Abstract
Background
We investigated the relationship between peripheral neuropathy and parameters of arterial stiffness and carotid intima media thickness (CIMT) in patients with type 2 diabetes mellitus (T2DM).
Material/Methods
The study included 161 patients (80 females and 81 males), 69 of whom had peripheral neuropathy. All patients underwent 24-h blood pressure monitoring, and arterial stiffness parameters were measured. The CIMT was measured using B-mode ultrasonography and patients also underwent transthoracic echocardiographic examination.
Results
Patients with peripheral neuropathy, compared with those without it, were older (54.68±8.35 years vs. 51.04±7.89 years; p=0.005) and had T2DM for longer periods (60 vs. 36 months; p=0.004). Glycated hemoglobin (HbA1c) values (8.55±1.85 mg/dL vs. 7.30±1.51 mg/dL; p<0.001), pulse wave velocity (PWV) (7.74±1.14 m/s vs. 7.15±1.10 m/s; p=0.001), CIMT (anterior 0.74±0.15 mm vs. 0.67±0.13 mm; p=0.01), and left ventricular mass (LVM) index (98.68±26.28 g/m2 vs. 89.71±19.70 g/m2; p=0.02) were all significantly increased in the group with peripheral neuropathy compared to the group without peripheral neuropathy. We determined that duration of diabetes, HbA1c, and LVM index were predictors of peripheral neuropathy.
Conclusions
A significant relationship was found between diabetic neuropathy and increased PWV, a parameter of arterial stiffness, as well as CIMT, a marker of systemic atherosclerosis. Diabetic peripheral neuropathy may be a determinant of subclinical atherosclerosis in T2DM.
MeSH Keywords: Carotid Intima-Media Thickness, Diabetes Mellitus, Type 2, Diabetic Neuropathies, Vascular Stiffness
Background
Type 2 diabetes mellitus (T2DM) is a worldwide epidemic, particularly in developed and developing countries [1]. Peripheral neuropathy, which unfavorably influences patient quality of life quite, is an early and common complication of T2DM. Although reported prevalence rates have differed across studies due to differences in study populations and diagnostic criteria, peripheral neuropathy is involved in 30–50% of patients with T2DM [1,2]. Chronic sensorimotor distal symmetric polyneuropathy is the most common form of diabetic peripheral neuropathy [1]. Hyperglycemia is a primary risk factor associated with diabetic peripheral neuropathy, and other independent predictors include T2DM duration, smoking, alcohol consumption, hypertension, hypertriglyceridemia, and increased body mass index [2,3].
Cardiovascular diseases are the leading causes of death in patients with diabetes, and T2DM is considered as a coronary artery disease equivalent [4]. Vascular changes due to microvascular disease and subclinical atherosclerosis are associated with the development of T2DM-related complications such as nephropathy, retinopathy, and autonomic neuropathy [5–7]. Recent studies have demonstrated the relationship between peripheral neuropathy, which is a frequent complication of T2DM, and atherosclerotic vascular changes [8,9]. These studies consistently demonstrated the relationship between functional parameters of arterial stiffness and peripheral neuropathy. However, results regarding the relationship between carotid intima-media thickness (CIMT) and peripheral neuropathy are contradictory [8,9].
T2DM is a major risk factors for carotid atherosclerosis [10]. The relationship between carotid atherosclerosis and cerebrovascular and coronary artery diseases has been demonstrated in many studies [11,12]. It has been reported that increased CIMT, which is measured by B-mode ultrasonography, is correlated with increased aortic intima-media thickness, which is measured by echocardiography, and is a marker of systemic atherosclerosis [10,12].
The present study investigated the relationship between peripheral neuropathy and functional parameters of arterial stiffness, which were measured by 24-h blood pressure monitoring, and CIMT, which was measured by B-mode ultrasonography, in patients with T2DM.
Material and Methods
Patients
The study population included 161 patients (81 males, mean age 52.60±8.26 years) with T2DM, who were examined using ambulatory blood pressure monitoring, carotid ultrasonography, transthoracic echocardiography, and electromyography between April 2013 and January 2014 at Selcuk University. The diagnosis of T2DM was made based on the criteria of the American Diabetes Association. Medical history was obtained and physical examination was performed in all patients. Individuals with clinically proven coronary heart disease, cerebrovascular disease, peripheral vascular disease, congestive heart failure, valvular heart disease, chronic kidney disease, and neuropathy due to other reasons (e.g., alcoholic neuropathy, carpal tunnel syndrome, and cerebrovascular disease sequel) were excluded from the study. Patients were divided into 2 groups: those with (n=69) and without (n=92) peripheral neuropathy. Venous blood samples for biochemical analyses and hematologic parameters were drawn after 12-h fasting before the patients received any medication. Fasting blood glucose, serum creatinine, total cholesterol, high-density lipoprotein cholesterol (HDL-C), low-density lipoprotein cholesterol (LDL-C), triglyceride, white blood cell (WBC), hemoglobin (Hb), and glycated hemoglobin (HbA1c) levels were recorded. Glomerular filtration rate (GFR) was estimated by MDRD method. The study protocol was approved by the local ethics committee, and all patients gave their written informed consent to participate in the study.
