Abstract
Hemangioblastomas (HBMs) in the cerebellopontine angle (CPA) have rarely been reported. When they are within the CPA, they may be misdiagnosed as vestibular schwannoma (VS) or cystic meningioma. Therefore, differential diagnosis is important for the safe treatment of the lesion. Large solid HBMs, similar to intracranial arteriovenous malformations (AVMs), are difficult to surgically remove from an eloquent area because of their location and hypervascularity. We report a case of an HBM in the CPA, which manifested as a hearing impairment or VS. Similar to AVM surgery, the tumor was widely opened and removed en bloc without a new neurological complication using the modified transcondylar fossa approach without resection of the jugular tubercle. Accurate diagnosis, pre-operative embolization, and a tailored approach were essential for the safe treatment of the HBM in the CPA.
Keywords: Hemangioblastoma, Cerebellopontine angle
INTRODUCTION
Hemangioblastoma (HBM), which is a highly vascular tumor of the central nervous system, accounts for 1.5-2.5% of all intracranial tumors and 7-12% of posterior fossa tumors in the cerebellum, brainstem, and spinal cord [1,9,10]. Cases of HBM in the cerebellopontine angle (CPA) have rarely been reported [2,7,8,9]; only 10 cases have been reported in English literature [2,3,4,5,7,8,9]. An HBM looks like an extra-axial lesion, such as a vestibular schwannoma (VS) or a meningioma, on a radiological image. Seventy to 75% of HBMs have cystic or multi-cystic forms [2]. The characteristics of solid variants of HBMs are similar to those of intracranial arteriovenous malformations (AVMs) [4,5]. Internal decompression and piecemeal resection may have devastating complications. Safe resection of large solid HBMs especially in the CPA requires surgical techniques similar to those for AVM surgery: pre-operative embolization, wide exposure, circumferential dissection, and en bloc removal. We present a case of HBM in the CPA, in which complete resection was achieved without morbidity using the modified transcondylar fossa approach (TCFA).
CASE REPORT
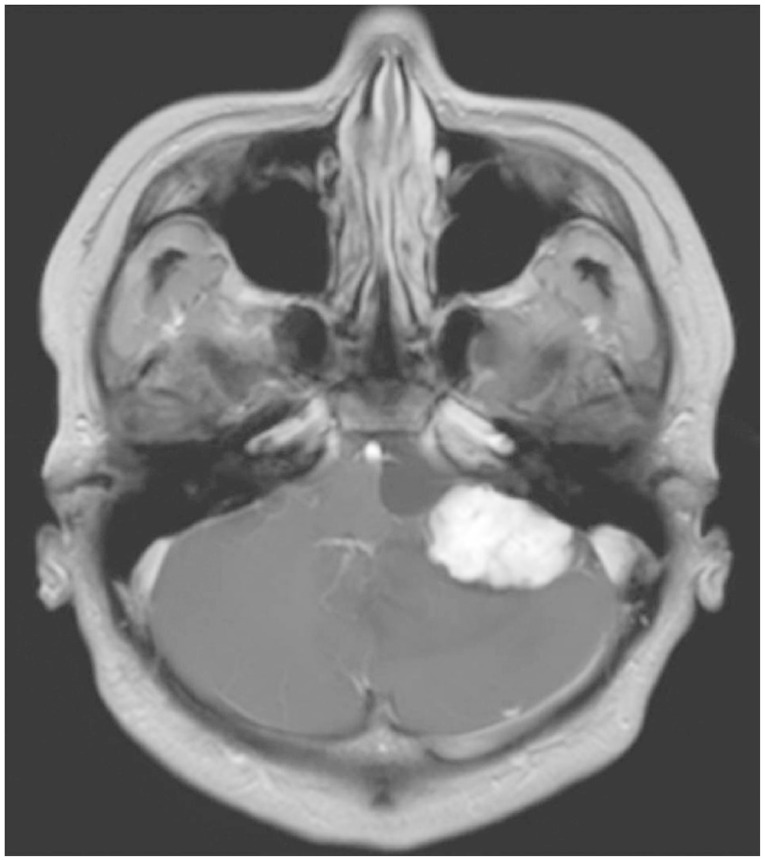
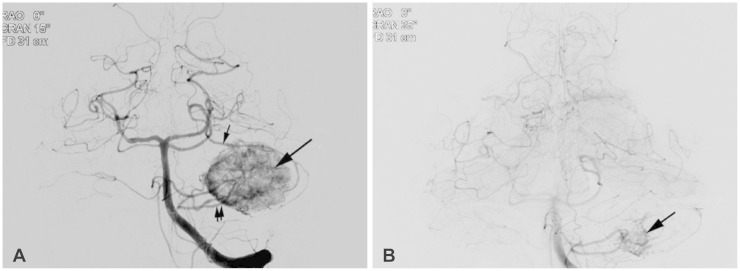
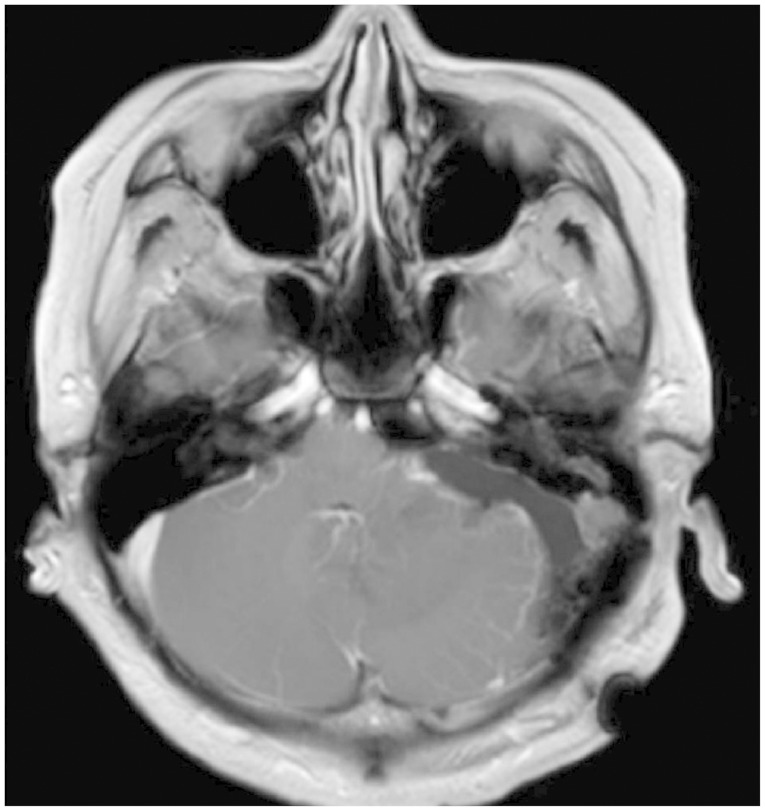
