Abstract
Airborne particulate matter (PM) exposure is a major environmental health concern and is linked to metabolic disorders, such as cardiovascular diseases (CVD) and diabetes, which are on the rise in the Kingdom of Saudi Arabia. This study investigated changes in mouse lung gene expression produced by administration of PM10 collected from Jeddah, Saudi Arabia. FVB/N mice were exposed to 100 µg PM10 or water by aspiration and euthanized 24 hr later. The bronchoalveolar lavage fluid (BALF) was collected and analyzed for neutrophil concentration and TNF-α and IL-6 levels. RNA was extracted from the lungs and whole transcript was analyzed using Affymetrix Mouse Gene 1.0 ST Array. Mice exposed to PM10 displayed an increase in neutrophil concentration and elevated TNF-α and IL-6 levels. Gene expression analysis revealed that mice exposed to PM10 displayed 202 genes that were significantly up-regulated and 40 genes that were significantly down-regulated. PM10 induced genes involved in inflammation, cholesterol and lipid metabolism, as well as atherosclerosis. This is the first study to demonstrate that Saudi Arabia PM10 increases in vivo expression of genes located in pathways associated with diseases involving metabolic syndrome and atherosclerosis.
Keywords: gene expression, metabolic diseases, lungs, cholesterol, particulate matter, mouse
Introduction
Airborne particulate matter (PM) exposures are a major environmental health concern. Epidemiological studies have linked air pollution with a number of detrimental outcomes such as, hospital admissions (Colucci et al. 2006; Vigotti et al. 2007), chronic obstructive pulmonary disease (Gan et al. 2013), lung cancer (Yanagi et al. 2012), asthma (Karakatsani et al. 2012) and cardiovascular disease (Beckerman et al. 2012; Brook et al. 2010; Chang et al. 2013; Chiu et al. 2013; Mazzoli-Rocha et al. 2010; Nishiwaki et al. 2012). PM has been found to be the major component of air pollution that has the most deleterious effects on human health (Colucci et al. 2006; Samet and Krewski 2007). PM exposure has been associated with premature mortality (Tagaris et al. 2010), decreased birth weight (Pedersen et al. 2013), and increased asthma morbidity (Delfino et al. 2014). PM may be composed of a number of constituents depending on the source, including organic matter, mineral oxides, liquid droplets, soil, dust, smoke, and gaseous chemicals (Araujo and Nel 2009; Simkhovich et al. 2008). PM may vary in size, chemical composition, and origin (Ghio et al. 2012). Coarse particulates (2.5– 10 µm) deposit in the nasopharyngeal region and the upper regions of the lung, while fine (≤ 2.5 µm) and ultrafine (≤1 µm) particulates may penetrate deep into the alveolar region (Ghio et al. 2012).
Several studies have reported on in vivo gene expression changes after particulate matter exposure. (Thomson et al. 2013) investigated the gene expression changes in kidney, heart, lungs, liver, and spleen in rats exposed to 5 or 50 mg/m3 urban particulate matter and found that mRNA profiles were similar across organs for many genes including redox/glucocorticoid-sensitive genes and inflammatory genes. An interesting study by (Motta et al. 2013) found that human subjects exposed to metal-rich PM displayed a unique expression of microRNAs (miRNAs) in their blood. The study found four differentially expressed PM- responsive miRNAs and eleven miRNA-mRNA pairs that regulate inflammatory gene expression.
Recently, (Khodeir et al. 2012) conducted a multi-week, multiple site sampling campaign to study the source apportionment and elemental composition of PM10 in Jeddah, the second largest city in Saudi Arabia. The major source factors for PM10 were soil re-suspensions, oil combustion, mixed industrial sources, traffic sources, and marine aerosols. Components of the PM10 from Jeddah have been characterized in (Khodeir et al. 2012).
There are many factors that indicate Jeddah, SA as a setting for high PM exposure. The city has stationary (power plant, airport, oil refinery, industries) and mobile (over 1.4 million cars) sources of air pollution. While an early study by (Nasralla 1983) concluded that concentrations of airborne particulates and other pollutants in Jeddah often exceeded air quality standards, a more recent study by (Elassouli et al. 2007) reported that PM10 in the city of Jeddah routinely exceeds the average hourly standard for PM10 established by the Presidency of Meteorology and Environment in Saudi Arabia, which is 80 µg/m3.
In previous studies, a 24 hr exposure of PM10 collected from Jeddah induced genes involved in NRF2-mediated response to oxidative stress in human bronchial epithelial cells (BEAS-2b) (Sun et al. 2012). (Huang et al. 2011) found that airway epithelial cells treated with an acute exposure of fine or ultrafine PM also displayed altered mRNA profiles involving genes in the NRF2-mediated oxidative stress response pathway. A 4-day exposure in BEAS-2b revealed that genes related to cholesterol and lipid synthesis pathways were also increased (Sun et al. 2012), thus prompting further investigation to assess whether such a metabolic response would be observed in vivo.
Metabolic diseases, such as cardiovascular disease and diabetes, are often associated with obesity and have become major health concerns in Saudi Arabia. A study by (Al-Othaimeen AI 2007) involving19,598 Saudi Arabian citizens found that 38% of males and 28% of females were overweight and a number of other studies highlighted the obesity problem in Saudi Arabia (Al-Malki et al. 2003; Madani et al. 2000). Given the previous in vitro findings that Saudi Arabia PM10 induces cholesterol and lipid metabolism genes, investigations were conducted to determine if PM10 would induce these genes in vivo in order to further evaluate the possibility that exposure to PM10 may contribute to the development of metabolic disorders.
Metabolic syndrome is a combination of risk factors, when occurring together, increase the risk of cardiovascular disease and diabetes. Many studies linked PM2.5 exposure with metabolic disorders (Brook et al. 2013; Brook et al. 2010); however, it is still unclear how ambient particulates mediate adverse health effects. While metabolic disorders are commonly associated with changes in the liver, various studies demonstrated that after a toxic insult, lungs and liver display similar changes in mRNA levels (Crespo et al. 1999; Flohe et al. 1999; Ghoshal et al. 2001; Rosen et al. 2007; Zhang et al. 1998).
Materials and Methods
Animals
Male FVB/N mice (11 weeks old, body weight 22–30 g) were obtained from Taconic Farms (German-town, NY) and housed in our AAALAC accredited housing facility in Tuxedo, NY. FVB/N mice respond well to perturbations of the airways and have been used in PM studies (Keith et al. 2004). Approval was given by NYU Institutional Animal Care and Use Committee (IACUC) for the use of animals in experimental studies. After a 16 day acclimation period, treatment mice (n=6) were exposed via aspiration to 100 µg PM10 (3.92 mg/kg) collected from Jeddah, Saudi Arabia and control mice (n=3) were exposed to an equivalent volume of distilled water. The PM2.5/PM10 ratio of the PM was 0.33. The dose of PM2.5 received by each mouse was 1.29 mg/kg. All procedures were conducted in compliance with New York University’s guidelines for ethical animal research. All procedures were conducted in compliance with New York University’s guidelines for ethical animal research. Details regarding the particle collection and extraction techniques, as well as, the components of PM10 have been previously described (Khodeir et al. 2012; Sun et al. 2012). PM10 was analyzed by XRF for the concentration of 27 elements. Re-suspended soil and oil combustion contributed 82% of the mass and mixed industrial sources, traffic sources, and marine aerosols were also present. The PM was heavily concentrated with silicon, calcium, sulfur, aluminum and iron. Some of the other metals present include nickel, vanadium, arsenic, lead, cadmium, manganese, titanium and magnesium.
