Abstract
Objective
HIV patients on antiretroviral therapy (HIV/ART) exhibit a unique atherogenic dyslipidemic profile with hypertriglyceridemia (HTG) and low plasma concentrations of high density lipoprotein-cholesterol (HDL-C). In the Heart Positive Study of HIV/ART patients, a hypolipidemic therapy of fenofibrate, niacin, diet, and exercise reduced HTG and plasma non-HDL-C concentrations and raised plasma HDL-C and adiponectin concentrations. We tested the hypothesis that HIV/ART HDL have abnormal structures and properties and are dysfunctional.
Approach and Results
Hypolipidemic therapy reduced the TG contents of LDL and HDL. At baseline, HIV/ART low density lipoproteins (LDL) were more triglyceride (TG)-rich and HDL were more TG- and cholesteryl ester (CE)-rich than the corresponding lipoproteins from normolipidemic (NL) subjects. Very low density lipoproteins, LDL and HDL were larger than the corresponding lipoproteins from NL subjects; HIV/ART HDL were less stable than NL HDL. HDL-[3H]CE uptake by Huh7 hepatocytes was used to assess HDL functionality. HIV/ART plasma were found to contain significantly less competitive inhibition activity for hepatocyte HDL-CE uptake than did NL plasma (p<0.001).
Conclusion
Compared to NL subjects, lipoproteins from HIV/ART patients are larger and more neutral lipid-rich, and their HDL are less stable and less receptor-competent. Based on this work and previous studies of lipase activity in HIV, we present a model in which plasma lipolytic activities and/or hepatic CE uptake are impaired in HIV/ART patients. These findings provide a rationale to determine whether the distinctive lipoprotein structure, properties and function of HIV/ART HDL predict atherosclerosis as assessed by carotid artery intimal medial thickness.
Keywords: HIV dyslipidemia, HDL function, lipoprotein composition, hepatocyte CE uptake
Introduction
Human immunodeficiency virus (HIV)-infected patients receiving antiretroviral therapy (ART) present with a dyslipidemia and insulin resistance1 that places them at a high risk for accelerated cardiovascular disease (CVD).2-6 The increased CVD among HIV/ART patients is a persistent public health challenge for which current therapies are not adequate. HIV/ART dyslipidemia comprises hypertriglyceridemia, low plasma high-density lipoprotein (HDL)-cholesterol (HDL-C), and elevated plasma non-HDL-C concentrations.7, 8 Other distinctive pathogenic features of HIV/ART dyslipidemia include hypoadiponectinemia,9, 10 increased lipolysis11, 12 and hepatic free fatty acids (FFA) flux, which increases very low-density lipoprotein synthesis,13 in a way that is mechanistically linked to defects in HDL metabolism.8, 14 Given that the low plasma HDL-C concentrations place HIV/ART patients at a high CVD risk, we tested the hypothesis that the compositions and properties of HDL from these patients were distinct from control subjects in a way that impaired HDL metabolism. This hypothesis was tested by studying dyslipidemic HIV/ART patients with and without hypertriglyceridemia, and control non HIV normolipidemic (NL) subjects.
Materials and Methods
Materials and Methods are available in the online-only Data Supplement.
Results
Altered Plasma Lipid Profiles in HIV/ART
Lipoprotein compositions were determined in a large subset of the Heart Positive HIV patients15 (predominantly those who completed the 24 week trial). Subset plasma lipid profiles simulated those of the entire Heart Positive patient set (Supplemental Table I). In all five treatment groups, patient plasma lipid profiles at baseline satisfied the lipid criteria for metabolic syndrome (MetS), i.e., hypertriglyceridemia and a low plasma HDL-C concentration. Moreover, the HDL-C levels were lower and those for TG were higher than NL control. Analysis of variance confirmed that the lipid profiles at baseline for the five groups, assigned at random upon enrollment in the Heart Positive Study, were not significantly different. As reported for the entire Heart Positive study16 treatment with fenofibrate + D&E (Group 3), niacin + D&E (Group 4) and the combined therapy of fenofibrate + niacin + D&E (Group 5) significantly reduced plasma TG, with median reductions of 35 – 42 %. Treatments including niacin with or without fenofibrate increased median HDL-C by 21 – 24%. Treatments including fibrate with or without niacin reduced median non-HDL-C by 20 – 25%.
Altered lipoprotein composition in HIV/ART
Compared with NL controls, LDL-%TG values of Heart Positive HIV/ART patients at baseline were a median two-fold higher, whereas other constituents were not significantly different (Table 1). HDL compositions were also significantly different, with higher %TG, %CE and %FC (core components), and lower %PL and % protein (surface components) in Heart Positive HIV/ART patients vs. NL controls. The fraction of HDL composed of surface and core components respectively were lower and higher in HIV/ART patients with significant elevations of both the %CE and the %TG. The elevation of neutral lipid core components suggested that hypertriglyceridemic HIV/ART HDL is larger than NL HDL.
Table 1. Heart Positive HIV and NL Control HDL and LDL Compositions at Baselinea.
| High Density Lipoprotein Composition | |||||||
|---|---|---|---|---|---|---|---|
| Donor | n | % Protein | % PL | % FC | % CE | % TG | % (CE+TG) |
| HIV | 114 | 42.0 | 19.3 | 4.08 | 27.2 | 7.41 | 35.3 |
| (36.6-45.7) | (18.4-20.7) | (3.36-5.25) | (24.2-30.0) | (5.68-10.17) | (31.2-38.3) | ||
| NL | 12 | 47.7 | 23.1 | 2.08 | 21.8 | 4.56 | 26.6 |
| (45.3-50.7) | (21.6-24.6) | (1.92-2.41) | (18.9-23.7) | (3.21-6.19) | (24.7-29.0) | ||
| pb | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | |
| ratio | 0.9 | 0.8 | 2.0 | 1.3 | 1.6 | 1.3 | |
| Low Density Lipoprotein Composition | |||||||
| n | % Protein | % PL | % FC | % CE | % TG | % (CE+TG) | |
| HIV | 113 | 24.2 | 18.7 | 7.56 | 36.5 | 11.97 | 49.2 |
| (22.4-26.1) | (17.8-19.7) | (6.86-8.33) | (33.4-39.1) | (9.27-16.3) | (47.6-50.9) | ||
| NLc | 22 | 22 | 8 | 42 | 6 | 48 | |
| ratio | 1.1 | 0.9 | 0.9 | 0.9 | 2.0 | 1.0 | |
Values are median and (25 – 75)% range.
Mann-Whitney rank sum test p value for comparison of Heart Positive HIV to NL values.