A 31-year-old male was admitted to our hospital with headache and hearing difficulty in his left ear. His neurological examination upon his admission revealed no symptoms and signs of cerebellar dysfunction and other cranial nerve dysfunctions. His neuro-otological test showed no serviceable hearing in the affected ear. The magnetic resonance imaging (MRI) showed a well-enhancing lesion in the left CPA with a peritumoral cyst on the medial portion of the lesion (Fig. 1). The cerebral angiogram (Fig. 2A) showed the lesion fed by the left superior cerebellar artery (SCA) and the anterior inferior cerebellar artery (AICA). They were superselected with flow-directed microcatheters (Marathon™, EV3, Irvine, CA, USA), and the distal feeders were embolized with N-butyl cyanoacrylate (0.4 cc in the SCA and 0.6 cc in the AICA). The angiogram after the embolization revealed a 90% reduction in the tumor vascularity (Fig. 2B). Five days after the embolization, surgical resection was performed. The patient was positioned lateral to the head that was turned towards the contralateral side. After a curvilinear skin incision was made, TCFA was performed under navigation guided with electrophysiological monitoring. The tumor was bright red and was associated with intense vascularity (Fig. 3A). Intra-operative indocyanine green video angiography was performed to identify the feeders (Fig. 3B). A peritumoral cyst with a brownish fluid was observed on the medial portion of the tumor, at the prepontine cistern. The tumor was dissected while the draining vein was circumferentially preserved, and then en bloc resection was achieved (Fig. 3C). Intra-operative indocyanine green videoangiography was used to confirm the angio-architecture of the tumor. The MRI obtained a day post-operatively showed complete resection of the tumor (Fig. 4). The post-operative neurological examination did not reveal a neurological complication, such as facial palsy or auditory function and lower cranial nerve symptoms. The pathological examination revealed a highly vascular neoplasm composed of atypical stromal cells that consisted of HBMs (Fig. 5). The index of the Ki-67 protein was low-approximately 3%.
Fig. 1.
Pre-operative gadolinium-enhanced magnetic resonance images showing the highly enhanced mass in the cerebellopontine angle with multiple flow voids and peritumoral edema.
Fig. 2.