24-h blood pressure monitoring
24-h noninvasive ambulatory blood pressure monitoring was performed in each subject using a Mobile O Graph 24h PWA (I.E.M. GmbH Stolberg Germany), which yields a simultaneous measure of brachial BP, PWV, and augmentation index. The device is supported with an expert software package, Hospital Management System (Hypertension Management System Client Server Company, IEM, GmbH Germany), for analysis of all registered measurements. In addition, the device is able to measure central BP and Alx@75 by an integrated pulse-wave analysis device. Waveforms were recorded with a regular oscillatory brachial cuff suitable for ambulatory measurement the using Austrian Research Centers (ARC) Solver method (Austrian Institute of Technology, Vienna, Austria). The ARC Solver method is a novel method for the determination of aortic systolic BP and Aix based on oscillometric blood pressure measurement with a common cuff. The method uses the pulse-waves assessed at the brachial artery [13,14]. Blood pressure was measured every 15 min during the day and evening (from 6:00 to 22:00) and every 30 min at night (from 22:00 to 06:00). Presence of at least 50 acceptable measurements was considered as an acceptable 24-h blood pressure monitoring recording for our study. The mean systolic blood pressure, diastolic blood pressure, mean arterial pressure, pulse-wave velocity (PWV), and Alx@75 were calculated for 24 h. The distribution of PWV with age and BP category is described and reference values for PWV are established. The mean PWV found was 6.84 m/s±1.65 [15].
Echocardiographic assessment
All patients underwent transthoracic echocardiographic examination using the Vivid E9 system with a 1.5–4.6 MHz probe (GE-Vingmed Ultrasound AS, Horten, Norway). Left ventricular (LV) dimensions and wall thickness were obtained from the parasternal long axis view with the M-mode cursor positioned just below the mitral leaflet tips, perpendicular to the long axis of the LV. LV ejection fraction was measured in accordance with Simpson’s method. LV mass (LVM) was measured using the formula defined by Devereux. LVM index was calculated by dividing LVM by the body surface area. All standard conventional echocardiographic assessments were performed according to the published criteria of the American Society of Echocardiography.
Carotid ultrasonography
CIMT was measured recording ultrasonographic images of both the left and right common carotid arteries with a 4.5–12 MHz linear array transducer (Vivid E9 system; GE Medical Systems, Horten, Norway). A region 1 cm proximal to the carotid bifurcation was identified. The intima-media thickness was measured using automated edge detection software as the distance between the lumen-intima interface and the media-adventitia interface. All examinations were performed by a single experienced examiner, who was blind to the clinical and biochemical data.
Neurological examination
All patients were examined by a neurologist for the presence of peripheral neuropathy. Neuropathic pain was defined as pain in the limbs in the absence of history of trauma or any other external cause. Bilateral spontaneous pain, hypoesthesia including decreased sensation to pinprick and temperature (cold tuning fork), or paresthesia of the legs were considered as the symptoms of polyneuropathy. Response of the Achilles tendon reflex was investigated in the knee-standing position, ie, with reinforcement, with a standard reflex hammer. Results were considered abnormal when no reflex was seen in 1 or both extremities. Vibration perception was measured on the medial malleolus in the lower extremities. Nerve conduction velocity was measured using a Nihonkohden electromyography system (Nihonkohden MEB-9002K VMA EP/EMG Measuring System 2005, USA). Patients with 2 or more of the following 4 components were diagnosed with neuropathy: presence of symptoms, absence of ankle tendon reflexes, abnormal scores of vibration perception, and abnormal nerve conduction velocity.
Statistical analysis
SPSS 17.0 for Windows was used for statistical analyses (SPSS, Chicago, IL, USA). Continuous variables are presented as median or mean ±SD; categorical variables are presented as percentage. Differences in the continuous variables between groups were determined by t test or the Mann-Whitney U test, for variables with or without normal distribution, respectively. To test the normal distribution, the Kolmogorov-Smirnov test was used. Categorical variables are summarized as percentages and compared with the χ2 test. The Pearson correlation coefficient was computed to determine the association between 2 continuous variables. Logistic regression analysis with enter method was performed including independent variables being significantly different between patients with T2DM having a possible causative role for neuropathy (+). All tests of significance were 2-tailed. Statistical significance was defined as a p value of less than 0.05.
