Abstract
This is the protocol for a review and there is no abstract. The objectives are as follows:
To compare the effectiveness and harmful effects of interventions, which target the epidermal growth factor receptor, in the treatment of ovarian cancer.
BACKGROUND
Description of the condition
Each year, world-wide, over 200,000 women are diagnosed with ovarian cancer and nearly 125,000 die, corresponding to an annual incidence of 6.6 cases per 100,000 women, an annual mortality rate of 4.0 deaths per 100,000 and a cumulative lifetime risk of 0.5% (GLOBOCAN 2002). In terms of incidence, it is the sixth most common cancer and it is the seventh most common cause of cancer death in women. The onset is often insidious; the symptoms are vague and may mimic other conditions. This may lead to a delay in diagnosis, and currently three-quarters of women with ovarian cancer are diagnosed when the disease has spread throughout the abdomen (stage III or IV) (Shepherd 1989) when the 5 year survival is 20 to 30% (Jemal 2008). Epithelial ovarian cancer, which arises from the surface of the ovary, accounts for 90% of all ovarian cancers and typically presents in post-menopausal women, with a peak incidence when women are in their early sixties, although it does occur in younger women, often associated with genetic predispositions (Quinn 2001).
Description of the intervention
Management of advanced ovarian cancer consists of debulking surgery, and platinum-based chemotherapy, with or without the addition of a taxane (Morrison 2007;Stewart 1999) and a recent randomised controlled trial (RCT) found that there was no difference in survival, if surgery were performed before or after the first three cycles of chemotherapy (Vergote 2008). However, in women presenting with advanced disease, there has been little change to the five-year survival for stage III to IV disease over the past 20 to 30 years (Engel 2002). Despite good initial responses to platinum agents and taxanes, most women have disease relapse, require further treatment with chemotherapy, and eventually develop resistance to conventional chemotherapeutic agents.
Conventional chemotherapeutic agents have activity on all rapidly dividing cells, hence the common side effects such as hair loss, bone marrow suppression and mucositis. Increasing knowledge of the genetic basis for cancer has lead to the development of novel reagents, which target cancer-specific pathways. It is hoped that these reagents will spare normal cells and reduce the toxic side effects of chemotherapy, in addition to having an enhanced therapeutic effect.
How the intervention might work
Cancer cells, just like normal cells, can respond to external stimulation via growth factor receptors. These pathways are often mutated in cancers and are therefore a potential target to control cancer cell growth.
Epidermal growth factor receptors and ovarian cancer
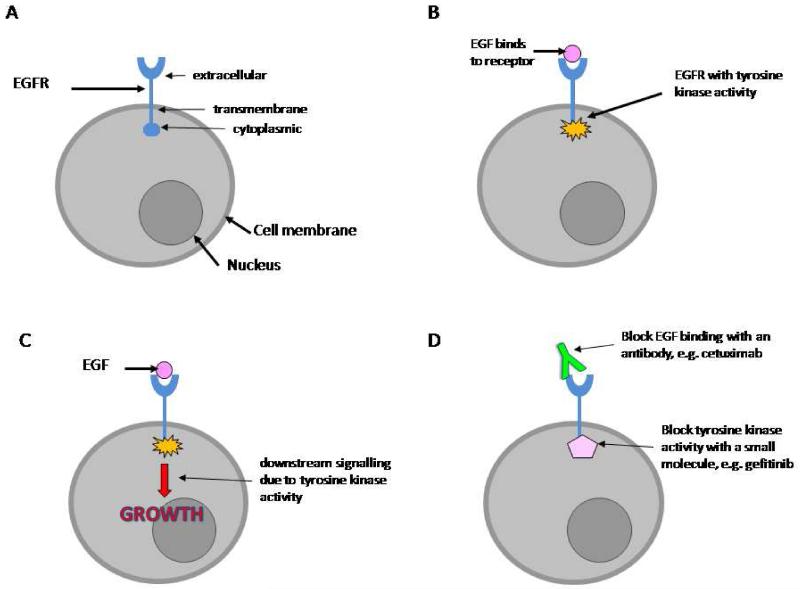
The epidermal growth factor receptor (EGFR or Erb1) is a cell surface molecule, which is normally involved in controlling cell growth. The EGFR is a tyrosine kinase enzyme that is made up of an extra-cellular ligand-binding domain, a cell membrane-spanning region, and an intracellular tyrosine kinase domain (Figure 1A). Following binding of its ligand, epidermal growth factor (EGF), the EGFR is activated; EGFR tyrosine kinase activity phosphorylates tyrosine residues on the EGFR and other proteins (Figure 1B), causing their activation and precipitating a sequence of down-stream events that lead to increased cell growth (Figure 1C). EGFR was first implicated in cancer aetiology when it was discovered that an oncogenic retrovirus encoded a mutated version of EGFR (Downward 1984). Abnormal EGFR activation has been demonstrated in ovarian cancer, is associated with a poorer prognosis (Nicholson 2001), and can happen through a variety of mechanisms. EGFR mutation occurs in some ovarian cancers (Moscatello 1995); the most common EGFR mutation is in the extra-cellular region and has been shown to result in EGF-independent activation (Ekstrand 1992). Over-expression of EGFR is common in many cancers (Bartlett 1996; Slamon 1989). EGFR activity can also be stimulated by increased production of EGF by the tumour cells (Bandera 2003). EGFR is central to the promotion of cell growth and has a role in the development of cancer. Preventing EGFR activity is therefore an attractive target for novel therapeutic agents. Anti-EGFR agents have been developed to prevent either the extra-cellular EGF binding, or inhibit the tyrosine kinase activity (Figure 1D). EGFR is a member of a family of similar molecules, called the epidermal growth factor receptor family. This family also includes Her2/neu, Erb3 and Erb4.
