Abstract
Background
Plasmodium ovale is one of the causative agents of human malaria. Plasmodium ovale infection has long been thought to be non-fatal. Due to its lower morbidity, P. ovale receives little attention in malaria research.
Methods
Two Malaysians went to Nigeria for two weeks. After returning to Malaysia, they fell sick and were admitted to different hospitals. Plasmodium ovale parasites were identified from blood smears of these patients. The species identification was further confirmed with nested PCR. One of them was successfully treated with no incident of relapse within 12-month medical follow-up. The other patient came down with malaria-induced respiratory complication during the course of treatment. Although parasites were cleared off the circulation, the patient’s condition worsened. He succumbed to multiple complications including acute respiratory distress syndrome and acute renal failure.
Results
Sequencing of the malaria parasite DNA from both cases, followed by multiple sequence alignment and phylogenetic tree construction suggested that the causative agent for both malaria cases was P. ovale curtisi.
Discussion
In this report, the differences between both cases were discussed, and the potential capability of P. ovale in causing severe complications and death as seen in this case report was highlighted.
Conclusion
Plasmodium ovale is potentially capable of causing severe complications, if not death. Complete travel and clinical history of malaria patient are vital for successful diagnoses and treatment. Monitoring of respiratory and renal function of malaria patients, regardless of the species of malaria parasites involved is crucial during the course of hospital admission.
Keywords: Plasmodium ovale curtisi, Imported malaria, Acute respiratory distress syndrome, Acute renal failure, Death
Background
Acute respiratory distress syndrome (ARDS) is one of the severe complications of malaria [1]. ARDS in falciparum malaria has been intensively studied [2-4]. However, ARDS is not restricted solely to Plasmodium falciparum infection. This potentially grave complication has also been reported in malaria caused by Plasmodium vivax, Plasmodium malariae, Plasmodium knowlesi and Plasmodium ovale[5-12]. Due to its limited geographical distribution [13], as well as the much lower morbidity [14], P. ovale has been overshadowed by other human malaria parasites in the field of medicine and medical research. Nevertheless, recent studies have shown that ovale malaria is caused by two genetically distinct subspecies, P. ovale curtisi and P. ovale wallikeri[15-18].
In this report, two cases of P. ovale infection acquired from the same location were presented. Both cases ended with different outcome, and two interesting turning points were ARDS complication and acute renal failure.
Methods
Case presentation
Two Malaysian acquaintances (patients A and B) went to Victoria Island, Nigeria together for a two-week working trip. Lariam® (mefloquine) was used as anti-malarial prophylaxis for the trip. They fell sick after returning to Malaysia and were admitted to different hospitals. Their cases are presented as follows:
Case A (Isolate MAL-2)
About two months after the trip to Nigeria, patient A (52-year-old Chinese male) was admitted to a hospital due to five consecutive days of fever with chills and rigours. He was jaundiced, anorexic and febrile with body temperature of 37.7°C upon admission. He had mild cough, blood pressure of 110/66 mm Hg, pulse rate of 98 beats per minute (BPM) with peak bilirubin level of 45 μmol/L and hepatosplenomegaly. His lung examination was normal. His urine was tea-coloured. Ultrasound study confirmed the findings of hepatosplenomegaly with signs of chronic cholecystitis and cholelithiasis. Initial haematological investigation showed that he was thrombocytopaenic (37,000/μl) with normal white blood cell (WBC) count (5,800 cells/μl) and haemoglobin level of 13.9 g/dL. He had not travelled to any other places after the trip to Nigeria. The patient had a past history of malaria for three times. The last episode of malaria was six months prior to present admission. However, the species of malaria parasites for the previous malaria episodes was not known. The patient also had an underlying condition of hypertension. Besides, he was a heavy alcohol consumer. Clinical findings on patient A upon admission are summarized in Table 1.
Table 1.
Summary of initial clinical findings on patient A and B upon admission
|
Test (unit) |
Patient A |
Patient B |
|---|---|---|
| {normal range} | (Isolate MAL-2) | (Isolate MAL-1) |
|
Blood Pressure (mmHg) {90/60 - 130/80} |
110/66 |
102/55 |
|
Pulse rate (BPM) {60 - 100} |
98 |
60 |
|
Parasitemia (%) |
0.10# |
0.18 |
|
Haemoglobin (g/dL) {male: 13.5 - 17.5} |
13.9 |
12.4 |
|
TWBC (× 103cells/μl) {4.5 - 11.0} |
5.8 |
3.1 |
|
Platelet (× 103/μl) {150.0 - 450.0} |
37.0 |
65.0 |
|
Serum creatinine (μmol/L) {60 - 110} |
82.0 |
107.0 |
|
Serum urea (mmol/L) {2.5 - 6.4} |
9.0 |
6.5 |
|
Total serum bilirubin (μmol/L) {<17} |
45.0 |
16.0 |
|
AST (IU/L) {15.0 - 37.0} |
47.0 |
47.0 |
|
ALT (IU/L) {30.0 - 65.0} |
29.0 |
39.0 |
| Random plasma glucose (mmol/L) {<11.1} | 6.4 | 9.5 |
Values in {} indicate the normal range of the respective event investigated, corresponding to the age and gender of patient.
TWBC = total white blood cell count; AST = aspartate aminotransferase; ALT = alanine aminotransferase.
Value marked with “#” was not obtained upon patient’s admission to the hospital.
Patient A was treated immediately for cholecystitis with intravenous (IV) ceftriaxone 2 g daily and IV metronidazole 500 mg thrice daily by the attending gastroenterologist. However, his fever and thrombocytopaenia persisted, and WBC count dropped progressively. On day 5 of admission, blood smears were prepared and examined under the microscope. “Plasmodium vivax-like” parasites were found with parasitaemia of 0.10%. Further microscopic examination by a referral diagnostic centre subsequently indicated that this was a mono-infection of P. ovale. This was confirmed with nested PCR technique using primers developed from the 18S ribosomal RNA (18S rRNA) gene as applied by previous reports [19-21], coupled with sequencing analysis using Basic Local Alignment Search Tool (BLAST) [22]. Meanwhile, bacteriological culture diagnoses from patient’s blood samples were negative.