Results
In the present study, data of a total of 161 patients (80 females and 81 males) were evaluated. Patients with peripheral neuropathy were older (54.68±8.35 years vs. 51.04±7.89 years; p=0.005) and had T2DM for longer time (month) (60 months vs. 36 months; p=0.004). Likewise, elevated HbA1c value, which indicates poorly controlled T2DM, was higher in the peripheral neuropathy group (8.55±1.85 mg/dL vs. 7.30±1.51 mg/dL; p<0.001). Body mass index, serum lipid concentrations, hematological parameters, and serum thyroid stimulating hormone levels were similar in both groups. Prevalence of hypertension and type of antihypertensive drugs received were similar in both groups. GFR was lower in the peripheral neuropathy group, but the difference was not statistically significant (Table 1).
Table 1.
Demographic and clinical characteristics of peripheral neuropathy (+) and peripheral neuropathy (−) groups.
| Peripheral neuropathy (+) n=69 (mean ±SD) |
Peripheral neuropathy (−) n=92 (mean ±SD) |
p | |
|---|---|---|---|
| Age, years | 51.04±7.89 | 54.68±8.35 | 0.005 |
| Male, n (%) | 43 (62.3) | 38 (41.3) | 0.01 |
| BMI, kg/m2 | 32.16±5.73 | 32.37±4.92 | 0.79 |
| Duration of diabetes (month) | 60 (12–300) | 36 (12–276) | 0.004 |
| Hypertension, n (%) | 26 (37.7) | 35 (38) | 1.0 |
| Smoking, n (%) | 21 (30.4) | 23 (25) | 0.47 |
| FBG (mg/dL) | 190.08±71.01 | 144.65±45.19 | <0.001 |
| Hb (g/dL) | 14.55±1.50 | 14.05±1.50 | 0.03 |
| Cr (mg/dL) | 0.85±0.19 | 0.81±0.14 | 0.20 |
| HbA1c (mg/dL) | 8.55±1.85 | 7.30±1.51 | <0.001 |
| LDL-C (mg/dL) | 119.49±32.21 | 116.10±32.21 | 0.51 |
| HDL-C (mg/dL) | 41.79±10.27 | 41.00±8.77 | 0.59 |
| TG (mg/dL) | 159.5 (58–668) | 185 (76–630) | 0.09 |
| eGFR (mL/min/1.73 m2) | 96.01±25.87 | 91.30±16.91 | 0.16 |
| Insulin, n (%) | 27 (39.1) | 14 (15.2) | 0.001 |
| Oral anti-diabetic, n (%) | 68 (98.6) | 90 (97.8) | 1.0 |
| Hypolipidemic therapy, n (%) | 31 (44.9) | 41 (44.6) | 1.0 |
| Aspirin, n (%) | 27 (39.7) | 35 (38) | 0.87 |
| Beta-blockers, n (%) | 10 (14.5) | 9 (9.8) | 0.46 |
| Ace-inhibitors, n (%) | 7 (10.1) | 8 (8.7) | 0.78 |
| Angiotensin-receptor blockers, n (%) | 26 (37.7) | 34 (37) | 1.0 |
| Diuretics, n (%) | 17 (24.6) | 24 (26.1) | 0.85 |
| Ca-antagonists, n (%) | 9 (13) | 9 (9.8) | 0.61 |
BMI – body mass index; Cr – serum creatinine; eGFR – estimated glomerular filtration rate; FBG – fasting blood glucose; Hb – hemoglobin; HbA1c – glycosylated hemoglobin A; HDL-C – high-density lipoprotein-cholesterol; LDL-C – low-density lipoprotein-cholesterol; TG – triglyceride.
