To the Editor:
Primary graft dysfunction (PGD) is a form of acute lung injury occurring within 72 hours of lung transplantation (1, 2). We have previously demonstrated an increase in early mortality in subjects with PGD (2). Understanding of the mechanisms leading to PGD and early mortality is important in identifying potential therapeutic targets and interventions.
The complement system defines a group of plasma and cell membrane proteins that play a key role in innate immunity as well as regulating adaptive immune response. C3a, C4a, and C5a, downstream products of the activated complement cascade, are potent neutrophil and lymphocyte chemoattractants (3–5). In animal models, activation of the complement system during lung ischemia and reperfusion can lead to cellular injury and lung allograft failure (6, 7).
Based on the relationship between complement activation and ischemia reperfusion injury in the lung allograft, we hypothesized that plasma levels of C3a, C4a, and C5a would be associated with both PGD and mortality after lung transplantation.
We performed a prospective cohort study of 190 lung transplant recipients from 10 centers enrolled between 2008 and 2010 in the Lung Transplant Outcomes Group (LTOG) cohort. We included consecutive subjects who had plasma samples available at the preoperative, 6-hour, and 24-hour time points (see Figure E1 in the online supplement). Clinical and mortality data were collected as described elsewhere (2). Informed consent was obtained from each subject. The Investigational Review Board at each center approved our study.
The primary outcome was grade 3 PGD within 72 hours after transplantation, which has demonstrated construct validity for survival (8). Grade 3 PGD at 24 hours was used to evaluate the relationship between concurrent lung injury, and grade 3 PGD at 48 or 72 hours was used to evaluate a severe lung injury phenotype (2). We evaluated all-cause mortality as an additional end point.
Plasma C3a, C4a, and C5a concentrations were measured using a commercially available cytometric bead array (BD Biosciences, San Jose, CA) in a manner devised to ensure stability of complement (see online supplement). We evaluated absolute differences in levels between each time point to assess changes during the early transplant period.
The association between complement and PGD was determined using Wilcoxon rank sum testing. We used Cox regression to evaluate associations between complement levels and time to death, conditioned on 90-day survival, to exclude bias from the effect of PGD and sepsis on early mortality. We adjusted for recipient, donor, and surgical variables (see online supplement).
Of the 190 subjects enrolled, 82 developed PGD (43%) within 72 hours post-transplant, and 33 had PGD at 48 or 72 hours (17%). Demographics of the cohort are in Table E1. There were no differences between subjects enrolled with blood samples at all three time points and other subjects enrolled in LTOG (Table E2).
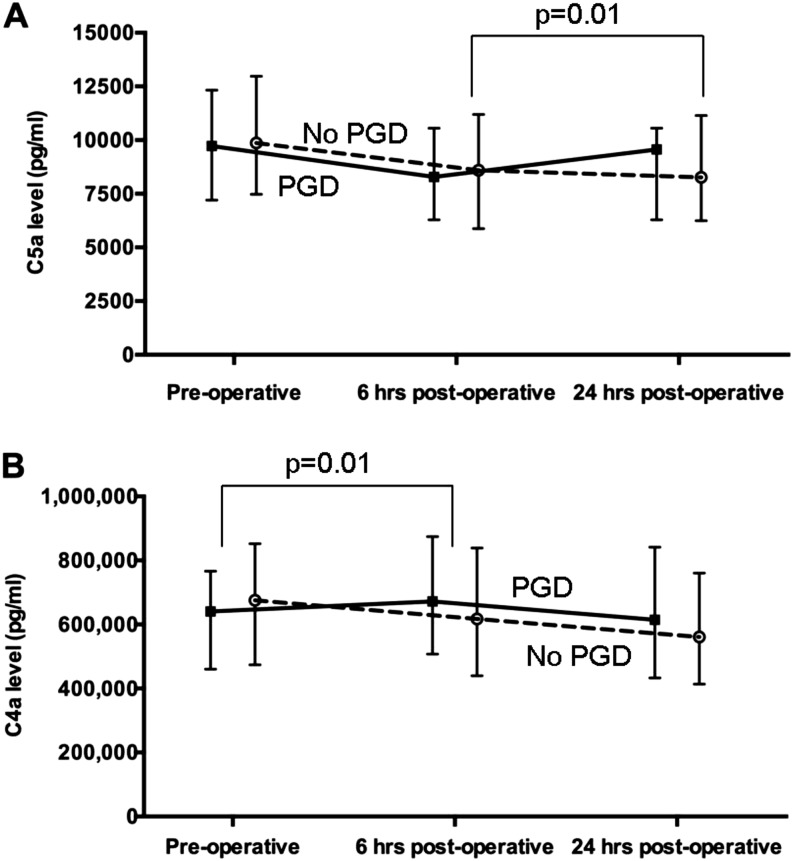
The median change in plasma C5a levels between 6 and 24 hours post transplantation was significantly greater in subjects with PGD than in those without (867 vs. −156 pg/ml, P = 0.01) (Figure 1A). The median change in plasma C4a levels between preoperative and 6 hours was greater in subjects with PGD (72,075 vs. −79,501, P = 0.01) (Figure 1B). There was no significant difference in change in plasma C3a levels between the PGD and non-PGD group at any individual time point (Table E3). In sensitivity analyses, there was no difference in complement levels using grade 3 PGD at 24 hours (Table E4). However, there was a similar relationship between change in plasma C5a levels between 6 and 24 hours (1,164 vs.113, P = 0.24) and change in plasma C4a levels between preoperative and 6 hours (109,654 vs. −8,185, P = 0.04) using grade 3 PGD at 48 or 72 hours.