Animals were anesthetized in a chamber containing 3 ml isoflurane. After 5 min, the animal was removed from the chamber and attached to a suspension slide with its tongue held down by a wire and its upper mouth held open by a rubber band. The appropriate volume of PM10 or distilled water was injected in the back of the animal’s mouth between breathes. Prior to exposure, PM10 was re-suspended by sonication for 10 min.
At 24 hr post-exposure, animals were euthanized with sodium pentobarbital (150–200 mg/kg) via an intraperitoneal injection. Animals were injected with ketamine (0.1 mg/ 10 g) 5–6 minutes prior to euthanasia. Blood was taken from the heart immediately following euthanasia. The trachea was cannulated and the lungs lavaged twice with 1.2 ml phosphate buffered saline without calcium and magnesium (PBS, Invitrogen, Carlsbad, CA). Bronchoaveolar lavage fluid (BALF) was stored on ice until analysis. Both lungs were extracted from the chest cavity and placed in a cryogenic tube and frozen in liquid nitrogen for future analysis of mRNA expression.
BALF Assays
For cell differentials, an aliquot of lavage fluid was prepared using a cytospin (Shandon, Southern Products, UK). An aliquot from each lavage sample (100 µl) was placed into the cytospin for 7 min. Slides were subsequently stained with Hemacolor® staining (EM Science, Gibbstown, NJ). Neutrophil and macrophage populations were enumerated by counting 100 total cells. The remaining lavage fluid was centrifuged at 400 g for 10 min and the collected supernatant was frozen at −20° C until further analysis. BALF supernatants were analyzed for tumor necrosis factor-alpha (TNF-α) and interleukin-6 (IL-6) by Enzyme-Linked Immunoassays (ELISA) using commercially available kits (R&D Systems, Minneapolis, MN). ELISA assays were run in duplicates. OD values were transferred to Microsoft Excel and a two- tailed t- test was performed. The coefficients of variation within each assay were 0.049 for TNF-α and 0.291 for IL-6. The amount of each cytokine was derived from standard curves specific to each cytokine.
RNA extraction and microarray hybridization
Total RNA was extracted from lungs of control and PM-exposed mice using Trizol (Invitrogen) and further purified using RNeasy Plus Micro Kit (Qiagen). One hundred ng of total RNA was used to synthesize double-stranded cDNA (dsDNA). cRNA was synthesized from dsDNA template, and subsequently used to produce sense single-stranded cDNA (ssDNA) with incorporated deoxyuridine triphosphate. The ssDNAs were fragmented, end-labeled, and hybridized to Affymetrix Mouse Gene 1.0 ST Array (Affymetrix). Hybridization and scanning of the arrays were performed using a standard procedure.
Microarray data analysis
Gene expression analyses were performed using R. Gene expression data were imported and normalized in batches using the Affymetrix package version 1.36.1. in R 2.15.1 GUI 1.42 Leopard build 64-bit and robust multichip average (RMA) (Bolstad et al. 2003; Gautier et al. 2004). Significance of gene expression changes between controls and PM10- treated mice were assessed using a gene-wise linear model approach with LIMMA 3.14.3, which utilizes an empirical Bayes approach to generate moderated t-statistics by taking into account the standard errors and estimated log-fold changes (Smythe et al. 2005). P-values were subjected to FDR correction for multiple hypothesis testing and adjusted p-values with p<0.05 were considered significant (Benjamini Y 1995). Gene network and pathway analyses were performed using Ingenuity Pathway Analysis software (http://www.ingenuity.com).
Real time PCR
Total RNA extracted from control and treated lung tissue was converted to single stranded cDNA using Superscrip® III (Invitrogen). Quantitative real-time PCR analysis was performed using SYBR green PCR system (Applied Biosystems) on ABI prism 7900HT system (Applied Biosystems). Relative gene expression levels were normalized to ACTB expression. All PCR reactions were performed in duplicate.
Results
Neutrophil, TNF- α and IL-6 Concentrations in BALF
During the acute phase of inflammation, neutrophils are one of the first responders that migrate to the inflammatory site and increased neutrophil concentration is a reliable indication that an inflammatory response has been elicited (Scapini et al. 2000). In order to measure the neutrophil concentration in bronchoalveolar lavage fluid (BALF) after PM10 exposure, FVB/N mice were treated via aspiration with either 100 µg PM10 or distilled water and BALF was collected 24 hr later and analyzed for neutrophil concentration (Table 1). The dose was generated from a previous experiment (Table 2). Our aim was to select a dose that would generate immune cell counts of 40–50 % neutrophils in the BALF. Mice treated with PM10 or water contained an average of 59.6 and 0.67% neutrophils/total immune cells in BALF, respectively. Other immune cells identified included macrophages, while eosinophils and basophils were not present. Mice treated with PM10 or water contained an average of 3.79 × 105 and 2.45 × 105 total immune cells/ ml, respectively.
Table 1.
Percent neutrophils and total immune cells present in aBALF.
| SAMPLE | % Neutrophils |
Total Immune Cells |
|---|---|---|
| Control | 0.67% | 2.45 × 105cells/ml |
| bPM10 | 59.6% | 3.79 × 105 cells/ml |
Bronchoalveolar lavage fluid
PM10- particulate matter collected from Jeddah, Saudi Arabia
Table 2.
| Dose | % Neutrophils | Total Immune Cells |
|---|---|---|
| 25 µg | 8% | 2.71 × 105cells/ ml |
| 50 µg | 14% | 2.65 × 105cells/ ml |
| c100 µg | 43% | 3.39 × 105cells/ ml |
| 250 µg | 100% | 5.05 × 105cells/ml |
| 500 µg | 99% | 5.6 × 105cells/ml |
Bronchoalveolar lavage fluid
PM10- particulate matter collected from Jeddah, Saudi Arabia
Dose chosen for acute exposure
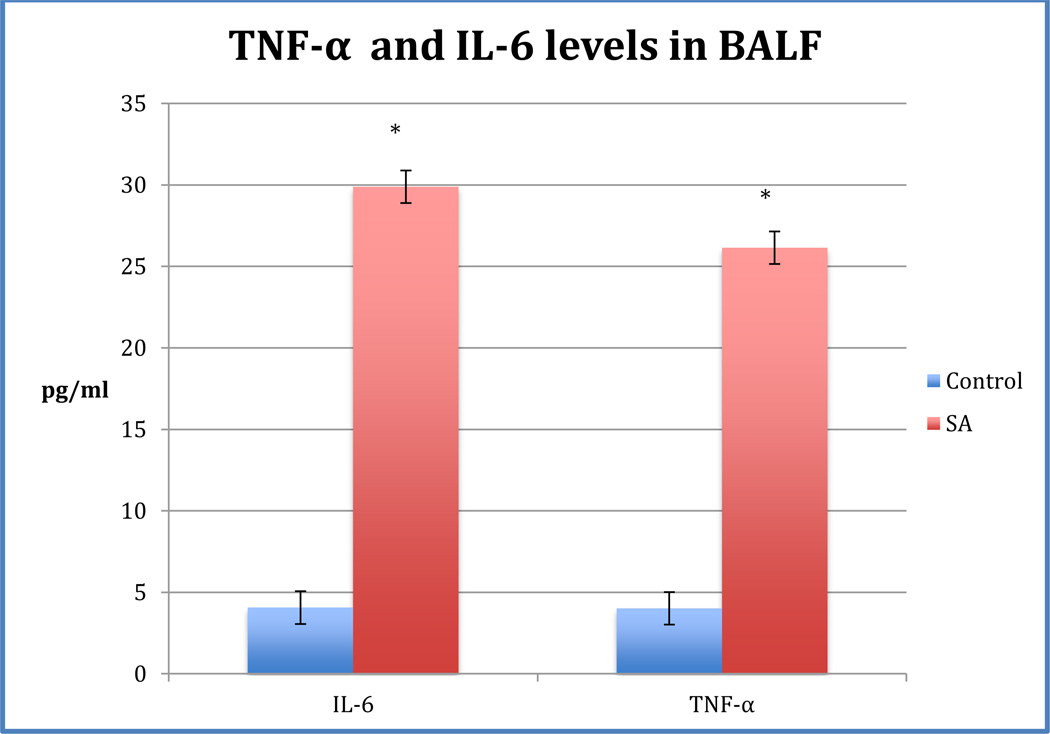
Tumor necrosis factor-alpha (TNF-α) and interleukin-6 (IL-6) are produced by lung epithelial cells, alveolar macrophages and other immune cells after an insult (Kaplanski et al. 2003; Rosenblum and Donato 1989). In order to determine the effects of PM10 on TNF- α and IL-6 levels, an ELISA was performed using BALF supernatant of PM10-exposed and control mice. TNF-α levels were higher in PM10-treated mice compared to control (Figure 1). The average TNF-α levels were 26.2 ± 7.0 pg/ml in PM10 and 4.01±1.9 pg/ml in control BALF, respectively. The TNF-α levels in the PM10 BALF were significantly increased compared to control. IL-6 levels were also significantly higher in PM10 BALF compared to control (Figure 1). Average IL-6 levels were 29.9 ± 9.9 pg/ml in PM10 and 4.06 ± 2.7 pg/ml in control BALF, respectively.