Figure 1.
(A) The EGFR is a transmembrane protein. (B) Following binding to its ligand, EGF, the EGFR is stimulated and develops tyrosine kinase activity. (C) Tyrosine kinase activity sets of a sequence of downstream events that lead to stimulation of cell growth. (D) EGFR activity can be blocked by antibodies, which prevent EGF binding to the receptor, or using chemicals, which inhibit the tyrosine kinase enzyme activity.
EGFR tyrosine kinase inhibitors
A number of small-molecule inhibitors of the EGFR tyrosine-kinase are in clinical trials for ovarian cancer, including gefitinib and erlotinib.
Gefitinib (Iressa/ZD1839) is a small molecule, which specifically inhibits EGFR tyrosine kinase activity (Moulder 2001). Pre-clinical studies have shown anti-tumour activity, and the orally active agent was well tolerated in a phase I clinical trial in a range of tumour types, including ovarian (Baselga 2002). A phase II study of gefitinib demonstrated poor response rates in women with platinum-resistant ovarian cancer who had not had the EGFR-status of their ovarian cancer tested. However, there was a 9% response rate in patients with EGFR-positive tumours, highlighting the need for selecting patients likely to benefit from treatment (Schilder 2005). Although response rates were modest, the treatment was well tolerated with rash and diarrhoea the main toxicities. Erlotinib (Tarceva/OSI-774) is another EGFR tyrosine kinase inhibitor, which has been through phase I and II clinical trials. Results from a recent phase II trial suggest that Erlotinib may have activity in ovarian carcinoma (Gordon 2005). Erlotinib was given to thirty-four patients with EGFR-positive recurrent, refractory, chemotherapy-resistant ovarian cancer;although responses were modest, the treatment was well tolerated. Further phase II and phase III trials for erlotinib are ongoing (NCT00263822 2005;NCT00520013 2007).
Neither gefitinib nor erlotinib has yet been licensed for use in ovarian cancer, outside of clinical trials. However, both have shown promise in other types of cancer: gefitinib has been licensed by the Food and Drug Administration in the USA for use in certain types of non-small cell lung cancer (NSCLC), and erlotinib has been licensed by both the FDA and the European Medicines Agency (EMEA) for use in some types of NSCLC and pancreatic cancer.
Antibodies against EGFR
It is also possible to block the EGFR pathway by means of specific antibodies.
Monoclonal antibodies are antibodies that have a specific target pattern to which they bind. Monoclonal antibodies have been developed against the extra-cellular portion of EGFR (Figure 1D). These antibodies are able to reduce EGFR activity, either by directly blocking EGF binding, or by causing the EGFR to be taken into the cell and degraded, so reducing the number of receptors available for stimulation at the cell surface. The monoclonal antibodies developed for clinical trials are humanized. This means that in addition to binding to the EGFR, they have a portion of the antibody, which is identical to a normal human antibody, so that it can stimulate the patient’s own immune system. When the antibody binds to the EGFR on a cell, it labels the cancer cell, so that it is recognized as foreign and is then destroyed by cells of the patient’s own immune system. HER2/neu is another member of the EGFR family and over-expression is related to poor outcome in breast cancer. A monoclonal antibody, Trastuzumab (Herceptin), has been developed that binds to HER2/neu (Baselga 2001;Cooley 1999). In women with recurrent ovarian cancer treated with Herceptin, a 7.3% response rate was demonstrated, with no significant toxicity (Bookman 2003).