Figure 1.
(A) Plasma C5a concentration at each time point in subjects without primary graft dysfunction (PGD) (dashed line) and with grade 3 PGD (solid line). The P value indicates that the difference in C5a concentration (represented by the slope of the line) between the 6-hour time point and the 24-hour time point is significantly greater in those with PGD compared with those without. (B) Plasma C4a concentration at each time point in subjects without PGD (dashed line) and with grade 3 PGD (solid line). The P value indicates that the difference in C4a concentration between the preoperative time point and the 6-hour time point (represented by the slope of the line) is significantly greater in those with PGD compared with those without.
Of 175 patients with available mortality data, 32 died (17%). The change in C5a levels from 6 to 24 hours was associated with an increased risk of death (hazard ratio [HR], 1.81; 95% confidence interval [CI], 1.16, 2.83; P = 0.01). C5a levels measured preoperatively and 24 hours after transplantation were also associated with an increased risk of death (HR, 1.64; 95% CI, 1.29, 2.09; P < 0.01; and HR, 1.94; 95% CI, 1.39, 2.72; P < 0.01; respectively). C3a levels measured at 6 hours were associated with an increased risk of death (HR, 1.68; 95% CI, 1.04, 2.71; P = 0.03). There was no association between change in C3a or C4a levels and mortality (Tables E5 and E6). These associations were independent of adjustment for all tested confounders, including PGD (Table 1).
Table 1.
Association of C3a and C5a at Different Time Points with Mortality
| Variable | HR C5a before Transplant (n = 175) | P Value | HR for C5a 24 h after Transplant (n = 175) | P Value | HR for Change in C5a between 6 and 24 h (n = 171) | P Value | HR for C3a at 6 h (n = 159) | P Value |
|---|---|---|---|---|---|---|---|---|
| Unadjusted (per 1 SD) | 1.64 (1.29–2.09) | <0.01 | 1.81 (1.16–2.83) | <0.01 | 1.94 (1.39–2.72) | <0.01 | 1.68 (1.04–2.71) | 0.03 |
| Adjusted for | ||||||||
| Grade 3 PGD | 1.65 (1.29–2.11) | <0.01 | 1.82 (1.17–2.95) | <0.01 | 1.95 (1.38–2.75) | <0.01 | 1.68 (1.04–2.70) | 0.03 |
| CBP | 1.73 (1.28–2.28) | <0.01 | 1.86 (1.28–2.28) | <0.01 | 1.97 (1.39–2.80) | <0.01 | 1.71 (1.06–2.76) | 0.03 |
| Recipient race | 1.63 (1.28–2.09) | <0.01 | 1.86 (1.20–2.89) | <0.01 | 1.93 (1.36–2.74) | <0.01 | 1.70 (1.04–2.77) | 0.03 |
| Recipient diagnosis | 1.82 (1.33–2.49) | <0.01 | 1.92 (1.21–3.04) | <0.01 | 1.95 (1.36–2.80) | <0.01 | 1.73 (1.07–2.80) | 0.03 |
| Recipient sex | 1.57 (1.23–1.99) | <0.01 | 1.79 (1.81–2.72) | <0.01 | 1.86 (1.34–2.59) | <0.01 | 1.71 (1.04–2.80) | 0.03 |
| Recipient age | 1.95 (1.41–2.70) | <0.01 | 1.97 (1.26–3.09) | <0.01 | 2.00 (1.41–2.86) | <0.01 | 1.78 (1.09–2.89) | 0.02 |
| Donor sex | 1.64 (1.28–2.10) | 0.01 | 1.82 (1.16–2.88) | <0.01 | 1.94 (1.38–2.74) | <0.01 | 1.68 (1.04–2.72) | 0.03 |
| Donor age | 1.64 (1.28–2.09) | <0.01 | 1.88 (1.22–2.91) | <0.01 | 1.94 (1.37–2.73) | <0.01 | 1.68 (1.04–2.73) | 0.03 |
| Donor race | 1.66 (1.29–2.14) | <0.01 | 1.81 (1.15–2.83) | <0.01 | 2.02 (1.43–2.85) | <0.01 | 1.66 (1.00–2.75) | 0.05 |
| Donor smoking | 1.69 (1.32–2.17) | <0.01 | 1.96 (1.27–3.04) | <0.01 | 2.06 (1.46–2.90) | <0.01 | 2.06 (1.46–2.90) | <0.01 |
| PRBC | 1.54 (1.18–2.01) | 0.02 | 1.70 (1.10–2.67) | <0.01 | 1.68 (1.10–2.56) | 0.02 | 1.63 (1.01–2.64) | 0.05 |