Fig. 1. TNF-α and IL-6 levels in BALF of mice treated with PM10.
Mice were exposed to 100 µg PM10 or distilled water.TNF-α. The average TNF-α levels were 26.2 ± 7.0 and 4.01±1.9 pg/ml in PM10 BALF and control BALF, respectively. TNF- α levels in PM10-treated mice were significantly higher than control mice (p< 0.0012).IL-6. Average IL-6 levels (p< 0.05) were 29.9 ± 9.9 and 4.06 ± 2.7 pg/ml in PM10 BALF and control BALF. IL-6 levels in PM10- treated mice were significantly higher than control mice (p< 0.05).
Gene expression profiles of PM10-exposed mice
PM is known to affect the expression of many genes involved in various molecular pathways that contribute to the inflammatory microenvironment as well as cholesterol and lipid metabolism (Huang et al. 2011; Kooter et al. 2005; Sun et al. 2012). Whole lung gene expression profiles of 4 mice exposed to PM10 and 3 control mice exposed to distilled water were examined. Whole transcript analysis using Affymetrix Mouse Gene 1.0 ST Array was performed and data analyzed using R and Ingenuity Pathway Analysis (IPA) software.
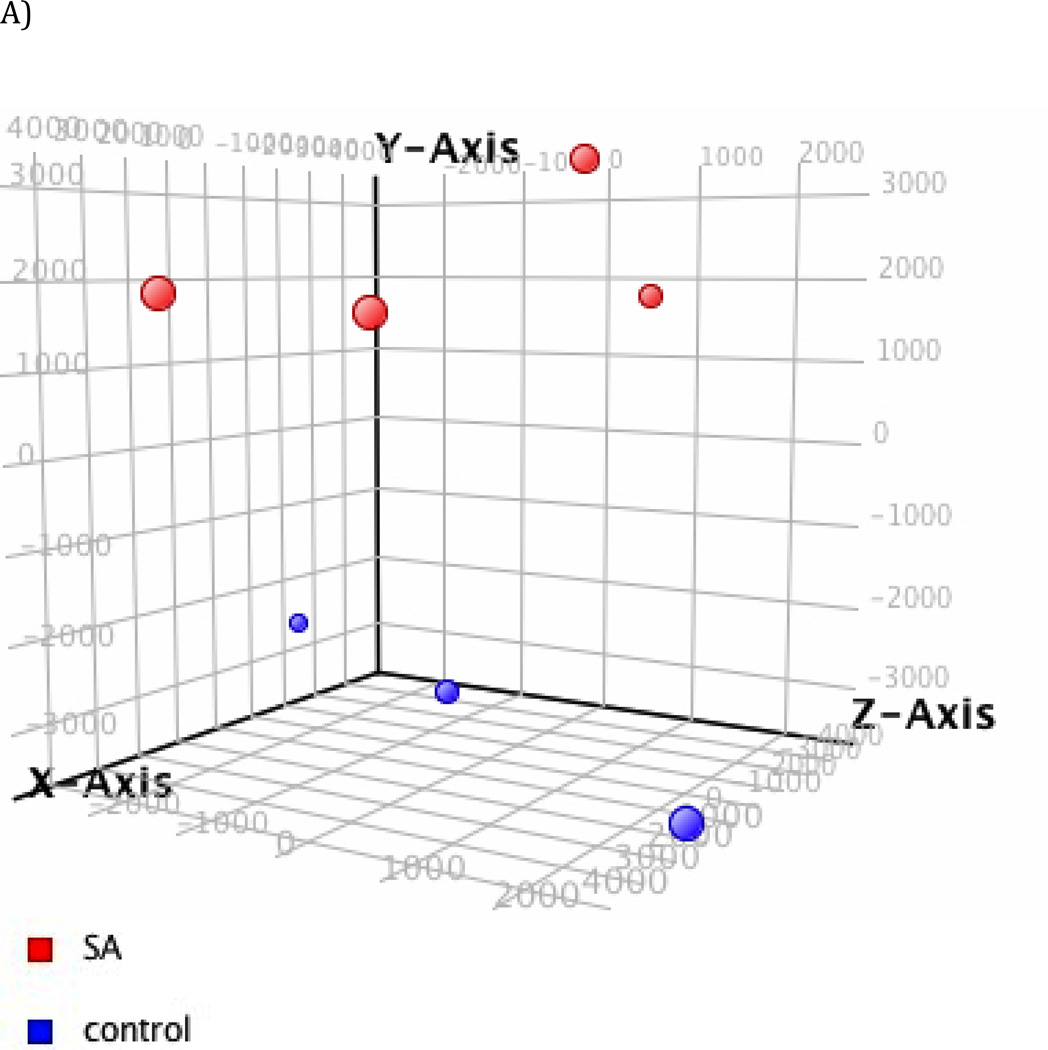
To explore the global impact of PM10 on gene expression, principal component analysis (PCA) was first performed to visualize the profile of all genes without any filtering. The PCA in Figure 2a demonstrates that gene expression in mice treated with PM10 or distilled water exhibits distinct differences in clustering based on exposure.
