recent findings in mice.36 Interestingly, E2 has been shown to repress Hes-1 expression in breast cancer cells.45 Should similar regulation occur in the RP3V Kiss1 neurons, rising E2 levels at puberty could cause Hes-1 to leave the Kiss1 promoter, increasing levels of Kiss1 to increase GnRH release. However, the functional relationship among E2 levels, Kiss1 promoter methylation, and transcription is unclear. Given the multiple functions of E2 and its assorted signaling pathways, it may both drive epigenetic changes and respond to them. Determining the exact role of E2 in the differential methylation patterns of Kiss1 and Kiss1r associated with puberty, be it causal or casual, will required further study.
For Kiss1r, a number of differentially methylated sites were identified to which several transcriptional regulators putatively bind. NF-E was shown to interact with cyclic AMP response element-binding protein (CREB) binding protein (CREB-BP), a transcriptional activator involved in estrogen-responsiveness.46 Previously, it was shown that E2- dependent methylation can modulate where CREB-BP binds the genome.46 Another identified factor was Sp1, which has been previously reported to be a positive regulator of Kiss1r expression in mouse pituitary cells.41 Finally, putative binding of the GCF and CTCF negative regulators was identified in a region of the Kiss1r promoter that undergoes increased methylation across puberty. If this methylation blocks binding, GCF and CTCF may leave the promoter at the time of or after puberty. However, concomitant with this putative loss of binding, another site also bound by GCF loses methylation, and thus likely acquires GCF binding after puberty. The net result would be that negative regulation lost in one region of the promoter would be taken over by negative regulation at another region of the promoter, and thus no change in expression would occur. It is important to note that most of the Kiss1r promoter is unmethylated and it is actively expressed both before and after puberty. Thus, it is likely to be bound by a large number of transcription factors, making the role of any one factor difficult to determine from this analysis alone. Understanding whether these regions were differentially bound in vivo by the putative factors before versus after puberty would add compelling evidence for the significance of the differentially methylated sites in gene regulation at the time of puberty.
Importantly, this study did not address whether the methylation changes drive puberty directly or are related to activation of some other regulatory pathways (eg, via sex steroids), but rather cataloged how/whether methylation is different before and after puberty. One remaining key question that this study did not examine is whether gene expression changes that take place during the transition period are associated with specific methylation patterns that are different from those either before or after puberty. The regulation of all methylation changes—before, during, and after puberty —is likely to be quite complex and attributable to multiple pathways that act in concert to bring about the pubertal transition.
In conclusion, this study highlights key regions within both Kiss1 and Kiss1r promoters that are differentially methylated within cells in the RP3V and POA, respectively, before and after puberty. The data presented here serve as a starting point for further investigation of the role of epigenetics in the transcriptional modulation of these genes by sex steroids and other environmental factors. It also offers the opportunity to examine the role of epigenetics in the differential regulation of Kiss1 transcription between the RP3V and ARC at puberty and across the female reproductive cycle.
Materials and Methods
Animals
All experimental procedures were conducted with the approval of the University of Otago animal ethics committee. All experiments used female Sprague-Dawley rat brain tissue. Tissue was collected at two ages that spanned puberty: prepubertal (postnatal day 29 (PN29); n = 3) and postpubertal (PN80; n = 3), on diestrus. Animals in this study received common food and were reared and lived in identical environmental conditions (same room). In our colonies, female rats went through puberty, as judged by vaginal opening and first estrus, at an average age of 49 days.
Preparation of brain tissue and DNA and RNA extraction
Animals were killed by decapitation, and whole brains were removed, snap frozen on dry ice, and then stored at −80°C until use. Brains were cut into 300-μm sections using a cryostat and two hypothalamic regions were microdissected (the preoptic area (POA) and the rostral periventricular region of the third ventricle (RP3V)) for DNA and RNA extraction. The POA, which contains numerous GnRH neurons,1 was located and then dissected from two consecutive sections using the anatomical landmarks set out in a stereotaxic coordinate atlas (Figs. 1A, B). The RP3V, encompassing both the anteroventral periventricular (AVPV) nucleus and the periventricular nucleus (PeN), and which contains numerous kisspeptin neurons,20 was similarly dissected from two consecutive sections (Figs. 1C, D). Tissue was microdissected and then digested in 250 μL of lysis buffer following manufacturer’s protocol (PureLink® genomic DNA kit, Invitrogen, Carlsbad, CA, USA). Concentrations of extracted DNA were measured using a nanospectrophotometer (NanoDrop® DN-1000, NanoDrop Products, Wilmington, DE, USA) and DNA was then stored at −20°C. Matched samples were also collected using the same dissection criteria (Figs. 1A–D) for preparation of mRNA. RNA was extracted using the Trizol reagent (Invitrogen, Australia) according to the manufacturer’s protocol. RNA yield was determined on a Nanodrop spectrophotometer. Extracted RNA was then treated with DNase I (DNA-free ™ kit, Ambion, Austin, TX, USA) according to the manufacturer’s protocol to remove residual genomic DNA and reduce the potential for false-positive results in subsequent qPCR.
