Abstract
A hexanucleotide (GGGGCC) expansion in C9ORF72 gene is the most common genetic change seen in familial Frontotemporal Lobar Degeneration (FTLD) and familial Motor Neurone Disease (MND). Pathologically, expansion bearers show characteristic p62 positive, TDP-43 negative inclusion bodies within cerebellar and hippocampal neurons which also contain dipeptide repeat proteins (DPR) formed from sense and antisense RAN (repeat associated non ATG-initiated) translation of the expanded repeat region itself. ‘Inappropriate’ formation, and aggregation, of DPR might therefore confer neurotoxicity and influence clinical phenotype. Consequently, we compared the topographic brain distribution of DPR in 8 patients with Frontotemporal dementia (FTD), 6 with FTD + MND and 7 with MND alone (all 21 patients bearing expansions in C9ORF72) using a polyclonal antibody to poly-GA, and related this to the extent of TDP-43 pathology in key regions of cerebral cortex and hippocampus. There were no significant differences in either the pattern or severity of brain distribution of DPR between FTD, FTD + MND and MND groups, nor was there any relationship between the distribution of DPR and TDP-43 pathologies in expansion bearers. Likewise, there were no significant differences in the extent of TDP-43 pathology between FTLD patients bearing an expansion in C9ORF72 and non-bearers of the expansion. There were no association between the extent of DPR pathology and TMEM106B or APOE genotypes. However, there was a negative correlation between the extent of DPR pathology and age at onset. Present findings therefore suggest that although the presence and topographic distribution of DPR may be of diagnostic relevance in patients bearing expansion in C9ORF72 this has no bearing on the determination of clinical phenotype. Because TDP-43 pathologies are similar in bearers and non-bearers of the expansion, the expansion may act as a major genetic risk factor for FTLD and MND by rendering the brain highly vulnerable to those very same factors which generate FTLD and MND in sporadic disease.
Electronic supplementary material
The online version of this article (doi:10.1186/2051-5960-2-70) contains supplementary material, which is available to authorized users.
Keywords: Frontotemporal lobar degeneration, Motor neurone disease, C9ORF72, Dipeptide repeat proteins
Introduction
Frontotemporal Lobar Degeneration (FTLD) is a clinical, pathological and genetically heterogeneous condition. The major clinical syndromes principally involve personality and behavioural change (behavioural variant frontotemporal dementia, or bvFTD) or language alterations of a fluent (semantic dementia) or non-fluent (progressive non-fluent aphasia) nature [1]. All three syndromes can be accompanied by Motor Neurone Disease (MND) though the combination of FTD and MND is most common [1]. Causative mutations have been identified in tau (MAPT) [2], progranulin (GRN) [3, 4], CHMP2B [5], and most recently in C9ORF72 [6–8]. This latter genetic change is characterized by an expansion of a hexanucleotide (GGGGCC) repeat region in the first intron or the promoter region of C9ORF72 gene, occurring in patients with either FTLD or MND, or a combination of both [6–8], and can number in excess of as many as 1500 repeats [9]. The expansion is found in about one in every twelve patients with FTLD and 1 in 10 patients with MND.
Pathologically, most FTLD cases with the expansion [6, 8, 10–13], like many non-mutational cases of FTLD [14, 15], show inclusion bodies within neurones (NCI) and glial cells of the cerebral cortex and hippocampus that contain the nuclear transcription factor, TDP-43. However, they also show a unique pathology within the hippocampus [10, 16, 17] and cerebellum [10–12, 16, 17] characterised by NCI that are TDP-43 negative, but immunoreactive to p62 protein. At least some of the target protein(s) within these p62-positive NCI are dipeptide repeat proteins (DPR) [17–20] formed from sense and antisense RAN (repeat associated non ATG-initiated) translation of the expanded repeat region itself [18–23]. ‘Inappropriate’ formation, and aggregation, of DPR may therefore confer neurotoxicity and influence clinical phenotype.
Previous studies on DPR in FTLD and MND have been largely limited to investigations on the hippocampus and cerebellum [17, 18], though one previous study [24] performed a wider topographic screen for DPR on 35 expansion cases with FTLD (n = 9) or MND (n = 8), or FTLD with MND (n = 18). This study failed to detect any significant differences in regional distribution or severity of DPR between each clinical group. In the present study, we too have determined how widely DPR are distributed throughout the brain in patients with FTLD and others with MND, and what cell types are affected, comparing the topographic distribution of DPR in patients with FTLD and MND in order to assess to what extent this distribution relates to the clinical expression of each disorder, and how it might relate to the underlying TDP-43 proteinopathy. Furthermore, because TMEM106B genotype has been claimed to be a genetic modifier of FTLD [25–29], and to protect C9ORF72 carriers from FTD [25, 28, 29], we investigated whether this might influence both the distribution and severity of both DPR and TDP-43 pathologies in FTLD cases. We also performed a similar analysis in respect of Apolipoprotein E (APOE) genotype since there have many studies claiming an increased frequency of APOE ϵ4 allele in FTLD (see [30–33] for examples) and this might operate by facilitating pathological changes, as it does in Alzheimer’s disease where possession of APOE ϵ4 allele is associated with increased deposition of amyloid β protein [34] and cerebral amyloid angiopathy [35].
Materials and methods
Patients
Sixty seven patients were investigated in total. Fourteen patients with FTLD (cases#1-14, 9 males, 5 females), and 7 with MND (cases#41-47, 6 males, 1 female) bore expansions in C9ORF72 (as evidenced by Southern blot and/or repeat primed PCR) (see Table 1 and Additional file 1: Table S1). We also investigated a further 14 other patients with FTLD (cases#27-40, 11 males, 3 females), and 20 other patients with MND (cases#48-67, 12 males, 8 females), all with no known mutation, and 12 patients with FTLD bearing a mutation in GRN (cases#15-26, 7 males, 5 females) (see Table 1 and Additional file 1: Table S1). All genetic analyses have been reported by us elsewhere [2, 3, 13, 36, 37]. Tissues from 39/40 FTLD and 23/27 of the MND cases were obtained from the Manchester Brain Bank through appropriate consenting procedures for the collection and use of the human brain tissues. All cases were from the North West of England and North Wales. The other FTLD case (case#14) and the further 4 MND cases (cases#44-47) were obtained from Institute of Psychiatry Brain Bank (London). Again, these were obtained through appropriate consenting procedures for the collection and use of the human brain tissues. The 40 patients with FTLD fulfilled Lund-Manchester clinical diagnostic criteria for FTLD [38, 39] and were consistent with recent consensus criteria [40, 41]. The 27 patients with MND fulfilled El Escorial criteria [42].
Table 1.
Mean (±SD) age at onset, death and duration of illness for groups of patients with FTD, FTD + MND or MND associated with expansions in C9ORF72 , or mutations in GRN , or not known to be associated with any known FTLD or MND associated genes
| Group | M/F | Age at onset (y) | Age at Death (y) | Duration of illness (y) |
|---|---|---|---|---|
| C9ORF72 FTD | 7/1 | 61.1 ± 8.5 | 67.6 ± 7.0 | 6.5 ± 3.0 |
| C9ORF72 FTD + MND | 2/4 | 58.8 ± 6.1 | 64.7 ± 5.8 | 5.8 ± 6.2 |
| C9ORF72 MND | 6/1 | 55.8 ± 8.6 | 57.6 ± 7.6 | 2.8 ± 1.7 |
| All C9ORF72 cases | 15/6 | 58.9 ± 7.8 | 63.4 ± 7.9 | 5.2 ± 4.1 |
| GRN FTD | 7/5 | 60.8 ± 5.9 | 69.7 ± 3.9 | 8.8 ± 3.8 |
| Non-mutational FTD | 2/2 | 60.8 ± 11.9 | 68.0 ± 15.6 | 7.3 ± 4.1 |
| Non-mutational FTD + MND | 9/1 | 59.3 ± 7.0 | 65.4 ± 8.2 | 6.2 ± 3.8 |
| All non-mutational FTD | 11/3 | 59.6 ± 12.7 | 63.4 ± 13.6 | 2.4 ± 1.1 |
| Non-mutational MND | 12/8 | 59.7 ± 8.2 | 66.1 ± 10.2 | 6.5 ± 3.8 |
FTD = Frontotemporal Dementia, FTD + MND + Frontotemporal dementia and Motor Neurone disease, MND = Motor Neurone Disease.
From clinical and neuropsychological assessments, 8 of the 14 FTLD cases with an expansion in C9ORF72 had a pure/predominant bvFTD phenotype, whereas the other 6 showed a mixed bvFTD and MND phenotype. Five of the FTLD patients without known mutation showed bvFTD phenotype, 3 had a progressive non-fluent aphasia (PNFA) phenotype and 6 showed a mixed bvFTD and MND phenotype. Pathologically, among the C9ORF72 expansion bearers with FTLD, all 8 patients with bvFTD had FTLD-TDP type A histology, whereas all 6 patients with FTD + MND had FTLD-TDP type B histology (according to Mackenzie et al. 2011 [43]). Of the 14 FTLD patients with no known mutation, 4 had FTLD-TDP type A histology (2 with bvFTD phenotype and 2 with PNFA), and 10 had FTLD-TDP type B histology (3 with bvFTD phenotype, 1 with PNFA phenotype and 6 with FTD + MND phenotype). All 12 patients bearing GRN mutation displayed FTLD-TDP type A histology; 6 had bvFTD phenotype and 6 had PNFA phenotype. All 27 patients with MND displayed characteristic TDP-43 pathology in mid brain and brainstem motor nerve nuclei, and in spinal cord (where this was available for study). These were usually skein-like in appearance, but occasionally more rounded, solid-appearing inclusions were also present.