NL values for LDL composition are from Havel et al.38
Effect of treatment on lipoprotein composition
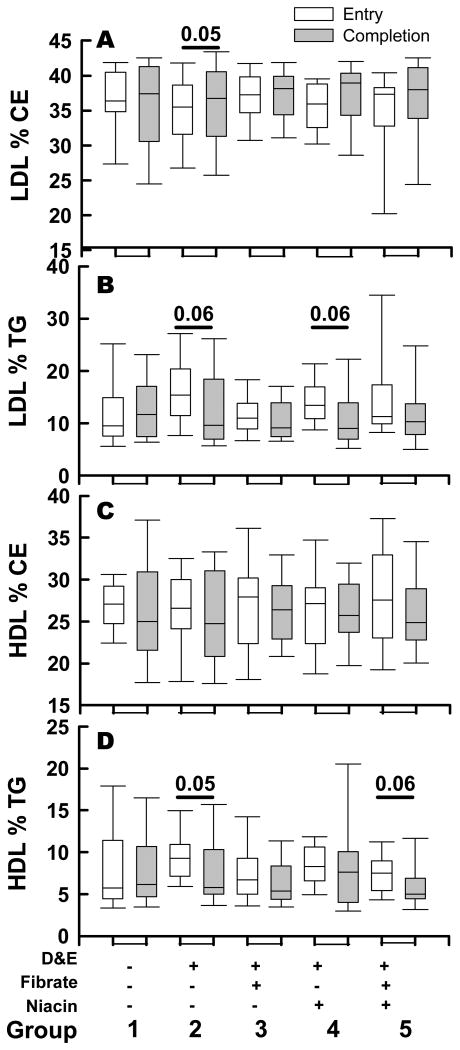
The five treatment arms differentially affected HDL and LDL neutral lipid compositions. As expected, the placebo group showed no change in HDL and LDL composition (Figure 1; Group 1). The various treatments did not have much effect on lipoprotein CE content (Figure 1A, 1C). While increased for all treatments, median LDL %CE (average of 4%) were not significantly different except for Group 2 (D&E). Similarly, while all treatments trended towards decreased HDL %CE (average 6%), this was not significant. In contrast, all four interventions reduced the median LDL %TG contents (Figure 1B) and HDL %TG (Figure 1D). Due to the limited sample size and large individual variability in %TG values, the differences in paired entry vs. after therapy values for any single treatment group only approached p<0.05 significance. However for all treatments combined there was a 24% decrease in LDL %TG (p=0.003) and a 25% decrease in HDL %TG (p=0.004). These results indicate that while the elevated TG contents of both LDL and HDL were responsive to treatment, the elevated CE content of HTG-HIV/ART HDL was more resistant to these treatment protocols.
Figure 1.
Comparison of the Effects of Four Different Anti Lipidemic Therapies vs. Placebo Control on LDL and HDL Composition. LDL and HDL %CE and % TG weight composition are shown as median, box plot of (25 – 75)% range and bar (10 – 90)% range at entry (white bars) and after therapy (gray bars). Therapy Groups 1 – 5 are denoted on the abscissa. Wilcoxon signed rank sum analysis of paired entry and after therapy values are indicated for p<0.06. For all treatments combined, both LDL and HDL %TG decrease significantly: p=0.003 and 0.004, respectively.
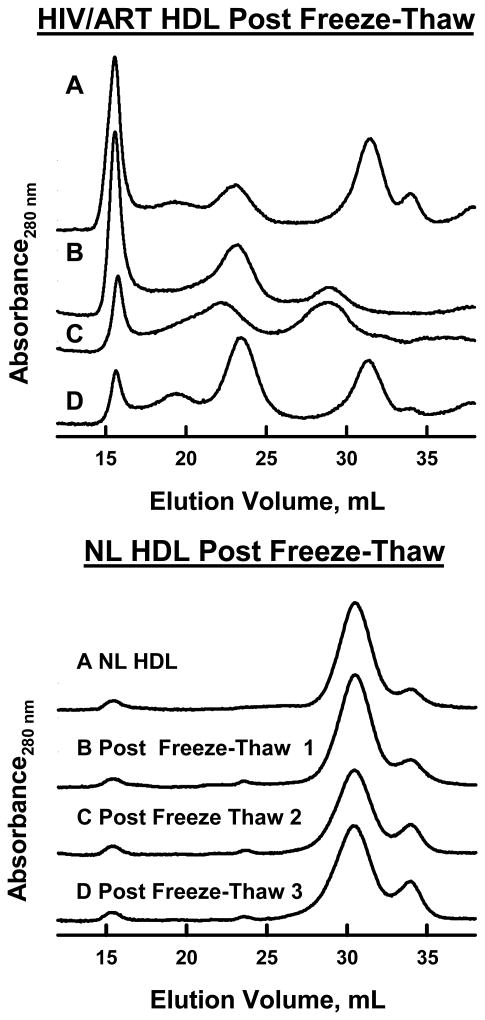
Hypertriglyceridemic HIV/ART HDL are not stable to freezing
As the neutral core to surface lipid ratio of lipoproteins increases, the particle size increases.17 Assessment of lipoprotein particle size by SEC revealed that Heart Positive HIV/ART HDL were unstable to freezing and exhibited other SEC peaks eluting earlier (larger particle size) that were absent from the chromatograms of normal HDL (Figure 2 A). We assigned these large species to HDL fusion.18 As shown in Figure 2 B, SEC uncovers freezing-dependent differences in the profile of NL control HDL after multiple freeze thaw cycles. However, the effect of freeze-thaw is much more profound for the HDL from four Heart Positive HIV/ART patients. Thus, the frozen-while-stored samples could not be used for studies of HDL sizing or functionality.
Figure 2.
Effects of Freezing on HDL as Assessed by SEC. Top Panel, A – D, Four different HIV/ART HDL samples after three freeze-thaw cycles. Bottom Panel, A – D, a single NL Control HDL sample analyzed before freezing and after one to three freeze-thaw cycles.
HDL Composition of non-HTG HIV/ART
HIV/ART HDL from a second HIV/ART patient group were stored at 4°C. The plasma lipid values for HIV/ART patients are compared to NL controls and the Heart Positive HIV/ART patients in Table 2. Unlike the Heart Positive Clinical Trial, which had completed by this time, the second HIV/ART patient group was not selected for hypertriglyceridemia. According to their plasma lipid concentrations, these patients presented with isolated low plasma HDL-C (p=0.004 vs. NL controls) and reduced total plasma cholesterol (p=0.025 vs. NL controls) but no hypertriglyceridemia. The HDL compositions for this group (Table 3) show that HDL-CE contents were lower than those of NL control, the opposite of what was observed for the HTG HIV/ART patients. However, similar to the hypertriglyceridemic Heart Positive patients, this group of normotriglyceridemic HIV/ART patients also have HDL neutral lipid cores enriched in TG relative to CE compared to NL control HDL. HDL samples from this group were studied further for stability, size and functionality.
