Abstract
Despite the completion of the Arabidopsis genome sequence, for only a relatively low percentage of the encoded proteins experimental evidence concerning their function is available. Plant proteins that harbour a single PLAT (Polycystin, Lipoxygenase, Alpha-toxin and Triacylglycerol lipase) domain and belong to the PLAT-plant-stress protein family are ubiquitously present in monocot and dicots. However, the function of PLAT-plant-stress proteins is still poorly understood. Therefore, we have assessed the function of the uncharacterised Arabidopsis PLAT-plant-stress family members through a combination of functional genetic and physiological approaches. PLAT1 overexpression conferred increased abiotic stress tolerance, including cold, drought and salt stress, while loss-of-function resulted in opposite effects on abiotic stress tolerance. Strikingly, PLAT1 promoted growth under non-stressed conditions. Abiotic stress treatments induced PLAT1 expression and caused expansion of its expression domain. The ABF/ABRE transcription factors, which are positive mediators of abscisic acid signalling, activate PLAT1 promoter activity in transactivation assays and directly bind to the ABRE elements located in this promoter in electrophoretic mobility shift assays. This suggests that PLAT1 represents a novel downstream target of the abscisic acid signalling pathway. Thus, we showed that PLAT1 critically functions as positive regulator of abiotic stress tolerance, but also is involved in regulating plant growth, and thereby assigned a function to this previously uncharacterised PLAT domain protein. The functional data obtained for PLAT1 support that PLAT-plant-stress proteins in general could be promising targets for improving abiotic stress tolerance without yield penalty.
Introduction
The PLAT domain (PS50095; Polycystin-1, Lipoxygenase, Alpha-toxin and Triacylglycerol lipase) forms a β-sandwich composed of two sheets of four strands each and is an intracellular domain. It occurs in a variety of membrane or lipid associated proteins that are multi-domain proteins, but also in proteins harbouring either a single PLAT domain or repeats [1]–[5]. Because of its similarity to the C2 domain, the PLAT domain was proposed to function in protein-protein interactions as well as protein-membrane interactions [2], [4], [6]. Indeed, the PLAT domain of Caenorhabditis elegans polycystin LOV-1 and human polycystin-1 interact with ATP-2, an ATP synthase F1 subunit [7], while the human polycystin-1L2 interacts with different types of G-proteins [8]. Importantly, the association with membranes is essential for the proper function of PLAT domain proteins [2], [7]. Further, the membrane targeted 11R-Lipoxygenase from Gersemia fruticosa was shown to bind calcium, required to induce its activity [9]. The PLAT domain regulates the catalytic activity in multi-domain proteins, but also in proteins interacting with the PLAT domain [2], [6], and was shown to regulate substrate specificity [10]. Whereas substantial experimental data on PLAT domain proteins is available for the animal field, PLAT domain proteins from plants were only poorly studied, despite the fact that genes encoding PLAT domain proteins were isolated from several plant species [3], [4], [11]–[16].
Transgenic approaches to improve abiotic stress tolerance often resulted in yield penalties under optimal growth conditions [17], [18], while only few studies reported an associated improved plant growth [4], [19]. Interestingly, one of these studies addressed the so far only studied PLAT-plant-stress protein CaTIN1 from Capsicum annuum, however this protein was only studied using heterologous expression in tobacco [4]. Because both gain-of-function and antisense CaTIN1 expression promoted abiotic and biotic stress tolerance, CaTIN1 function remained elusive. Proteins that belong to the PLAT-plant-stress protein family (Conserved Domain cd1754) are ubiquitously present in monocot and dicot plant species and harbour a single PLAT domain. Our analyses of the limited in silico expression data available for PLAT-plant-stress proteins indicate transcriptional induction by different abiotic and biotic stimuli. This suggests that PLAT-plant-stress proteins in general could promote tolerance towards stress responses, although no data from functional studies in homologous systems are available for these proteins.
The plant hormone abscisic acid (ABA) regulates different aspects of plant development, such as stomatal aperture [20] and seed germination [21]. ABA production is increased by abiotic stresses and ABA regulated genes strongly overlap with those induced under drought, salinity and less prominently cold stress conditions [22]–[25]. The ABA stimulated stomatal closure has been shown to serve as primary defence mechanism during the initial phase of biotic stress responses [26], [27]. In contrast, ABA mostly negatively regulates the subsequent phases in biotic stress responses by repressing the salicylic acid, ethylene, jasmonic acid and cytokinin signalling pathways [26]–[29]. ABA-deficient mutants showed enhanced defence responses against Botrytis cinerea [30], and virulent bacteria in tomato [31] and Arabidopsis [32]. Although these findings suggest that ABA is involved in the crosstalk between abiotic and biotic stress responses, no direct link in the antagonistic interaction between these stresses is available.
Based on comparative genomic analysis, we identified three Arabidopsis genes (PLAT1 AT4G39730, PLAT2 AT2G22170 and PLAT3 AT5G65158) that belong to the PLAT-plant-stress subgroup and submitted this annotation to the TAIR database. PLAT1 and PLAT2 are orthologs of CaTIN1 and CaTIN1-2, respectively [4], [16]. Our in silico analysis of published experimental data [33] revealed that cold stress induced the expression levels for the PLAT1 ortholog in Thlaspi arvense, which is a close relative of Arabidopsis. Based on these findings we hypothesised that the Arabidopsis members from this PLAT-plant-stress subgroup, similar to CaTIN1 [4], also promote tolerance towards various stress responses. Here we report on the molecular characterisation and functional analysis of the PLAT-plant-stress subgroup family member AT4G39730 that we designated as Arabidopsis PLAT domain protein 1 (PLAT1). We showed that PLAT1 critically functions as positive regulator of abiotic stress tolerance, also promotes plant growth and is a direct target of the ABF transcription factors, which are positive mediators of the ABA signalling pathway [34], [35]. The possible practical application to increase abiotic stress tolerance without yield penalty in crop species is discussed.
Materials and Methods
Plasmid construction and plant transformation
Total RNA isolated from Arabidopsis (TRIR reagent from Thermo Fischer Scientific, Germany) was reverse-transcribed using the ReverAid™ First strand cDNA synthesis kit (Thermo Fischer Scientific, Germany). Using this cDNA as template, the full-length PLAT1 cDNA was amplified by PCR with the PLAT1-F cDNA and PLAT1-R cDNA primers (Table S1 in File S1). The dexamethasone inducible overexpression construct, 35S>>PLAT1 was created by cloning the PLAT1 cDNA PCR product in the OP shuttle vector pEG647. The resulting OP:PLAT1 cassette was transferred to the binary vector pEG618 harbouring the 35S:LhGR activator component, resulting in 35S>>PLAT1. The PLAT1 rescue/reporter constructs were generated by PCR amplification from genomic DNA with the PLAT1-F and PLAT1-R rescue primers (Table S1 in File S1) to isolate the genomic fragment harbouring 2039 bp PLAT1 promoter sequence, including the 5′-UTR region, and the PLAT1 coding region without stop codon. Subsequently, the venus YFP or GUS reporter proteins were fused in frame to the C-terminus of the PLAT1 protein resulting in PLAT1:PLAT1-YFP and PLAT1:PLAT1-GUS, respectively. The different 35S:ABF1–4 overexpression constructs were created by cloning the ABF1–4 PCR products (ABF1–4-F OX and ABF1–4-R OX primers, Table S1 in File S1) into the pPS1 binary vector. The MBP-ABF1–4 fusion proteins for the EMSA experiments were created by cloning the ABF1–4 PCR products (ABF1–4-F and ABF1–4-R primers; Table S1 in File S1) into the pMA-c2xL vector harbouring the maltose binding protein as tag for protein purification. Binary vectors were introduced into Agrobacterium tumefaciens LBA4404 by electroporation and used to transform Arabidopsis plants using the floral dip method [36].
Phylogenetic analysis
To identify members of the PLAT-plant-stress family from other plant species, multiple database searches were performed using the Basic Local Alignment Search Tool (BLAST) algorithms BLASTp and tBLASTn available on the public databases, PLAZA 2.0 (bioinformatics.psb.ugent.be/plaza) and Phytozome v8.0 (www.phytozome.net) with cutoff value of E<10−5. We used nucleotide and amino acid sequences of PLAT1 from the TAIR database (www.arabidopsis.org) to BLAST all databases. Phylogenetic analysis was performed by using CLUSTALW alignment in PHYLIP format clustal algorithm, and displayed in a phylogram tree format with locus name of each protein. Bootstrap values were presented as a percent of 100 resampled trees at each tree node using default settings of the TreeTop-Phylogenetic Tree (www.genebee.msu.su/services/phtreereduced.html).
Plant materials and growth conditions
Nicotiana benthamiana plants were grown under greenhouse conditions as described previously [37]. Arabidopsis plants (Col-0 ecotype) were grown in soil at 8 h light/16 h darkness at 22°C (light intensity: 180 µmol m−2 s−1) or on half strength MS medium under continuous light at 22°C (light intensity: 180 µmol m−2 s−1) in growth cabinets. T3 homozygous T-DNA insertion lines were obtained for the PLAT1 gene, plat1-1 SALK-112728c and plat1-2 SALK-1283454c, and the PLAT2 gene, plat2 SAIL-1171C06. The T-DNA insertions were verified with the primers PLAT1-1-F, PLAT1-1-R and SALK LB2 (plat1-1), PLAT1-2-F, PLAT1-1-R and SALK LB2 (plat1-2), and PLAT2-F, PLAT2-R and pROK2 LB1 (plat2) (Table S1 in File S1). For all plant experiments T3 or T4 homozygous plant lines were employed, based on the segregation of the respective antibiotic selection marker, except for the experiments shown in Figure S4, for which segregating T2 Arabidopsis lines were employed.
Abiotic stress conditions
The abiotic stress experiments in soil were performed as 3 biological replicates (cold stress as 2 biological replicates) with at least 10 plants each. For the Arabidopsis germination experiment, seeds were directly germinated on half strength MS medium including the respective chemicals as indicated. At least 100 seeds per treatment/genotype were used in 3 independent experiments. For the salt stress tolerance in plates, seeds were germinated and grown for 6 d on half strength MS medium, transferred to half strength MS medium including NaCl (either 0, 150 or 200 mM) and optionally 5 µM dexamethasone (35S>>PLAT1 lines), and grown for another 4 d. These experiments were performed as 2 biological replicates, each with 2 technical replicates and with >12 Col-0 and >26 mutant/transgenics seedlings per plate. For the tunicamycin (TM) experiments, seeds were directly germinated on half strength MS medium including different TM concentrations. At least 70 seeds per treatment/genotype were used in 3 independent experiments, with 2 technical replicates each.
