Abstract
Multiple myeloma (MM) is a clonal malignancy characterized by the proliferation of malignant plasma cells in the bone marrow and the production of monoclonal immunoglobulin. In addition to genetic changes, gene hypermethylation is an alternative mechanism of tumor suppressor gene inactivation in MM. The cyclin-dependent kinase inhibitor 1 (CDKN2B or p15INK4B) gene lies adjacent to the tumor suppressor gene, cyclin-dependent kinase inhibitor 2 (CDKN2A), and is frequently mutated and deleted in a wide variety of tumors, including MM. However, there is a lack of systematic analysis of p15 epigenetic modification such as methylation in MM from different studies that can provide more powerful estimation of an effect. In this study, we have systematically reviewed the studies of p15INK4B promoter methylation in MM and quantified the association between p15INK4B promoter methylation and MM using meta-analysis methods. We observed that the frequency of p15INK4B methylation is significantly higher in MM patients than in normal healthy controls. The pooled odds ratio (OR) from ten studies including 394 MM and 99 normal individuals is 0.08, while confidence interval (CI) is 0.03–0.21 (P<0.00001). This indicates that p15INK4B inactivation through methylation plays an important role in the pathogenesis of MM. In addition, the frequency of p15INK4B methylation was significantly higher in patients with MM than in those with asymptomatic monoclonal gammopathy of undetermined significance. The pooled OR from four studies is 0.40, 95% CI =0.21–0.78 (P=0.007). These results suggest that silencing of p15INK4B gene expression by epigenetic modification such as promoter hypermethylation plays a role not only in the initiation of MM but also in plasma cell malignant transformation, disease progression, and development.
Keywords: multiple myeloma, asymptomatic monoclonal gammopathy of undetermined significance (MGUS), p15, methylation, meta-analysis
Introduction
In tumor cells, the pattern of methylation is changed. There is a growing list of tumor suppressor genes (TSGs) with increased methylation frequency at promoter region1 with detectable gene–body DNA hypomethylation.2 Since aberrant hypermethylation of CpG dinucleotides in the promoter of TSG is a commonly observed epigenetic modification in human tumors,3–5 analysis of specific gene promoter methylation as a tool for diagnosis and prognosis of tumors has been widely applied in many different tumors, including multiple myeloma (MM).6,7
MM is a clonal malignancy characterized by the proliferation of malignant plasma cells in the bone marrow and the production of monoclonal immunoglobulin.8 In the US, MM accounts for ~1% of all malignant diseases or ~15% of all hematological malignancies and is the second most common form of hematological malignancies.9 Clinically, MM starts with immortalization of a postgerminal center B-cell and presents as asymptomatic monoclonal gammopathy of undetermined significance (MGUS). MGUS is considered as the precursor of MM, since it is able to progress to symptomatic MM at a rate of 1% per year.10 MGUS plasma cells share many abnormal characteristics with MM plasma cells. Cytogenetic abnormalities, such as 14q32 translocations, deletions of 13q, and numeric gains of multiple chromosomes, have been discovered at the level of MGUS, indicating early onset of karyotypic instability in monoclonal gammopathies.11 Genetically, MM develops and progresses due to the accumulation of multiple defined pathological genetic events. By genome-wide methylation microarrays or gene-specific hypermethylation analysis, aberrant DNA methylation in MM has been found to inactivate a number of protein-coding TSGs in the process of regulating cell cycle progression, cell growth, or apoptosis, which include cyclin-dependent kinase inhibitor 2A (p16CDKN2A), cyclin-dependent kinase inhibitor 2B (p15CDKN2B or p15INK4B), death-associated protein kinase (DAP), secreted frizzled-related protein 2, suppressor of cytokine signaling 1 (SOCS1),12,13 and microribonucleic acid.14 However, there is a lack of a thorough summary of different studies on p15INK4B epigenetic methylation in MM that can provide the reader with extensive information on whether an effect exists and what size the effect is. In the present study, we will carry out a meta-analysis of p15INK4B methylation status in MM and in its precursor MGUS.