Table 2. Plasma Lipid Values for HIV Patients with Isolated Low HDL-C (nonHTG HIV) compared to Heart Positive HIV and NL Controlsa.
| Cholesterol | Triglyceride | HDL-C | Calc LDL-C | Non-HDL-C | ||
|---|---|---|---|---|---|---|
| Donor | n | mg/dL | mg/dL | mg/dL | mg/dL | mg/dL |
| Heart Positive HIV | 114 | 195 | 248 | 37 | 108 | 158 |
| (172-230) | (171-329) | (31-42) | (77-137) | (136-187) | ||
| pb | 0.914 | <0.001 | <0.001 | 0.755 | 0.010 | |
| nonHTG HIV | 10 | 125 | 109 | 33 | 44 | 80 |
| (72-208) | (91-228) | (20-58) | (34-128) | (53-159) | ||
| pb | 0.025 | 0.668 | 0.004 | 0.052 | 0.106 | |
| NL Control | 12 | 196 | 118 | 67 | 103 | 124 |
| (166-233) | (90-142) | (52-92) | (90-120) | (111-153) |
Values are median and (25 – 75)% range.
Mann-Whitney rank sum test p value for comparison of HIV values to NL values.
Table 3. HDL Composition of Heart Positive HIV, HIV with Isolated Low HDL-C (nonHTG HIV) and NL Control Donorsa.
| High Density Lipoprotein Composition | ||||||||
|---|---|---|---|---|---|---|---|---|
| Donor | n | % Protein | % PL | % FC | % CE | % TG | % (CE+TG) | TG (TG+CE) |
| Heart Positive HIV | 114 | 42.0 | 19.3 | 4.08 | 27.2 | 7.41 | 35.3 | 0.22 |
| (36.6-45.7) | (18.4-20.7) | (3.36-5.25) | (24.2-30.0) | (5.68-10.17) | (31.2-38.3) | (0.16-0.29) | ||
| pb | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | 0.076 | |
| nonHTG HIV | 10 | 49.3 | 24.8 | 2.22 | 15.2 | 5.34 | 20.3 | 0.25 |
| (47.1-59.0) | (18.8-27.4) | (2.03-2.70) | (10.8-18.7) | (3.39-6.70) | (16.6-24.2) | (0.17-0.35) | ||
| pb | 0.156 | 0.817 | 0.448 | 0.006 | 0.621 | 0.008 | 0.049 | |
| NL | 12 | 47.7 | 23.1 | 2.08 | 21.8 | 4.56 | 26.6 | 0.17 |
| (45.3-50.7) | (21.6-24.6) | (1.92-2.41) | (18.9-23.7) | (3.21-6.19) | (24.7-29.0) | (0.12-0.25) | ||
Values are median and (25 – 75)% range.
Mann-Whitney rank sum or Student t-test p value for comparison of HIV values to NL values.
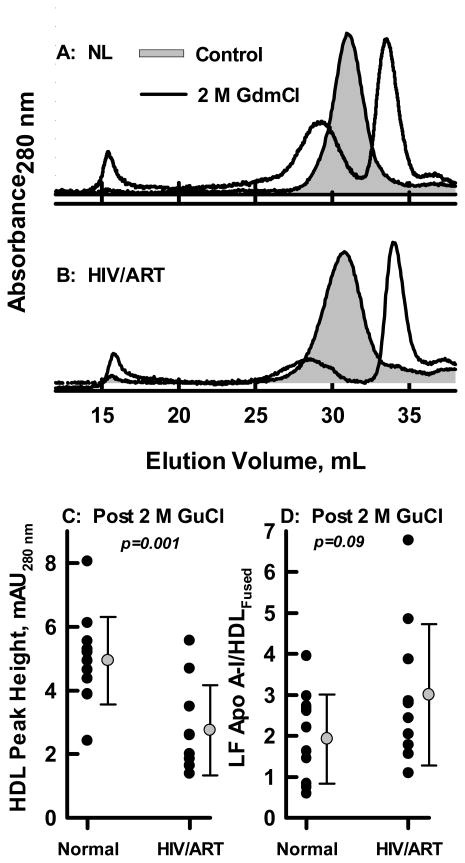
HIV/ART HDL are less stable to chaotropic perturbation than NL control HDL
Incubation of HDL with GdmCl induces the release of lipid-free apo A-I and the formation of a larger fused HDL (Figure 3).18, 19 Comparison of the effects of GdmCl on the lipoprotein profiles of HDL from NL control and HIV/ART patients revealed notable differences in the amounts of the released products (Figure 3 A, B). Although GdmCl converted the HDL from both NL control and HIV/ART to the expected products, the relative amounts of fused HDL and lipid-free apo A-I formed were different (Figure 3 C, D). Post GdmCl, the relative amount of HDL remaining is lower and the amount of lipid-free apo A-I is higher in HIV/ART HDL vs. NL control HDL. Accordingly, even non HTG HIV/ART HDL is less stable than NL HDL.
Figure 3.
Comparison of the Stability of HDL from HIV/ART and NL Control Subjects. A, B: Examples of SEC profiles of NL and HIV/ART HDL before (gray shaded curve) and after (line) treatment with 2M GdmCl. Stability was assessed on the basis of the HDL peak height after treatment (C) and the ratio of lipid-free apo A-I/HDLfused ratio. Less fused (lipidated) HDL and more lipid-free apo A-I is formed from the HDL of the HIV/ART patients than from those of the the NL Control.