Biotic stress conditions
To determine pathogen susceptibility, leaves from 8-w-old Arabidopsis plants were infected with Pseudomonas syringae pv. tomato DC3000 with or without the avrRpm1 gene by infiltration using a needleless syringe as described previously [38]. Visual evaluation of disease symptoms were conducted at 3 to 5 d. For expression analysis, 8-w-old plants were infected with P. syringae pv. tomato DC3000 or Sclerotinia sclerotiorum. Subsequently, the S. sclerotiorum infected plants were kept in a clear plastic box under saturating humidity. The biotic stress experiments were performed as 3 biological replicates with at least 10 plants each.
Expression analysis
Total RNA isolation and Northern-blot analysis was carried out as described previously [37]. Filters were exposed to a screen for 4 d, which was scanned with a Phosphor imager (Fuji BAS2000, Ray-test, Germany). The probes for the PLAT1, PLAT2 and PLAT3 genes were generated by PCR from cDNA with the primers, PLAT1-F probe and PLAT1-R probe, PLAT2-F probe and PLAT2-R probe, and PLAT3-F probe and PLAT3-R probe (Table S1 in File S1), respectively. The RT-PCR analysis was performed essentially as described before [37]. At least 10 seedlings per genotype were grown for 14 d on control plates before transfer to the respective stress and control medium. The optimal cycle number was determined for each primer pair (Table S2 in File S1). Expression values were corrected for the ACTIN and UBIQUITIN signal intensities and expressed as relative values compared to Col-0. For expression analysis, 3 independent experiments were performed.
ABA determination
The extraction and analysis of ABA was carried out as described previously [39].
PLAT1-YFP localisation
For transient PLAT1-YFP expression a single colony of A. tumefaciens LBA4404 containing either PLAT1:PLAT1-YFP or the ER-rk CD3-959 ER-marker [40] construct was inoculated into 5 ml induction medium with antibiotics and grown overnight at 28°C. The bacteria were collected by centrifugation and resuspended in 10 mM MES and 10 mM MgCl2 containing 200 µM acetosyringone to an OD600 of 1.0. Aliquots (1 ml) of A. tumefaciens cells containing PLAT1:PLAT1-YFP and ER-marker construct were mixed together, and then a syringe was used to infiltrate the mixture into the lower surface of N. benthamiana leaves. YFP and mCherry fluorescence was visualized 48 h post infiltration, using Olympus confocal laser scanning microscope (model FV1000, Tokyo, Japan). For stable PLAT-YFP expression following ABA and salt treatment, the PLAT1:PLAT1-YFP line YFP13-1 was employed, 10 plants per treatment.
Transactivation assay
Suspensions of A. tumefaciens carrying the respective 35S:ABF1–4 overexpression constructs and the PLAT1:PLAT1-GUS rescue/reporter construct were mixed in a ratio of 1∶1. The resulting mixed suspensions were used to infiltrate leaves of 6-w-old greenhouse grown N. benthamiana in soil. As control, leaves were infiltrated with A. tumefaciens carrying the PLAT1:PLAT1-GUS rescue construct only, or 10 mM MgCl2. The GUS fluorometric assays were carried out as described previously [41]. Samples were isolated from the infiltrated regions 2 d after infiltration for 5 independent plants and ground in 500 µl of extraction buffer containing 50 mM sodium phosphate, 10 mM EDTA, 10 mM β-mercaptoethanol and 0.1% N-lauroylsarcosine (pH = 7.4). After centrifugation at 4°C for 10 min at 13,000 rpm, the supernatant was used for the determination of GUS enzyme activity. 50 µl of supernatants was transferred into one slot of a black 96-well plate and 50 µl of a 2 mM MUG (methylumbellifery-β-D-glucuronide, Sigma) solution was added. For each sample 3 technical replicates were measured. The samples were incubated at 37°C for 30, 60 and 90 min, before the reaction was stopped with 1 M sodium carbonate. A standard curve was prepared with MU (4-methylumbelliferone, Sigma) in a concentration range from 0 to 16 µM. Excitation was measured at 365 nm, emission at 455 nm. Total protein amount was determined by the Bradford assay. GUS enzyme activity was calculated in pmol Mu min−1 mg−1 protein.
Expression and purification of ABFs
The ABFs were prepared employing a MBP-fusion purification procedure. Five ml of an overnight bacterial culture was incubated with 500 ml rich broth medium containing glucose and ampicillin. The cells were grown to 2×108 cells ml−1 (A600 = 0.5). IPTG was added to a final concentration of 0.3 mM and a further incubation at 37°C for 2 h followed. The cells were harvested by centrifugation at 4,000 g for 20 min. The supernatant was discarded and the pellet re-suspended in 25 ml of column buffer (20 mM Tris-HCl, 200 mM NaCl, 1 mM EDTA, 1 mM DTT and 0.1 mM PMSF (pH = 7.4). The pellet was kept at −20°C overnight and thawed in cold water the next morning. The sample was placed in an ice-water bath and sonicated in short pulses of 15 s for at least 2 min. The suspension was centrifuged at 9,000 g for 30 min and the supernatant diluted 1/3 (v/v) with column buffer before purification. Amylase resin was poured in a 5 ml column and washed with 8 column volumes of column buffer. The diluted sample was loaded and slowly ran over the column. The column was then washed with 12 volumes of column buffer and the proteins subsequently eluted with column buffer containing 10 mM maltose. 10 to 15 fractions containing 2 ml each were collected. Proteins were checked via SDS-page.
Electrophoretic mobility shift assay
For EMSA, the 200 bp PLAT1 promoter, generated by PCR with the primers pPLAT1-F and pPLAT1-R (Table S1 in File S1) was used as positive probe. The mutated version lacking the 2 ABRE elements was generated in 2 steps by PCR using the primers WIP-F1 and WIP-R1, and WIP-F2 and WIP-R2. The 2 PCR fragments were joined to create the negative 200 bp probe WIP1. The 200 bp PLAT1 promoter fragment was labelled with [γ-32P]ATP using T4 polynucleotide kinase (5′ end labelling). The reaction was incubated for 1 h at 37°C, purified and eluted with 10 mM Tris, pH = 8.0. The labelled probe was incubated 30 min at room temperature with 5 µg of the respective ABFx protein extracts alone, with 100 fold molar excess of “cold” specific competitor (200 bp PLAT1 promoter), and with 100 fold molar excess of “cold” negative probe (WIP1), including poly-dIdC as nonspecific competitor. The EMSA samples were run on a 5% native poly-acrylamide gel (10×10 cm). Before loading the samples, the gel was pre-run 40 min at 80 V and 4°C and the samples were run at 120 V and 4°C. After electrophoresis, radioactivity was detected in the dried gel as described above. Functionality of the EMSA assay and ABFx preparation using the MBP fusion protein was proven using the ABF1 protein extract and the published [34] positive ABRE-F and ABRE-R, and negative mABRE-F and mABRE-R control primers (Table S1 in File S1).
Statistical analysis
Standard deviations and average values were calculated in excel. Statistical significance for differences between treatments was analysed using the unpaired two sided Student's t-test in excel. ***, ** or * indicate statistical significance at p<0.001, p<0.01 or p<0.05, respectively.
Accession numbers
The AGI locus identifiers for the Arabidopsis PLAT-plant-stress family members are: PLAT1, AT4G39730; PLAT2, AT2G22170 and PLAT3, AT5G65158.
Results
PLAT1 expression is induced by abiotic stress conditions
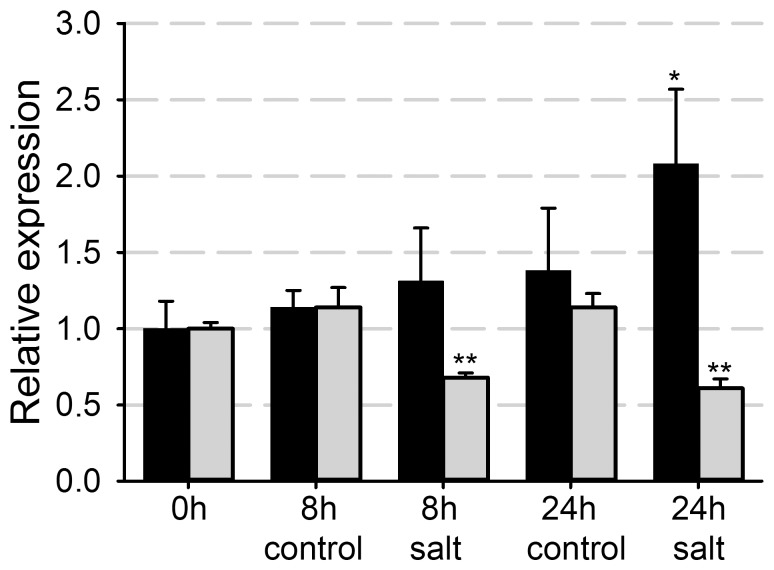
The PLAT-plant-stress subgroup of PLAT domain proteins comprises three Arabidopsis family members. Phylogenetic analysis of these Arabidopsis PLAT-plant-stress proteins using the neighbour-joining method showed that PLAT3 falls outside the other members (Figure S1A). In silico expression analysis by the eFP Browser [42] showed PLAT1 and PLAT2 expression during many developmental stages, which is affected under different stress conditions, whereas PLAT3 is not represented on the Affymetrix ATH1 arrays. To corroborate these data, we investigated expression of the PLAT family members by northern blot and RT-PCR analysis. This showed that PLAT1 was indeed expressed throughout development (Figure S1B). In contrast, PLAT2 expression was only detected in young seedlings (Figure S1E), whereas PLAT3 expression could not be detected in any of the analysed organs and developmental stages, neither by northern blot nor RT-PCR analysis at 35 cycles. PLAT1 expression was induced both by salt, following one day of watering with 200 mM NaCl (Figure S1C), and cold treatment, following incubation of 3-w-old plants at 8°C for 2 d (Figure S1D). PLAT1 expression was also induced following the transfer of young seedlings to medium with 200 mM NaCl (Figure 1). However, both the extent of PLAT1 induction and the temporal dynamics were different from that following salt stress using older plants grown in soil (Figure S1C), probably because PLAT1 expression is highest in young seedlings (Figure S1B). PLAT2 expression was repressed following the transfer of young seedlings to medium with 200 mM NaCl (Figure 1). These results confirmed the in silico expression data and suggested that among the Arabidopsis PLAT family members mainly PLAT1 is involved in abiotic stress responses.
Figure 1. PLAT1 expression is induced by salt stress conditions.
