Abstract
The developmental regulation of globin gene expression has served as an important model for understanding higher eukaryotic transcriptional control mechanisms. During human erythroid development there is a sequential switch from expression of the embryonic ε-globin gene to the fetal ɣ-globin gene in utero, and post-partum the ɣ-globin gene is silenced as the β-globin gene becomes the predominantly expressed locus. Because the expression of normally silenced fetal ɣ-type globin genes, and resultant production of fetal hemoglobin (HbF) in adult erythroid cells can ameliorate the pathophysiologic consequences of both abnormal β-globin chains in sickle cell anemia and deficient β-globin chain production in β-thalassemia, understanding the complex mechanisms of this developmental switch has direct translational clinical relevance. Of particular interest for translational research are the factors that mediate silencing of the ɣ-globin gene in adult stage erythroid cells. In addition to the regulatory roles of transcription factors and their cognate DNA sequence motifs, there has been a growing appreciation of the role of epigenetic signals and their cognate factors in gene regulation, and in particular in gene silencing through chromatin. Much of the information about epigenetic silencing stems from studies of globin gene regulation. As discussed here, the term epigenetics refers to post-synthetic modifications of DNA and chromosomal histone proteins that affect gene expression and can be inherited through somatic cell replication. A full understanding of the molecular mechanisms of epigenetic silencing of fetal hemoglobin expression should facilitate development of more effective treatment of β-globin chain hemoglobinopathies.
Introduction
DNA methylation was the first well described epigenetic signal and was long posited to have a role in gene regulation (1-3). Vertebrate globin genes were among the first in which an inverse relationship between cytosine methylation and transcription was demonstrated (4-7). Both histone and non-histone chromosomal protein post-synthetic modifications have also been shown to have important roles in gene regulation, a concept formalized as the histone code (8-10). These relationships have been described in detail in a recent review (11). The current discussion will focus primarily on the epigenetic mechanisms involved in developmental human β-type globin gene silencing (and hence fetal hemoglobin silencing) and the preclinical and potential clinical translational avenues for overcoming this silencing in context of the treatment of inherited β-globin gene disorders.
In all vertebrates that have been studied, a switch from embryonic, or primitive, to definitive hemoglobin production occurs in erythroid cells during development. In humans and old world primates, as well as certain ruminants, an intermediate fetal hemoglobin (HbF) predominates during mid to late gestational stages and persists at a low level post-partum in definitive erythroid cells after adult hemoglobin (HbA) predominates (Table 1). The details of this switch have been reviewed extensively (12, 13).
Table 1. Developmental stage-specific human and mouse β-type globin gene and corresponding hemoglobin expression patterns.
| Developmental Stage | Predominant β-type Globin (Hemoglobin) | |
|---|---|---|
| Mouse | Primitive | βH1, εγ (Hb E) I-III |
| Definitive | βmaj (Hb βmaj) | |
| βmin (Hb βmin) | ||
| Human | Primitive | ε (Hb Gower-1, Hb Gower-2) |
| Definitive | ||
| Fetal Gestational | ɣ (HbF), (Hb Portland-1) | |
| Post-partum | β (HbA) | |
| δ (HbA2) |
As with much of human biology, the ability to identify important regulatory mechanisms that are physiologically relevant is a major challenge requiring robust pre-clinical models for understanding ɣ-globin gene silencing in adults and successfully targeting those mechanisms therapeutically. Because of a high degree of evolutionary conservation of gene regulatory mechanisms in erythroid cells, transgenic mice bearing a yeast artificial chromosome containing an intact human β-globin gene locus (β-globin YAC), have provided a valuable model system for studying developmental globin gene regulation. The transgenic mouse model also allows for testing the effects of modulating epigenetic processes in the context of whole animal physiology. At the same time the β-globin YAC mouse model is limited by the fact that the mouse lacks a true analog of the human fetal erythroid compartment, such that the transgenic human ɣ-globin gene is regulated like the murine embryonic β-type globin genes, which are repressed several orders of magnitude more than the human ɣ-globin gene in adult humans (14), (Table 1).
Cultured primary human erythroid cells derived from CD34+ progenitors induced to erythroid differentiation provide another powerful model for studying human ɣ-globin gene silencing (15, 16). The limitations of cultured primary erythroid cells include their limited life span and the fact that achieving terminal erythroid differentiation while maintaining cell viability is often challenging.
The primate baboon model has also been quite useful given that the developmental β-type globin gene repertoire of the baboon is very similar to humans, including a fetal hemoglobin (17). Other vertebrate models and cultured cell systems have provided important early insights into epigenetic control of globin gene silencing, but the discussion of pre-clinical translational studies is directed primarily at the above models.
Much of the focus of research on developmental ɣ-globin gene silencing has been on trans-acting transcription factors. The discovery of the quantitative trait locus BCL11A on chromosome 2p16 (18, 19), identified this factor as an important regulator of fetal hemoglobin expression. Subsequent studies have shown that BCL11A binds to an intergenic region in the β-globin locus and has a dominant silencing effect on murine embryonic β-type H1 and εγ-globin, as well as human ε- and ɣ-globin gene expression in β-YAC transgenic mice (12, 20).
Knockdown of BCL11A in cultured primary human adult erythroid cells also results in a significant upregulation of ɣ-globin gene expression, although the magnitude of this effect is much less than in the β-YAC mouse model (19). The transcription factor SOX6 also mediates embryonic βH1 and εγ-globin gene silencing in the mouse, and it is known to interact with BCL11A (21, 22).
Krüppel-Like Factor 1(KLF1), originally known as Erythroid krüppel-like factor (EKLF), was initially shown to be critical for adult β-globin gene transcription (23), and to increase the ability of the β-globin promoter to compete with the ɣ-globin promoter for the enhancer function of the erythroid-specific β-globin Locus Control Region (LCR) (24, 25). A more direct role of KLF1 in ɣ-globin gene silencing occurs through its stimulation of BCL11A expression (26, 27).
The MYB gene has also been implicated in regulating fetal hemoglobin levels through both quantitative trait locus studies and functional assays (18, 28-30).
A number of other transcription factors having been implicated in embryonic/fetal β-type globin gene silencing. These include GATA1 in association with FOG1 and the NuRD complex (31-34), NF-E4 (35), the TR2/TR4/DRED complex (36, 37), Ikaros in association with the PYR co-regulatory complex (38).
As the transcription factors involved in fetal globin gene silencing have been recently reviewed, the remainder of this review will focus on epigenetic silencing mechanisms (39).
Epigenetic Regulators – Writers and Readers
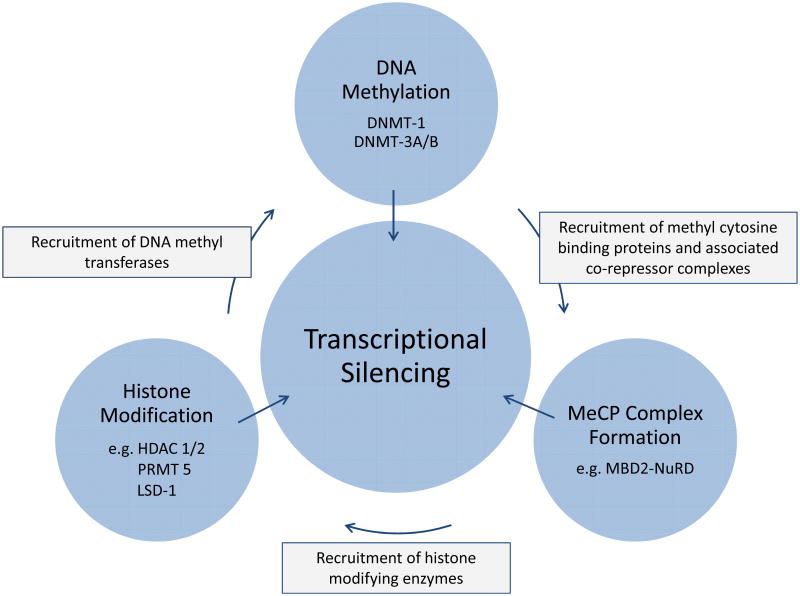
There are only a few examples in which an epigenetic modification of DNA or a chromosomal protein has a direct effect on structure or function (40). An exception is histone acetylation which does appear to directly alter chromatin structure (11, 41). In most cases, epigenetic marks serve as a recognition signal for a protein or protein complex which ultimately carries out the specific associated regulatory function. A useful organizing concept for identifying potential targets for perturbing epigenetic fetal globin gene silencing is that of writers and readers. Writers are the enzymes that deposit or remove an epigenetic mark, while readers are the proteins or complexes that interpret those marks and carry out the associated regulatory function.