Histological methods
Paraffin sections were cut (at a thickness of 6 μm) from formalin fixed blocks of representative regions of brain to include (where available) frontal cortex (BA8/9), temporal cortex (BA21/22), cingulate gyrus, insular cortex, motor cortex, inferior parietal and occipital (BA17/18) cortex. Blocks were also cut from the amygdala and posterior hippocampus, basal ganglia (to include caudate nucleus, putamen, globus pallidus and thalamus), substantia nigra (to include oculomotor nucleus), pons (to include locus caeruleus and V cranial nerve nucleus), medulla (to include inferior olives and XII cranial nerve nucleus), cerebellum (with dentate nucleus) and cervical and lumbar spinal cord (where available).
Sections from each brain region were immunostained with a poly-GA antibody (courtesy of M Hasegawa), as described previously [44]. The antibody was used at dilution of 1:1000–1:3000. This antibody was raised against poly-(GA)8 peptide with cysteine at N-terminus, conjugated to m-maleimidobenzoyl-N-hydrosuccinimide ester-activated thyroglobulin. The thyroglobulin-peptide complex (200 μg) emulsified in Freund’s complete adjuvant was injected subcutaneously into a New Zealand White rabbit, followed by 4 weekly injections of peptide complex emulsified in Freund’s incomplete adjuvant, starting after 2 weeks after the first immunization. Immunoreactivity of the antisera was characterized by ELISA as follows. The peptide immunogens were coated onto microtiter plates. The plates were blocked with 10% fetal bovine serum (FBS) in PBS, incubated with the rabbit antisera diluted in 10% FBS/ PBS at room temperature for 1.5 h, followed by incubation with HRP-goat anti-rabbit IgG (Bio-Rad) at 1:3000 dilution, and reacted with the substrate, 0.4 mg/mL o-phenylenediamine, in citrate phosphate buffer (24 mM citric acid, 51 mM Na2HPO4). The absorbance at 490 nm was measured using Plate Chameleon (HIDEX). In addition, sections of temporal cortex with hippocampus from 11 (non-expansion bearing) cases with other histological and genetic forms of FTLD, other neurodegenerative disorders and healthy controls (see Table 1) were also immunostained for DRP with anti poly-GA antibody as ‘negative controls’. Antibodies were employed in standard IHC protocol, though antigen unmasking was performed by pressure cooking in citrate buffer (pH 6, 10 mM) for 30 minutes, reaching 120 degrees Celsius and >15 kPa pressure.
Further sections of frontal cortex and temporal cortex with hippocampus were immunostained for both phosphorylated (at Ser 409/410), and non-phosphorylated, TDP-43 (rabbit polyclonal antibodies (pS409/410-2 antibody, Cosmo Biotech Ltd, Tokyo, Japan and 10782-2-AP antibody, Proteintech, Manchester, UK, respectively – at 1:3000 and 1:1000, respectively).
Pathological assessment
The presence of DPR immunostained NCI within nerve cells was assessed at ×20 magnification in those brain regions where all cases could be represented, according to:
0 = no DPR immunostained NCI present in any field.
0.5 = rare/single DPR immunostained NCI present in entire section.
1 = a few DPR immunostained NCI present, in some but not all fields.
2 = a moderate number of DPR immunostained NCI present in each field.
3 = many DPR immunostained NCI present affecting many cells in each field.
4 = very many DPR immunostained NCI present, affecting nearly all cells in every field.
Scores per assessed area were summated across those regions where these were available for all 21 individuals with C9ORF72 expansions. Brain regions were grouped on an anatomical or a ‘functional’ basis. Hence, scores from frontal, temporal, cingulate, insular, parietal and occipital cortical regions were summated to generate a total ‘cortical’ score for each case. Scores from hippocampus and adjacent regions of subiculum, entorhinal cortex and fusiform gyrus were summated to give a total medial temporal lobe score for each case. Scores for caudate nucleus, putamen, globus pallidus, thalamus, substantia nigra, locus caeruleus and dorsal raphe were summated to give a total ‘subcortical’ score. Scores in motor cortex and in X and XII cranial nerve nuclei were summated to give a total ‘motor’ score. Scores in cerebellar granule and Purkinje cells, and in cells of the dentate nucleus, inferior olives and pontine nuclei were summated to give a total ‘cerebellar’ score. Due to the unavailability of tissue in all cases, it was not possible to include scores for amygdala (absent from 5/21 cases) or spinal cord (absent from 10/21 cases) within the medial temporal lobe or motor region analyses, respectively.
The frequency of TDP-43 pathological changes (as NCI and neurites, where present) in each of frontal and temporal cortex (pyramidal cells of layers II) and hippocampus (dentate gyrus granule cells), was scored semi-quantitatively according to:
0 = no TDP-43 immunostained NCI and/or neurites.
0.5 = rare/single TDP-43 immunostained NCI and/or neurite present in entire section.
1 = very few TDP-43 immunostained NCI and/or neurites.
2 = a moderate number of TDP-43 immunostained NCI and/or neurites.
3 = many TDP-43 immunostained NCI and/or neurites, affecting many cells in every field.
4 = very many immunostained NCI and/or neurites present, affecting nearly all cells in every field.
TDP-43 pathology cores per assessed area were summated across those regions where these were available for all individuals. Hence, scores from dentate gyrus of hippocampus, and from frontal and temporal cortex, were summated (in all except 4 cases where dentate gyrus was not available) to give a total TDP-43 pathology score for each case.
Genetic analysis
DNA was extracted from blood or frozen brain tissue by routine phenol-chloroform extraction. The TMEM106B assay was genotyped by allelic discrimination using the Applied Biosystems pre-developed assay cat number C_7604953_10. Genotyping was carried out using the Applied Biosystems 7900, and genotypes were assigned automatically using the SDS 2.3 software. APOE was genotyped according to Wenham [45].
Statistical analysis
Rating data was entered into an excel spreadsheet and analyzed using Statistical Package for Social Sciences (SPSS) software (version 17.0). Kruskal-Wallis or Mann–Whitney test was used to compare inclusion scores between several groups or pairs of groups, respectively. All correlations were performed using Spearman rank correlation test. In all instances, a p-value of less than 0.05 was considered statistically significant.
Results
Demographics
Mean ages at onset, death and duration of illness for C9ORF72 associated FTD, FTD + MND and MND groups, FTD associated with GRN mutation, non-mutational FTD, FTD + MND and MND groups are shown in Table 1.
ANOVA comparison of age at onset, death and duration of illness between all 7 diagnostic groups revealed no significant differences in age at onset (F6,51 = 0.27, p = 0.946) or death (F6,56 = 1.4, p = 0.227), though duration of illness differed between the groups (F6,51 = 4.3, p = 0.001). As would be expected, post-hoc analysis showed that duration of illness was significantly less in C9ORF72 cases with MND than C9ORF72 associated FTD (p = 0.014), but not less than C9ORF72 associated FTD + MND (p = 0.281), nor was there any difference in disease duration between C9ORF72 associated FTD and C9ORF72 associated FTD + MND (p = 0.815). Again, as expected, non-mutational MND showed a shorter duration of illness than non-mutational FTD (p = 0.022) and non-mutational FTD + MND (p = 0.013). However, there were no significant differences in duration of illness between C9ORF72 associated FTD, non-mutational FTD or GRN associated FTD (F2,37 = 1.6, p = 0.215), or between C9ORF72 associated FTD + MND and non-mutational FTD + MND (p = 0.900), or between C9ORF72 associated MND and non-mutational MND (p = 0.605).
ANOVA comparison of age at onset, death and duration of illness between C9ORF72 associated FTD, FTD + MND and MND groups alone revealed no significant group differences in age at onset (F2,17 = 0.77, p = 0.477) or duration of illness (F2,17 = 1.4, p = 0.227), though duration of illness tended to differ between the 3 groups (F2,18 = 4.1, p = 0.045).
Cytological observations
All cases had been previously classified on the basis of the type and distribution of TDP-43 immunoreactive changes according to Mackenzie et al. 2011 [43], and hence showed TDP-43 histological changes typical of the group in which they had been placed. These have been well described previously, both by ourselves and others, and are therefore not further detailed in the present study.
DPR were characteristically present in all FTLD and MND cases previously known to bear expansions in C9ORF72, but none were seen in any of the cases bearing GRN mutations, nor in any of the other FTLD cases or MND cases not known to be associated with any FTLD or MND linked mutation.In cases of FTD and FTD + MND, DPR were observed to be most frequent within the cerebral neocortex, hippocampus and cerebellum. They were infrequent or absent in basal ganglia regions, and were rare or usually absent in mid brain, brainstem, medulla and spinal cord regions. DPR were present in neuronal cytoplasmic inclusions (NCI) throughout all cortical layers in all regions of the cerebral cortex examined. In outer cortical layers they were mostly present in small non-pyramidal neurons appearing as dots or clusters of granules (Figure 1a), whereas in the deeper cortical layers DPR were again present in smaller non-pyramidal neurons as clusters of granules, but in the larger pyramidal cells they often adopted a more star-shaped or spicular appearance (Figure 1b). Granular type DPR were particular numerous in parietal and occipital cortex and in motor cortical regions in most cases, and common in frontal and temporal cortex when these areas were not severely degenerated, though in cases where these regions were badly degenerated DPR were much less frequent.In the hippocampus, DPR were present as abundant, small, rounded NCI within granule cells of the dentate gyrus (Figure 1c), though more spicular or granular inclusions were commonly seen within pyramidal cells of areas CA4 and CA2/3 (Figure 1d), becoming less frequent in CA1 region and subiculum. Again neurons containing small clusters of DPR were present throughout all layers of the entorhinal cortex. Small granular or spicular NCI were widespread within the amygdala, though usually only to a moderate extent, and less severely than in the hippocampus.Generally, DPR were sparse within basal ganglia regions. They were occasionally present as spicular NCI in small neurons in the caudate nucleus and putamen, and in larger neurons of the globus pallidus but were more common in the thalamus, especially those of the ventrolateral nuclei (Figure 1e). DPR were rarely (if ever) seen in cells of the substantia nigra, locus caeruleus, nucleus basalis of Meynert, dorsal raphe nucleus, basalis pontis, in motor nuclei of III, IV, V, X or XII cranial nerves, or in anterior horn cells of the spinal cord (where available for study). However, in a few cases DPR were occasionally seen as spicular NCI in neurons of the inferior olives.