Relative PLAT1 and PLAT2 expression in 14-d-old Col-0 seedlings following transfer to salt stress medium compared to control conditions. PLAT1 black bars, PLAT2 grey bars. Values are means of 3 replicates ± standard deviation. n≥10 per replicate. ** or * indicate statistical significance calculated using the unpaired Student's t-test at p<0.01 or p<0.05, respectively.
PLAT1 promotes tolerance towards abiotic stress conditions
To analyse PLAT function in stress responses and plant development, we obtained T-DNA insertion mutants for PLAT1 and PLAT2. Two independent homozygous Arabidopsis loss-of-function mutants were obtained for PLAT1 (plat1-1 and plat1-2) and one promoter insertion mutant was obtained for PLAT2 (plat2) from the SALK and SAIL mutant collections, respectively [43], [44]. Since both plat1 mutants exhibited similar phenotypic defects, only the characterisation of the plat1-1 mutant is described in detail. The plat1-1 and plat2 mutants exhibited no obvious growth defects under control conditions and most likely represent null alleles because PLAT1 and PLAT2 expression was not detected in the respective insertion mutants (Figure S1E).
The plat1-1 mutant was more sensitive to salt and drought stress, as well as cold stress conditions (Figure 2), evident by a reduction in root length upon growth at 8°C from 9.41±1.25 mm for Col-0 to 7.02±1.27 mm or 6.20±0.94 mm for plat1-1 and plat1-2, respectively (p<0.001, n = 14). In contrast, the plat2 mutant did not show obvious changes in salt stress tolerance (Figure 3). Together with the differential effect of salt stress on expression of the PLAT family members (Figure 1), these data support that only PLAT1 is involved in abiotic stress tolerance.
Figure 2. PLAT1 loss-of-function reduces abiotic stress tolerance.
(A) Salt stress tolerance in wild-type (Col-0) and plat1-1 seedlings irrigated with 200 mM NaCl for 14 d. n≥10 (B) Drought stress tolerance in wild-type and plat1-1 seedlings, following 14 d without watering. n≥10 (C) Cold stress tolerance in 7-d-old wild-type, plat1-1 and plat1-2 seedlings following 14 d of incubation at 8°C. n = 14. Scale bar = 1 cm.
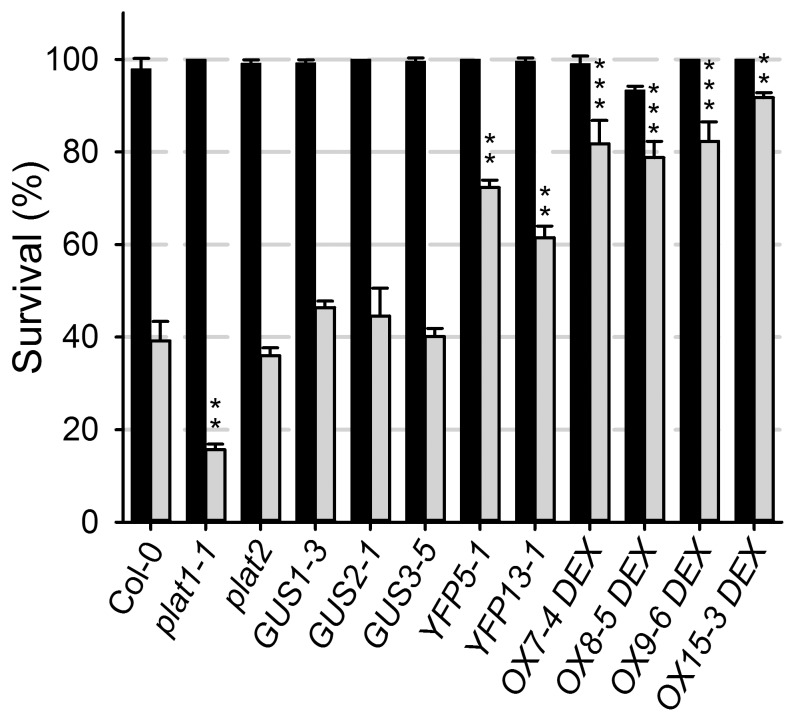
Figure 3. PLAT1 promotes tolerance towards salt stress conditions.
Survival, expressed as the percentage of the seedlings transferred at the 6-d-old stage that developed (pale) green leaves during 4 d of salt stress conditions for the different mutant and transgenic lines (grey bars, n≥26 per replicate) compared to Col-0 control plants (black bars, n≥12 per replicate) that were grown on the same plates. Values are means of 3 replicates ± standard deviation. ***, ** or * indicate statistical significance calculated using the unpaired Student's t-test at p<0.001, p<0.01 or p<0.05, respectively.
To analyse whether PLAT1 also plays a role in biotic stress responses, we studied PLAT1 expression following inoculation of plants with the hemibiotrophic pathogen P. syringae pv. tomato DC3000 or DC3000 avrRpm1 (RPM1, Figure S2A), and the necrotrophic fungal pathogen S. sclerotiorum (Figure S2B). This showed that PLAT1 expression was not specifically affected by these pathogens since increased expression was also observed for the respective control treatments (10 mM MgCl2 and PDA medium). Next, disease symptom development following inoculation with P. syringae pv. tomato DC3000 was investigated. Disease symptom development was not obviously affected in plat1-1 (Figure S2C), which is in agreement with the fact that biotic stress conditions did not significantly affect PLAT1 expression.
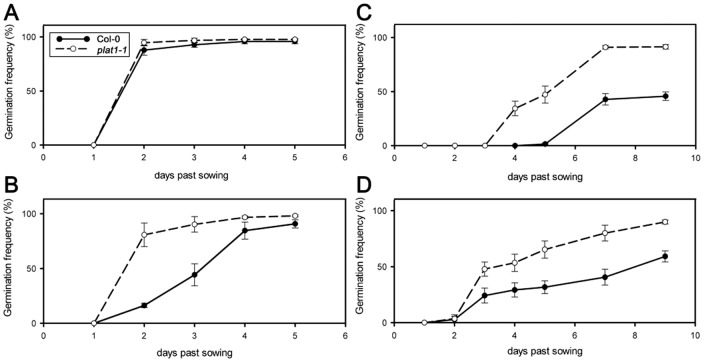
Because ABA is strongly involved in abiotic stress responses, we analysed whether PLAT1 function is correlated with ABA signalling. One characteristic effect of ABA is the inhibition of seed germination [45]. Further, seed germination frequency is reduced by salt and osmotic stress conditions, resulting from increased ABA signalling. Under control conditions the germination frequency of the plat1-1 seeds was similar to that of wild-type (Figure 4A). However, under osmotic stress using 300 mM mannitol (Figure 4B) and salt stress conditions employing 200 mM NaCl (Figure 4C), the plat1-1 seeds exhibited a higher germination frequency compared to wild-type, suggesting that ABA signalling is reduced in plat1-1. Indeed, plat1-1 seed germination is less severely reduced by ABA (1.5 µM) compared to wild-type (Figure 4D).
Figure 4. PLAT1 loss-of-function reduces ABA sensitivity during seed germination.
(A) Seed germination of plat1-1 and wild-type (Col-0). (B) Seed germination of plat1-1 and wild-type on 300 mM mannitol (osmotic stress). (C) Seed germination of plat1-1 and wild-type on 200 mM NaCl (salt stress). (D) Seed germination of plat1-1 and wild-type on 1.5 µM ABA. Values are means of 3 replicates ± standard deviation. n≥100 per replicate.
Since a change in seed dormancy could have influenced the seed germination assays and thus interpretation for ABA sensitivity, we analysed germination for the different PLAT lines on control medium without prior stratification. This showed that seed dormancy was only affected by PLAT2 loss-of-function (Figure S3). Further, ABA levels were not significantly affected in plat1-1 plants compared to wild-type control (Table S3 in File S1). Together, these results indicate that PLAT1 loss-of-function reduced ABA sensitivity, which could have caused increased sensitivity towards abiotic stress conditions.
We generated genomic PLAT1 rescue constructs, where the PLAT1 protein was fused to either the YFP or GUS reporter proteins. Following genetic complementation of the plat1-1 mutant, seed responses towards ABA could be completely restored back to that of wild-type employing the genomic rescue/reporter constructs: 5 out of 6 lines for PLAT1:PLAT1-GUS (Figure S4A) and 15 out of 20 lines PLAT1:PLAT1-YFP (Figure S4B). This confirmed that the defects in abiotic stress tolerance were indeed caused by the T-DNA insertion in the plat1-1 mutant and that the PLAT1 fusion proteins are functional in these rescued lines. Transgenic expression levels can be influenced to a large extend by plant sequences flanking the respective T-DNA integration sites, causing variation between individual transgenic lines harbouring the same T-DNA construct. In addition to transformed plat1-1 lines with restored (wild-type) ABA sensitivity, also transgenics were obtained exhibiting increased ABA sensitivity and thus tissue specific PLAT1 overexpression phenotype (Figure S4). To study whether PLAT1 overexpression would confer an opposite phenotype compared to the plat1 mutant, we selected these lines with tissue specific PLAT1 overexpression for further analysis and also generated ectopic PLAT1 overexpression lines. Because we could not obtain transgenics harbouring constitutive PLAT1 overexpression, we generated lines with inducible PLAT1 overexpression (35S>>PLAT1) employing the dexamethasone inducible OP-LhGR two component system [46]. From 17 transgenic PLAT1 OX lines, the majority exhibited delayed germination on 1.5 µM ABA (Figure S4C).
To compare tissue specific with ectopic overexpression, which could cause considerable differences in overexpression phenotypes [47], we selected the GUS1-3, GUS2-1, GUS3-5, YFP5-1 and YFP13-1 genomic rescue lines with increased ABA sensitivity in the seed germination assays for further analysis. In these selected tissue specific (GUS and YFP) PLAT1 overexpression lines, PLAT1 transcript levels were increased to a similar extend as the ectopic (OX) overexpression lines, compared to wild-type control (Table S4 in File S1). The different types of PLAT1 overexpression lines promoted salt stress tolerance after transfer to medium containing both 200 mM NaCl and 5 µM dexamethasone (DEX) (Figure 3). Thus, PLAT1 overexpression resulted in higher PLAT1 expression levels and reciprocal phenotypic effects compared to the plat1-1 mutant, confirming that PLAT1 functions in promoting tolerance towards abiotic stress conditions.