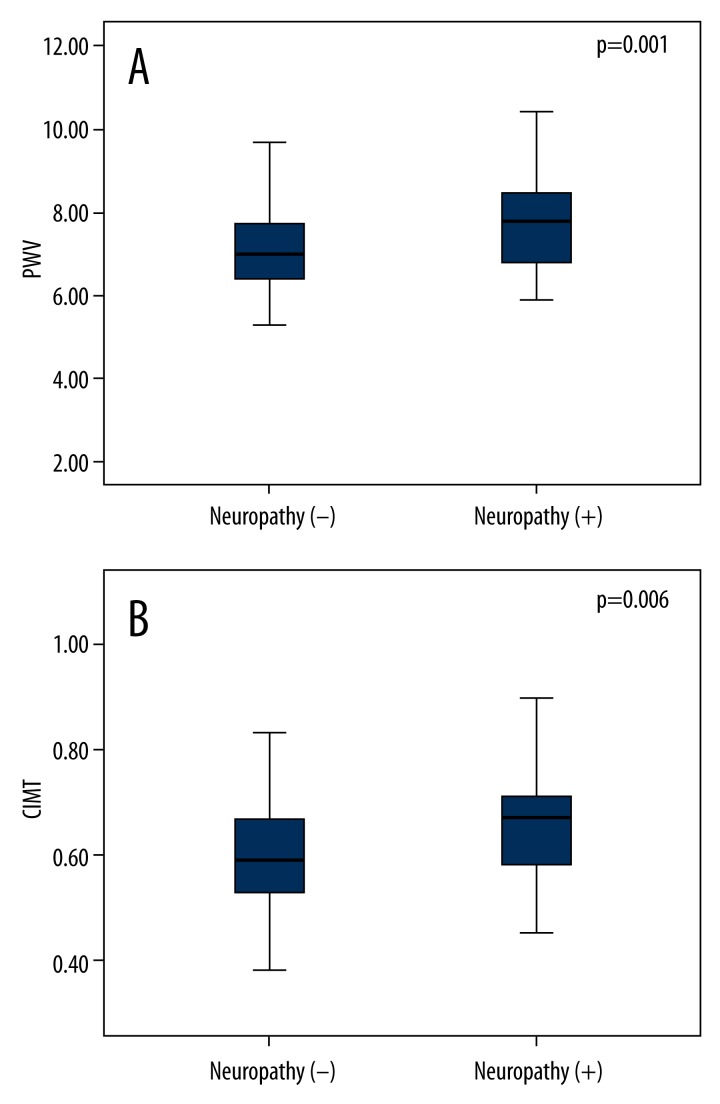
With regard to the results of 24-h blood pressure monitoring, PWV, which is the parameter of arterial stiffness, was statistically increased in the group with peripheral neuropathy as compared to the group without peripheral neuropathy (7.74±1.14 m/s vs. 7.15±1.10 m/s; p=0.001) (Figure 1). Pulse pressure, which is another important parameter of arterial stiffness, was higher in the peripheral neuropathy group, but the difference was not statistically significant. Other results of 24-h blood pressure monitoring were similar in both groups (Table 2).
Figure 1.
Comparisons of PWV (A) and CIMT (B) between peripheral neuropathy (+) and peripheral neuropathy (−) groups.
Table 2.
Comparison of echocardiographic, carotid artery ultrasonographic, and 24-h noninvasive ambulatory blood pressure monitoring parameters between peripheral neuropathy (+) and peripheral neuropathy (−) groups.
| Peripheral neuropathy (+) n=69 (mean ±SD) |
Peripheral neuropathy (−) n=92 (mean ±SD) |
P | |
|---|---|---|---|
| LVEDD (mm) | 48.00±5.91 | 46.54±5.90 | 0.14 |
| LVESD (mm) | 30.42±6.46 | 28.97±4.30 | 0.10 |
| IVS (mm) | 10.01±1.54 | 9.65±1.35 | 0.12 |
| PW (mm) | 9.80±1.23 | 9.41±1.36 | 0.07 |
| LVMI (g/m2) | 98.68±26.28 | 89.71±19.70 | 0.02 |
| EF (%) | 65.61±8.11 | 67.22±5.85 | 0.16 |
| LA diameter (mm) | 35.08±3.68 | 34.16±3.19 | 0.10 |
| Aortic diameter (mm) | 28.38±3.03 | 27.86±2.89 | 0.28 |
| CIMTanterior, (mm) | 0.74±0.15 | 0.67±0.13 | 0.01 |
| CIMTposterior, (mm) | 0.65±0.10 | 0.60±0.10 | 0.006 |
| Mean office SBP (mmHg) | 129.33±20.23 | 125.98±16.61 | 0.25 |
| Mean office DBP (mmHg) | 77.50±12.59 | 78.22±10.51 | 0.69 |
| Mean 24-h SBP (mmHg) | 120.60±13.77 | 119.10±13.92 | 0.49 |
| Mean 24-h DBP (mmHg) | 73.62±8.97 | 73.40±11.86 | 0.89 |
| Mean 24-h MAP (mmHg) | 95.60±9.35 | 93.61±13.93 | 0.30 |
| Mean 24-h PP (mmHg) | 47.37±8.55 | 45.54±9.41 | 0.20 |
| PWV (m/s) | 7.74±1.14 | 7.15±1.10 | 0.001 |
| AIx@75 (%) | 27.70±6.05 | 28.23±7.87 | 0.64 |
AIx@75 – aortic augmentation index corrected for heart rate 75; CIMT – carotid intima media thickness; DBP – diastolic blood pressure; EF – ejection fraction; IVS – interventricular septum; LA – left atrium; LVEDD – left ventricular end-diastolic diameter; LVESD – left ventricular end-systolic diameter; LVMI – left ventricular mass index; MAP – mean arterial pressure; PD – posterior wall; PP – pulse pressure; PWV – pulse wave velocity; SBP – systolic blood pressure.