Methods
Search strategy
Medline, PubMed, Web of Science, Scopus, and Embase were searched in April 2014 using the search terms “p15”, “p15INK4B”, “cyclin-dependent kinase inhibitor 2B”, “methylation”, “multiple myeloma”, and “clinical studies”. Investigations identified through the search approach as described were screened by titles first, then by abstracts of the publications. After exclusion of nonrelevant publications and identification of duplicates from the different databases, the remaining papers were evaluated in the full-text version for inclusion and exclusion criteria and for relevant articles in the reference lists. All clinical studies except case reports were chosen: eg, randomized controlled trials, cohort studies, case control studies, and case series. The language of publication was restricted to English and Chinese. All searched data were retrieved. Authors’ bibliographies and references of selected studies were also searched for other relevant studies. The most complete study was chosen to avoid duplication if the same patient populations were reported in several publications.
Selection criteria
We collected all eligible articles about the relationship between p15INK4B methylation and/or expression and clinicopathological features and clinical outcomes in MM patients in this meta-analysis. Diagnosis and staging classification of MM must follow the standard criteria as described previously.15,16 In detail, MM requires evidence of bone marrow plasmacytosis, M-protein in serum or urine, and osteolytic bone lesions and the presence of myeloma-related organ or tissue impairment. MGUS is characterized15,16 by monoclonal protein <30 g/L and bone marrow clonal cells <10% with no evidence of MM, other B-cell proliferative disorders, or amyloidosis. Normal controls are referred to as the normal peripheral blood samples or bone marrow aspirates obtained from the healthy donors. Studies meeting the following inclusion criteria were included: 1) p15INK4B methylation and/or expression evaluated in the circulation and/or bone marrow, 2) research that revealed the relationship between p15INK4B methylation and/or expression and MM clinicopathological parameters and prognosis, 3) p15INK4B methylation and/or expression examined by methylation-specific polymerase chain reaction (MSP) or Southern blot, 4) articles that were published as full papers in English or Chinese, and 5) articles that provided sufficient information to estimate hazard ratio (HR) about overall survival (OS) and 95% confidence interval (CI) and probabilities for progression-free survival (PFS) where applicable. The exclusion criteria included the following: 1) letters, reviews, case reports, conference abstracts, editorials, expert opinion, and non-English language papers; 2) articles with no information on OS or those that could not calculate the HR about OS from the given information; and 3) all publications regarding in vitro/ex vivo studies, cell lines, and human xenografts.
Data extraction
Two investigators independently extracted data from eligible studies. Disagreements were resolved by discussion and consensus. Two investigators reviewed all of the articles that fit inclusion and exclusion criteria. The following information was recorded for each study: the first author name, year of publication, sample source, number of cases, clinicopathological parameters, stage, p15INK4B methylation and/or expression, and patient survival. Data for study characteristics and clinical response were summarized and turned into table format. Heterogeneity of investigation was evaluated to determine whether the data of the various studies could be analyzed in a meta-analysis.
Statistical analysis
Analysis was conducted using Stata 12.0 (Stata Corporation, TX, USA) and Review Manager 5.2 (Cochrane Collaboration, Oxford, UK). Comparisons of dichotomous measures were done by pooled estimates of odds ratios (ORs) as well as their 95% CIs. P-value of <0.05 was considered to be statistically significant. Heterogeneity was examined by a chi-square test with significance being set at P<0.10; the total variation among studies was estimated by I square. If there was heterogeneity among studies, we used a random effect model to pool the ORs; otherwise, a fixed effect model was selected.
The database search generated 25 articles from Medline, PubMed, the Web of Science, Scopus, and Embase. After initial screening of all titles, abstracts, and eligibility, 12 full-text studies were retracted for more detailed assessment. The search of the article references did not produce additional publications. Eventually, 12 publications met the inclusion criteria for qualitative study and for meta-analysis. The article search and study selection are depicted in Figure 1.
Figure 1.
Flow chart of study selection.