IMC-C225 (Cetuximab/Erbitux) is a humanized mouse monoclonal antibody against EGFR (HER1/ErbB1) which has shown activity in combination with topotecan (a conventional chemotherapeutic agent) in pre-clinical studies (Ciardiello 1999;Ciardiello 2001; Goldstein 1995). Cetuximab binds to the EGFR and blocks EGF binding, thereby preventing down-stream signalling and growth stimulation, in addition to antibody-directed cell killing by the immune system.
Why it is important to do this review
Novel types of treatment strategies, which work in different ways to conventional chemotherapeutic agents, have been developed. It is therefore important to establish whether the addition of these new drugs to conventional chemotherapy regimens gives added benefit, in terms of survival, and if so, at what cost, in terms of additional harmful effects. In addition, since these compounds may be less toxic compared to conventional chemotherapy agents, it may be possible to give these newer treatments to patients who are not currently taking chemotherapy (so called maintenance treatment), to reduce the chance of, or delay, the recurrence of their ovarian cancer.
OBJECTIVES
To compare the effectiveness and harmful effects of interventions, which target the epidermal growth factor receptor, in the treatment of ovarian cancer.
METHODS
Criteria for considering studies for this review
Types of studies
RCTs
Types of participants
Adult women with histologically proven ovarian cancer. Women with other concurrent malignancies will be excluded.
Types of interventions
anti-EGFR agents (tyrosine kinase inhibitors and/or monoclonal antibodies) plus conventional chemotherapy versus conventional chemotherapy
anti-EGFR agents (tyrosine kinase inhibitors and/or monoclonal antibodies) versus no treatment
Types of outcome measures
Primary outcomes
Overall survival (OS)
Secondary outcomes
Progression-free survival (PFS)
Quality of life (QOL), measured by a validated scale
- Toxicity. Grades of toxicity will be extracted and grouped (CTEP 2006) as follows:
- haematological (leucopenia, anaemia, thrombocytopenia, neutropenia, haemorrhage)
- gastrointestinal (nausea, vomiting, anorexia, diarrhoea, liver, proctitis)
- genitourinary
- skin (stomatitis, mucositis, alopecia, allergy)
- neurological (peripheral and central)
- other side effects not categorised above
Search methods for identification of studies
Papers in all languages will be sought and translations carried out if necessary.
Electronic searches
See: Cochrane Gynaecological Cancer Group methods used in reviews.
The following electronic databases will be searched:
The Cochrane Gynaecological Cancer Collaborative Review Group’s Trial Register
Cochrane Central Register of Controlled Trials (CENTRAL)
MEDLINE
EMBASE
The MEDLINE search strategy is presented in Appendix 1.
For databases other than MEDLINE, the search strategy will be adapted accordingly. Databases will be searched from 1990 until 2009. These novel agents have been developed recently and so searches before 1990 would not be relevant.
All relevant articles found will be identified on PubMed and using the ‘related articles’ feature, a further search will be carried out for newly published articles.
Searching other resources
Physicians Data Query, www.controlled-trials.com/rct, www.clinicaltrials.gov, www.cancer.gov/clinicaltrials and the National Research Register (NRR) will be searched for ongoing trials. Details of ongoing or unpublished trials will also be sought from the FDA (Food and Drug Administration, the regulatory body for medicines within the USA, www.fda.gov and EMEA (European Medicines Agency, the drug regulatory body for within Europe,www.emea.europa.eu, and from pharmaceutical company sources. The main investigators of the relevant ongoing trials will be contacted for further information, as will be the major co-operative trials groups active in this area.
The reference lists of all included trials will be searched for further relevant trials.
Correspondence
Authors of relevant trials will be contacted to ask if they know of further data which may or may not have been published.