Echocardiographic evaluation was performed in both groups. LVM index was significantly increased in the group with peripheral neuropathy (98.68±26.28 g/m2 vs. 89.71±19.70 g/m2; p=0.02). Other echocardiographic parameters were found to be similar in both groups (Table 2).
CIMT was measured in the patients. Anterior measurement (0.74±0.15 mm vs. 0.67±0.13 mm; p=0.01) and posterior measurement (0.65±0.10 mm vs. 0.60±0.10 mm; p=0.006) were statistically significantly increased in the group with peripheral neuropathy (Table 2, Figure 1).
Bivariate correlation analysis revealed a significant correlation between PWV with CIMT and LVM index (Figures 2 and 3).
Figure 2.
Correlation between PWV with CIMT.
Figure 3.
Correlation between PWV with LV mass index.
Retrospective stepwise analysis was performed in multivariate analysis to detect the predictors of peripheral neuropathy. Parameters that were considered to be associated with peripheral neuropathy (sex, age, PWV, CIMT, smoking, hypertension, diabetes duration, and HbA1c and LVM index) were evaluated. As the result of analysis, duration of diabetes, HbA1c, and LVM index were determined to be the predictors of peripheral neuropathy (Table 3).
Table 3.
Statistically significant final conditions after the parameters (Sex, Age, PWV, CIMT, Smoking, Hypertension, Diabetes duration, HbA1C, and LV Mass index) thought to be associated with peripheral neuropathy were included in the retrospective stepwise multivariate analysis.
| Independent variables | β±SE | Wald | p |
|---|---|---|---|
| HbA1c | 0.544±0.130 | 17.669 | <0.001 |
| Diabetes duration | 0.010±0.003 | 10.107 | 0.001 |
| LVMI | 0.028±0.010 | 7.955 | 0.005 |
β±SE – beta ± standard error. Significant p values are demonstrated in bold.
Discussion
Increased PWV, which is a parameter of arterial stiffness, and increased CIMT, which is an early sign of systemic atherosclerosis, were significantly higher in the peripheral neuropathy group than in the group without peripheral neuropathy. LVM index was higher in diabetic patients with peripheral neuropathy independently of the prevalence of hypertension and antihypertensive medications administered. As expected, we demonstrated a significant relationship of peripheral neuropathy with the duration of T2DM and with HbA1c values.
T2DM is an endocrine disease that occurs secondary to insulin deficiency and that impairs carbohydrate metabolism as well as lipid and protein metabolism [16]. The chronic course of the disease is associated with an increased risk for complications such as retinopathy, nephropathy, neuropathy, and atherosclerosis [1,3,5,6,16]. Many pathophysiological mechanisms, including decreased Na+/K+ ATPase activity, increased levels of vasoconstrictors such as thromboxane A2 and endothelin, decreased levels of vasodilators such as prostaglandin I2 and nitric oxide, increased aldose reductase activity, increased production of reactive oxygen species and free radicals, increased protein glycation, altered lipoprotein metabolism, and increased protein kinase C activity are responsible for the development of microvascular and macrovascular complications of diabetes [16–18]. In the present study, a strong correlation was detected between the development of peripheral neuropathy and increasing age, duration of T2DM, and HbA1c level, which is a glycemic control marker. Dyck et al. conducted a study in 264 diabetic patients and found a strong correlation between exposure to hyperglycemia and peripheral neuropathy [19]. Many studies have demonstrated a relationship among age, diabetes duration, and HbA1c levels, supporting the results of the present study [1–3,8,20]. However, although previous studies demonstrated a relationship among peripheral neuropathy, hypertension, and nephropathy, we found no relationship between hypertension and peripheral neuropathy; however, a weak relationship was detected between peripheral neuropathy and decreased eGFR, which is a marker of nephropathy [3,8,20].