DNA Methylation
DNA methyltransferases (DNMTs) are a major category of epigenetic writers, as DNA methylation was the first well established epigenetic regulatory signal. The most well-characterized of these are the denovo methylases, DNMT 3A and 3B, which symmetrically methylate cytosines in the dinucleotide CpG on both strands of unmethylated DNA, and DNMT1, a so-called maintenance methylase, that adds a methyl group to the symmetric CpG on the unmethylated strand of DNA following DNA replication. Since the discovery that silent embryonic and fetal β-type globin genes are methylated, and that the cytidine analog, 5-azacytidine inhibits the processive methylation of hemi-methylated DNA following replication, many studies have focused on DNMT1 as a target for reversing globin gene silencing. Initial studies in animal models (42) were followed by clinical interventions that demonstrated increased fetal hemoglobin expression in patients with both sickle cell anemia and β-thalassemia who were treated with 5-azacytidine (43-45). The mechanism by which 5-azacytidine actually induces increased human fetal gamma globin gene expression has been debated, and mechanisms such as generalized cytotoxicity and induced erythroid cellular stress have been proposed (13, 46-50). Nonetheless in well-characterized primate and human β-globin gene locus-bearing transgenic mouse models, disruption of DNA methylation appears to be its major mechanism of relieving ɣ-globin gene silencing, although perhaps indirectly in part (51-56). Despite the development of more specific inhibitors of DNMT1, such as decitabine which, unlike 5-azacytidine, lacks effects on RNA metabolism, concerns about the safety of this class of agents have limited clinical application in β-hemoglobinopathies. However, a recent study of low dose decitabine in β-thalassemia patients reported an increase in HbF without detectable short term cytotoxicity or genotoxicity (57).
The readers of DNA methylation are a group of proteins that preferentially bind to DNA containing symmetrically methylated CpG dinucleotides. The largest family of these are the Methyl cytosine Binding Domain (MBD) proteins, which include MBD1, MBD2, MECP2, and MBD4 (58). Of these, the role of MBD2 in regulating embryonic/fetal β-type globin gene silencing in adult erythroid cells is the most-well characterized. MBD2 binds preferentially to DNA containing a high density of methylated CpGs. MBD2 has been shown to bind directly to the avian embryonic ρ -globin gene and knockdown of MBD2 de-represses the gene in adult erythroid cells in culture (59). Knockdown of MBD2 has also been shown to induce a large increase in expression of the silent human ɣ-globin gene in human β-globin locus bearing transgenic mice (53), and in human primary CD34 precursor derived adult erythroid cells (60). Importantly, complete knockout of MBD2 in transgenic mice does not result in any major deleterious phenotype (61), suggesting that its disruption postnatally in somatic cells may have minimal toxicity.
MBD2 mediates silencing by recruiting the Nucleosome Remodeling and Deacetylase (NuRD) complex to methylated DNA (62, 63). Structural studies of the MBD2-NuRD complex have identified a critical coiled-coil protein interaction between MBD2 and p66α/β, another NuRD complex component. Enforced expression of the p66 coiled-coil protein results in release of the Mi2β chromatin remodeling ATPase from the NuRD complex, and de-repression of the silenced embryonic and fetal β-type globin genes, presumably by decoupling MBD2 from the NuRD chromatin remodeling function (60).
A closely related member of the MBD family, MBD3, also associates with a NuRD complex, but does not bind to methylated versus non-methylated DNA with high affinity (58, 64). Moreover, the presence of MBD2 and MBD3 in association with NuRD complex appears to be mutually exclusive (65). MBD3-NuRD is associated with the ɣ-globin gene promoter primarily through association with the GATA1 transcription factor associated protein, Friend of GATA1 (FOG1), (32, 33) or other complexes (66). Disruption of expression of the Mi2β subunit of NuRD results in increased ɣ-globin gene expression in transgenic mice (34), cultured mouse CID hematopoietic cells bearing a human β-globin gene locus and cultured primary human erythroid cells (67).
Recently it was shown that as little as a 50% knockdown of Mi2β in primary human erythroid cells results in a ∼10-fold increase in ɣ-globin gene expression without affecting erythroid differentiation, compared to control CD34+ progenitor derived erythroid cells treated with scramble shRNA (67). The degree of differentiation in control cells in these studies leads to a level of 1% ɣ/ɣ+β RNA, which is comparable to normal adult reticulocyte levels. Interestingly, in these studies, the effect of Mi2β on ɣ-globin gene silencing did not appear to be due largely to an effect on MBD2-NuRD or MBD3-NuRD. Rather at least part of the effect was through down regulation of BCL11A and KLF1 in Mi2β knockdown erythroid cells. The purposed relationships of MBD2-NuRD, MBD3-NuRD and Mi2β in ɣ-globin gene silencing in the context of other major epigenetic regulatory factors are depicted in Figure 1, (67). Based on the preponderance of evidence, it appears that MBD2 plays a greater role than MBD3 but not than Mi2β, in silencing ɣ-globin gene expression, while Mi2β plays a greater role than either MBD2 or MBD3.
Figure 1. Mi2β-mediated epigenetic globin gene silencing through multiple mechanisms.
Mi2β is a critical component of the MBD2/NuRD complex which regulates developmental globin gene silencing independently of BCL11A and KLF1/EKLF. Mi2β also binds to the distal promoter region of the γ-globin gene as part of the MBD3/NuRD/GATA-1/FOG-1 silencing complex. Importantly, Mi2β binds to and activates expression of BCL11A and KLF1/EKLF which in turn silence ɣ-globin gene expression. In each of these processes, Mi2β has been shown to directly promote ɣ-globin gene silencing. As depicted, Mi2β is also associated with the BCL11A complex and the TR2/TR4/DRED complex through its association with NuRD, but as indicated by the non-colored symbol, its role in the action of these complexes has not been demonstrated directly. Likewise, other non-colored symbols designate epigenetic modulators that have not been shown directly to mediate silencing in association with factors designated by colored symbols.
Histone Acetylation
Increased histone acetylation has long been posited to be associated with decompressed chromatin and active gene expression (68, 69). The writers for histone acetylation are histone acetyltransferases including P300/CBP; PCAF and TAF(11)250, (70) as well as histone deacetylases (which might be more properly thought of as “erasers”). The complexity of histone acetylation and its relationship to gene regulation has been intensively studied and will not be reviewed in detail here. The consensus is that acetylation of lysines at histone tails results in charge neutralization and loosening of the interaction of nucleosomes with their associated DNA (11).
