Abstract
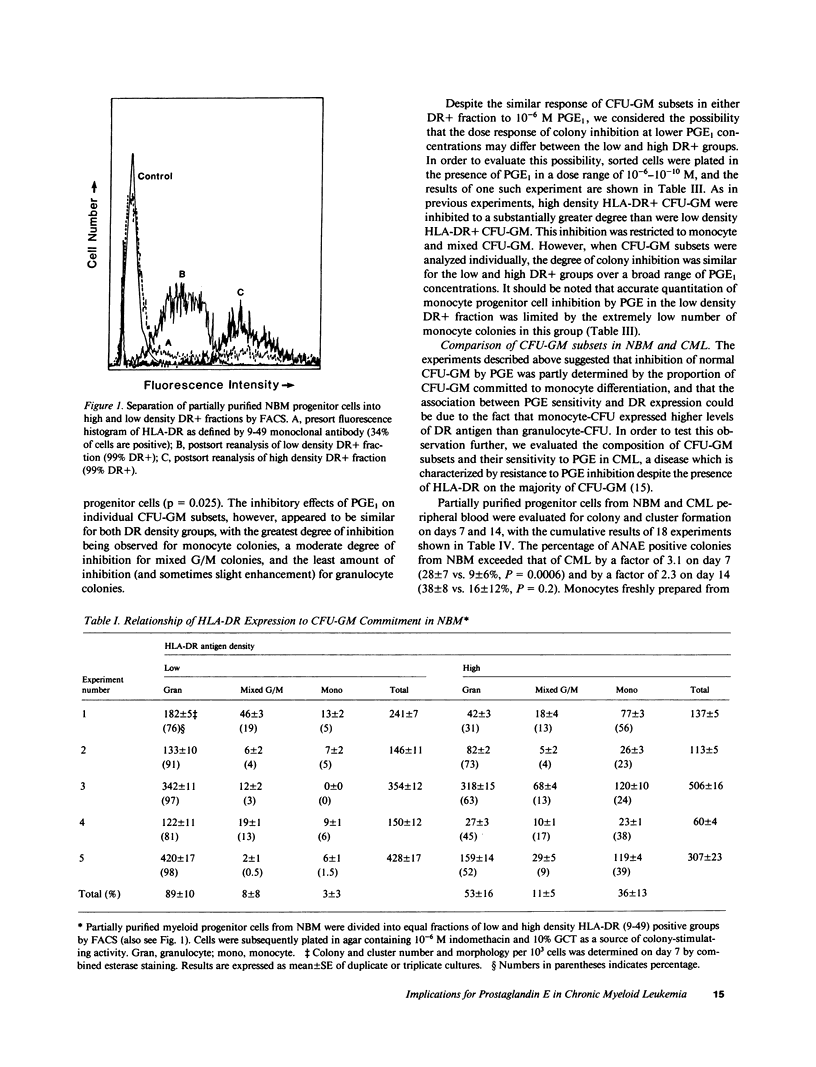
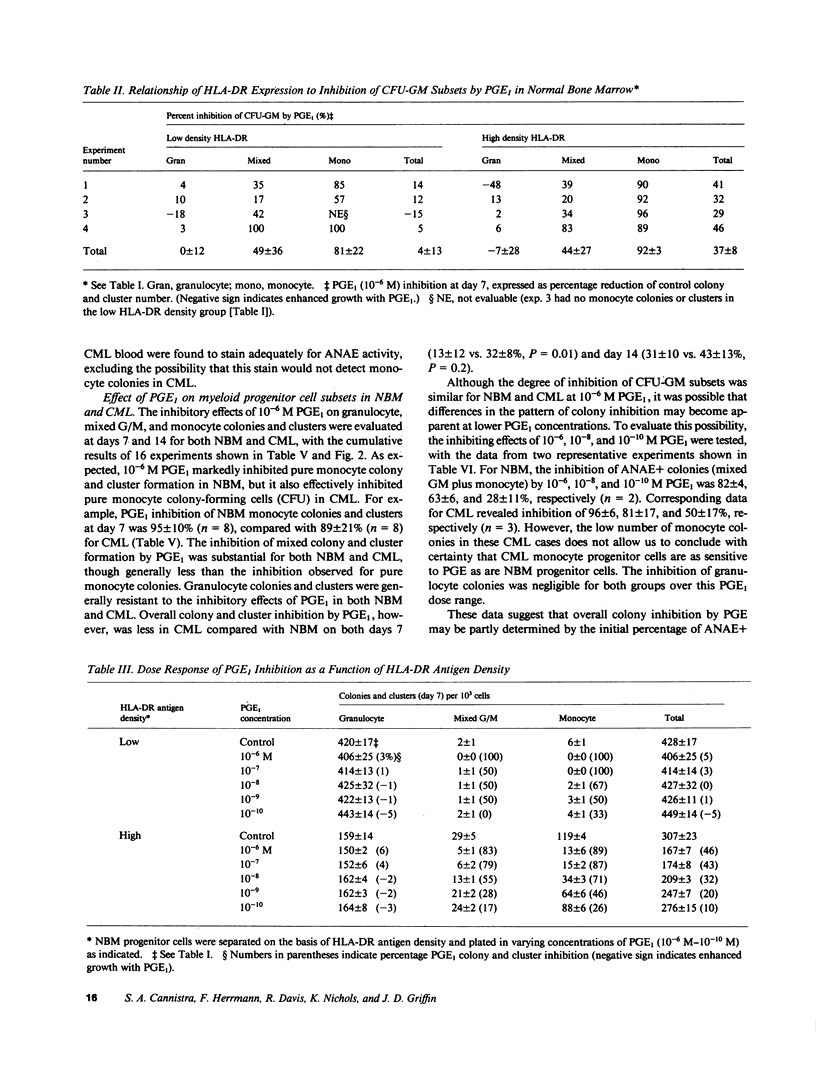
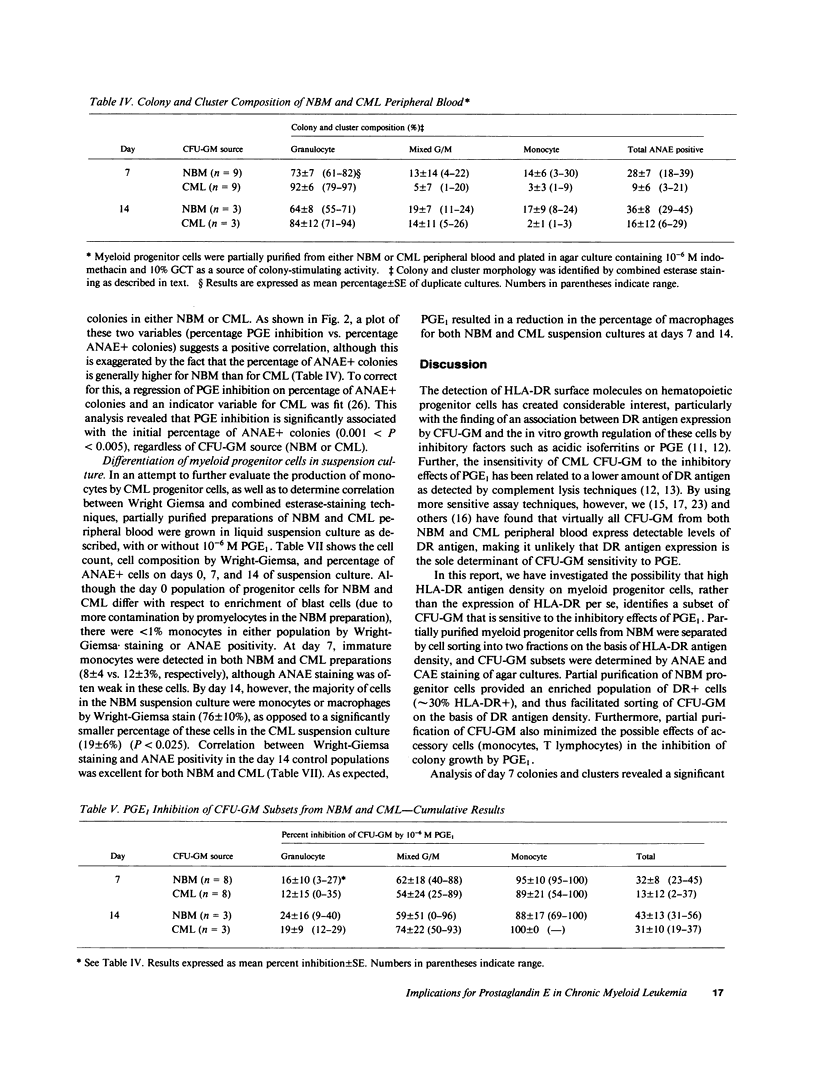
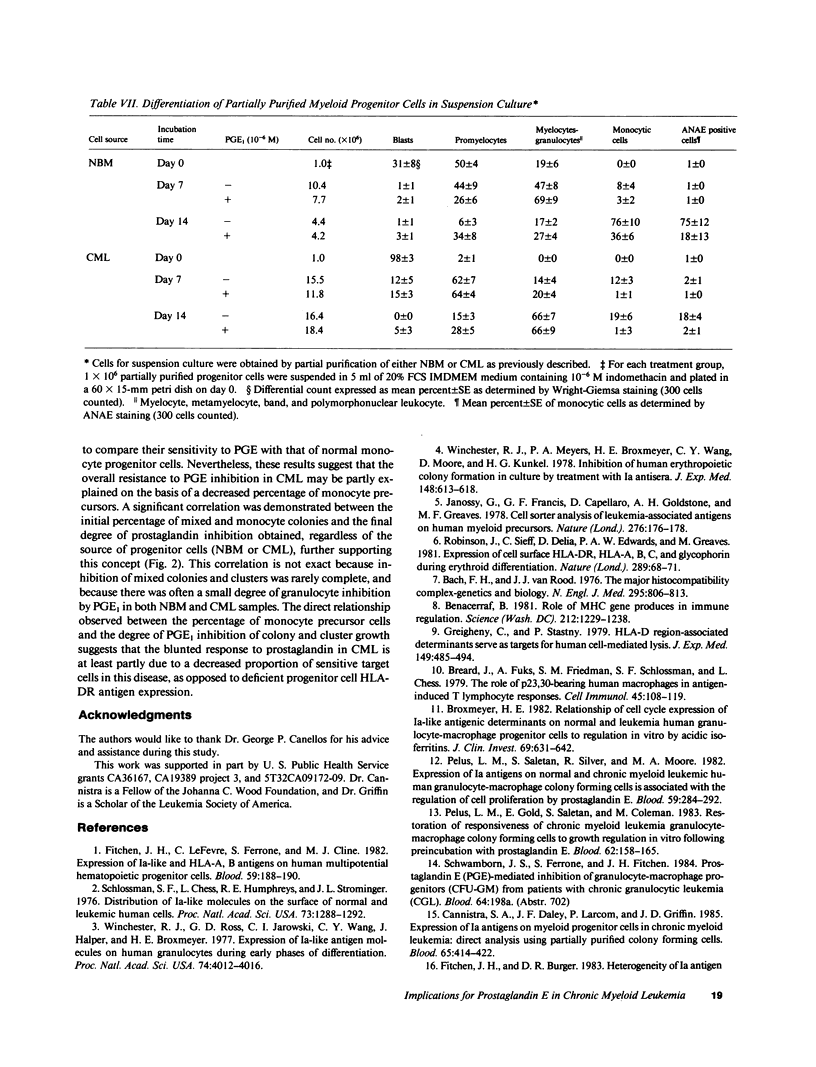
The expression of HLA-DR antigens by normal myeloid progenitor cells (CFU-GM) has been linked to inhibition of colony growth by prostaglandin E (PGE), while resistance to the inhibitory effects of PGE in chronic myeloid leukemia (CML) has been attributed to a lower fraction of HLA-DR+ CFU-GM in this disease. However, we have previously shown that virtually all CFU-GM in normal bone marrow (NBM) as well as CML peripheral blood express HLA-DR antigens, which raises the possibility that these surface molecules may not be the sole determinants of a progenitor cell's sensitivity to PGE. In order to evaluate the relationship between HLA-DR expression and prostaglandin inhibition, we partially purified NBM progenitor cells using fluorescence-activated cell sorting to prepare cell fractions with high and low HLA-DR antigen density. Normal progenitor cells with high DR density tended to form monocyte colonies in agar culture, whereas the low DR density fraction was enriched for granulocyte colony-forming cells. Inhibition by PGE was greatest in the high DR+ fraction and was largely restricted to monocyte progenitor cells. Inhibition of CFU-GM by PGE was less in CML than in NBM, but this decreased inhibition correlated with a significantly lower number of monocyte-CFU in CML. These data suggest that high HLA-DR antigen density may select for normal progenitor cells that are committed to monocyte differentiation and are, therefore, more likely to be inhibited by PGE. The relative deficit of monocyte progenitor cells in CML may partially explain the phenomenon of PGE resistance in this disease.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aglietta M., Piacibello W., Gavosto F. Insensitivity of chronic myeloid leukemia cells to inhibition of growth by prostaglandin E1. Cancer Res. 1980 Jul;40(7):2507–2511. [PubMed] [Google Scholar]