Figure 1.

Topographic brain distribution of dipeptide repeat proteins (poly-GA) in patient #9 with Frontotemporal dementia associated with an expansion in C9ORF72. Regions shown are frontal cortex layer II (a), frontal cortex layer V (b), dentate gyrus (c) and area CA4 (d) of hippocampus, ventrolateral nucleus of thalamus (e), granule cells (f) and Purkinje cells (g) of cerebellum, dentate nucleus (h). Immunoperoxidase-haematoxylin ×40 microscope magnification.
In the cerebellum, DPR were widespread within granule cells of the cerebellum (Figure 1f). As with the cerebral cortex, more granular looking NCI were usually present in basket cells, with occasional spicular NCI being seen in Purkinje cells (Figure 1g) and neurones in the dentate nucleus (Figure 1h), but none were seen within Golgi neurones, or within Bergmann glia. A punctate, or filamentous, staining was also seen within the molecular layer of the cerebellum, this probably relating to parallel projection fibres (Figure 1g). A similar distribution of DPR was observed in the 7 MND cases bearing expansions in C9ORF72 to that seen in the FTD and FTD + MND cases (Figure 2).
Figure 2.
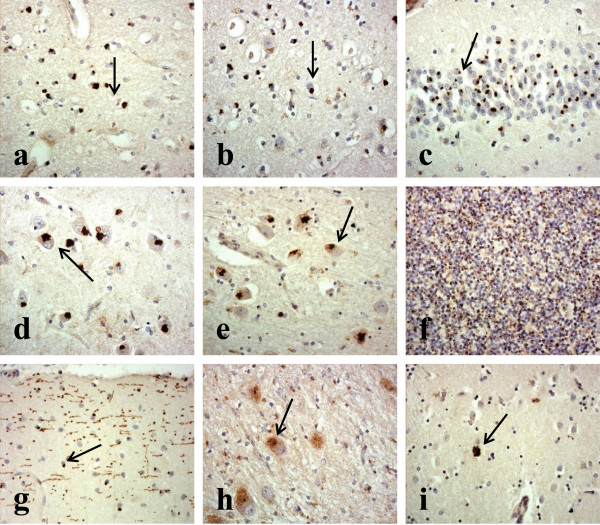
Topographic brain distribution of dipeptide repeat proteins (poly-GA) in patient #23 with Motor Neurone Disease associated with an expansion in C9ORF72. Regions shown are frontal cortex layer II (a), frontal cortex layer V (b), dentate gyrus (c) and area CA4 (d) of hippocampus, ventrolateral nucleus of thalamus (e), granule cells (f) and Purkinje cells (g) of cerebellum, dentate nucleus (h) and putamen (i). Immunoperoxidase-haematoxylin ×40 microscope magnification.
Comparisons of DPR ratings
Composite rating scores for DPR pathology are shown for each case in Table 2. When Comparisons of rating scores for DPR, showed no significant differences in total severity scores for DPR between FTD, FTD + MND and MND cases for cortical (χ2 = 0.19, p = 0.911), hippocampal (χ2 = 3.7, p = 0.160), motor (χ2 = 2.3, p = 0.323), subcortical (χ2 = 2.56, p = 0.279) or cerebellar (χ2 = 2.3, p = 0.318) regions, or for total score summated across all 5 subregions (χ2 = 1.4, p = 0.487) (Table 2). Similarly, when analysed by region, there were no significant differences in DPR scores between any of the cortical regions investigated, either over all cases, or according to clinical subgroups.
Table 2.
Summated scores for DPR pathology ratings in Cortical, Medial Temporal, Motor, Subcortical and Cerebellar regions, as well as total score across all 5 regions, for patients with FTD (cases #1-8), FTD + MND (cases#9-14) and MND (cases#41-47) associated with expansion in C9ORF72
| Case ID# | Summated DPR score | ||||||
|---|---|---|---|---|---|---|---|
| Pathology | Cortex | Medial temporal | Motor | Subcortical | Cerebellar | Total | |
| 1 | FTLD-TDP A | 16 | 12 | 3 | 3 | 5 | 39 |
| 2 | FTLD-TDP A | 12 | 14 | 3 | 1 | 5.5 | 35.5 |
| 3 | FTLD-TDP A | 15 | 18 | 3 | 5 | 7.5 | 48.5 |
| 4 | FTLD-TDP A | 13 | 12 | 2 | 2.5 | 3.5 | 33 |
| 5 | FTLD-TDP A | 12 | 11 | 1 | 3 | 3 | 30 |
| 6 | FTLD-TDP A | 11 | 9 | 1.5 | 3 | 2 | 26.5 |
| 7 | FTLD-TDP A | 12 | 9.5 | 1 | 5.5 | 3.5 | 31.5 |
| 8 | FTLD-TDP A | 22 | 23 | 2 | 4 | 5.5 | 55.5 |
| 9 | FTLD-TDP B | 20 | 15.5 | 2 | 1 | 4.5 | 43 |
| 10 | FTLD-TDP B | 12 | 11 | 2 | 2 | 4 | 31 |
| 11 | FTLD-TDP B | 11 | 10.5 | 2 | 1.5 | 4 | 29 |
| 12 | FTLD-TDP B | 18 | 16 | 3.5 | 4.5 | 5 | 47 |
| 13 | FTLD-TDP B | 11 | 11 | 2 | 3.5 | 2 | 29.5 |
| 14 | FTLD-TDP B | 16 | 15 | 3 | 4.5 | 6.5 | 45 |
| 41 | MND | 16 | 13 | 3 | 6 | 5.5 | 43.5 |
| 42 | MND | 15 | 13 | 3 | 3 | 1 | 35 |
| 43 | MND | 14 | 18 | 3 | 4 | 3 | 42 |
| 44 | MND | 12 | 18 | 2.5 | 5 | 3 | 40.5 |
| 45 | MND | 13 | 12 | 3 | 1.5 | 4.5 | 34 |
| 46 | MND | 11 | 19 | 2 | 4.5 | 3 | 39.5 |
| 47 | MND | 23 | 23 | 3 | 5 | 3.5 | 57.5 |
When all cases were grouped, there were significant correlations between the severity of DPR pathology and age at onset of disease for cortical (rs = −0.620, p = 0.004), hippocampal (rs = −0.537, p = 0.015), motor (rs = −0.482, p = 0.031) and for total score summated across all 5 subregions (rs = −0.583, p = 0.016), but not for subcortical (rs = −0.218, p = 0.356) or cerebellar (rs = −0.137, p = 0.318) regions. However, there was no significant correlation between the severity of DPR pathology and duration of disease, except weakly so in cerebellum (rs = −0.493, p = 0.027).
Comparisons of DPR severity scores according to possession of at least one copy of APOE ϵ4 allele were performed on 12/14 FTLD cases and 3/7 MND cases (for whom DNA was available for genotyping). No significant differences in DPR severity scores were found between ϵ4 allele bearers and non-bearers, either overall (p = 0.240) or according to subregion (cortical (p = 0.130), hippocampal (p = 0.189), motor (p = 0.887), subcortical (p = 0.506) or cerebellar (p = 0.640).
Due to availability of DNA, TMEM106B genotypes were only available for 7 of the 14 FTLD cases, and 1 of the 7 MND cases, bearing C9ORF72 expansions, 9 of the 12 GRN mutation carriers, and 10 of the 14 FTLD cases and 1 of the 20 MND cases without known mutation. Because TMEM106B SNPs rs1020004, rs1990622 and rs6966915 were in complete linkage disequilibrium, pathological analyses were confined to rs 1990622. Unfortunately, only one of the FTLD, and none of the MND cases, irrespective of type or presence of mutation, was homozygous for the minor allele at any SNP. Of the C9ORF72 expansion bearers, 4 of the FTLD cases were homozygotes for the major allele and 3 were heterozygotes, with the MND case being homozygous for the major allele. Of the GRN mutation bearers, 5 of the FTLD cases were homozygotes for the major allele and 4 were heterozygotes. Of the non-mutational FTLD cases, 6 were homozygotes for the major allele, 2 were heterozygotes and 1 was homozygous for the minor allele.
Nonetheless, comparisons of DPR severity scores were performed between FTLD cases heterozygous (TC) and homozygous (TT) for the major allele. No significant differences in DPR severity scores were found between bearers of TC or TT genotypes, either overall (p = 0.881) or according to subregion (cortical (p = 0.881), hippocampal (p = 0.549), motor (p = 0.495), subcortical (p = 0.546) or cerebellar (p = 0.878).
Comparisons of TDP-43 ratings
Composite rating scores for TDP-43 pathology are shown for each case in Table 3. There were no significant differences in the severity of TDP-43 pathology scores between FTLD cases bearing expansions in C9ORF72, FTLD cases without expansions, and FTLD cases with GRN mutation for either frontal cortex (χ2 = 1.95, p = 0.377), temporal cortex (χ2 = 1.94, p = 0.379) or dentate gyrus of hippocampus (χ2 = 1.55, p = 0.461), or as total TDP-43 severity score (χ2 = 1.82, p = 0.403). Moreover, there were no significant differences in the severity of TDP-43 pathology scores between FTLD cases with type A histology bearing an expansion in C9ORF72, FTLD cases with type A histology but without expansion in C9ORF72 mutation, and FTLD cases with GRN mutation and type A histology for either frontal cortex (χ2 = 2.27, p = 0.321), temporal cortex (χ2 = 0.66, p = 0.719) or dentate gyrus of hippocampus (χ2 = 5.45, p = 0.065), or as total TDP-43 severity score (χ2 = 2.19, p = 0.334). Similarly, there were no significant differences in the severity of TDP-43 pathology scores between FTLD cases with type B histology bearing an expansion in C9ORF72 and FTLD cases with type B histology but without expansion in C9ORF72 mutation for either frontal cortex (p = 0.570), temporal cortex (p = 0.147) or dentate gyrus of hippocampus (p = 0.344), or as total TDP-43 severity score (p = 0.254).
