Abstract
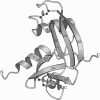
The capacity of angiogenin (Ang) to induce blood vessel growth is critically dependent on its ribonucleolytic activity. Crystallography and mutagenesis of human Ang have previously shown that its pyrimidine binding site is obstructed by Gln-117, implying that a conformational change is a key part of the mechanism of Ang action. The 1.5-A-resolution crystal structure of bovine Ang, in which glutamic acid is substituted for Gln-117, now confirms that a blocked active site is characteristic of these proteins. Indeed, the inactive conformation of bovine Ang is stabilized by a more extensive set of interactions than is that of human Ang. The three-dimensional structure of the putative receptor binding site is also well conserved in the two proteins. The Arg-Gly-Asp segment of this site in bovine Ang, which is replaced by Arg-Glu-Asn in human Ang, does not have a conformation typical of an integrin recognition site.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Acharya K. R., Shapiro R., Allen S. C., Riordan J. F., Vallee B. L. Crystal structure of human angiogenin reveals the structural basis for its functional divergence from ribonuclease. Proc Natl Acad Sci U S A. 1994 Apr 12;91(8):2915–2919. doi: 10.1073/pnas.91.8.2915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Acharya K. R., Subramanian V., Shapiro R., Riordan J. F., Vallee B. L. Crystallization and preliminary X-ray analysis of human angiogenin. J Mol Biol. 1992 Dec 20;228(4):1269–1270. doi: 10.1016/0022-2836(92)90333-f. [DOI] [PubMed] [Google Scholar]
- Artymiuk P. J., Blake C. C. Refinement of human lysozyme at 1.5 A resolution analysis of non-bonded and hydrogen-bond interactions. J Mol Biol. 1981 Nov 15;152(4):737–762. doi: 10.1016/0022-2836(81)90125-x. [DOI] [PubMed] [Google Scholar]
- Bicknell R., Vallee B. L. Angiogenin stimulates endothelial cell prostacyclin secretion by activation of phospholipase A2. Proc Natl Acad Sci U S A. 1989 Mar;86(5):1573–1577. doi: 10.1073/pnas.86.5.1573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bond M. D., Strydom D. J., Vallee B. L. Characterization and sequencing of rabbit, pig and mouse angiogenins: discernment of functionally important residues and regions. Biochim Biophys Acta. 1993 Mar 5;1162(1-2):177–186. doi: 10.1016/0167-4838(93)90145-h. [DOI] [PubMed] [Google Scholar]
- Bond M. D., Vallee B. L. Isolation of bovine angiogenin using a placental ribonuclease inhibitor binding assay. Biochemistry. 1988 Aug 23;27(17):6282–6287. doi: 10.1021/bi00417a013. [DOI] [PubMed] [Google Scholar]
- Borah B., Chen C. W., Egan W., Miller M., Wlodawer A., Cohen J. S. Nuclear magnetic resonance and neutron diffraction studies of the complex of ribonuclease A with uridine vanadate, a transition-state analogue. Biochemistry. 1985 Apr 9;24(8):2058–2067. doi: 10.1021/bi00329a038. [DOI] [PubMed] [Google Scholar]
- Curran T. P., Shapiro R., Riordan J. F. Alteration of the enzymatic specificity of human angiogenin by site-directed mutagenesis. Biochemistry. 1993 Mar 9;32(9):2307–2313. doi: 10.1021/bi00060a023. [DOI] [PubMed] [Google Scholar]
- Fett J. W., Olson K. A., Rybak S. M. A monoclonal antibody to human angiogenin. Inhibition of ribonucleolytic and angiogenic activities and localization of the antigenic epitope. Biochemistry. 1994 May 10;33(18):5421–5427. doi: 10.1021/bi00184a010. [DOI] [PubMed] [Google Scholar]
- Fett J. W., Strydom D. J., Lobb R. R., Alderman E. M., Bethune J. L., Riordan J. F., Vallee B. L. Isolation and characterization of angiogenin, an angiogenic protein from human carcinoma cells. Biochemistry. 1985 Sep 24;24(20):5480–5486. doi: 10.1021/bi00341a030. [DOI] [PubMed] [Google Scholar]
- Hallahan T. W., Shapiro R., Vallee B. L. Dual site model for the organogenic activity of angiogenin. Proc Natl Acad Sci U S A. 1991 Mar 15;88(6):2222–2226. doi: 10.1073/pnas.88.6.2222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harper J. W., Auld D. S., Riordan J. F., Vallee B. L. Enzymatically active angiogenin/ribonuclease A hybrids formed by peptide interchange. Biochemistry. 1988 Jan 12;27(1):219–226. doi: 10.1021/bi00401a033. [DOI] [PubMed] [Google Scholar]
- Harper J. W., Vallee B. L. A covalent angiogenin/ribonuclease hybrid with a fourth disulfide bond generated by regional mutagenesis. Biochemistry. 1989 Feb 21;28(4):1875–1884. doi: 10.1021/bi00430a067. [DOI] [PubMed] [Google Scholar]