A number of inhibitors of histone deacetylases have been identified or synthesized, the prototype being butyric acid (69). Butyric acid and derivatives were shown to induce expression of silenced embryonic and fetal β-type globin genes in several animal models (71, 72). While increased fetal hemoglobin expression was associated with increased histone acetylation in the vicinity of the ɣ-globin gene, (54) it is important to recognize that histone deacetylases might potentially affect acetylation of transcription factors and other non-histone proteins. Moreover, butyrate and other HDAC inhibitors have been shown to affect other signaling pathways including the STAT5, cAMP and MAP kinase systems (73-75). Thus the exact molecular mechanism(s) of ɣ-globin gene activation by HDACs are not fully known. Nonetheless, treatment of patients with sickle cell anemia and β-thalassemia with sodium butyrate and butyric acid was shown to induce increased HbF expression (76, 77). The effect of naturally occurring butyrates is somewhat variable, possibly reflecting phenotypic differences in their metabolism or in the factors that are responsible for the mechanisms of action. Extensive efforts have been made to improve on the effectiveness of histone deacetylase inhibitors while decreasing unwanted side effects. Recent large scale chemical genetic studies independently identified HDAC1 and HDAC2 inhibitors as inducers of ɣ-globin gene expression, (78) affirming the likely mechanism of action of butyric acid and some of its derivatives.
Histone Methylation
Unlike histone acetylation, which is generally associated with both active chromatin configuration and gene expression, histone methylation can signal either gene activation, gene silencing, or a bivalent state. For example, histone H3K4me3 methylation is usually associated with open chromatin and gene transcription while histone H3K9 and H3K27me3 methylation are most frequently associated with gene silencing (8, 79, 80). The presence of both H3K4me3 and H3K27me3 is associated with a poised bivalent state (81).
The major writers of histone methylation are the SET domain lysine specific methylases and the protein arginine methyltransferases (PRMT). A PRMT5-dependant multi-protein complex has been shown to contribute to human ɣ-globin gene silencing. Moreover, symmetric methylation of histone H4 arginine 3 (H4R3 Me2s) serves as a binding target for DNMT3A leading to methylation at the ɣ-globin gene promoter. The histone lysine methyl transferase Suv4-20h1 and components of the NuRD complex are also associated with these complex (66, 82). Recently, the methyltransferase inhibitor, adenosine-2′, 3′ dialdehyde (Adox) was shown to induce ɣ-globin gene expression in human primary erythroid cells in culture, suggesting that PRMT5 enzymatic activity may be mechanistically linked to silencing. As with most inhibitor studies, the possibility remains that the effects of Adox may be through another methyltransferase (83).
Another member of the protein arginine methyltransferase family, PRMT1, has been associated with human ɣ-globin gene silencing through association with a protein named friend of PRMT1 (FoP) (84). Knockdown of FoP resulted in increased ɣ-globin gene expression in cultured primary human erythroid cells. Interestingly, PRMT1 has also been shown to facilitate a number of histone acetylation events including acetylation of Lys9/Lys14 and subsequent transcription of the adult β-globin gene (85). This result suggests that the enzymatic activity of PRMT1 also may contribute to ɣ-globin gene silencing through increasing the β-globin gene's ability to compete for the β-globin locus control region enhancer activity.
Specific lysine demethylases are also involved in ɣ-globin gene silencing in both murine and human adult erythroid cells. The Lysine-specific demethylase 1 (LSD1) has been shown to associate with the transcription factor BCL11A through a complex containing the repressor element-1 silencing factor co-repressor-1 (CoREST) (86), and to mediate part of BCL11A's strong ɣ-globin gene silencing activity. LSD1 also has been shown to associate with the TR2/TR4/DRED complex, along with several other co-repressor complexes (87). Inhibition of LSD1 by either RNA interference, or the LSD1 enzymatic activity inhibitor, tranylcypromine results in increased ɣ-globin gene expression in β-globin locus-bearing transgenic mice and cultured primary human erythroid cells (86, 88). However, since LSD1 is required for normal erythroid maturation, it has been suggested that its inhibition potentially might adversely affect that process (86).
The Interplay between DNA Methylation and Histone Modification in Regulating Gene Expression
Studies in vertebrate model systems have demonstrated a close and often reinforcing relationship between DNA methylation and repressive histone modifications in gene silencing (89, 90). In some instances DNA methylation and associated Methyl binding domain proteins recruit co-repressor complexes that contain SET domain proteins that catalyze H3K9 methylation (91). Other studies have demonstrated that repressive histone marks such as H3K9 methylation may recruit DNA methyltransferases (92). Conversely, histone acetylation has been shown to prevent extinction of gene expression and subsequent DNA methylation (41, 93). The self-reinforcing nature of these interactions are depicted in Figure 2.
Figure 2. Interdependence of epigenetic gene silencing.
Existing data suggest that DNA methylation and postsynthetic histone modification are dependent on one another and in turn reinforce each other. Either mark appears capable of establishing the other by recruiting epigenetic writer enzymes, thus resulting in a cycle that reinforces gene silencing. For example, as illustrated, DNA methylation recruits methyl cytosine binding proteins which recruit co-repressor complexes that contain histone modifying enzymes that catalyze loss of activating marks (Eg. Histone acetylation and methylation) or addition of repressive marks (Eg. Histone methylation). In turn, repressive histone modifications such as H3K9 methylation can recruit DNA methyltransferases that deposit the repressive methylation mark at CpG dinucleotides.
MicroRNA and RNA Interference
Frequently microRNA (miRNA) and small inhibitory RNA (siRNA) are included in the category of epigenetic regulatory mechanisms. These small RNAs are capable of well-characterized post transcriptional gene silencing, but also have been shown to direct epigenetic modifications in plants and animals (94). Several miRNAs have been implicated in the regulation of ɣ-globin gene expression. Lin28B and the associated let-7 miRNA family are regulated during fetal to adult erythroid development. Enforced expression of LIN28B resulted in increased ɣ-globin gene expression in cultured adult primary human erythroid cells, while LIN28B knockdown decreased ɣ-globin gene expression in fetal cord blood derived human erythroid cells which normally express high levels of HbF (95). At least part of this effect was attributed to the effect of LIN28B on expression of BCL11A. Similarly, microRNA-486-3p was shown to bind to the BCL11A mRNA 3′untranslated region and down regulate its expression concomitant with upregulation of ɣ-globin gene expression in cultured human erythroid cells (96). The role of direct epigenetic changes in the actions of either LIN28B or microRNA-486-3p remains unknown.
Interplay Between Transcription Factors and Epigenetic Regulators
Any discussion of epigenetic regulation of globin gene expression must account for the interplay between transcription factors and co-regulatory complexes with which they interact and which in turn often contain both “writers” (eg. Histone acetylases and deacetylases), and “readers” (eg. Methyl-cytosine binding proteins) of epigenetic chromatin marks. Several transcription factors that are involved in embryonic fetal β-type globin gene silencing are known to associate with one or more co-repressor complexes. Among these, BCL11A has emerged as a dominant regulator of developmental globin gene silencing in mice and is also implicated as a strong mediator of ɣ-globin gene silencing in cultured human primary erythroid cells (19). BCL11A has been shown to associate with the MBD3-NuRD complex, as well as the LSD1/CoREST complex, Sin3A, NCoR/SMRT, and DNMT1 (86). Another transcription factor complex associated with embryonic globin gene silencing, the TR2/TR4/DRED orphan nuclear receptor complex, has been shown to associate with a number of epigenetic co-regulatory proteins, including the MBD3-NuRD, LSD1/CoREST, Sin3A complexes and DNMT1 (87). Thus the effectors of these transcription factors may be in large part epigenetic.
Another connection between epigenetic regulators and transcription factors that are involved in ɣ-globin gene silencing is through epigenetic regulation of expression of the transcription factors themselves. It was recently shown that Mi2β/CHD4, independently of the NuRD complex, is required for high level expression of both KLF1 and BCL11A in primary human adult erythroid cells and that Mi2β/CHD4 binds directly to BCL11A (67), (See Figure 1).
It is important to note that virtually all of the epigenetic and transcriptional regulatory factors that are discussed here and depicted in Figure 1, have been shown to play a role in normal developmental globin gene switching. However, the relative effect of a given factor in the totality of ɣ-globin gene silencing appears to vary considerably in developmental globin silencing or “switching” versus maintenance of silencing in the adult erythroid compartment.