Table 3.
Summated scores for TDP-43 pathology ratings in Frontal and Temporal cortex, and Dentate Gyrus, as well as total score across all 3 regions, for patients with FTD (cases #1-8) or FTD + MND (cases#9-13) associated with C9ORF72 expansions, and for patients with FTLD due to GRN mutation (case#15-26) or not associated with any known mutation (cases#27-40)
| Case ID# | Gene | Pathology | TDP-43 score | |||
|---|---|---|---|---|---|---|
| DG | TCX | FCX | Total | |||
| 1 | C9ORF72 | FTLD-TDP A | 1 | 4 | 3 | 8 |
| 2 | C9ORF72 | FTLD-TDP A | 2 | 4 | 3 | 8 |
| 3 | C9ORF72 | FTLD-TDP A | 2 | 1 | 3 | 6 |
| 4 | C9ORF72 | FTLD-TDP A | 1 | 3 | 3 | 7 |
| 5 | C9ORF72 | FTLD-TDP A | 1 | 1 | 1 | 3 |
| 6 | C9ORF72 | FTLD-TDP A | 2 | 2 | 2 | 6 |
| 7 | C9ORF72 | FTLD-TDP A | 1 | 1 | 2 | 4 |
| 8 | C9ORF72 | FTLD-TDP A | 2 | 2 | 3 | 7 |
| 9 | C9ORF72 | FTLD-TDP B | 3 | 2 | 2 | 7 |
| 10 | C9ORF72 | FTLD-TDP B | 4 | 4 | 4 | 12 |
| 11 | C9ORF72 | FTLD-TDP B | 4 | 3 | 3 | 10 |
| 12 | C9ORF72 | FTLD-TDP B | 1 | 2 | 2 | 5 |
| 13 | C9ORF72 | FTLD-TDP B | na | 3 | 3 | na |
| 15 | GRN | FTLD-TDP A | na | 4 | 4 | na |
| 16 | GRN | FTLD-TDP A | na | 2 | 2 | na |
| 17 | GRN | FTLD-TDP A | 4 | 3 | 3 | 10 |
| 18 | GRN | FTLD-TDP A | 2 | 2 | 2 | 6 |
| 19 | GRN | FTLD-TDP A | 3 | 3 | 3 | 9 |
| 20 | GRN | FTLD-TDP A | 1 | 4 | 4 | 9 |
| 21 | GRN | FTLD-TDP A | 2 | 4 | 4 | 10 |
| 22 | GRN | FTLD-TDP A | 4 | 2 | 1 | 7 |
| 23 | GRN | FTLD-TDP A | 2 | 1 | 3 | 6 |
| 24 | GRN | FTLD-TDP A | 2 | 0 | 1 | 3 |
| 25 | GRN | FTLD-TDP A | 3 | 4 | 4 | 11 |
| 26 | GRN | FTLD-TDP A | na | 3 | 4 | na |
| 27 | No mutation | FTLD-TDP A | 2 | 3 | 1 | 6 |
| 28 | No mutation | FTLD-TDP A | 3 | 4 | 4 | 11 |
| 29 | No mutation | FTLD-TDP A | 2 | 2 | 2 | 6 |
| 30 | No mutation | FTLD-TDP A | 1 | 1 | 1 | 3 |
| 31 | No mutation | FTLD-TDP B | 4 | 4 | 4 | 12 |
| 32 | No mutation | FTLD-TDP B | 0 | 3 | 4 | 7 |
| 33 | No mutation | FTLD-TDP B | 3 | 0 | 1 | 4 |
| 34 | No mutation | FTLD-TDP B | 2 | 1 | 1 | 4 |
| 35 | No mutation | FTLD-TDP B | 3 | 2 | 4 | 9 |
| 36 | No mutation | FTLD-TDP B | 4 | 1 | 1 | 6 |
| 37 | No mutation | FTLD-TDP B | 2 | 2 | 2 | 6 |
| 38 | No mutation | FTLD-TDP B | 1 | 0 | 1 | 2 |
| 39 | No mutation | FTLD-TDP B | 1 | 2 | 3 | 6 |
| 40 | No mutation | FTLD-TDP B | 3 | 3 | 3 | 9 |
Within the C9ORF72 expansion carriers, there were no significant correlations between the severity of TDP-43 pathology and either age at onset of disease, or duration of illness, for either cortical, hippocampal or total scores. Neither was there any correlation between TDP-43 scores and clinical phenotype (ie FTD versus FTD + MND).
Comparisons of TDP-43 severity scores according to possession of at least one copy of APOE ϵ4 allele were performed on 36/39 FTLD cases (for whom DNA was available for genotyping), irrespective of the presence or absence of mutation. No significant differences in the severity of TDP-43 pathology scores were seen between FTLD cases with APOE ϵ4 allele and FTLD cases without APOE ϵ4 allele for either frontal cortex (p = 0.187), temporal cortex (p = 0.293) or dentate gyrus of hippocampus (p = 0.171), or as total TDP-43 severity score (p = 0.261). Similarly, comparisons of TDP-43 severity scores according to heterozygosity (TC) or homozygosity (TT) for major allele in TMEM106B were performed on 24/39 FTLD cases (where these data were available), irrespective of the presence or absence of mutation. No significant differences in the severity of TDP-43 pathology scores between FTLD cases with TC and TT genotypes were seen for either frontal cortex (p = 0.301), temporal cortex (p = 0.592) or dentate gyrus of hippocampus (p = 0.768), or as total TDP-43 severity score (p = 0.519).
Discussion
There are two major outcomes from this study. Firstly, that in cases of FTLD or MND bearing expansions in C9ORF72, neither the topographical distribution, nor the relative severity, of DPR differs between cases of FTD alone, FTD + MND or MND alone. Secondly, neither the morphological appearance, nor relative severity, of TDP-43 immunoreactive changes differ in cases of FTLD bearing an expansion in C9ORF72 from that in non-expansion bearing cases of FTLD (ie sporadic forms of FTLD), or from cases of FTLD bearing mutations in GRN. These findings support the observations of Mackenzie and colleagues [24] who likewise noted no differences in either DPR or TDP-43 pathology between expansion bearing cases of FTD alone, FTD + MND or MND alone. Such observations call into question the relevance of DPR pathology in determining clinical phenotype, and the role of the expansion per se in causing FTLD or MND.
Expansions in C9ORF72 have been postulated to invoke disease by 3, mutually non-exclusive, mechanisms: firstly involving a loss of C9orf72 protein through haploinsufficiency [6], secondly by RNA toxicity through sequestration of RNA species by the expanded sequences [6, 21], and thirdly through cytotoxicity from formation and accumulation of DPR [18–20, 22, 23]. While this latter suggestion is attractive, and has parallels with other aggregating brain proteins such as tau, TDP-43, α-synuclein, there is strong evidence that argues against such an effect.
Firstly, in both the present, and in other studies [24], the distribution of DPR, as evidenced by poly-GA immunostaining, does not parallel that of neurodegeneration (ie the TDP-43 proteinopathy) in either FTLD or MND. Indeed, in both disorders the greatest severity of DPR lies in brain regions such as the granule cell layer of the cerebellum, the hippocampal dentate gyrus and CA4 pyramidal cells, and the parietal and occipital cortex, with similar observations being recorded for poly-GP and poly-GR antibodies [17]. Such affected regions of the brain have hitherto not been considered to be involved in the (TDP-43) pathological process, and are brain regions which do not show obvious clinical repercussions, which might be anticipated if the DPR were principal in driving a neurodegenerative process, although there have been reports of cerebellar atrophy in expansion bearers [46, 47].
Secondly, it is hard to conceive how a seemingly identical topographical pattern of DPR pathology could determine, or even predispose towards, such diverse clinical phenotypes as FTD or MND. To explain such an apparent paradox, Mackenzie et al. 2013 [24] invoked the argument that the visible DPR protein aggregates conferred a neuroprotective effect in functionally preserved brain regions with high DPR loads, such as cerebellum, hippocampus and occipital cortex, through sequestration of soluble toxic species. In the present study, we were unable to show any significant differences in DPR load between any of the neocortical regions investigated, these being as equally high in functionally disturbed areas such as frontal, temporal and motor cortex, as those in apparently functionally normal regions such as parietal and occipital cortex. It is therefore difficult to reconcile such observations with any putative neuroprotective effect, since if this were indeed the case then it might be anticipated that visible DPR should be lower in cortical regions, such as frontal and temporal cortex, showing functional change and neurodegeneration, in which any putative neurotoxic effects of soluble (oligomeric) precursors had not been so restricted. Nonetheless, there does appear to be some clinical distinctions between expansion bearers and non-bearers in both FTLD and MND, in as much as expansion bearers in both conditions are more likely to display psychosis [13, 48, 49]. Concerning the present cases, there was no difference between the level of DPR pathology in those cases presenting with a florid psychosis compared to those where psychosis was not apparent (data not shown), suggesting that the presence and/or extent of DPR pathology is neither a determinant, nor a modulator, of psychosis.
The second observation from this study indicates that as far as the TDP-43 proteinopathy is concerned, there are again no distinctions in pathology between expansion bearers and non-bearers of the expansion, either overall or more specifically in respect of FTLD-TDP type A or type B histologies. This also supports observations by Mackenzie et al. [24] and further calls into question the role of the expansion per se. Although, there is no a priori reason to suspect that patients with an expansion in C9ORF72 should ‘naturally’ carry a higher burden of TDP-43 pathology than non-expansion carriers, it is nonetheless important to observe that such differences do indeed not exist. Moreover, if the expansion were directly driving a TDP-43 proteinopathy, it is hard to reconcile this with observations of different TDP-43 histologies. FTLD-TDP Type B (involving a preponderance of neuronal cytoplasmic inclusions) is the most common TDP-43 histological subtype associated with the expansion [12, 24], though a significant number of cases display type A histology [17], and rare cases with type C histology have been described [12]. How such diverse histological patterns could stem from a common genetic root is puzzling.