Quantitative reverse transcription-PCR
All qPCR was carried out with the LightCycler 480 Real Time PCR System (Roche, Basel, Switzerland), using white 96-well plates (Roche). SYBR Green chemistry was used for Kiss1r and Taqman chemistry was used for Kiss1 PCRs. Kiss1r primers were designed using NCBI’s primerBLAST function (http://www.ncbi.nlm.nih.gov/tools/primerblast), and Kiss1 primers and Taqman probes were designed using Primer Express software (PE, Applied Biosystems, Foster City, CA, USA). Sequences of all primers and probes are shown in Table 1. For SYBR Green each individual reaction was run in triplicate for each RNA preparation and contained: 10 straliey app = “EN”mastermix (Applied Biosystems), 2 μm, forward and reverse mixed primer, 6 μL H2O (Molecular grade, Invitrogen, Australia), and 2 μM cDNA (final cDNA concentration = 2 ng/mL). These reactions were run on the LightCycler using the following protocol: 95°C for 10 minutes (pre- incubation); 45 cycles of 95°C for 10 seconds, 60°C for 1 minute and 72°C for 30 seconds (amplification); 95°C for 1 second, 50°C for 1 minute (melting curve); 40°C for 30 seconds (cooling). Taqman reactions (20 μL) were prepared by adding 2 μL of template cDNA, in duplicate, together with LightCycler 480 Probes Master Mix (Roche) to 96-well LightCycler 480 Multiwell plates with adhesive covers (Roche). A reaction mix was prepared containing primers and probes at optimized concentrations of 500 nM and 200 nM, respectively, except the Kiss1 forward primer was used at a concentration of 300 nM. Samples were heated to 50°C for 2 minutes, then to 90°C for 10 minutes before 45 cycles of 95°C for 15 seconds and 60°C for 1 minute, as per the manufacturers instructions. All qPCR runs also included two types of negative controls: (i) no reverse transcriptase; (ii) no cDNA. All qPCR data were analyzed using the 2-ΔΔCt relative quantification method47 to calculate fold-changes in gene expression relative to the internal standards (β2-microglobulin and β-actin) in the pre-pubertal and post-pubertal groups. Student’s t-tests were performed to determine significance in gene expression change between the two groups.
Table 1.
qPCR primer sequences (5′–3′).
| GENE | FORWARD | REVERSE | PROBE |
|---|---|---|---|
| Kiss1 | GCCGCTGGCAAAAGTGAA | GATTCCTTTTCCCAGGCATTAA | ATCCACAGGCCAGCAGTCCGGA |
| Kiss1r | GCTGGGAGACTTCATGTGCAA | AGCGGGAACACAGTTCACATACC | NA |
| beta-actin (Taqman) | AGGCCAACCGTGAAAAGATG | GCCTGGATGGCTACGTACATG | CCCAGATCATGTTTGAGACCTTCAAC |
| beta2-microglobulin | TTCTGGTGCTTGTCTCACTGA | CAGTATGTTCGGCTTCCCATTC | NA |
| beta-actin | AGATGACCCAGATCATGTTTGAGA | ACCAGAGGCATACAGGGACAA | NA |
Bisulfite conversion and PCR
To preserve the methylation status of the DNA, all extracted samples underwent bisulfite conversion as per manufacturer’s instructions (Invitrogen™ MethylCode™ Bisulfite Conversion Kit). Bisulfite converted DNA was then stored at −20°C until use. Methylation-specific primer sets were designed using MethPrimer (http://www.urogene.org/methprimer/index1.html) to amplify portions of the upstream promoter regions of the Kiss1 (2.7 kb, 6 primer sets) and Kiss1r genes (1.9 kb, 5 primer sets) that included the greatest number of CpG dinucleotides (for primer sequences see Table 2). Kiss-1 primer sets were then used with RP3V samples and Kiss1r sets with POA samples, and subjected to PCR (PCR conditions for each primer set are detailed in Table 1) using Platinum Taq polymerase (Invitrogen). The PCR products were verified on a 1% agarose gel stained with ethidium bromide, and purified using a Qiagen Min Elute PCR Purification Kit following manufacturer’s protocol (Qiagen, Hilden, Germany).
