Abstract
The goal of endodontic treatment is the prevention and control of pulpal and periradicular infections. Calcium hydroxide (Ca(OH)2) has been widely used in endodontics as an intracanal medicament to eliminate the remaining microorganisms after chemomechanical preparation. The purpose of this article is to review the antimicrobial properties of Ca(OH)2 as an intracanal medicament in root canal treatment. The first part of this review details the characteristics of Ca(OH)2 and summarizes the results of in vitro studies related to its antimicrobial effect. The antimicrobial effect of Ca(OH)2 results from the release of hydroxyl ions when it comes into contact with aqueous fluids. Ca(OH)2 has a wide range of antimicrobial effects against common endodontic pathogens, but is less effective against Enterococcus faecalis and Candida albicans. The addition of vehicles or other agents might contribute to the antimicrobial effect of Ca(OH)2.
Keywords: Antimicrobial effect, Calcium hydroxide, Endodontics, Intracanal medicament, Microorganism
Introduction
Microorganisms are the cause of apical inflammatory lesions, and the goal of endodontic treatment is the prevention and control of pulpal and periradicular infections.1 Numerous measures have been introduced to reduce the number of microorganisms from the root canal system, including various mechanical instrumentation techniques, irrigation regimes, and intracanal medicaments.2,3,4,5,6 It is difficult to eliminate all microorganisms from an infected root canal system by mechanical instrumentation alone.7,8,9 Therefore, chemical irrigation and disinfection are necessary to remove microorganisms, their byproducts, pulp tissue remnants, and other debris from the root canal. Intracanal medicaments may perform these roles by remaining in the root canal between treatment appointments.10
Since Hermann introduced it in 1920 as a pulp-capping agent, calcium hydroxide (Ca(OH)2) has been widely used in endodontics.11 Various biological properties of Ca(OH)2, such as antimicrobial activity, tissue-dissolving ability, inhibition of tooth resorption, and hard tissue formation, have been investigated, and its wide use in root canal treatment has been associated with periradicular healing and few adverse reactions.3,12,13,14,15,16
Currently, Ca(OH)2 is considered the first choice of root canal dressing materials. The purpose of this article is to review the antimicrobial effect of Ca(OH)2 as an intracanal medicament in root canal treatment. A PubMed search was performed to identify laboratory and clinical studies that investigated the antimicrobial effect of Ca(OH)2 from 1970 to 2013 and was limited to English-language papers. Studies that included Ca(OH)2 as one of the comparative groups as well as the main subject were all reviewed. The articles were classified and analyzed according to their experimental methods. The first part of this review will detail the characteristics of Ca(OH)2 and summarize the results of in vitro studies related to its antimicrobial effect.
Review
Chemical characteristics of calcium hydroxide and mechanisms of antimicrobial effect
Calcium hydroxide is a white odorless powder with the formula Ca(OH)2. It has low solubility in water and releases calcium (Ca2+) and hydroxyl (OH-) ions slowly. The low solubility is a good clinical characteristic because a long period is necessary for Ca(OH)2 to become soluble in tissue fluids when in direct contact with vital tissues.17 Ca(OH)2 has a high pH (12.5 - 12.8) and is chemically classified as a strong base. It dissociates into calcium and hydroxyl ions on contact with an aqueous solution, and the main actions of Ca(OH)2 are attributed to the effect of these ions on vital tissues, such as inducing hard tissue deposition and being antibacterial.17 Hydroxyl ions are responsible for the highly alkaline nature of Ca(OH)2. Most of the pathogens are unable to survive in the highly alkaline environment provided by Ca(OH)2.18 Since the pH of Ca(OH)2 is about 12.5, bacteria in the infected root canal are eliminated when in direct contact with this substance.3
The antimicrobial activity of Ca(OH)2 is related to the release of hydroxyl ions in contact with aqueous fluids. Hydroxyl ions are highly oxidant free radicals that show extreme reactivity with biomolecules.19 The lethal effect on microorganisms has been attributed to the following mechanisms - damage to the bacterial cytoplasmic membrane, protein denaturation, and/or damage to the DNA - yet, it is difficult to establish the main mechanism involved in the death of bacteria.20,21,22,23 Kontakiotis et al. suggested that the ability of Ca(OH)2 to absorb carbon dioxide may contribute to its antibacterial activity.24
Laboratory studies
1. Antimicrobial susceptibility tests
A series of studies demonstrated the antimicrobial effect of Ca(OH)2 (Table 1). Several researchers investigated the effect of root canal medicaments by a direct exposure test, and they found that Ca(OH)2 was effective in killing bacteria.24,25,26,27,28,29,30 However, it should be noted that they did not evaluate the susceptibility of Enterococcus faecalis (E. faecalis). Leonardo et al. evaluated the antimicrobial activity of two Ca(OH)2 pastes against seven bacterial strains, and both of them were effective for all strains.31 Some studies have compared various Ca(OH)2 pastes.32,33 Mehrvarzfar et al. compared bioactive glass with Ca(OH)2 and found that both exhibited antimicrobial effects against E. faecalis and that Ca(OH)2 showed a superior disinfecting effect.34 Blanscet et al. found that the higher the concentration of the Ca(OH)2 paste was, the larger were the zones of inhibition observed.35
Table 1.
Studies reporting antimicrobial effect of Ca(OH)2
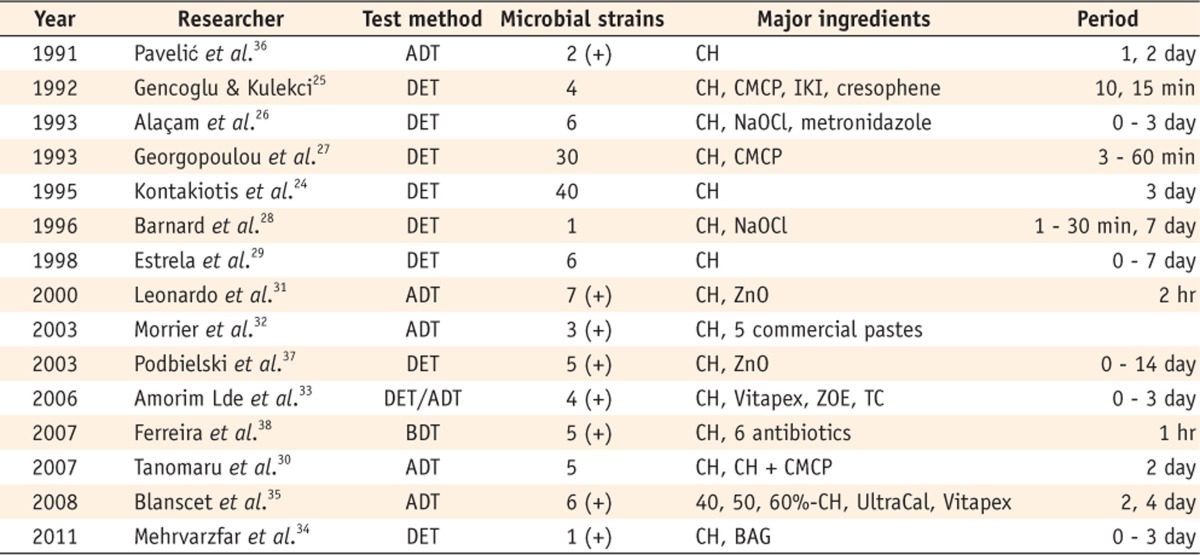
(+), E. faecalis was included as a subject of the experiment.
ADT, Agar diffusion test; DET, Direct exposure test; BDT, Broth dilution test, CH, Calcium hydroxide; CMCP, Camphorated paramonochlorophenol; IKI, Iodine potassium iodide; NaOCl, Sodium hypochlorite; ZnO, Zinc oxide; ZOE, Zinc oxide eugenol; TC, Tetracycline; BAG, Bioactive glass.
Vitapex, Neo-Dental Int., Federal Way, WA, USA; UltraCal, Ultradent Products Inc., South Jordan, UT, USA.
Some authors have insisted that E. faecalis is less susceptible to Ca(OH)2 than other bacteria. Pavelic et al. evaluated the antimicrobial effect of Ca(OH)2 by using an agar diffusion method.36 They found that Ca(OH)2 effectively inhibited the growth of all three microorganisms after 24 hours, but there was a difference in the sensitivity of each microorganism, such that Streptococcus mutans was the most sensitive and E. faecalis was the least. In the studies of Podbielski et al. and Ferreira et al., Ca(OH)2 was effective against all experimental strains and E. faecalis was the least susceptible.37,38
On the other hand, some articles have presented results that bring into question the antimicrobial effect of Ca(OH)2 (Table 2). Ohara et al. evaluated the antibacterial effect of several irrigants and found that Ca(OH)2 was totally ineffective as saline.39 Studies that compared the effect of various agents showed that Ca(OH)2 had only a weak effect.40,41,42,43 More recent studies that compared Ca(OH)2 with other agents reported that Ca(OH)2 was less effective than others.44,45,46 Adl et al. compared a triple antibiotic paste with Ca(OH)2 and found that Ca(OH)2 exhibited the smallest inhibition zones against E. faecalis.47
Table 2.