An alternative explanation of the role of C9ORF72 could be that the expansion acts as a risk factor for FTLD and MND, though does not drive the (TDP-43) pathological process directly, acting more as a ‘gatekeeper’ to disease, rendering the brain susceptible to the ‘development’ of all sporadic forms of FTLD-TDP, and sporadic forms of MND associated with TDP-43. Such a scenario could accommodate the present observations of the different histological types of TDP-43 proteinopathy being associated with disease, and the relative proportions with which they occur, and with findings that the extent of TDP-43 pathology is the same in expansion and non-expansion bearers both in terms of either type A or type B histologies.
There is other evidence in support of this latter line of argument. Firstly, although rare, pathologies other than FTLD or MND, such as corticobasal degeneration [13, 17], Alzheimer’s disease ([50–52], but see [44]), Parkinson’s disease [53] and Huntington disease-like conditions [9], have been associated with expansions in C9ORF72. Indeed, there has been one Belgian case with C9ORF72 expansion and clinical FTLD which lacks detectable TDP-43 pathology, but shows FTLD-UPS pathology [7]. Secondly, there appears to be no link between expansion size and TDP-43 histological type [17], or clinical phenotype [9]. Thirdly, there appears to be a higher than expected coincidence of repeat expansions in individuals carrying other genetic variants involving mutations in GRN with C9ORF72 [54, 55] or MAPT with C9ORF72 [54–57] (so called oligogenic inheritance), suggesting that another ‘hit’ may be necessary for clinical disease, yet in these dual mutation cases, apart from the DPR changes, either a TDP-43 proteinopathy, or tauopathy, typical of the accompanying (GRN or MAPT) mutation prevails.
The correlation between age at onset and severity of DPR pathology, unrelated to disease duration, is interesting though the reason for this is unclear. It is possible that there is a more severe ‘expression’ of the expansion in younger individuals leading to a greater level of production and accumulation of DPR, or that the dipeptides aggregate more efficiently into NCI perhaps due to variations in length of DPR monomers formed as a result of differences in translation efficiency with age during RAN translation.
TMEM106B genotype (ie homozygosity for the minor allele) has been claimed to be a genetic modifier of FTLD in both GRN and C9ORF72 mutation carriers [25, 26, 28, 29], and to protect C9ORF72 carriers from FTD [25, 28, 29]. Indeed, van Blitterwijk et al. [28] reported that homozygous carriers of the minor protective TMEM106B allele with FTLD-TDP type A histology appeared to have a lower TDP-43 burden than homozygous carriers of the major allele. Therefore, we investigated whether variations at this locus might influence both the distribution and severity of both DPR and TDP-43 pathologies in FTLD cases associated with either C9ORF72 expansions or GRN mutations. Unfortunately, no homozygous bearers of TMEM106B minor allele were available in the present study for either mutation, but we did not find any differences in DPR and TDP-43 pathology between heterozygotes and those homozygous for the major allele suggesting that heterozygosity, at least, does not confer a ‘partial’ protection in either C9ORF72 or GRN mutation carriers. Similarly, although possession of at least one copy of APOE ϵ4 allele has been (variably) claimed to increase risk of FTLD [30–33], we found no significant differences in extent of either DPR or TDP proteinopathy between ϵ4 allele bearers and non-bearers, implying that any putative risk associated with ϵ4 allele does not operate through facilitating the pathological changes of FTLD.
How the expansion might act in a ‘gatekeeper’ role is not clear. Much attention has been levied towards a determination of the nature and effect of DPR in vivo, and in vitro, as well as the role RNA foci might play in causing the disease. However, there is evidence of C9orf72 haploinsufficiency in expansion carriers [6], though given the paucity of knowledge surrounding the role and function of C9orf72, it is difficult to ascribe precise meaning to such a putative loss of protein. A haploinsufficiency state might impair vital ‘protective’ brain functions. Alternatively, the formation of toxic RNA foci could likewise render cells vulnerable [6, 21, 23, 58], though against this are observations that such a process, like DPR formation, is disseminated widely throughout the brain, and not solely confined to those neuronal populations vulnerable to TDP-43 proteinopathy. Present knowledge suggests C9orf72 belongs to the differentially expressed in normal and neoplastic (DENN)-like family of proteins, these being GDP/GTP exchange factors which lead to activation of Rab-GTPases and maintenance of vesicular trafficking [59]. In this regard, it is interesting that the most recent GWAS for FTLD has highlighted variations in RAB38 as a risk factor for bvFTD [60]. RAB38 has been suggested to mediate protein trafficking to lysosomal-related organelles [61, 62] and maturation of phagosomes [63], and impairment in these processes might elicit cargo accumulation in early endosomes, with downstream effects on recycling/degradative pathways [61]. Indeed, an association with lysosomal processes in FTLD has previously been suggested by two studies on GRN [64] and TMEM106B [65]. Considering that endolysosomal homeostasis is essential for the health of neurons, functional links between RAB38, TMEM106B, PGRN and FTLD imply disturbances in C9orf72 may trigger or promote autophagosomal/lysosomal dysfunctions, thereby playing a key role in the onset and/or progression of the disease.
Nonetheless, if such a ‘gatekeeper role’ is true, then the almost complete penetrance of the mutation needs to be explained. In this respect the expansion might be viewed as providing an ‘open door’ to disease, but without actually causing disease directly, and in this regard the DPR pathology may be a ‘red herring’ without pathogenetic consequence. Clearly, until we know more about the normal function of C9orf72 protein, what cells it is present in, and where in the cell it is located, all is mere speculation. The concept of the expansion being a risk factor for disease, rather than a cause, is tempting since it would neatly accommodate the observed diversity of clinical and histological subtypes associated with the expansion. However, were the expansion to act as a risk factor, it appears to act (almost) selectively for FTLD and MND since expansions are only infrequently, and then maybe only coincidentally, seen in more common disorders such as Alzheimer’s and Parkinson’s disease [50–53]. Therefore, while it is clear that expansions in C9ORF72 are not without ‘pathological expression’, what DPR might translate into in clinical terms is not clear, and whether these structures confer anything beyond diagnostic utility still remains to be demonstrated.
Electronic supplementary material
Additional file 1: Selected clinical, pathological and genetic details for 40 cases of Frontotemporal Lobar degeneration and 27 cases of Motor Neurone disease. (XLSX 18 KB)
Acknowledgements
We acknowledge the support of Alzheimer’s Research UK and Alzheimer’s Society through their funding of the Manchester Brain Bank under the Brains for Dementia Research (BDR) initiative. DMAM and SPB also receive funding from Medical Research Council and Wellcome Trust (Grant number 089701/Z/09/Z), which supported this study. The Institute of Psychiatry Brain Bank receives funding from Medical Research Council.
Footnotes
Competing interests
The authors declare that they have no competing interests.
Contributor Information
Yvonne S Davidson, Email: yvonne.davidson@manchester.ac.uk.
Holly Barker, Email: holly.barker@student.manchester.ac.uk.
Andrew C Robinson, Email: andrew.c.robinson@manchester.ac.uk.
Jennifer C Thompson, Email: jennifer.c.thompson@manchester.ac.uk.
Jenny Harris, Email: jenny.harris@manchester.ac.uk.
Claire Troakes, Email: troakes.claire@kcl.ac.uk.
Bradley Smith, Email: smith.bradley@kcl.ac.uk.
Safa Al-Saraj, Email: Al-Sarraj.safa@kcl.ac.uk.
Chris Shaw, Email: shaw.chris@kcl.ac.uk.
Sara Rollinson, Email: sara.rollinson@manchester.ac.uk.
Masami Masuda-Suzukake, Email: masuda-ms@igakuken.or.jp.
Masato Hasegawa, Email: hasegawa-ms@igakuken.or.jp.
Stuart Pickering-Brown, Email: SPB@manchester.ac.uk.
Julie S Snowden, Email: Julie.snowden@manchester.ac.uk.
David M Mann, Email: david.mann@manchester.ac.uk.