Cardiovascular disease is a major complication of T2DM, and cardiac diseases are the cause of death in 68% of patients with T2DM [21]. Although quite important, atherosclerosis is usually asymptomatic, particularly in patients with T2DM [12]. In a study performed using myocardial perfusion scintigraphy, silent myocardial ischaemia was found in 22% of the patients with T2DM [12,22]. Atherosclerosis is a multifactorial disease frequently involving the entire arterial system. Therefore, the severity of atherosclerotic changes in any segment of the arterial system can provide information about other atherosclerotic involvements in the arterial system [23]. Easy visualization of carotid arteries (and thereby determination of the extensiveness of atherosclerosis in these arteries) may also provide information about the presence and severity of accompanying coronary atherosclerosis [11,12,23]. It has been demonstrated that intima-media thickening, an early sign of atherosclerosis in the vascular bed, is associated with cardiovascular risk factors and the extensiveness of symptomatic coronary artery disease [23,24]. Moreover, long-term follow-up of asymptomatic patients revealed that increased CIMT is associated with an increased risk of stroke and silent cerebral infarction [11,25]. The relationship between T2DM and increased CIMT has been demonstrated in many studies. Yamasaki et al. showed that CIMT increases in diabetic versus non-diabetic subjects [26]. A meta-analysis including 4019 patients with diabetes demonstrated a relationship among impaired glucose tolerance, diabetes, and increased CIMT [27]. Diabetic peripheral neuropathy is a common microvascular complication with high mortality rates, but little is known about the association between diabetic peripheral neuropathy and atherosclerotic vascular changes [9]. A previous study demonstrated that CIMT increases in patients with versus without peripheral neuropathy, which is among the chronic complications of diabetes [8]. However, another study failed to demonstrate such a relationship [9]. In the present study, we demonstrated increased CIMT in patients with peripheral neuropathy compared with those without peripheral neuropathy.
Arterial stiffness is a sensitive marker indicative of decreased arterial elasticity and vascular wall injury. A relationship has been demonstrated between carotid atherosclerosis, which is measured by angiography, and increased PWV, an arterial stiffness parameter [28]. PWV is a non-invasive, relatively cheap, easily applicable, and reliable test for measuring arterial stiffness [20,29]. Studies revealed that arterial stiffness measured by PWV increases in patients with diabetes with peripheral neuropathy compared with those without peripheral neuropathy [7,8,30]. Consistent with previous studies, the present study measured PWV, indicating that arterial stiffness increased in patients with diabetes with peripheral neuropathy. However, the underlying mechanism linking peripheral neuropathy in diabetic patients to arterial stiffness is not well understood. One possible explanation is that large artery stiffness may cause microvascular damage via high pulse pressure, leading to diminished blood flow to nerve tissues vulnerable to hypoxic damage, and thereby to the development of neuropathy [9].
LV hypertrophy is an important cardiovascular risk factor. The incidence and mortality rate of cardiovascular events can decrease with treatment of LV hypertrophy [31]. Therefore, identifying and controlling the factors that cause LV hypertrophy are quite important. Studies have revealed a relationship among age, hypertension, obesity, and the LVM index and, accordingly, LV hypertrophy [32]. The micro-vascular damage, which causes the elevation of central blood pressure, might have a closer relationship with LV hypertrophy and LVM index [33]. In the present study, although blood pressure, prevalence of hypertension and anti-hypertensive medication usage were similar among the patients, the LVM index was increased in the peripheral neuropathy group. This difference can be explained by the micro-vascular damage of the patients with peripheral neuropathy group. Also, a relationship between the LVM index and PWV was demonstrated in this study.
Limitations
The main limitation of the present study arises from its cross-sectional design. It was not possible to establish a cause-and-effect relationship between peripheral neuropathy, CIMT, and arterial stiffness. Moreover, we did not determine predictors of peripheral neuropathy, since neither the patient number nor study design used were suitable for this.