A summary of the co-regulatory complexes that contain known epigenetic readers and writers and that are associated with transcription factors involved in developmental regulation of ɣ-globin gene expression is presented in Table 2.
Table 2. Association of ɣ-globin gene silencing transcription factors with epigenetic modulators.
The epigenetic modulator factors may either act as effectors of transcription factor-mediated silencing (designated by †), or act to regulate expression of a specific transcription factor (designated by *).
| Transcription Factors | Associated Epigenetic Modulators |
|---|---|
| BCL11A | MBD3-NuRD (HDAC1/2)†, CoREST†, N-CoR/SMRT†, LSD1†, Sin3A†, DNMT1†, Mi2β/CHD4* |
| TR2/TR4 | MBD3-NuRD (HDAC1/2) † LSD1†, Sin3A†, DNMT1† |
| GATA1/FOG1 | MBD3-NuRD (HDAC1/2)† |
| NFE4 | DNMT3A† |
| KLF-1 | BRG1/SWI/SNF†, Mi2β/CHD4* |
| Ikaros | SWI/SNF† |
Translational Clinical Trials Targeting Epigenetic HbF Silencing
The first clinical trials aimed at reversing fetal hemoglobin silencing in patients with sickle cell anemia and β-thalassemia targeted DNA methylation with 5-azacytidine (43-45). While subsequent trials with cytotoxic agents raised questions as to the exact mechanism of 5-azacytidine induced HbF expression (13), a number of preclinical studies support a major role for DNA hypo-methylation (51-53, 55, 56). As noted previously, concerns about adverse effects of hypo-methylating agents with known cytotoxicity have limited the widespread use of 5-azacytidine and decitabine in the clinic.
The use of histone deacetylase inhibitors represents the other major example of clinical trials aimed at targeting epigenetic silencing of HbF expression in patients with β-globin gene disorders (76, 77, 97). Recent trials with oral butyrate derivatives have shown activity in patients with β-thalassemia. One such agent Sodium 2, 2 dimethyl butyrate was shown to be tolerated in Phase I/II trial (98). While butyrate and derivative compounds have demonstrated effectiveness in some patients, the effects are variable. The nature of this variability remains unknown and could involve differences in metabolism of various HDAC inhibitors or genetic heterogeneity in acetylated protein targets or downstream regulatory factors in different patients.
Based on the preclinical studies described previously, a number of epigenetic modulators are either in early phase clinical trial testing or such trials are being planned. Among these are inhibitors of the histone lysine demethylase, LSD1 (86, 88), the histone arginine methylase PRMT5 (83), as well as selective HDAC1 and HDAC2 inhibitors (78). The development of more selective HDAC inhibitors may increase their effectiveness while decreasing the unwanted side effects of these agents.
In the face of a large number of epigenetic targets for inducing HbF expression in patients with β-thalassemia and sickle cell anemia, consideration of several factors should guide the further development of targeting strategies. The same considerations also apply to therapeutic targeting of transcription factors such as BCL11A and KLF1. The first is selection of the most informative preclinical model systems to identify promising agents. Human β-globin locus bearing transgenic mice have provided a valuable model to identify important epigenetic and transcription factor silencers of embryonic/fetal β-globin gene expression. However, as noted previously because mice only have embryonic and adult β-globin expression, this model may either underestimate or overestimate the effect that a given epigenetic or genetic fetal globin gene silencer will have in humans.
Cultured human primary erythroid cells derived from CD34+ progenitors also provide a valuable model for identifying epigenetic targets for inducing fetal hemoglobin expression. One important caveat for studies using these cells is that it is very important that the level of ɣ-globin gene expression be measured after extensive erythroid differentiation when the ɣ/ɣ+β expression level in control cells approaches that in normal adult erythroid cells. Agents that interfere with differentiation might result in a sufficient increase in ɣ-globin gene expression in this model to be clinically useful, but may have deleterious effects on erythropoiesis. Variation in the level of erythroid differentiation achieved in studies of agents that disrupt ɣ-globin gene silencing in this cell model system must be taken in to consideration when assessing their relative therapeutic potential.
Another consideration is how specific the effect of a given type of epigenetic targeting might be. Clearly epigenetic regulatory factors have global effects on gene expression in all cell types, so that complete inhibition or ablation would likely be catastrophic in many instances. One exception might be the methyl binding domain protein MBD2, whose complete absence is tolerated in knockout mice with only a minimal phenotype (63). It is also generally believed that only genes that are in a poised state can be readily transcriptionally activated. Thus, if partial inhibition of multiple fetal globin gene silencing mechanisms can be achieved epigenetically, this might be highly effective with acceptable short and long term off target effects. Finally, the feasibility of targeting a given epigenetic regulator must be considered. Those that function through enzymatic activity such as DNA methylases, HDACs, histone demethylases, and histone methylases, and potentially the ATPase activity of Mi2β/CHD4, are more readily druggable. This is largely why clinical trials targeting these regulators already have been carried out or are underway. Like transcription factors, those epigenetic regulators such as MBD2-NuRD that function through protein-protein or protein DNA interactions have been considered “undruggable” in the past. However, recent success in developing agents such as covalently stapled peptides capable of disrupting protein-protein interactions in animal models (99-101), and targeting specific proteins for degradation in the proteasome (102, 103) suggest that this class of epigenetic regulators may be targeted successfully in the future (Table 3).
Table 3. Potential epigenetic therapeutic targets of ɣ-globin gene silencing.
| DNA methyl transferases |
| Histone deacetylases |
| Histone demethylases |
| Methyl-CpG binding domain protein 2 (MBD2) |
| Mi2p/CHD4 ATPase chromatin remodeler |
| PRMT5 |
| Friend of PRMT1 (FoP) |
In summary, epigenetic mechanisms play a key role in fetal globin gene silencing, both independently and in cooperation with specific transcription factor silencers such as BCL11A and KLF1. Among the first proof of principle targeted treatment trials in patients with β- hemoglobinopathies were those aimed at DNA methylation and histone acetylation, two key epigenetic marks of globin gene transcriptional activity. With further understanding of the specificity of epigenetic fetal globin gene silencing mechanisms, it is likely that targeting of this process will result in more successful future treatment of patients with β-globin disorders through induction of increased HbF levels.
Acknowledgments
The author's work presented in this article was supported by NIH grants R01 DK 29902 and R56 DK029902, and Massey Cancer Center Core Grant P30 CA016059
The contributions of Maria Amaya, Megha Desai, Shou Zhen Wang, Sheng Zu Zhu, and many past students and fellows to the work done in the author's laboratory is gratefully acknowledged.
The helpful assistance of Amy Jones in preparation of this manuscript is most appreciated.
All authors have read this journal article and signed.
All authors have read the journal's authorship agreement.