Studies reporting Ca(OH)2 to be ineffective
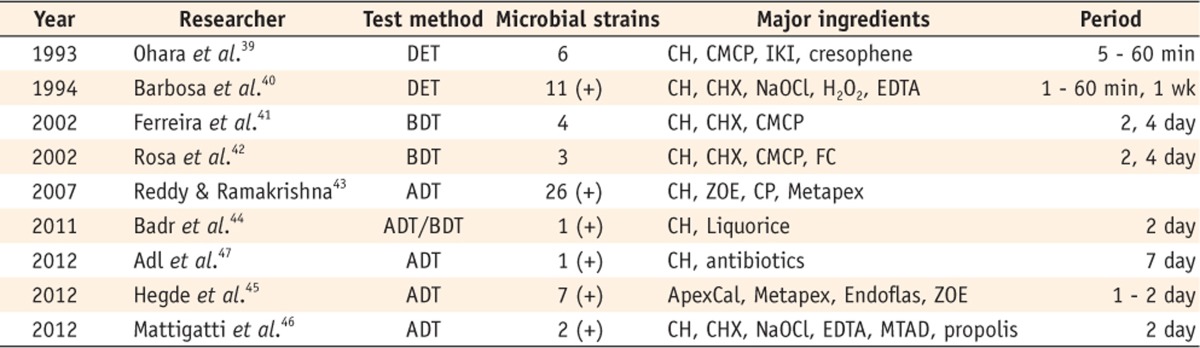
(+), E. faecalis was included as a subject of the experiment.
DET, Direct exposure test; BDT, Broth dilution test; ADT, Agar diffusion test, CH, Calcium hydroxide; CMCP, Camphorated paramonochlorophenol; IKI, Iodine potassium iodide; CHX, Chlorhexidine; NaOCl, sodium hypochlorite; H2O2, Hydrogen peroxide; EDTA, Ethylenediaminetetraacetic acid; FC, Formocresol; ZOE, Zinc oxide euginol; CP, Camphorated phenol.
Metapex, Meta Biomed Co., Ltd., Cheongju, Korea; ApexCal, Ivoclar Vivadent, Schaan, Liechtenstein; Endoflas, Sanlor Laboratory, Miami, FL, USA; MTAD, Dentsply Tulsa Dental Specialties, Tulsa, OK, USA.
Several researchers have assessed the influence of vehicles or agents mixed with Ca(OH)2 (Table 3). Estrela et al. examined the antimicrobial efficacy of Ca(OH)2 with certain vehicles and concluded that the vehicles did not influence the antimicrobial activity of Ca(OH)2.48,49 However, different results were observed by Gomes et al.50,51 They reported that Ca(OH)2 mixed with water or glycerin showed little or no effect, whereas Ca(OH)2 mixed with camphorated paramonochlorophenol (CMCP) was significantly more effective. The pastes with oily vehicles showed larger zones of inhibition than those with aqueous or viscous vehicles. In Siqueira and Uzeda's study, Ca(OH)2 mixed with distilled water or glycerin was ineffective against all bacterial strains even after 7 days of incubation.52 However, in another study using a broth dilution test, Ca(OH)2 with saline or glycerin showed antibacterial activity after 1 - 3 days.53 Vianna et al. stated that all tested Ca(OH)2 pastes were able to kill bacteria, but the paste prepared with CMCP was more effective in eliminating E. faecalis and Candida albicans (C. albicans).54 This coincided with the result reported by Gangwar.55 Turk et al. and Pacios et al. reported that E. faecalis and C. albicans were not inhibited by Ca(OH)2 mixed with distilled water.56,57
Table 3.
Studies on the effect of Ca(OH)2 mixed with vehicles or other agents
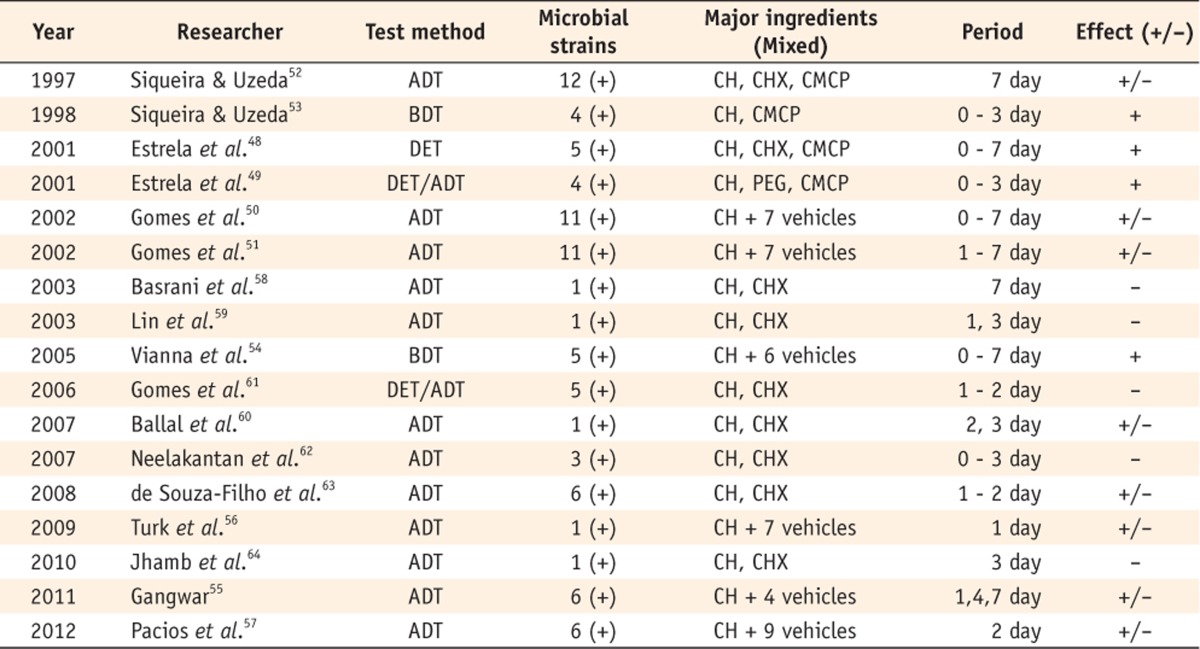
(+), E. faecalis was included as a subject of the experiment; +/-, The result showed a limited effect.
ADT, Agar diffusion test; BDT, Broth dilution test; DET, Direct exposure test, CH, Calcium hydroxide; CHX, Chlorhexidine; CMCP, Camphorated paramonochlorophenol; PEG, Polyethylene glycol.
Some have studied Ca(OH)2, chlorhexidine (CHX), and their mixtures. Basrani et al., Lin et al. and Ballal et al. found that the CHX gel was more effective than the Ca(OH)2 paste against E. faecalis and C. albicans.58,59,60 Several other authors reported that the antimicrobial effect of Ca(OH)2 alone was significantly lower than that of Ca(OH)2 mixed with CHX.61,62,63,64
Some studies have reported other factors that could affect the antimicrobial effect of Ca(OH)2. Portenier et al. found that the susceptibility of E. faecalis differed with its various phases such as the starvation, stationary, or exponential growth phases.65 Oliveira et al. reported that the existence of serum or necrotic tissue slowed down the antimicrobial activity of intracanal medicaments.66 Irrespective of the results, we have to take care in using phenolic compounds such as CMCP, because they may have toxic and/or antigenic effects.67
2. Infected dentin models - bovine teeth
In 1987, Haapasalo and Orstavik introduced an in vitro model for a dentinal tubule infection of the root canal.68 Cylindrical dentin specimens made from extracted bovine incisors were infected with E. faecalis, and medicaments were applied to the lumen of the dentin blocks. After a certain period, bacterial samples were taken with sterile round burs, and the dentin chips obtained with each bur were immediately collected in separate test tubes. These tubes were then incubated and inspected. In this study, liquid CMCP rapidly and completely disinfected the dentinal tubules, whereas Ca(OH)2 failed to eliminate them even superficially. Since then, a number of experiments have been performed using this method (Table 4). This model enabled an evaluation of the infection status at different depths of the dentinal tubules. Even though some researchers have modified the details of the model, the main objective of these studies, to evaluate the effect of antimicrobial agents in the root canal system with its own structures and components, was the same.
Table 4.