Table 2.
Methylation analysis primers (5′–3′) and cycling parameters.
| PRIMER NAME | SEQUENCE | ANNEALING TEMPERATURE (°C) | [MgCl2] (mM) | PRIMER CONCENTRATION (μM) |
|---|---|---|---|---|
| Kiss1 region1, For | GAATTATTTGAAGGGAGTTTGGTAAG | 58 | 1.5 | 0.5 |
| Kiss1 region 1, Rev | CCTCTAACTCCCCTAAATACTCTAC | |||
| Kiss1 region 2, For | GTTATGTGGGTTTAAGGGGTTAAAT | 62 | 4 | 0.5 |
| Kiss1 region 2, Rev | AATAATCCAAACTTCTACCTCTCATC | |||
| Kiss1 region 3, For | TTTTATTATTTTTTTTGTTTAGAGA | 54 | 3.5 | 0.8 |
| Kiss1 region 3, Rev | ACAACTTATATAAATAAATCATTCC | |||
| Kiss1 region 4, For | TAGTTGGGGGTTAGGTTAATTAATG | 56 | 3 | 0.5 |
| Kiss1 region 4, Rev | ACCCAAAACTTCTATTCAAAAACAA | |||
| Kiss1, region 5, For | AGTTAAATTGATGAGGTTAATTTAGTT | 60 | 4 | 0.5 |
| Kiss1 region 5, Rev | TCAACTATTCTCTTAAAAACCTAAAAC | |||
| Kiss1, region 6, For | TTTGTTTTGTAAATTTTTTATTTAGAG | 58 | 3.5 | 0.5 |
| Kiss1 region 6, Rev | CAACAACTACCAAAAAACCAAC | |||
| Kiss1r region 1, For | TTTAGTTTAGGGAGGAGATTTTAGTT | 58 | 0.75 | 0.5 |
| Kiss1r region 1, Rev | AATTTTATCCTCAACCCTTCTAATC | |||
| Kiss1r region 2, For | TTAGAAGGGTTGAGGATAAAATTTG | 60 | 3 | 0.5 |
| Kiss1r region 2, Rev | CTCCAATAATCCTCCTACCTCAAC | |||
| Kiss1r region 3, For | TTTAGAGTTTTTAGATTTTTAATTTTT | 58 | 3 | 0.5 |
| Kiss1r region 3, Rev | CCTTATCAAACTACCAAAATACTATAC | |||
| Kiss1r region 4, For | TTTTGAGATATATTTATTAGGGATGTTAGG | 60 | 3 | 0.5 |
| Kiss1r region 4, Rev | AAAAAATCATAACCCTTTCAAAAAC | |||
| Kiss1r region 5, For | TTGAAAGGGTTATGATTTTTTTT | 56 | 1.5 | 0.2 |
| Kiss1r region 5, Rev | CCAAACAACCTTCACAAATTAC |
Barcoding and 454 sequencing
For each animal, all eleven purified PCR products, covering the Kiss1 and Kiss1r promoters, were pooled according to Meyer et al.48 The six pools were then barcoded using blunt-ended ligation.48 For designing the barcodes, we used 8-bp long tags with a minimum substitution distance of 3, selected from the supplementary material in Meyer et al.48 A pool of barcoded samples was then used to construct a sequencing library using the RLMID12 adaptors and sequenced on the 454 Life Sciences (Roche Diagnostics) GS Junior following the manufacturer’s instructions.