PLAT1 promotes growth
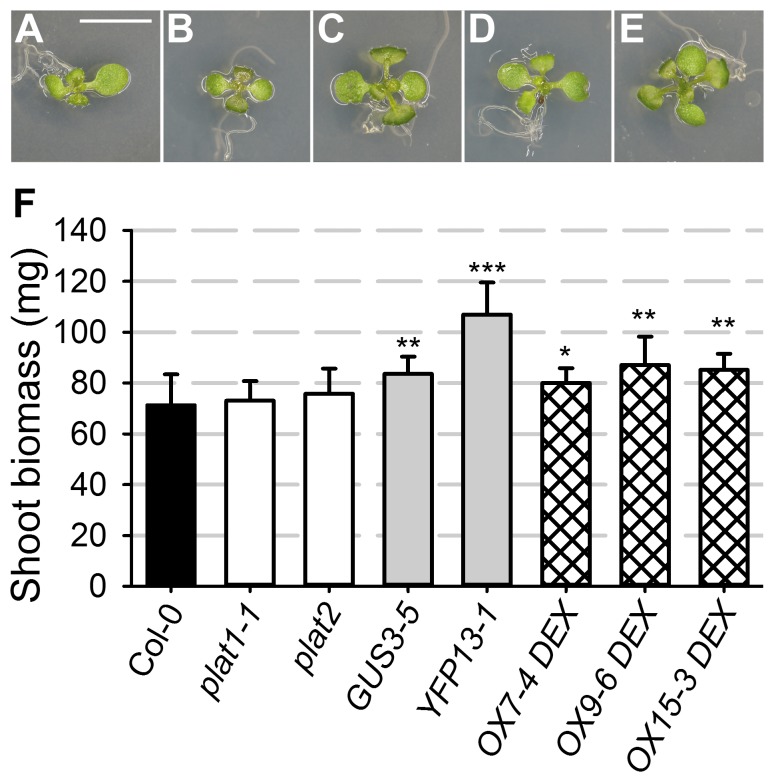
The DEX induced PLAT1 OX lines from the salt stress experiment shown in Figure 3, appeared to show faster development on control medium including DEX (Figure 5A-E). Therefore, shoot and root growth was analysed in the plat1-1 mutant and PLAT1 overexpression lines. This showed that the different PLAT1 overexpression lines produced more shoot biomass compared to wild-type control (Figure 5F), while the length of the root apical meristem and total root length were not affected (Table S5 in File S1). Interestingly, the number of emerged lateral roots was increased, but only for the ectopic PLAT1 (OX) overexpression lines (Table S5 in File S1). This effect on lateral root formation was the only difference observed between the tissue specific and ectopic overexpression lines, and could reflect the difference between increased expression at its natural expression site (GUS and YFP) and broader expression in new (ectopic) cell types (OX). In contrast, PLAT1 loss-of-function did not affect either shoot or root growth, except for a reduced number of emerged lateral roots (Figure 5F, Table S5 in File S1).
Figure 5. PLAT1 promotes plant growth.
Phenotypes of plants from control medium including 5 µM DEX from the salt stress experiment shown in Figure 3. (A) Wild-type. (B) plat1-1. (C-E) Three independent PLAT1 ectopic overexpression lines (OX). Scale bar = 1 cm. (F) Shoot biomass production (weight per 5 shoots) for the different PLAT1 lines. Values are means of 8 replicates ± standard deviation. ***, ** or * indicate statistical significance calculated using the unpaired Student's t-test at p<0.001, p<0.01 or p<0.05, respectively.
PLAT1 is expressed in structures related to the regulation of water household
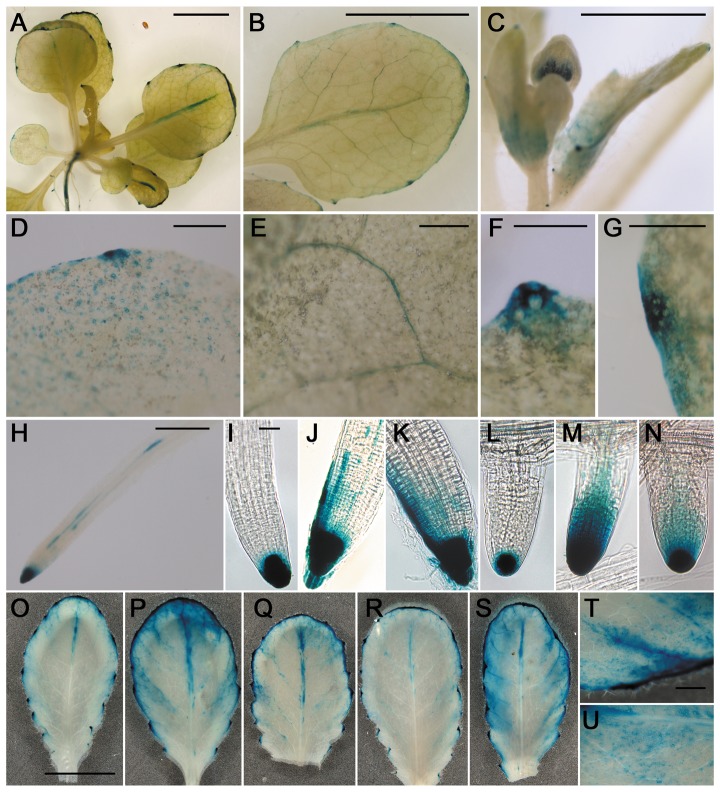
The PLAT1:PLAT1-GUS rescue/reporter construct conferred GUS activity (4 out of 6 lines analysed) in vascular tissue, leaf edges, hydathodes and stomata, which are structures related to the regulation of water household. The expression in stomata corresponds to the identification of PLAT1 in guard cells [48]. GUS activity could also be observed in floral organs, root tips, pericycle cells and lateral root primordia (Figure 6A-6H). The basal PLAT1 expression pattern in the shoot correlated with the abiotic stress tolerance, while that of the root correlated with the changes in lateral root number for the plat1-1 mutant and PLAT1 overexpression lines. Both treatment with salt and ABA resulted in an expanded PLAT1 expression domain in root tips (Figure 6I-6N) and leaves (Figure 6O-6U), evident by the GUS activity in leaf mesophyll cells (Figure 6T and 6U), as well as in root pericycle cells (Figure S5).
Figure 6. PLAT1 is expressed in the vasculature, hydathodes and stomata of aerial organs.
PLAT1 expression is reflected by GUS activity in plat1-1 mutant plants rescued by the PLAT1:PLAT1-GUS rescue/reporter construct (line GUS3-5). The PLAT1 promoter confers expression in the leaf vasculature (A, B, E), hydathodes (B, F, G), floral organs (C), stomata (D) and the primary root tip and root pericycle cells (H). PLAT1 expression in primary root tips, 24 h following transfer to control (I), 200 mM NaCl (J) or 1.5 µM ABA plates (K). PLAT1 expression in lateral root tips, 24 h following transfer to control (L), 200 mM NaCl (M) or 1.5 µM ABA plates (N). PLAT1 expression in fully expanded rosette leaf from 4-w-old seedling, control (O), following 24 h (P), or 48 h (Q) of treatment with 1.5 µM ABA and following 24 h (R), or 48 h (S) watering with 200 mM NaCl. Detail of rosette leaf showing expansion of expression domain in leaf mesophyll following 24 h ABA treatment (T), or 48 h watering with NaCl (U). Scale bar = 1 cm (A-C) and (O-S), 1 mm (D-H), (T), (U), 0.1 mm (I-N). n≥10.
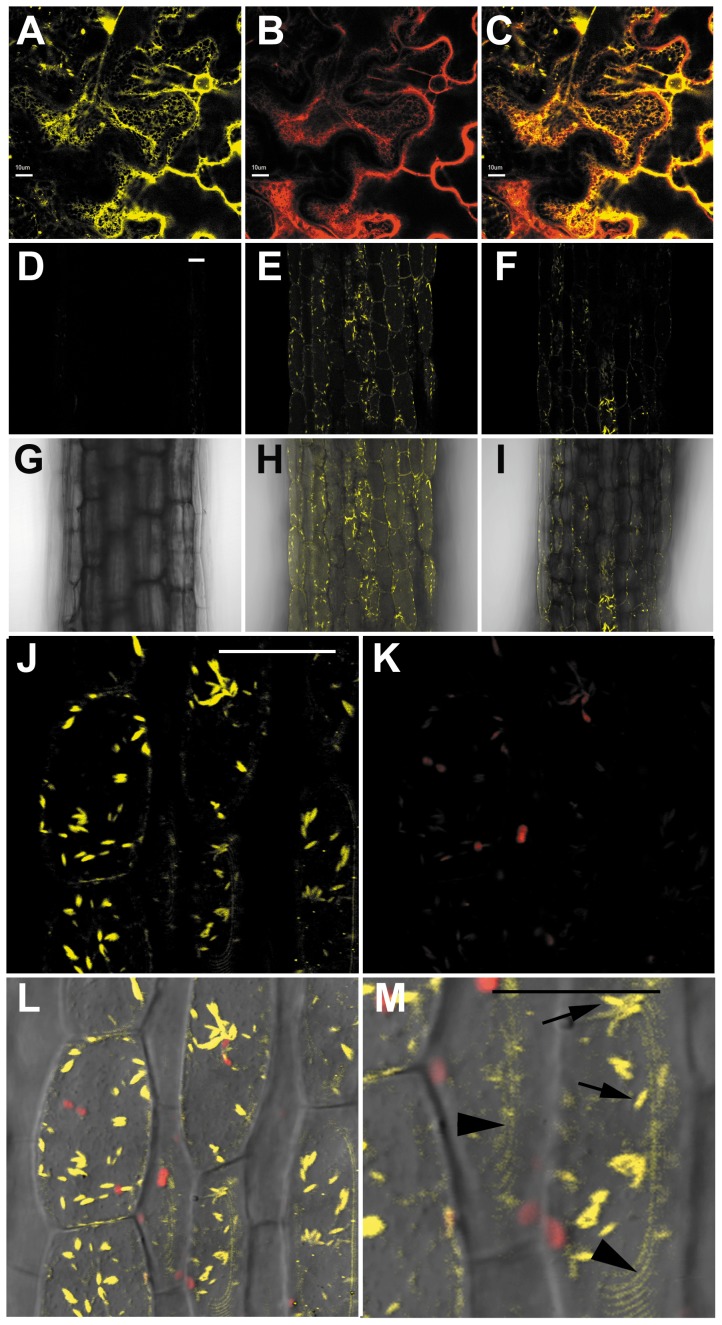
To understand protein function, knowledge on its sub-cellular localisation is essential, because this influences access to and availability of interaction partners [49]. According to the Aramemnon membrane protein database [50], PLAT1 was predicted to contain a signal peptide involved in the secretory pathway and one transmembrane spanning domain. To analyse the sub-cellular localisation we transiently transformed the PLAT1:PLAT1-YFP construct together with different organelle markers [40] to N. benthamiana, which showed that the PLAT1-YFP fusion protein co-localised with the mCherry ER-marker ER-rk CD3-959 (Figure 7A-7C). The analysis of YFP reporter activity in the stable rescued plat1-1 transformants harbouring the PLAT1:PLAT1-YFP construct reflected the expression pattern evident from the GUS rescue reporter lines, confirmed PLAT1 induction by salt and ABA (Figure 7D-7I), and PLAT1 localisation to the ER (Figure 7J-7M). Further, PLAT1 is localised to rod shaped ER structures that resemble ER bodies (Figure 7M).