Studies on the antimicrobial effect of Ca(OH)2 using infected dentin models with bovine teeth
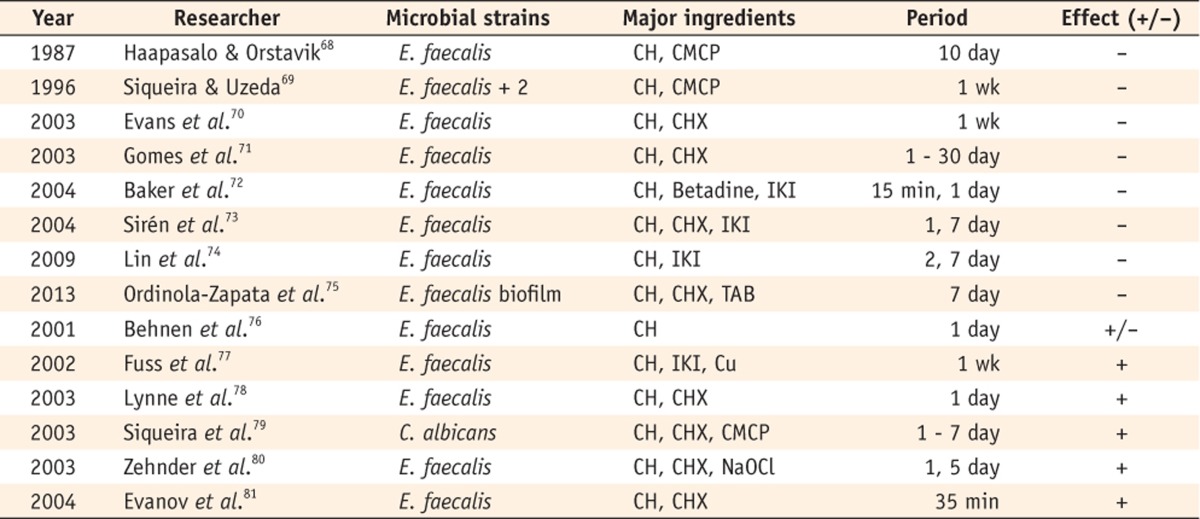
+/-, The result showed limited effect.
CH, Calcium hydroxide; CMCP, Camphorated paramonochlorophenol; CHX, Chlorhexidine; IKI, Iodine potassium iodide; TAB, Triple antibiotic paste; Cu, Copper; NaOCl, Sodium hypochlorite.
The results have been controversial. Siqueira & Uzeda reported that the Ca(OH)2/saline paste was ineffective against E. faecalis and Fusobacterium nucleatum (F. nucleatum) even after 1 week of exposure.69 The results of other experiments supported that Ca(OH)2 had little or no antimicrobial effect.70,71,72,73,74 A recent study used an infected dentin biofilm model that showed only a minimal inhibition of bacterial growth by Ca(OH)2 for 1 week.75 Behnen et al. found that a thin preparation of Ca(OH)2 was more effective than thick preparations.76 Fuss et al. reported that Ca(OH)2 significantly reduced bacterial viability in dentinal tubules up to a depth of 300 µm.77 Lynne et al. found that a 10% Ca(OH)2 paste effectively eliminated E. faecalis (89 - 94%).78 Siqueira et al. and Zehnder et al. reported that Ca(OH)2 eliminated C. albicans and E. faecalis after 7 days.79,80
Evanov et al. investigated whether the antimicrobial action of Ca(OH)2 or CHX was enhanced by heat.81 Both agents at 46℃ produced significantly less growth than either group at 37℃.
To be effective against the bacteria located inside the dentinal tubules, hydroxyl ions from Ca(OH)2 must diffuse through the dentin and reach sufficient levels to be lethal. Dentin has the buffering property of alkaline substances because of the proton donors in the hydrated layer of the hydroxyapatite.82 It has been demonstrated that Ca(OH)2 alkalinizes dentin, but the pH values may be insufficient to kill some bacterial strains, particularly E. faecalis, which can survive at a pH value of 11.5.3 Some authors supposed that excessively large numbers of bacteria might be used, which may have mechanically blocked the dentinal tubules, preventing Ca(OH)2 from entering.
3. Infected dentin models - human teeth
Considerably more studies have been performed with extracted human teeth than with bovine teeth.
Safavi et al. used the roots of extracted human teeth.83 The specimens were infected with Streptococcus faecium and exposed to Ca(OH)2 or iodine potassium iodide (IKI) for a duration ranging from 1 minute to 24 hours and the viability of the microorganisms was determined by the incubation of the entire roots in a culture medium. IKI disinfected dentin effectively, but bacteria remained viable in the dentin after extended periods of Ca(OH)2 treatment. This study was followed by several researchers who supported the ineffectiveness of Ca(OH)2 as an intracanal medicament.84,85,86 Some authors have reported that Ca(OH)2 showed almost no antimicrobial effect at all (Table 5).58,87,88,89,90
Table 5.
Studies reporting Ca(OH)2 to be ineffective using infected dentin models with human teeth
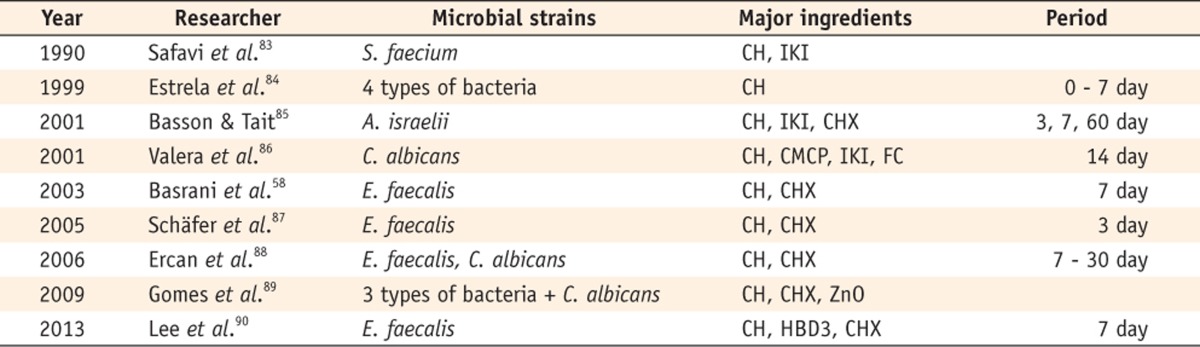
CH, Calcium hydroxide; IKI, Iodine potassium iodide; CHX, Chlorhexidine; CMCP, Camphorated paramonochlorophenol; FC, Formocresol; ZnO, Zinc oxide; HBD3, Human β-defensin 3.
Nevertheless, the opposite results have also been exhibited (Table 6). The study of Stuart et al. that compared the antimicrobial effectiveness of three medicaments in the root canals of extracted human teeth showed a 99.9% reduction against four species of bacteria in the teeth treated with Ca(OH)2.91 Han et al. found that Ca(OH)2 with an aqueous vehicle could successfully reduce the bacteria in the dentinal tubules.92 The results of other experiments showed that the bacteria were almost eliminated by Ca(OH)2.93,94,95,96,97,98 Noetzel et al. and Valera et al. found that Ca(OH)2 effectively reduced microorganisms that remained even after cleaning and shaping procedures.99,100
Table 6.
Studies reporting antimicrobial effect of Ca(OH)2 using infected dentin models with human teeth
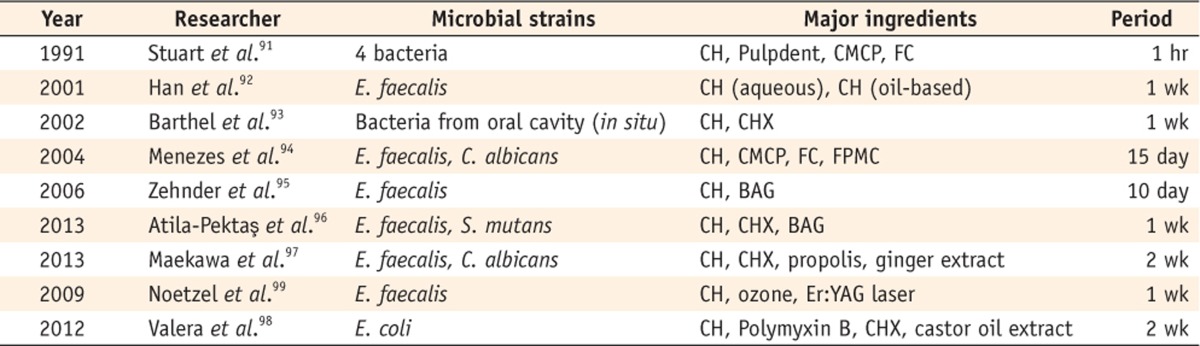
CH, Calcium hydroxide; CMCP, Camphorated paramonochlorophenol; FC, Formocresol; CHX, Chlorhexidine; FPMC, Furacin paramonochlorophenol; BAG, Bioactive glass.
Pulpdent, Pulpdent Corp., Watertown, MA, USA.