Sequencing data analysis
454 sequencing produced 36,384 sequencing reads in a single standard flowgram format (SFF). Obtained reads were initially examined using the Genome Sequencer FLX System Software version 2.3 and its SFF Tools commands (Roche Diagnostics). Only reads that contained complete individual-specific barcodes (with no mismatch) were extracted and separated into six individual SFF files for further analysis. The number of reads per individual ranged from 3826 to 6398. Individual SFF files were then imported into CLC Genomics Workbench 5.1 software (CLC Bio, Aarhus, Denmark), where reads were mapped to each of the eleven reference sequences. These reference sequences were modified using the assumption that all cytosine residues had been converted to thymines during bisulfite conversion. The CLC mapping parameters were set to the following: mismatch, insertion and deletion cost = 2, length fraction = 0.5, similarity fraction = 0.8. The mappings were then used for single-nucleotide polymorphism (SNP) detection. The criteria for SNP detection were set to minimum central and neighboring Phred quality score = 20, minimum coverage = 1, and minimum variant frequency = 0%. The list of detected SNPs was then narrowed to focus on previously determined CpG dinucleotides, and the percentage of C and T for each region was recorded. The reverse comparison was then completed, which mapped reads to references with the original cytosines still in place in order to identify CpG dinucleotides where 100% of the sequences had been converted to thymines, and thus would not be picked up in the initial mapping. The statistical differences in C/T percentages within each region of Kiss1 and Kiss1r promoters between prepubertal and postpubertal groups were determined using unpaired, two-tailed t-test analysis (Prism, Graphpad Software, San Diego, CA, USA).
In silico transcription factor binding site analysis
Transcription Element Search System (TESS) (http://www.cbil.upenn.edu/cgibin/tess/tess)34 software was used to determine putative transcription factors that bind the promoter regions of Kiss1 and Kiss1r genes. Genomic sequences corresponding to each amplicon plus an additional flanking ten base pairs were input into TESS for analysis using default parameter settings.
Acknowledgments
The authors would like to thank Dr Sharon Ladyman for expert animal handling and tissue collection.
Footnotes
Author Contributions
Conceived and designed the experiments: CLJ, AKW, JLV, NJG. Analyzed the data: CLJ, AKW, JLV, MZ, JLS. Wrote the first draft of the manuscript: CLJ, AKW. Contributed to the writing of the manuscript: AKW, MZ, JLV, JLS, NJG, CLJ. Agree with manuscript results and conclusions: AKW, MZ, JLV, JLS, NJG, CLJ. Jointly developed the structure and arguments for the paper: AKW, CLJ, NJG. Made critical revisions and approved final version: AKW, MZ, JLV, JLS, NJG, CLJ. All authors reviewed and approved of the final manuscript.
DISCLOSURES AND ETHICS
As a requirement of publication the authors have provided signed confirmation of their compliance with ethical and legal obligations including but not limited to compliance with ICMJE authorship and competing interests guidelines, that the article is neither under consideration for publication nor published elsewhere, of their compliance with legal and ethical guidelines concerning human and animal research participants (if applicable), and that permission has been obtained for reproduction of any copyrighted material. This article was subject to blind, independent, expert peer review. The reviewers reported no competing interests.
FUNDING: Authors disclose no funding sources.
COMPETING INTERESTS: Authors disclose no potential conflicts of interest.