Data collection and analysis
Selection of studies
All titles and abstracts retrieved by electronic searching will be downloaded to the reference management database Endnote, duplicates will be removed and the remaining references will be examined by two review authors (KG, KH) independently. Those studies which clearly do not meet the inclusion criteria will be excluded and copies of the full text of potentially relevant references will be obtained. The eligibility of retrieved papers will be assessed independently by two review authors (KG, KH). Disagreements will be resolved by discussion between the two review authors and if necessary by a third review author (JM or SN). Reasons for exclusion will be documented.
Data extraction and management
For included studies, data will be abstracted as follows:
Author, year of publication and journal citation (including language)
Country
Setting
Inclusion and exclusion criteria
Study design, methodology
-
Study population
-
○
Total number enrolled
-
○
Patient characteristics
-
○
Age
-
○
Co-morbidities
-
○
Previous treatment
-
○
Total study duration
Total number of intervention groups
-
Ovarian cancer details at diagnosis
-
○
FIGO stage
-
○
Histological cell type
-
○
Tumour grade
-
○
Extent of disease
-
○
-
Intervention details
-
○
Type of EGFR inhibitor
-
○
Dose
-
○
Duration of treatment
-
○
Consolidation treatment or treatment of active disease
-
○
-
Comparison details
-
○
Type of control: conventional chemotherapy or no treatment
-
○
Dose (if appropriate)
-
○
Duration (if appropriate)
-
○
Deviations from protocol
Risk of bias in study (see below)
Duration of follow-up
-
Outcomes: OS, PFS, QOL, toxicity;
-
○
For each outcome: Outcome definition (with diagnostic criteria if relevant);
-
○
Unit of measurement (if relevant);
-
○
For scales: upper and lower limits, and whether high or low score is good
-
○
Results: Number of participants allocated to each intervention group;
-
○
For each outcome of interest: Sample size; Missing participants.
-
○
Data on outcomes will be extracted as below:
For time to event (OS and PFS) data, we will extract the log of the hazard ratio [log(HR)] and its standard error from trial reports; if these are not reported, we will attempt to estimate them from other reported statistics using the methods of Parmar 1998.
For dichotomous outcomes (e.g. toxicity or deaths if it was not possible to use a HR), we will extract the number of patients in each treatment arm who experienced the outcome of interest and the number of patients assessed at endpoint, in order to estimate a relative risk (RR).
For continuous outcomes (e.g. QOL measures), we will extract the final value and standard deviation of the outcome of interest and the number of patients assessed at endpoint in each treatment arm at the end of follow-up, in order to estimate the mean difference (if trials measured outcomes on the same scale) or standardised mean differences (if trials measured outcomes on different scales) between treatment arms and its standard error.
Both unadjusted and adjusted statistics will be extracted, if reported. Where adjusted results are extracted, the variables that were adjusted for will be recorded.
Where possible, all data extracted will be those relevant to an intention-to-treat analysis, in which participants are analysed in groups to which they were assigned.
The time points at which outcomes were collected and reported will be noted.
Data will be abstracted independently by two review authors (KH, KG) onto a data abstraction form specially designed for the review. Differences between review authors will be resolved by discussion or by appeal to a third review author (JM or SN) if necessary.
Assessment of risk of bias in included studies
The risk of bias in included RCTs will be assessed using the Cochrane Collaboration’s tool (Cochrane 2008). This will include assessment of:
sequence generation
allocation concealment
blinding of patients, treatment providers and outcome assessors
-
incomplete outcome data: We will record the proportion of participants whose outcomes were not reported at the end of the study; we will note if loss to follow-up was not reported. We will code a satisfactory level of loss to follow-up for each outcome as:
-
○
Yes, if fewer than 20% of patients were lost to follow-up and reasons for loss to follow-up were similar in both treatment arms
-
○
No, if more than 20% of patients were lost to follow-up or reasons for loss to follow-up differed between treatment arms
-
○
Unclear if loss to follow-up was not reported
-
○
selective reporting of outcomes
other possible sources of bias
The risk of bias tool will be applied independently by two review authors (KH, KG) and differences resolved by discussion or by appeal to a third review author (JM or SN). Results will be presented in both a risk of bias graph and a risk of bias summary. Results of meta-analyses will be interpreted in light of the findings with respect to risk of bias.
Measures of treatment effect
We will use the following measures of the effect of treatment:
For time to event data, we will use the HR, if possible.
For dichotomous outcomes, we will use the RR.
For continuous outcomes, we will use the mean difference between treatment arms if all trials measured the outcome on the same scale, otherwise standardised mean differences will be used.