Figure 7. PLAT1 is localised to the ER in Arabidopsis and Nicotiana benthamiana.
Transient transformation of the PLAT1:PLAT1-YFP construct to N. benthamiana. YFP channel showing PLAT1-YFP expression (A), RFP channel showing ER-rk CD3-959 mCherry marker expression (B), co-localisation of PLAT1-YFP with the ER-rk CD3-959 marker (C). PLAT1 expression in stable plat1-1 transgenics (line YFP13-1) rescued by the PLAT1:PLAT1-YFP rescue/reporter construct (D-I) 48 h following transfer of 3-d-old seedlings to control, NaCl or ABA plates, with YFP channel (D-F) and merged YFP and bright field channels (G-I). PLAT1 expression following transfer to control medium (D, G), following transfer to 200 mM NaCl (E, H), and expression following transfer to 1.5 µM ABA (F, I). Hypocotyl section from line YFP13-1 with YFP channel showing PLAT1:PLAT1-YFP reporter activity (J), red autofluorescence of chloroplasts (K), merged image of bright field, YFP and red autofluorescence (L). (M) Detail of (L), showing PLAT1 localisation to putative ER bodies (arrows) and the ER (arrow heads). Scale bar = 10 µm (A-C), 0.1 mm (D-L), 0.05 mm (M). n≥10.
The PLAT1 promoter is a direct target of the ABF transcription factors
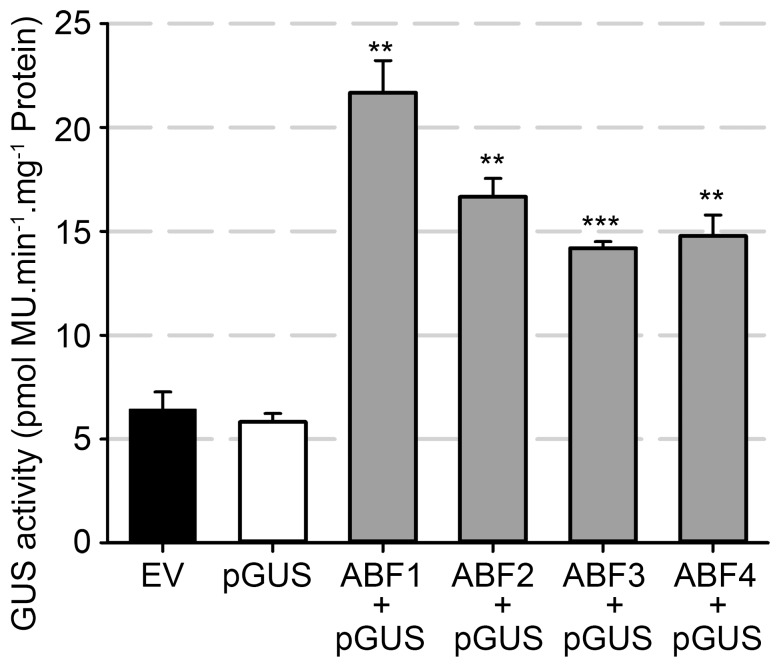
Sequence analysis of the PLAT1 promoter identified 2 G-boxes/ABRE elements (CACGTG motif) located at positions −165 to −156 and −134 to −126 relative to the transcription start site. ABRE elements are direct binding sites of the ABF/AREB (bZIP) transcription factors, which are positive mediators of the ABA signalling pathway [34], [35]. We employed a transactivation assay to investigate whether the PLAT1 promoter is indeed regulated by these ABF transcription factors. Leaves from wild-type tobacco N. benthamiana plants were simultaneously infiltrated with A. tumefaciens harbouring either one of the 35S:ABF1–4 overexpression constructs (ABF1–4) and the PLAT1:PLAT1-GUS genomic rescue/reporter construct (pGUS). Each of the double infiltrations resulted in significantly higher GUS activity levels, compared to infiltration with the PLAT1:PLAT1-GUS reporter construct only (Figure 8). This indicated that the ABF transcription factors activate the PLAT1 promoter.
Figure 8. The PLAT1 promoter is activated by the ABF transcription factors.
GUS activity in 8-w-old N. benthamiana leaves infiltrated with A. tumefaciens harbouring the PLAT1:PLAT1-GUS reporter (pGUS) or an empty vector (EV) as negative control, compared to the simultaneous infiltration of the 35S:ABF1–4 and PLAT1:PLAT1-GUS constructs. Values are means of 3 replicates ± standard deviation. *** or ** indicate statistical significance calculated using the unpaired Student's t-test at p<0.001 or p<0.01, respectively.
Electrophoretic mobility shift assays (EMSA) confirmed these transactivation results and showed that the ABF1, ABF3 and ABF4 transcription factors directly bind to the PLAT1 200 bp promoter fragment (−200 to +1), which contains the 2 ABRE elements. This ABF binding was specifically competed with a 100-fold molar excess of unlabelled 200 bp PLAT1 promoter fragment (PLAT1; Figure S6), but not with the mutated 200 bp promoter fragment WIP1, where point mutations were introduced in the two ABRE elements (WIP1; Figure S6). Thus, PLAT1 expression is regulated by the ABA signalling pathway as direct target of the ABF transcription factors.
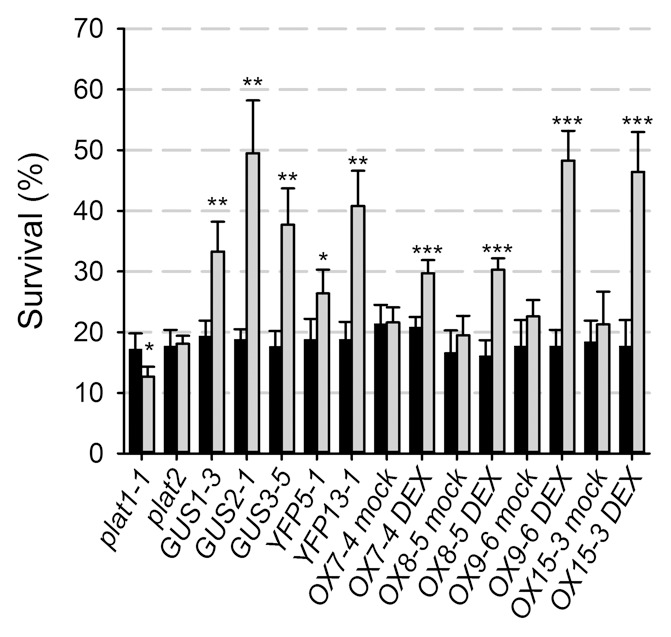
PLAT1 promotes tolerance towards ER stress elicited by tunicamycin
The PLAT1 subcellular localisation in the ER (Figure 7) suggested that PLAT1 function is related to ER stress. Under stress conditions, misfolded proteins accumulate in the ER that eventually cause ER stress [51], resulting in reduced growth and induction of the unfolded protein response (UPR) to compensate for this increased accumulation of misfolded proteins. TM is used as ER stress agent and interferes with N-linked glycosylation of secreted glycoproteins, which prevents protein folding in the ER [51]. The tolerance of the different PLAT1 overexpression lines towards TM was improved compared to Col-0 wild-type (Figure 9). In contrast, the plat1-1 mutant was more sensitive, while the response towards TM was not affected in the plat2 mutant (Figure 9), which supports a function for PLAT1 in ER stress responses and/or UPR. The basal expression levels for the ER stress markers BIP1,2, CNX1, CRT1 and PDIL [52] was higher for the different PLAT1 overexpression lines (Figure S7), which suggests that the capacity of these lines to deal with unprocessed and/or misfolded proteins in the ER is increased, which could contribute to an increased abiotic stress tolerance.
Figure 9. PLAT1 promotes tolerance towards tunicamycin elicited ER stress.
Survival is expressed as the percentage of the plated seeds that developed (pale) green seedlings. Control conditions black bars, ER stress (0.05 µg l−1 TM) grey bars. Values are means of 3 replicates ± standard deviation. n≥70 per replicate. *** or ** indicate statistical significance calculated using the unpaired Student's t-test at p<0.001 or p<0.01, respectively.
Discussion
The complete Arabidopsis genome sequence is available, nevertheless only for a relatively low percentage of Arabidopsis proteins experimental evidence concerning their function is available. While among the PLAT-plant-stress family only CaTIN1 function has been studied by heterologous expression in tobacco [4], for other family members only limited in silico expression data is available. This showed that these members were induced by different abiotic and biotic stimuli, suggesting that PLAT-plant-stress proteins in general could promote tolerance towards stress responses and thus can be of great importance for developing stress tolerant crops. Nevertheless, little information is available on the function of these proteins. We assigned a function to the previously uncharacterised Arabidopsis PLAT domain proteins, which are members of this PLAT-plant-stress subgroup. The combination of genetic and physiological approaches supported functional diversification within this Arabidopsis PLAT protein family. PLAT1 critically functions as positive regulator of abiotic stress tolerance and also confers increased plant growth. Because PLAT1 is a direct activated target of the ABF transcription factors and PLAT1 levels affect ABA sensitivity based on seed germination assays, PLAT1 represents a novel downstream target of the ABA signalling pathway. PLAT2 appears to function specifically in seed dormancy, while PLAT3 might represent a non-expressed pseudogene.
Analysis of the plat1-1 loss-of-function mutant, PLAT1 overexpression lines and genetic complementation of the plat1-1 mutant, showed that PLAT1 critically functions as positive regulator of abiotic stress tolerance. Abiotic stress treatments induced PLAT1 expression, PLAT1 loss-of-function resulted in reduced abiotic stress tolerance, whereas PLAT1 overexpression conferred an inverse response, evident from increased cold, drought and salt stress tolerance. No change in pathogen susceptibility was detected for the plat1-1 loss-of-function mutant, which correlates with the fact that pathogen infection did not significantly affect PLAT1 expression. It is however possible that an altered pathogen resistance in plat1-1 could have been too weak to be detected in the employed pathosystem. Otherwise, functional redundancy among the PLAT family members (partially) could have compensated for PLAT1 loss-of-function in biotic stress tolerance.