Abbreviations
- HbF
Fetal hemoglobin
- HbA
Adult hemoglobin
- β-YAC
Beta-globin Yeast Artificial Chromosome
- LCR
Locus Control Region
- NuRD
Nucleosome Remodeling and Deacetylase
- DNMT
DNA methyl transferase
- MBD
Methyl cytosine Binding Domain
- shRNA
Short hairpin RNA
- HDAC
Histone deacetylase
- PRMT
Protein arginine methyl transferase
- Adox
Adenosine-2′, 3′-dialdehyde
- FoP
Friend of PRMT1 (Protein arginine methyl transferase)
- siRNA
small inhibitory RNA, BCL11A, B-Cell Lymphoma/Leukemia A
- EKLF
Erythroid krüppel-like factor
- KLF-1
Krüppel-like factor 1
- TR2/TR4/DRED
Nuclear receptor TR2/TR4 complex
- Mi2β/CHD4
Chromodomain-helicase DNA binding protein 4
- CID
Chemical inducer of dimerization
- PCAF
P300-associated factor
- CoREST
REST-co-repressor
- NCoR/SMRT
Nuclear receptor co-repressor-1/Silencing Mediator for Retinoid and Thyroid Receptors
- LSD1
Lysine-Specific Demethylase-1
Footnotes
The authors declare that they have no conflict of interest.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Scarano E, Iaccarino M, Grippo P, Parisi E. The heterogeneity of thymine methyl group origin in DNA pyrimidine isostichs of developing sea urchin embryos. Proc Natl Acad Sci U S A. 1967 May;57(5):1394–400. doi: 10.1073/pnas.57.5.1394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Holliday R, Pugh JE. DNA modification mechanisms and gene activity during development. Science. 1975 Jan 24;187(4173):226–32. [PubMed] [Google Scholar]
- 3.Riggs AD. X inactivation, differentiation, and DNA methylation. Cytogenet Cell Genet. 1975;14(1):9–25. doi: 10.1159/000130315. [DOI] [PubMed] [Google Scholar]
- 4.McGhee JD, Ginder GD. Specific DNA methylation sites in the vicinity of the chicken beta-globin genes. Nature. 1979 Aug 2;280(5721):419–20. doi: 10.1038/280419a0. [DOI] [PubMed] [Google Scholar]
- 5.Razin A, Riggs AD. DNA methylation and gene function. Science. 1980 Nov 7;210(4470):604–10. doi: 10.1126/science.6254144. [DOI] [PubMed] [Google Scholar]
- 6.Shen CK, Maniatis T. Tissue-specific DNA methylation in a cluster of rabbit beta-like globin genes. Proc Natl Acad Sci U S A. 1980 Nov;77(11):6634–8. doi: 10.1073/pnas.77.11.6634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.van der Ploeg LH, Flavell RA. DNA methylation in the human gamma delta beta-globin locus in erythroid and nonerythroid tissues. Cell. 1980 Apr;19(4):947–58. doi: 10.1016/0092-8674(80)90086-0. [DOI] [PubMed] [Google Scholar]
- 8.Jenuwein T, Allis CD. Translating the histone code. Science. 2001 Aug 10;293(5532):1074–80. doi: 10.1126/science.1063127. [DOI] [PubMed] [Google Scholar]
- 9.Berger SL. Histone modifications in transcriptional regulation. Curr Opin Genet Dev. 2002 Apr;12(2):142–8. doi: 10.1016/s0959-437x(02)00279-4. [DOI] [PubMed] [Google Scholar]
- 10.Britton LM, Gonzales-Cope M, Zee BM, Garcia BA. Breaking the histone code with quantitative mass spectrometry. Expert Rev Proteomics. 2011 Oct;8(5):631–43. doi: 10.1586/epr.11.47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Zentner GE, Henikoff S. Regulation of nucleosome dynamics by histone modifications. Nat Struct Mol Biol. 2013 Mar;20(3):259–66. doi: 10.1038/nsmb.2470. [DOI] [PubMed] [Google Scholar]
- 12.Sankaran VG, Xu J, Ragoczy T, Ippolito GC, Walkley CR, Maika SD, et al. Developmental and species-divergent globin switching are driven by BCL11A. Nature. 2009 Aug 27;460(7259):1093–7. doi: 10.1038/nature08243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Stamatoyannopoulos G. Prospects for developing a molecular cure for thalassemia. Hematology. 2005;10(Suppl 1):255–7. doi: 10.1080/10245330512331390627. [DOI] [PubMed] [Google Scholar]
- 14.Porcu S, Kitamura M, Witkowska E, Zhang Z, Mutero A, Lin C, et al. The human beta globin locus introduced by YAC transfer exhibits a specific and reproducible pattern of developmental regulation in transgenic mice. Blood. 1997 Dec 1;90(11):4602–9. [PubMed] [Google Scholar]
- 15.Pope SH, Fibach E, Sun J, Chin K, Rodgers GP. Two-phase liquid culture system models normal human adult erythropoiesis at the molecular level. Eur J Haematol. 2000 May;64(5):292–303. doi: 10.1034/j.1600-0609.2000.90032.x. [DOI] [PubMed] [Google Scholar]
- 16.Fibach E, Prus E. Differentiation of human erythroid cells in culture. Curr Protoc Immunol. 2005 Nov;Chapter 22 doi: 10.1002/0471142735.im22f07s69. Unit 22F.7. [DOI] [PubMed] [Google Scholar]
- 17.DeSimone J, Biel SI, Heller P. Stimulation of fetal hemoglobin synthesis in baboons by hemolysis and hypoxia. Proc Natl Acad Sci U S A. 1978 Jun;75(6):2937–40. doi: 10.1073/pnas.75.6.2937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Thein SL, Menzel S, Peng X, Best S, Jiang J, Close J, et al. Intergenic variants of HBS1L-MYB are responsible for a major quantitative trait locus on chromosome 6q23 influencing fetal hemoglobin levels in adults. Proc Natl Acad Sci U S A. 2007 Jul 3;104(27):11346–51. doi: 10.1073/pnas.0611393104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Sankaran VG, Menne TF, Xu J, Akie TE, Lettre G, Van Handel B, et al. Human fetal hemoglobin expression is regulated by the developmental stage-specific repressor BCL11A. Science. 2008 Dec 19;322(5909):1839–42. doi: 10.1126/science.1165409. [DOI] [PubMed] [Google Scholar]
- 20.Sankaran VG, Xu J, Byron R, Greisman HA, Fisher C, Weatherall DJ, et al. A functional element necessary for fetal hemoglobin silencing. N Engl J Med. 2011 Sep 1;365(9):807–14. doi: 10.1056/NEJMoa1103070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Yi Z, Cohen-Barak O, Hagiwara N, Kingsley PD, Fuchs DA, Erickson DT, et al. Sox6 directly silences epsilon globin expression in definitive erythropoiesis. PLoS Genet. 2006 Feb;2(2):e14. doi: 10.1371/journal.pgen.0020014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Xu J, Sankaran VG, Ni M, Menne TF, Puram RV, Kim W, et al. Transcriptional silencing of {gamma}-globin by BCL11A involves long-range interactions and cooperation with SOX6. Genes Dev. 2010 Apr 15;24(8):783–98. doi: 10.1101/gad.1897310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Miller IJ, Bieker JJ. A novel, erythroid cell-specific murine transcription factor that binds to the CACCC element and is related to the kruppel family of nuclear proteins. Mol Cell Biol. 1993 May;13(5):2776–86. doi: 10.1128/mcb.13.5.2776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Wijgerde M, Gribnau J, Trimborn T, Nuez B, Philipsen S, Grosveld F, et al. The role of EKLF in human beta-globin gene competition. Genes Dev. 1996 Nov 15;10(22):2894–902. doi: 10.1101/gad.10.22.2894. [DOI] [PubMed] [Google Scholar]
- 25.Zhou D, Pawlik KM, Ren J, Sun CW, Townes TM. Differential binding of erythroid krupple-like factor to embryonic/fetal globin gene promoters during development. J Biol Chem. 2006 Jun 9;281(23):16052–7. doi: 10.1074/jbc.M601182200. [DOI] [PubMed] [Google Scholar]