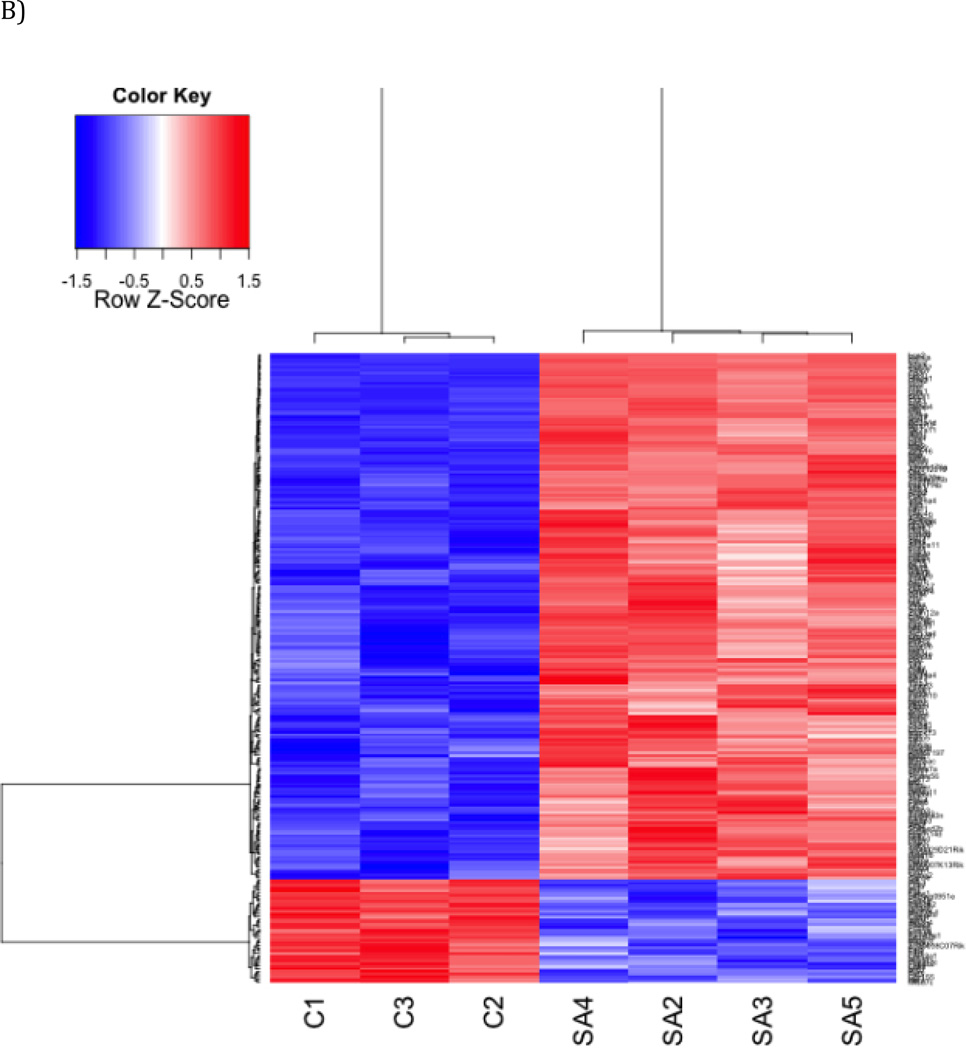
Fig. 2. Gene expression profiles of PM10-exposed mice.
(a) Principal component analysis (PCA) PCA revealed distinct separation between control mice vs. treatment mice Red: PM10; Blue: control (b) Heat Map. Hierarchical cluster analysis of significantly differentially expressed genes in a PM10- treated group compared to an untreated control group. The bar relates the color code to the expression value after quantile normalization and baseline transformation to the median levels of control samples.
Similar effects were seen in hierarchical clustering analysis of genes significantly changed by PM10 (Figure 2b). Gene expression changes between controls and PM10-treated mice were assessed using a gene-wise linear model approach with LIMMA 3.14.3 (Smythe et al. 2005). 202 genes were identified that were significantly up-regulated and 40 genes that were significantly down-regulated in PM10- treated mice. Table 3 displays the top 10 up-regulated and down-regulated genes for the treatment group versus the control.
Table 3.
Top ten up- and down-regulated genes (p< 0.05) in the lungs of PM10-treated mice versus control mice.
| Affymetrix ID | Gene Symbol | Gene Name |
Fold Change |
|---|---|---|---|
| 10563597 | Saa3 | serum amyloid A 3 | 42.4 |
| 10429560 | Ly6i | lymphocyte antigen 6 complex, locus I | 11.0 |
| 10505451 | Orm2 | orosomucoid 2 | 10.6 |
| 10436095 | Retnla | resistin like alpha | 8.72 |
| 10598976 | Timp1 | tissue inhibitor of metalloproteinase 1 | 8.46 |
| 10545569 | Reg3g | regenerating islet-derived 3 gamma | 8.11 |
| 10389231 | Ccl3 | chemokine (C-C motif) ligand 3 | 7.85 |
| 10523156 | Cxcl2 | chemokine (C-X-C motif) ligand 2 | 7.84 |
| 10505438 | Orm1 | orosomucoid 1 | 6.72 |
| 10416837 | Irg1 | immunoresponsive gene 1 | 6.31 |
| 10529937 | Kcnip4 | Kv channel interacting protein 4 | −1.47 |
| 10542929 | Calcr | calcitonin receptor | −1.48 |
| 10509577 | Pla2g2d | phospholipase A2, group IID | −1.51 |
| 10493449 | Thbs3 | Thrombospondin3 | −1.54 |
| 10538802 | A930038C07Rik | RIKEN cDNA A930038C07 gene | −1.55 |
| 10595211 | Col12a1 | collagen, type XII, alpha 1 | −1.55 |
| 10384223 | Igfbp3 | insulin-like growth factor binding protein 3 | −1.58 |
| 10580663 | Ces1f | carboxylesterase 1F | −1.65 |
| 10580678 | Ces1g | carboxylesterase 1G | −1.74 |
| 10523483 | Prdm8 | PR domain containing 8 | −1.79 |
To assess the biological relevance of the differentially expressed genes in PM10- treated versus control the Ingenuity Pathway Analysis (IPA) tool was used. IPA identified many significantly up-regulated genes that participated in lung inflammation: serum amyloid-A3 (SAA3) (42.4-fold), tissue inhibitor of metalloproteinase 1 (TIMP1) (8.46-fold), chemokine (C-X-C motif) ligand 2 (CXCL2) (7.84-fold), tumor necrosis factor receptor superfamily, member 9 (TNFRSF9) (2.59-fold) and chemokine (C-C motif) ligand 2 (CCL2) (3.00-fold). The top molecular and physiological function categories (ranked by p-value) were “cell-to-cell signaling and interaction” and “immune cell trafficking” suggesting that one of the early events in PM10 –exposed mice is the migration of immune cells to the lungs due to a cascade of signaling and recruiting events. Genes associated with top molecular and physiological function categories included: CCL2, CXCL2 and TNFRSF9. Interestingly, the top pathway identified in each toxicity category – such as cardiotoxicity, hepatotoxicity, nephrotoxicity, was inflammation.
Along with many inflammatory genes, PM10 induced genes involved in lipid and cholesterol metabolism, as well as, others associated with metabolic disorders. SAA3 (42.4-fold), CH25H (cholesterol 25 hydroxylase) (2.29-fold), STEAP4 (six transmembrane epithelial antigen of prostate 4) (3.76-fold), LBP (1.46-fold) (lipopolysaccharide binding protein) and PLA2G2D (phospholipase A2, group IID) (−1.51- fold) were differentially expressed between the control and treated mice. RT-PCR revealed another up-regulated gene: OLR1 (oxidized low density lipoprotein (lectin-like) receptor 1) (6.50-fold).
Some of the top IPA canonical pathways were involved in CVD, diabetes and obesity. LXR/RXR activation pathway (p= 7.65 × 10−10) was largely influenced by PM10 exposure. The liver X receptor (LXR) and retinoid X receptor form heterodimers and regulate cholesterol, lipid, and glucose metabolism (Wente et al. 2007). Atherosclerosis signaling (p= 5 × 10−6) was another pathway affected by the exposure. IPA identified TNF- α, Il-6 and PLA2G2D to be involved in this pathway. Other pathways that were induced that are related to liver function include, hepatic cholestasis pathway (p= 2.26 × 10−5) and hepatic fibrosis (p= 3.81 × 10−5).
Induced genes and their upstream regulators
TNF-α and IL-6 were significantly elevated in the BALF of PM10-treated mice. The gene expression profiles of PM10-treated mice contained numerous genes that were downstream of TNF-α and IL-6. The differentially expressed genes, therefore, may have been attributed to the interaction of upstream regulators.