If adjusted results are available, they will be preferred; otherwise unadjusted results will be used.
Dealing with missing data
We will not impute missing outcome data; if only imputed outcome data are reported, we will contact trial authors to request data on the outcomes only among participants who were assessed.
Assessment of heterogeneity
Heterogeneity between studies will be assessed by visual inspection of forest plots, by estimation of the percentage heterogeneity between trials which cannot be ascribed to sampling variation (Higgins 2003), and by a formal statistical test of the significance of the heterogeneity (Deeks 2001) and, if possible, by sub-group analyses Subgroup analysis and investigation of heterogeneity. If there is evidence of substantial heterogeneity, the possible reasons for this will be investigated and reported.
Assessment of reporting biases
Funnel plots corresponding to meta-analysis of the primary outcome will be examined to assess the potential for small study effects. When there is evidence of small-study effects, publication bias will be considered as only one of a number of possible explanations. If these plots suggest that treatment effects may not be sampled from a symmetric distribution, as assumed by the random effects model, sensitivity analyses will be performed using fixed effects models.
Data synthesis
If sufficient, clinically similar studies are available, their results will be pooled in meta-analyses.
The generic inverse variance facility of RevMan 5 will be used for meta-analyses of HRs and for any meta-analyses that include adjusted results. Random effects models with inverse variance weighting will be used for all meta-analyses (DerSimonian 1986). If any trials have multiple treatment groups, the control group will be divided between the treatment groups and comparisons between each treatment group and a split control group will be treated as independent comparisons.
If possible, studies making different comparisons will be synthesised using the methods of Bucher 1997.
Subgroup analysis and investigation of heterogeneity
As we expect to find few trials, we do not plan any sub-group analyses. However, factors such as type of intervention (e.g. use as early stage consolidation therapy in chemo-sensitive cancers or use in late stage chemo-resistant cancers) and stage of disease will be considered in interpretation of any heterogeneity.
Sensitivity analysis
Sensitivity analyses will be performed excluding (i) studies at high risk of bias, (ii) unadjusted results.
ACKNOWLEDGEMENTS
We thank Chris Williams for clinical and editorial advice, Jane Hayes for advice on the search strategy and Gail Quinn and Clare Jess for their contribution to the editorial process. We thank the referees for their many helpful suggestions.
SOURCES OF SUPPORT
Internal sources
No sources of support supplied
External sources
NIHR CCRCD, UK. JM is a Walport Clinical Lecturer, 50% academic component is funded by NIHR CCRCD
Macmillan Cancer Supoort, UK.JM is a subspecialist trainee in gynaecological oncology. This 50% clinical post is funded by a grant from Macmillan Cancer Support.
Department of Health, UK. NHS Cochrane Collaboration Programme Grant Scheme CPG-506
Appendix 1. MEDLINE search strategy
Medline RCT search filter:
-
1
randomized controlled trial.pt.
-
2
controlled clinical trial.pt.
-
3
randomized.ab.
-
4
placebo.ab.
-
5
drug therapy.fs.
-
6
randomly.ab.
-
7
trial.ab.
-
8
groups.ab.
-
9
1 or 2 or 3 or 4 or 5 or 6 or 7 or 8
-
10
animals.sh. not (humans.sh. and animals.sh.)
-
11
9 not 10
Medline search for patient population:
-
12
Ovar$
-
13
Cancer$ or carcinoma$ or neoplasm$ or tumor$ or tumour$ or malignan$
-
14
12 and 13
-
15
Exp ovarian neoplasms/
-
16
14 or 15
Medline search for intervention:
-
17
Epidermal growth factor receptor.mp.
-
18
(EGFR adj5 inhibit$).mp.
-
19
exp Antibodies, Monoclonal/
-
20
monoclonal antibodies.mp.
-
21
Trastuzumab or herceptin
-
22
Cetuximab or IMC-C225 or erbitux
-
23
EMD 72000 or matuzumab
-
24
Panitumumab or ABX-EGF or vectibix
-
25
Pertuzumab or rhumab 2C4 or omnitarg
-
26
exp Protein-Tyrosine Kinases/
-
27
(tyrosine kinase adj5 inhibit$).mp.