REFERENCES
- 1.Herbison AE. Physiology of the GnRH neuronal network. In: Neill JD, editor. Knobil and Neill’s Physiology of Reproduction. 3rd ed. Waltham, MA: Academic Press; pp. 1415–1482. [Google Scholar]
- 2.Wray S. From nose to brain: development of gonadotrophin-releasing hormone-1 neurones. J Neuroendocrinol. 2010;22(7):743–753. doi: 10.1111/j.1365-2826.2010.02034.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Jasoni CL, Porteous RW, Herbison AE. Anatomical location of mature GnRH neurons corresponds with their birthdate in the developing mouse. Dev Dyn. 2009;238(3):524–531. doi: 10.1002/dvdy.21869. [DOI] [PubMed] [Google Scholar]
- 4.Low VF, Fiorini Z, Fisher L, Jasoni CL. Netrin-1 stimulates developing GnRH neurons to extend neurites to the median eminence in a calcium- dependent manner. PLoS One. 2012;7(10):e46999. doi: 10.1371/journal.pone.0046999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Fiorini Z, Jasoni CL. A novel developmental role for kisspeptin in the growth of gonadotrophin-releasing hormone neurites to the median eminence in the mouse. J Neuroendocrinol. 2010;22(10):1113–1125. doi: 10.1111/j.1365-2826.2010.02059.x. [DOI] [PubMed] [Google Scholar]
- 6.Clarkson J, Herbison AE. Development of GABA and glutamate signaling at the GnRH neuron in relation to puberty. Mol Cell Endocrinol. 2006;(254–255):32–38. doi: 10.1016/j.mce.2006.04.036. [DOI] [PubMed] [Google Scholar]
- 7.Clarkson J, Boon WC, Simpson ER, Herbison AE. Postnatal development of an estradiol-kisspeptin positive feedback mechanism implicated in puberty onset. Endocrinology. 2009;150(7):3214–3220. doi: 10.1210/en.2008-1733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Han SK, Gottsch ML, Lee KJ, et al. Activation of gonadotropin-releasing hormone neurons by kisspeptin as a neuroendocrine switch for the onset of puberty. J Neurosci. 2005;25(49):11349–11356. doi: 10.1523/JNEUROSCI.3328-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kauffman AS, Park JH, McPhie-Lalmansingh AA, et al. The kisspeptin receptor GPR54 is required for sexual differentiation of the brain and behavior. J Neurosci. 2007;27(33):8826–8835. doi: 10.1523/JNEUROSCI.2099-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Navarro VM, Gottsch ML, Chavkin C, Okamura H, Clifton DK, Steiner RA. Regulation of gonadotropin-releasing hormone secretion by kisspeptin/dynorphin/neurokinin B neurons in the arcuate nucleus of the mouse. J Neurosci. 2009;29(38):11859–11866. doi: 10.1523/JNEUROSCI.1569-09.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Dudas B, Merchenthaler I. Three-dimensional representation of the neurotransmitter systems of the human hypothalamus: inputs of the gonadotrophin hormone-releasing hormone neuronal system. J Neuroendocrinol. 2006;18(2):79–95. doi: 10.1111/j.1365-2826.2005.01398.x. [DOI] [PubMed] [Google Scholar]
- 12.Seminara SB, Messager S, Chatzidaki EE, et al. The GPR54 gene as a regulator of puberty. N Engl J Med. 2003;349(17):1614–1627. doi: 10.1056/NEJMoa035322. [DOI] [PubMed] [Google Scholar]
- 13.Funes S, Hedrick JA, Vassileva G, et al. The KiSS-1 receptor GPR54 is essential for the development of the murine reproductive system. Biochem Biophys Res Commun. 2003;312(4):1357–1363. doi: 10.1016/j.bbrc.2003.11.066. [DOI] [PubMed] [Google Scholar]
- 14.de Roux N, Genin E, Carel JC, Matsuda F, Chaussain JL, Milgrom E. Hypogonadotropic hypogonadism due to loss of function of the KiSS1-derived peptide receptor GPR54. Proc Natl Acad Sci U S A. 2003;100(19):10972–10976. doi: 10.1073/pnas.1834399100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Semple RK, Achermann JC, Ellery J, et al. Two novel missense mutations in g protein-coupled receptor 54 in a patient with hypogonadotropic hypogonadism. J Clin Endocrinol Metab. 2005;90(3):1849–1855. doi: 10.1210/jc.2004-1418. [DOI] [PubMed] [Google Scholar]
