Abstract
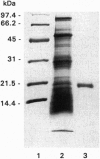
A cyclophilin (CyP) purified to homogeneity from the polycentric anaerobic rumen fungus Orpinomyces sp. strain PC-2 had a molecular mass of 20.5 kDa and a pI of 8.1. The protein catalyzed the isomerization of the prolyl peptide bond of N-succinyl-Ala-Ala-(cis,trans)-Pro-Phe p-nitroanilide with a kcat/Km value of 9.3 x 10(6) M-1.s-1 at 10 degrees C and pH 7.8. Cyclosporin A strongly inhibited this peptidylprolyl cis-trans isomerase activity with an IC50 of 19.6 nM. The sequence of the first 30 N-terminal amino acids of this CyP had high homology with the N-terminal sequences of other eukaryotic CyPs. By use of a DNA hybridization probe amplified by PCR with degenerate oligonucleotide primers designed based on the amino acid sequences of the N terminus of this CyP and highly conserved internal regions of other CyPs, a full-length cDNA clone was isolated. It possessed an open reading frame encoding a polypeptide of 203 amino acids with a calculated molecular weight of 21,969, containing a putative hydrophobic signal peptide sequence of 22 amino acids preceding the N terminus of the mature enzyme and a C-terminal sequence, Lys-Ala-Glu-Leu, characteristic of an endoplasmic reticulum retention signal. The Orpinomyces PC-2 CyP is a typical type B CyP. The amino acid sequence of the Orpinomyces CyP exhibits striking degrees of identity with the corresponding human (70%), bovine (69%), mouse (68%), chicken (66%), maize (61%), and yeast (54%) proteins. Phylogenetic analysis based on the CyP sequences indicated that the evolutionary origin of the Orpinomyces CyP was closely related with CyPs of animals.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Barichievich E. M., Calza R. E. Supernatant protein and cellulase activities of the anaerobic ruminal fungus Neocallimastix frontalis EB188. Appl Environ Microbiol. 1990 Jan;56(1):43–48. doi: 10.1128/aem.56.1.43-48.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bartling D., Heese A., Weiler E. W. Nucleotide sequence of a cDNA encoding an Arabidopsis cyclophilin-like protein. Plant Mol Biol. 1992 Jun;19(3):529–530. doi: 10.1007/BF00023405. [DOI] [PubMed] [Google Scholar]
- Borneman W. S., Akin D. E., Ljungdahl L. G. Fermentation products and plant cell wall-degrading enzymes produced by monocentric and polycentric anaerobic ruminal fungi. Appl Environ Microbiol. 1989 May;55(5):1066–1073. doi: 10.1128/aem.55.5.1066-1073.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bose S., Mücke M., Freedman R. B. The characterization of a cyclophilin-type peptidyl prolyl cis-trans-isomerase from the endoplasmic-reticulum lumen. Biochem J. 1994 Jun 15;300(Pt 3):871–875. doi: 10.1042/bj3000871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bossard M. J., Koser P. L., Brandt M., Bergsma D. J., Levy M. A. A single Trp121 to Ala121 mutation in human cyclophilin alters cyclosporin A affinity and peptidyl-prolyl isomerase activity. Biochem Biophys Res Commun. 1991 May 15;176(3):1142–1148. doi: 10.1016/0006-291x(91)90404-u. [DOI] [PubMed] [Google Scholar]
- Bram R. J., Crabtree G. R. Calcium signalling in T cells stimulated by a cyclophilin B-binding protein. Nature. 1994 Sep 22;371(6495):355–358. doi: 10.1038/371355a0. [DOI] [PubMed] [Google Scholar]
