Abstract
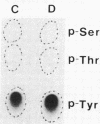
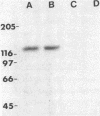
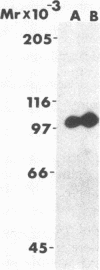
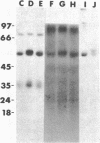
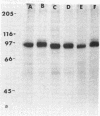
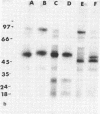
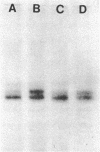
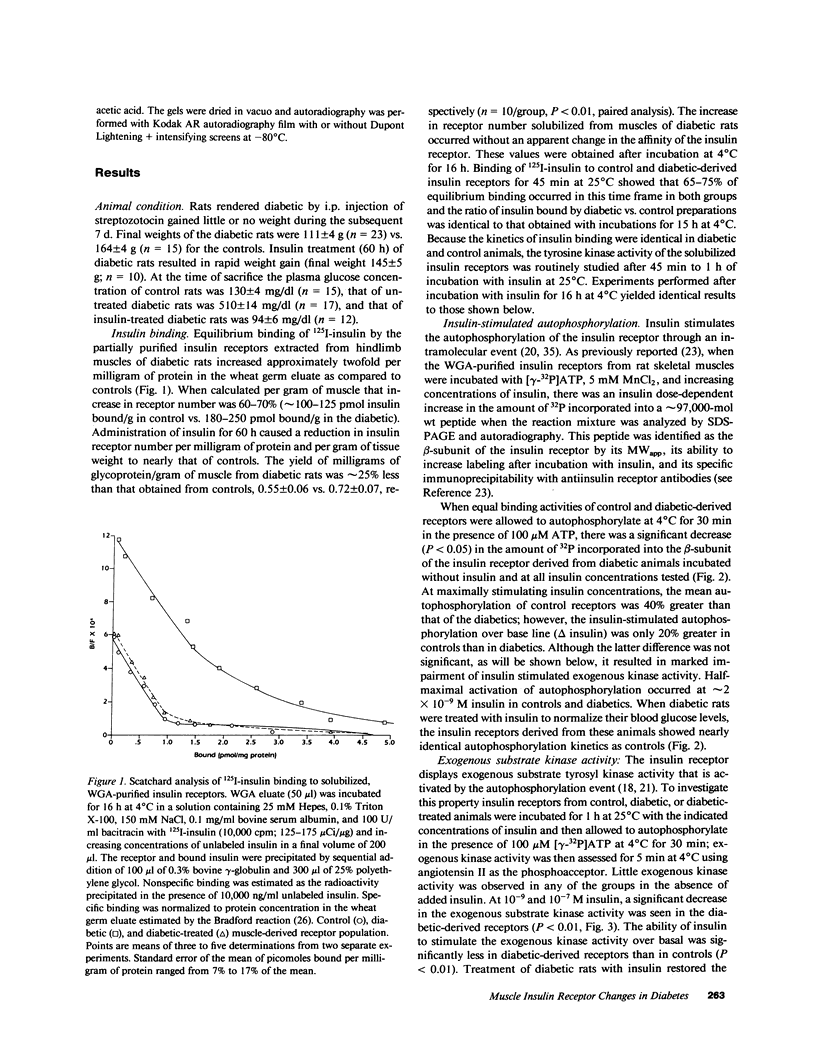
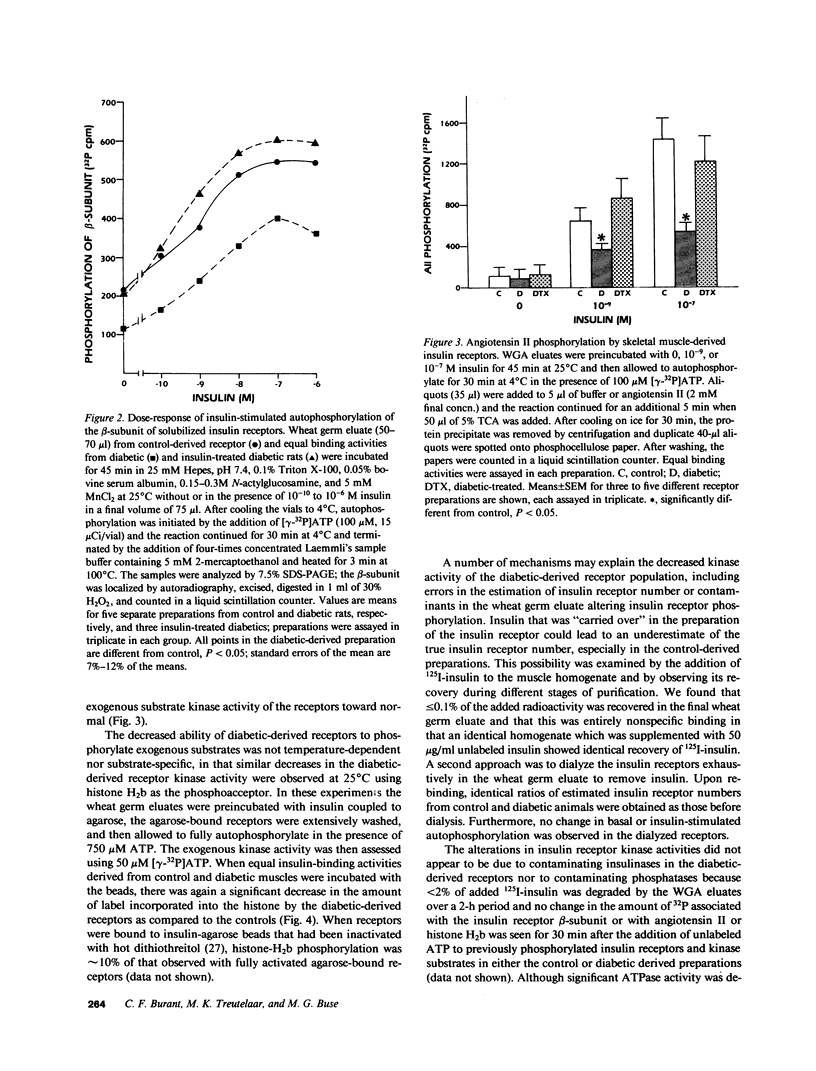
The effect of diabetes on the structure and function of insulin receptors was studied in rats 7 d after streptozotocin injection, using solubilized, partially purified receptors from rat hindlimb muscles. Diabetes increased the number of insulin receptors per gram of muscle 60-70% without apparent change in insulin binding affinity. Incubation of receptors at 4 degrees C with [gamma-32P]ATP and insulin resulted in dose-dependent autophosphorylation of the beta-subunit on tyrosine residues; receptors from diabetic rats showed decreased base-line phosphorylation, as well as a decrease in autophosphorylation at maximally stimulating insulin concentrations. These receptors also showed diminished exogenous substrate kinase activity using histone H2b and angiotensin II as phosphoacceptors. The electrophoretic mobility (sodium dodecyl sulfate-polyacrylamide gel electrophoresis) of a subpopulation of beta-subunits derived from diabetics was slightly decreased; differences in electrophoretic mobility between control- and diabetic-derived beta-subunits were enhanced by generating fragments by partial Staphylococcus aureus V8 protease digestion. Endoglycosidase-H or neuraminidase treatment increased the electrophoretic mobility of beta-subunits in both groups, but only neuraminidase appeared to decrease or abolish differences in electrophoretic mobility between controls and diabetics, suggesting that excess sialilation may account, in part, for the altered mobility of diabetic derived beta-subunits. All structural and functional alterations in insulin receptors were prevented by treating diabetic rats with insulin for 60 h. Peripheral insulin resistance associated with insulinopenic diabetes may be related to modifications in insulin receptor structure, resulting in impaired signal transmission.