Results
Identification of relevant studies
Twenty-five publications were identified by the search method as described. Thirteen of those were excluded due to laboratory studies, nonoriginal articles (review), or studies irrelevant to the current analysis. Eventually, there were 12 studies included in the final meta-analysis (Figure 1).17–28
Study characteristics
Twelve studies published from 1997 to 2010 were eligible for meta-analysis. A total of 488 MM and 99 MGUS patients from the People’s Republic of China, France, Germany, Austria, Spain, Brazil, and Argentina were enrolled. Their basic characteristics are summarized in Table 1.
Table 1.
Basic characteristics of the included studies
| Study/country | Patients
|
Methods | Primary aim | Methylation site | |
|---|---|---|---|---|---|
| MGUS | MM | ||||
| Ng et al25/People’s Republic of China | 12 | MSP | Investigate whether p16/p15 genes are inactivated by hypermethylation of the 5′ CpG islands | Promoter, CpG islands | |
| Fan et al20/People’s Republic of China | 24 | MSP | Study the action, characteristics, and expression of high methylation of p15 and p16 genes in MM | Promoter, CpG islands | |
| Wu et al28/People’s Republic of China | 12 | MSP | Study the effect of operative region hypermethylation gene in human malignant hematopoietic tumors | Promoter, CpG islands | |
| Guillerm et al23/France | 33 | 33 | MSP | Determine how and when the alterations of p15 and p16 contribute to tumorigenesis | Promoter, CpG islands |
| Chen et al18/People’s Republic of China | 22 | MSP | Investigate the frequency of p16 and p15 methylation and its relationship with bone marrow cell apoptosis and clinical outcome | Promoter, CpG islands | |
| Guillerm et al22/France | 61 | MSP | Investigate different prognostic values of p15 and p16 gene methylation in MM | Promoter, CpG islands | |
| Galm et al21/Germany | 3 | 56 | MSP | Gain insight into specific pathways that are relevant in the multiple step transformation process of normal plasma cells to MM | Promoter, CpG islands |
| Seidl et al26/Austria | 29 | 113 | MSP | Investigate the methylation frequencies of ten genes in MGUS and compare with MM | Promoter, CpG islands |
| Chim et al19/People’s Republic of China | 13 | MSP | Study the frequency and whether multiple genes are methylated in any one individual | Promoter, CpG islands | |
| Martin et al24/Spain | 13 | 30 | MSP | Explore the role of DNA methylation in plasma cell disorders | Promoter, CpG islands |
| Braggio et al17/Brazil | 68 | MSP | Gain insights on the prognostic implication resulting from epigenetic silencing | Promoter, CpG islands | |
| Stanganelli et al27/Argentina | 21 | 44 | MSP | Determine the role of aberrant promoter methylation as one of the steps involved in the progression of MGUS to MM | Promoter, CpG islands |
Abbreviations: MGUS, monoclonal gammopathy of undetermined significance; MM, multiple myeloma; MSP, methylation-specific polymerase chain reaction.
p15 methylation and clinicopathological features
The inactivation of p15INK4B through methylation in MGUS
We observed that p15INK4B methylation is higher in MGUS than in normal individual controls, but it did not reach significant difference. The pooled ORs from three studies including 63 patients with MGUS and 19 healthy individuals are shown in Figure 2 (OR=0.30, 95% CI=0.05–1.72, P=0.18). These findings indicate that although MGUS is considered as a premalignant condition, p15INK4B gene methylation is not the only one determinant factor for its potential malignancy. We excluded the other two studies that showed that p15INK4B methylation in MGUS is 1.8%21 and 6.5%,22 because no healthy individual controls were available.
Figure 2.
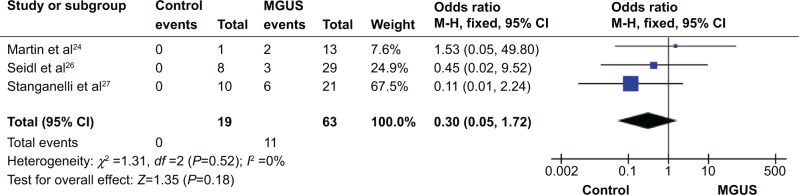
p15 methylation in MGUS.