He was treated with a course of six doses of Riamet® (artemether and lumefantrine), four tablets per dose, and primaquine for two weeks. Patient A responded well to the anti-malarial treatment clinically and biochemically. Patient’s parasitaemia dropped to 0.06% the following day. Malaria parasites were cleared in less than 48 hours after initiation of Riamet® treatment. He was discharged well on day 8 of hospitalization. He remained well without relapse of malaria throughout his medical follow-up of 12 months with the hospital.
Case B (Isolate MAL-1)
Around six months after the trip to Nigeria, patient B (59-year-old Chinese male) fell sick and went to a private hospital. He was then referred to a tertiary referral hospital. Upon admission to the referral hospital, he gave a history of intermittent fever with rigours, myalgia and nausea for ten days. His blood pressure upon admission was 102/55 mm Hg, with pulse rate of 60 BPM. Plasma glucose level was 9.5 mmol/L. Jaundice and hepatosplenomegaly were not detected. He was alert and conscious. His lung examination was normal. He made a one-day-trip to Kota Kinabalu, Sabah, three months before the admission. He had no known medical illness and no known history of acquiring malaria. Initial haematological investigation revealed that he was thrombocytopaenic (65,000/μl) with low WBC count (3,100 cells/μl) and haemoglobin level of 12.4 g/dL. Malaria parasites were detected in his blood, with parasitaemia of 0.18%. The species was identified as P. ovale, which was further ascertained with nested PCR as mentioned in the previous section. Clinical findings on patient B upon admission are summarized in Table 1, and more clinical details on patient B throughout his hospital stay is available in Table 2.
Table 2.
Clinical details on patient B (Isolate MAL-1) throughout his hospital stay
| Day | BP (mmHg) | Hb (g/dL) | Plt (×10 3 /μl) | TSB (μmol/L) | AST (IU/L) | ALT (IU/L) | SCr (μmol/L) | Blood bacteriological & fungal culture | Anti-malarials | Antibiotics | Additional notes |
|---|---|---|---|---|---|---|---|---|---|---|---|
|
1 |
102/55 |
12.4 |
65 |
16 |
47 |
39 |
100 |
N/A |
N/A |
N/A |
PR 60 BPM; SPO2 97% RA; loss of appetite; Lungs: clear |
|
2 |
92/52 |
N/A |
N/A |
N/A |
N/A |
N/A |
107 |
N/A |
Chloroquine |
N/A |
PR 70 BPM; Lungs: clear; |
| Primaquine |
Dengue IgM negative |
||||||||||
|
3 |
118/66 |
N/A |
107 |
13 |
88 |
71 |
112 |
N/A |
Chloroquine |
N/A |
PR 70 BPM; SPO2 98% RA; |
| Primaquine |
Lungs: minimal basal crepitations |
||||||||||
|
4 |
106/60 |
10.5 |
120 |
13 |
50 |
90 |
101 |
Negative |
Artesunate |
Ceftriaxone |
PR 66 BPM; Haemoptysis, epistaxis, shortness of breath; CXR: bilateral haziness |
| Quinine | |||||||||||
|
5 |
115/56 |
11.2 |
170 |
8 |
56 |
62 |
114 |
N/A |
Artesunate |
Ceftriaxone |
Blood smear: negative for malaria parasites; Transferred to ICU |
|
6 |
104/50 |
9.4 |
183 |
6 |
58 |
43 |
139 |
Negative |
Artesunate |
Tazocin® |
Blood smear: negative for malaria |
|
7 |
95/56 |
10.3 |
197 |
9 |
46 |
42 |
215 |
Negative |
Artesunate |
Tazocin® |
Blood smear: negative for malaria |
|
8 |
102/70 |
9.4 |
178 |
8 |
88 |
43 |
291 |
|
Artesunate |
Tazocin® |
Blood smear: negative for malaria; respiratory acidosis |
| Primaquine | |||||||||||
|
9 |
143/64 |
9.0 |
184 |
7 |
49 |
39 |
297 |
Negative |
Artesunate |
Tazocin® |
Blood smear: negative for malaria |
|
10 |
170/60 |
8.5 |
199 |
10 |
54 |
48 |
316 |
N/A |
Artesunate |
Tazocin® |
Blood smear: negative for malaria |
|
11 |
165/68 |
8.1 |
231 |
8 |
99 |
81 |
301 |
N/A |
N/A |
Tazocin® |
Ferritin blood test: 2,118 ng/ml |
|
12 |
200/78 |
8.1 |
288 |
13 |
128 |
148 |
309 |
Negative |
N/A |
Vancomycin |
|
| Imipenem | |||||||||||
|
13 |
160/60 |
6.8 |
311 |
16 |
137 |
190 |
392 |
N/A |
N/A |
Vancomycin |
Hemolysis screening: negative; |
| Imipenem |
CXR: worsening |
||||||||||
|
14 |
170/63 |
9.1 |
265 |
13 |
47 |
106 |
408 |
N/A |
N/A |
Vancomycin |
|
| Imipenem | |||||||||||
|
15 |
135/51 |
9.5 |
270 |
22 |
65 |
74 |
472 |
Positive for Enterobacter cloacae |
N/A |
Vancomycin |
Rigours; acidosis (resp. & met.) |
| Imipenem |
ABG: pH 7.128; pCO2 59.5 mmHg; |
||||||||||
| SLED 4 hours | |||||||||||
|
16 |
111/45 |
9.1 |
225 |
19 |
43 |
51 |
413 |
N/A |
N/A |
Vancomycin |
CVVHD |
| Imipenem | |||||||||||
|
17 |
134/57 |
7.5 |
179 |
29 |
109 |
65 |
424 |
N/A |
N/A |
Vancomycin |
Seizures; SLED 8 hours |
| Meropenem | |||||||||||
|
18 |
135/55 |
7.3 |
178 |
30 |
38 |
40 |
320 |
N/A |
N/A |
Meropenem |
Gentle HD 4 hours |
|
19 |
130/56 |
9.0 |
204 |
34 |
40 |
30 |
404 |
N/A |
N/A |
Meropenem |
Gentle HD 4 hours |
|
20 |
147/61 |
8.6 |
207 |
25 |
43 |
26 |
403 |
N/A |
N/A |
Meropenem |
Severe resp. acidosis; met. acidosis; gentle HD 4 hours |
|
21 |
140/54 |
N/A |
N/A |
N/A |
N/A |
N/A |
427 |
N/A |
N/A |
Meropenem |
Gentle HD 3 hours |
|
22 |
102/48 |
9.3 |
166 |
39 |
296 |
119 |
401 |
N/A |
N/A |
Meropenem |
Worsening hypoxia; hypotensive; Gentle HD 6 hours |