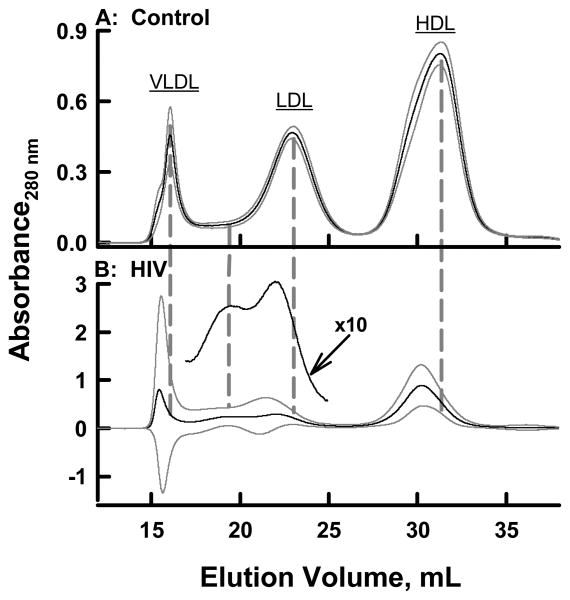
HIV/ART Lipoproteins are Larger than NL Control Lipoproteins
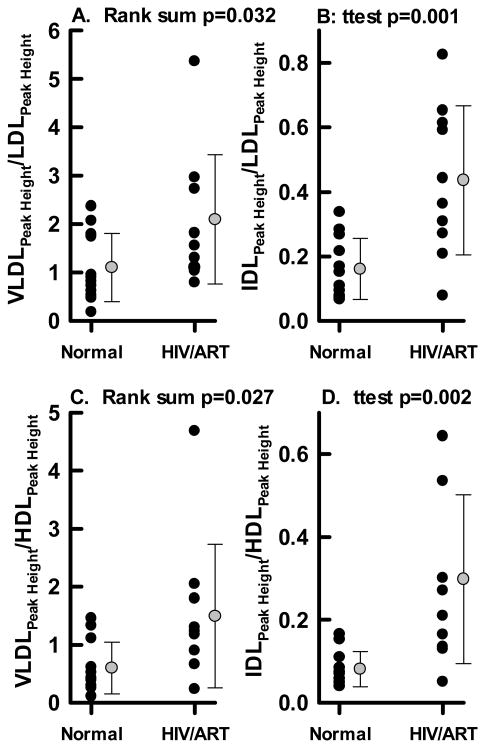
The high neutral lipid content of LDL and HDL of Heart Positive patients suggested these might be larger lipoproteins.17 The sizes of lipoproteins in the TLP from non HTG HIV patients and controls were compared by SEC (Supplemental Figure II). The means ± SE of these data, calculated and plotted as shown in Figure 4, reveal profound differences between HIV/ART and NL control TLP. NL control TLP contains prominent peaks for VLDL, LDL, and HDL with relatively small standard errors (Figure 4 A). SEC profiles for HIV/ART TLP differ (Figure 4 B). First, peaks for the three major lipoproteins are shifted to earlier elution volumes corresponding to a larger particle sizes (compare grey vertical lines). Second, (Figure 4 B, 10X insert), the SEC of HIV/ART TLP contains a peak between LDL and VLDL that we assign to IDL. Lastly, the standard errors at each elution volume of the HIV/ART TLP chromatogram are much larger than those of control TLP. The relative amounts of the lipoproteins are also altered. Based on the ratios of the peak heights, HIV/ART lipoproteins are altered relative to NL control, with higher amounts of VLDL and IDL relative to LDL and HDL (Figure 5). The ratios relative to LDL for NL control and HIV/ART are as follows: VLDL/LDL = 1.10 vs. 2.10 (p = 0.032); IDL/LDL = 0.160 vs. 0.436 (p = 0.001). These larger sizes, and the greater amounts of VLDL and IDL relative to LDL and HDL, suggest delayed processing of apo B lipoproteins in HIV plasma.
Figure 4.
Comparison of Lipoprotein Sizes by SEC of TLP from NL Control and HIV/ART Subjects. Plots in A and B respectively are the average SEC profiles of the TLP from 12 NL Control and 10 HIV/ART Subjects. Individual chromatograms are in Supplemental Figure III. The insert in B is a 10X expansion of that region of the chromatogram to better reveal peaks for IDL and LDL. The black and grey lines represent the mean and standard error of the chromatograms. The dashed grey lines demonstrate that the average size of VLDL, LDL and HDL are larger in HIV/ART than in NL subjects.
Figure 5.
Relative amounts of VLDL and IDL in HIV/ART and NL Control Plasma. Peak heights for each lipoprotein were determined from the individual donor TLP chromatograms shown in Supplemental Figure II. The ratios of VLDL and IDL relative to LDL are shown in A and B, and relative to HDL in C and D. HIV/ART plasma contains significantly more VLDL (p<0.03) and especially IDL (p<0.002) relative to both LDL and HDL than does NL Control plasma. The ratio of LDL/HDL was not significantly different (data not shown).
Validation of a Functional Assessment of NL Control vs. HIV/ART HDL
Because of the altered CE content and the lower stability of the HIV/ART HDL samples, we hypothesized that HIV/ART HDL are dysfunctional in the final step of RCT, transfer of HDL CE to hepatocytes. The most straightforward method for measuring hepatocyte HDL-CE uptake requires preparation of HDL-[3H]CE, a procedure that can take 10 days and requires significant quantities of starting HDL20 to yield HDL-[3H]CE with >99% radiochemical purity,21, 22 followed by uptake assays according to Acton.23 This assay is impractical in the context of large patient numbers and limited plasma volumes, so we modified and validated a competitive assay of hepatic CE uptake that we have used to compare the HDL-[3H]CE uptake before and after remodeling by streptococcal serum opacity factor.20 This assay uses NL control and HIV/ART patient HDL to compete with the uptake of a standard stock HDL-[3H]CE (Supplemental Figure III). Inhibition constants, ID50%, were calculated from the inhibitor HDL dose-response curves. In this assay, a large ID50% corresponds to HDL that interact poorly with hepatic HDL receptors and vice versa. Our assay is reproducible; each panel in Supplemental Figure III contains assays on the same HDL sample repeated on different days, with up to 17 weeks between assays. Assays in triplicate showed a within-day variability of 10.6% (n = 28) and day-to-day variability of 10.4% (n = 4).