- 26.Borg J, Papadopoulos P, Georgitsi M, Gutierrez L, Grech G, Fanis P, et al. Haploinsufficiency for the erythroid transcription factor KLF1 causes hereditary persistence of fetal hemoglobin. Nat Genet. 2010 Sep;42(9):801–5. doi: 10.1038/ng.630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Zhou D, Liu K, Sun CW, Pawlik KM, Townes TM. KLF1 regulates BCL11A expression and gamma- to beta-globin gene switching. Nat Genet. 2010 Sep;42(9):742–4. doi: 10.1038/ng.637. [DOI] [PubMed] [Google Scholar]
- 28.Jiang J, Best S, Menzel S, Silver N, Lai MI, Surdulescu GL, et al. cMYB is involved in the regulation of fetal hemoglobin production in adults. Blood. 2006 Aug 1;108(3):1077–83. doi: 10.1182/blood-2006-01-008912. [DOI] [PubMed] [Google Scholar]
- 29.Lettre G, Sankaran VG, Bezerra MA, Araujo AS, Uda M, Sanna S, et al. DNA polymorphisms at the BCL11A, HBS1L-MYB, and beta-globin loci associate with fetal hemoglobin levels and pain crises in sickle cell disease. Proc Natl Acad Sci U S A. 2008 Aug 19;105(33):11869–74. doi: 10.1073/pnas.0804799105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Sankaran VG, Menne TF, Scepanovic D, Vergilio JA, Ji P, Kim J, et al. MicroRNA-15a and -16-1 act via MYB to elevate fetal hemoglobin expression in human trisomy 13. Proc Natl Acad Sci U S A. 2011 Jan 25;108(4):1519–24. doi: 10.1073/pnas.1018384108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Harju-Baker S, Costa FC, Fedosyuk H, Neades R, Peterson KR. Silencing of agamma-globin gene expression during adult definitive erythropoiesis mediated by GATA1-FOG1-Mi2 complex binding at the -566 GATA site. Mol Cell Biol. 2008 May;28(10):3101–13. doi: 10.1128/MCB.01858-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Miccio A, Wang Y, Hong W, Gregory GD, Wang H, Yu X, et al. NuRD mediates activating and repressive functions of GATA1 and FOG1 during blood development. EMBO J. 2010 Jan 20;29(2):442–56. doi: 10.1038/emboj.2009.336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Miccio A, Blobel GA. Role of the GATA1/FOG1/NuRD pathway in the expression of human beta-like globin genes. Mol Cell Biol. 2010 Jul;30(14):3460–70. doi: 10.1128/MCB.00001-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Costa FC, Fedosyuk H, Chazelle AM, Neades RY, Peterson KR. Mi2beta is required for gamma-globin gene silencing: Temporal assembly of a GATA1-FOG1-Mi2 repressor complex in beta-YAC transgenic mice. PLoS Genet. 2012;8(12):e1003155. doi: 10.1371/journal.pgen.1003155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Zhou W, Zhao Q, Sutton R, Cumming H, Wang X, Cerruti L, et al. The role of p22 NF-E4 in human globin gene switching. J Biol Chem. 2004 Jun 18;279(25):26227–32. doi: 10.1074/jbc.M402191200. [DOI] [PubMed] [Google Scholar]
- 36.Tanabe O, McPhee D, Kobayashi S, Shen Y, Brandt W, Jiang X, et al. Embryonic and fetal beta-globin gene repression by the orphan nuclear receptors, TR2 and TR4. EMBO J. 2007 May 2;26(9):2295–306. doi: 10.1038/sj.emboj.7601676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Campbell AD, Cui S, Shi L, Urbonya R, Mathias A, Bradley K, et al. Forced TR2/TR4 expression in sickle cell disease mice confers enhanced fetal hemoglobin synthesis and alleviated disease phenotypes. Proc Natl Acad Sci U S A. 2011 Nov 15;108(46):18808–13. doi: 10.1073/pnas.1104964108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Bank A. Regulation of human fetal hemoglobin: New players, new complexities. Blood. 2006 Jan 15;107(2):435–43. doi: 10.1182/blood-2005-05-2113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Sankaran VG, Xu J, Orkin SH. Advances in the understanding of haemoglobin switching. Br J Haematol. 2010 Apr;149(2):181–94. doi: 10.1111/j.1365-2141.2010.08105.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Tate PH, Bird AP. Effects of DNA methylation on DNA-binding proteins and gene expression. Curr Opin Genet Dev. 1993 Apr;3(2):226–31. doi: 10.1016/0959-437x(93)90027-m. [DOI] [PubMed] [Google Scholar]
- 41.Pikaart MJ, Recillas-Targa F, Felsenfeld G. Loss of transcriptional activity of a transgene is accompanied by DNA methylation and histone deacetylation and is prevented by insulators. Genes Dev. 1998 Sep 15;12(18):2852–62. doi: 10.1101/gad.12.18.2852. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.DeSimone J, Heller P, Hall L, Zwiers D. 5-azacytidine stimulates fetal hemoglobin synthesis in anemic baboons. Proc Natl Acad Sci U S A. 1982 Jul;79(0027-8424; 14):4428–31. doi: 10.1073/pnas.79.14.4428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Ley TJ, DeSimone J, Anagnou NP, Keller GH, Humphries RK, Turner PH, et al. 5-azacytidine selectively increases gamma-globin synthesis in a patient with beta+ thalassemia. N Engl J Med. 1982 Dec 9;307(24):1469–75. doi: 10.1056/NEJM198212093072401. [DOI] [PubMed] [Google Scholar]
- 44.Charache S, Dover G, Smith K, Talbot CC, Jr, Moyer M, Boyer S. Treatment of sickle cell anemia with 5-azacytidine results in increased fetal hemoglobin production and is associated with nonrandom hypomethylation of DNA around the gamma-delta-beta-globin gene complex. Proc Natl Acad Sci U S A. 1983 Aug;80(15):4842–6. doi: 10.1073/pnas.80.15.4842. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Lowrey CH, Nienhuis AW. Brief report: Treatment with azacitidine of patients with end-stage beta-thalassemia. N Engl J Med. 1993 Sep 16;329(12):845–8. doi: 10.1056/NEJM199309163291205. [DOI] [PubMed] [Google Scholar]
- 46.Letvin NL, Linch DC, Beardsley GP, McIntyre KW, Nathan DG. Augmentation of fetal-hemoglobin production in anemic monkeys by hydroxyurea. N Engl J Med. 1984 Apr 5;310(14):869–73. doi: 10.1056/NEJM198404053101401. [DOI] [PubMed] [Google Scholar]
- 47.Papayannopoulou T, Torrealba de Ron A, Veith R, Knitter G, Stamatoyannopoulos G. Arabinosylcytosine induces fetal hemoglobin in baboons by perturbing erythroid cell differentiation kinetics. Science. 1984 May 11;224(4649):617–9. doi: 10.1126/science.6200940. [DOI] [PubMed] [Google Scholar]
- 48.Platt OS, Orkin SH, Dover G, Beardsley GP, Miller B, Nathan DG. Hydroxyurea enhances fetal hemoglobin production in sickle cell anemia. J Clin Invest. 1984 Aug;74(2):652–6. doi: 10.1172/JCI111464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Torrealba-de Ron AT, Papayannopoulou T, Knapp MS, Fu MF, Knitter G, Stamatoyannopoulos G. Perturbations in the erythroid marrow progenitor cell pools may play a role in the augmentation of HbF by 5-azacytidine. Blood. 1984 Jan;63(1):201–10. [PubMed] [Google Scholar]
- 50.Mabaera R, Greene MR, Richardson CA, Conine SJ, Kozul CD, Lowrey CH. Neither DNA hypomethylation nor changes in the kinetics of erythroid differentiation explain 5-azacytidine's ability to induce human fetal hemoglobin. Blood. 2008 Jan 1;111(1):411–20. doi: 10.1182/blood-2007-06-093948. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Lavelle DE. The molecular mechanism of fetal hemoglobin reactivation. Semin Hematol. 2004 Oct;41(4 Suppl 6):3–10. doi: 10.1053/j.seminhematol.2004.08.002. [DOI] [PubMed] [Google Scholar]