| 23 | N/A | N/A | N/A | 22 | 136 | 109 | 314 | N/A | N/A | N/A | Asystole; death |
N/A = not available; BP = blood pressure; Hb = haemoglobin; Plt = platelet; TSB = total serum bilirubin; AST = aspartate aminotransferase; ALT = alanine aminotransferase; SCr = serum creatinine; BP = blood pressure; BPM = beats per minute; PR = pulse rate; SPO2 = blood oxygen saturation; RA = room air; IgM = immunoglobulin M; CXR = chest X-ray; ICU = intensive care unit; resp. = respiratory; met. = metabolic; ABG = arterial blood gas; pCO2 = partial pressure of carbon dioxide; SLED = sustained low efficiency dialysis; CVVHD = continuous veno-venous haemodialysis; HD = haemodialysis.
Anti-malaria therapy course of chloroquine (chloroquine phosphate 150 mg base) and primaquine (30 mg) was started on patient B. However, he was still febrile (38.4°C) 24 hours later. On day 3 of the admission, patient B developed loose stools and lung examination revealed fine basal crepitations. Blood smear examination showed that the malaria parasites were not cleared. In the morning of day 4, he complained of feeling breathless and lethargic. His body temperature surged to 39.2°C. With presence of basal crepitations, an initial diagnosis of pneumonia was made and IV ceftriaxone was added into the course of treatment.
However, later that day in the afternoon, the patient developed worsening dyspnea, haemoptysis, and subsequently epistaxis. Chest X-ray examination showed bilateral haziness up to the upper zone, which was suggestive of pulmonary haemorrhage. Haematological investigation showed that his platelet count was 120,000/μl and the malaria parasite load was reduced to 0.03%. Despite the lowering of parasitaemia, patient B progressed into respiratory failure. He was intubated and ventilated. His anti-malarial treatment was changed to IV quinine 850 mg (1 dose) and subsequently to IV artesunate 160 mg (for 7 days). Furosemide was given to the patient for presumed pulmonary oedema.
On day 5 of the admission, malaria parasites were completely cleared. However, patient was still febrile with temperature of 40.8°C. He was transferred to intensive care unit. On day 6, patient B was oliguric. Increased level of creatinine and worsening of respiratory acidosis were noted. The patient had developed acute kidney injury (AKI) secondary to overwhelming sepsis. Hypotensive episodes were encountered. Therefore inotropic support was initiated. His antibiotic regime was changed to IV Tacocin® (tazobactam and piperacilin).
On day 12, patient B was still febrile with little improvement on his lung function. He was still dependent on the ventilator. IV Tacocin® was replaced by IV vancomycin and imipenem empirical treatment. All the five sets of bacteriological blood cultures and one set of fungal culture requested earlier on different days came back as negative. Nevertheless, dialysis was started on day 15 due to oliguric AKI with worsening acidosis (mixed respiratory and metabolic). Dialysis was performed on daily basis till the end of his life.
On day 17, bacteriological culture from patient’s blood sample that was collected on day 15 was positive for Enterobacter cloacae, which was sensitive to carbapenems. Hence, the imipenem therapy was continued. However, due to epileptic seizures suffered by the patient on day 17, the antibiotic regime was subsequently replaced with meropenem. A computerized tomography (CT) scanning on the patient’s brain showed no abnormality. Another thorax CT scanning showed extensive bilateral lung consolidation and loculated pleural effusion. His fever persisted, and he was on prolonged ventilation with difficulty in weaning off. His condition continued to deteriorate. He went into recalcitrant atrial fibrillation on day 22 and required tripleinotropic support. Further blood bacteriological and fungal cultures were all negative. On the 23rd day of hospital admission, he went into asystole and succumbed to P. ovale infection with ARDS, acute renal failure, metabolic acidosis, and nosocomial sepsis.
Consent
Consent was granted by patient/ patient’s family for the publication of these case reports.
Post-clinical analysis and interpretation
Plasmodium ovale is not found locally in Malaysia. Alignment of short segments from 18S rRNA gene sequences of these two cases, i.e. isolate MAL-1 (GenBank accession number KF192072) and isolate MAL-2 (GenBank accession number KF192073) with 18S rRNA gene sequences of other isolates of P. ovale spp. that were available in GenBank (accession number L48986, JF894403, JF894405, GQ183051, JF894407, JF894410, M19172) were conducted. The multiple sequence alignment showed that the aetiological agents of the two cases in this case report were P. ovale curtisi (Figure 1). The clustering of these two isolates with P. ovale curtisi can be visualized clearly via a simple phylogenetic tree constructed using neighbour-joining method (bootstrap = 1000) as described previously [23] (Figure 2).
Figure 1.