High ABA levels confer resistance towards abiotic stress conditions like drought and salinity, which is linked to its role in regulating stomatal aperture, while ABA shows antagonistic interaction with pathogen defence signalling pathways [26], [28]–[31]. The ABF/ABRE transcription factors, which are positive mediators of the ABA signalling pathway [34], [35], were shown to be induced by cold (ABF1), salinity (ABF2 and ABF3) and drought stress (ABF4) [34], while ABF2 or ABF3 overexpression enhanced abiotic stress tolerance [53]. PLAT1 overexpression increased and loss-of-function reduced sensitivity towards ABA during seed germination. Because ABA levels were not significantly affected in the different PLAT1 lines, PLAT1 overexpression resembled the effect of increased ABA signalling, including the differential regulation of abiotic and biotic stress tolerance.
Although it is widely speculated that the effect of ABA signalling on stress tolerance in higher plants is regulated by the complex (antagonistic) interactions with other phytohormones, our understanding of the ABA signalling pathway leading to the adaptation of naturally occurring multi-stress responses remains unclear [54]. Genes harbouring two ABRE elements in their promoter were predicted to be direct (activated) target genes of the ABF/ABRE transcription factors [34] and the expression for a large number of such genes was affected by a triple ABF loss-of-function mutant [35]. However, only the DREB2C and RD29B genes were functionally shown to be direct ABF target genes [55], [56]. The PLAT1 promoter harbours two ABRE elements, located at positions −165 to −156 and −134 to −126 relative to the transcription start site. In agreement with these findings, transactivation and EMSA experiments showed that the ABF transcription factors directly bind to and activate the PLAT1 promoter. Analysis of plat1-1 lines complemented with the PLAT1:PLAT1-GUS rescue/reporter construct showed that PLAT1 is expressed in the leaf vasculature, hydathodes and stomata, which are structures linked to the regulation of water household, but also at specific regions in the root. Together with the induced PLAT1 expression levels as well as expanded expression domain following ABA treatment and abiotic stress conditions, PLAT1 expression correlates with PLAT1 function in abiotic stress tolerance and its regulation by ABA signalling. Together, these data showed that PLAT1 represents a novel component of the ABA signalling as direct target of the ABF transcription factors, which might explain its function in stress tolerance.
ABA in general negatively affects plant growth, mainly through crosstalk with the brassinosteroid pathway [57], and promotes quiescence of stem cells resulting in reduced root growth and lateral root formation [58]–[60]. In addition to the effects on stress tolerance, which could be correlated with PLAT1 being a downstream target of the ABA signalling pathway, PLAT1 promotes plant growth, evident by a faster development and consequently increased shoot biomass. Therefore, PLAT1 function exhibits both expected and unexpected ABA related responses. Heterologous CaTIN1 overexpression in tobacco also resulted in increased abiotic stress tolerance and plant growth, but additionally conferred increased biotic resistance, probably through influencing the redox state. Further, CaTIN1 expression was induced by ethylene treatment and infection by tobacco mosaic virus, but not following ABA treatment [4]. Thus, despite the fact that PLAT1 and CaTIN1 are orthologs, partially convergent evolution occurred in the different plant species on protein function and transcriptional regulation.
Transient expression experiments and analysis of plat1-1 lines complemented with the PLAT1:PLAT1-YFP rescue/reporter construct showed that PLAT1 is localised to the ER, but also in rod shaped structures resembling ER bodies. This is supported by the induction of these structures with PLAT1-YFP signals following ABA treatment or salt stress conditions. ER bodies are specific to Brassicaceae and induced following stress conditions and wounding [61]–[63], but no direct correlation between ER bodies and abiotic stress responses has been shown. PLAT1 promotes tolerance towards the ER stress elicitor TM and the basal expression levels of ER stress markers, representing chaperonins functioning in ER stress and/or UPR.
Together our results indicate that PLAT1 functions in abiotic stress tolerance, either directly through promoting abiotic stress responses, and/or indirectly through improving basal tolerance/fitness. A direct promotion of abiotic stress responses could result from promoting ABA signaling, which is related to its function as novel direct target gene of the ABA signaling pathway. An indirect effect could result from stimulating ER stress responses for a higher basal tolerance/fitness. ER stress responses were shown to be indispensable for abiotic stress responses [64], [65] and the ER appears to play a prominent role in ABA-mediated stress signalling since ABA release from the ER is important for plants coping with stress [66]. The PLAT1 protein essentially harbours one transmembrane spanning domain and one large PLAT domain that covers the rest of the protein sequence and which has been shown to function in protein interaction. Therefore, PLAT1 most likely does not possess enzymatic activity, but rather functions as ‘docking site’ for interacting proteins with enzymatic or signalling activity functioning in ABA regulated pathways, enabling PLAT1 to regulate their activity.
To obtain plants through biotechnology or breeding approaches with increased tolerance towards adverse conditions, but without yield penalties under optimal growth conditions, it is important to identify all genes involved in stress responses and understand their function. Therefore, the identification and assignment of a function to the previously uncharacterised PLAT-plant-stress family member PLAT1 contributes to this important goal. The improved plant growth associated with the increased tolerance towards cold, drought and salt stress mediated by PLAT1 overexpression could be an important asset in crop improvement. To enable the application of PLAT1 or other members from the PLAT-plant-stress family in crop improvement, future studies will be needed to address the multifaceted role of these proteins in stress tolerance and plant development.
Supporting Information
PLAT1 expression patterns under different conditions. (A) Phylogenetic tree of the PLAT-plant-stress subgroup. Phylogenetic analysis was carried out using the neighbour-joining method with 100 bootstraps and displayed using TreeTop. Glycine max (Glyma), Zea mays (GRMZM), Oryza sativa (Os), Populus trichocarpa (POPTR), Sorghum bicolor (Sb). (B) PLAT1 expression in different organs from 3-w-old, 6-w-old and 12-w-old wild-type (Col-0) plants: F, Flower; H, hypocotyl; L, leaf; R, root; S, inflorescence stem and W, whole plant. (C) PLAT1 expression following salt treatment (right) compared to control watering (left). (D) PLAT1 expression following cold treatment. (E) PLAT1 and PLAT2 expression by RT-PCR in the respective T-DNA insertion mutants plat1-1 and plat2. Bottom panels, rRNA for loading control (A-D).
(TIF)
PLAT1 loss-of-function does not affect biotic stress tolerance. (A) PLAT1 expression following leaf infiltration of 107 cfu ml−1 of P. syringae pv. tomato DC3000 or DC3000 avrRpm1 in 10 mM MgCl2, compared to control treatment (MgCl2). (B) PLAT1 expression following infection with S. sclerotiorum compared to control treatment (PDA). (C) Leaves from wild-type (Col-0) plants (Top panel) and plat1-1 plants (Bottom panel), 3 d after infection with 107 cfu ml−1 P. syringae pv. tomato DC3000. Scale bar = 1 cm, n≥10.
(TIF)
PLAT2 functions in seed dormancy. Seed germination of plat1-1, PLAT1:PLAT1-GUS line GUS3-5, 35S>>PLAT1 line OX9-6, plat2 and wild-type (Col-0) on control medium including 5 µM DEX without prior stratification. Values are means of 3 replicates ± standard deviation. n≥100 per replicate.
(TIF)
Increased ABA sensitivity by tissue specific or ectopic PLAT1 overexpression. (A, B) Seed germination of plat1-1, wild-type (Col-0) and plat1-1 lines complemented with the PLAT1:PLAT1-GUS rescue construct (GUS) (A), or plat1-1 lines complemented with the PLAT1:PLAT1-YFP rescue construct (YFP) (B) on medium supplemented with 1.5 µM ABA. (C) Seed germination of wild-type and transgenic lines harbouring the 35S>>PLAT1 ectopic overexpression construct (OX) on medium supplemented with 1.5 µM ABA and 5 µM DEX. Values are means of 3 replicates ± standard deviation. n≥100 per replicate.
(TIF)
PLAT1 expression is induced in adult roots by ABA treatment and salt stress. PLAT1:PLAT1-GUS seedlings were monitored for PLAT1 expression 8 h (A-C) and 24 h (D-F) following transfer to control, NaCl or ABA plates. (A, D) Detail of 2-w-old adult root with PLAT1 expression in emerging lateral root primordia following transfer to control medium. (B, E) Detail of adult root with expanded expression domain following transfer to 200 mM NaCl. (C, F) Detail of adult root with expanded expression domain following transfer to 1.5 µM ABA. Scale bar = 0.1 mm, n≥10.
(TIF)
PLAT1 functions as direct ABF target in ABA signalling. EMSA assay showing that the ABF transcription factors bind to the 200 bp PLAT1 promoter region containing two ABRE elements PLAT1 (*). This binding was specifically competed with a 100 molar excess of unlabelled PLAT1 promoter fragment (PLAT1), but not the negative probe lacking the two ABRE elements (WIP1). Arrowhead indicates shifted band. Bracket indicates free probe.
(TIF)
PLAT1 overexpression lines exhibit higher basal expression levels for ER stress markers. Relative expression levels for ER stress markers in the different PLAT1 overexpression lines compared to wild-type (Col-0) and the plat1-1 mutant. (A) BIP1,2 (HSP70), (B) CNX1 (CALNEXIN1), (C) CRT1 (CALRETICULIN1) and (D) PDIL (PROTEIN DISULFIDE ISOMERASE-like). Values are means of 3 replicates ± standard deviation. n≥10 per replicate. ***, ** or * indicate statistical significance calculated using the unpaired Student's t-test at p<0.001, p<0.01 or p<0.05, respectively.
(TIF)
Combined file containing supporting tables. Table S1: List of primers used for cloning, genotyping of T-DNA insertion mutants and EMSA controls. Table S2: List of primers used for RT-PCR. Table S3: ABA levels are not affected by PLAT1. Table S4: PLAT1 transcript levels in the different overexpression lines. Table S5: PLAT1 promotes lateral root formation.
(DOC)
Acknowledgments
We thank A. Luidold for help with the seed germination assays, K. Remele and W. Pfeifhofer for help with ABA determination.
Data Availability
The authors confirm that all data underlying the findings are fully available without restriction. All relevant data are within the paper and its Supporting Information files.