- Caroni P., Rothenfluh A., McGlynn E., Schneider C. S-cyclophilin. New member of the cyclophilin family associated with the secretory pathway. J Biol Chem. 1991 Jun 15;266(17):10739–10742. [PubMed] [Google Scholar]
- Compton L. A., Davis J. M., Macdonald J. R., Bächinger H. P. Structural and functional characterization of Escherichia coli peptidyl-prolyl cis-trans isomerases. Eur J Biochem. 1992 Jun 15;206(3):927–934. doi: 10.1111/j.1432-1033.1992.tb17002.x. [DOI] [PubMed] [Google Scholar]
- Fischer G., Bang H., Mech C. Nachweis einer Enzymkatalyse für die cis-trans-Isomerisierung der Peptidbindung in prolinhaltigen Peptiden. Biomed Biochim Acta. 1984;43(10):1101–1111. [PubMed] [Google Scholar]
- Freskgård P. O., Bergenhem N., Jonsson B. H., Svensson M., Carlsson U. Isomerase and chaperone activity of prolyl isomerase in the folding of carbonic anhydrase. Science. 1992 Oct 16;258(5081):466–468. doi: 10.1126/science.1357751. [DOI] [PubMed] [Google Scholar]
- Frigerio G., Pelham H. R. A Saccharomyces cerevisiae cyclophilin resident in the endoplasmic reticulum. J Mol Biol. 1993 Sep 5;233(1):183–188. doi: 10.1006/jmbi.1993.1497. [DOI] [PubMed] [Google Scholar]
- Gasser C. S., Gunning D. A., Budelier K. A., Brown S. M. Structure and expression of cytosolic cyclophilin/peptidyl-prolyl cis-trans isomerase of higher plants and production of active tomato cyclophilin in Escherichia coli. Proc Natl Acad Sci U S A. 1990 Dec;87(24):9519–9523. doi: 10.1073/pnas.87.24.9519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haendler B., Hofer-Warbinek R., Hofer E. Complementary DNA for human T-cell cyclophilin. EMBO J. 1987 Apr;6(4):947–950. doi: 10.1002/j.1460-2075.1987.tb04843.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haendler B., Keller R., Hiestand P. C., Kocher H. P., Wegmann G., Movva N. R. Yeast cyclophilin: isolation and characterization of the protein, cDNA and gene. Gene. 1989 Nov 15;83(1):39–46. doi: 10.1016/0378-1119(89)90401-0. [DOI] [PubMed] [Google Scholar]
- Handschumacher R. E., Harding M. W., Rice J., Drugge R. J., Speicher D. W. Cyclophilin: a specific cytosolic binding protein for cyclosporin A. Science. 1984 Nov 2;226(4674):544–547. doi: 10.1126/science.6238408. [DOI] [PubMed] [Google Scholar]
- Harding M. W., Galat A., Uehling D. E., Schreiber S. L. A receptor for the immunosuppressant FK506 is a cis-trans peptidyl-prolyl isomerase. Nature. 1989 Oct 26;341(6244):758–760. doi: 10.1038/341758a0. [DOI] [PubMed] [Google Scholar]
- Harding M. W., Handschumacher R. E., Speicher D. W. Isolation and amino acid sequence of cyclophilin. J Biol Chem. 1986 Jun 25;261(18):8547–8555. [PubMed] [Google Scholar]
- Hasel K. W., Glass J. R., Godbout M., Sutcliffe J. G. An endoplasmic reticulum-specific cyclophilin. Mol Cell Biol. 1991 Jul;11(7):3484–3491. doi: 10.1128/mcb.11.7.3484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hayano T., Takahashi N., Kato S., Maki N., Suzuki M. Two distinct forms of peptidylprolyl-cis-trans-isomerase are expressed separately in periplasmic and cytoplasmic compartments of Escherichia coli cells. Biochemistry. 1991 Mar 26;30(12):3041–3048. doi: 10.1021/bi00226a009. [DOI] [PubMed] [Google Scholar]