-
28
gefitinib or ZD1839 or iressa
-
29
erlotinib or OSI-774 or tarceva
-
30
lapatinib or gw 572016 or tykerb
-
31
canertinib or CI-1033
-
32
EKB-569
-
33
PKI-166
-
34
BMS 599626
-
35
Vandetanib or rinn or zactima or ZD6474
-
36
17 or 18 or 19 or 20 or 21 or 22 or 23 or 24 or 25 or 26 or 27 or 28 or 29 or 30 or 31 or 32 or 33 or 34 or 35
-
37
11 and 16 and 36
Key
pt = publication type
ab = abstract
fs = floating sub-heading
mp = title, original title, abstract, name of substance word, subject heading word
sh = medical subject heading
WHAT’S NEW
| 21 July 2009 | New citation required and minor changes | Author citation amended |
HISTORY
Protocol first published: Issue 3, 2009
Footnotes
DECLARATIONS OF INTEREST
None
Additional references
- Bandera 2003 .Bandera CA, Tsui HW, Mok SC, Tsui FW. Expression of cytokines and receptors in normal, immortalized, and malignant ovarian epithelial cell lines. Anticancer Research. 2003;23(4):3151–7. [PubMed] [Google Scholar]
- Bartlett 1996 .Bartlett JM, Langdon SP, Simpson BJ, Stewart M, Katsaros D, Sismondi P, et al. The prognostic value of epidermal growth factor receptor mRNA expression in primary ovarian cancer. British Journal of Cancer. 1996;73(3):301–6. doi: 10.1038/bjc.1996.53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baselga 2001 .Baselga J, Albanell J, Molina MA, Arribas J. Mechanism of action of trastuzumab and scientific update. Seminars in Oncology. 2001;28(5 Suppl 16):4–11. doi: 10.1016/s0093-7754(01)90276-3. [DOI] [PubMed] [Google Scholar]
- Baselga 2002 .Baselga J, Rischin D, Ranson M, Calvert H, Raymond E, Kieback DG, et al. Phase I safety, pharmacokinetic, and pharmacodynamic trial of ZD1839, a selective oral epidermal growth factor receptor tyrosine kinase inhibitor, in patients with five selected solid tumor types. Journal of Clinical Oncology. 2002;20(21):4292–302. doi: 10.1200/JCO.2002.03.100. [DOI] [PubMed] [Google Scholar]
- Bookman 2003 .Bookman MA, Darcy KM, Clarke-Pearson D, Boothby RA, Horowitz IR. Evaluation of monoclonal humanized anti-HER2 antibody, trastuzumab, in patients with recurrent or refractory ovarian or primary peritoneal carcinoma with overexpression of HER2: a phase II trial of the Gynecologic Oncology Group. Journal of Clinical Oncology. 2003;21(2):283–90. doi: 10.1200/JCO.2003.10.104. [DOI] [PubMed] [Google Scholar]
- Bucher 1997 .Bucher HC, Guyatt GH, Griffith LE, Walter SD. The Results of Direct and Indirect Treatment Comparisons in Meta-Analysis of Randomized Controlled Trials. Journal of Clinical Epidemiology. 1997;50(6):683–91. doi: 10.1016/s0895-4356(97)00049-8. [DOI] [PubMed] [Google Scholar]
- Ciardiello 1999 .Ciardiello F, Bianco R, Damiano V, De Lorenzo S, Pepe S, De Placido S, et al. Antitumor activity of sequential treatment with topotecan and anti-epidermal growth factor receptor monoclonal antibody C225. Clinical Cancer Research. 1999;5(4):909–16. [PubMed] [Google Scholar]
- Ciardiello 2001 .Ciardiello F, Caputo R, Bianco R, Damiano V, Fontanini G, Cuccato S, et al. Inhibition of growth factor production and angiogenesis in human cancer cells by ZD1839 (Iressa), a selective epidermal growth factor receptor tyrosine kinase inhibitor. Clinical Cancer Research. 2001;7(5):1459–65. [PubMed] [Google Scholar]
- Cochrane 2008 .Higgins JPT, Green S, editors. Cochrane Handbook for Systematic Reviews of Interventions Version 5.0.0 [updated September 2008] The Cochrane Collaboration; 2008. Available from www.cochrane-handbook.org. [Google Scholar]