| Transplant type | 1.66 (1.29–2.13) | <0.01 | 1.81 (1.16–2.82) | <0.01 | 1.94 (1.38–2.73) | <0.01 | 1.71 (1.05–2.77) | 0.03 |
| PASP | 1.64 (1.27–2.11) | 0.02 | 1.79 (1.11–2.89) | <0.01 | 1.99 (1.37–2.90) | <0.01 | 1.74 (1.03–2.95) | 0.04 |
Definition of abbreviations: HR = hazard ratio; PASP = pulmonary artery systolic pressure at the time of transplantation; PGD = primary graft dysfunction within 72 h; PRBC = packed red blood cell transfusion within 24 h.
HRs with 95% confidence intervals are presented per 1-SD change in complement level.
Our study demonstrates that early changes in plasma C4a and C5a are associated with PGD, and there is an association between C3a and C5a levels with mortality, independent of PGD. Collectively, these data suggest that systemic complement activation is important in both short- and long-term outcomes after transplantation.
The increase in C5a between 6 and 24 hours is associated with both PGD and mortality. This suggests a role for an early systemic complement response in both early and late outcomes. Prior work has implicated complement activation in lung ischemia–reperfusion injury (9). The change in C5a level was observed early after transplant and was not associated with concurrent lung injury (grade 3 at 24 h), indicating it may be driving lung injury and not just a marker of injury. Complement may be important in the pathogenesis PGD by attracting neutrophils and accelerating both the innate and adaptive immune responses within the first 24 hours after injury (10, 11).
The association of complement with mortality was independent of PGD. Prior work found an association between C3a, C5a, and obliterative bronchiolitis in a mouse model (12). Our findings in humans suggest a role for complement activation that continues after the early reperfusion period, potentially a signal of chronic immune activation starting in the early post-transplant period and leading to formation of autoantibodies, leading to chronic graft dysfunction and mortality. These findings support the study of blockade of complement activation to improve clinical outcomes after transplantation.
Our study had limitations. We were unable to measure complement levels in bronchoalveolar lavage; therefore, we could not link the plasma and lung compartments. The incidence of PGD in this study was 40%, which is higher than previous reports; however, we observed a similar trend in sensitivity analyses using a more restrictive definition of PGD. Finally, we were unable to adjust for center effect given small numbers.
In conclusion, our study suggests a role for early, systemic activation of the complement pathway in development of severe PGD and mortality after lung transplantation.
Footnotes
Author Contributions: Conception and design, R.J.S., A.M.E., J.M.D., S.M.P., L.B.W., J.D.C., and D.S.W. Acquisition of data, R.J.S., A.M.E., P.A.S., D.W.R., J.B.O., E.C., V.N.L., S.M.B., M.C., K.M.W., J.B.O., A.W., D.J.L., D.S.W., L.B.W., and J.D.C. Analysis and interpretation of data, R.J.S., A.M.E., G.J.E., J.M.D., D.S.W., and J.D.C. Drafting or revising the manuscript for important intellectual content, R.J.S., A.M.E., J.M.D., P.A.S., D.W.R., L.B.W., A.W., S.M.P., G.J.E., V.N.L., S.M.B., J.D.C., and D.S.W. Final approval of the version to be published, all authors.