Preoperative angiographic imaging showing the angio-arcitecture of the tumor. A: Left vertebral artery angiogram anterior-posterior views of the tumor (large arrow) fed by the superior cerebellar artery (small arrow) and the anterior inferior cerebellar artery (small double arrow). B: After the embolization, angiogram showing a 90% reduction in the tumor vascularity (arrow).
Fig. 3.
These photographs showing the intraoperative videoimaging (A and C), and intraoperative indocyanine green videoangiography (B). A: Operative photograph after the craniotomy showing the orange tumor. Large arrow: mastoid bone; small black arrows: tumor; and small white arrows: transverse sinus. B: Indocyanine green videoangiography showing the hypervascular tumor (arrow) fed by the anterior inferior cerebellar artery (double arrow). C: Photograph of the coagulated draining vein. Arrow: draining vein.
Fig. 4.
Gadolinium-enhanced magnetic resonance images obtained a day post-operatively confirming complete resection of the hemangioblastoma.
Fig. 5.
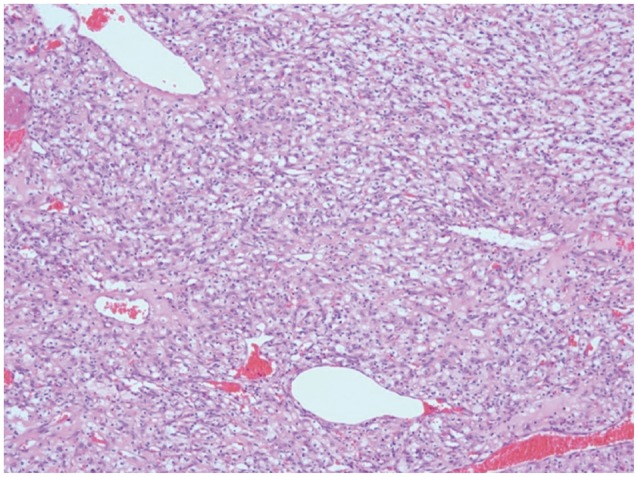
Photograph of a highly vascular tumor consisting of numerous blood vessels and intervening stromal cells (hematoxylineosin, magnification ×100).
DISCUSSION
Hemangioblastomas of the posterior fossa typically arise within the substance of the cerebellum and the brainstem [2]. However, unusual HBMs involving the cranial nerve and the CPA have rarely been reported [2,3,4,5,7,8,9,10]. HBMs are generally intra-axial in origin, so the CPA is very rarely involved [2,3,4,5,7,8,9]. In English literature, only 10 cases of CPA HBM have been reported [2,7]. In this case, the tumor was placed in the CPA. However, we identified the tumor attached to the surface of the cerebellum around the foramen of Luschka. Therefore, that areas was seemingly the site of origin. In our opinion, the tumor derived from the cerebellar pial vessels was intra-axial in origin. However, the tumor growth was directed into the CPA, an extra-axial location, and not into the cerebellar parenchyme. Although 6-10% of all intracranial tumors were found in the CPA, most of them are VSs and meningiomas [2,3,4]. Because of the differences in the surgical strategies used for these tumors, differential diagnosis is crucial to the safe management of HBMs. Classically, about two-thirds of HBMs appear as well-circumscribed cystic masses with hypervascular mural nodules [2]. Radiological findings of HBMs show multiple signal voids in the lesion, a peritumoral cyst, and peritumoral edema. Although there are no histological differences between the cystic and solid tumor subtypes [9], solid subtypes have been regarded as difficult to treat surgically because of their AVM-like characteristics [4,7,9]. Usually, after aspiration of the cystic fluid, sufficient exposure for surgery can be achieved in cystic HBMs. However, solid subtypes may require a more extended approach to achieve an adequate work space, not only due to their solid nature but also due to their AVM-like character. Therefore, we used modified TCFA. Various approaches that included the retrosigmoid [5,7,9], translabyrinthine [2], and transcochlear approaches [4] have been used with or without pre-operative embolization. Dow et al. [4] used the trans-cochlear far lateral approach in patients with large (>3 cm) solid HBMs in the CPA. This approach provided a wide enough exposure to achieve early control of proximal feeding vessels and to dissect the lesion circumferentially [4]. For cystic HBMs in the CPA, Bush et al. [2] removed the tumor via the translabyrinthine approach because they first thought the lesion was an atypical cystic VS. They suggested that had an HBM been considered, the suboccipital approach might have provided adequate exposure and potentially preserved hearing [2]. To achieve adequate exposure, we used modified TCFA. Compared to retrosigmoid craniotomy, TCFA extended the bony opening to approach the midline lower clival lesion by drilling the bone over the condylar fossa and the jugular tubercle [6]. For midline access in the CPA, the jugular tubercle should be partially resected in the conventional TCFA [6]. However, in our case, the tumor already made the space. Therefore, careful approach of the proximal feeding vessels such as AICA and their early control were possible without resection of the jugular tubercle. The more significantly extended bony removal provided a wider space for the circumferential dissection. To reduce the vascularity of the tumor, pre-operative embolization [11] was carried out in our case. To reduce the vascularity, Kamitani et al. [5] used pre-operative radiosurgery in a 3.5 cm-diameter solid hypervascular HBM. They reported that radiosurgery nine months before craniotomy significantly reduced the vascularity and subsequently enabled complete and safe tumor removal [5]. Solid HBMs are minor subtypes that mimic AVMs, and for which a careful strategy that includes pre-operative embolization and/or radiosurgery should be considered, besides a tailored approach, as with surgery on AVMs.