Notes: Three of the included studies investigated p15INK4B methylation status between 63 multiple myeloma patients and 19 normal individuals. The combined odds ratio was 0.30 (95% CI: 0.05–1.72; Z=1.35; P=0.18).
Abbreviations: MGUS, monoclonal gammopathy of undetermined significance; CI, confidence interval.
The inactivation of p15INK4B through methylation in MM
We observed that p15INK4B methylation is significantly higher in MM than in normal healthy controls. The pooled ORs from ten studies including 394 MM and 99 normal individuals are shown in Figure 3 (OR=0.08, 95% CI=0.03–0.21, P<0.00001), indicating that p15 inactivation through methylation plays an important role in the pathogenesis of myeloma.
Figure 3.
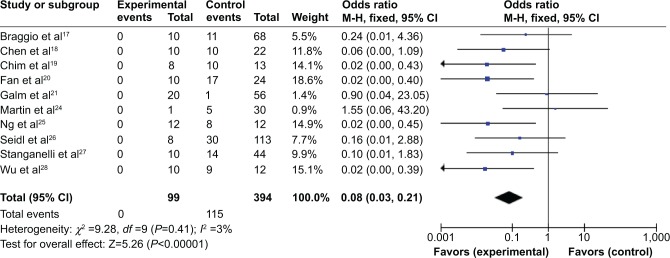
p15 methylation in multiple myeloma.
Notes: Ten of included studies investigated p15INK4B methylation status between 394 patients with multiple myeloma and 99 normal individuals. The combined odds ratio was 0.08 (95% confidence interval [CI]: 0.03–0.21; Z=5.26; P<0.00001).
The role of p15INK4B methylation in plasma cell malignant transformation
We analyzed 220 MM and 96 MGUS patients pooled in four studies to assess whether the aberrant p15INK4B methylation/expression in serum/bone marrow DNA was associated with advanced stage. P15INK4B methylation/expression estimated in blood/bone marrow samples in early-stage MGUS and late MM stage was examined. The frequency of p15INK4B inactivation was significantly higher in patients with MM than in those with MGUS (Figure 4), OR=0.40, 95% CI=0.21–0.78, P=0.007. These results suggest that silencing of p15INK4B gene expression by promoter hypermethylation may contribute to plasma cell malignant transformation, disease progression, and development.
Figure 4.
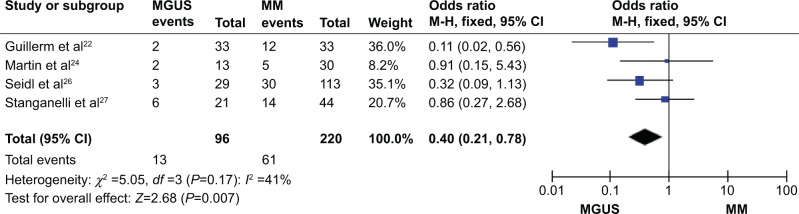
p15 methylation in MM versus MGUS.
Notes: Four of the included studies investigated p15INK4B methylation status between 220 patients with MM and 96 patients with MGUS. The combined odds ratio was 0.40 (95% CI: 0.21–0.78; Z=2.68; P=0.007).
Abbreviations: MM, multiple myeloma; MGUS, monoclonal gammopathy of undetermined significance; CI, confidence interval.
Prognostic values of p15INK4B gene methylation in MM
Only three included studies21,22,26 estimated the relationship between OS/PFS and p15INK4B methylation/expression. All of them showed the absence of prognostic impact of p15INK4B gene methylation on MM patients. The data from Guillerm et al22 demonstrated that there is no significant difference in OS/PFS of 61 MM patients with (n=13; median survival 8.6 years) or without (n=16; median survival 8.6 years) p15INK4B gene methylation (P=0.59/P=0.28, respectively). Two other studies21,26 contained just narrative description; therefore, we were unable to calculate the pooled HR for OS/PFS.