- Helm K. W., LaFayette P. R., Nagao R. T., Key J. L., Vierling E. Localization of small heat shock proteins to the higher plant endomembrane system. Mol Cell Biol. 1993 Jan;13(1):238–247. doi: 10.1128/mcb.13.1.238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herrler M., Bang H., Marahiel M. A. Cloning and characterization of ppiB, a Bacillus subtilis gene which encodes a cyclosporin A-sensitive peptidyl-prolyl cis-trans isomerase. Mol Microbiol. 1994 Mar;11(6):1073–1083. doi: 10.1111/j.1365-2958.1994.tb00384.x. [DOI] [PubMed] [Google Scholar]
- High K. P., Joiner K. A., Handschumacher R. E. Isolation, cDNA sequences, and biochemical characterization of the major cyclosporin-binding proteins of Toxoplasma gondii. J Biol Chem. 1994 Mar 25;269(12):9105–9112. [PubMed] [Google Scholar]
- Kallen J., Spitzfaden C., Zurini M. G., Wider G., Widmer H., Wüthrich K., Walkinshaw M. D. Structure of human cyclophilin and its binding site for cyclosporin A determined by X-ray crystallography and NMR spectroscopy. Nature. 1991 Sep 19;353(6341):276–279. doi: 10.1038/353276a0. [DOI] [PubMed] [Google Scholar]
- Kallen J., Walkinshaw M. D. The X-ray structure of a tetrapeptide bound to the active site of human cyclophilin A. FEBS Lett. 1992 Apr 6;300(3):286–290. doi: 10.1016/0014-5793(92)80865-e. [DOI] [PubMed] [Google Scholar]
- Ke H. M., Zydowsky L. D., Liu J., Walsh C. T. Crystal structure of recombinant human T-cell cyclophilin A at 2.5 A resolution. Proc Natl Acad Sci U S A. 1991 Nov 1;88(21):9483–9487. doi: 10.1073/pnas.88.21.9483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kofron J. L., Kuzmic P., Kishore V., Colón-Bonilla E., Rich D. H. Determination of kinetic constants for peptidyl prolyl cis-trans isomerases by an improved spectrophotometric assay. Biochemistry. 1991 Jun 25;30(25):6127–6134. doi: 10.1021/bi00239a007. [DOI] [PubMed] [Google Scholar]
- Koser P. L., Livi G. P., Levy M. A., Rosenberg M., Bergsma D. J. A Candida albicans homolog of a human cyclophilin gene encodes a peptidyl-prolyl cis-trans isomerase. Gene. 1990 Dec 15;96(2):189–195. doi: 10.1016/0378-1119(90)90252-m. [DOI] [PubMed] [Google Scholar]
- Koser P. L., Sylvester D., Livi G. P., Bergsma D. J. A second cyclophilin-related gene in Saccharomyces cerevisiae. Nucleic Acids Res. 1990 Mar 25;18(6):1643–1643. doi: 10.1093/nar/18.6.1643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li X. L., Ljungdahl L. G. Cloning, sequencing, and regulation of a xylanase gene from the fungus Aureobasidium pullulans Y-2311-1. Appl Environ Microbiol. 1994 Sep;60(9):3160–3166. doi: 10.1128/aem.60.9.3160-3166.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu J., Chen C. M., Walsh C. T. Human and Escherichia coli cyclophilins: sensitivity to inhibition by the immunosuppressant cyclosporin A correlates with a specific tryptophan residue. Biochemistry. 1991 Mar 5;30(9):2306–2310. doi: 10.1021/bi00223a003. [DOI] [PubMed] [Google Scholar]
- Liu J., Farmer J. D., Jr, Lane W. S., Friedman J., Weissman I., Schreiber S. L. Calcineurin is a common target of cyclophilin-cyclosporin A and FKBP-FK506 complexes. Cell. 1991 Aug 23;66(4):807–815. doi: 10.1016/0092-8674(91)90124-h. [DOI] [PubMed] [Google Scholar]
- Liu J., Walsh C. T. Peptidyl-prolyl cis-trans-isomerase from Escherichia coli: a periplasmic homolog of cyclophilin that is not inhibited by cyclosporin A. Proc Natl Acad Sci U S A. 1990 Jun;87(11):4028–4032. doi: 10.1073/pnas.87.11.4028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Müller M. The hydrogenosome. J Gen Microbiol. 1993 Dec;139(12):2879–2889. doi: 10.1099/00221287-139-12-2879. [DOI] [PubMed] [Google Scholar]