Multiple sequence alignment of short segments from 18S rRNA gene sequences of Plasmodium ovale spp. isolates. The isolate MAL-1 (GenBank accession number KF192072) and isolate MAL-2 (GenBank accession number KF192073) in this report were indicated as P. ovale curtisi, as shown by the high nucleotide sequence similarity shared between these two isolates and gene sequences of other P. ovale curtisi isolates (GenBank accession number JF894403, JF894405, L48986) that were available in GenBank. Gene sequences of P. ovale wallikeri from GenBank (GenBank accession number JF894407, JF894410) were used in this multiple sequence alignment as well. Position of nucleotides is based on sequences of isolate MAL-1.
Figure 2.
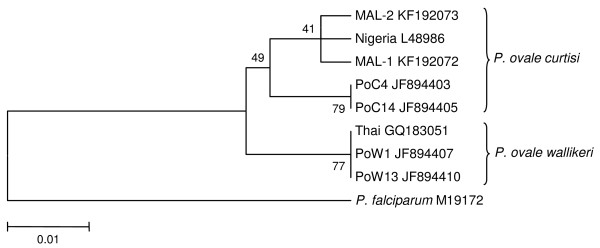
Phylogenetic tree based on a short segment of 18S rRNA gene sequences of Plasmodium spp. isolates. Isolate MAL-1 (accession number KF192072) and isolate MAL-2 (accession number KF192073) in this report clustered with P. ovale curtisi isolates, clearly indicating that these two cases were caused by P. ovale curtisi. The tree is constructed using the Neighbor-Joining method (bootstrap = 1000). GenBank accession number is given after each isolate’s name. The 18S rRNA gene sequences of P.falciparum isolate (GenBank accession number M19172) was used as outgroup.
A case of ovale malaria imported from Nigeria was previously reported in Malaysia [24], and these are another two imported cases of P. ovale infection. Unlike the previous case report, which involved a Nigerian student (imported patient) who showed no complications [24], both cases reported here involved Malaysians that acquired malaria from a trip to Nigeria (imported infection), and one showed severe complications that were ultimately fatal. Interestingly, both patients had taken anti-malarial prophylaxis during their trip to Nigeria. Nevertheless, they still contracted malaria. This may be due to usage of the prophylactic drugs without properly following the instructions or low compliance during the trip. Another reason for failed protective effect of anti-malaria chemoprophylaxis against P. ovale infection is the ability of P. ovale to form hypnozoites, which can survive chemoprophylaxis [18].
In case A, there was a delay in malaria diagnosis. This case is a good example of clinical setting where malaria tends to be overlooked at the initial stage of medical investigation due to low level of suspicion. From the initial haematological investigation, all but one (platelet count) showed values within the normal range. In many hospital settings, especially those of urban areas and non-malaria endemic regions, such investigation outcome usually carries a thin possibility of instigating a blood film microscopic examination. Indeed, a case of traveller’s malaria that developed into severe complications due to delayed diagnosis was reported in Croatia recently [25]. The available reported malaria with delayed diagnoses may just be a tip of the iceberg; and such incident may happen more frequently than expected. Thus malaria should have been suspected if patient presented with fever, thrombocytopaenia, and showed history of travelling to malaria-endemic areas within the past one year. The long period between travel to malaria endemic country and disease onset may also contribute to delay in diagnosis, and subsequently the appropriate treatment. The late onset of symptoms due to long latency period provides challenges to clinical diagnoses upon admission. As mentioned previously, P. ovale spp. can form hypnozoites and result in long latency [18,26], which can be seen in the two cases presented here.
Patient B succumbed to the malaria infection and he did not have any medical problems prior to the illness. Patient A, who is a heavy drinker, was known to have hypertension even before the infection, survived. One possible explanation for this is the partial immunity gained by patient A from previous incidents of malaria. People who have past history of acquiring falciparum malaria and those staying in malaria-endemic regions are known to show less severe symptoms during subsequent malaria infections compared to people who are from non-malaria endemic regions (malaria-naïve) [27]. Indeed, such protective effect of acquired partial immunity via previous exposure to malaria was noted and mentioned in previous reports on falciparum and vivax malaria-related ARDS [5,28,29]. Furthermore, malaria related pulmonary complications are often seen in non-immune patients [30]. Patient A might be partially protected against severe complications due to previous exposure to malaria. Patient B did not have prior exposure to malaria, and therefore, he suffered and succumbed to the severe complications in his first attack of malaria infection. Even so, pathogenesis of ovale malaria is not intensively studied and protection against ovale malaria complications conferred by previous exposure has not been investigated. Therefore, more investigations are needed to validate this postulation.
One important note worth mentioning here is the trend of ARDS onset and development in this case. Similar to the situation of malaria-induced ARDS reported previously [10,31,32], patient B showed normal breathing without wheezing and crepitations upon admission, but his respiratory function deteriorated towards the grave complication of ARDS afterwards. Therefore, physicians should pay close attention to the respiratory condition of every admitted malaria case. Moreover, malaria is regarded as one of the important causes of ARDS [1].
In case B, nosocomial Enterobacter cloacae infection was diagnosed during the course of his hospital stay. Here, bacterial infection was ruled out as the primary cause of ARDS in patient B as the bacteriological culture showed positive results as late as day 15, whereas he showed symptoms of acute lung injury as early as day 4. Undoubtedly, the bacterial infection that occurred at such critical timing would worsen the health condition of the patient, which acted as the secondary contributing factor towards the fatal end for this case. Another point worth mentioning was the onset of the ARDS symptoms after the initiation of anti-malarial therapy, which was similar to many cases of malaria-associated ARDS reported previously [9,10,30-34]. The pulmonary injury seen in this case was likely to be a post-treatment related pulmonary inflammation. Nonetheless, a few case reports of malaria-induced ARDS with pre-treatment onset were described previously [5,35]. In addition, quinine was known to be capable of inducing pulmonary injury [34]. Hence, the quinine therapy that was briefly applied on the patient may contribute to the worsening of his lung injury. Nevertheless, quinine therapy was immediately replaced with artesunate within the same day.