- Aglietta M., Piacibello W., Gavosto F. Prostaglandins and myelopoiesis: effect of prostaglandin E1 on normal and chronic myeloid leukemia colony forming cells (CFU-GM) subpopulations. Cell Biol Int Rep. 1981 Sep;5(9):836–836. doi: 10.1016/0309-1651(81)90196-x. [DOI] [PubMed] [Google Scholar]
- Bach F. H., van Rood J. J. The major histocompatibility complex--genetics and biology. (First of three parts). N Engl J Med. 1976 Oct 7;295(15):806–813. doi: 10.1056/NEJM197610072951504. [DOI] [PubMed] [Google Scholar]
- Benacerraf B. Role of MHC gene products in immune regulation. Science. 1981 Jun 12;212(4500):1229–1238. doi: 10.1126/science.6165083. [DOI] [PubMed] [Google Scholar]
- Breard J., Fuks A., Friedman S. M., Schlossman S. F., Chess L. The role of p23,30-bearing human macrophages in antigen-induced T lymphocyte responses. Cell Immunol. 1979 Jun;45(1):108–119. doi: 10.1016/0008-8749(79)90366-6. [DOI] [PubMed] [Google Scholar]
- Broxmeyer H. E. Relationship of cell-cycle expression of Ia-like antigenic determinants on normal and leukemia human granulocyte-macrophage progenitor cells to regulation in vitro by acidic isoferritins. J Clin Invest. 1982 Mar;69(3):632–642. doi: 10.1172/JCI110490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cannistra S. A., Daley J. F., Larcom P., Griffin J. D. Expression of Ia antigens on myeloid progenitor cells in chronic myeloid leukemia: direct analysis using partially purified colony-forming cells. Blood. 1985 Feb;65(2):414–422. [PubMed] [Google Scholar]
