Exocytosis and endocytosis need to be tightly balanced to keep cell surface area constant. This is of particular relevance for presynaptic terminals, where endocytosis needs to compensate for the ongoing exocytosis of synaptic vesicle to ultimately guarantee reliable synaptic transmission. How the endocytotic machinery manages to balance exocytosis is poorly understood. In principle, several mechanisms coupling endo- to exocytosis are conceivable: The membrane and proteins of the fused vesicles could diffuse laterally in the plasma membrane and represent a trigger for the endocytotic machinery (Dittman & Ryan, 2009). Protein scaffolds at the release site seem to coordinate this process (Haucke et al. 2011). Thereby, endocytosis could be directly coupled to exocytosis (Fig.1A). There is also compelling evidence that presynaptic terminals rely, in addition, on more indirect ways to couple exo- and endocytosis. At various types of synapses an acceleration of endocytosis was observed upon increasing the intracellular calcium concentration (Wu et al. 2007). Using calcium, the trigger of exocytosis, to trigger endocytosis provides a straightforward strategy to couple exo- and endocytosis. It is an ongoing challenge to understand how the spatiotemporal inhomogeneous presynaptic calcium concentration triggers endocytosis. In principle, the increase of residual calcium concentration during synaptic activity (up to a few micromolar, ∼100 ms) provides a rather long-lasting signal from which the amount of exocytosis might be inferred (Fig.1B). In addition, the short but large increase in local calcium concentration near calcium channels (tens of micromolar, <1 ms) could provide a trigger for endocytosis (Fig.1C). The local calcium has the advantage that the same signal triggers both exo- and endocytosis. However, using local calcium to trigger endocytosis is complicated by the fact that endocytosis occurs presumably at different sites from exocytosis and on a much slower time scale. Nevertheless, the local calcium is the main trigger for endocytosis at the mature calyx of Held synapse, a large presynaptic terminal of the auditory system (Yamashita et al. 2010). Finally, it has been shown at the Drosophila neuromuscular junction (Kuromi et al. 2004) that specific types of calcium channels are functionally coupled to endocytosis (Fig.1D). Whether such a mechanism is also used by vertebrate central synapses was unclear up to now. In this issue of The Journal of Physiology, Midorikawa et al. (2014) provide convincing evidence that some calcium channels are rather coupled to endocytosis than exocytosis – at least in the immature calyx of Held synapse.
Figure 1. Triggering endocytosis.
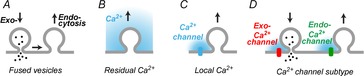
Endocytosis could be triggered by vesicle fusion (A), residual calcium concentration (B), or local calcium concentration near a calcium channel (C). The study by Midorikawa et al. (2014) shows that some mammalian central synapses use ‘endocytotic’ calcium channels (D; Endo-Ca2+ channels) to specifically trigger endocytosis.
The authors performed presynaptic capacitance measurements and analysed the impact of pharmacological calcium channel blockers on exocytosis, endocytosis and the calcium channel current at immature and mature rat calyces of Held. They found that blocking R-type calcium channels slows endocytosis in young rats (in particular the slow mode of endocytosis), while calcium current and exocytosis were only slightly reduced. To rule out that the slight reduction in exocytosis was causing the slowing of endocytosis, the authors perform the following experiment: They reduced the driving force of the calcium influx (by depolarization to +40 or +20 mV instead of 0 mV) and observed a reduction of the calcium current and the exocytosis while the time course of endocytosis was unchanged (Fig. 3 in Midorikawa et al. 2014). These convincing results confirm the specific effect of R-type calcium channels on endocytosis. Inhibition of R-type calcium channels still had an effect on the time course of endocytosis, even with high concentrations of the intracellular calcium buffer EGTA, suggesting that local microdomains of R-type calcium channels accelerate the slow mode of endocytosis. In addition, the authors found that the specific role of R-type calcium channels for endocytosis does not exist at the more mature calyx of Held, despite a remaining fraction of R-type calcium channels. Thus, the study by Midorikawa et al. (2014) introduces a new concept for mammalian central synapses, namely, the regulation of endocytosis by specialized ‘endocytotic’ calcium channels (cf. Fig.1D). At the immature calyx of Held, these ‘endocytotic’ calcium channels represent a specific molecular subtype of calcium channels, which enabled the authors to dissect ‘exocytotic’ from ‘endocytotic’ calcium channels with pharmacological blockers.