Funded by National Institutes of Health grants HL096845 (J.D.C. and D.S.W.), HL087115 (J.D.C.), HL081619 (J.D.C.), HL115354 (J.D.C.), HL 114626 (J.D.C. and D.J.L.), T32 HL007568 (R.J.S.), and P01AI084853 (D.S.W.).
This letter has an online supplement, which is accessible from this issue's table of contents at www.atsjournals.org
Author disclosures are available with the text of this letter at www.atsjournals.org
References
- 1.Christie JD, Carby M, Bag R, Corris P, Hertz M, Weill D ISHLT Working Group on Primary Lung Graft Dysfunction. Report of the ISHLT Working Group On Primary Lung Graft Dysfunction Part II: definition. A consensus statement of The International Society for Heart and Lung Transplantation. J Heart Lung Transplant. 2005;24:1454–1459. doi: 10.1016/j.healun.2004.11.049. [DOI] [PubMed] [Google Scholar]
- 2.Diamond JM, Lee JC, Kawut SM, Shah RJ, Localio AR, Bellamy SL, Lederer DJ, Cantu E, Kohl BA, Lama VN, et al. Lung Transplant Outcomes Group. Clinical risk factors for primary graft dysfunction after lung transplantation. Am J Respir Crit Care Med. 2013;187:527–534. doi: 10.1164/rccm.201210-1865OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Strainic MG, Shevach EM, An F, Lin F, Medof ME. Absence of signaling into CD4⁺ cells via C3aR and C5aR enables autoinductive TGF-β1 signaling and induction of Foxp3⁺ regulatory T cells. Nat Immunol. 2013;14:162–171. doi: 10.1038/ni.2499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Frank MM. Complement in the pathophysiology of human disease. N Engl J Med. 1987;316:1525–1530. doi: 10.1056/NEJM198706113162407. [DOI] [PubMed] [Google Scholar]
- 5.Asgari E, Zhou W, Sacks S. Complement in organ transplantation. Curr Opin Organ Transplant. 2010;15:486–491. doi: 10.1097/MOT.0b013e32833b9cb7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Naka Y, Marsh HC, Scesney SM, Oz MC, Pinsky DJ. Complement activation as a cause for primary graft failure in an isogeneic rat model of hypothermic lung preservation and transplantation. Transplantation. 1997;64:1248–1255. doi: 10.1097/00007890-199711150-00004. [DOI] [PubMed] [Google Scholar]
- 7.Bishop MJ, Giclas PC, Guidotti SM, Su ML, Chi EY. Complement activation is a secondary rather than a causative factor in rabbit pulmonary artery ischemia/reperfusion injury. Am Rev Respir Dis. 1991;143:386–390. doi: 10.1164/ajrccm/143.2.386. [DOI] [PubMed] [Google Scholar]
- 8.Christie JD, Bellamy S, Ware LB, Lederer D, Hadjiliadis D, Lee J, Robinson N, Localio AR, Wille K, Lama V, et al. Construct validity of the definition of primary graft dysfunction after lung transplantation. J Heart Lung Transplant. 2010;29:1231–1239. doi: 10.1016/j.healun.2010.05.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Keshavjee S, Davis RD, Zamora MR, de Perrot M, Patterson GA. A randomized, placebo-controlled trial of complement inhibition in ischemia-reperfusion injury after lung transplantation in human beings. J Thorac Cardiovasc Surg. 2005;129:423–428. doi: 10.1016/j.jtcvs.2004.06.048. [DOI] [PubMed] [Google Scholar]
- 10.Peng Q, Li K, Sacks SH, Zhou W. The role of anaphylatoxins C3a and C5a in regulating innate and adaptive immune responses. Inflamm Allergy Drug Targets. 2009;8:236–246. doi: 10.2174/187152809788681038. [DOI] [PubMed] [Google Scholar]
- 11.Pratt JR, Basheer SA, Sacks SH. Local synthesis of complement component C3 regulates acute renal transplant rejection. Nat Med. 2002;8:582–587. doi: 10.1038/nm0602-582. [DOI] [PubMed] [Google Scholar]
- 12.Suzuki H, Lasbury ME, Fan L, Vittal R, Mickler EA, Benson HL, Shilling R, Wu Q, Weber DJ, Wagner SR, et al. Role of complement activation in obliterative bronchiolitis post-lung transplantation. J Immunol. 2013;191:4431–4439. doi: 10.4049/jimmunol.1202242. [DOI] [PMC free article] [PubMed] [Google Scholar]