Thrombocytopaenia was seen in both cases. Such phenomenon has been observed in patients infected with P. falciparum, P. vivax, and P. knowlesi[8,21,36-39]. Thrombocytopaenia may serve as a contributing factor, instead of a primary cause of malaria-related severe pulmonary injury. Indeed, the platelet reading of patient A (survived case) was lower than that of patient B (fatal case) upon admission. Other events that are believed to contribute towards acute pulmonary injuries include: host immunologic responses [34], rosetting phenomenon, which is found in all four species of human malaria parasites [40-44], and sequestration that is found in P. falciparum and to a lesser, but significant extent, P. vivax[45]. This event may account for the much higher prevalence of ARDS complications seen in falciparum malaria. Nonetheless, the pathophysiology of malaria-associated ARDS is not completely understood [1,10,30].
Another aspect of this fatal case of P. ovale infection that deserves attention is the acute renal failure. Acute renal failure is another potentially fatal complication in malaria [25,46,47]. However, such complication is reported mostly in falciparum malaria [47-49], the newly emerging knowlesi malaria [21,50], and occasionally, in a few cases of P. vivax and P. malariae infections [51,52]. A fatal case of vivax malaria with acute renal failure was reported recently [53]. This is by far the first reported case of P. ovale infection with acute renal injury. In case B, the patient suffered renal injury a few days before he came down with nosocomial sepsis. Therefore, the acute renal injury seen in this case was less likely to be caused by nosocomial sepsis. Nevertheless, the nosocomial sepsis might act as the secondary contributing factor towards the renal failure in this patient. Besides, the medications used on the patient such as vancomycin is potentially nephrotoxic [54], thus serving as another potential contributing factor that worsened the acute renal injury. Hence, such therapy was discontinued.
Apart from the differences in pathological development of both cases, the anti-malaria treatment regimes used in both cases were dissimilar as well. For patient A, Riamet® with primaquine was used whereas for patient B, chloroquine and primaquine were prescribed at the initial stage of treatment. Parasites were not detected from patient A’s blood smear in less than 48 hours after initiation of antimalarial treatment. On the other hand, complete parasite clearance could only be declared on patient B after switching the medication to quinine and artesunate. Consequently, the parasite clearance took as long as five days to accomplish. Parasite clearance was much faster in treatment strategy using the artemether-lumefantrine-primaquine combination than that of chloroquine-primaquine combination. Indeed, artemisinin-based combination therapy (ACT) has been proven to show high efficacy in treating falciparum malaria [55] and non-falciparum malaria [56]. The much slower parasite clearance by chloroquine-based treatment may be due to the stage-specific anti-malarial property of chloroquine. As shown by previous studies, the trophozoites of P. vivax, P. malariae and P. ovale are relatively insusceptible to chloroquine [57-59]. Therefore, ACT such as Riamet® can serve as an effective first line treatment even for non-falciparum malaria.
Severe ovale malaria is not commonly found. There are a few reports of P. ovale infection with ARDS [9-12], and one case of ovale malaria with splenic complication [60]. Fatal ovale malaria is even rarer. A fatal case of ovale malaria involving a young Moroccan soldier was reported recently [12]. Interestingly, this ovale malaria casualty also suffered ARDS complication. This recent case report and our report show that P. ovale is capable of causing severe complications and death. For that reason, patients with P. ovale infection should not be regarded as “ultimately benign” and taken lightly, particularly in the non-immune travellers. In view of this, travel history as well as past history of acquiring malaria in patients with fever must be recorded in detail to assist in the diagnosis and management of travellers’ malaria and severe malaria.
Conclusion
Two imported cases of P. ovale infections with different pathological progress were reported. Plasmodium ovale is potentially capable of causing severe complications, if not death. In view of the differences between these two cases, complete clinical history of malaria patient, especially the travel history and history of malaria exposure are vital for successful treatment. Monitoring of respiratory and renal function of malaria patients, regardless of the species of malaria parasites involved is important during the course of hospital admission. In addition, ACT such as Riamet® should be applied to malaria patients regardless of the species of aetiological agents for prompt and efficient treatment.
Competing interests
The authors declared that they have no competing interests.
Authors’ contributions
WCL, YLL, and RM collected, analysed and interpreted the data. LHT, AK and SFSO collected blood samples, conducted clinical diagnoses and treatment intervention. FWC conducted and processed molecular diagnoses. MYF constructed and analysed phylogenetic tree. WCL, YLL, LHT and RM arranged the data, conceptualized and prepared the manuscript. All authors read and approved the final manuscript.
Contributor Information
Yee-Ling Lau, Email: lauyeeling@um.edu.my.
Wenn-Chyau Lee, Email: leewc_88@hotmail.com.
Lian-Huat Tan, Email: hutan07@gmail.com.
Adeeba Kamarulzaman, Email: adeeba@um.edu.my.
Sharifah Faridah Syed Omar, Email: shfaridah@um.edu.my.
Mun-Yik Fong, Email: fongmy@um.edu.my.
Fei-Wen Cheong, Email: fwcheong18@hotmail.com.
Rohela Mahmud, Email: rohela@ummc.edu.my.
Acknowledgements
YLL, MYF, RM and FWC were supported by University of Malaya High Impact Research (HIR) Grant UM-MOHE (UM.C/625/1/HIR/MOHE/CHAN/14/3) from the Ministry of Higher Education, Malaysia. WCL was supported by University of Malaya Student Research Grant PV044/2012A. We would like to express our gratitude to the medical staff in University of Malaya Medical Centre and Sunway Medical Centre.