IPA’s upstream regulator analysis represented a novel approach to predict the activation or inhibition of upstream regulators based on the expression pattern of genes downstream to those regulators. In the PM10- treated mice, TNF-α was one of the top upstream regulators that was activated (p= 5.81 × 10−35). TNF-α played a role in the up-regulation of several genes including SAA3, CCL2, TIMP1, STEAP4, CH25H, and LPB. IL-6 was also found to be an upstream regulator that played a role in inducing gene expression (p= 2.76 × 10−26). IL-6 activation in PM10- treated mice contributed to the up-regulation of many of the same genes that were also up-regulated by TNF-α, such as, CCL2, SAA3, CXCL2, and LPB.
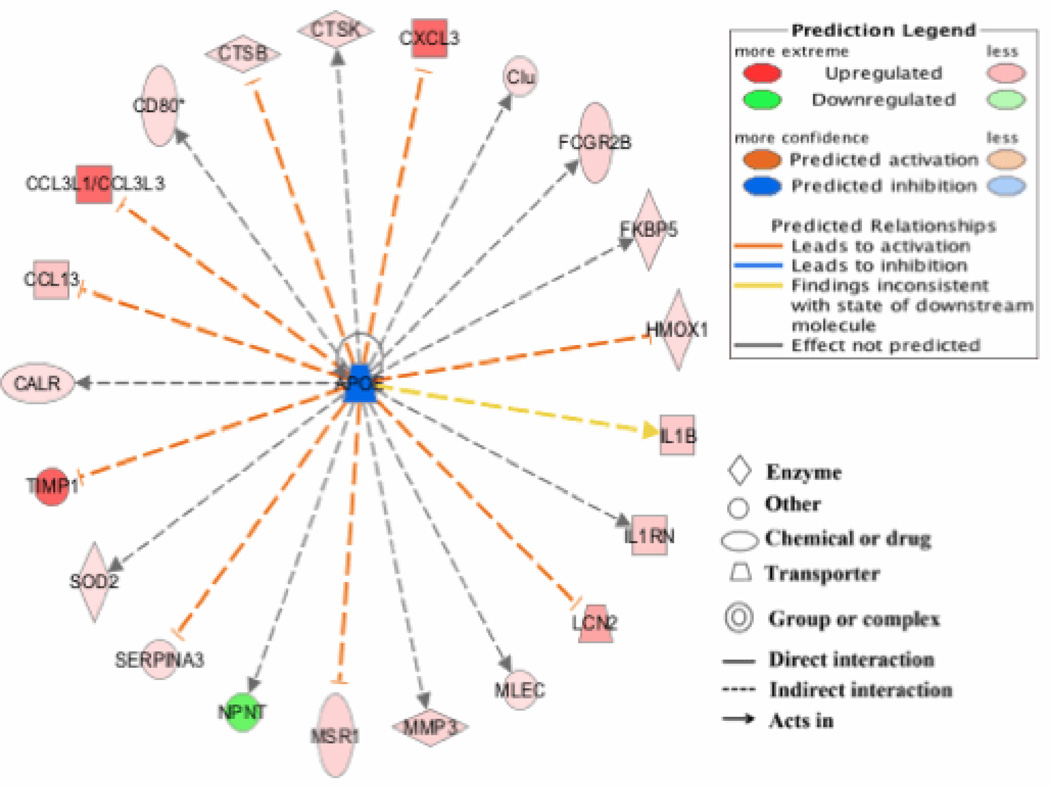
Interestingly, APOE (p= 1.08 × 10−12) was identified by IPA to be activated upstream of many of the differentially expressed genes (Figure 3). Apolipoprotein E (APOE) is produced by the liver and macrophages and is involved in cholesterol metabolism (Frikke-Schmidt 2000). According to IPA, induction of TIMP1, CXCL3, and CCL3 were attributed to upstream activation of APOE.
Fig. 3. APOE is a top upstream regulator influencing altered gene expression in PM10-treated mice.
APOE was identified as an upstream regulator of many of the genes induced by PM10. Most of the genes affected by this regulator are up-regulated and only a few are down-regulated.
Another interesting upstream regulator that may have contributed to the differentially expressed genes was NFκB (p= 6.46 × 10−31). According to IPA, NFκB promotes expression of many of the same genes that are activated by both TNF-α and IL-6 such as TIMP-1, CCL13 and CH25H. NFκB was also involved in the down-regulation of PLA2G2D. Our analysis suggested that following PM10 exposure, TNF-α, IL-6, APOE, and NFκB were upstream regulators that worked together to activate genes involved in inflammation, cholesterol, and lipid metabolism.
Gene expression validation
To validate the gene expression changes observed in the microarray analysis, RNA from lungs of mice was used to perform quantitative real-time PCR (qRT-PCR) of selected genes and the fold changes with qRT-PCR were compared to those from microarrays (Table 4).
Table 4.
Real-time validation of microarray results.
| qRT-PCRa | Microarray | |
|---|---|---|
| TNFRSF9 | 92.5 | 2.59 |
| SAA3 | 299.5 | 42.4 |
| CXCL3 | 16.9 | 2.09 |
| CES1G | −45.8 | −1.74 |
| CH25H | 45.1 | 2.29 |
| CA13 | 18.2 | 1.55 |
| STEAP4 | 18.1 | 3.76 |
| OLR1 | 6.5 | - |
The RT-PCR data represent the means of duplicates and are presented as fold change to the level expressed in control mice lungs.
Discussion
The objective of this study was to characterize the inflammatory responses and gene expression changes in the lungs of mice exposed to PM10 collected from Jeddah, Saudi Arabia. This study demonstrated that acute exposure to PM10 in mice stimulated neutrophil influx into the respiratory tract, elevated TNF-α and IL-6 levels in BALF, and altered the gene expression profiles of lung cells. Many of the induced genes function to promote the inflammatory microenvironment by participating in pathways, such as, immune cell trafficking and cell-to-cell signaling. Along with up-regulated inflammatory genes, genes involved in cholesterol and lipid metabolism were found to have increased expression levels in PM10-treated mice. This is the first study to demonstrate that PM10 collected from Saudi Arabia raises in vivo expression of genes involved in pathways associated with metabolic diseases.
The PM collected from Jeddah, SA is different than other PM sources in a number of ways. A large fraction of the Saudi Arabia PM was coarse PM. The PM’s average ratio of PM2.5/PM10 was 0.33, which was significantly lower than in most other urban locations (Marcazzan et al. 2003; Rodriguez et al. 2004; Yatkin and Bayram 2008). The large portion of coarse PM in the Jeddah PM sample is likely due to the high level of wind-blown dust and sand occurring in that region. Also, nickel (Ni) and vanadium (V) are higher in the PM sample than some of the other metals probably due to the large amount of fossil fuel combustion in the area. Ni and V are common components in many particulate matter samples and studies investigating the effects of PM containing Ni and V have observed CVD endpoints (Afridi et al. 2011; Zhang et al. 2009). (Lippmann et al. 2006) exposed ApoE−/− mice to PM containing high concentrations of Ni. Electrocardiograms of the mice revealed that Ni was significantly associated with acute changes in heart rate and their variability. A study by (Campen et al. 2001) demonstrated that Ni and V combined produced increased toxic effects than just Ni alone.
Metabolic disorders have become major health concerns in Saudi Arabia. Metabolic syndrome is a combination of risk factors, when occurring together, increase the risk of cardiovascular disease and diabetes. Some of the risk factors include: obesity, high blood pressure, high glucose, low high-density cholesterol levels, and high serum triglycerides (Schivo M 2013). Several studies have demonstrated the association of PM exposure with risk factors of metabolic syndrome. A recent study by (Liu et al. 2013) found that PM mediates insulin resistance by regulating visceral adipose tissue inflammation, hepatic lipid metabolism, and glucose utilization in skeletal muscle. An epidemiological study by (Nascimento and Francisco 2013) found that in a city in Brazil as PM exposure increased so did hospitalization due to hypertension. A 10µg/m3 increase in concentration of particulate matter was associated with a 13% increase in risk of hospitalization. In 2002, 35% of deaths in Saudi Arabia were due to cardiovascular disease (CVD) (Taha Abdullah Kumosani 2011). Diabetes mellitus is another chronic disease that occurs at high rates among Saudi Arabians (Alqurashi et al. 2011). Studies have shown that individuals with diabetes are at a higher risk for the toxic effects of particulate matter (Kappos AD 2004). Obesity increases an individual’s chances of developing a chronic disease such as CVD and diabetes. Several studies were conducted documenting high rates of obesity among Saudi Arabian citizens (Al-Malki et al. 2003; Madani et al. 2000). One shocking study revealed that the prevalence of obesity in Saudi Arabian adults was 83% (Madani et al. 2000).