HIV HDL is a poor competitor for hepatocyte HDL-CE uptake
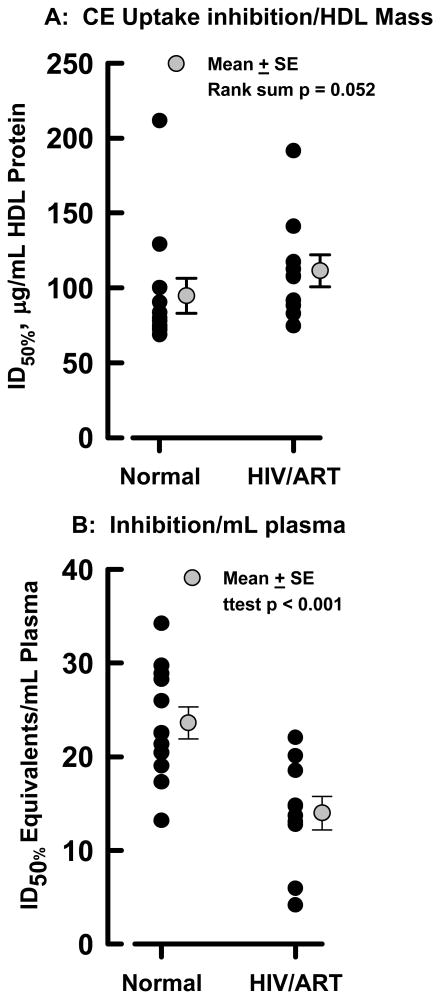
Using this competitive inhibition assay, we compared the ID50% of HDL from HIV/ART patients with those from NL controls. As shown in Figure 6 A, ID50% for individual NL and HIV/ART HDL distributed over a wide range, 72 – 211 and 75 – 191 μg/mL HDL protein for HIV/ART and NL respectively. These ranges, which are far greater than the assay variability, illustrate the individual heterogeneity of donor HDL with respect to selective hepatic CE uptake as well as the power of this assay to distinguish HDL functionality on the basis of hepatic CE uptake. Our data show that the mean ID50% for the NL group was lower than that of the HIV/ART subjects (rank sum p = 0.052), indicating the HIV/ART HDL was a poorer competitor, i.e., required more HDL to reach its ID50%. Importantly, adjustment of these data for each donor's HDL-C level, which were lower for the HIV/ART patients (Figure 6 B), revealed the dysfunctionality of HIV/ART HDL. The difference between HIV/ART and NL plasma to affect HDL-CE uptake was profound—CE uptake inhibition by HIV/ART HDL expressed as plasma volume-equivalents was much lower. NL plasma contains 23.6 ± 1.7 ID50% units/mL vs. 14.0 ± 1.8 ID50% units/mL for HIV/ART plasma (p <0.001). The mean rate of CE uptake from HDL-[3H]CE is inhibited on average twice more effectively by NL HDL than by HIV/ART HDL when measured at plasma concentrations of the respective HDL. Thus, at plasma concentrations HIV/ART HDL interact with hepatocytes less effectively than NL HDL.
Figure 6.
Inhibition of Hepatocyte HDL CE Uptake by HDL from NL and HIV/ART Subjects. A, ID50% for individual donor HDL indicating inhibitory activity per mg HDL protein. B, Inhibitory potency of each patient's plasma expressed in terms of the volume of plasma needed to achieve 50% inhibition. Black dots: individual values; gray dot and bars: mean ± SE. The combination of low plasma HDL-C and the higher median ID50% in the HIV/ART versus NL Control subjects results in an average ID50% equivalent that is 41% lower for the HIV/ART vs NL subjects, p<0.001.
Discussion
Plasma Lipid Profiles of HIV/ART Patients with HTG or Isolated Low HDL-C versus NL Controls
The Heart Positive HIV/ART patients in the four therapeutic and placebo groups have similar lipid profiles (Supplemental Table I). The higher LDL- and HDL-TG contents within the HTG HIV/ART group (Table 1) are consistent with the CETP-mediated transfer of TG from VLDL, which are elevated, to LDL and HDL. This effect is dose-dependent with respect to fasting plasma TG levels and is expected to yield less stable TG-rich LDL and HDL.24 TG-enrichment may underlie the greater freeze instability of Heart Positive vs. control subjects. The effects of the interventions on LDL and HDL compositions were consistent with this model (Figure 1). LDL and HDL TG from patients on D & E and receiving niacin and/or fenofibrate were lower. The plasma lipoprotein profile of the non HTG HIV/ART patients was characterized by isolated low HDL-C (Table 2), low HDL %CE, and a TG-enriched neutral lipid core (Table 3).
Metabolism of HIV/ART Lipoproteins is Impaired
According to the SEC profiles, the dyslipidemia in HIV/ART TLP patients is highly heterogeneous (large standard errors) and is associated with the appearance of lipoproteins that are larger than those of NL control (Figure 4). Consistent with this, the data of Aragones et al showed greater heterogeneity in HIV HDL and increased CE in the larger HDL subfractions.25 Two observations suggest that increased lipoprotein heterogeneity is due impaired lipoprotein hydrolysis. First, the SEC profiles (Figure 4) showed that all HIV/ART lipoproteins elute earlier than their NL control counterparts; second, these data also show that unlike control TLP, HIV/ART TLP contains a prominent peak for IDL (Figure 4). Increased amounts of VLDL and IDL in HIV/ART fasting plasma indicate delayed processing of these lipoproteins (Figure 5). Given that both IDL- and HDL-TG are substrates for hepatic lipase (HL), which reduces their size by hydrolysis, we hypothesize that HL activity is impaired under the conditions of HIV/ART dyslipidemia, a hypothesis that is supported by reports that HL and to a lesser extent lipoprotein lipase (LPL) activities are lower in HIV/ART-associated dyslipidemia26; lower LPL activity would also explain the larger HIV/ART vs. NL control VLDL. While endothelial lipase activity has not been reported in HIV samples, low endothelial lipase activity could also cause larger HDL as well as reduced HDL phospholipid,27 but would be expected to cause higher rather than lower plasma HDL-C.28
Interaction of HIV/ART HDL with Hepatocytes is Impaired
According to our competitive assay of HDL-CE uptake by Huh7 cells, differences in the ID50% of NL Control vs. HIV/ART HDL were nearly significant (p = 0.052; Figure 6 A). Moreover, when the ID50% data were normalized to the plasma HDL-C levels of each individual, HIV/ART plasma contains significantly less competitive inhibition activity for hepatocyte HDL-CE uptake than does NL plasma (p<0.001). Decreased interaction of HIV/ART HDL with hepatic receptors may contribute to the observed elevated HDL-%CE in the Heart Positive patient population (Table 1). It would suggest that the higher HDL-CE may result from an inability of HIV/ART HDL to deliver its CE to hepatocyte receptors for uptake and clearance. The molecular basis for this is not known but may be due to altered surface protein structure or configuration in response to an altered core composition.