References
- Mohan A, Sharma SK, Bollineni S. Acute lung injury and acute respiratory distress syndrome in malaria. J Vector Borne Dis. 2008;45:179–193. [PubMed] [Google Scholar]
- Lucas R, Lou J, Morel DR, Ricou B, Suter PM, Grau GE. TNF receptors in the microvascular pathology of acute respiratory distress syndrome and cerebral malaria. J Leukoc Biol. 1997;61:551–558. doi: 10.1002/jlb.61.5.551. [DOI] [PubMed] [Google Scholar]
- Vásquez AM, Tobón A. Pathogenic mechanisms in Plasmodium falciparum malaria. Biomedica. 2012;32(Suppl 1):106–120. doi: 10.1590/S0120-41572012000500012. [DOI] [PubMed] [Google Scholar]
- Deroost K, Tyberghein A, Lays N, Noppen S, Schwarzer E, Vanstreels E, Komuta M, Prato M, Lin JW, Pamplona A, Janse CJ, Arese P, Roskams T, Daelemans D, Opdenakker G, Van den Steen PE. Hemozoin induces lung inflammation and correlates with malaria-associated acute respiratory distress syndrome. Am J Respir Cell Mol Biol. 2013;48:589–600. doi: 10.1165/rcmb.2012-0450OC. [DOI] [PubMed] [Google Scholar]
- Lomar AV, Vidal JE, Lomar FP, Barbas CV, de Matos GJ, Boulos M. Acute respiratory distress syndrome due to vivax malaria: case report and literature review. Braz J Infect Dis. 2005;9:425–430. doi: 10.1590/s1413-86702005000500011. [DOI] [PubMed] [Google Scholar]
- Agarwal R, Nath A, Gupta D. Noninvasive ventilation in Plasmodium vivax related ALI/ARDS. Intern Med. 2007;46:2007–2011. doi: 10.2169/internalmedicine.46.0401. [DOI] [PubMed] [Google Scholar]
- Lozano F, Leal M, Lissen E, Munoz J, Bautista A, Regordan C. [P. falciparum and P. malariae malaria complicated by pulmonary edema with disseminated intravascular coagulation] (in French) Presse Med. 1983;12:3004–3005. [PubMed] [Google Scholar]
- Daneshvar C, Davis TM, Cox-Singh J, Rafa’ee MZ, Zakaria SK, Divis PC, Singh B. Clinical and laboratory features of human Plasmodium knowlesi infection. Clin Infect Dis. 2009;49:852–860. doi: 10.1086/605439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee EY, Maguire JH. Acute pulmonary edema complicating ovale malaria. Clin Infect Dis. 1999;29:697–698. doi: 10.1086/598667. [DOI] [PubMed] [Google Scholar]
- Rojo-Marcus G, Cuadros-González J, Mesa-Latorre JM, Culebras-López AM, de Pablo-Sánchez R. Case report: acute respiratory distress syndrome in a case of Plasmodium ovale malaria. Am J Trop Med Hyg. 2008;79:391–393. [PubMed] [Google Scholar]
- Rozé B, Lambert Y, Gelin E, Geffroy F, Hutin P. [Plasmodium ovale malaria severity] (in French) Med Mal Infect. 2011;41:213–220. doi: 10.1016/j.medmal.2010.07.006. [DOI] [PubMed] [Google Scholar]
- Hachimi MA, Hatim EA, Moudden MK, Elkartouti A, Errami M, Louzi L, Hanafi SM, Mahmoudi A. [The acute respiratory distress syndrome in malaria: is it always the prerogative of Plasmodium falciparum?] (in French) Rev Pneumol Clin. 2013;S0761–8417(13):00071. doi: 10.1016/j.pneumo.2013.03.001. [DOI] [PubMed] [Google Scholar]
- Collins WE, Jeffery GM. Plasmodium ovale: parasite and disease. Clin Microbiol Rev. 2005;18:570–581. doi: 10.1128/CMR.18.3.570-581.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Snounou G, Pinheiro L, Antunes AM, Ferreira C, do Rosario VE. Non-immune patients in the Democratic Republic of São Tomé e Principe reveal a high level of transmission of P. ovale and P. vivax despite low frequency in immune patients. Acta Trop. 1998;70:197–203. doi: 10.1016/S0001-706X(98)00021-7. [DOI] [PubMed] [Google Scholar]
- Sutherland CJ, Tanomsing N, Nolder D, Oguike M, Jennison C, Pukrittayakamee S, Dolecek C, Hien TT, do Rosário VE, Arez AP, Pinto J, Michon P, Escalante AA, Nosten F, Burke M, Lee R, Blaze M, Dan Otto T, Barnwell JW, Pain A, Williams J, White NJ, Day NPJ, Snounou G, Lockhart PJ, Chiodini PL, Imwong M, Polley SD. Two nonrecombining sympatric forms of the human malaria parasite Plasmodium ovale occur globally. J Infect Dis. 2010;201:1544–1550. doi: 10.1086/652240. [DOI] [PubMed] [Google Scholar]
- Sutherland CJ, Polley SD. In: Genetics and Evolution of the Infectious Diseases. Tibayrenc M, editor. London: Elsevier; 2011. Genomic Insights into the Past, Current and Future Evolution of Human Parasites of the Genus Plasmodium; pp. 607–635. [Google Scholar]
- Calderaro A, Piccolo G, Gorrini C, Montecchini S, Rossi S, Medici MC, Chezzi C, Snounou G. A new real-time PCR for the detection of Plasmodium ovale wallikeri. PLoS One. 2012;7:e48033. doi: 10.1371/journal.pone.0048033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nolder D, Oguike MC, Maxwell-Scott H, Niyazi HA, Smith V, Chiodini PL, Sutherland CJ. An observational study of malaria in British travellers: Plasmodium ovale wallikeri and Plasmodium ovale curtisi differ significantly in the duration of latency. BMJ Open. 2013;3:e002711. doi: 10.1136/bmjopen-2013-002711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Singh B, Kim Sung L, Matusop A, Radhakrishnan A, Shamsul SS, Cox-Singh J, Thomas A, Conway DJ. A large focus of naturally acquired Plasmodium knowlesi infections in human being. Lancet. 2004;363:1017–1024. doi: 10.1016/S0140-6736(04)15836-4. [DOI] [PubMed] [Google Scholar]