Funding Statement
This research was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (NRF-2014R1A1A2004432), Republic of Korea (T.K.H.), and by the Fundación Séneca, Comunidad Autónoma de la Región de Murcia (A.A.). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1. Minor W, Steczko J, Stec B, Otwinowski Z, Bolin JT, et al. (2006) Crystal structure of soybean lipoxygenase L-1 at 1.4 å resolution. Biochemistry 35: 10687–10701. [DOI] [PubMed] [Google Scholar]
- 2. Bateman A, Sandford R (1999) The PLAT domain: a new piece in the PKD1 puzzle. Curr Biol 9: R588–590. [DOI] [PubMed] [Google Scholar]
- 3. Hong Y, Wang TW, Hudak KA, Schade F, Froese CD, et al. (2000) An ethylene-induced cDNA encoding a lipase expressed at the onset of senescence. Proc Natl Acad Sci U S A 97: 8717–8722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Shin R, An JM, Park CJ, Kim YJ, Joo S, et al. (2004) Capsicum annuum tobacco mosaic virus-induced clone 1 expression perturbation alters the plant's response to ethylene and interferes with the redox homeostasis. Plant Physiol 135: 561–573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Tomchick DR, Phan P, Cymborowski M, Minor W, Holman TR (2001) Structural and functional characterization of second-coordination sphere mutants of soybean lipoxygenase-1. Biochemistry 40: 7509–7517. [DOI] [PubMed] [Google Scholar]
- 6. Allard JB, Brock TG (2005) Structural organization of the regulatory domain of human 5-lipoxygenase. Curr Protein Pept Sci 6: 125–131. [DOI] [PubMed] [Google Scholar]
- 7. Hu J, Barr MM (2005) ATP-2 interacts with the PLAT domain of LOV-1 and is involved in Caenorhabditis elegans polycystin signaling. Mol Biol Cell 16: 458–469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Yuasa T, Takakura A, Denker BM, Venugopal B, Zhou J (2004) Polycystin-1L2 is a novel G-protein-binding protein. Genomics 84: 126–138. [DOI] [PubMed] [Google Scholar]
- 9. Eek P, Jarving R, Jarving I, Gilbert NC, Newcomer ME, et al. (2012) Structure of a calcium-dependent 11R-lipoxygenase suggests a mechanism for Ca2+ regulation. J Biol Chem 287: 22377–22386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Joshi N, Hoobler EK, Perry S, Diaz G, Fox B, et al. (2013) Kinetic and structural investigations into the allosteric and pH effect on the substrate specificity of human epithelial 15-lipoxygenase-2. Biochemistry 52: 8026–8035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Li ST, Zhang M, Fu CH, Xie S, Zhang Y, et al. (2012) Molecular cloning and characterization of two 9-lipoxygenase genes from Taxus chinensis . Plant Molecular Biology Reporter 30: 1283–1290. [Google Scholar]
- 12. Bhardwaj PK, Kaur J, Sobti RC, Ahuja PS, Kumar S (2011) Lipoxygenase in Caragana jubata responds to low temperature, abscisic acid, methyl jasmonate and salicylic acid. Gene 483: 49–53. [DOI] [PubMed] [Google Scholar]
- 13. Inamdar MI, Bhat RS, Kuruvinashetti MS (2010) Cloning a drought-inducible cDNA (PjPLAT1) from Prosopis juliflora . Karnataka J Agric Sci 23: 350–351. [Google Scholar]
- 14. Leal GA Jr, Albuquerque PS, Figueira A (2007) Genes differentially expressed in Theobroma cacao associated with resistance to witches' broom disease caused by Crinipellis perniciosa . Mol Plant Pathol 8: 279–292. [DOI] [PubMed] [Google Scholar]
- 15. Lee SC, Kim SH, An SH, Yi SY, Hwang BK (2006) Identification and functional expression of the pepper pathogen-induced gene, CaPIP2, involved in disease resistance and drought and salt stress tolerance. Plant Mol Biol 62: 151–164. [DOI] [PubMed] [Google Scholar]
- 16. Shin R, Kim MJ, Paek KH (2003) The CaTin1 (Capsicum annuum TMV-induced clone 1) and CaTin1-2 genes are linked head-to-head and share a bidirectional promoter. Plant Cell Physiol 44: 549–554. [DOI] [PubMed] [Google Scholar]
- 17. Hussain SS, Kayani MA, Amjad M (2011) Transcription factors as tools to engineer enhanced drought stress tolerance in plants. Biotechnol Prog 27: 297–306. [DOI] [PubMed] [Google Scholar]
- 18. Golldack D, Luking I, Yang O (2011) Plant tolerance to drought and salinity: stress regulating transcription factors and their functional significance in the cellular transcriptional network. Plant Cell Rep 30: 1383–1391. [DOI] [PubMed] [Google Scholar]
- 19. Islam S, Griffiths CA, Blomstedt CK, Le TN, Gaff DF, et al. (2013) Increased biomass, seed yield and stress tolerance is conferred in Arabidopsis by a novel enzyme from the resurrection grass Sporobolus stapfianus that glycosylates the strigolactone analogue GR24. PLoS One 8: e80035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Joshi-Saha A, Valon C, Leung J (2011) Abscisic acid signal off the STARting block. Mol Plant 4: 562–580. [DOI] [PubMed] [Google Scholar]
- 21. Finkelstein R, Reeves W, Ariizumi T, Steber C (2008) Molecular aspects of seed dormancy. Annu Rev Plant Biol 59: 387–415. [DOI] [PubMed] [Google Scholar]
- 22. Padmalatha KV, Dhandapani G, Kanakachari M, Kumar S, Dass A, et al. (2012) Genome-wide transcriptomic analysis of cotton under drought stress reveal significant down-regulation of genes and pathways involved in fibre elongation and up-regulation of defense responsive genes. Plant Mol Biol 78: 223–246. [DOI] [PubMed] [Google Scholar]
- 23. Hsu FC, Chou MY, Peng HP, Chou SJ, Shih MC (2011) Insights into hypoxic systemic responses based on analyses of transcriptional regulation in Arabidopsis. PLoS One 6: e28888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Seki M, Narusaka M, Ishida J, Nanjo T, Fujita M, et al. (2002) Monitoring the expression profiles of 7000 Arabidopsis genes under drought, cold and high-salinity stresses using a full-length cDNA microarray. Plant J 31: 279–292. [DOI] [PubMed] [Google Scholar]
- 25. Shinozaki K, Yamaguchi-Shinozaki K, Seki M (2003) Regulatory network of gene expression in the drought and cold stress responses. Curr Opin Plant Biol 6: 410–417. [DOI] [PubMed] [Google Scholar]
- 26. Lee SC, Luan S (2012) ABA signal transduction at the crossroad of biotic and abiotic stress responses. Plant Cell Environ 35: 53–60. [DOI] [PubMed] [Google Scholar]
- 27. Ton J, Flors V, Mauch-Mani B (2009) The multifaceted role of ABA in disease resistance. Trends Plant Sci 14: 310–317. [DOI] [PubMed] [Google Scholar]
- 28. Mosher S, Moeder W, Nishimura N, Jikumaru Y, Joo SH, et al. (2010) The lesion-mimic mutant cpr22 shows alterations in abscisic acid signaling and abscisic acid insensitivity in a salicylic acid-dependent manner. Plant Physiol 152: 1901–1913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Großkinsky DK, van der Graaff E, Roitsch T (2014) Abscisic acid-cytokinin antagonism modulates resistance against Pseudomonas syringae in tobacco. Phytopathology 10.1094/PHYTO-03-14-0076-R [DOI] [PubMed] [Google Scholar]
- 30. Audenaert K, Pattery T, Cornelis P, Hofte M (2002) Induction of systemic resistance to Botrytis cinerea in tomato by Pseudomonas aeruginosa 7NSK2: role of salicylic acid, pyochelin, and pyocyanin. Mol Plant Microbe Interact 15: 1147–1156. [DOI] [PubMed] [Google Scholar]
- 31. Thaler JS, Bostock RM (2004) Interactions between abscisic-acid-mediated responses and plant resistance to pathogens and insects. Ecology 85: 48–58. [Google Scholar]
- 32. Fan J, Hill L, Crooks C, Doerner P, Lamb C (2009) Abscisic acid has a key role in modulating diverse plant-pathogen interactions. Plant Physiol 150: 1750–1761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Sharma N, Cram D, Huebert T, Zhou N, Parkin IA (2007) Exploiting the wild crucifer Thlaspi arvense to identify conserved and novel genes expressed during a plant's response to cold stress. Plant Mol Biol 63: 171–184. [DOI] [PubMed] [Google Scholar]
- 34. Choi H, Hong J, Ha J, Kang J, Kim SY (2000) ABFs, a family of ABA-responsive element binding factors. J Biol Chem 275: 1723–1730. [DOI] [PubMed] [Google Scholar]
- 35. Yoshida T, Fujita Y, Sayama H, Kidokoro S, Maruyama K, et al. (2010) AREB1, AREB2, and ABF3 are master transcription factors that cooperatively regulate ABRE-dependent ABA signaling involved in drought stress tolerance and require ABA for full activation. Plant J 61: 672–685. [DOI] [PubMed] [Google Scholar]
- 36. Clough SJ, Bent AF (1988) Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana . Plant Journal 16: 735–743. [DOI] [PubMed] [Google Scholar]
- 37. Großkinsky DK, Naseem M, Abdelmohsen UR, Plickert N, Engelke T, et al. (2011) Cytokinins mediate resistance against Pseudomonas syringae in tobacco through increased antimicrobial phytoalexin synthesis independent of salicylic acid signaling. Plant Physiol 157: 815–830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Großkinsky DK, Koffler BE, Roitsch T, Maier R, Zechmann B (2012) Compartment specific antioxidative defense in Arabidopsis against virulent and avirulent Pseudomonas syringae . Phytopathology 102: 662–673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Großkinsky DK, Albacete A, Jammer A, Krbez P, van der Graaff E, et al. (2014) A rapid phytohormone and phytoalexin screening method for physiological phenotyping. Mol Plant 7: 1053–1056. [DOI] [PubMed] [Google Scholar]
- 40. Nelson BK, Cai X, Nebenfuhr A (2007) A multicolored set of in vivo organelle markers for co-localization studies in Arabidopsis and other plants. Plant J 51: 1126–1136. [DOI] [PubMed] [Google Scholar]
- 41. Hyun TK, Hoffmann A, Sinha AK, Roitsch T (2009) Tomato mitogen activated protein kinases regulate the expression of extracellular invertase Lin 6 in response to stress related stimuli. Funct Plant Biol 36: 1088–1097. [DOI] [PubMed] [Google Scholar]