HDL Stability
HIV/ART HDL were found to be less stable than NL HDL both to freezing and to chaotropic perturbation. Importantly, HDL instability has been uncovered by LCAT, CETP, PLTP, and serum opacity factor, all of which induce fusion and/or the release of lipid-free apo A-I.22, 29-32 Although there have been many studies of the effects of GdmCl on HDL,18, 19, 33, 34 this is the first to show that the effects of GdmCl on HDL vary among dyslipidemic patients and NL controls, and that the NL control and HIV/ART HDL respond differently to this denaturant. According to several studies, HDL resides in a kinetic trap from which it can escape via both physico-chemical18, 19 and biological perturbations—lecithin:cholesterol acyltransferase, cholesteryl ester transfer protein, phospholipid transfer protein, and serum opacity factor. 29-31, 35 Thus, in the context of the kinetic model of Gursky,18 HIV/ART HDL is less stable than NL control HDL. Given that a major marker of instability is the release of lipid-free apo A-I, one could speculate that the plasma factors cited above mediate production of more lipid-free apo A-I in HIV/ART patients compared to NL controls. A negative consequence of this would be increased renal clearance of apo A-I, which would have the effect of lowering plasma HDL-C levels; this hypothesis needs more rigorous testing.
A metabolic model of dysfunctional lipoprotein processing in HIV/ART dyslipidemia
According to our data, HIV/ART dyslipidemia is associated with larger HDL, LDL and VLDL, and impaired HDL hepatocyte binding, which likely affects hepatocyte HDL-CE uptake. Other studies in HIV patients have demonstrated peripheral tissue hyperlipolytic activity11 and lower lipoprotein and hepatic lipase activities.26, 36 In contrast, the initial step in reverse cholesterol transport, macrophage cholesterol efflux to HDL, is unaltered by HIV and its treatment. Rose et al found similar rates of activated RAW 264.7 macrophage-cholesterol efflux to the plasma of HIV-infected patients, currently treated HIV-infected patients, and HIV-negative subjects.8 Taken together, these data support a metabolic model for HIV dyslipidemia (Figure 7) that begins with peripheral tissue hyperlipolytic activity11 that results in the release of higher amounts of free fatty acids that are extracted by the liver and used for the production of more VLDL, thereby producing a hypertriglyceridemic state. In the presence of high VLDL-TG concentrations, CETP mediates the exchange of VLDL TG for HDL and LDL-CE, thereby producing TG-rich HDL and LDL. CETP activity has been reported to be elevated8 or within normal limits37 in HIV/ART subjects. Lipoprotein-TG hydrolysis by hepatic lipase (HL) reduces their sizes by removing core lipids and when this process is inhibited, larger TG-rich lipoprotein species are observed. In our study, there is even some residual IDL, which under normolipemic conditions is converted to LDL by HL. All lipoprotein classes from HIV/ART are larger than those form NL subjects suggesting resistance to the activities of hepatic and/or lipoprotein lipases. As a consequence of the high TG content of the HDL particles, we speculate that they are less stable and that the HIV/ART apo A-I is more labile than that of NL HDL. Whether the instability influences function remains to be investigated. Nevertheless, the CE of HIV/ART HDL are hepatically extracted at a lower rate than those of NL HDL because of their intrinsically lower uptake and the lower plasma HDL-C levels in HIV/ART patients.
Figure 7.
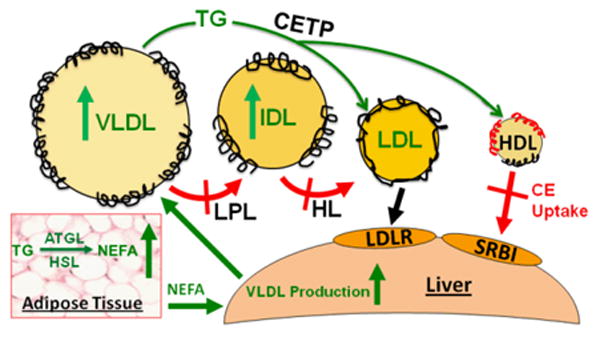
Metabolic model for the production of dysfunctional lipoproteins in HIV/ART dyslipidemia. See text for discussion.
Supplementary Material
Significance.
Patients with human immunodeficiency virus (HIV) receiving antiretroviral therapy (ART), present with a unique dyslipidemia that places them at risk for cardiovascular disease (CVD). We compared HIV/ART patients with normolipidemic (NL) subjects. All HIV/ART lipoproteins were larger than the corresponding NL lipoproteins; the HIV/ART HDL are richer in neutral lipids, less stable, and bind poorly to hepatocytes. These findings were based on new assays of HDL stability and functionality; this study was the first to apply these assays to a clinical population. These findings are unprecedented in HIV/ART research and show that the underlying mechanisms for the unique HIV/ART dyslipidemia are impaired lipoprotein clearance by plasma lipases and/or hepatic receptors. This research brings a new perspective on the pathogenesis of HIV/ART-associated dyslipidemia, with high relevance to some other metabolic diseases—Cushing's syndrome, polycystic ovarian disease, and obesity-linked diabetes.
Acknowledgments
The authors thank Hu Yu Alice Lin for excellent technical assistance, Xaioyuan Perrard for the mRNA analyses, and Lele Li for initial work on HDL stability to GdmCl treatment.
Sources of Funding: This work was supported by National Institutes of Health grants HL73696 (A.B.), RO1-HL-30914 and RO1-HL56865 (H.J.P.), and P30DK079638 for support of the Diabetes Research Center at Baylor College of Medicine.
Abbreviations
- C
cholesterol
- CE
cholesteryl ester
- CETP
cholesteryl ester transfer protein
- FC
free cholesterol
- FPLC
fast protein liquid chromatography
- HDL
high density lipoprotein
- HL
hepatic lipase
- HP
Heart Positive HIV study patients
- HIV/ART HIV
patients on highly active antiretroviral therapy
- HTG
hypertriglyceridemic
- IDL
intermediate density lipoprotein
- GdmCl
guanidinium chloride
- LCAT
lecithin:cholesterol acyltransferase
- LDL
low density lipoprotein
- LPL
lipoprotein lipase
- MetS
metabolic syndrome
- PLTP
phospholipid transfer protein
- SEC
size exclusion chromatography
- TG
triglyceride
- VLDL
very low density lipoprotein
- TLP
total lipoproteins
Footnotes
Disclosures: None of the authors have disclosures to report.