Lastly, there were authors who reported the limited antimicrobial effect of Ca(OH)2 that could partially reduce the number of bacteria (Table 7). Tanriverdi et al. found that the effectiveness of Ca(OH)2 was inferior to that of the other agents.101 However, a significant reduction in the bacterial number was observed. Similar results were exhibited in other studies.102,103,104,105 Lana et al. said that a Ca(OH)2 paste induced a 70% elimination of E. faecalis for 14 days.106 Delgado et al., Harrison et al., Kandaswamy et al., and Madhubala et al. reported 75%, 83%, 55%, and 59% of bacterial reduction, respectively.107,108,109,110 Almyroudi et al., Oncag et al. and Pavaskar et al. said that the antimicrobial efficacy of Ca(OH)2 might decrease over time.111,112,113
Table 7.
Studies reporting limited effect of Ca(OH)2 using infected dentin models with human teeth
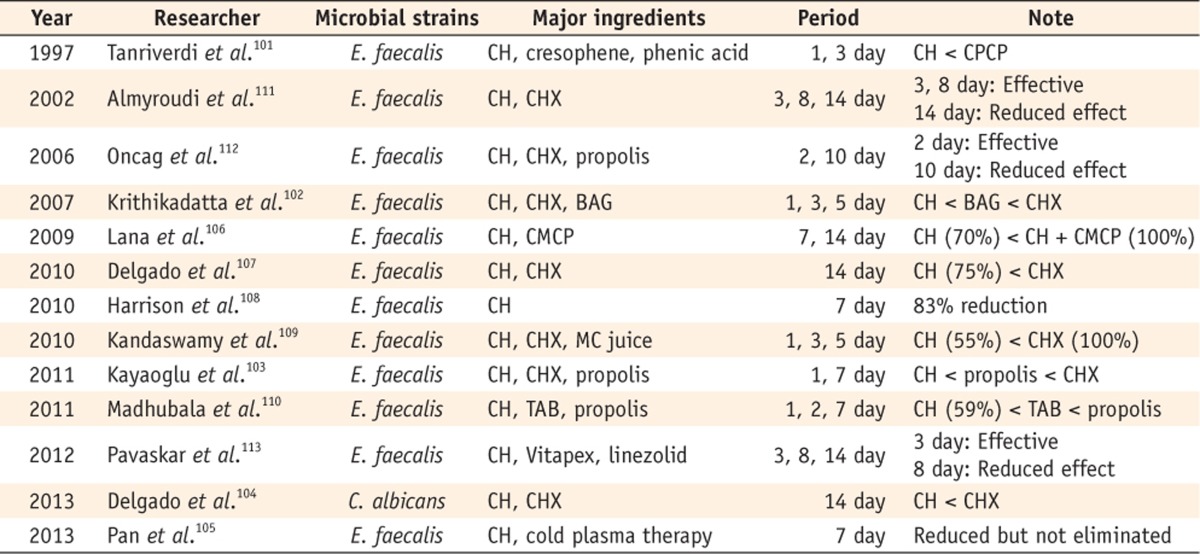
CH, Calcium hydroxide; CPCP, Camphorated parachlorophenol; CHX, Chlorhexidine; BAG, Bioactive glass; CMCP, Camphorated paramonochlorophenol; MC juice, Morinda citrifolia juice; TAB, Triple antibiotic paste.
Vitapex, Neo-Dental Int., Federal Way, WA, USA.
Some have compared the effect of Ca(OH)2 with different vehicles (Table 8). Sukawat and Srisuwan found that Ca(OH)2 mixed with CMCP killed all of the E. faecalis inside the tubules, whereas Ca(OH)2 mixed with distilled water or CHX was ineffective against these bacteria.114 Cwikla et al. said that there were significant differences in the disinfecting ability among the three Ca(OH)2 formulations.115 Lima et al. reported that all Ca(OH)2-based medicaments used in the study were able to significantly reduce the colony-forming units (CFU) of E. faecalis in the extracted teeth.116 Prabhakar et al. said that a combination of IKI or CHX with Ca(OH)2 may be beneficial.117
Table 8.
Studies on the effect of Ca(OH)2 mixed with vehicles or other agents using infected dentin models with human teeth
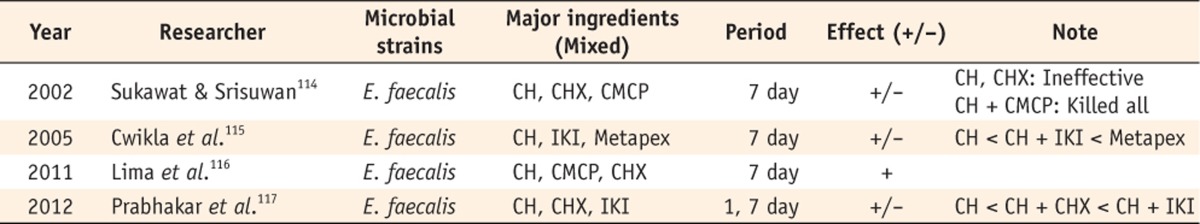
+/-, The result showed that Ca(OH)2 alone was less effective than when mixed with others.
CH, Calcium hydroxide; CHX, Chlorhexidine; CMCP, Camphorated paramonochlorophenol; IKI, Iodine potassium iodide.
Metapex, Meta Biomed Co., Ltd., Cheongju, Korea.
One study applied molecular techniques to the infected dentin model. Cook et al. evaluated the effect of Ca(OH)2 or CHX prior to root canal obturation on the survival of E. faecalis by using culture and polymerase chain reaction (PCR) techniques.118 A significant finding was exhibited such that no bacterial growth was seen on any of the cultures. However, PCR results showed a positive result on most of the experimental groups. These molecular techniques have been actively used for in vivo studies, which we shall discuss in the next part of this article.
Conclusions
Studies on the antimicrobial effect of Ca(OH)2 have differed with regard to methodology, inoculum size and age, culture medium, and bacterial strains used. Furthermore, experimental conditions completely equivalent to the root canal environment could not be ensured. Therefore, the studies showed varied, even conflicting, results. Although some studies have supported the antimicrobial effect of Ca(OH)2, others have questioned its efficacy.
In summary of the first part of this review, the antimicrobial effect of Ca(OH)2 is related to the hydroxyl ions released in an aqueous environment, which affects cytoplasmic membranes, proteins, and the DNA of microorganisms. Ca(OH)2 has a wide range of antimicrobial effects against common endodontic pathogens, but it is less effective against specific species such as E. faecalis or C. albicans. The addition of vehicles or other agents might contribute to the antimicrobial effect of Ca(OH)2. Although it remains controversial, it seems that by mixing Ca(OH)2 with CHX, the antimicrobial activity of Ca(OH)2 can be increased.
Footnotes
No potential conflict of interest relevant to this article was reported.