Sensitivity analyses and publication bias
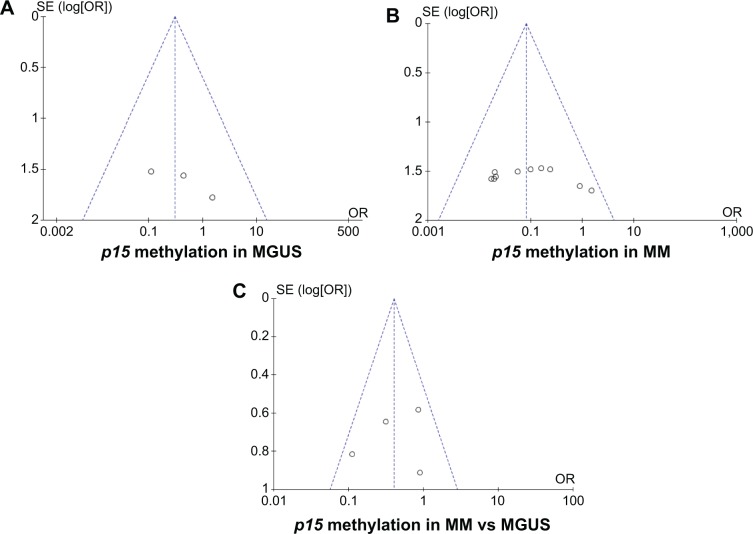
A sensitivity analysis in which one study was removed at a time was conducted to assess the result stability. The pooled ORs were not significantly changed, indicating the stability of our analyses. The funnel plots were largely symmetric (Figure 5), suggesting there were no publication biases in the meta-analysis of p15INK4B methylation/expression and clinicopathological features.
Figure 5.
p15 methylation in MM versus MGUS.
Notes: Funnel plot of publication bias in the meta-analysis of p15INK4B expression and clinicopathological features. (A) p15INK4B methylation in MGUS. (B) p15INK4B methylation in MM. (C) p15INK4B methylation in MM versus MGUS.
Abbreviations: MM, multiple myeloma; MGUS, monoclonal gammopathy of undetermined significance; SE, standard error; OR, odds ratio.
Discussion
MM is an incurable B-cell tumor. Pathogenesis involves upregulation of D-type cyclins, activation of oncogenes, and inactivation of TSGs.12 A better understanding of inactivation of TSGs by gene hypermethylation in MM pathogenesis will lead to more rational combinations of drugs targeting true epigenetic changes and cytotoxic actions. Since gene hypermethylation is a common mechanism of TSG inactivation, various approaches have been used to elucidate the role of gene hypermethylation in MM, including a candidate gene approach, microarray approach for genes upregulated by hypomethylating agents, and a cancer pathway approach, which enables a comprehensive picture of the involvement of multiple TSGs in MM.12 The main TSGs inactivated by hypermethylation in MM are as follows: 1) the INK4 and CIP/KIP families of cyclin-dependent kinase inhibitors, CDKN2B (p16INK4A) and CDKN2A (p15INK4B); 2) SHP1 protein-tyrosine phosphatase and soluble Wnt inhibitors, which are associated with constitutive activation of JAK/STAT and Wnt signaling; and 3) the DAPK/P14/HDM2/P53/Apaf-1 pathway.12 The present study focused on the involvement of epigenetic changes of p15INK4B in MM pathogenesis and prognosis.
CD138 is a plasma biomarker. Several included studies in this meta-analysis sorted CD138+ plasma cells22,23 for MSP analysis, while others used unsorted mononuclear cells.26 By reviewing various studies on methylation examination in B-cell malignancies for different TSGs such as p16INKAB and p15INK4B, we found that the reported methylation frequencies of these tested genes were comparable in both types of studies.22,29–34 Therefore, we concluded that CD138+ cells sorting is not mandatory for methylation evaluation by MSP technique. In other words, sorting CD138+ or not does not affect the included data analysis.