These new findings also raise interesting questions. Do different types of calcium channels contribute to a spatial segregation of exo- and endocytotic sites (Hosoi et al. 2009)? R-type calcium channels contribute relatively little to synaptic transmission at the calyx and have been suggested to be distant from release sites (Wu et al. 1999). For the immature calyx, Midorikawa et al. (2014) therefore suggest that R-type calcium channels are located close to endocytotic sites. The situation at the mature calyx is less clear. The data might indicate that during development endocytotic sites move spatially closer to exocytotic sites where P/Q-type channels are concentrated. However, remote endocytotic sites equipped with a subpopulation of P/Q-type calcium channels also seem consistent with the data. Finally, it will be interesting to investigate why immature calyces rely on R-type calcium channels for endocytosis while the remaining R-type calcium channels in mature calyces loose the ability to trigger endocytosis. This is of particular interest because endocytotic capacity increases during development (Renden & von Gersdorff, 2007).
In summary, the study by Midorikawa et al. (2014) provides a new concept of how endocytosis is regulated at mammalian central synapses. The authors show that some calcium channels are devoted to trigger endocytosis, but not exocytosis. It remains to be determined whether other synapses also utilize metabolically expensive calcium influx to trigger ‘only’ endocytosis.
Additional Information
Competing Interests
None declared
References
- Dittman J, Ryan TA. Molecular circuitry of endocytosis at nerve terminals. Annu Rev Cell Dev Biol. 2009;25:133–160. doi: 10.1146/annurev.cellbio.042308.113302. [DOI] [PubMed] [Google Scholar]
- Haucke V, Neher E, Sigrist SJ. Protein scaffolds in the coupling of synaptic exocytosis and endocytosis. Nat Rev Neurosci. 2011;12:127–138. doi: 10.1038/nrn2948. [DOI] [PubMed] [Google Scholar]
- Hosoi N, Holt M, Sakaba T. Calcium dependence of exo- and endocytotic coupling at a glutamatergic synapse. Neuron. 2009;63:216–229. doi: 10.1016/j.neuron.2009.06.010. [DOI] [PubMed] [Google Scholar]
- Kuromi H, Honda A, Kidokoro Y. Ca2+ influx through distinct routes controls exocytosis and endocytosis at Drosophila presynaptic terminals. Neuron. 2004;41:101–111. doi: 10.1016/s0896-6273(03)00815-8. [DOI] [PubMed] [Google Scholar]
- Midorikawa M, Okamoto Y, Sakaba T. Developmental changes in Ca channel subtypes regulating endocytosis at the calyx of Held. J Physiol. 2014;592:3495–3510. doi: 10.1113/jphysiol.2014.273243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Renden R, von Gersdorff H. Synaptic vesicle endocytosis at a CNS nerve terminal: faster kinetics at physiological temperatures and increased endocytotic capacity during maturation. J Neurophysiol. 2007;98:3349–3359. doi: 10.1152/jn.00898.2007. [DOI] [PubMed] [Google Scholar]
- Wu LG, Ryan TA, Lagnado L. Modes of vesicle retrieval at ribbon synapses, calyx-type synapses, and small central synapses. J Neurosci. 2007;27:11793–11802. doi: 10.1523/JNEUROSCI.3471-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu LG, Westenbroek RE, Borst JG, Catterall WA, Sakmann B. Calcium channel types with distinct presynaptic localization couple differentially to transmitter release in single calyx-type synapses. J Neurosci. 1999;19:726–736. doi: 10.1523/JNEUROSCI.19-02-00726.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamashita T, Eguchi K, Saitoh N, von Gersdorff H, Takahashi T. Developmental shift to a mechanism of synaptic vesicle endocytosis requiring nanodomain Ca2+ Nat Neurosci. 2010;13:838–844. doi: 10.1038/nn.2576. [DOI] [PMC free article] [PubMed] [Google Scholar]