References
- 1.Neary D, Snowden JS, Mann DMA. Frontotemporal lobar degeneration: clinical and pathological relationships. Acta Neuropathologica. 2007;114:31–38. doi: 10.1007/s00401-007-0236-3. [DOI] [PubMed] [Google Scholar]
- 2.Hutton M, Lendon CL, Rizzu P, Baker M, Froelich S, Houlden M, Pickering-Brown SM, Chakraverty S, Isaacs A, Grover A, Hackett J, Adamson J, Lincoln S, Dickson D, Davies P, Petersen RC, Stevens M, de Graaf E, Wauters E, van Baren J, Hillebrand M, Joosse M, Kwon JM, Nowotny P, Che LK, Norton J, Morris JC, Reed LA, Trojanowski JQ, Basun H, et al. Coding and splice donor site mutations in tau cause autosomal dominant dementia (FTDP-17) Nature. 1998;393:702–705. doi: 10.1038/31508. [DOI] [PubMed] [Google Scholar]
- 3.Baker M, Mackenzie IRA, Pickering-Brown SM, Gass J, Rademakers R, Lindholm C, Snowden J, Adamson J, Sadovnick AD, Rollinson S, Cannon A, Dwosh E, Neary D, Melquist S, Richardson A, Dickson D, Eriksen J, Robinson T, Zehr C, Dickey CA, Crook R, McGowan E, Mann D, Boeve B, Feldman H, Hutton M. Mutations in Progranulin cause tau-negative frontotemporal dementia linked to chromosome 17. Nature. 2006;442:916–919. doi: 10.1038/nature05016. [DOI] [PubMed] [Google Scholar]
- 4.Cruts M, Gijselinck I, van der Zee J, Engelborghs S, Wils H, Pirici D, Rademakers R, Vandenberghe R, Dermaut B, Martin JJ, van Duijn C, Peeters K, Sciot R, Santens P, De Pooter T, Mattheijssens M, Van den Broeck M, Cuijt I, Vennekens K, De Deyn PP, Kumar-Singh S, Van Broeckhoven C. Null mutations in progranulin cause ubiquitin-positive frontotemporal dementia linked to chromosome 17q21. Nature. 2006;442:920–924. doi: 10.1038/nature05017. [DOI] [PubMed] [Google Scholar]
- 5.Skibinski G, Parkinson NJ, Brown JM, Chakrabarti L, Lloyd SL, Hummerich H, Nielsen JE, Hodges JR, Spillantini MG, Thusgaard T, Brandner S, Brun A, Rossor MN, Gade A, Johannsen P, Sørensen SA, Gydesen S, Fisher EM, Collinge J. Mutations in the endosomal ESCRTIII-complex subunit CHMP2B in frontotemporal dementia. Nat Genet. 2005;37:806–808. doi: 10.1038/ng1609. [DOI] [PubMed] [Google Scholar]
- 6.DeJesus-Hernandez M, Mackenzie IR, Boeve BF, Boxer AL, Baker M, Rutherford NJ, Nicholson AM, Finch NA, Flynn H, Adamson J, Kouri N, Wojtas A, Sengdy P, Hsiung GY, Karydas A, Seeley WW, Josephs KA, Coppola G, Geschwind DH, Wszolek ZK, Feldman H, Knopman DS, Petersen RC, Miller BL, Dickson DW, Boylan KB, Graff-Radford NR, Rademakers R. Expanded GGGGCC hexanucleotide repeat in noncoding region of C9ORF72 causes chromosome 9p-linked FTD and ALS. Neuron. 2011;72:245–256. doi: 10.1016/j.neuron.2011.09.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Gijselinck I, Van Langenhove T, van der Zee J, Sleegers K, Philtjens S, Kleinberger G, Janssens J, Bettens K, Van Cauwenberghe C, Pereson S, Engelborghs S, Sieben A, De Jonghe P, Vandenberghe R, Santens P, De Bleecker J, Maes G, Baumer V, Dillen L, Joris G, Cuijt I, Corsmit E, Elinck E, Van Dongen J, Vermeulen S, Van den Broeck M, Vaerenberg C, Mattheijssens M, Peeters K, Robberecht W, et al. A C9orf72 promoter repeat expansion in a Flanders-Belgian cohort with disorders of the frontotemporal lobar degeneration-amyotrophic lateral sclerosis spectrum: a gene identification study. Lancet Neurol. 2012;11:54–65. doi: 10.1016/S1474-4422(11)70261-7. [DOI] [PubMed] [Google Scholar]
- 8.Renton AE, Majounie E, Waite A, Simon-Sanchez J, Rollinson S, Gibbs JR, Schymick JC, Laaksovirta H, van Swieten JC, Myllykangas L, Kalimo H, Paetau A, Abramzon Y, Remes AM, Kaganovich A, Scholz SW, Duckworth J, Ding J, Harmer DW, Hernandez DG, Johnson JO, Mok K, Ryten M, Trabzuni D, Guerreiro RJ, Orrell RW, Neal J, Murray A, Pearson J, Jansen IE, et al. A hexanucleotide repeat expansion in C9ORF72 is the cause of chromosome 9p21-linked ALS-FTD. Neuron. 2011;72:257–268. doi: 10.1016/j.neuron.2011.09.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Beck J, Poulter M, Hensman D, Rohrer JD, Mahoney CJ, Adamson G, Campbell T, Uphill J, Borg A, Fratta P, Orrell RW, Malaspina A, Rowe J, Brown J, Hodges J, Sidle K, Polke JM, Houlden H, Schott JM, Fox NC, Rossor MN, Tabrizi SJ, Isaacs AM, Hardy J, Warren JD, Collinge J, Mead S. Large C9orf72 hexanucleotide repeat expansions are seen in multiple neurodegenerative syndromes and are more frequent than expected in the UK population. Am J Hum Genet. 2013;92:345–353. doi: 10.1016/j.ajhg.2013.01.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Al-Sarraj S, King A, Troakes C, Smith B, Maekawa S, Bodi I, Rogelj B, Al-Chalabi A, Hortobagyi T, Shaw CE. p62 positive, TDP-43 negative, neuronal cytoplasmic and intranuclear inclusions in the cerebellum and hippocampus define the pathology of C9orf72-linked FTLD and MND/ALS. Acta Neuropathol. 2011;122:691–702. doi: 10.1007/s00401-011-0911-2. [DOI] [PubMed] [Google Scholar]
- 11.Boxer AL, Mackenzie IR, Boeve BF, Baker M, Seeley WW, Crook R, Feldman H, Hsiung GY, Rutherford N, Laluz V, Whitwell J, Fote D, McDade E, Molano J, Karydas A, Wojtas A, Goldman J, Mirsky J, Sengdy P, DeArmond S, Miller BL, Rademakers R. Clinical, neuroimaging and neuropathological features of a new chromosome 9p-linked FTD-ALS family. J Neurol Neurosurg Psychiatry. 2011;82:196–203. doi: 10.1136/jnnp.2009.204081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Murray ME, DeJesus-Hernandez M, Rutherford NJ, Baker M, Duara R, Graff-Radford NR, Wszolek ZK, Ferman TJ, Josephs KA, Boylan KB, Rademakers R, Dickson DW. Clinical and neuropathologic heterogeneity of c9FTD/ALS associated with hexanucleotide repeat expansion in C9ORF72. Acta Neuropathol. 2011;122:673–690. doi: 10.1007/s00401-011-0907-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Snowden JS, Rollinson S, Thompson JC, Harris JM, Stopford CL, Richardson AM, Jones M, Gerhard A, Davidson YS, Robinson A, Gibbons L, Hu Q, DuPlessis D, Neary D, Mann DM, Pickering-Brown SM. Distinct clinical and pathological characteristics of frontotemporal dementia associated with C9ORF72 mutations. Brain. 2012;135:693–708. doi: 10.1093/brain/awr355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Cairns NJ, Bigio EH, Mackenzie IRA, Neumann M, Lee VMY, Hatanpaa KJ, White CL, III, Schneider JA, Tenenholz Grinberg L, Halliday G, Duyckaerts C, Lowe JS, Holm IE, Tolnay M, Okamoto K, Yokoo H, Murayama S, Woulfe J, Munoz DG, Dickson DW, Ince PG, Trojanowski JQ, Mann DMA. Neuropathologic diagnostic and nosologic criteria for frontotemporal lobar degeneration: consensus of the Consortium for Frontotemporal Lobar Degeneration. Acta Neuropathol. 2007;114:2–22. doi: 10.1007/s00401-007-0237-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Mackenzie IRA, Baborie A, Pickering-Brown SM, Du Plessis D, Jaros E, Perry RH, Neary D, Snowden JS, Mann DMA. Heterogeneity of ubiquitin pathology in frontotemporal lobar degeneration: classification and relation to clinical phenotype. Acta Neuropathol. 2006;112:539–549. doi: 10.1007/s00401-006-0138-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.King A, Maekawa S, Bodi I, Troakes C, Al-Sarraj S. Ubiquitinated, p62 immunopositive cerebellar cortical neuronal inclusions are evident across the spectrum of TDP-43 proteinopathies but are only rarely additionally immunopositive for phosphorylation-dependent TDP-43. Neuropathology. 2011;31:239–249. doi: 10.1111/j.1440-1789.2010.01171.x. [DOI] [PubMed] [Google Scholar]
- 17.Mann DM, Rollinson S, Robinson A, Bennion Callister J, Thompson JC, Snowden JS, Gendron T, Petrucelli L, Masuda-Suzukake M, Hasegawa M, Davidson Y, Pickering-Brown S. Dipeptide repeat proteins are present in the p62 positive inclusions in patients with frontotemporal lobar degeneration and motor neurone disease associated with expansions in C9ORF72. Acta Neuropathol Commun. 2013;1:68. doi: 10.1186/2051-5960-1-68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ash PE, Bieniek KF, Gendron TF, Caulfield T, Lin WL, Dejesus-Hernandez M, van Blitterswijk MM, Jansen-West K, Paul JW, 3rd, Rademakers R, Boylan KB, Dickson DW, Petrucelli L. Unconventional translation of C9ORF72 GGGGCC expansion generates insoluble polypeptides specific to c9FTD/ALS. Neuron. 2013;77:639–646. doi: 10.1016/j.neuron.2013.02.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Mori K, Weng SM, Arzberger T, May S, Rentzsch K, Kremmer E, Schmid B, Kretzschmar HA, Cruts M, Van Broeckhoven C, Haass C, Edbauer D. The C9orf72 GGGGCC repeat is translated into aggregating dipeptide-repeat proteins in FTLD/ALS. Science. 2013;339:1335–1338. doi: 10.1126/science.1232927. [DOI] [PubMed] [Google Scholar]