- Feighery C., Stastny P. HLA-D region-associated determinants serve as targets for human cell-mediated lysis. J Exp Med. 1979 Feb 1;149(2):485–494. doi: 10.1084/jem.149.2.485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fitchen J. H., Le Fèvre C., Ferrone S., Cline M. J. Expression of Ia-like and HLA-A,B antigens on human multipotential hematopoietic progenitor cells. Blood. 1982 Jan;59(1):188–190. [PubMed] [Google Scholar]
- Gentile P., Byer D., Pelus L. M. In vivo modulation of murine myelopoiesis following intravenous administration of prostaglandin E2. Blood. 1983 Nov;62(5):1100–1107. [PubMed] [Google Scholar]
- Griffin J. D., Beveridge R. P., Schlossman S. F. Isolation of myeloid progenitor cells from peripheral blood of chronic myelogenous leukemia patients. Blood. 1982 Jul;60(1):30–37. [PubMed] [Google Scholar]
- Griffin J. D., Hercend T., Beveridge R., Schlossman S. F. Characterization of an antigen expressed by human natural killer cells. J Immunol. 1983 Jun;130(6):2947–2951. [PubMed] [Google Scholar]
- Griffin J. D., Ritz J., Nadler L. M., Schlossman S. F. Expression of myeloid differentiation antigens on normal and malignant myeloid cells. J Clin Invest. 1981 Oct;68(4):932–941. doi: 10.1172/JCI110348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Janossy G., Francis G. E., Capellaro D., Goldstone A. H., Greaves M. F. Cell sorter analysis of leukaemia-associated antigens on human myeloid precursors. Nature. 1978 Nov 9;276(5684):176–178. doi: 10.1038/276176a0. [DOI] [PubMed] [Google Scholar]
- Kubota K., Mizoguchi H., Miura Y., Suda T., Takaku F. A new technique for the cytochemical examination of human hemopoietic cells grown in agar gel. Exp Hematol. 1980 Mar;8(3):339–344. [PubMed] [Google Scholar]
- Meuer S. C., Hussey R. E., Fabbi M., Fox D., Acuto O., Fitzgerald K. A., Hodgdon J. C., Protentis J. P., Schlossman S. F., Reinherz E. L. An alternative pathway of T-cell activation: a functional role for the 50 kd T11 sheep erythrocyte receptor protein. Cell. 1984 Apr;36(4):897–906. doi: 10.1016/0092-8674(84)90039-4. [DOI] [PubMed] [Google Scholar]