References
- 1.Carr A, Samaras K, Burton S, Law M, Freund J, Chisholm DJ, Cooper DA. A syndrome of peripheral lipodystrophy, hyperlipidaemia and insulin resistance in patients receiving HIV protease inhibitors. AIDS. 1998;12:F51–58. doi: 10.1097/00002030-199807000-00003. [DOI] [PubMed] [Google Scholar]
- 2.Worm SW, Sabin C, Weber R, Reiss P, El-Sadr W, Dabis F, De Wit S, Law M, Monforte AD, Friis-Moller N, Kirk O, Fontas E, Weller I, Phillips A, Lundgren J. Risk of myocardial infarction in patients with HIV infection exposed to specific individual antiretroviral drugs from the 3 major drug classes: The data collection on adverse events of anti-HIV drugs (d:A:D) study. J Infect Dis. 2010;201:318–330. doi: 10.1086/649897. [DOI] [PubMed] [Google Scholar]
- 3.d'Arminio A, Sabin CA, Phillips AN, Reiss P, Weber R, Kirk O, El-Sadr W, De Wit S, Mateu S, Petoumenos K, Dabis F, Pradier C, Morfeldt L, Lundgren JD, Friis-Moller N. Cardio- and cerebrovascular events in HIV-infected persons. AIDS. 2004;18:1811–1817. doi: 10.1097/00002030-200409030-00010. [DOI] [PubMed] [Google Scholar]
- 4.Flynn TE, Bricker LA. Myocardial infarction in HIV-infected men receiving protease inhibitors. Ann Iinternal Med. 1999;131:548. doi: 10.7326/0003-4819-131-7-199910050-00032. [DOI] [PubMed] [Google Scholar]
- 5.Friis-Moller N, Weber R, Reiss P, Thiebaut R, Kirk O, d'Arminio Monforte A, Pradier C, Morfeldt L, Mateu S, Law M, El-Sadr W, De Wit S, Sabin CA, Phillips AN, Lundgren JD. Cardiovascular disease risk factors in HIV patients--association with antiretroviral therapy. Results from the DAD study. AIDS. 2003;17:1179–1193. doi: 10.1097/01.aids.0000060358.78202.c1. [DOI] [PubMed] [Google Scholar]
- 6.Varriale P, Saravi G, Hernandez E, Carbon F. Acute myocardial infarction in patients infected with human immunodeficiency virus. Am Heart J. 2004;147:55–59. doi: 10.1016/j.ahj.2003.07.007. [DOI] [PubMed] [Google Scholar]
- 7.Samaras K, Wand H, Law M, Emery S, Cooper D, Carr A. Prevalence of metabolic syndrome in HIV-infected patients receiving highly active antiretroviral therapy using international diabetes foundation and adult treatment panel III criteria: Associations with insulin resistance, disturbed body fat compartmentalization, elevated C-reactive protein, and [corrected] hypoadiponectinemia. Diabetes Care. 2007;30:113–119. doi: 10.2337/dc06-1075. [DOI] [PubMed] [Google Scholar]
- 8.Rose H, Hoy J, Woolley I, Tchoua U, Bukrinsky M, Dart A, Sviridov D. HIV infection and high density lipoprotein metabolism. Atherosclerosis. 2008;199:79–86. doi: 10.1016/j.atherosclerosis.2007.10.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Tong Q, Sankale JL, Hadigan CM, Tan G, Rosenberg ES, Kanki PJ, Grinspoon SK, Hotamisligil GS. Regulation of adiponectin in human immunodeficiency virus-infected patients: Relationship to body composition and metabolic indices. The Journal of Clinical Endocrinology and Metabolism. 2003;88:1559–1564. doi: 10.1210/jc.2002-021600. [DOI] [PubMed] [Google Scholar]
- 10.Vigouroux C, Maachi M, Nguyen TH, Coussieu C, Gharakhanian S, Funahashi T, Matsuzawa Y, Shimomura I, Rozenbaum W, Capeau J, Bastard JP. Serum adipocytokines are related to lipodystrophy and metabolic disorders in HIV-infected men under antiretroviral therapy. AIDS. 2003;17:1503–1511. doi: 10.1097/00002030-200307040-00011. [DOI] [PubMed] [Google Scholar]
- 11.Sekhar RV, Jahoor F, White AC, Pownall HJ, Visnegarwala F, Rodriguez-Barradas MC, Sharma M, Reeds PJ, Balasubramanyam A. Metabolic basis of HIV-lipodystrophy syndrome. Am J Physiol Endocrinol and Metab. 2002;283:E332–337. doi: 10.1152/ajpendo.00058.2002. [DOI] [PubMed] [Google Scholar]
- 12.Hadigan C, Borgonha S, Rabe J, Young V, Grinspoon S. Increased rates of lipolysis among human immunodeficiency virus-infected men receiving highly active antiretroviral therapy. Metabolism. 2002;51:1143–1147. doi: 10.1053/meta.2002.34704. [DOI] [PubMed] [Google Scholar]
- 13.Reeds DN, Mittendorfer B, Patterson BW, Powderly WG, Yarasheski KE, Klein S. Alterations in lipid kinetics in men with HIV-dyslipidemia. Am J Physiol Endocrinol and Metab. 2003;285:E490–497. doi: 10.1152/ajpendo.00118.2003. [DOI] [PubMed] [Google Scholar]
- 14.Mujawar Z, Rose H, Morrow MP, Pushkarsky T, Dubrovsky L, Mukhamedova N, Fu Y, Dart A, Orenstein JM, Bobryshev YV, Bukrinsky M, Sviridov D. Human immunodeficiency virus impairs reverse cholesterol transport from macrophages. PLoS Biol. 2006;4:e365. doi: 10.1371/journal.pbio.0040365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Asztalos BF, Tani M, Schaefer EJ. Metabolic and functional relevance of HDL subspecies. Curr Opin Lipidol. 2011;22:176–185. doi: 10.1097/MOL.0b013e3283468061. [DOI] [PubMed] [Google Scholar]
- 16.Balasubramanyam A, Coraza I, Smith EO, Scott LW, Patel P, Iyer D, Taylor AA, Giordano TP, Sekhar RV, Clark P, Cuevas-Sanchez E, Kamble S, Ballantyne CM, Pownall HJ. Combination of niacin and fenofibrate with lifestyle changes improves dyslipidemia and hypoadiponectinemia in HIV patients on antiretroviral therapy: Results of “Heart Positive,” a randomized, controlled trial. J Clin Endocrinol Metab. 2011;96:2236–2247. doi: 10.1210/jc.2010-3067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Shen BW, Scanu AM, Kezdy FJ. Structure of human serum lipoproteins inferred from compositional analysis. Proc Natl Acad Sci USA. 1977;74:837–841. doi: 10.1073/pnas.74.3.837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Mehta R, Gantz DL, Gursky O. Human plasma high-density lipoproteins are stabilized by kinetic factors. J Mol Biol. 2003;328:183–192. doi: 10.1016/s0022-2836(03)00155-4. [DOI] [PubMed] [Google Scholar]