- 20.Xu Z, Poidevin M, Li X, Li Y, Shu L, Nelson DL, Li H, Hales CM, Gearing M, Wingo TS, Jin P. Expanded GGGGCC repeat RNA associated with amyotrophic lateral sclerosis and frontotemporal dementia causes neurodegeneration. Proc Natl Acad Sci U S A. 2013;110:7778–7783. doi: 10.1073/pnas.1219643110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Mizielinska S, Lashley T, Norona FE, Clayton EL, Ridler CE, Fratta P, Isaacs AM. C9orf72 frontotemporal lobar degeneration is characterised by frequent neuronal sense and antisense RNA foci. Acta Neuropathol. 2013;126:845–858. doi: 10.1007/s00401-013-1200-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Mori K, Arzberger T, Grasser FA, Gijselinck I, May S, Rentzsch K, Weng SM, Schludi MH, van der Zee J, Cruts M, Van Broeckhoven C, Kremmer E, Kretzschmar HA, Haass C, Edbauer D. Bidirectional transcripts of the expanded C9orf72 hexanucleotide repeat are translated into aggregating dipeptide repeat proteins. Acta Neuropathol. 2013;126:881–894. doi: 10.1007/s00401-013-1189-3. [DOI] [PubMed] [Google Scholar]
- 23.Zu T, Liu Y, Banez-Coronel M, Reid T, Pletnikova O, Lewis J, Miller TM, Harms MB, Falchook AE, Subramony SH, Ostrow LW, Rothstein JD, Troncoso JC, Ranum LP. Proc Natl Acad Sci USA. 2013. RAN proteins and RNA foci from antisense transcripts in C9ORF72 ALS and frontotemporal dementia. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Mackenzie IR, Arzberger T, Kremmer E, Troost D, Lorenzl S, Mori K, Weng SM, Haass C, Kretzschmar HA, Edbauer D, Neumann M. Dipeptide repeat protein pathology in C9ORF72 mutation cases: clinico-pathological correlations. Acta Neuropathol. 2013;126:859–879. doi: 10.1007/s00401-013-1181-y. [DOI] [PubMed] [Google Scholar]
- 25.Finch N, Carrasquillo MM, Baker M, Rutherford NJ, Coppola G, Dejesus-Hernandez M, Crook R, Hunter T, Ghidoni R, Benussi L, Crook J, Finger E, Hantanpaa KJ, Karydas AM, Sengdy P, Gonzalez J, Seeley WW, Johnson N, Beach TG, Mesulam M, Forloni G, Kertesz A, Knopman DS, Uitti R, White CL, 3rd, Caselli R, Lippa C, Bigio EH, Wszolek ZK, Binetti G, et al. TMEM106B regulates progranulin levels and the penetrance of FTLD in GRN mutation carriers. Neurology. 2011;76:467–474. doi: 10.1212/WNL.0b013e31820a0e3b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Gallagher MD, Suh E, Grossman M, Elman L, McCluskey L, Van Swieten JC, Al-Sarraj S, Neumann M, Gelpi E, Ghetti B, Rohrer JD, Halliday G, Van Broeckhoven C, Seilhean D, Shaw PJ, Frosch MP, Alafuzoff I, Antonell A, Bogdanovic N, Brooks W, Cairns NJ, Cooper-Knock J, Cotman C, Cras P, Cruts M, De Deyn PP, Decarli C, Dobson-Stone C, Engelborghs S, Fox N, et al. Acta Neuropathol. 2014. TMEM106B is a genetic modifier of frontotemporal lobar degeneration with C9orf72 hexanucleotide repeat expansions. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.van Blitterswijk M, Mullen B, Nicholson AM, Bieniek KF, Heckman MG, Baker MC, Dejesus-Hernandez M, Finch NA, Brown PH, Murray ME, Hsiung GY, Stewart H, Karydas AM, Finger E, Kertesz A, Bigio EH, Weintraub S, Mesulam M, Hatanpaa KJ, White Iii CL, Strong MJ, Beach TG, Wszolek ZK, Lippa C, Caselli R, Petrucelli L, Josephs KA, Parisi JE, Knopman DS, Petersen RC, et al. Acta Neuropathol. 2014. TMEM106B protects C9ORF72 expansion carriers against frontotemporal dementia. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Van Deerlin VM, Sleiman PM, Martinez-Lage M, Chen-Plotkin A, Wang LS, Graff-Radford NR, Dickson DW, Rademakers R, Boeve BF, Grossman M, Arnold SE, Mann DM, Pickering-Brown SM, Seelaar H, Heutink P, van Swieten JC, Murrell JR, Ghetti B, Spina S, Grafman J, Hodges J, Spillantini MG, Gilman S, Lieberman AP, Kaye JA, Woltjer RL, Bigio EH, Mesulam M, Al-Sarraj S, Troakes C, et al. Common variants at 7p21 are associated with frontotemporal lobar degeneration with TDP-43 inclusions. Nat Genet. 2010;42:234–239. doi: 10.1038/ng.536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.van der Zee J, Van Langenhove T, Kleinberger G, Sleegers K, Engelborghs S, Vandenberghe R, Santens P, Van den Broeck M, Joris G, Brys J, Mattheijssens M, Peeters K, Cras P, De Deyn PP, Cruts M, Van Broeckhoven C. TMEM106B is associated with frontotemporal lobar degeneration in a clinically diagnosed patient cohort. Brain. 2011;134:808–815. doi: 10.1093/brain/awr007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Bernardi L, Maletta RG, Tomaino C, Smirne N, Di Natale M, Perri M, Longo T, Colao R, Curcio SA, Puccio G, Mirabelli M, Kawarai T, Rogaeva E, St George Hyslop PH, Passarino G, De Benedictis G, Bruni AC. The effects of APOE and tau gene variability on risk of frontotemporal dementia. Neurobiol Aging. 2006;27:702–709. doi: 10.1016/j.neurobiolaging.2005.03.008. [DOI] [PubMed] [Google Scholar]
- 31.Fabre SF, Forsell C, Viitanen M, Sjögren M, Wallin A, Blennow K, Blomberg M, Andersen C, Wahlund LO, Lannfelt L. Clinic-based cases with frontotemporal dementia show increased cerebrospinal fluid tau and high apolipoprotein E epsilon4 frequency, but no tau gene mutations. Exp Neurol. 2001;168:413–418. doi: 10.1006/exnr.2000.7613. [DOI] [PubMed] [Google Scholar]
- 32.Rosso SM, Roks G, Cruts M, van Broeckhoven C, Heutink P, van Duijn CM, van Swieten JC. Apolipoprotein E4 in the temporal variant of frontotemporal dementia. J Neurol Neurosurg Psychiatry. 2002;72:820. doi: 10.1136/jnnp.72.6.820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Stevens M, van Duijn CM, Kamphorst W, de Knijff P, Heutink P, van Gool WA, Scheltens P, Ravid R, Oostra BA, Niermeijer MF, van Swieten JC. Familial aggregation in frontotemporal dementia. Neurology. 1998;50:1541–1545. doi: 10.1212/WNL.50.6.1541. [DOI] [PubMed] [Google Scholar]
- 34.Mann DMA, Iwatsubo T, Pickering-Brown SM, Owen F, Saido TC, Perry RH. Preferential deposition of amyloid ß protein (Aß) in the form Aß40 in Alzheimer’s disease is associated with a gene dosage effect of the apolipoprotein E E4 allele. Neurosci Lett. 1997;221:81–84. doi: 10.1016/S0304-3940(96)13294-8. [DOI] [PubMed] [Google Scholar]
- 35.Allen N, Robinson AC, Snowden S, Davidson YS, Mann DMA. Patterns of cerebral amyloid angiopathy define histopathological phenotypes in Alzheimer’s disease. Neuropathol Appl Neurobiol. 2014;40:136–148. doi: 10.1111/nan.12070. [DOI] [PubMed] [Google Scholar]
- 36.Pickering-Brown SM, Richardson AMT, Snowden JS, McDonagh AM, Burns A, Braude W, Baker M, Liu W-K, Yen S-H, Hardy J, Hutton M, Davies Y, Allsop D, Craufurd D, Neary D, Mann DMA. Inherited frontotemporal dementia in 9 British families associated with intronic mutations in the tau gene. Brain. 2002;125:732–751. doi: 10.1093/brain/awf069. [DOI] [PubMed] [Google Scholar]
- 37.Pickering-Brown SM, Baker M, Gass J, Boeve BF, Loy CT, Brooks WS, Mackenzie IR, Martins RN, Kwok JB, Halliday GM, Kril JJ, Schofield PR, Mann DM, Hutton M. Mutations in progranulin explain atypical phenotypes with variants in MAPT. Brain. 2006;129:3124–3126. doi: 10.1093/brain/awl289. [DOI] [PubMed] [Google Scholar]
- 38.Brun A, Englund E, Gustafson L, Passant U, Mann DMA, Neary D, Snowden JS. Clinical, neuropsychological and neuropathological criteria of fronto-temporal dementia. J Neurol Neurosurg Psychiatry. 1994;57:416–418. doi: 10.1136/jnnp.57.4.416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Neary D, Snowden JS, Gustafson L, Passant U, Stuss D, Black S, Freedman M, Kertesz A, Robert PH, Albert M, Boone K, Miller BL, Cummings J, Benson DF. Frontotemporal lobar degeneration: a consensus on clinical diagnostic criteria. Neurology. 1998;51:1546–1554. doi: 10.1212/WNL.51.6.1546. [DOI] [PubMed] [Google Scholar]
- 40.Harris JM, Gall C, Thompson JC, Richardson AMT, Neary D, du Plessis D, Pal P, Mann DMA, Snowden JS, Jones M. Sensitivity and specificity of FTDC criteria for behavioral variant Frontotemporal dementia. Neurology. 2013;80:1881–1887. doi: 10.1212/WNL.0b013e318292a342. [DOI] [PubMed] [Google Scholar]
- 41.Rascovsky K, Hodges JR, Knopman D, Mendez MF, Kramer JH, Neuhaus J, van Swieten JC, Seelaar H, Dopper EG, Onyike CU, Hillis AE, Josephs KA, Boeve BF, Kertesz A, Seeley WW, Rankin KP, Johnson JK, Gorno-Tempini ML, Rosen H, Prioleau-Latham CE, Lee A, Kipps CM, Lillo P, Piguet O, Rohrer JD, Rossor MN, Warren JD, Fox NC, Galasko D, Salmon DP, et al. Sensitivity of revised diagnostic criteria for the behavioural variant of frontotemporal dementia. Brain. 2011;134:2456–2477. doi: 10.1093/brain/awr179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Brooks BR. El Escorial World Federation of Neurology criteria for the diagnosis of amyotrophic lateral sclerosis. Subcommittee on Motor Neuron Diseases/Amyotrophic Lateral Sclerosis of the World Federation of Neurology Research Group on Neuromuscular Diseases and the El Escorial “Clinical limits of amyotrophic lateral sclerosis” workshop contributors. J NeurolSci. 1994;124(suppl):96–107. doi: 10.1016/0022-510x(94)90191-0. [DOI] [PubMed] [Google Scholar]