- Cox-Singh J, Davis TM, Lee KS, Shamsul SS, Matusop A, Ratnam S, Rahman HA, Conway DJ, Singh B. Plasmodium knowlesi in humans is widely distributed and potentially life threatening. Clin Infect Dis. 2008;46:165–171. doi: 10.1086/524888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee WC, Chin PW, Lau YL, Chin LC, Fong MY, Yap CJ, Supramaniam RR, Mahmud R. Hyperparasitaemic human Plasmodium knowlesi infection with atypical morphology in peninsular Malaysia. Malar J. 2013;12:88. doi: 10.1186/1475-2875-12-88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Basic Local Alignment Search Tool (BLAST) [ http://blast.ncbi.nlm.nih.gov] [DOI] [PubMed]
- Tamura K, Dudley J, Nei M, Kumar S. MEGA 4: evolutionary genetics analysis (MEGA) software version 4.0. Mol Biol Evol. 2007;24:1596–1599. doi: 10.1093/molbev/msm092. [DOI] [PubMed] [Google Scholar]
- Lim YA, Mahmud R, Chew CH, Thiruventhiran T, Chua KH. Plasmodium ovale infection in Malaysia: first imported case. Malar J. 2010;9:272. doi: 10.1186/1475-2875-9-272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Troselj-Vukić B, Vuksanović-Mikulicić S, Sladoje-Martinović B, Milotić I, Slavuljica I. Unrecognized malaria and its consequences: a case report of severe malaria with acute renal failure. Coll Antropol. 2013;37:611–613. [PubMed] [Google Scholar]
- Miller MJ, Marcus DM, Cameron DG. Latent infections with Plasmodium ovale malaria. Canad Med Ass J. 1965;92:1241–1247. [PMC free article] [PubMed] [Google Scholar]
- Høgh B. Clinical and parasitological studies on immunity to Plasmodium falciparum malaria in children. Scand J Infect Dis Suppl. 1996;102:1–53. [PubMed] [Google Scholar]
- Tanios MA, Kogelman L, McGovern B, Hassoun PM. Acute respiratory distress syndrome complicating Plasmodium vivax malaria. Crit Care Med. 2001;29:665–667. doi: 10.1097/00003246-200103000-00037. [DOI] [PubMed] [Google Scholar]
- Sarkar S, Saha K, Das CS. Three cases of ARDS: an emerging complication of Plasmodium vivax malaria. Lung India. 2010;27:154–157. doi: 10.4103/0970-2113.68323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taylor WR, Cañon V, White NJ. Pulmonary manifestations of malaria: recognition and management. Treat Respir Med. 2006;5:419–428. doi: 10.2165/00151829-200605060-00007. [DOI] [PubMed] [Google Scholar]
- Anstey NM, Jacups SP, Cain T, Pearson T, Ziesing PJ, Fisher DA, Currie BJ, Marks PJ, Maguire GP. Pulmonary manifestations of uncomplicated falciparum and vivax malaria: cough, small airways obstruction, impaired gas transfer, and increased pulmonary phagocytic activity. J Infect Dis. 2002;185:1326–1334. doi: 10.1086/339885. [DOI] [PubMed] [Google Scholar]
- Price L, Planche T, Rayner C, Krishna S. Acute respiratory distress syndrome in Plasmodium vivax malaria: case report and review of the literature. Trans R Soc Trop Med Hyg. 2007;101:655–659. doi: 10.1016/j.trstmh.2007.02.014. [DOI] [PubMed] [Google Scholar]
- Martell RW, Kallenbach J, Zwi S. Pulmonary oedema in the falciparum malaria. Br Med J. 1979;1:1763–1764. doi: 10.1136/bmj.1.6180.1763-a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taylor WR, White NJ. Malaria and the lung. Clin Chest Med. 2002;23:457–468. doi: 10.1016/S0272-5231(02)00004-7. [DOI] [PubMed] [Google Scholar]
- Rahman AK, Sulaiman FN. Plasmodium vivax malaria presenting as acute respiratory distress syndrome: a case report. Trop Doct. 2013;43:83–85. doi: 10.1177/0049475513485733. [DOI] [PubMed] [Google Scholar]
- Kakar A, Bhoi S, Prakash V, Kakar S. Profound thrombocytopenia in Plasmodium vivax malaria. Diag Microbiol Infect Dis. 1999;35:234–244. doi: 10.1016/s0732-8893(99)00069-3. [DOI] [PubMed] [Google Scholar]
- Ansari S, Khoharo HK, Abro A, Akhund IA, Qureshi F. Thrombocytopenia in Plasmodium falciparum malaria. J Ayub Med Coll Abbottabad. 2009;21:145–147. [PubMed] [Google Scholar]
- Thapa R, Biswas B, Mallick D, Sardar S, Modak S. Childhood Plasmodium vivax malaria with severe thrombocytopenia and bleeding manifestations. J Pediatr Hematol Oncol. 2009;31:758–759. doi: 10.1097/MPH.0b013e3181b7eb12. [DOI] [PubMed] [Google Scholar]
- Lacerda MV, Mourão MP, Coelho HC, Santos JB. Thrombocytopenia in malaria: who cares? Mem Inst Oswaldo Cruz. 2011;106(Suppl 1):52–63. doi: 10.1590/s0074-02762011000900007. [DOI] [PubMed] [Google Scholar]
- David PH, Handunnetti SM, Leech JH, Gamage P, Mendis KN. Rosetting: a new cytoadherence property of malaria-infected erythrocytes. Am J Trop Med Hyg. 1988;38:289–297. doi: 10.4269/ajtmh.1988.38.289. [DOI] [PubMed] [Google Scholar]