Exposure to air pollution may influence the current problems of obesity and obesity-related diseases in Saudi Arabia. Previously, our lab reported that BEAS-2b cells (human bronchial epithelial cells) exposed to PM10 collected from Saudi Arabia for 4 days contained increased gene expression profiles of genes involved in cholesterol and lipid synthesis pathways (Sun et al. 2012). Interestingly, our current study reports similar findings in vivo and supports the conclusion that PM from Saudi Arabia may be involved in the etiology of metabolic syndromes.
Other studies exposing mice to PM demonstrated that PM affects parameters associated with metabolic syndrome – insulin resistance (Xu et al. 2011) and adipocyte inflammation (Sun et al. 2009). One investigation exposed mice to PM2.5 by inhalation 5 days/week for 10 months and found insulin resistance and a decrease tolerance to glucose. Mice exposed to PM contained a decreased amount of circulating leptin and adiponectin and reduced mitochondrial number and size (Xu et al. 2011). Another study found that mice exposed to a high- fat diet and PM displayed increased systemic inflammation, insulin resistance, and elevation in visceral fat with higher levels of adipose tissue macrophages (Sun et al. 2009). A strain of rabbits that naturally develop atherosclerosis was exposed to 1.5 mg/kg PM10 by intratracheal instillation and the results demonstrated that PM promotes the recruitment of circulating monocytes into atherosclerotic plaques (Yatera et al. 2008).
While metabolic disorders are commonly associated with changes in liver, various studies demonstrated that after exposure to chemical or biological stress, lungs and liver display similar changes in mRNA levels (Crespo et al. 1999; Flohe et al. 1999; Ghoshal et al. 2001; Rosen et al. 2007; Zhang et al. 1998). A study reported that after in utero exposure to perfluorooctanoic acid (PFOA), full term mouse fetuses displayed similar changes in gene expression in lungs and liver, with the majority of changes occurring in genes involved in lipid homeostasis (Rosen et al. 2007).
Many of the PM10-induced genes are involved in cholesterol and lipid metabolism and have been implicated in metabolic disorders- SAA3, CH25H, STEAP4, OLR1, and PLA2G2D. SAA3 (serum amyloid A- 3) was the most up-regulated gene and is an apolipoprotein associated with high density lipoprotein (HDL) in plasma (Uhlar and Whitehead 1999). This gene is primarily expressed in liver and involved in transport of cholesterol to the liver for secretion into bile (de Beer et al. 1994; Uhlar and Whitehead 1999) and both TNF-α and IL-6 were noted to activate SAA3 (Bombini et al. 2004; Fasshauer et al. 2004). SAA protein plays a critical role in inflammatory responses and mediates neutrophil infiltration into the lungs after exposure. SAA was shown to activate the NLRP inflammasome (Ather et al. 2011) and its serum concentrations are increased during inflammatory disorders (Menschikowski et al. 2013). The role of SAA protein in inflammation may be contributing to metabolic syndrome and atherosclerosis since both of these conditions are influenced by inflammation. SAA has been identified as an active player in the development of atherosclerosis. (Dong et al. 2011) reported that ApoE−/− mice overexpressing SAA contained a larger aortic atherosclerotic lesion than that found in control mice. SAA also induced the expression of VCAM-1 (vascular adhesion molecule- 1) (Dong et al. 2011), which is also involved in the development of atherosclerosis (Cybulsky et al. 2001; Nakashima et al. 1998). It should be noted here that SAA3 is an acute response protein that is synthesized during the acute phase of inflammation. Whether or not it was directly activated due to PM-induction of pathways involved in metabolic syndrome is unknown. It may have just been a bystander observation and long-term studies are needed to confirm its continued expression throughout PM exposure and its ability to promote the development of metabolic syndrome.
CH25H (cholesterol 25-hydroxylase) was significantly up-regulated in treated mice and its protein product functions to inhibit the synthesis of cholesterol by indirectly blocking the function of SREBP (sterol regulatory element binding proteins). SREBP are transcription factors that mediate a network of genes involved in cholesterol synthesis. Sterols in turn inhibit SREBP and thus SREBP regulation of sterols is controlled by a negative feedback mechanism (Eberle et al. 2004). SREBF1 and 2 genes were found to be up-regulated by Saudi Arabia PM10 in vitro after 4 days of exposure (Sun et al., 2012). Ultimately, CH25H lowers cholesterol synthesis by negatively regulating SREBP and inducing degradation of HMG-CoA reductase (Gil et al. 1985), which is the enzyme targeted by the group of cholesterol –lowering drugs known as statins. Since Saudi Arabia PM10 affects the levels of SREBF and CH25H, it would be of interest to conduct intermediate and long- term animal investigations in order to determine if the levels of CH25H decrease and SREBF increase.
STEAP4 (six transmembrane epithelial antigen of prostate 4) was also found to be significantly up-regulated in mice treated with Saudi Arabia PM and the gene may be involved in development of metabolic disease. An investigation found that a genetic variation of STEAP4 in Chinese Uygur patients was associated with metabolic syndrome (Nanfang et al. 2010). STEAP4 may be involved in obesity via its role in adipocyte development and metabolism. The protein encoded by STEAP4 is termed TIARP (tumor necrosis factor-alpha-induced adipose-related protein) and acts as a transporter or channel on fat cells which mediate TNF- α effects on adipocyte development (Moldes et al. 2001). Future chronic studies will investigate whether or not PM-induced STEAP4 is correlated with phenotypic changes of fat in exposed mice.
Other interesting genes altered by PM include- OLR1 and PLA2G2D. These genes are associated with cholesterol metabolism (OLR1) (Khaidakov et al. 2011) and lipid metabolism (PLA2G2D) (Degousee et al. 2002). OLR1 (oxidized low density lipoprotein (lectin-like) receptor 1) encodes a protein that binds, internalizes and degrades oxidized low-density lipoprotein. A polymorphism in this gene has been associated with metabolic syndrome (Palmieri et al. 2013). A study found that deletion of the OLR1 gene in LDL receptor-deficient mice fed a high cholesterol diet decreases the outcome of atherogenesis in these mice. The fall in atherogenesis was marked by a reduction in inflammation: a decrease in the proinflammatory signal MAPK P38 and NFκβ, and an elevation in anti-inflammatory levels. A PM- induced up-regulation of OLR1 may promote atherogenesis by increasing inflammatory responses (Mehta et al. 2007). Moreover, OLR1 is up-regulated in the atherosclerotic arteries of humans and animals and has been implicated in diabetes in animals (Mehta et al. 2006). Based on the aforementioned findings, PM-induced OLR1 may play a role in the development of metabolic disease. PLA2G2D (phospholipase A2, group IID) encodes a phospholipase that creates free fatty acids and lysophospholipids by hydrolyzing glycerophospholipids (Degousee et al. 2002). An increase in glycerophospholipids leads to the enrichment of lipid mediators which may promote insulin resistance and impair glucose tolerance in metabolic syndrome (Farooqui 2013). Thus, down-regulation of PLA2G2D in PM-treated mice may lead to metabolic syndrome by increasing glycerophospholipids. However, it should be noted here that another reason for PLAG2D down-regulation may be a cellular response to combat damage to the plasma membrane. Glycerophospholipids are structural components of the plasma membrane and increased synthesis of these lipids may be involved in other pathways besides promotion of metabolic syndrome. PM- induced alterations of lipid metabolism genes may be occurring to reinforce structural integrity to the damaged cell membrane of lung epithelial cells.