- 52.Goren A, Simchen G, Fibach E, Szabo PE, Tanimoto K, Chakalova L, et al. Fine tuning of globin gene expression by DNA methylation. PLoS One. 2006 Dec 20;1:e46. doi: 10.1371/journal.pone.0000046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Rupon JW, Wang SZ, Gaensler K, Lloyd J, Ginder GD. Methyl binding domain protein 2 mediates gamma-globin gene silencing in adult human betaYAC transgenic mice. Proc Natl Acad Sci U S A. 2006 Apr 25;103(17):6617–22. doi: 10.1073/pnas.0509322103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Fathallah H, Weinberg RS, Galperin Y, Sutton M, Atweh GF. Role of epigenetic modifications in normal globin gene regulation and butyrate-mediated induction of fetal hemoglobin. Blood. 2007 Nov 1;110(9):3391–7. doi: 10.1182/blood-2007-02-076091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Chin J, Singh M, Banzon V, Vaitkus K, Ibanez V, Kouznetsova T, et al. Transcriptional activation of the gamma-globin gene in baboons treated with decitabine and in cultured erythroid progenitor cells involves different mechanisms. Exp Hematol. 2009 Oct;37(10):1131–42. doi: 10.1016/j.exphem.2009.06.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Banzon V, Ibanez V, Vaitkus K, Ruiz MA, Peterson K, DeSimone J, et al. siDNMT1 increases gamma-globin expression in chemical inducer of dimerization (CID)-dependent mouse betaYAC bone marrow cells and in baboon erythroid progenitor cell cultures. Exp Hematol. 2011 Jan;39(1):26, 36.e1. doi: 10.1016/j.exphem.2010.10.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Olivieri NF, Saunthararajah Y, Thayalasuthan V, Kwiatkowski J, Ware RE, Kuypers FA, et al. A pilot study of subcutaneous decitabine in beta-thalassemia intermedia. Blood. 2011 Sep 8;118(10):2708–11. doi: 10.1182/blood-2011-03-341909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Hendrich B, Bird A. Identification and characterization of a family of mammalian methyl-CpG binding proteins. Mol Cell Biol. 1998 Nov;18(11):6538–47. doi: 10.1128/mcb.18.11.6538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Kransdorf EP, Wang SZ, Zhu SZ, Langston TB, Rupon JW, Ginder GD. MBD2 is a critical component of a methyl cytosine-binding protein complex isolated from primary erythroid cells. Blood. 2006 Oct 15;108(8):2836–45. doi: 10.1182/blood-2006-04-016394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Gnanapragasam MN, Scarsdale JN, Amaya ML, Webb HD, Desai MA, Walavalkar NM, et al. p66Alpha-MBD2 coiled-coil interaction and recruitment of mi-2 are critical for globin gene silencing by the MBD2-NuRD complex. Proc Natl Acad Sci U S A. 2011 May 3;108(18):7487–92. doi: 10.1073/pnas.1015341108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Hendrich B, Guy J, Ramsahoye B, Wilson VA, Bird A. Closely related proteins MBD2 and MBD3 play distinctive but interacting roles in mouse development. Genes Dev. 2001 Mar 15;15(6):710–23. doi: 10.1101/gad.194101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Ng HH, Zhang Y, Hendrich B, Johnson CA, Turner BM, Erdjument-Bromage H, et al. MBD2 is a transcriptional repressor belonging to the MeCP1 histone deacetylase complex. Nat Genet. 1999 Sep;23(1):58–61. doi: 10.1038/12659. [DOI] [PubMed] [Google Scholar]
- 63.Feng Q, Zhang Y. The MeCP1 complex represses transcription through preferential binding, remodeling, and deacetylating methylated nucleosomes. Genes Dev. 2001 Apr 1;15(7):827–32. doi: 10.1101/gad.876201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Cramer JM, Scarsdale JN, Walavalkar NM, Buchwald WA, Ginder GD, Williams DC., Jr Probing the dynamic distribution of bound states for methylcytosine-binding domains on DNA. J Biol Chem. 2014 Jan 17;289(3):1294–302. doi: 10.1074/jbc.M113.512236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Le Guezennec X, Vermeulen M, Brinkman AB, Hoeijmakers WA, Cohen A, Lasonder E, et al. MBD2/NuRD and MBD3/NuRD, two distinct complexes with different biochemical and functional properties. Mol Cell Biol. 2006 Feb;26(3):843–51. doi: 10.1128/MCB.26.3.843-851.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Rank G, Cerruti L, Simpson RJ, Moritz RL, Jane SM, Zhao Q. Identification of a PRMT5-dependent repressor complex linked to silencing of human fetal globin gene expression. Blood. 2010 Sep 2;116(9):1585–92. doi: 10.1182/blood-2009-10-251116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Amaya M, Desai M, Gnanapragasam MN, Wang SZ, Zu Zhu S, Williams DC, Jr, et al. Mi2beta-mediated silencing of the fetal gamma-globin gene in adult erythroid cells. Blood. 2013 Apr 25;121(17):3493–501. doi: 10.1182/blood-2012-11-466227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Allfrey VG, Faulkner R, Mirsky AE. Acetylation and methylation of histones and their possible role in the regulation of rna synthesis. Proc Natl Acad Sci U S A. 1964 May;51:786–94. doi: 10.1073/pnas.51.5.786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Vidali G, Boffa LC, Bradbury EM, Allfrey VG. Butyrate suppression of histone deacetylation leads to accumulation of multiacetylated forms of histones H3 and H4 and increased DNase I sensitivity of the associated DNA sequences. Proc Natl Acad Sci U S A. 1978 May;75(5):2239–43. doi: 10.1073/pnas.75.5.2239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Marmorstein R. Structure and function of histone acetyltransferases. Cell Mol Life Sci. 2001 May;58(5-6):693–703. doi: 10.1007/PL00000893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Ginder GD, Whitters MJ, Pohlman JK. Activation of a chicken embryonic globin gene in adult erythroid cells by 5-azacytidine and sodium butyrate. Proc Natl Acad Sci U S A. 1984 Jul;81(13):3954–8. doi: 10.1073/pnas.81.13.3954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Perrine SP, Rudolph A, Faller DV, Roman C, Cohen RA, Chen SJ, et al. Butyrate infusions in the ovine fetus delay the biologic clock for globin gene switching. Proc Natl Acad Sci U S A. 1988 Nov;85(22):8540–2. doi: 10.1073/pnas.85.22.8540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Boosalis MS, Bandyopadhyay R, Bresnick EH, Pace BS, Van DeMark K, Zhang B, et al. Short-chain fatty acid derivatives stimulate cell proliferation and induce STAT-5 activation. Blood. 2001 May 15;97(10):3259–67. doi: 10.1182/blood.v97.10.3259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Witt O, Monkemeyer S, Ronndahl G, Erdlenbruch B, Reinhardt D, Kanbach K, et al. Induction of fetal hemoglobin expression by the histone deacetylase inhibitor apicidin. Blood. 2003 Mar 1;101(5):2001–7. doi: 10.1182/blood-2002-08-2617. [DOI] [PubMed] [Google Scholar]
- 75.Sangerman J, Lee MS, Yao X, Oteng E, Hsiao CH, Li W, et al. Mechanism for fetal hemoglobin induction by histone deacetylase inhibitors involves gamma-globin activation by CREB1 and ATF-2. Blood. 2006 Nov 15;108(10):3590–9. doi: 10.1182/blood-2006-01-023713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Perrine SP, Ginder GD, Faller DV, Dover GH, Ikuta T, Witkowska HE, et al. A short-term trial of butyrate to stimulate fetal-globin-gene expression in the beta-globin disorders. N Engl J Med. 1993 Jan 14;328(2):81–6. doi: 10.1056/NEJM199301143280202. [DOI] [PubMed] [Google Scholar]