- Pelus L. M., Broxmeyer H. E., Clarkson B. D., Moore M. A. Abnormal responsiveness of granulocyte-macrophage committed colony-forming cells from patients with chronic myeloid leukemia to inhibition by prostaglandin E1. Cancer Res. 1980 Jul;40(7):2512–2515. [PubMed] [Google Scholar]
- Pelus L. M., Broxmeyer H. E., Kurland J. I., Moore M. A. Regulation of macrophage and granulocyte proliferation. Specificities of prostaglandin E and lactoferrin. J Exp Med. 1979 Aug 1;150(2):277–292. doi: 10.1084/jem.150.2.277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pelus L. M., Gold E., Saletan S., Coleman M. Restoration of responsiveness of chronic myeloid leukemia granulocyte-macrophage colony-forming cells to growth regulation in vitro following preincubation with prostaglandin E. Blood. 1983 Jul;62(1):158–165. [PubMed] [Google Scholar]
- Pelus L. M., Saletan S., Silver R. T., Moore M. A. Expression of Ia-antigens on normal and chronic myeloid leukemic human granulocyte-macrophage colony-forming cells (CFU-GM) is associated with the regulation of cell proliferation by prostaglandin E. Blood. 1982 Feb;59(2):284–292. [PubMed] [Google Scholar]
- Reinherz E. L., Meuer S., Fitzgerald K. A., Hussey R. E., Levine H., Schlossman S. F. Antigen recognition by human T lymphocytes is linked to surface expression of the T3 molecular complex. Cell. 1982 Oct;30(3):735–743. doi: 10.1016/0092-8674(82)90278-1. [DOI] [PubMed] [Google Scholar]
- Robinson J., Sieff C., Delia D., Edwards P. A., Greaves M. Expression of cell-surface HLA-DR, HLA-ABC and glycophorin during erythroid differentiation. Nature. 1981 Jan 1;289(5793):68–71. doi: 10.1038/289068a0. [DOI] [PubMed] [Google Scholar]