Full text
PDF








Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Amatruda J. M., Roncone A. M. Normal hepatic insulin receptor autophosphorylation in nonketotic diabetes mellitus. Biochem Biophys Res Commun. 1985 May 31;129(1):163–170. doi: 10.1016/0006-291x(85)91417-2. [DOI] [PubMed] [Google Scholar]
- Avruch J., Nemenoff R. A., Blackshear P. J., Pierce M. W., Osathanondh R. Insulin-stimulated tyrosine phosphorylation of the insulin receptor in detergent extracts of human placental membranes. Comparison to epidermal growth factor-stimulated phosphorylation. J Biol Chem. 1982 Dec 25;257(24):15162–15166. [PubMed] [Google Scholar]
- Beemon K., Hunter T. Characterization of Rous sarcoma virus src gene products synthesized in vitro. J Virol. 1978 Nov;28(2):551–566. doi: 10.1128/jvi.28.2.551-566.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- Burant C. F., Treutelaar M. K., Landreth G. E., Buse M. G. Phosphorylation of insulin receptors solubilized from rat skeletal muscle. Diabetes. 1984 Jul;33(7):704–708. doi: 10.2337/diab.33.7.704. [DOI] [PubMed] [Google Scholar]
- Carlin C. R., Knowles B. B. Biosynthesis of the epidermal growth factor receptor in human epidermoid carcinoma-derived A431 cells. J Biol Chem. 1984 Jun 25;259(12):7902–7908. [PubMed] [Google Scholar]
- Cooper J. A., Sefton B. M., Hunter T. Detection and quantification of phosphotyrosine in proteins. Methods Enzymol. 1983;99:387–402. doi: 10.1016/0076-6879(83)99075-4. [DOI] [PubMed] [Google Scholar]
- DeFronzo R. A., Hendler R., Simonson D. Insulin resistance is a prominent feature of insulin-dependent diabetes. Diabetes. 1982 Sep;31(9):795–801. doi: 10.2337/diab.31.9.795. [DOI] [PubMed] [Google Scholar]
- Foss M. C., Vlachokosta F. V., Cunningham L. N., Aoki T. T. Restoration of glucose homeostasis in insulin-dependent diabetic subjects. An inducible process. Diabetes. 1982 Jan;31(1):46–52. doi: 10.2337/diab.31.1.46. [DOI] [PubMed] [Google Scholar]
- Fujita-Yamaguchi Y., Sato Y., Kathuria S. Removal of sialic acids from the purified insulin receptor results in enhanced insulin-binding and kinase activities. Biochem Biophys Res Commun. 1985 Jun 28;129(3):739–745. doi: 10.1016/0006-291x(85)91954-0. [DOI] [PubMed] [Google Scholar]
- Galbraith R. A., Wise C., Buse M. G. Insulin binding and response in erythroblastic leukemic cells. Diabetes. 1980 Jul;29(7):571–578. doi: 10.2337/diab.29.7.571. [DOI] [PubMed] [Google Scholar]
- Grigorescu F., Flier J. S., Kahn C. R. Defect in insulin receptor phosphorylation in erythrocytes and fibroblasts associated with severe insulin resistance. J Biol Chem. 1984 Dec 25;259(24):15003–15006. [PubMed] [Google Scholar]
- Grunberger G., Zick Y., Gorden P. Defect in phosphorylation of insulin receptors in cells from an insulin-resistant patient with normal insulin binding. Science. 1984 Mar 2;223(4639):932–934. doi: 10.1126/science.6141638. [DOI] [PubMed] [Google Scholar]
- Haft D. E. Studies of the metabolism of isolated livers of normal and alloxan-diabetic rats perfused with insulin. Diabetes. 1968 May;17(5):244–250. doi: 10.2337/diab.17.5.244. [DOI] [PubMed] [Google Scholar]
- Harano Y., Ohgaku S., Hidaka H., Haneda K., Kikkawa R., Shigeta Y., Abe H. Glucose, insulin and somatostatin infusion for the determination of insulin sensitivity. J Clin Endocrinol Metab. 1977 Nov;45(5):1124–1127. doi: 10.1210/jcem-45-5-1124. [DOI] [PubMed] [Google Scholar]
- Hedo J. A., Harrison L. C., Roth J. Binding of insulin receptors to lectins: evidence for common carbohydrate determinants on several membrane receptors. Biochemistry. 1981 Jun 9;20(12):3385–3393. doi: 10.1021/bi00515a013. [DOI] [PubMed] [Google Scholar]
