Abstract
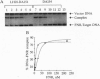
In the facultative anaerobe Escherichia coli, the transcription factor FNR (fumarate nitrate reduction) regulates gene expression in response to oxygen deprivation. To investigate how the activity of FNR is regulated by oxygen availability, two mutant proteins, DA154 and LH28-DA154, which have enhanced in vivo activity in the presence of oxygen, were purified and compared. Unlike other previously examined FNR preparations, the absorption spectrum of LH28-DA154 had two maxima at 324 nm and 419 nm, typical of iron-sulfur (Fe-S)-containing proteins. Consistent with these data, metal analysis showed that only the LH28-DA154 protein contained a significant amount of iron and acid-labile sulfide, and, by low temperature EPR spectroscopy, a signal typical of a [3Fe-4S]+ cluster was detected. The LH28-DA154 protein that contained the Fe-S cluster also contained a higher proportion of dimers and had a 3- to 4-fold higher apparent affinity for the target DNA than the DA154 protein. In agreement with this, we found that when the LH28-DA154 protein was treated with an iron chelator (alpha,alpha'-dipyridyl), it lost its characteristic absorption and the apparent affinity for DNA was reduced 6-fold. However, increased DNA binding and the characteristic absorption spectrum could be restored by in vitro reconstitution of the Fe-S center. DNA binding of the LH28-DA154 protein was also affected by the redox state of the Fe-S center, since protein exposed to oxygen bound 1/10th as much DNA as the protein reduced anaerobically with dithionite. The observation that DNA binding is enhanced when the Fe-S center is reduced indicates that the redox state of the Fe-S center affects the DNA-binding activity of this protein and suggests a possible mechanism for regulation of the wild-type protein.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Beinert H. Recent developments in the field of iron-sulfur proteins. FASEB J. 1990 May;4(8):2483–2491. doi: 10.1096/fasebj.4.8.2185975. [DOI] [PubMed] [Google Scholar]
- Beinert H., Thomson A. J. Three-iron clusters in iron-sulfur proteins. Arch Biochem Biophys. 1983 Apr 15;222(2):333–361. doi: 10.1016/0003-9861(83)90531-3. [DOI] [PubMed] [Google Scholar]
- Bernlohr D. A., Switzer R. L. Reaction of Bacillus subtilis glutamine phosphoribosylpyrophosphate amidotransferase with oxygen: chemistry and regulation by ligands. Biochemistry. 1981 Sep 29;20(20):5675–5681. doi: 10.1021/bi00523a006. [DOI] [PubMed] [Google Scholar]
- Engel P., Trageser M., Unden G. Reversible interconversion of the functional state of the gene regulator FNR from Escherichia coli in vivo by O2 and iron availability. Arch Microbiol. 1991;156(6):463–470. doi: 10.1007/BF00245393. [DOI] [PubMed] [Google Scholar]
- Green J., Guest J. R. Activation of FNR-dependent transcription by iron: an in vitro switch for FNR. FEMS Microbiol Lett. 1993 Oct 15;113(2):219–222. doi: 10.1111/j.1574-6968.1993.tb06517.x. [DOI] [PubMed] [Google Scholar]
- Green J., Trageser M., Six S., Unden G., Guest J. R. Characterization of the FNR protein of Escherichia coli, an iron-binding transcriptional regulator. Proc Biol Sci. 1991 May 22;244(1310):137–144. doi: 10.1098/rspb.1991.0062. [DOI] [PubMed] [Google Scholar]
- Hidalgo E., Demple B. An iron-sulfur center essential for transcriptional activation by the redox-sensing SoxR protein. EMBO J. 1994 Jan 1;13(1):138–146. doi: 10.1002/j.1460-2075.1994.tb06243.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kennedy M. C., Kent T. A., Emptage M., Merkle H., Beinert H., Münck E. Evidence for the formation of a linear [3Fe-4S] cluster in partially unfolded aconitase. J Biol Chem. 1984 Dec 10;259(23):14463–14471. [PubMed] [Google Scholar]
- Kuo C. F., McRee D. E., Fisher C. L., O'Handley S. F., Cunningham R. P., Tainer J. A. Atomic structure of the DNA repair [4Fe-4S] enzyme endonuclease III. Science. 1992 Oct 16;258(5081):434–440. doi: 10.1126/science.1411536. [DOI] [PubMed] [Google Scholar]
- Lazazzera B. A., Bates D. M., Kiley P. J. The activity of the Escherichia coli transcription factor FNR is regulated by a change in oligomeric state. Genes Dev. 1993 Oct;7(10):1993–2005. doi: 10.1101/gad.7.10.1993. [DOI] [PubMed] [Google Scholar]
- Mulliez E., Fontecave M., Gaillard J., Reichard P. An iron-sulfur center and a free radical in the active anaerobic ribonucleotide reductase of Escherichia coli. J Biol Chem. 1993 Feb 5;268(4):2296–2299. [PubMed] [Google Scholar]
- Niehaus F., Hantke K., Unden G. Iron content and FNR-dependent gene regulation in Escherichia coli. FEMS Microbiol Lett. 1991 Dec 1;68(3):319–323. doi: 10.1016/0378-1097(91)90376-l. [DOI] [PubMed] [Google Scholar]
- Rydén L., Ofverstedt L. G., Beinert H., Emptage M. H., Kennedy M. C. Molecular weight of beef heart aconitase and stoichiometry of the components of its iron-sulfur cluster. J Biol Chem. 1984 Mar 10;259(5):3141–3144. [PubMed] [Google Scholar]
- Shaw D. J., Rice D. W., Guest J. R. Homology between CAP and Fnr, a regulator of anaerobic respiration in Escherichia coli. J Mol Biol. 1983 May 15;166(2):241–247. doi: 10.1016/s0022-2836(83)80011-4. [DOI] [PubMed] [Google Scholar]
- Spiro S., Guest J. R. Inactivation of the FNR protein of Escherichia coli by targeted mutagenesis in the N-terminal region. Mol Microbiol. 1988 Nov;2(6):701–707. doi: 10.1111/j.1365-2958.1988.tb00080.x. [DOI] [PubMed] [Google Scholar]
- Unden G., Becker S., Bongaerts J., Schirawski J., Six S. Oxygen regulated gene expression in facultatively anaerobic bacteria. Antonie Van Leeuwenhoek. 1994;66(1-3):3–22. doi: 10.1007/BF00871629. [DOI] [PubMed] [Google Scholar]
- Unden G. Differential roles for menaquinone and demethylmenaquinone in anaerobic electron transport of E. coli and their fnr-independent expression. Arch Microbiol. 1988;150(5):499–503. doi: 10.1007/BF00422294. [DOI] [PubMed] [Google Scholar]
- Zheng L., White R. H., Cash V. L., Jack R. F., Dean D. R. Cysteine desulfurase activity indicates a role for NIFS in metallocluster biosynthesis. Proc Natl Acad Sci U S A. 1993 Apr 1;90(7):2754–2758. doi: 10.1073/pnas.90.7.2754. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ziegelhoffer E. C., Kiley P. J. In vitro analysis of a constitutively active mutant form of the Escherichia coli global transcription factor FNR. J Mol Biol. 1995 Jan 27;245(4):351–361. doi: 10.1006/jmbi.1994.0029. [DOI] [PubMed] [Google Scholar]