- 42. Winter D, Vinegar B, Nahal H, Ammar R, Wilson GV, et al. (2007) An “Electronic Fluorescent Pictograph” browser for exploring and analyzing large-scale biological data sets. PLoS One 2: e718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Sessions A, Burke E, Presting G, Aux G, McElver J, et al. (2002) A high-throughput Arabidopsis reverse genetics system. Plant Cell 14: 2985–2994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Alonso JM, Stepanova AN, Leisse TJ, Kim CJ, Chen H, et al. (2003) Genome-wide insertional mutagenesis of Arabidopsis thaliana . Science 301: 653–657. [DOI] [PubMed] [Google Scholar]
- 45. Barrero JM, Millar AA, Griffiths J, Czechowski T, Scheible WR, et al. (2010) Gene expression profiling identifies two regulatory genes controlling dormancy and ABA sensitivity in Arabidopsis seeds. Plant J 61: 611–622. [DOI] [PubMed] [Google Scholar]
- 46. Craft J, Samalova M, Baroux C, Townley H, Martinez A, et al. (2005) New pOp/LhG4 vectors for stringent glucocorticoid-dependent transgene expression in Arabidopsis. Plant J 41: 899–918. [DOI] [PubMed] [Google Scholar]
- 47. van der Graaff E, den Dulk-Ras A, Hooykaas PJJ, Keller B (2000) Activation tagging of the LEAFY PETIOLE gene affects leaf petiole development in Arabidopsis thaliana . Development 127: 4971–4980. [DOI] [PubMed] [Google Scholar]
- 48. Zhao Z, Zhang W, Stanley BA, Assmann SM (2008) Functional proteomics of Arabidopsis thaliana guard cells uncovers new stomatal signaling pathways. Plant Cell 20: 3210–3226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Scott MS, Calafell SJ, Thomas DY, Hallett MT (2005) Refining protein subcellular localization. PLoS Comput Biol 1: e66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Schwacke R, Schneider A, van der Graaff E, Fischer K, Catoni E, et al. (2003) ARAMEMNON, a novel database for Arabidopsis integral membrane proteins. Plant Physiol 131: 16–26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Howell SH (2013) Endoplasmic reticulum stress responses in plants. Annu Rev Plant Biol 64: 477–499. [DOI] [PubMed] [Google Scholar]
- 52. Liu JX, Srivastava R, Che P, Howell SH (2007) An endoplasmic reticulum stress response in Arabidopsis is mediated by proteolytic processing and nuclear relocation of a membrane-associated transcription factor, bZIP28. Plant Cell 19: 4111–4119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Kim JB, Kang JY, Kim SY (2004) Over-expression of a transcription factor regulating ABA-responsive gene expression confers multiple stress tolerance. Plant Biotechnol J 2: 459–466. [DOI] [PubMed] [Google Scholar]
- 54. Atkinson NJ, Urwin PE (2012) The interaction of plant biotic and abiotic stresses: from genes to the field. J Exp Bot 63: 3523–3543. [DOI] [PubMed] [Google Scholar]
- 55. Uno Y, Furihata T, Abe H, Yoshida R, Shinozaki K, et al. (2000) Arabidopsis basic leucine zipper transcription factors involved in an abscisic acid-dependent signal transduction pathway under drought and high-salinity conditions. Proc Natl Acad Sci U S A 97: 11632–11637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Kim JS, Mizoi J, Yoshida T, Fujita Y, Nakajima J, et al. (2011) An ABRE promoter sequence is involved in osmotic stress-responsive expression of the DREB2A gene, which encodes a transcription factor regulating drought-inducible genes in Arabidopsis. Plant Cell Physiol 52: 2136–2146. [DOI] [PubMed] [Google Scholar]
- 57. Choudhary SP, Yu JQ, Yamaguchi-Shinozaki K, Shinozaki K, Tran LS (2012) Benefits of brassinosteroid crosstalk. Trends Plant Sci 17: 594–605. [DOI] [PubMed] [Google Scholar]
- 58. Fukaki H, Tasaka M (2009) Hormone interactions during lateral root formation. Plant Mol Biol 69: 437–449. [DOI] [PubMed] [Google Scholar]
- 59. Zhang H, Han W, De Smet I, Talboys P, Loya R, et al. (2010) ABA promotes quiescence of the quiescent centre and suppresses stem cell differentiation in the Arabidopsis primary root meristem. Plant J 64: 764–774. [DOI] [PubMed] [Google Scholar]
- 60. Duan L, Dietrich D, Ng CH, Chan PM, Bhalerao R, et al. (2013) Endodermal ABA signaling promotes lateral root quiescence during salt stress in Arabidopsis seedlings. Plant Cell 25: 324–341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Matsushima R, Hayashi Y, Yamada K, Shimada T, Nishimura M, et al. (2003) The ER body, a novel endoplasmic reticulum-derived structure in Arabidopsis. Plant Cell Physiol 44: 661–666. [DOI] [PubMed] [Google Scholar]
- 62. Hara-Nishimura I, Matsushima R (2003) A wound-inducible organelle derived from endoplasmic reticulum: a plant strategy against environmental stresses? Curr Opin Plant Biol 6: 583–588. [DOI] [PubMed] [Google Scholar]
- 63. Yamada K, Hara-Nishimura I, Nishimura M (2011) Unique defense strategy by the endoplasmic reticulum body in plants. Plant Cell Physiol 52: 2039–2049. [DOI] [PubMed] [Google Scholar]
- 64. Liu L, Cui F, Li Q, Yin B, Zhang H, et al. (2011) The endoplasmic reticulum-associated degradation is necessary for plant salt tolerance. Cell Res 21: 957–969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Liu JX, Howell SH (2010) Endoplasmic reticulum protein quality control and its relationship to environmental stress responses in plants. Plant Cell 22: 2930–2942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. Lee KH, Piao HL, Kim HY, Choi SM, Jiang F, et al. (2006) Activation of glucosidase via stress-induced polymerization rapidly increases active pools of abscisic acid. Cell 126: 1109–1120. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
PLAT1 expression patterns under different conditions. (A) Phylogenetic tree of the PLAT-plant-stress subgroup. Phylogenetic analysis was carried out using the neighbour-joining method with 100 bootstraps and displayed using TreeTop. Glycine max (Glyma), Zea mays (GRMZM), Oryza sativa (Os), Populus trichocarpa (POPTR), Sorghum bicolor (Sb). (B) PLAT1 expression in different organs from 3-w-old, 6-w-old and 12-w-old wild-type (Col-0) plants: F, Flower; H, hypocotyl; L, leaf; R, root; S, inflorescence stem and W, whole plant. (C) PLAT1 expression following salt treatment (right) compared to control watering (left). (D) PLAT1 expression following cold treatment. (E) PLAT1 and PLAT2 expression by RT-PCR in the respective T-DNA insertion mutants plat1-1 and plat2. Bottom panels, rRNA for loading control (A-D).
(TIF)
PLAT1 loss-of-function does not affect biotic stress tolerance. (A) PLAT1 expression following leaf infiltration of 107 cfu ml−1 of P. syringae pv. tomato DC3000 or DC3000 avrRpm1 in 10 mM MgCl2, compared to control treatment (MgCl2). (B) PLAT1 expression following infection with S. sclerotiorum compared to control treatment (PDA). (C) Leaves from wild-type (Col-0) plants (Top panel) and plat1-1 plants (Bottom panel), 3 d after infection with 107 cfu ml−1 P. syringae pv. tomato DC3000. Scale bar = 1 cm, n≥10.
(TIF)
PLAT2 functions in seed dormancy. Seed germination of plat1-1, PLAT1:PLAT1-GUS line GUS3-5, 35S>>PLAT1 line OX9-6, plat2 and wild-type (Col-0) on control medium including 5 µM DEX without prior stratification. Values are means of 3 replicates ± standard deviation. n≥100 per replicate.
(TIF)
Increased ABA sensitivity by tissue specific or ectopic PLAT1 overexpression. (A, B) Seed germination of plat1-1, wild-type (Col-0) and plat1-1 lines complemented with the PLAT1:PLAT1-GUS rescue construct (GUS) (A), or plat1-1 lines complemented with the PLAT1:PLAT1-YFP rescue construct (YFP) (B) on medium supplemented with 1.5 µM ABA. (C) Seed germination of wild-type and transgenic lines harbouring the 35S>>PLAT1 ectopic overexpression construct (OX) on medium supplemented with 1.5 µM ABA and 5 µM DEX. Values are means of 3 replicates ± standard deviation. n≥100 per replicate.
(TIF)
PLAT1 expression is induced in adult roots by ABA treatment and salt stress. PLAT1:PLAT1-GUS seedlings were monitored for PLAT1 expression 8 h (A-C) and 24 h (D-F) following transfer to control, NaCl or ABA plates. (A, D) Detail of 2-w-old adult root with PLAT1 expression in emerging lateral root primordia following transfer to control medium. (B, E) Detail of adult root with expanded expression domain following transfer to 200 mM NaCl. (C, F) Detail of adult root with expanded expression domain following transfer to 1.5 µM ABA. Scale bar = 0.1 mm, n≥10.
(TIF)
PLAT1 functions as direct ABF target in ABA signalling. EMSA assay showing that the ABF transcription factors bind to the 200 bp PLAT1 promoter region containing two ABRE elements PLAT1 (*). This binding was specifically competed with a 100 molar excess of unlabelled PLAT1 promoter fragment (PLAT1), but not the negative probe lacking the two ABRE elements (WIP1). Arrowhead indicates shifted band. Bracket indicates free probe.
(TIF)
PLAT1 overexpression lines exhibit higher basal expression levels for ER stress markers. Relative expression levels for ER stress markers in the different PLAT1 overexpression lines compared to wild-type (Col-0) and the plat1-1 mutant. (A) BIP1,2 (HSP70), (B) CNX1 (CALNEXIN1), (C) CRT1 (CALRETICULIN1) and (D) PDIL (PROTEIN DISULFIDE ISOMERASE-like). Values are means of 3 replicates ± standard deviation. n≥10 per replicate. ***, ** or * indicate statistical significance calculated using the unpaired Student's t-test at p<0.001, p<0.01 or p<0.05, respectively.
(TIF)
Combined file containing supporting tables. Table S1: List of primers used for cloning, genotyping of T-DNA insertion mutants and EMSA controls. Table S2: List of primers used for RT-PCR. Table S3: ABA levels are not affected by PLAT1. Table S4: PLAT1 transcript levels in the different overexpression lines. Table S5: PLAT1 promotes lateral root formation.
(DOC)
Data Availability Statement
The authors confirm that all data underlying the findings are fully available without restriction. All relevant data are within the paper and its Supporting Information files.