- Schlossman S. F., Chess L., Humphreys R. E., Strominger J. L. Distribution of Ia-like molecules on the surface of normal and leukemic human cells. Proc Natl Acad Sci U S A. 1976 Apr;73(4):1288–1292. doi: 10.1073/pnas.73.4.1288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stashenko P., Nadler L. M., Hardy R., Schlossman S. F. Characterization of a human B lymphocyte-specific antigen. J Immunol. 1980 Oct;125(4):1678–1685. [PubMed] [Google Scholar]
- Taetle R., Li-en S. Further studies on mechanisms of abnormal prostaglandin response by chronic myelogenous leukaemia granulocyte/macrophage progenitors. Leuk Res. 1984;8(5):833–842. doi: 10.1016/0145-2126(84)90104-8. [DOI] [PubMed] [Google Scholar]
- Todd R. F., 3rd, Meuer S. C., Romain P. L., Schlossman S. F. A monoclonal antibody that blocks class II histocompatibility-related immune interactions. Hum Immunol. 1984 May;10(1):23–40. doi: 10.1016/0198-8859(84)90083-1. [DOI] [PubMed] [Google Scholar]
- Todd R. F., 3rd, Nadler L. M., Schlossman S. F. Antigens on human monocytes identified by monoclonal antibodies. J Immunol. 1981 Apr;126(4):1435–1442. [PubMed] [Google Scholar]
- Williams N. Preferential inhibition of murine macrophage colony formation by prostaglandin E. Blood. 1979 Jun;53(6):1089–1094. [PubMed] [Google Scholar]
- Winchester R. J., Meyers P. A., Broxmeyer H. E., Wang C. Y., Moore M. A., Kunkel H. G. Inhibition of human erythropoietic colony formation in culture by treatment with Ia antisera. J Exp Med. 1978 Aug 1;148(2):613–618. doi: 10.1084/jem.148.2.613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Winchester R. J., Ross G. D., Jarowski C. I., Wang C. Y., Halper J., Broxmeyer H. E. Expression of Ia-like antigen molecules on human granulocytes during early phases of differentiation. Proc Natl Acad Sci U S A. 1977 Sep;74(9):4012–4016. doi: 10.1073/pnas.74.9.4012. [DOI] [PMC free article] [PubMed] [Google Scholar]