Conclusions
The study determined a significant relationship of peripheral neuropathy with increased PWV and CIMT in patients with T2DM. The duration of diabetes, increased HbA1C, and increased LVM index were predictors of diabetic peripheral neuropathy.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Source of support: Departmental sources
References
- 1.Boulton AJM, Vinik AI, Arezzo JC, et al. Diabetic neuropathies: a statement by the American Diabetes Association. Diabetes Care. 2005;28:956–62. doi: 10.2337/diacare.28.4.956. [DOI] [PubMed] [Google Scholar]
- 2.Won JC, Kim SS, Ko KS, et al. Current Status of Diabetic Peripheral Neuropathy in Korea: Report of a Hospital-Based Study of Type 2 Diabetic Patients in Korea by the Diabetic Neuropathy Study Group of the Korean Diabetes Association. Diabetes Metab J. 2014;38:25–31. doi: 10.4093/dmj.2014.38.1.25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Tesfaye S, Chaturvedi N, Eaton SE, et al. Vascular risk factors and diabetic neuropathy. N Engl J Med. 2005;352:341–50. doi: 10.1056/NEJMoa032782. [DOI] [PubMed] [Google Scholar]
- 4.Ryden L, Standl E, Bartnik M, et al. Guidelines on diabetes, pre-diabetes, and cardiovascular diseases: executive summary. The Task Force on Diabetes and Cardiovascular Diseases of the European Society of Cardiology (ESC) and of the European Association for the Study of Diabetes (EASD) Eur Heart J. 2007;28:88–136. doi: 10.1093/eurheartj/ehl260. [DOI] [PubMed] [Google Scholar]
- 5.Yun YW, Shin MH, Lee YH, et al. Arterial stiffness is associated with diabetic retinopathy in Korean type 2 diabetic patients. J Prev Med Public Health Yebang Ŭihakhoe Chi. 2011;44:260–66. doi: 10.3961/jpmph.2011.44.6.260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bouchi R, Babazono T, Mugishima M, et al. Arterial stiffness is associated with incident albuminuria and decreased glomerular filtration rate in type 2 diabetic patients. Diabetes Care. 2011;34:2570–75. doi: 10.2337/dc11-1020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bagherzadeh A, Nejati-Afkham A, Tajallizade-Khoob Y, et al. Association of cardiac autonomic neuropathy with arterial stiffness in type 2 diabetes mellitus patients. J Diabetes Metab Disord. 2013;12:55. doi: 10.1186/2251-6581-12-55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Yokoyama H, Yokota Y, Tada J, et al. Diabetic neuropathy is closely associated with arterial stiffening and thickness in Type 2 diabetes. Diabet Med J Br Diabet Assoc. 2007;24:1329–35. doi: 10.1111/j.1464-5491.2007.02278.x. [DOI] [PubMed] [Google Scholar]
- 9.Kim ES, Moon S, Kim H-S, et al. Diabetic peripheral neuropathy is associated with increased arterial stiffness without changes in carotid intima-media thickness in type 2 diabetes. Diabetes Care. 2011;34:1403–5. doi: 10.2337/dc10-2222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Yuan C, Lai CWK, Chan LWC, et al. Cumulative effects of hypertension, dyslipidemia, and chronic kidney disease on carotid atherosclerosis in chinese patients with type 2 diabetes mellitus. J Diabetes Res. 2014;2014:179686. doi: 10.1155/2014/179686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.O’Leary DH, Polak JF, Kronmal RA, et al. Carotid-artery intima and media thickness as a risk factor for myocardial infarction and stroke in older adults. Cardiovascular Health Study Collaborative Research Group. N Engl J Med. 1999;340:14–22. doi: 10.1056/NEJM199901073400103. [DOI] [PubMed] [Google Scholar]
- 12.Katakami N, Kaneto H, Shimomura I. Carotid ultrasonography: A potent tool for better clinical practice in diagnosis of atherosclerosis in diabetic patients. J Diabetes Investig. 2014;5:3–13. doi: 10.1111/jdi.12106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Wei W, Tölle M, Zidek W, et al. Validation of the mobil-O-Graph: 24h-blood pressure measurement device. Blood Press Monit. 2010;15:225–28. doi: 10.1097/MBP.0b013e328338892f. [DOI] [PubMed] [Google Scholar]
- 14.Wassertheurer S, Kropf J, Weber T, et al. A new oscillometric method for pulse wave analysis: comparison with a common tonometric method. J Hum Hypertens. 2010;24:498–504. doi: 10.1038/jhh.2010.27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Díaz A, Galli C, Tringler M, et al. Reference values of pulse wave velocity in healthy people from an urban and rural Argentinean population. Int J Hypertens. 2014;2014:653239. doi: 10.1155/2014/653239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ostenson CG. The pathophysiology of type 2 diabetes mellitus: an overview. Acta Physiol Scand. 2001;171:241–47. doi: 10.1046/j.1365-201x.2001.00826.x. [DOI] [PubMed] [Google Scholar]