Many studies have linked PM exposure with metabolic disorders; however it is still unclear how ambient particulates mediate adverse health effects. Some of the gene expression changes that occur after an acute exposure to PM10 that may influence the development of metabolic diseases were characterized. Our studies to-date characterizing the gene expression changes induced by PM10 collected from Jeddah, Saudi Arabia have generated in vitro and in vivo findings that suggest that PM10 may be mediating or exacerbating metabolic diseases- a health concern that is increasing rapidly in Saudi Arabia. While this study provides data that acute exposure to PM10 affects pathways involved in cholesterol and lipid metabolism, long- term studies are needed to confirm an association of PM10 exposure and the occurrence of metabolic syndrome risk factors.
Acknowledgements
Thanks to Chin Chiu, Lisa Passantino, Yana Chervona, and Alexandra Munoz for help with experimental procedures and data analysis.
Funding Information
This work was funded by King Abdulaziz University (KAU), Jeddah, under grant number 4/00/00/252 and by NIEHS under grant # P30ES000260. The sponsors did not have any role in the study design.
Footnotes
Institution where work was performed: Department of Environmental Medicine, NYU School of Medicine, New York, NY, USA
REFERENCES
- Afridi HI, Kazi TG, Kazi N, Kandhro GA, Baig JA, Jamali MK, Arain MB, Shah AQ, Shah F, Khan S, Kolachi NF. Association of environmental toxic elements in biological samples of myocardial infarction patients at different stages. Biol Trace Elem Res. 2011;141:26–40. doi: 10.1007/s12011-010-8713-2. [DOI] [PubMed] [Google Scholar]
- Al-Malki JS, Al-Jaser MH, Warsy AS. Overweight and obesity in Saudi females of childbearing age. Int J Obes Relat Metab Disord. 2003;27:134–139. doi: 10.1038/sj.ijo.0802181. [DOI] [PubMed] [Google Scholar]
- Al-Othaimeen AI A-NM, Osman AK. Obesity: an emerging problem in Saudi Arabia. Analysis of data from the National Nutrition Survey. East Mediterr Health J. 2007;13:441–448. [PubMed] [Google Scholar]
- Alqurashi KA, Aljabri KS, Bokhari SA. Prevalence of diabetes mellitus in a Saudi community. Ann Saudi Med. 2011;31:19–23. doi: 10.4103/0256-4947.75773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Araujo JA, Nel AE. Particulate matter and atherosclerosis: Role of particle size, composition and oxidative stress. Part Fibre Toxicol. 2009;6:24. doi: 10.1186/1743-8977-6-24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ather JL, Ckless K, Martin R, Foley KL, Suratt BT, Boyson JE, Fitzgerald KA, Flavell RA, Eisenbarth SC, Poynter ME. Serum amyloid A activates the NLRP3 inflammasome and promotes Th17 allergic asthma in mice. J Immunol. 2011;187:64–73. doi: 10.4049/jimmunol.1100500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beckerman BS, Jerrett M, Finkelstein M, Kanaroglou P, Brook JR, Arain MA, Sears MR, Stieb D, Balmes J, Chapman K. The association between chronic exposure to traffic-related air pollution and ischemic heart disease. J Toxicol Environ Health A. 2012;75:402–411. doi: 10.1080/15287394.2012.670899. [DOI] [PubMed] [Google Scholar]
- Benjamini Y HY. Controlling the false discovery rate: A practical and powerful approach to multiple testing. J Roy Stat Soc Series B. 1995:289–300. [Google Scholar]
- Bolstad BM, Irizarry RA, Astrand M, Speed TP. A comparison of normalization methods for high density oligonucleotide array data based on variance and bias. Bioinformatics. 2003;19:185–193. doi: 10.1093/bioinformatics/19.2.185. [DOI] [PubMed] [Google Scholar]
- Bombini G, Canetti C, Rocha FA, Cunha FQ. Tumour necrosis factor-alpha mediates neutrophil migration to the knee synovial cavity during immune inflammation. Eur J Pharmacol. 2004;496:197–204. doi: 10.1016/j.ejphar.2004.06.003. [DOI] [PubMed] [Google Scholar]
- Brook RD, Cakmak S, Turner MC, Brook JR, Crouse DL, Peters PA, van Donkelaar A, Villeneuve PJ, Brion O, Jerrett M, Martin RV, Rajagopalan S, Goldberg MS, Pope CA, 3rd, Burnett RT. Long-Term fine particulate matter exposure and mortality from diabetes mellitus in canada. Diabetes Care. 2013 Oct;36(10):3313–3320. doi: 10.2337/dc12-2189. 2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brook RD, Rajagopalan S, Pope CA, 3rd, Brook JR, Bhatnagar A, Diez-Roux AV, Holguin F, Hong Y, Luepker RV, Mittleman MA, Peters A, Siscovick D, Smith SC, Jr, Whitsel L, Kaufman JD. Particulate matter air pollution and cardiovascular disease: An update to the scientific statement from the American Heart Association. Circulation. 2010;121:2331–2378. doi: 10.1161/CIR.0b013e3181dbece1. [DOI] [PubMed] [Google Scholar]
- Campen MJ, Nolan JP, Schladweiler MC, Kodavanti UP, Evansky PA, Costa DL, Watkinson WP. Cardiovascular and thermoregulatory effects of inhaled PM-associated transition metals: a potential interaction between nickel and vanadium sulfate. Toxicol Sci. 2001;64:243–252. doi: 10.1093/toxsci/64.2.243. [DOI] [PubMed] [Google Scholar]
- Chang CC, Kuo CC, Liou SH, Yang CY. Fine particulate air pollution and hospital admissions for myocardial infarction in a subtropical city: Taipei, Taiwan. J Toxicol Environ Health A. 2013;76:440–448. doi: 10.1080/15287394.2013.771559. [DOI] [PubMed] [Google Scholar]
- Chiu HF, Tsai SS, Weng HH, Yang CY. Short-term effects of fine particulate air pollution on emergency room visits for cardiac arrhythmias: a case-crossover study in Taipei. J Toxicol Environ Health A. 2013;76:614–623. doi: 10.1080/15287394.2013.801763. [DOI] [PubMed] [Google Scholar]
- Colucci ME, Veronesi L, Roveda AM, Marangio E, Sansebastiano G. Particulate matter (PM10) air pollution, daily mortality, and hospital admissions: recent findings. Ig Sanita Pubbl. 2006;62:289–304. [PubMed] [Google Scholar]
- Crespo E, Macias M, Pozo D, Escames G, Martin M, Vives F, Guerrero JM, Acuna-Castroviejo D. Melatonin inhibits expression of the inducible NO synthase II in liver and lung and prevents endotoxemia in lipopolysaccharide-induced multiple organ dysfunction syndrome in rats. FASEB J. 1999;13:1537–1546. [PubMed] [Google Scholar]
- Cybulsky MI, Iiyama K, Li H, Zhu S, Chen M, Iiyama M, Davis V, Gutierrez-Ramos JC, Connelly PW, Milstone DS. A major role for VCAM-1, but not ICAM-1, in early atherosclerosis. J Clin Invest. 2001;107:1255–1262. doi: 10.1172/JCI11871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Beer MC, Kindy MS, Lane WS, de Beer FC. Mouse serum amyloid A protein (SAA5) structure and expression. J Biol Chem. 1994;269:4661–4667. [PubMed] [Google Scholar]