- Pahl A., Uhlein M., Bang H., Schlumbohm W., Keller U. Streptomycetes possess peptidyl-prolyl cis-trans isomerases that strongly resemble cyclophilins from eukaryotic organisms. Mol Microbiol. 1992 Dec;6(23):3551–3558. doi: 10.1111/j.1365-2958.1992.tb01790.x. [DOI] [PubMed] [Google Scholar]
- Pelham H. R. Control of protein exit from the endoplasmic reticulum. Annu Rev Cell Biol. 1989;5:1–23. doi: 10.1146/annurev.cb.05.110189.000245. [DOI] [PubMed] [Google Scholar]
- Price E. R., Zydowsky L. D., Jin M. J., Baker C. H., McKeon F. D., Walsh C. T. Human cyclophilin B: a second cyclophilin gene encodes a peptidyl-prolyl isomerase with a signal sequence. Proc Natl Acad Sci U S A. 1991 Mar 1;88(5):1903–1907. doi: 10.1073/pnas.88.5.1903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rinfret A., Collins C., Ménard R., Anderson S. K. The N-terminal cyclophilin-homologous domain of a 150-kilodalton tumor recognition molecule exhibits both peptidylprolyl cis-trans-isomerase and chaperone activities. Biochemistry. 1994 Feb 22;33(7):1668–1673. doi: 10.1021/bi00173a008. [DOI] [PubMed] [Google Scholar]
- Schreiber S. L. Chemistry and biology of the immunophilins and their immunosuppressive ligands. Science. 1991 Jan 18;251(4991):283–287. doi: 10.1126/science.1702904. [DOI] [PubMed] [Google Scholar]
- Schumacher A., Schröter H., Multhaup G., Nordheim A. Murine cyclophilin-S1: a variant peptidyl-prolyl isomerase with a putative signal sequence expressed in differentiating F9 cells. Biochim Biophys Acta. 1991 Dec 2;1129(1):13–22. doi: 10.1016/0167-4781(91)90207-3. [DOI] [PubMed] [Google Scholar]
- Schönbrunner E. R., Mayer S., Tropschug M., Fischer G., Takahashi N., Schmid F. X. Catalysis of protein folding by cyclophilins from different species. J Biol Chem. 1991 Feb 25;266(6):3630–3635. [PubMed] [Google Scholar]
- Siekierka J. J., Hung S. H., Poe M., Lin C. S., Sigal N. H. A cytosolic binding protein for the immunosuppressant FK506 has peptidyl-prolyl isomerase activity but is distinct from cyclophilin. Nature. 1989 Oct 26;341(6244):755–757. doi: 10.1038/341755a0. [DOI] [PubMed] [Google Scholar]
- Spitzfaden C., Weber H. P., Braun W., Kallen J., Wider G., Widmer H., Walkinshaw M. D., Wüthrich K. Cyclosporin A-cyclophilin complex formation. A model based on X-ray and NMR data. FEBS Lett. 1992 Apr 6;300(3):291–300. doi: 10.1016/0014-5793(92)80866-f. [DOI] [PubMed] [Google Scholar]
- Stamnes M. A., Rutherford S. L., Zuker C. S. Cyclophilins: a new family of proteins involved in intracellular folding. Trends Cell Biol. 1992 Sep;2(9):272–276. doi: 10.1016/0962-8924(92)90200-7. [DOI] [PubMed] [Google Scholar]
- Tropschug M., Nicholson D. W., Hartl F. U., Köhler H., Pfanner N., Wachter E., Neupert W. Cyclosporin A-binding protein (cyclophilin) of Neurospora crassa. One gene codes for both the cytosolic and mitochondrial forms. J Biol Chem. 1988 Oct 5;263(28):14433–14440. [PubMed] [Google Scholar]
- Wainright P. O., Hinkle G., Sogin M. L., Stickel S. K. Monophyletic origins of the metazoa: an evolutionary link with fungi. Science. 1993 Apr 16;260(5106):340–342. doi: 10.1126/science.8469985. [DOI] [PubMed] [Google Scholar]
- von Heijne G. A new method for predicting signal sequence cleavage sites. Nucleic Acids Res. 1986 Jun 11;14(11):4683–4690. doi: 10.1093/nar/14.11.4683. [DOI] [PMC free article] [PubMed] [Google Scholar]