Epigenetic changes contribute to tumorigenesis and affect initial steps in malignant transformation by altering genome stability and regulating gene expression.35 Analysis of the methylome of CD138+ cells by ligation-mediated polymerase chain reaction assay from different stages of myelomagenesis demonstrated qualitative epigenetic differences between premalignant and malignant stages.35 Plasma cells from MGUS, a premalignant and early stage of myeloma, were characterized by striking widespread hypomethylation, while gene-specific hypermethylation was found to occur in the advanced stages of myeloma. Furthermore, aberrant demethylation in MGUS occurred primarily in CpG islands, whereas differentially methylated loci in cases of myeloma occurred predominantly outside of CpG islands and affected distinct sets of gene pathways.35 These results indicate widespread stage-specific epigenetic changes during myelomagenesis and that early demethylation can be a potential contributor to genome instability seen in myeloma.35 In our included studies, there was a significant difference in hypermethylation status of p15INK4B genes at promoter region between patients with MGUS and MM, which reflects the stage-specific epigenetic changes during myelomagenesis. However, all the studies fail to give information about the methylation situation outside promoter region.
Methylation patterns may be useful as prognostic indicators in MM. Despite possible variations between methods utilized, heterogeneity of patients, and other unknown factors, p15INK4B methylation analysis in MM provides interesting prognostic information. Patients with methylated p15INK4B gene demonstrated significantly poorer OS (P=0.01, log-rank test 8.6 years)21,22 and PFS (P=0.005, 8.6 years).22 By univariate analysis, hypermethylation of DAPK (P<0.001) and RARβ (P=0.01) genes was also identified as an adverse prognostic factor. Median OS of MM patients with hypermethylation in DAPK (4 months) and RARβ (34 months) was significantly lower than in those without hypermethylation.17
Combined with the findings, our meta-analysis data suggest that p15INK4B along with p16INK4A, DAPK, and RARβ methylation might play a role in the initial transformation of plasma cells. However, p15INK4B methylation might exert a lesser influence on subsequent tumor progression.22
Recently, Wang et al36 performed a meta-analysis to reveal the potential contribution of p15INK4B and p16INK4A methylation in the pathogenesis of MM. Their results indicated that the rates of p15INK4B and p16INK4A promoter methylation in MM samples were significantly higher compared with normal controls. Furthermore, they also showed that the aberrant methylation of p15INK4B was significantly correlated with the risk of both Caucasian and Asian MM patients. Our current meta-analysis is consistent with their findings and further strengthens the conclusion that p15INK4B promoter methylation may be an important contributor to the pathogenesis of MM.
Footnotes
Disclosure
The authors report no conflicts of interest in this work.
References
- 1.Esteller M. Profiling aberrant DNA methylation in hematologic neoplasms: a view from the tip of the iceberg. Clin Immunol. 2003;109:80–88. doi: 10.1016/s1521-6616(03)00208-0. [DOI] [PubMed] [Google Scholar]
- 2.Feinberg AP, Tycko B. The history of cancer epigenetics. Nat Rev Cancer. 2004;4:143–153. doi: 10.1038/nrc1279. [DOI] [PubMed] [Google Scholar]