- Cooley 1999 .Cooley S, Burns LJ, Repka T, Miller JS. Natural killer cell cytotoxicity of breast cancer targets is enhanced by two distinct mechanisms of antibody-dependent cellular cytotoxicity against LFA-3 and HER2/neu. Experimental Hematology. 1999;27(10):1533–41. doi: 10.1016/s0301-472x(99)00089-2. [DOI] [PubMed] [Google Scholar]
- CTEP 2006 .Cancer Therapy Evaluation Program (CTEP) Common Terminology Criteria for Adverse Events, Version 3.0. DCTD, NCI, NIH, DHHS; 2006. http://ctep.cancer.gov [Google Scholar]
- Deeks 2001 .Deeks JJ, Altman DG, Bradburn MJ. Statistical methodsfor examining heterogeneity and combining results from several studies in meta-analysis. In: Egger M, Davey Smith G, Altman DG, editors. Systematic Reviews in Health Care: Meta-Analysis in Context. 2nd Edition BMJ Publication group; London: 2001. [Google Scholar]
- DerSimonian 1986 .DerSimonian R, Laird N. Meta-analysis in clinical trials. Controlled Clinical Trials. 1986;7(3):177–88. doi: 10.1016/0197-2456(86)90046-2. [DOI] [PubMed] [Google Scholar]
- Downward 1984 .Downward J, Yarden Y, Mayes E, Scrace G, Totty N, Stockwell P, et al. Close similarity of epidermal growth factor receptor and v-erb-B oncogene protein sequences. Nature. 1984;307(5951):521–7. doi: 10.1038/307521a0. [DOI] [PubMed] [Google Scholar]
- Ekstrand 1992 .Ekstrand AJ, Sugawa N, James CD, Collins VP. Amplified and rearranged epidermal growth factor receptor genes in human glioblastomas reveal deletions of sequences encoding portions of the N-and/or C-terminal tails. Proceedings of the National Academy of Science U S A. 1992;89(10):4309–13. doi: 10.1073/pnas.89.10.4309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Engel 2002 .Engel J, Eckel R, Schubert-Fritschle G, Kerr J, Kuhn W, Diebold J, et al. Moderate progress for ovarian cancer in the last 20 years: prolongation of survival, but no improvement in the cure rate. 2002;38(18):2435–45. doi: 10.1016/s0959-8049(02)00495-1. http://info.cancerresearchuk.org/cancerstats/types/ovary/survival/ [DOI] [PubMed] [Google Scholar]
- GLOBOCAN 2002 .Ferlay J, Bray F, Pisani P, Parkin DM. IARC CancerBase. 5. IARCPress; Lyon: 2004. GLOBOCAN 2002: Cancer Incidence, Mortality and Prevalence Worldwide. version 2.0. [Google Scholar]
- Goldstein 1995 .Goldstein NI, Prewett M, Zuklys K, Rockwell P, Mendelsohn J. Biological efficacy of a chimeric antibody to the epidermal growth factor receptor in a human tumor xenograft model. Clinical Cancer Research. 1995;1(11):1311–8. [PubMed] [Google Scholar]
- Gordon 2005 .Gordon AN, Finkler N, Edwards RP, Garcia AA, Crozier M, Irwin DH, et al. Efficacy and safety of erlotinib HCl, an epidermal growth factor receptor (HER1/EGFR) tyrosine kinase inhibitor, in patients with advanced ovarian carcinoma: results from a phase II multicenter study. International Journal Gynecological Cancer. 2005;15(5):785–92. doi: 10.1111/j.1525-1438.2005.00137.x. [DOI] [PubMed] [Google Scholar]
- Higgins 2003 .Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003;327(7414):557–60. doi: 10.1136/bmj.327.7414.557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jemal 2008 .Jemal A, Siegel R, Ward E, Hao Y, Xu J, Murray T, et al. Cancer Statistics. A Cancer Journal for Clinicians. 2008;58(2):71–96. doi: 10.3322/CA.2007.0010. [DOI] [PubMed] [Google Scholar]
- Morrison 2007 .Morrison J, Swanton A, Collins S, Kehoe S. Chemotherapy versus surgery for initial treatment in advanced ovarian epithelial cancer. Cochrane Database of Systematic Reviews. 2007;(4) doi: 10.1002/14651858.CD005343.pub2. DOI: 10.1002/14651858.CD005343. [DOI] [PubMed] [Google Scholar]