- Hu G., Riordan J. F., Vallee B. L. Angiogenin promotes invasiveness of cultured endothelial cells by stimulation of cell-associated proteolytic activities. Proc Natl Acad Sci U S A. 1994 Dec 6;91(25):12096–12100. doi: 10.1073/pnas.91.25.12096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kabsch W., Sander C. Dictionary of protein secondary structure: pattern recognition of hydrogen-bonded and geometrical features. Biopolymers. 1983 Dec;22(12):2577–2637. doi: 10.1002/bip.360221211. [DOI] [PubMed] [Google Scholar]
- Lee F. S., Vallee B. L. Structure and action of mammalian ribonuclease (angiogenin) inhibitor. Prog Nucleic Acid Res Mol Biol. 1993;44:1–30. doi: 10.1016/s0079-6603(08)60215-9. [DOI] [PubMed] [Google Scholar]
- Logan D., Abu-Ghazaleh R., Blakemore W., Curry S., Jackson T., King A., Lea S., Lewis R., Newman J., Parry N. Structure of a major immunogenic site on foot-and-mouth disease virus. Nature. 1993 Apr 8;362(6420):566–568. doi: 10.1038/362566a0. [DOI] [PubMed] [Google Scholar]
- Maes P., Damart D., Rommens C., Montreuil J., Spik G., Tartar A. The complete amino acid sequence of bovine milk angiogenin. FEBS Lett. 1988 Dec 5;241(1-2):41–45. doi: 10.1016/0014-5793(88)81027-5. [DOI] [PubMed] [Google Scholar]
- Olson K. A., Fett J. W., French T. C., Key M. E., Vallee B. L. Angiogenin antagonists prevent tumor growth in vivo. Proc Natl Acad Sci U S A. 1995 Jan 17;92(2):442–446. doi: 10.1073/pnas.92.2.442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reisdorf C., Abergel D., Bontems F., Lallemand J. Y., Decottignies J. P., Spik G. Proton resonance assignments and secondary structure of bovine angiogenin. Eur J Biochem. 1994 Sep 15;224(3):811–822. doi: 10.1111/j.1432-1033.1994.00811.x. [DOI] [PubMed] [Google Scholar]
- Ruoslahti E., Pierschbacher M. D. New perspectives in cell adhesion: RGD and integrins. Science. 1987 Oct 23;238(4826):491–497. doi: 10.1126/science.2821619. [DOI] [PubMed] [Google Scholar]
- Russo N., Shapiro R., Acharya K. R., Riordan J. F., Vallee B. L. Role of glutamine-117 in the ribonucleolytic activity of human angiogenin. Proc Natl Acad Sci U S A. 1994 Apr 12;91(8):2920–2924. doi: 10.1073/pnas.91.8.2920. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shapiro R., Riordan J. F., Vallee B. L. Characteristic ribonucleolytic activity of human angiogenin. Biochemistry. 1986 Jun 17;25(12):3527–3532. doi: 10.1021/bi00360a008. [DOI] [PubMed] [Google Scholar]
- Shapiro R., Strydom D. J., Olson K. A., Vallee B. L. Isolation of angiogenin from normal human plasma. Biochemistry. 1987 Aug 11;26(16):5141–5146. doi: 10.1021/bi00390a037. [DOI] [PubMed] [Google Scholar]
- Shapiro R., Vallee B. L. Identification of functional arginines in human angiogenin by site-directed mutagenesis. Biochemistry. 1992 Dec 15;31(49):12477–12485. doi: 10.1021/bi00164a026. [DOI] [PubMed] [Google Scholar]
- Shapiro R., Vallee B. L. Identification of functional arginines in human angiogenin by site-directed mutagenesis. Biochemistry. 1992 Dec 15;31(49):12477–12485. doi: 10.1021/bi00164a026. [DOI] [PubMed] [Google Scholar]
- Shapiro R., Vallee B. L. Site-directed mutagenesis of histidine-13 and histidine-114 of human angiogenin. Alanine derivatives inhibit angiogenin-induced angiogenesis. Biochemistry. 1989 Sep 5;28(18):7401–7408. doi: 10.1021/bi00444a038. [DOI] [PubMed] [Google Scholar]
- Soncin F. Angiogenin supports endothelial and fibroblast cell adhesion. Proc Natl Acad Sci U S A. 1992 Mar 15;89(6):2232–2236. doi: 10.1073/pnas.89.6.2232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Strydom D. J., Fett J. W., Lobb R. R., Alderman E. M., Bethune J. L., Riordan J. F., Vallee B. L. Amino acid sequence of human tumor derived angiogenin. Biochemistry. 1985 Sep 24;24(20):5486–5494. doi: 10.1021/bi00341a031. [DOI] [PubMed] [Google Scholar]
- Stuart D. I., Levine M., Muirhead H., Stammers D. K. Crystal structure of cat muscle pyruvate kinase at a resolution of 2.6 A. J Mol Biol. 1979 Oct 15;134(1):109–142. doi: 10.1016/0022-2836(79)90416-9. [DOI] [PubMed] [Google Scholar]
- Tarragona-Fiol A., Eggelte H. J., Harbron S., Sanchez E., Taylorson C. J., Ward J. M., Rabin B. R. Identification by site-directed mutagenesis of amino acids in the B2 subsite of bovine pancreatic ribonuclease A. Protein Eng. 1993 Nov;6(8):901–906. doi: 10.1093/protein/6.8.901. [DOI] [PubMed] [Google Scholar]
- Wang B. C. Resolution of phase ambiguity in macromolecular crystallography. Methods Enzymol. 1985;115:90–112. doi: 10.1016/0076-6879(85)15009-3. [DOI] [PubMed] [Google Scholar]