- 3.Ghavifekr Fakhr M, Farshdousti Hagh M, Shanehbandi D, Baradaran B. DNA methylation pattern as important epigenetic criterion in cancer. Genet Res Int. 2013;2013:317569. doi: 10.1155/2013/317569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Delpu Y, Cordelier P, Cho WC, Torrisani J. DNA methylation and cancer diagnosis. Int J Mol Sci. 2013;14:15029–15058. doi: 10.3390/ijms140715029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ma X, Wang YW, Zhang MQ, Gazdar AF. DNA methylation data analysis and its application to cancer research. Epigenomics. 2013;5:301–316. doi: 10.2217/epi.13.26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Dimopoulos K, Gimsing P, Gronbaek K. The role of epigenetics in the biology of multiple myeloma. Blood Cancer J. 2014;4:e207. doi: 10.1038/bcj.2014.29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Sharma A, Heuck CJ, Fazzari MJ, et al. DNA methylation alterations in multiple myeloma as a model for epigenetic changes in cancer. Wiley Interdiscip Rev Syst Biol Med. 2010;2:654–669. doi: 10.1002/wsbm.89. [DOI] [PubMed] [Google Scholar]
- 8.Rollig C, Illmer T. The efficacy of arsenic trioxide for the treatment of relapsed and refractory multiple myeloma: a systematic review. Cancer Treat Rev. 2009;35:425–30. doi: 10.1016/j.ctrv.2009.04.007. [DOI] [PubMed] [Google Scholar]
- 9.Siegel R, Ward E, Brawley O, Jemal A. Cancer statistics, 2011: the impact of eliminating socioeconomic and racial disparities on premature cancer deaths. CA Cancer J Clin. 2011;61:212–236. doi: 10.3322/caac.20121. [DOI] [PubMed] [Google Scholar]
- 10.Kyle RA, Therneau TM, Rajkumar SV, et al. A long-term study of prognosis in monoclonal gammopathy of undetermined significance. N Engl J Med. 2002;346:564–569. doi: 10.1056/NEJMoa01133202. [DOI] [PubMed] [Google Scholar]
- 11.Fonseca R, Bailey RJ, Ahmann GJ, et al. Genomic abnormalities in monoclonal gammopathy of undetermined significance. Blood. 2002;100:1417–1424. [PubMed] [Google Scholar]
- 12.Chim CS, Kwong YL, Liang R. Gene hypermethylation in multiple myeloma: lessons from a cancer pathway approach. Clin Lymphoma Myeloma. 2008;8:331–339. doi: 10.3816/CLM.2008.n.048. [DOI] [PubMed] [Google Scholar]
- 13.Walker BA, Wardell CP, Chiecchio L, et al. Aberrant global methylation patterns affect the molecular pathogenesis and prognosis of multiple myeloma. Blood. 2011;117:553–562. doi: 10.1182/blood-2010-04-279539. [DOI] [PubMed] [Google Scholar]
- 14.Wong KY, Huang X, Chim CS. DNA methylation of microRNA genes in multiple myeloma. Carcinogenesis. 2012;33:1629–1638. doi: 10.1093/carcin/bgs212. [DOI] [PubMed] [Google Scholar]
- 15.International Myeloma Working Group Criteria for the classification of monoclonal gammopathies, multiple myeloma and related disorders: a report of the International Myeloma Working Group. Br J Haematol. 2003;121:749–757. [PubMed] [Google Scholar]
- 16.Durie BG, Salmon SE. A clinical staging system for multiple myeloma. Correlation of measured myeloma cell mass with presenting clinical features, response to treatment, and survival. Cancer. 1975;36:842–854. doi: 10.1002/1097-0142(197509)36:3<842::aid-cncr2820360303>3.0.co;2-u. [DOI] [PubMed] [Google Scholar]
- 17.Braggio E, Maiolino A, Gouveia ME, et al. Methylation status of nine tumor suppressor genes in multiple myeloma. Int J Hematol. 2010;91:87–96. doi: 10.1007/s12185-009-0459-2. [DOI] [PubMed] [Google Scholar]
- 18.Chen W, Wu Y, Zhu J, Liu J, Tan S, Xia C. Methylation of p16 and p15 genes in multiple myeloma. Chin Med Sci J. 2002;17:101–105. [PubMed] [Google Scholar]
- 19.Chim CS, Kwong YL, Fung TK, Liang R. Methylation profiling in multiple myeloma. Leuk Res. 2004;28:379–385. doi: 10.1016/j.leukres.2003.08.008. [DOI] [PubMed] [Google Scholar]
- 20.Fan H, Guo X, Wu Q, et al. Detection of high methylation of p15(INK4B) and p16(INK4A) genes in multiple myeloma. Zhongguo Shi Yan Xue Ye Xue Za Zhi. 2000;8:271–274. Chinese. [PubMed] [Google Scholar]