References
- 1.Kakehashi S, Stanley HR, Fitzgerald RJ. The effects of surgical exposures of dental pulps in germ-free and conventional laboratory rats. Oral Surg Oral Med Oral Pathol. 1965;20:340–349. doi: 10.1016/0030-4220(65)90166-0. [DOI] [PubMed] [Google Scholar]
- 2.Byström A, Sundqvist G. Bacteriologic evaluation of the effect of 0.5 percent sodium hypochlorite in endodontic therapy. Oral Surg Oral Med Oral Pathol. 1983;55:307–312. doi: 10.1016/0030-4220(83)90333-x. [DOI] [PubMed] [Google Scholar]
- 3.Byström A, Claesson R, Sundqvist G. The antibacterial effect of camphorated paramonochlorophenol, camphorated phenol and calcium hydroxide in the treatment of infected root canals. Endod Dent Traumatol. 1985;1:170–175. doi: 10.1111/j.1600-9657.1985.tb00652.x. [DOI] [PubMed] [Google Scholar]
- 4.Byström A, Sundqvist G. The antibacterial action of sodium hypochlorite and EDTA in 60 cases of endodontic therapy. Int Endod J. 1985;18:35–40. doi: 10.1111/j.1365-2591.1985.tb00416.x. [DOI] [PubMed] [Google Scholar]
- 5.Sjögren U, Sundqvist G. Bacteriologic evaluation of ultrasonic root canal instrumentation. Oral Surg Oral Med Oral Pathol. 1987;63:366–370. doi: 10.1016/0030-4220(87)90208-8. [DOI] [PubMed] [Google Scholar]
- 6.Shuping GB, Orstavik D, Sigurdsson A, Trope M. Reduction of intracanal bacteria using nickel-titanium rotary instrumentation and various medications. J Endod. 2000;26:751–755. doi: 10.1097/00004770-200012000-00022. [DOI] [PubMed] [Google Scholar]
- 7.Byström A, Sundqvist G. Bacteriologic evaluation of the efficacy of mechanical root canal instrumentation in endodontic therapy. Scand J Dent Res. 1981;89:321–328. doi: 10.1111/j.1600-0722.1981.tb01689.x. [DOI] [PubMed] [Google Scholar]
- 8.Metzler RS, Montgomery S. Effectiveness of ultrasonics and calcium hydroxide for the debridement of human mandibular molars. J Endod. 1989;15:373–378. doi: 10.1016/s0099-2399(89)80076-7. [DOI] [PubMed] [Google Scholar]
- 9.Peters OA, Laib A, Göhring TN, Barbakow F. Changes in root canal geometry after preparation assessed by high-resolution computed tomography. J Endod. 2001;27:1–6. doi: 10.1097/00004770-200101000-00001. [DOI] [PubMed] [Google Scholar]
- 10.Walton RE. Intracanal medicaments. Dent Clin North Am. 1984;28:783–796. [PubMed] [Google Scholar]
- 11.Hermann BW. Calcium hydroxyd als Mittelzurn, Behandeln und Fullen von Wurzelkanalen [Thesis] Wurzburg: 1920. [Google Scholar]
- 12.Hasselgren G, Olsson B, Cvek M. Effects of calcium hydroxide and sodium hypochlorite on the dissolution of necrotic porcine muscle tissue. J Endod. 1988;14:125–127. doi: 10.1016/S0099-2399(88)80212-7. [DOI] [PubMed] [Google Scholar]
- 13.Tronstad L. Root resorption-etiology, terminology and clinical manifestations. Endod Dent Traumatol. 1988;4:241–252. doi: 10.1111/j.1600-9657.1988.tb00642.x. [DOI] [PubMed] [Google Scholar]
- 14.Foreman PC, Barnes IE. Review of calcium hydroxide. Int Endod J. 1990;23:283–297. doi: 10.1111/j.1365-2591.1990.tb00108.x. [DOI] [PubMed] [Google Scholar]
- 15.Sjogren U, Hagglund B, Sundqvist G, Wing K. Factors affecting the long-term results of endodontic treatment. J Endod. 1990;16:498–504. doi: 10.1016/S0099-2399(07)80180-4. [DOI] [PubMed] [Google Scholar]
- 16.De Moor RJ, De Witte AM. Periapical lesions accidentally filled with calcium hydroxide. Int Endod J. 2002;35:946–958. doi: 10.1046/j.1365-2591.2002.00597.x. [DOI] [PubMed] [Google Scholar]
- 17.Farhad A, Mohammadi Z. Calcium hydroxide: a review. Int Dent J. 2005;55:293–301. doi: 10.1111/j.1875-595x.2005.tb00326.x. [DOI] [PubMed] [Google Scholar]
- 18.Heithersay GS. Calcium hydroxide in the treatment of pulpless teeth with associated pathology. J Br Endod Soc. 1975;8:74–93. doi: 10.1111/j.1365-2591.1975.tb01000.x. [DOI] [PubMed] [Google Scholar]
- 19.Freeman BA, Crapo JD. Biology of disease: free radicals and tissue injury. Lab Invest. 1982;47:412–426. [PubMed] [Google Scholar]
- 20.Siqueira JF, Jr, Lopes HP. Mechanisms of antimicrobial activity of calcium hydroxide: a critical review. Int Endod J. 1999;32:361–369. doi: 10.1046/j.1365-2591.1999.00275.x. [DOI] [PubMed] [Google Scholar]
- 21.Halliwell B. Oxidants and human disease: some new concepts. FASEB J. 1987;1:358–364. [PubMed] [Google Scholar]
- 22.Estrela C, Holland R. Calcium hydroxide: study based on scientific evidences. J Appl Oral Sci. 2003;11:269–282. doi: 10.1590/s1678-77572003000400002. [DOI] [PubMed] [Google Scholar]
- 23.Imlay JA, Linn S. DNA damage and oxygen radical toxicity. Science. 1988;240:1302–1309. doi: 10.1126/science.3287616. [DOI] [PubMed] [Google Scholar]
- 24.Kontakiotis E, Nakou M, Georgopoulou M. In vitro study of the indirect action of calcium hydroxide on the anaerobic flora of the root canal. Int Endod J. 1995;28:285–289. doi: 10.1111/j.1365-2591.1995.tb00317.x. [DOI] [PubMed] [Google Scholar]
- 25.Gencoglu N, Külekçi G. Antibacterial efficacy of root canal medicaments. J Nihon Univ Sch Dent. 1992;34:233–236. doi: 10.2334/josnusd1959.34.233. [DOI] [PubMed] [Google Scholar]
- 26.Alaçam T, Omürlü H, Ozkul A, Görgül G, Misirligil A. Cytotoxicity versus antibacterial activity of some antiseptics in vitro. J Nihon Univ Sch Dent. 1993;35:22–27. doi: 10.2334/josnusd1959.35.22. [DOI] [PubMed] [Google Scholar]
- 27.Georgopoulou M, Kontakiotis E, Nakou M. In vitro evaluation of the effectiveness of calcium hydroxide and paramonochlorophenol on anaerobic bacteria from the root canal. Endod Dent Traumatol. 1993;9:249–253. doi: 10.1111/j.1600-9657.1993.tb00281.x. [DOI] [PubMed] [Google Scholar]
- 28.Barnard D, Davies J, Figdor D. Susceptibility of Actinomyces israelii to antibiotics, sodium hypochlorite and calcium hydroxide. Int Endod J. 1996;29:320–326. doi: 10.1111/j.1365-2591.1996.tb01392.x. [DOI] [PubMed] [Google Scholar]
- 29.Estrela C, Pimenta FC, Ito IY, Bammann LL. In vitro determination of direct antimicrobial effect of calcium hydroxide. J Endod. 1998;24:15–17. doi: 10.1016/S0099-2399(98)80205-7. [DOI] [PubMed] [Google Scholar]
- 30.Tanomaru JM, Pappen FG, Tanomaru Filho M, Spolidorio DM, Ito IY. In vitro antimicrobial activity of different gutta-percha points and calcium hydroxide pastes. Braz Oral Res. 2007;21:35–39. doi: 10.1590/s1806-83242007000100006. [DOI] [PubMed] [Google Scholar]
- 31.Leonardo MR, da Silva LA, Tanomaru Filho M, Bonifácio KC, Ito IY. In vitro evaluation of antimicrobial activity of sealers and pastes used in endodontics. J Endod. 2000;26:391–394. doi: 10.1097/00004770-200007000-00003. [DOI] [PubMed] [Google Scholar]
- 32.Morrier JJ, Benay G, Hartmann C, Barsotti O. Antimicrobial activity of Ca(OH)2 dental cements: an in vitro study. J Endod. 2003;29:51–54. doi: 10.1097/00004770-200301000-00014. [DOI] [PubMed] [Google Scholar]
- 33.Amorim Lde F, Toledo OA, Estrela CR, Decurcio Dde A, Estrela C. Antimicrobial analysis of different root canal filling pastes used in pediatric dentistry by two experimental methods. Braz Dent J. 2006;17:317–322. doi: 10.1590/s0103-64402006000400010. [DOI] [PubMed] [Google Scholar]
- 34.Mehrvarzfar P, Akhavan H, Rastgarian H, Mohammadzade Akhlagi N, Soleymanpour R, Ahmadi A. An in vitro comparative study on the antimicrobial effects of bioglass 45S5 vs. calcium hydroxide on Enterococcus faecalis. Iran Endod J. 2011;6:29–33. [PMC free article] [PubMed] [Google Scholar]
- 35.Blanscet ML, Tordik PA, Goodell GG. An agar diffusion comparison of the antimicrobial effect of calcium hydroxide at five different concentrations with three different vehicles. J Endod. 2008;34:1246–1248. doi: 10.1016/j.joen.2008.07.012. [DOI] [PubMed] [Google Scholar]