- Hedo J. A., Kasuga M., Van Obberghen E., Roth J., Kahn C. R. Direct demonstration of glycosylation of insulin receptor subunits by biosynthetic and external labeling: evidence for heterogeneity. Proc Natl Acad Sci U S A. 1981 Aug;78(8):4791–4795. doi: 10.1073/pnas.78.8.4791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herzberg V. L., Grigorescu F., Edge A. S., Spiro R. G., Kahn C. R. Characterization of insulin receptor carbohydrate by comparison of chemical and enzymatic deglycosylation. Biochem Biophys Res Commun. 1985 Jun 28;129(3):789–796. doi: 10.1016/0006-291x(85)91961-8. [DOI] [PubMed] [Google Scholar]
- KIPNIS D. M., CORI C. F. Studies of tissue permeability. V. The penetration and phosphorylation of 2-deoxyglucose in the rat diaphragm. J Biol Chem. 1959 Jan;234(1):171–177. [PubMed] [Google Scholar]
- Kadowaki T., Kasuga M., Akanuma Y., Ezaki O., Takaku F. Decreased autophosphorylation of the insulin receptor-kinase in streptozotocin-diabetic rats. J Biol Chem. 1984 Nov 25;259(22):14208–14216. [PubMed] [Google Scholar]
- Kasuga M., Fujita-Yamaguchi Y., Blithe D. L., Kahn C. R. Tyrosine-specific protein kinase activity is associated with the purified insulin receptor. Proc Natl Acad Sci U S A. 1983 Apr;80(8):2137–2141. doi: 10.1073/pnas.80.8.2137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kasuga M., Karlsson F. A., Kahn C. R. Insulin stimulates the phosphorylation of the 95,000-dalton subunit of its own receptor. Science. 1982 Jan 8;215(4529):185–187. doi: 10.1126/science.7031900. [DOI] [PubMed] [Google Scholar]
- Kasuga M., Zick Y., Blith D. L., Karlsson F. A., Häring H. U., Kahn C. R. Insulin stimulation of phosphorylation of the beta subunit of the insulin receptor. Formation of both phosphoserine and phosphotyrosine. J Biol Chem. 1982 Sep 10;257(17):9891–9894. [PubMed] [Google Scholar]
- Kobayashi M., Olefsky J. M. Effects of streptozotocin-induced diabetes on insulin binding, glucose transport, and intracellular glucose metabolism in isolated rat adipocytes. Diabetes. 1979 Feb;28(2):87–95. doi: 10.2337/diab.28.2.87. [DOI] [PubMed] [Google Scholar]
- Le Marchand-Brustel Y., Freychet P. Effect of fasting and streptozotocin diabetes on insulin binding and action in the isolated mouse soleus muscle. J Clin Invest. 1979 Nov;64(5):1505–1515. doi: 10.1172/JCI109609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Le Marchand-Brustel Y., Grémeaux T., Ballotti R., Van Obberghen E. Insulin receptor tyrosine kinase is defective in skeletal muscle of insulin-resistant obese mice. Nature. 1985 Jun 20;315(6021):676–679. doi: 10.1038/315676a0. [DOI] [PubMed] [Google Scholar]
- Le Marchand Y., Loten E. G., Assimacopoulos-Jeannet F., Forgue M. E., Freychet P., Jeanrenaud B. Effect of fasting and streptozotocin in the obese-hyperglycemic (ob/ob) mouse. Apparent lack of a direct relationship between insulin binding and insulin effects. Diabetes. 1977 Jun;26(6):582–590. doi: 10.2337/diab.26.6.582. [DOI] [PubMed] [Google Scholar]
- MORGAN H. E., CADENAS E., REGEN D. M., PARK C. R. Regulation of glucose uptake in muscle. II. Rate-limiting steps and effects of insulin and anoxia in heart muscle from diabetic rats. J Biol Chem. 1961 Feb;236:262–268. [PubMed] [Google Scholar]
- Martin F. I., Stocks A. E. Insulin sensitivity and 131-I insulin metabolism in juvenile-type diabetics. Australas Ann Med. 1967 Nov;16(4):289–296. doi: 10.1111/imj.1967.16.4.289. [DOI] [PubMed] [Google Scholar]
- Mayfield R. K., Sullivan F. M., Colwell J. A., Wohltmann H. J. Predicting insulin requirements for a portable insulin pump using the Biostator. Evidence for reversible insulin resistance in poorly controlled type I diabetics. Diabetes. 1983 Oct;32(10):908–914. doi: 10.2337/diab.32.10.908. [DOI] [PubMed] [Google Scholar]