- Handunnetti SM, David PH, Perera KL, Mendis KN. Uninfected erythrocytes form “rosettes” around Plasmodium falciparum infected erythrocytes. Am J Trop Med Hyg. 1989;40:115–118. doi: 10.4269/ajtmh.1989.40.115. [DOI] [PubMed] [Google Scholar]
- Udomsangpetch R, Thanikkul K, Pukrittayakamee S, White NJ. Rosette formation by Plasmodium vivax. Trans R Soc Trop Med Hyg. 1995;89:635–637. doi: 10.1016/0035-9203(95)90422-0. [DOI] [PubMed] [Google Scholar]
- Angus BJ, Thanikkul K, Silamut K, White NJ, Udomsangpetch R. Short report: rosette formation in P. ovale infection. Am J Trop Med Hyg. 1996;55:560–561. doi: 10.4269/ajtmh.1996.55.560. [DOI] [PubMed] [Google Scholar]
- Lowe BS, Mosobo M, Bull PC. All four species of human malaria parasites form rosettes. Trans R Soc Trop Med Hyg. 1998;92:526. doi: 10.1016/S0035-9203(98)90901-4. [DOI] [PubMed] [Google Scholar]
- Carvalho BO, Lopes SC, Mogueira PA, Orlandi PP, Bargieri DY, Blanco YC, Mamomi R, Leite JA, Rodrigues MM, Araújo MO, Russell B, Suwanarusk R, Snounou G, Rénia L, Costa FT. On the cytoadhesion of Plasmodium vivax-infected erythrocytes. J infect Dis. 2010;202:638–647. doi: 10.1086/654815. [DOI] [PubMed] [Google Scholar]
- Trampuz A, Jereb M, Muzlovic I, Prabhu RM. Clinical review: severe malaria. Crit Care. 2003;7:315–323. doi: 10.1186/cc2183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Das BS. Renal failure in malaria. J Vector Borne Dis. 2008;45:83–97. [PubMed] [Google Scholar]
- Eiam-Ong S, Sitprija V. Falciparum malaria and the kidney: a model of inflammation. Am J Kidney Dis. 1998;32:361–375. doi: 10.1053/ajkd.1998.v32.pm9740151. [DOI] [PubMed] [Google Scholar]
- Sinniah R, Lye W. Acute renal failure from myoglobinuria secondary to myositis from severe falciparum malaria. Am J Nephrol. 2000;20:339–343. doi: 10.1159/000013611. [DOI] [PubMed] [Google Scholar]
- Singh B, Daneshvar C. Human infections and detection of Plasmodium knowlesi. Clin Microbiol Rev. 2013;26:165–184. doi: 10.1128/CMR.00079-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prakash J, Singh AK, Kumar NS, Saxena RK. Acute renal failure in Plasmodium vivax malaria. J Assoc Physicians India. 2003;51:265–267. [PubMed] [Google Scholar]
- Hendrickse RG, Adeniyi A, Edington GM, Glasgow EF, White RH, Houba V. Quartan malarial nephrotic syndrome. Collaborative clinicopathological study in Nigerian children. Lancet. 1972;1:1143–1149. doi: 10.1016/s0140-6736(72)91373-6. [DOI] [PubMed] [Google Scholar]
- Patel MP, Kute VB, Gumber MR, Gera DN, Shah PR, Patel HV, Trivedi HL, Vanikar AV. An unusual case of Plasmodium vivax malaria monoinfection associated with crescentic glomerulonephritis: a need for vigilance. Parasitol Res. 2013;112:427–430. doi: 10.1007/s00436-012-3040-5. [DOI] [PubMed] [Google Scholar]
- Gupta A, Biyani M, Khaira A. Vancomycin nephrotoxicity: myths and facts. Neth J Med. 2011;69:379–383. [PubMed] [Google Scholar]
- Bhattarai A, Ali AS, Kachur SP, Mártensson A, Abbas AK, Khatib R, Al-mafazy A, Ramsan G, Rotliant G, Gerstenmaier JF, Molteni F, Abdulla S, Montgomery SM, Kaneko A, Björkman A. Impact of artemisinin-based combination therapy and insecticide-treated nets on malaria burden in Znazibar. PLoS Med. 2007;4:e309. doi: 10.1371/journal.pmed.0040309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mombo-Ngoma G, Kleine C, Basra A, Wűrbel H, Diop DA, Capan M, Adegnika AA, Kurth F, Mordműller B, Joanny F, Kremsner PG, Ramharter M, Bélard S. Prospective evaluation of artemether-lumefantrine for the treatment of non-falciparum and mixed-species malaria in Gabon. Malar J. 2012;11:120. doi: 10.1186/1475-2875-11-120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Russell B, Chalfein F, Prasetyorini B, Kenangalem E, Piera K, Suwanarusk R, Brockman A, Prayoga P, Sugiarto P, Cheng Q, Tjitra E, Anstey NM, Price RN. Determinants of in vitro drug susceptibility testing of Plasmodium vivax. Antimicrob Agents Chemother. 2008;52:1040–1045. doi: 10.1128/AAC.01334-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sharrock WW, Suwanarusk R, Lek-Uthai U, Edstein MD, Kosaisavee V, Travers T, Jaidee A, Sriprawat K, Price RN, Nosten F, Russell B. Plasmodium vivax trophozoites insensitive to chloroquine. Malar J. 2008;7:94. doi: 10.1186/1475-2875-7-94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Siswantoro H, Russell B, Ratcliff A, Prasetyorini B, Chalfein F, Marfurt J, Kenangalem E, Wuwung M, Piera KA, Ebsworth EP, Anstey NM, Tjitra E, Price RN. In vivo and in vitro efficacy of chloroquine against Plasmodium malariae and P. ovale in Papua, Indonesia. Antimicrob Agents Chemother. 2011;55:197–202. doi: 10.1128/AAC.01122-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cinquetti G, Banal F, Rondel C, Plancade D, de Saint Roman C, Adriamanantena D, Ragot C, Védy C, Graffin B. Splenic infarction during Plasmodium ovale acute malaria: first case reported. Malar J. 2010;9:288. doi: 10.1186/1475-2875-9-288. [DOI] [PMC free article] [PubMed] [Google Scholar]