- 43.Mackenzie IRA, Neumann M, Baborie A, Sampathu DM, Du Plessis D, Jaros E, Perry RH, Trojanowski JQ, Mann DMA, Lee VM-Y. A harmonized classification system for FTLD-TDP pathology. Acta Neuropathol. 2011;122:111–113. doi: 10.1007/s00401-011-0845-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Davidson YS, Robinson AC, Snowden JS, Mann DM. Pathological assessments for the presence of hexanucleotide repeat expansions in C9ORF72 in Alzheimer's disease. Acta Neuropathol Commun. 2013;1:50. doi: 10.1186/2051-5960-1-50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Wenham PR, Price WH, Blundell G. Apolipoprotein E genotyping by one-stage PCR. Lancet. 1991;337:1158–1159. doi: 10.1016/0140-6736(91)92823-K. [DOI] [PubMed] [Google Scholar]
- 46.Mahoney CJ, Beck J, Rohrer JD, Lashley T, Mok K, Shakespeare T, Yeatman T, Warrington EK, Schott JM, Fox NC, Rossor MN, Hardy J, Collinge J, Revesz T, Mead S, Warren JD. Frontotemporal dementia with the C9ORF72 hexanucleotide repeat expansion: clinical, neuroanatomical and neuropathological features. Brain. 2012;135:736–750. doi: 10.1093/brain/awr361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Whitwell JL, Weigand SD, Boeve BF, Senjem ML, Gunter JL, DeJesus-Hernandez M, Rutherford NJ, Baker M, Knopman DS, Wszolek ZK, Parisi JE, Dickson DW, Petersen RC, Rademakers R, Jack CR, Jr, Josephs KA. Neuroimaging signatures of frontotemporal dementia genetics: C9ORF72, tau, progranulin and sporadics. Brain. 2012;135:794–806. doi: 10.1093/brain/aws001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Galimberti D, Fenoglio C, Serpente M, Villa C, Bonsi R, Arighi A, Fumagalli GG, Del Bo R, Bruni AC, Anfossi M, Clodomiro A, Cupidi C, Nacmias B, Sorbi S, Piaceri I, Bagnoli S, Bessi V, Marcone A, Cerami C, Cappa SF, Filippi M, Agosta F, Magnani G, Comi G, Franceschi M, Rainero I, Giordana MT, Rubino E, Ferrero P, Rogaeva E, et al. Autosomal dominant Frontotemporal Lobar degeneration due to C9ORF72 hexanucleotide repeat expansion: late onset psychotic clinical presentation. Biol Psychiatr. 2013;74:384–91. doi: 10.1016/j.biopsych.2013.01.031. [DOI] [PubMed] [Google Scholar]
- 49.Snowden JS, Harris J, Richardson A, Rollinson S, Thompson JC, Neary D, Mann DMA, Pickering-Brown S. Frontotemporal dementia with amyotrophic lateral sclerosis: a clinical comparison of patients with and without repeat expansions in C9ORF72. Amyotrophic Lateral Sclerosis and Frontotemporal Degeneration. 2013;14:172–176. doi: 10.3109/21678421.2013.765485. [DOI] [PubMed] [Google Scholar]
- 50.Cacace R, Van Cauwenberghe C, Bettens K, Gijselinck I, van der Zee J, Engelborghs S, Vandenbulcke M, Van Dongen J, Baumer V, Dillen L, Mattheijssens M, Peeters K, Cruts M, Vandenberghe R, De Deyn PP, Van Broeckhoven C, Sleegers K. C9orf72 G4C2 repeat expansions in Alzheimer's disease and mild cognitive impairment. Neurobiol Aging. 2013;34(1712):e1711–1717. doi: 10.1016/j.neurobiolaging.2012.12.019. [DOI] [PubMed] [Google Scholar]
- 51.Harms M, Benitez BA, Cairns N, Cooper B, Cooper P, Mayo K, Carrell D, Faber K, Williamson J, Bird T, Diaz-Arrastia R, Foroud TM, Boeve BF, Graff-Radford NR, Mayeux R, Chakraverty S, Goate AM, Cruchaga C, NIA-LOAD/NCRAD Family Study Consortium C9orf72 hexanucleotide repeat expansions in clinical Alzheimer disease. JAMA Neurol. 2013;70:736–741. doi: 10.1001/2013.jamaneurol.537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Kohli MA, John-Williams K, Rajbhandary R, Naj A, Whitehead P, Hamilton K, Carney RM, Wright C, Crocco E, Gwirtzman HE, Lang R, Beecham G, Martin ER, Gilbert J, Benatar M, Small GW, Mash D, Byrd G, Haines JL, Pericak-Vance MA, Züchner S. Repeat expansions in the C9ORF72 gene contribute to Alzheimer's disease in Caucasians. Neurobiol Aging. 2013;34(1519):e1515–1512. doi: 10.1016/j.neurobiolaging.2012.10.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Cooper-Knock J, Frolov A, Highley JR, Charlesworth G, Kirby J, Milano A, Hartley J, Ince PG, McDermott CJ, Lashley T, Revesz T, Shaw PJ, Wood NW, Bandmann O. C9ORF72 expansions, parkinsonism, and Parkinson’s disease. Neurology. 2013;81:808–811. doi: 10.1212/WNL.0b013e3182a2cc38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Ferrari R, Mok K, Moreno JH, Cosentino S, Goldman J, Pietrini P, Mayeux R, Tierney MC, Kapogiannis D, Jicha GA, Murrell JR, Ghetti B, Wassermann EM, Grafman J, Hardy J, Huey ED, Momeni P. Screening for C9ORF72 repeat expansion in FTLD. Neurobiol Aging. 2012;33(1850):e1851–1811. doi: 10.1016/j.neurobiolaging.2012.02.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.van Blitterswijk M, Baker M, DeJesus-Hernandez M, Ghidoni R, Benussi L, Finger E, Hsiung G-Y R, Kelley BJ, Murray ME, Rutherford NJ, Brown PE, Ravenscroft T, Mullen B, Ash PEA, Bienik KF, Hatanpaa KJ, McCarty Wood E, Coppola G, Bigio EH, Lippa C, Strong MJ, Beach TG, Knopman DS, Huey ED, Mesulam M, Bird T, White CL, III, Kertesz A, Geschwind DH, Van Deerlin VM, et al. C9ORF72 repeat expansions in cases with previously identified pathogenic mutations. Neurology. 2013;81:1–10. doi: 10.1212/WNL.0b013e3182a8250c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.King A, Al-Sarraj S, Troakes C, Smith BN, Maekawa S, Iovino M, Spillantini MG, Shaw C. Mixed tau, TDP-43 and p62 pathology in FTLD associated with a C9ORF72 repeat expansion and p.Ala239Thr MAPT (tau) variant. Acta Neuropathol. 2012;125:303–310. doi: 10.1007/s00401-012-1050-0. [DOI] [PubMed] [Google Scholar]
- 57.Lashley T, Rohrer JD, Mahoney C, Gordon E, Beck J, Mead S, Warren J, Rossor M, Revesz T. Neuropathol Appl Neurobiol. 2013. A pathogenic progranulin mutation and C9orf72 repeat expansion in a family with frontotemporal dementia. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Lee YB, Chen HJ, Peres JN, Gomez-Deza J, Attig J, Stalekar M, Troakes C, Nishimura AL, Scotter EL, Vance C, Adachi Y, Sardone V, Miller JW, Smith BN, Gallo JM, Ule J, Hirth F, Rogelj B, Houart C, Shaw CE. Cell Rep. 2013. Hexanucleotide repeats in ALS/FTD form length-dependent RNA foci, sequester RNA binding Proteins, and are neurotoxic. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Levine TP, Daniels RD, Gatta AT, Wong LH, Hayes MJ. The product of C9orf72, a gene strongly implicated in neurodegeneration, is structurally related to DENN Rab-GEFs. Bioinformatics. 2013;29:499–503. doi: 10.1093/bioinformatics/bts725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Ferrari R, Hernandez DG, Nalls MA, Rohrer JD, Ramasamy A, Kwok JBJ, Dobson-Stone C, Brooks WS, Schofield PR, Halliday GM, Hodges JR, Piguet O, Bartley L, Thompson E, Haan E, Hernández I, Ruiz A, Boada M, Borroni B, Padovani A, Cruchaga C, Cairns NJ, Benussi L, Binetti G, Ghidoni R, Forloni G, Galimberti D, Fenoglio C, Serpente M, Scarpini E, et al. Lancet Neurol. 2014. Genome wide association study reveals lysosomal and immune system involvement in frontotemporal dementia. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Bultema JJ, Ambrosio AL, Burek CL, Di Pietro SM. BLOC-2, AP-3, and AP-1 proteins function in concert with Rab38 and Rab32 proteins to mediate protein trafficking to lysosome-related organelles. J Biol Chem. 2012;287:19550–19563. doi: 10.1074/jbc.M112.351908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Wasmeier C, Romao M, Plowright L, Bennett DC, Raposo G, Seabra MC. Rab38 and Rab32 control post-Golgi trafficking of melanogenic enzymes. J Cell Biol. 2006;175:271–281. doi: 10.1083/jcb.200606050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Seto S, Tsujimura K, Koide Y. Rab GTPAses regulating phagosome maturation are differentially recruited to mycobacterial phagosomes. Traffic. 2011;12:407–420. doi: 10.1111/j.1600-0854.2011.01165.x. [DOI] [PubMed] [Google Scholar]
- 64.Hu F, Padukkavidana T, Vægter CB, Brady OA, Zheng Y, Mackenzie IR, Feldman HH, Nykjaer A, Strittmatter SM. Sortilin-mediated endocytosis determines levels of the frontotemporal dementia protein, progranulin. Neuron. 2010;68:654–667. doi: 10.1016/j.neuron.2010.09.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Brady OA, Zheng Y, Murphy K, Huang M, Hu F. The frontotemporal lobar degeneration risk factor, TMEM106B, regulates lysosomal morphology and function. Hum Mol Genet. 2013;22:685–695. doi: 10.1093/hmg/dds475. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1: Selected clinical, pathological and genetic details for 40 cases of Frontotemporal Lobar degeneration and 27 cases of Motor Neurone disease. (XLSX 18 KB)