- 16.d’Anglemont de Tassigny X, Fagg LA, Dixon JP, et al. Hypogonadotropic hypogonadism in mice lacking a functional Kiss1 gene. Proc Natl Acad Sci U S A. 2007;104(25):10714–10719. doi: 10.1073/pnas.0704114104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Tenenbaum-Rakover Y, Commenges-Ducos M, Iovane A, Aumas C, Admoni O, de Roux N. Neuroendocrine phenotype analysis in five patients with isolated hypogonadotropic hypogonadism due to a L102P inactivating mutation of GPR54. J Clin Endocrinol Metab. 2007;92(3):1137–1144. doi: 10.1210/jc.2006-2147. [DOI] [PubMed] [Google Scholar]
- 18.Lapatto R, Pallais JC, Zhang D, et al. Kiss1-/- mice exhibit more variable hypogonadism than Gpr54-/- mice. Endocrinology. 2007;148(10):4927–4936. doi: 10.1210/en.2007-0078. [DOI] [PubMed] [Google Scholar]
- 19.Ahmed EI, Zehr JL, Schulz KM, Lorenz BH, DonCarlos LL, Sisk CL. Pubertal hormones modulate the addition of new cells to sexually dimorphic brain regions. Nat Neurosci. 2008;11(9):995–997. doi: 10.1038/nn.2178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Clarkson J, Herbison AE. Postnatal development of kisspeptin neurons in mouse hypothalamus; sexual dimorphism and projections to gonadotropin-releasing hormone neurons. Endocrinology. 2006;147(12):5817–5825. doi: 10.1210/en.2006-0787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Poling MC, Kauffman AS. Organizational and activational effects of sex steroids on kisspeptin neuron development. Front Neuroendocrinol. 2013;34(1):3–17. doi: 10.1016/j.yfrne.2012.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Mayer C, Acosta-Martinez M, Dubois SL, et al. Timing and completion of puberty in female mice depend on estrogen receptor alpha-signaling in kiss-peptin neurons. Proc Natl Acad Sci U S A. 2010;107(52):22693–22698. doi: 10.1073/pnas.1012406108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Herbison AE, Porteous R, Pape JR, Mora JM, Hurst PR. Gonadotropin- releasing hormone neuron requirements for puberty, ovulation, and fertility. Endocrinology. 2008;149(2):597–604. doi: 10.1210/en.2007-1139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Khan AR, Kauffman AS. The role of kisspeptin and RFamide-related peptide-3 neurones in the circadian-timed preovulatory luteinising hormone surge. J Neuroendocrinol. 2012;24(1):131–143. doi: 10.1111/j.1365-2826.2011.02162.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Lomniczi A, Loche A, Castellano JM, et al. Epigenetic control of female puberty. Nat Neurosci. 2013;16(3):281–289. doi: 10.1038/nn.3319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Herbison AE, de Tassigny Xd, Doran J, Colledge WH. Distribution and postnatal development of Gpr54 gene expression in mouse brain and gonadotropin-releasing hormone neurons. Endocrinology. 2010;151(1):312–321. doi: 10.1210/en.2009-0552. [DOI] [PubMed] [Google Scholar]
- 27.Navarro VM, Castellano JM, Fernández-Fernández R, et al. Developmental and hormonally regulated messenger ribonucleic acid expression of KiSS-1 and its putative receptor, GPR54, in rat hypothalamus and potent luteinizing hormone-releasing activity of KiSS-1 peptide. Endocrinology. 2004;145(10):4565–4574. doi: 10.1210/en.2004-0413. [DOI] [PubMed] [Google Scholar]
- 28.Moore LD, Le T, Fan G. DNA methylation and its basic function. Neuropsychopharmacology. 2013;38(1):23–38. doi: 10.1038/npp.2012.112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Schroeder JW, Conneely KN, Cubells JC, et al. Neonatal DNA methylation patterns associate with gestational age. Epigenetics. 2011;6(12):1498–1504. doi: 10.4161/epi.6.12.18296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Murgatroyd C, Patchev AV, Wu Y, et al. Dynamic DNA methylation programs persistent adverse effects of early-life stress. Nat Neurosci. 2009;12(12):1559–1566. doi: 10.1038/nn.2436. [DOI] [PubMed] [Google Scholar]
- 31.Zilberman D, Henikoff S. Genome-wide analysis of DNA methylation patterns. Development. 2007;134(22):3959–3965. doi: 10.1242/dev.001131. [DOI] [PubMed] [Google Scholar]