In conclusion, HBMs can present within the CPA, mimicking other pathologies such as VS and meningioma. Although unusual, HBMs should be considered in differential diagnosis of CPA tumors. Large solid HBM subtypes are similar in clinical character to intracranial AVMs. Therefore, for the safe resection of the lesion, a specific strategy for reducing the vascularity of the tumor and the tailored skull base approach should be considered.
Footnotes
The authors have no financial conflicts of interest.
References
- 1.Amano T, Tokunaga S, Shono T, et al. Cerebellar hemangioblastoma manifesting as hearing disturbance. Neurol Med Chir (Tokyo) 2009;49:418–420. doi: 10.2176/nmc.49.418. [DOI] [PubMed] [Google Scholar]
- 2.Bush ML, Pritchett C, Packer M, Ray-Chaudhury A, Jacob A. Hemangioblastoma of the cerebellopontine angle. Arch Otolaryngol Head Neck Surg. 2010;136:734–738. doi: 10.1001/archoto.2010.98. [DOI] [PubMed] [Google Scholar]
- 3.Deb P, Sahni H, Bhatoe HS. Cystic angiomatous meningioma in the cerebellopontine angle mimicking hemangioblastoma. J Cancer Res Ther. 2010;6:560–563. doi: 10.4103/0973-1482.77074. [DOI] [PubMed] [Google Scholar]
- 4.Dow GR, Sim DW, O'Sullivan MG. Excision of large solid haemangioblastomas of the cerebellopontine angle by a skull base approach. Br J Neurosurg. 2002;16:168–171. doi: 10.1080/02688690220131804. [DOI] [PubMed] [Google Scholar]
- 5.Kamitani H, Hirano N, Takigawa H, et al. Attenuation of vascularity by preoperative radiosurgery facilitates total removal of a hypervascular hemangioblastoma at the cerebello-pontine angle: case report. Surg Neurol. 2004;62:238–243. doi: 10.1016/j.surneu.2003.09.030. discussion 243-4. [DOI] [PubMed] [Google Scholar]
- 6.Matsushima T, Kawashima M, Masuoka J, Mineta T, Inoue T. Transcondylar fossa (supracondylar transjugular tubercle) approach: anatomic basis for the approach, surgical procedures, and surgical experience. Skull Base. 2010;20:83–91. doi: 10.1055/s-0029-1242193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Nair BR, Joseph V, Chacko G, Keshava SN. Giant solid hemangioblastoma of the cerebellopontine angle: a technically challenging case. Neurol India. 2014;62:228–229. doi: 10.4103/0028-3886.132450. [DOI] [PubMed] [Google Scholar]
- 8.Qiao PF, Niu GM, Han XD. Hemangioblastoma originating from the right cerebellopontine angle. Neurosciences (Riyadh) 2011;16:372–374. [PubMed] [Google Scholar]
- 9.Rachinger J, Buslei R, Prell J, Strauss C. Solid haemangioblastomas of the CNS: a review of 17 consecutive cases. Neurosurg Rev. 2009;32:37–47. doi: 10.1007/s10143-008-0166-0. discussion 47-8. [DOI] [PubMed] [Google Scholar]
- 10.Roberti F, Jones RV, Wright DC. Cranial nerve hemangioblastomas. Report of a rare case and review of literature. Surg Neurol. 2007;67:640–646. doi: 10.1016/j.surneu.2006.08.085. discussion 646. [DOI] [PubMed] [Google Scholar]
- 11.Sakamoto N, Ishikawa E, Nakai Y, et al. Preoperative endovascular embolization for hemangioblastoma in the posterior fossa. Neurol Med Chir (Tokyo) 2012;52:878–884. doi: 10.2176/nmc.52.878. [DOI] [PubMed] [Google Scholar]