- 77.Perrine SP, Olivieri NF, Faller DV, Vichinsky EP, Dover GJ, Ginder GD. Butyrate derivatives. new agents for stimulating fetal globin production in the beta-globin disorders. Am J Pediatr Hematol Oncol. 1994 Feb;16(1):67–71. [PubMed] [Google Scholar]
- 78.Bradner JE, Mak R, Tanguturi SK, Mazitschek R, Haggarty SJ, Ross K, et al. Chemical genetic strategy identifies histone deacetylase 1 (HDAC1) and HDAC2 as therapeutic targets in sickle cell disease. Proc Natl Acad Sci U S A. 2010 Jul 13;107(28):12617–22. doi: 10.1073/pnas.1006774107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Lachner M, Jenuwein T. The many faces of histone lysine methylation. Curr Opin Cell Biol. 2002 Jun;14(3):286–98. doi: 10.1016/s0955-0674(02)00335-6. [DOI] [PubMed] [Google Scholar]
- 80.Wozniak RJ, Bresnick EH. Epigenetic control of complex loci during erythropoiesis. Curr Top Dev Biol. 2008;82:55–83. doi: 10.1016/S0070-2153(07)00003-8. [DOI] [PubMed] [Google Scholar]
- 81.Voigt P, Tee WW, Reinberg D. A double take on bivalent promoters. Genes Dev. 2013 Jun 15;27(12):1318–38. doi: 10.1101/gad.219626.113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Zhao Q, Rank G, Tan YT, Li H, Moritz RL, Simpson RJ, et al. PRMT5-mediated methylation of histone H4R3 recruits DNMT3A, coupling histone and DNA methylation in gene silencing. Nat Struct Mol Biol. 2009 Mar;16(3):304–11. doi: 10.1038/nsmb.1568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.He Y, Rank G, Zhang M, Ju J, Liu R, Xu Z, et al. Induction of human fetal hemoglobin expression by adenosine-2′,3′-dialdehyde. J Transl Med. 2013 Jan 14;11:14. doi: 10.1186/1479-5876-11-14. 5876-11-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.van Dijk TB, Gillemans N, Pourfarzad F, van Lom K, von Lindern M, Grosveld F, et al. Fetal globin expression is regulated by friend of Prmt1. Blood. 2010 Nov 18;116(20):4349–52. doi: 10.1182/blood-2010-03-274399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Li X, Hu X, Patel B, Zhou Z, Liang S, Ybarra R, et al. H4R3 methylation facilitates beta-globin transcription by regulating histone acetyltransferase binding and H3 acetylation. Blood. 2010 Mar 11;115(10):2028–37. doi: 10.1182/blood-2009-07-236059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Xu J, Bauer DE, Kerenyi MA, Vo TD, Hou S, Hsu YJ, et al. Corepressor-dependent silencing of fetal hemoglobin expression by BCL11A. Proc Natl Acad Sci U S A. 2013 Apr 16;110(16):6518–23. doi: 10.1073/pnas.1303976110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Cui S, Kolodziej KE, Obara N, Amaral-Psarris A, Demmers J, Shi L, et al. Nuclear receptors TR2 and TR4 recruit multiple epigenetic transcriptional corepressors that associate specifically with the embryonic beta-type globin promoters in differentiated adult erythroid cells. Mol Cell Biol. 2011 Aug;31(16):3298–311. doi: 10.1128/MCB.05310-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Shi L, Cui S, Engel JD, Tanabe O. Lysine-specific demethylase 1 is a therapeutic target for fetal hemoglobin induction. Nat Med. 2013 Mar;19(3):291–4. doi: 10.1038/nm.3101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Fuks F. DNA methylation and histone modifications: Teaming up to silence genes. Curr Opin Genet Dev. 2005 Oct;15(5):490–5. doi: 10.1016/j.gde.2005.08.002. [DOI] [PubMed] [Google Scholar]
- 90.Johnson LM, Bostick M, Zhang X, Kraft E, Henderson I, Callis J, et al. The SRA methyl-cytosine-binding domain links DNA and histone methylation. Curr Biol. 2007 Feb 20;17(4):379–84. doi: 10.1016/j.cub.2007.01.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Sarraf SA, Stancheva I. Methyl-CpG binding protein MBD1 couples histone H3 methylation at lysine 9 by SETDB1 to DNA replication and chromatin assembly. Mol Cell. 2004 Aug 27;15(4):595–605. doi: 10.1016/j.molcel.2004.06.043. [DOI] [PubMed] [Google Scholar]
- 92.Dodge JE, Kang YK, Beppu H, Lei H, Li E. Histone H3-K9 methyltransferase ESET is essential for early development. Mol Cell Biol. 2004 Mar;24(6):2478–86. doi: 10.1128/MCB.24.6.2478-2486.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Mutskov VJ, Farrell CM, Wade PA, Wolffe AP, Felsenfeld G. The barrier function of an insulator couples high histone acetylation levels with specific protection of promoter DNA from methylation. Genes Dev. 2002 Jun 15;16(12):1540–54. doi: 10.1101/gad.988502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Castel SE, Martienssen RA. RNA interference in the nucleus: Roles for small RNAs in transcription, epigenetics and beyond. Nat Rev Genet. 2013 Feb;14(2):100–12. doi: 10.1038/nrg3355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Lee YT, de Vasconcellos JF, Yuan J, Byrnes C, Noh SJ, Meier ER, et al. LIN28B-mediated expression of fetal hemoglobin and production of fetal-like erythrocytes from adult human erythroblasts ex vivo. Blood. 2013 Aug 8;122(6):1034–41. doi: 10.1182/blood-2012-12-472308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Lulli V, Romania P, Morsilli O, Cianciulli P, Gabbianelli M, Testa U, et al. MicroRNA-486-3p regulates gamma-globin expression in human erythroid cells by directly modulating BCL11A. PLoS One. 2013 Apr 4;8(4):e60436. doi: 10.1371/journal.pone.0060436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Atweh G, Fathallah H. Pharmacologic induction of fetal hemoglobin production. Hematol Oncol Clin North Am. 2010 Dec;24(6):1131–44. doi: 10.1016/j.hoc.2010.08.001. [DOI] [PubMed] [Google Scholar]
- 98.Fucharoen S, Inati A, Siritanaratku N, Thein SL, Wargin WC, Koussa S, et al. A randomized phase I/II trial of HQK-1001, an oral fetal globin gene inducer, in beta-thalassaemia intermedia and HbE/beta-thalassaemia. Br J Haematol. 2013 May;161(4):587–93. doi: 10.1111/bjh.12304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Moellering RE, Cornejo M, Davis TN, Del Bianco C, Aster JC, Blacklow SC, et al. Direct inhibition of the NOTCH transcription factor complex. Nature. 2009 Nov 12;462(7270):182–8. doi: 10.1038/nature08543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Cerchietti LC, Ghetu AF, Zhu X, Da Silva GF, Zhong S, Matthews M, et al. A small-molecule inhibitor of BCL6 kills DLBCL cells in vitro and in vivo. Cancer Cell. 2010 Apr 13;17(4):400–11. doi: 10.1016/j.ccr.2009.12.050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.LaBelle JL, Katz SG, Bird GH, Gavathiotis E, Stewart ML, Lawrence C, et al. A stapled BIM peptide overcomes apoptotic resistance in hematologic cancers. J Clin Invest. 2012 Jun 1;122(6):2018–31. doi: 10.1172/JCI46231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Kronke J, Udeshi ND, Narla A, Grauman P, Hurst SN, McConkey M, et al. Lenalidomide causes selective degradation of IKZF1 and IKZF3 in multiple myeloma cells. Science. 2014 Jan 17;343(6168):301–5. doi: 10.1126/science.1244851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Lu G, Middleton RE, Sun H, Naniong M, Ott CJ, Mitsiades CS, et al. The myeloma drug lenalidomide promotes the cereblon-dependent destruction of ikaros proteins. Science. 2014 Jan 17;343(6168):305–9. doi: 10.1126/science.1244917. [DOI] [PMC free article] [PubMed] [Google Scholar]