- 21.Galm O, Wilop S, Reichelt J, et al. DNA methylation changes in multiple myeloma. Leukemia. 2004;18:1687–1692. doi: 10.1038/sj.leu.2403434. [DOI] [PubMed] [Google Scholar]
- 22.Guillerm G, Depil S, Wolowiec D, Quesnel B. Different prognostic values of p15(INK4b) and p16(INK4a) gene methylations in multiple myeloma. Haematologica. 2003;88:476–478. [PubMed] [Google Scholar]
- 23.Guillerm G, Gyan E, Wolowiec D, et al. p16(INK4a) and p15(INK4b) gene methylations in plasma cells from monoclonal gammopathy of undetermined significance. Blood. 2001;98:244–246. doi: 10.1182/blood.v98.1.244. [DOI] [PubMed] [Google Scholar]
- 24.Martin P, Garcia-Cosio M, Santon A, Bellas C. Aberrant gene promoter methylation in plasma cell dyscrasias. Exp Mol Pathol. 2008;84:256–261. doi: 10.1016/j.yexmp.2008.02.003. [DOI] [PubMed] [Google Scholar]
- 25.Ng MH, Chung YF, Lo KW, Wickham NW, Lee JC, Huang DP. Frequent hypermethylation of p16 and p15 genes in multiple myeloma. Blood. 1997;89:2500–2506. [PubMed] [Google Scholar]
- 26.Seidl S, Ackermann J, Kaufmann H, et al. DNA-methylation analysis identifies the E-cadherin gene as a potential marker of disease progression in patients with monoclonal gammopathies. Cancer. 2004;100:2598–2606. doi: 10.1002/cncr.20295. [DOI] [PubMed] [Google Scholar]
- 27.Stanganelli C, Arbelbide J, Fantl DB, Corrado C, Slavutsky I. DNA methylation analysis of tumor suppressor genes in monoclonal gammopathy of undetermined significance. Ann Hematol. 2010;89:191–199. doi: 10.1007/s00277-009-0818-3. [DOI] [PubMed] [Google Scholar]
- 28.Wu Q, Guo X, Fan H, et al. P15(INK4B) gene methylation in malignant hematopoietic diseases. Zhonghua Xue Ye Xue Za Zhi. 2000;21:644–646. Chinese. [PubMed] [Google Scholar]
- 29.Gonzalez M, Mateos MV, Garcia-Sanz R, et al. De novo methylation of tumor suppressor gene p16/INK4a is a frequent finding in multiple myeloma patients at diagnosis. Leukemia. 2000;14:183–187. doi: 10.1038/sj.leu.2401617. [DOI] [PubMed] [Google Scholar]
- 30.Ng MH, To KW, Lo KW, et al. Frequent death-associated protein kinase promoter hypermethylation in multiple myeloma. Clin Cancer Res. 2001;7:1724–1729. [PubMed] [Google Scholar]
- 31.Li Y, Nagai H, Ohno T, et al. Aberrant DNA methylation of p57(KIP2) gene in the promoter region in lymphoid malignancies of B-cell phenotype. Blood. 2002;100:2572–2577. doi: 10.1182/blood-2001-11-0026. [DOI] [PubMed] [Google Scholar]
- 32.Chen CY, Tsay W, Tang JL, et al. SOCS1 methylation in patients with newly diagnosed acute myeloid leukemia. Genes Chromosomes Cancer. 2003;37:300–305. doi: 10.1002/gcc.10222. [DOI] [PubMed] [Google Scholar]
- 33.Tasaka T, Asou H, Munker R, et al. Methylation of the p16INK4A gene in multiple myeloma. Br J Haematol. 1998;101:558–564. doi: 10.1046/j.1365-2141.1998.00724.x. [DOI] [PubMed] [Google Scholar]
- 34.Voso MT, Scardocci A, Guidi F, et al. Aberrant methylation of DAP-kinase in therapy-related acute myeloid leukemia and myelodysplastic syndromes. Blood. 2004;103:698–700. doi: 10.1182/blood-2003-07-2249. [DOI] [PubMed] [Google Scholar]
- 35.Heuck CJ, Mehta J, Bhagat T, et al. Myeloma is characterized by stage-specific alterations in DNA methylation that occur early during myelomagenesis. J Immunol. 2013;190:2966–2975. doi: 10.4049/jimmunol.1202493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Wang X, Zhu YB, Cui HP, Yu TT. Aberrant promoter methylation of p15 and p16 genes may contribute to the pathogenesis of multiple myeloma: a meta-analysis. Tumour Biol. 2014 Jun 8; doi: 10.1007/s13277-014-2054-2. Epub. [DOI] [PubMed] [Google Scholar]