- 36.Pavelić B, Anić I, Najzar-Fleger D, Stilinović B, Temmer K. The antimicrobial efficiency of aqueous solutions of calcium hydroxide on Streptococcus mutans, Streptococcus faecalis and Candida albicans, in vitro. Acta Stomatol Croat. 1991;25:207–212. [PubMed] [Google Scholar]
- 37.Podbielski A, Spahr A, Haller B. Additive antimicrobial activity of calcium hydroxide and chlorhexidine on common endodontic bacterial pathogens. J Endod. 2003;29:340–345. doi: 10.1097/00004770-200305000-00006. [DOI] [PubMed] [Google Scholar]
- 38.Ferreira FB, Torres SA, Rosa OP, Ferreira CM, Garcia RB, Marcucci MC, Gomes BP. Antimicrobial effect of propolis and other substances against selected endodontic pathogens. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2007;104:709–716. doi: 10.1016/j.tripleo.2007.05.019. [DOI] [PubMed] [Google Scholar]
- 39.Ohara P, Torabinejad M, Kettering JD. Antibacterial effects of various endodontic irrigants on selected anaerobic bacteria. Endod Dent Traumatol. 1993;9:95–100. doi: 10.1111/j.1600-9657.1993.tb00258.x. [DOI] [PubMed] [Google Scholar]
- 40.Barbosa SV, Spangberg LS, Almeida D. Low surface tension calcium hydroxide solution is an effective antiseptic. Int Endod J. 1994;27:6–10. doi: 10.1111/j.1365-2591.1994.tb00221.x. [DOI] [PubMed] [Google Scholar]
- 41.Ferreira CM, da Silva Rosa OP, Torres SA, Ferreira FB, Bernardinelli N. Activity of endodontic antibacterial agents against selected anaerobic bacteria. Braz Dent J. 2002;13:118–122. doi: 10.1590/s0103-64402002000200008. [DOI] [PubMed] [Google Scholar]
- 42.Rosa OP, Torres SA, Ferreira CM, Ferreira FB. In vitro effect of intracanal medicaments on strict anaerobes by means of the broth dilution method. Pesqui Odontol Bras. 2002;16:31–36. doi: 10.1590/s1517-74912002000100006. [DOI] [PubMed] [Google Scholar]
- 43.Reddy S, Ramakrishna Y. Evaluation of antimicrobial efficacy of various root canal filling materials used in primary teeth: a microbiological study. J Clin Pediatr Dent. 2007;31:193–198. doi: 10.17796/jcpd.31.3.t73r4061424j2578. [DOI] [PubMed] [Google Scholar]
- 44.Badr AE, Omar N, Badria FA. A laboratory evaluation of the antibacterial and cytotoxic effect of Liquorice when used as root canal medicament. Int Endod J. 2011;44:51–58. doi: 10.1111/j.1365-2591.2010.01794.x. [DOI] [PubMed] [Google Scholar]
- 45.Hegde S, Lala PK, Dinesh RB, Shubha AB. An in vitro evaluation of antimicrobial efficacy of primary root canal filling materials. J Clin Pediatr Dent. 2012;37:59–64. doi: 10.17796/jcpd.37.1.v6305588xw525505. [DOI] [PubMed] [Google Scholar]
- 46.Mattigatti S, Ratnakar P, Moturi S, Varma S, Rairam S. Antimicrobial effect of conventional root canal medicaments vs propolis against Enterococcus faecalis, Staphylococcus aureus and Candida albicans. J Contemp Dent Pract. 2012;13:305–309. doi: 10.5005/jp-journals-10024-1142. [DOI] [PubMed] [Google Scholar]
- 47.Adl A, Shojaee NS, Motamedifar M. A comparison between the antimicrobial effects of triple antibiotic paste and calcium hydroxide against Entrococcus Faecalis. Iran Endod J. 2012;7:149–155. [PMC free article] [PubMed] [Google Scholar]
- 48.Estrela C, Bammann LL, Pimenta FC, Pécora JD. Control of microorganisms in vitro by calcium hydroxide pastes. Int Endod J. 2001;34:341–345. doi: 10.1046/j.1365-2591.2001.00368.x. [DOI] [PubMed] [Google Scholar]
- 49.Estrela C, Rodrigues de Araújo Estrela C, Bammann LL, Pecora JD. Two methods to evaluate the antimicrobial action of calcium hydroxide paste. J Endod. 2001;27:720–723. doi: 10.1097/00004770-200112000-00002. [DOI] [PubMed] [Google Scholar]
- 50.Gomes BP, Ferraz CC, Garrido FD, Rosalen PL, Zaia AA, Teixeira FB, de Souza-Filho FJ. Microbial susceptibility to calcium hydroxide pastes and their vehicles. J Endod. 2002;28:758–761. doi: 10.1097/00004770-200211000-00003. [DOI] [PubMed] [Google Scholar]
- 51.Gomes BP, Ferraz CC, Vianna ME, Rosalen PL, Zaia AA, Teixeira FB, Souza-Filho FJ. In vitro antimicrobial activity of calcium hydroxide pastes and their vehicles against selected microorganisms. Braz Dent J. 2002;13:155–161. doi: 10.1590/s0103-64402002000300002. [DOI] [PubMed] [Google Scholar]
- 52.Siqueira JF, Jr, de Uzeda M. Intracanal medicaments: evaluation of the antibacterial effects of chlorhexidine, metronidazole, and calcium hydroxide associated with three vehicles. J Endod. 1997;23:167–169. doi: 10.1016/S0099-2399(97)80268-3. [DOI] [PubMed] [Google Scholar]
- 53.Siqueira JF, Jr, de Uzeda M. Influence of different vehicles on the antibacterial effects of calcium hydroxide. J Endod. 1998;24:663–665. doi: 10.1016/S0099-2399(98)80151-9. [DOI] [PubMed] [Google Scholar]
- 54.Vianna ME, Gomes BP, Sena NT, Zaia AA, Ferraz CC, de Souza Filho FJ. In vitro evaluation of the susceptibility of endodontic pathogens to calcium hydroxide combined with different vehicles. Braz Dent J. 2005;16:175–180. doi: 10.1590/s0103-64402005000300001. [DOI] [PubMed] [Google Scholar]
- 55.Gangwar A. Antimicrobial effectiveness of different preparations of calcium hydroxide. Indian J Dent Res. 2011;22:66–70. doi: 10.4103/0970-9290.79986. [DOI] [PubMed] [Google Scholar]
- 56.Turk BT, Sen BH, Ozturk T. In vitro antimicrobial activity of calcium hydroxide mixed with different vehicles against Enterococcus faecalis and Candida albicans. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2009;108:297–301. doi: 10.1016/j.tripleo.2009.03.029. [DOI] [PubMed] [Google Scholar]
- 57.Pacios MG, Silva C, López ME, Cecilia M. Antibacterial action of calcium hydroxide vehicles and calcium hydroxide pastes. J Investig Clin Dent. 2012;3:264–270. doi: 10.1111/j.2041-1626.2012.00147.x. [DOI] [PubMed] [Google Scholar]
- 58.Basrani B, Tjaderhane L, Santos JM, Pascon E, Grad H, Lawrence HP, Friedman S. Efficacy of chlorhexidine- and calcium hydroxide-containing medicaments against Enterococcus faecalis in vitro. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2003;96:618–624. doi: 10.1016/s1079-2104(03)00166-5. [DOI] [PubMed] [Google Scholar]
- 59.Lin YH, Mickel AK, Chogle S. Effectiveness of selected materials against Enterococcus faecalis: part 3. The antibacterial effect of calcium hydroxide and chlorhexidine on Enterococcus faecalis. J Endod. 2003;29:565–566. doi: 10.1097/00004770-200309000-00006. [DOI] [PubMed] [Google Scholar]
- 60.Ballal V, Kundabala M, Acharya S, Ballal M. Antimicrobial action of calcium hydroxide, chlorhexidine and their combination on endodontic pathogens. Aust Dent J. 2007;52:118–121. doi: 10.1111/j.1834-7819.2007.tb00475.x. [DOI] [PubMed] [Google Scholar]
- 61.Gomes BP, Vianna ME, Sena NT, Zaia AA, Ferraz CC, de Souza Filho FJ. In vitro evaluation of the antimicrobial activity of calcium hydroxide combined with chlorhexidine gel used as intracanal medicament. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2006;102:544–550. doi: 10.1016/j.tripleo.2006.04.010. [DOI] [PubMed] [Google Scholar]
- 62.Neelakantan P, Sanjeev K, Subbarao CV. Duration-dependent susceptibility of endodontic pathogens to calcium hydroxide and chlorhexidene gel used as intracanal medicament: an in vitro evaluation. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2007;104:e138–e141. doi: 10.1016/j.tripleo.2007.04.035. [DOI] [PubMed] [Google Scholar]