- 19.Pownall HJ, Hosken BD, Gillard BK, Higgins CL, Lin HY, Massey JB. Speciation of human plasma high-density lipoprotein (HDL): HDL stability and apolipoprotein A-I partitioning. Biochemistry. 2007;46:7449–7459. doi: 10.1021/bi700496w. [DOI] [PubMed] [Google Scholar]
- 20.Gillard BK, Rosales C, Pillai BK, Lin HY, Courtney HS, Pownall HJ. Streptococcal serum opacity factor increases the rate of hepatocyte uptake of human plasma high-density lipoprotein cholesterol. Biochemistry. 2010;49:9866–9873. doi: 10.1021/bi101412m. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Rosales C, Gillard BK, Courtney HS, Blanco-Vaca F, Pownall HJ. Apolipoprotein modulation of streptococcal serum opacity factor activity against human plasma high-density lipoproteins. Biochemistry. 2009;48:8070–8076. doi: 10.1021/bi901087z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Gillard BK, Courtney HS, Massey JB, Pownall HJ. Serum opacity factor unmasks human plasma high-density lipoprotein instability via selective delipidation and apolipoprotein A-I desorption. Biochemistry. 2007;46:12968–12978. doi: 10.1021/bi701525w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Acton S, Rigotti A, Landschulz KT, Xu S, Hobbs HH, Krieger M. Identification of scavenger receptor SR-BI as a high density lipoprotein receptor. Science. 1996;271:518–520. doi: 10.1126/science.271.5248.518. [DOI] [PubMed] [Google Scholar]
- 24.Pownall HJ, Brauchi D, Kilinc C, Osmundsen K, Pao Q, Payton-Ross C, Gotto AM, Jr, Ballantyne CM. Correlation of serum triglyceride and its reduction by omega-3 fatty acids with lipid transfer activity and the neutral lipid compositions of high-density and low-density lipoproteins. Atherosclerosis. 1999;143:285–297. doi: 10.1016/s0021-9150(98)00301-3. [DOI] [PubMed] [Google Scholar]
- 25.Aragones G, Beltran-Debon R, Rull A, Rodriguez-Sanabria F, Fernandez-Sender L, Camps J, Joven J, Alonso-Villaverde C. Human immunodeficiency virus-infection induces major changes in high-density lipoprotein particle size distribution and composition: The effect of antiretroviral treatment and disease severity. Clin Chem Lab Med. 2010;48:1147–1152. doi: 10.1515/CCLM.2010.218. [DOI] [PubMed] [Google Scholar]
- 26.Yarasheski KE, Tebas P, Claxton S, Marin D, Coleman T, Powderly WG, Semenkovich CF. Visceral adiposity, c-peptide levels, and low lipase activities predict HIV-dyslipidemia. Am J Physiol Endocrinol Metab. 2003;285:E899–905. doi: 10.1152/ajpendo.00036.2003. [DOI] [PubMed] [Google Scholar]
- 27.Singaraja RR, Sivapalaratnam S, Hovingh K, et al. The impact of partial and complete loss-of-function mutations in endothelial lipase on high-density lipoprotein levels and functionality in humans. Circ Cardiovasc Genet. 2013;6:54–62. doi: 10.1161/CIRCGENETICS.111.962613. [DOI] [PubMed] [Google Scholar]
- 28.Edmondson AC, Brown RJ, Kathiresan S, et al. Loss-of-function variants in endothelial lipase are a cause of elevated HDL cholesterol in humans. J Clin Invest. 2009;119:1042–1050. doi: 10.1172/JCI37176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Silver ET, Scraba DG, Ryan RO. Lipid transfer particle-induced transformation of human high density lipoprotein into apolipoprotein A-I-deficient low density particles. J Biol Chem. 1990;265:22487–22492. [PubMed] [Google Scholar]
- 30.Lusa S, Jauhiainen M, Metso J, Somerharju P, Ehnholm C. The mechanism of human plasma phospholipid transfer protein-induced enlargement of high-density lipoprotein particles: Evidence for particle fusion. Biochem J. 1996;313(Pt 1):275–282. doi: 10.1042/bj3130275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Liang HQ, Rye KA, Barter PJ. Remodelling of reconstituted high density lipoproteins by lecithin: Cholesterol acyltransferase. J Lipid Res. 1996;37:1962–1970. [PubMed] [Google Scholar]
- 32.Rye KA, Hime NJ, Barter PJ. Evidence that cholesteryl ester transfer protein-mediated reductions in reconstituted high density lipoprotein size involve particle fusion. J Biol Chem. 1997;272:3953–3960. doi: 10.1074/jbc.272.7.3953. [DOI] [PubMed] [Google Scholar]
- 33.Nichols AV, Gong EL, Blanche PJ, Forte TM, Anderson DW. Effects of guanidine hydrochloride on human plasma high density lipoproteins. Biochim Biophys Acta. 1976;446:226–239. doi: 10.1016/0005-2795(76)90113-6. [DOI] [PubMed] [Google Scholar]
- 34.Rosseneu M, Van Tornout P, Lievens MJ, Schmitz G, Assmann G. Dissociation of apolipoprotein AI from apoprotein-lipid complexes and from high-density lipoproteins. A fluorescence study. Eur J Biochem. 1982;128:455–460. doi: 10.1111/j.1432-1033.1982.tb06986.x. [DOI] [PubMed] [Google Scholar]
- 35.Rosales C, Tang D, Gillard BK, Courtney HS, Pownall HJ. Apolipoprotein E mediates enhanced plasma high-density lipoprotein cholesterol clearance by low-dose streptococcal serum opacity factor via hepatic low-density lipoprotein receptors in vivo. Arterioscler Thromb Vasc Biol. 2011;31:1834–1841. doi: 10.1161/ATVBAHA.111.224360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Baril L, Beucler I, Valantin MA, Bruckert E, Bonnefont-Rousselot D, Coutellier A, Caumes E, Katlama C, Bricaire F. Low lipolytic enzyme activity in patients with severe hypertriglyceridemia on highly active antiretroviral therapy. AIDS. 2001;15:415–417. doi: 10.1097/00002030-200102160-00016. [DOI] [PubMed] [Google Scholar]
- 37.Johnston CS, Kim CM, Buller AJ. Vinegar improves insulin sensitivity to a high-carbohydrate meal in subjects with insulin resistance or type 2 diabetes. Diabetes Care. 2004;27:281–282. doi: 10.2337/diacare.27.1.281. [DOI] [PubMed] [Google Scholar]
- 38.Havel RJ, Goldstein JL, Brown MS. Lipoproteins and lipid transport. In: Bondy PK, Rosenberg LE, editors. The Metabolic Control of Disease. Philadephia, Penn: Saunders Publishing; 1980. pp. 398–494. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.