- Okamoto M., Kuzuya H., Imura H. Increased affinity of insulin receptor on hepatocytes from streptozotocin-induced diabetic rats. Endocrinol Jpn. 1984 Jun;31(3):235–243. doi: 10.1507/endocrj1954.31.235. [DOI] [PubMed] [Google Scholar]
- Pilch P. F., Czech M. P. Interaction of cross-linking agents with the insulin effector system of isolated fat cells. Covalent linkage of 125I-insulin to a plasma membrane receptor protein of 140,000 daltons. J Biol Chem. 1979 May 10;254(9):3375–3381. [PubMed] [Google Scholar]
- Reaven G. M., Sageman W. S., Swenson R. S. Development of insulin resistance in normal dogs following alloxan-induced insulin deficiency. Diabetologia. 1977 Sep;13(5):459–462. doi: 10.1007/BF01234496. [DOI] [PubMed] [Google Scholar]
- Ronnett G. V., Knutson V. P., Kohanski R. A., Simpson T. L., Lane M. D. Role of glycosylation in the processing of newly translated insulin proreceptor in 3T3-L1 adipocytes. J Biol Chem. 1984 Apr 10;259(7):4566–4575. [PubMed] [Google Scholar]
- Rosen O. M., Herrera R., Olowe Y., Petruzzelli L. M., Cobb M. H. Phosphorylation activates the insulin receptor tyrosine protein kinase. Proc Natl Acad Sci U S A. 1983 Jun;80(11):3237–3240. doi: 10.1073/pnas.80.11.3237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salzman A., Wan C. F., Rubin C. S. Biogenesis, transit, and functional properties of the insulin proreceptor and modified insulin receptors in 3T3-L1 adipocytes. Use of monensin to probe proreceptor cleavage and generate altered receptor subunits. Biochemistry. 1984 Dec 18;23(26):6555–6565. doi: 10.1021/bi00321a043. [DOI] [PubMed] [Google Scholar]
- Sherwin R. S., Tamborlane W. V., Genel M., Felig P. Treatment of juvenile-onset diabetes by subcutaneous infusion of insulin with a portable pump. Diabetes Care. 1980 Mar-Apr;3(2):301–308. doi: 10.2337/diacare.3.2.301. [DOI] [PubMed] [Google Scholar]
- Taylor S. I., Hedo J. A., Underhill L. H., Kasuga M., Elders M. J., Roth J. Extreme insulin resistance in association with abnormally high binding affinity of insulin receptors from a patient with leprechaunism: evidence for a defect intrinsic to the receptor. J Clin Endocrinol Metab. 1982 Dec;55(6):1108–1113. doi: 10.1210/jcem-55-6-1108. [DOI] [PubMed] [Google Scholar]
- Tepperman H. M., DeWitt J., Tepperman J. The effects of streptozotocin diabetes on the activities of rat liver glycosyltransferases. Diabetes. 1983 May;32(5):412–415. doi: 10.2337/diab.32.5.412. [DOI] [PubMed] [Google Scholar]
- Van Obberghen E., Rossi B., Kowalski A., Gazzano H., Ponzio G. Receptor-mediated phosphorylation of the hepatic insulin receptor: evidence that the Mr 95,000 receptor subunit is its own kinase. Proc Natl Acad Sci U S A. 1983 Feb;80(4):945–949. doi: 10.1073/pnas.80.4.945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- White M. F., Haring H. U., Kasuga M., Kahn C. R. Kinetic properties and sites of autophosphorylation of the partially purified insulin receptor from hepatoma cells. J Biol Chem. 1984 Jan 10;259(1):255–264. [PubMed] [Google Scholar]
- Yu K. T., Czech M. P. Tyrosine phosphorylation of the insulin receptor beta subunit activates the receptor-associated tyrosine kinase activity. J Biol Chem. 1984 Apr 25;259(8):5277–5286. [PubMed] [Google Scholar]
- Zick Y., Whittaker J., Roth J. Insulin stimulated phosphorylation of its own receptor. Activation of a tyrosine-specific protein kinase that is tightly associated with the receptor. J Biol Chem. 1983 Mar 25;258(6):3431–3434. [PubMed] [Google Scholar]