- 63.de Souza-Filho FJ, Soares Ade J, Vianna ME, Zaia AA, Ferraz CC, Gomes BP. Antimicrobial effect and pH of chlorhexidine gel and calcium hydroxide alone and associated with other materials. Braz Dent J. 2008;19:28–33. doi: 10.1590/s0103-64402008000100005. [DOI] [PubMed] [Google Scholar]
- 64.Jhamb S, Nikhil V, Singh V. An in vitro study of antibacterial effect of calcium hydroxide and chlorhexidine on Enterococcus faecalis. Indian J Dent Res. 2010;21:512–514. doi: 10.4103/0970-9290.74222. [DOI] [PubMed] [Google Scholar]
- 65.Portenier I, Waltimo T, Ørstavik D, Haapasalo M. The susceptibility of starved, stationary phase, and growing cells of Enterococcus faecalis to endodontic medicaments. J Endod. 2005;31:380–386. doi: 10.1097/01.don.0000145421.84121.c8. [DOI] [PubMed] [Google Scholar]
- 66.Oliveira JC, Alves FR, Uzeda M, Rôças IN, Siqueira JF., Jr Influence of serum and necrotic soft tissue on the antimicrobial effects of intracanal medicaments. Braz Dent J. 2010;21:295–300. doi: 10.1590/s0103-64402010000400001. [DOI] [PubMed] [Google Scholar]
- 67.Ribeiro DA. Do endodontic compounds induce genetic damage? A comprehensive review. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2008;105:251–256. doi: 10.1016/j.tripleo.2007.07.045. [DOI] [PubMed] [Google Scholar]
- 68.Haapasalo M, Orstavik D. In vitro infection and disinfection of dentinal tubules. J Dent Res. 1987;66:1375–1379. doi: 10.1177/00220345870660081801. [DOI] [PubMed] [Google Scholar]
- 69.Siqueira JF, Jr, de Uzeda M. Disinfection by calcium hydroxide pastes of dentinal tubules infected with two obligate and one facultative anaerobic bacteria. J Endod. 1996;22:674–676. doi: 10.1016/S0099-2399(96)80062-8. [DOI] [PubMed] [Google Scholar]
- 70.Evans MD, Baumgartner JC, Khemaleelakul SU, Xia T. Efficacy of calcium hydroxide: chlorhexidine paste as an intracanal medication in bovine dentin. J Endod. 2003;29:338–339. doi: 10.1097/00004770-200305000-00005. [DOI] [PubMed] [Google Scholar]
- 71.Gomes BP, Souza SF, Ferraz CC, Teixeira FB, Zaia AA, Valdrighi L, Souza-Filho FJ. Effectiveness of 2% chlorhexidine gel and calcium hydroxide against Enterococcus faecalis in bovine root dentine in vitro. Int Endod J. 2003;36:267–275. doi: 10.1046/j.1365-2591.2003.00634.x. [DOI] [PubMed] [Google Scholar]
- 72.Baker NE, Liewehr FR, Buxton TB, Joyce AP. Antibacterial efficacy of calcium hydroxide, iodine potassium iodide, betadine, and betadine scrub with and without surfactant against E faecalis in vitro. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2004;98:359–364. doi: 10.1016/j.tripleo.2004.04.023. [DOI] [PubMed] [Google Scholar]
- 73.Sirén EK, Haapasalo MP, Waltimo TM, Ørstavik D. In vitro antibacterial effect of calcium hydroxide combined with chlorhexidine or iodine potassium iodide on Enterococcus faecalis. Eur J Oral Sci. 2004;112:326–331. doi: 10.1111/j.1600-0722.2004.00144.x. [DOI] [PubMed] [Google Scholar]
- 74.Lin S, Kfir A, Laviv A, Sela G, Fuss Z. The in vitro antibacterial effect of iodine-potassium iodide and calcium hydroxide in infected dentinal tubules at different time intervals. J Contemp Dent Pract. 2009;10:59–66. [PubMed] [Google Scholar]
- 75.Ordinola-Zapata R, Bramante CM, Minotti PG, Cavenago BC, Garcia RB, Bernardineli N, Jaramillo DE, Hungaro Duarte MA. Antimicrobial activity of triantibiotic paste, 2% chlorhexidine gel, and calcium hydroxide on an intraoral-infected dentin biofilm model. J Endod. 2013;39:115–118. doi: 10.1016/j.joen.2012.10.004. [DOI] [PubMed] [Google Scholar]
- 76.Behnen MJ, West LA, Liewehr FR, Buxton TB, McPherson JC., 3rd Antimicrobial activity of several calcium hydroxide preparations in root canal dentin. J Endod. 2001;27:765–767. doi: 10.1097/00004770-200112000-00013. [DOI] [PubMed] [Google Scholar]
- 77.Fuss Z, Mizrahi A, Lin S, Cherniak O, Weiss EI. A laboratory study of the effect of calcium hydroxide mixed with iodine or electrophoretically activated copper on bacterial viability in dentinal tubules. Int Endod J. 2002;35:522–526. doi: 10.1046/j.1365-2591.2002.00517.x. [DOI] [PubMed] [Google Scholar]
- 78.Lynne RE, Liewehr FR, West LA, Patton WR, Buxton TB, McPherson JC. In vitro antimicrobial activity of various medication preparations on E. faecalis in root canal dentin. J Endod. 2003;29:187–190. doi: 10.1097/00004770-200303000-00006. [DOI] [PubMed] [Google Scholar]
- 79.Siqueira JF, Jr, Rôças IN, Lopes HP, Magalhães FA, de Uzeda M. Elimination of Candida albicans infection of the radicular dentin by intracanal medications. J Endod. 2003;29:501–504. doi: 10.1097/00004770-200308000-00003. [DOI] [PubMed] [Google Scholar]
- 80.Zehnder M, Grawehr M, Hasselgren G, Waltimo T. Tissue-dissolution capacity and dentin-disinfecting potential of calcium hydroxide mixed with irrigating solutions. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2003;96:608–613. doi: 10.1016/s1079-2104(03)00157-4. [DOI] [PubMed] [Google Scholar]
- 81.Evanov C, Liewehr F, Buxton TB, Joyce AP. Antibacterial efficacy of calcium hydroxide and chlorhexidine gluconate irrigants at 37 degrees C and 46 degrees C. J Endod. 2004;30:653–657. doi: 10.1097/01.don.0000121620.11272.22. [DOI] [PubMed] [Google Scholar]
- 82.Nerwich A, Figdor D, Messer HH. pH changes in root dentin over a 4-week period following root canal dressing with calcium hydroxide. J Endod. 1993;19:302–306. doi: 10.1016/s0099-2399(06)80461-9. [DOI] [PubMed] [Google Scholar]
- 83.Safavi KE, Spangberg LS, Langeland K. Root canal dentinal tubule disinfection. J Endod. 1990;16:207–210. doi: 10.1016/s0099-2399(06)81670-5. [DOI] [PubMed] [Google Scholar]
- 84.Estrela C, Pimenta FC, Ito IY, Bammann LL. Antimicrobial evaluation of calcium hydroxide in infected dentinal tubules. J Endod. 1999;25:416–418. doi: 10.1016/S0099-2399(99)80269-6. [DOI] [PubMed] [Google Scholar]
- 85.Basson NJ, Tait CM. Effectiveness of three root canal medicaments to eliminate Actinomyces israelii from infected dentinal tubules in vitro. SADJ. 2001;56:499–501. [PubMed] [Google Scholar]
- 86.Valera MC, de Moraes Rego J, Jorge AO. Effect of sodium hypochlorite and five intracanal medications on Candida albicans in root canals. J Endod. 2001;27:401–403. doi: 10.1097/00004770-200106000-00008. [DOI] [PubMed] [Google Scholar]
- 87.Schäfer E, Bössmann K. Antimicrobial efficacy of chlorhexidine and two calcium hydroxide formulations against Enterococcus faecalis. J Endod. 2005;31:53–56. doi: 10.1097/01.don.0000134209.28874.1c. [DOI] [PubMed] [Google Scholar]
- 88.Ercan E, Dalli M, Dülgergil CT. In vitro assessment of the effectiveness of chlorhexidine gel and calcium hydroxide paste with chlorhexidine against Enterococcus faecalis and Candida albicans. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2006;102:e27–e31. doi: 10.1016/j.tripleo.2006.02.022. [DOI] [PubMed] [Google Scholar]
- 89.Gomes BP, Montagner F, Berber VB, Zaia AA, Ferraz CC, de Almeida JF, Souza-Filho FJ. Antimicrobial action of intracanal medicaments on the external root surface. J Dent. 2009;37:76–81. doi: 10.1016/j.jdent.2008.09.009. [DOI] [PubMed] [Google Scholar]
- 90.Lee JK, Park YJ, Kum KY, Han SH, Chang SW, Kaufman B, Jiang J, Zhu Q, Safavi K, Spångberg L. Antimicrobial efficacy of a human β-defensin-3 peptide using an Enterococcus faecalis dentine infection model. Int Endod J. 2013;46:406–412. doi: 10.1111/iej.12002. [DOI] [PubMed] [Google Scholar]
- 91.Stuart KG, Miller CH, Brown CE, Jr, Newton CW. The comparative antimicrobial effect of calcium hydroxide. Oral Surg Oral Med Oral Pathol. 1991;72:101–104. doi: 10.1016/0030-4220(91)90198-l. [DOI] [PubMed] [Google Scholar]