- Moscatello 1995 .Moscatello DK, Holgado-Madruga M, Godwin AK, Ramirez G, Gunn G, Zoltick PW, et al. Frequent expression of a mutant epidermal growth factor receptor in multiple human tumors. Cancer Research. 1995;55(23):5536–9. [PubMed] [Google Scholar]
- Moulder 2001 .Moulder SL, Yakes FM, Muthuswamy SK, Bianco R, Simpson JF, Arteaga CL. Epidermal growth factor receptor (HER1) tyrosine kinase inhibitor ZD1839 (Iressa) inhibits HER2/neu (erbB2)-over expressing breast cancer cells in vitro and in vivo. Cancer Research. 2001;61(24):8887–95. [PubMed] [Google Scholar]
- NCT00263822 2005 . NCT00263822 . A Randomized, Multicenter, Phase III Study of Erlotinib Versus Observation in Patients With no Evidence of Disease Progression After First Line, Platinum-Based Chemotherapy For High-Risk Ovarian Epithelial, Primary Peritoneal, or Fallopian Tube Cancer. Dec 7, 2005. MetaRegister of Controlled Trials Protocol Registered. [Google Scholar]
- NCT00520013 2007 . NCT00520013 . A Randomized Phase II Trial of Avastin (A) or Avastin and Erlotinib (AE) as First Line Consolidation Chemotherapy After Carboplatin, Paclitaxel and Avastin (CTA) Induction Therapy for Newly Diagnosed Advanced Ovarian, Fallopian Tube and Primary Peritoneal Cancer & Papillary Serous Mullerian Tumors. Aug 22, 2007. MetaRegister of Controlled Trials Protocol Registered. [Google Scholar]
- Nicholson 2001 .Nicholson RI, Gee JM, Harper ME. EGFR and cancer prognosis. European Journal of Cancer. 2001;37(Suppl 4):S9–15. doi: 10.1016/s0959-8049(01)00231-3. [DOI] [PubMed] [Google Scholar]
- Parmar 1998 .Parmar MK, Torri V, Stewart L. Extracting summary statistics to perform meta-analyses of the published literature for survival endpoints. Statistics in Medicine. 1998;17(24):2815–34. doi: 10.1002/(sici)1097-0258(19981230)17:24<2815::aid-sim110>3.0.co;2-8. [DOI] [PubMed] [Google Scholar]
- Quinn 2001 .Quinn M, Babb B, Brock A, Jones J. In: Cancer Trends in England and Wales. Statistics OfN., editor. The Stationery Office; London: 2001. [Google Scholar]
- Schilder 2005 .Schilder RJ, Sill MW, Chen X, Darcy KM, Decesare SL, Lewandowski G, et al. Phase II study of gefitinib in patients with relapsed or persistent ovarian or primary peritoneal carcinoma and evaluation of epidermal growth factor receptor mutations and immunohistochemical expression: a Gynecologic Oncology Group Study. Clinical Cancer Research. 2005;11(15):5539–48. doi: 10.1158/1078-0432.CCR-05-0462. [DOI] [PubMed] [Google Scholar]
- Shepherd 1989 .Shepherd JH. Revised FIGO staging for gynaecological cancer. British Journal of Obstetrics and Gynaecology. 1989;96(8):889–92. doi: 10.1111/j.1471-0528.1989.tb03341.x. [DOI] [PubMed] [Google Scholar]
- Slamon 1989 .Slamon DJ, Godolphin W, Jones LA, Holt JA, Wong SG, Keith DE, et al. Studies of the HER-2/neu proto-oncogene in human breast and ovarian cancer. Science. 1989;244(4905):707–12. doi: 10.1126/science.2470152. [DOI] [PubMed] [Google Scholar]
- Stewart 1999 .Stewart L, Advanced Ovarian Cancer Trialists Group Chemotherapy for advanced ovarian cancer. Cochrane Database of Systematic Reviews. 1999;(1) doi: 10.1002/14651858.CD001418. Art. No.: CD001418 DOI: 10.1002/14651858.CD001418. [DOI] [PubMed] [Google Scholar]
- Vergote 2008 .Vergote I, Tropé CG, Amant F, Kristensen GB, Sardi JE, Ehlen T, et al. EORTC-GCG/NCIC-CTG Randomised trial comparing primary debulking surgery with neoadjuvant chemotherapy in stage IIIC-IV ovarian, fallopian tube and peritoneal cancer (OVCA); Proceedings of the 12th Biennial Meeting of the International Gynecologic Cancer Society - IGCS; Bangkok. 2008.Sep, [Google Scholar]
- * Indicates the major publication for the study