- 17.Komosińska-Vassev K, Olczyk K, Olczyk P, et al. Effects of metabolic control and vascular complications on indices of oxidative stress in type 2 diabetic patients. Diabetes Res Clin Pract. 2005;68:207–16. doi: 10.1016/j.diabres.2004.10.004. [DOI] [PubMed] [Google Scholar]
- 18.Sandireddy R, Yerra VG, Areti A, et al. Neuroinflammation and Oxidative Stress in Diabetic Neuropathy: Futuristic Strategies Based on These Targets. Int J Endocrinol. 2014;2014:674987. doi: 10.1155/2014/674987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Dyck PJ, Davies JL, Wilson DM, et al. Risk factors for severity of diabetic polyneuropathy: intensive longitudinal assessment of the Rochester Diabetic Neuropathy Study cohort. Diabetes Care. 1999;22:1479–86. doi: 10.2337/diacare.22.9.1479. [DOI] [PubMed] [Google Scholar]
- 20.Maser RE, Steenkiste AR, Dorman JS, et al. Epidemiological correlates of diabetic neuropathy. Report from Pittsburgh Epidemiology of Diabetes Complications Study. Diabetes. 1989;38:1456–61. doi: 10.2337/diab.38.11.1456. [DOI] [PubMed] [Google Scholar]
- 21.Adams JN, Raffield LM, Freedman BI, et al. Analysis of common and coding variants with cardiovascular disease in the Diabetes Heart Study. Cardiovasc Diabetol. 2014;13:77. doi: 10.1186/1475-2840-13-77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Wackers FJT, Chyun DA, Young LH, et al. Resolution of asymptomatic myocardial ischemia in patients with type 2 diabetes in the Detection of Ischemia in Asymptomatic Diabetics (DIAD) study. Diabetes Care. 2007;30:2892–98. doi: 10.2337/dc07-1250. [DOI] [PubMed] [Google Scholar]
- 23.Jadhav UM, Kadam NN. Carotid intima-media thickness as an independent predictor of coronary artery disease. Indian Heart J. 2001;53:458–62. [PubMed] [Google Scholar]
- 24.Mukherjee D, Yadav JS. Carotid artery intimal-medial thickness: indicator of atherosclerotic burden and response to risk factor modification. Am Heart J. 2002;144:753–59. doi: 10.1067/mhj.2002.124865. [DOI] [PubMed] [Google Scholar]
- 25.Hougaku H, Matsumoto M, Handa N, et al. Asymptomatic carotid lesions and silent cerebral infarction. Stroke J Cereb Circ. 1994;25:566–70. doi: 10.1161/01.str.25.3.566. [DOI] [PubMed] [Google Scholar]
- 26.Yamasaki Y, Kawamori R, Matsushima H, et al. Asymptomatic hyperglycaemia is associated with increased intimal plus medial thickness of the carotid artery. Diabetologia. 1995;38:585–91. doi: 10.1007/BF00400728. [DOI] [PubMed] [Google Scholar]
- 27.Brohall G, Odén A, Fagerberg B. Carotid artery intima-media thickness in patients with Type 2 diabetes mellitus and impaired glucose tolerance: a systematic review. Diabet Med J Br Diabet Assoc. 2006;23:609–16. doi: 10.1111/j.1464-5491.2005.01725.x. [DOI] [PubMed] [Google Scholar]
- 28.Shen L, Wu W, You B, et al. Relationship between pulse wave velocity and carotid atherosclerosis in geriatric people. Cerebrovasc Dis Basel Switz. 2011;32:16–20. doi: 10.1159/000330316. [DOI] [PubMed] [Google Scholar]
- 29.Song HG, Kim EJ, Seo HS, et al. Relative contributions of different cardiovascular risk factors to significant arterial stiffness. Int J Cardiol. 2010;139:263–68. doi: 10.1016/j.ijcard.2008.10.032. [DOI] [PubMed] [Google Scholar]
- 30.Kim ES, Moon S, Kim H-S, et al. Diabetic peripheral neuropathy is associated with increased arterial stiffness without changes in carotid intima-media thickness in type 2 diabetes. Diabetes Care. 2011;34:1403–5. doi: 10.2337/dc10-2222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Gardin JM, McClelland R, Kitzman D, et al. M-mode echocardiographic predictors of six- to seven-year incidence of coronary heart disease, stroke, congestive heart failure, and mortality in an elderly cohort (the Cardiovascular Health Study) Am J Cardiol. 2001;87:1051–57. doi: 10.1016/s0002-9149(01)01460-6. [DOI] [PubMed] [Google Scholar]
- 32.Levy D, Garrison RJ, Savage DD, et al. Prognostic implications of echocardiographically determined left ventricular mass in the Framingham Heart Study. N Engl J Med. 1990;322:1561–66. doi: 10.1056/NEJM199005313222203. [DOI] [PubMed] [Google Scholar]
- 33.Masugata H, Senda S, Inukai M, et al. Association between left ventricular hypertrophy and changes in arterial stiffness during hypertensive treatment. Clin Exp Hypertens. 2014;36:258–62. doi: 10.3109/10641963.2013.810229. [DOI] [PubMed] [Google Scholar]