- 92.Han GY, Park SH, Yoon TC. Antimicrobial activity of Ca(OH)2 containing pastes with Enterococcus faecalis in vitro. J Endod. 2001;27:328–332. doi: 10.1097/00004770-200105000-00004. [DOI] [PubMed] [Google Scholar]
- 93.Barthel CR, Zimmer S, Zilliges S, Schiller R, Göbel UB, Roulet JF. In situ antimicrobial effectiveness of chlorhexidine and calcium hydroxide: gel and paste versus gutta-percha points. J Endod. 2002;28:427–430. doi: 10.1097/00004770-200206000-00002. [DOI] [PubMed] [Google Scholar]
- 94.Menezes MM, Valera MC, Jorge AO, Koga-Ito CY, Camargo CH, Mancini MN. In vitro evaluation of the effectiveness of irrigants and intracanal medicaments on microorganisms within root canals. Int Endod J. 2004;37:311–319. doi: 10.1111/j.0143-2885.2004.00799.x. [DOI] [PubMed] [Google Scholar]
- 95.Zehnder M, Luder HU, Schätzle M, Kerosuo E, Waltimo T. A comparative study on the disinfection potentials of bioactive glass S53P4 and calcium hydroxide in contralateral human premolars ex vivo. Int Endod J. 2006;39:952–958. doi: 10.1111/j.1365-2591.2006.01173.x. [DOI] [PubMed] [Google Scholar]
- 96.Atila-Pektaş B, Yurdakul P, Gülmez D, Görduysus O. Antimicrobial effects of root canal medicaments against Enterococcus faecalis and Streptococcus mutans. Int Endod J. 2013;46:413–418. doi: 10.1111/iej.12004. [DOI] [PubMed] [Google Scholar]
- 97.Maekawa LE, Valera MC, Oliveira LD, Carvalho CA, Camargo CH, Jorge AO. Effect of Zingiber officinale and propolis on microorganisms and endotoxins in root canals. J Appl Oral Sci. 2013;21:25–31. doi: 10.1590/1678-7757201302129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Valera MC, Maekawa LE, Chung A, de Oliveira LD, Carvalho CA, Koga-Ito CY, Jorge AO. Effectiveness of castor oil extract on Escherichia coli and its endotoxins in root canals. Gen Dent. 2012;60:e204–e209. [PubMed] [Google Scholar]
- 99.Noetzel J, Nonhoff J, Bitter K, Wagner J, Neumann K, Kielbassa AM. Efficacy of calcium hydroxide, Er:YAG laser or gaseous ozone against Enterococcus faecalis in root canals. Am J Dent. 2009;22:14–18. [PubMed] [Google Scholar]
- 100.Valera MC, Silva KC, Maekawa LE, Carvalho CA, Koga-Ito CY, Camargo CH, Lima RS. Antimicrobial activity of sodium hypochlorite associated with intracanal medication for Candida albicans and Enterococcus faecalis inoculated in root canals. J Appl Oral Sci. 2009;17:555–559. doi: 10.1590/S1678-77572009000600003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Tanriverdi F, Esener T, Erganiş O, Belli S. An in vitro test model for investigation of disinfection of dentinal tubules infected with Enterococcus faecalis. Braz Dent J. 1997;8:67–72. [PubMed] [Google Scholar]
- 102.Krithikadatta J, Indira R, Dorothykalyani AL. Disinfection of dentinal tubules with 2% chlorhexidine, 2% metronidazole, bioactive glass when compared with calcium hydroxide as intracanal medicaments. J Endod. 2007;33:1473–1476. doi: 10.1016/j.joen.2007.08.016. [DOI] [PubMed] [Google Scholar]
- 103.Kayaoglu G, Ömürlü H, Akca G, Gürel M, Gençay Ö, Sorkun K, Salih B. Antibacterial activity of Propolis versus conventional endodontic disinfectants against Enterococcus faecalis in infected dentinal tubules. J Endod. 2011;37:376–381. doi: 10.1016/j.joen.2010.11.024. [DOI] [PubMed] [Google Scholar]
- 104.Delgado RJ, Gasparoto TH, Sipert CR, Pinheiro CR, de Moraes IG, Garcia RB, Duarte MA, Bramante CM, Torres SA, Garlet GP, Campanelli AP, Bernardineli N. Antimicrobial activity of calcium hydroxide and chlorhexidine on intratubular Candida albicans. Int J Oral Sci. 2013;5:32–36. doi: 10.1038/ijos.2013.12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Pan J, Sun K, Liang Y, Sun P, Yang X, Wang J, Zhang J, Zhu W, Fang J, Becker KH. Cold plasma therapy of a tooth root canal infected with Enterococcus faecalis biofilms in vitro. J Endod. 2013;39:105–110. doi: 10.1016/j.joen.2012.08.017. [DOI] [PubMed] [Google Scholar]
- 106.Lana PE, Scelza MF, Silva LE, Mattos-Guaraldi AL, Hirata Júnior R. Antimicrobial activity of calcium hydroxide pastes on Enterococcus faecalis cultivated in root canal systems. Braz Dent J. 2009;20:32–36. doi: 10.1590/s0103-64402009000100005. [DOI] [PubMed] [Google Scholar]
- 107.Delgado RJ, Gasparoto TH, Sipert CR, Pinheiro CR, Moraes IG, Garcia RB, Bramante CM, Campanelli AP, Bernardineli N. Antimicrobial effects of calcium hydroxide and chlorhexidine on Enterococcus faecalis. J Endod. 2010;36:1389–1393. doi: 10.1016/j.joen.2010.04.013. [DOI] [PubMed] [Google Scholar]
- 108.Harrison AJ, Chivatxaranukul P, Parashos P, Messer HH. The effect of ultrasonically activated irrigation on reduction of Enterococcus faecalis in experimentally infected root canals. Int Endod J. 2010;43:968–977. doi: 10.1111/j.1365-2591.2010.01715.x. [DOI] [PubMed] [Google Scholar]
- 109.Kandaswamy D, Venkateshbabu N, Gogulnath D, Kindo AJ. Dentinal tubule disinfection with 2% chlorhexidine gel, propolis, morinda citrifolia juice, 2% povidone iodine, and calcium hydroxide. Int Endod J. 2010;43:419–423. doi: 10.1111/j.1365-2591.2010.01696.x. [DOI] [PubMed] [Google Scholar]
- 110.Madhubala MM, Srinivasan N, Ahamed S. Comparative evaluation of propolis and triantibiotic mixture as an intracanal medicament against Enterococcus faecalis. J Endod. 2011;37:1287–1289. doi: 10.1016/j.joen.2011.05.028. [DOI] [PubMed] [Google Scholar]
- 111.Almyroudi A, Mackenzie D, McHugh S, Saunders WP. The effectiveness of various disinfectants used as endodontic intracanal medications: an in vitro study. J Endod. 2002;28:163–167. doi: 10.1097/00004770-200203000-00005. [DOI] [PubMed] [Google Scholar]
- 112.Oncag O, Cogulu D, Uzel A, Sorkun K. Efficacy of propolis as an intracanal medicament against Enterococcus faecalis. Gen Dent. 2006;54:319–322. [PubMed] [Google Scholar]
- 113.Pavaskar R, de Ataide Ide N, Chalakkal P, Pinto MJ, Fernandes KS, Keny RV, Kamath A. An in vitro study comparing the intracanal effectiveness of calcium hydroxide- and linezolid-based medicaments against Enterococcus faecalis. J Endod. 2012;38:95–100. doi: 10.1016/j.joen.2011.09.031. [DOI] [PubMed] [Google Scholar]
- 114.Sukawat C, Srisuwan T. A comparison of the antimicrobial efficacy of three calcium hydroxide formulations on human dentin infected with Enterococcus faecalis. J Endod. 2002;28:102–104. doi: 10.1097/00004770-200202000-00013. [DOI] [PubMed] [Google Scholar]
- 115.Cwikla SJ, Bélanger M, Giguère S, Progulske-Fox A, Vertucci FJ. Dentinal tubule disinfection using three calcium hydroxide formulations. J Endod. 2005;31:50–52. doi: 10.1097/01.don.0000134291.03828.d1. [DOI] [PubMed] [Google Scholar]
- 116.Lima RK, Guerreiro-Tanomaru JM, Faria-Júnior NB, Tanomaru-Filho M. Effectiveness of calcium hydroxide-based intracanal medicaments against Enterococcus faecalis. Int Endod J. 2012;45:311–316. doi: 10.1111/j.1365-2591.2011.01976.x. [DOI] [PubMed] [Google Scholar]
- 117.Prabhakar AR, Hadakar SG, Raju OS. Comparative evaluation of pH and antibacterial effect of various calcium hydroxide combinations on E. faecalis and its effect on root strength: an in vitro study. Contemp Clin Dent. 2012;3:42–47. doi: 10.4103/0976-237X.94545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Cook J, Nandakumar R, Fouad AF. Molecular- and culture-based comparison of the effects of antimicrobial agents on bacterial survival in infected dentinal tubules. J Endod. 2007;33:690–692. doi: 10.1016/j.joen.2007.01.022. [DOI] [PubMed] [Google Scholar]