- 32.Takase K, Uenoyama Y, Inoue N, et al. Possible role of oestrogen in pubertal increase of Kiss1/kisspeptin expression in discrete hypothalamic areas of female rats. J Neuroendocrinol. 2009;21(6):527–537. doi: 10.1111/j.1365-2826.2009.01868.x. [DOI] [PubMed] [Google Scholar]
- 33.Clarkson J, Herbison AE. Oestrogen, kisspeptin, GPR54 and the preovulatory luteinising hormone surge. J Neuroendocrinol. 2009;21(4):305–311. doi: 10.1111/j.1365-2826.2009.01835.x. [DOI] [PubMed] [Google Scholar]
- 34.Schug J. Using TESS to predict transcription factor binding sites in DNA sequence. Curr Protoc Bioinformatics. 2008 doi: 10.1002/0471250953.bi0206s21. Chapter 2:Unit 2.6. [DOI] [PubMed] [Google Scholar]
- 35.Sasai Y, Kageyama R, Tagawa Y, Shigemoto R, Nakanishi S. Two mammalian helix-loop-helix factors structurally related to Drosophila hairy and Enhancer of split. Genes Dev. 1992;6(12B):2620–2634. doi: 10.1101/gad.6.12b.2620. [DOI] [PubMed] [Google Scholar]
- 36.Semaan SJ, Dhamija S, Kim J, Ku EC, Kauffman AS. Assessment of epigenetic contributions to sexually-dimorphic Kiss1 expression in the anteroventral periventricular nucleus of mice. Endocrinology. 2012;153(4):1875–1886. doi: 10.1210/en.2011-1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Reed AL, Yamazaki H, Kaufman JD, Rubinstein Y, Murphy B, Johnson AC. Molecular cloning and characterization of a transcription regulator with homology to GC-binding factor. J Biol Chem. 1998;273(34):21594–21602. doi: 10.1074/jbc.273.34.21594. [DOI] [PubMed] [Google Scholar]
- 38.Peng Z, Shen R, Li YW, Teng KY, Shapiro CL, Lin HJ. Epigenetic repression of RARRES1 is mediated by methylation of a proximal promoter and a loss of CTCF binding. PLoS One. 2012;7(5):e36891. doi: 10.1371/journal.pone.0036891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Tomikawa J, Uenoyama Y, Ozawa M, et al. Epigenetic regulation of Kiss1 gene expression mediating estrogen-positive feedback action in the mouse brain. Proc Natl Acad Sci U S A. 2012;109(20):E1294–E1301. doi: 10.1073/pnas.1114245109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Mueller JK, Dietzel A, Lomniczi A, et al. Transcriptional regulation of the human KiSS1 gene. Mol Cell Endocrinol. 2011;342(1–2):8–19. doi: 10.1016/j.mce.2011.04.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.DeFino MC, Wacker JL, Lyssand JS, Wang EH, Hague C. Differential regulation of GPR54 transcription by specificity protein-1 and partial estrogen response element in mouse pituitary cells. Biochem Biophys Res Commun. 2010;393(4):603–608. doi: 10.1016/j.bbrc.2010.02.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Bibikova M, Laurent LC, Ren B, Loring JF, Fan JB. Unraveling epigenetic regulation in embryonic stem cells. Cell Stem Cell. 2008;2(2):123–134. doi: 10.1016/j.stem.2008.01.005. [DOI] [PubMed] [Google Scholar]
- 43.Deaton AM, Bird A. CpG islands and the regulation of transcription. Genes Dev. 2011;25(10):1010–1022. doi: 10.1101/gad.2037511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Suzuki MM, Bird A. DNA methylation landscapes: provocative insights from epigenomics. Nat Rev Genet. 2008;9(6):465–476. doi: 10.1038/nrg2341. [DOI] [PubMed] [Google Scholar]
- 45.Müller P, Merrell KW, Crofts JD, et al. Estrogen-dependent downregulation of hairy and enhancer of split homolog-1 gene expression in breast cancer cells is mediated via a 3’ distal element. J Endocrinol. 2009;200(3):311–319. doi: 10.1677/JOE-08-0094. [DOI] [PubMed] [Google Scholar]
- 46.Ceschin DG, Walia M, Wenk SS, et al. Methylation specifies distinct estrogen-induced binding site repertoires of CBP to chromatin. Genes Dev. 2011;25(11):1132–1146. doi: 10.1101/gad.619211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Schmittgen TD, Livak KJ. Analyzing real-time PCR data by the comparative C(T) method. Nat Protoc. 2008;3(6):1101–1108. doi: 10.1038/nprot.2008.73. [DOI] [PubMed] [Google Scholar]
- 48.Meyer M, Stenzel U, Hofreiter M. Parallel tagged sequencing on the 454 platform. Nat Protoc. 2008;3(2):267–278. doi: 10.1038/nprot.2007.520. [DOI] [PubMed] [Google Scholar]