- Degousee N, Ghomashchi F, Stefanski E, Singer A, Smart BP, Borregaard N, Reithmeier R, Lindsay TF, Lichtenberger C, Reinisch W, Lambeau G, Arm J, Tischfield J, Gelb MH, Rubin BB. Groups IV, V, and X phospholipases A2s in human neutrophils: Role in eicosanoid production and gram-negative bacterial phospholipid hydrolysis. J Biol Chem. 2002;277:5061–5073. doi: 10.1074/jbc.M109083200. [DOI] [PubMed] [Google Scholar]
- Delfino RJ, Wu J, Tjoa T, Gullesserian SK, Nickerson B, Gillen DL. Asthma morbidity and ambient air pollution: effect modification by residential traffic-related air pollution. Epidemiology. 2014;25:48–57. doi: 10.1097/EDE.0000000000000016. [DOI] [PubMed] [Google Scholar]
- Dong Z, Wu T, Qin W, An C, Wang Z, Zhang M, Zhang Y, Zhang C, An F. Serum amyloid A directly accelerates the progression of atherosclerosis in apolipoprotein E-deficient mice. Mol Med. 2011;17:1357–1364. doi: 10.2119/molmed.2011.00186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eberle D, Hegarty B, Bossard P, Ferre P, Foufelle F. SREBP transcription factors: Master regulators of lipid homeostasis. Biochimie. 2004;86:839–848. doi: 10.1016/j.biochi.2004.09.018. [DOI] [PubMed] [Google Scholar]
- Elassouli SM, Alqahtani MH, Milaat W. Genotoxicity of air borne particulates assessed by comet and the Salmonella mutagenicity test in Jeddah, Saudi Arabia. Int J Environ Res Public Health. 2007;4:216–233. doi: 10.3390/ijerph2007030004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Farooqui AA. Lipid mediators in metabolic syndrome and neurological disorders. Metab Syndrome. 2013:103–141. [Google Scholar]
- Fasshauer M, Klein J, Kralisch S, Klier M, Lossner U, Bluher M, Paschke R. Serum amyloid A3 expression is stimulated by dexamethasone and interleukin-6 in 3T3-L1 adipocytes. J Endocrinol. 2004;183:561–567. doi: 10.1677/joe.1.05699. [DOI] [PubMed] [Google Scholar]
- Flohe S, Dominguez Fernandez E, Ackermann M, Hirsch T, Borgermann J, Schade FU. Endotoxin tolerance in rats: expression of TNF-alpha, IL-6, IL-10, VCAM-1 AND HSP 70 in lung and liver during endotoxin shock. Cytokine. 1999;11:796–804. doi: 10.1006/cyto.1998.0490. [DOI] [PubMed] [Google Scholar]
- Frikke-Schmidt R. Context-dependent and invariant associations between APOE genotype and levels of lipoproteins and risk of ischemic heart disease: A review. Scand J Clin Lab Invest Suppl. 2000;233:3–25. [PubMed] [Google Scholar]
- Gan WQ, Fitzgerald JM, Carlsten C, Sadatsafavi M, Brauer M. Associations of ambient air pollution with chronic obstructive pulmonary disease hospitalization and mortality. Am J Respir Crit Care Med. 2013 Apr 1;187(7):721–727. doi: 10.1164/rccm.201211-2004OC. 2013. [DOI] [PubMed] [Google Scholar]
- Gautier L, Cope L, Bolstad BM, Irizarry RA. affy--analysis of Affymetrix GeneChip data at the probe level. Bioinformatics. 2004;20:307–315. doi: 10.1093/bioinformatics/btg405. [DOI] [PubMed] [Google Scholar]
- Ghio AJ, Carraway MS, Madden MC. Composition of air pollution particles and oxidative stress in cells, tissues, and living systems. J Toxicol Environ Health B. 2012;15:1–21. doi: 10.1080/10937404.2012.632359. [DOI] [PubMed] [Google Scholar]
- Ghoshal K, Majumder S, Zhu Q, Hunzeker J, Datta J, Shah M, Sheridan JF, Jacob ST. Influenza virus infection induces metallothionein gene expression in the mouse liver and lung by overlapping but distinct molecular mechanisms. Mol Cell Biol. 2001;21:8301–8317. doi: 10.1128/MCB.21.24.8301-8317.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gil G, Faust JR, Chin DJ, Goldstein JL, Brown MS. Membrane-bound domain of HMG CoA reductase is required for sterol-enhanced degradation of the enzyme. Cell. 1985;41:249–258. doi: 10.1016/0092-8674(85)90078-9. [DOI] [PubMed] [Google Scholar]
- Huang YC, Karoly ED, Dailey LA, Schmitt MT, Silbajoris R, Graff DW, Devlin RB. Comparison of gene expression profiles induced by coarse, fine, and ultrafine particulate matter. J Toxicol Environ Health A. 2011;74:296–312. doi: 10.1080/15287394.2010.516238. [DOI] [PubMed] [Google Scholar]
- Kaplanski G, Marin V, Montero-Julian F, Mantovani A, Farnarier C. IL-6: A regulator of the transition from neutrophil to monocyte recruitment during inflammation. Trends Immunol. 2003;24:25–29. doi: 10.1016/s1471-4906(02)00013-3. [DOI] [PubMed] [Google Scholar]
- Kappos AD BP, Eikmann T, Englert N, Heinrich U, Höppe P, Koch E, Krause GH, Kreyling WG, Rauchfuss K, Rombout P, Schulz-Klemp V, Thiel WR, Wichmann HE. Health effects of particles in ambient air. Int J Hyg Environ Health. 2004;207:399–407. doi: 10.1078/1438-4639-00306. [DOI] [PubMed] [Google Scholar]
- Karakatsani A, Analitis A, Perifanou D, Ayres JG, Harrison RM, Kotronarou A, Kavouras IG, Pekkanen J, Hameri K, Kos GP, de Hartog JJ, Hoek G, Katsouyanni K. Particulate matter air pollution and respiratory symptoms in individuals having either asthma or chronic obstructive pulmonary disease: a European multicentre panel study. Environ Health. 2012;11:75. doi: 10.1186/1476-069X-11-75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keith R, Miller Y, Hudish T, Girod C, Sotto-Santiago S, Franklin W, Nemenoff R, March T, Nana-Sinkam S, Geraci M. Pulmonary prostacyclin synthase overexpression chemoprevents tobacco smoke lung carcinogenesis in mice. Cancer Res. 2004 Aug 15;64(16):5897–5904. doi: 10.1158/0008-5472.CAN-04-1070. 2004, 64. [DOI] [PubMed] [Google Scholar]
- Khaidakov M, Mitra S, Kang BY, Wang X, Kadlubar S, Novelli G, Raj V, Winters M, Carter WC, Mehta JL. Oxidized LDL receptor 1 (OLR1) as a possible link between obesity, dyslipidemia and cancer. PLoS One. 2011;6:e20277. doi: 10.1371/journal.pone.0020277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khodeir M, Shamy M, Alghamdi M, Zhong M, Sun H, Costa M, Chen LC, Maciejczyk P. Source apportionment and elemental composition of PM2.5 and PM10 in Jeddah City, Saudi Arabia. Atmos Pollut Res. 2012;3:331–340. doi: 10.5094/apr.2012.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kooter I, Pennings J, Opperhuizen A, Cassee F. Gene expression pattern in spontaneously hypertensive rats exposed to urban particulate matter (EHC-93) Inhal Toxicol. 2005;17:53–65. doi: 10.1080/08958370590885717. [DOI] [PubMed] [Google Scholar]
- Lippmann M, Ito K, Hwang JS, Maciejczyk P, Chen LC. Cardiovascular effects of nickel in ambient air. Environ Health Perspect. 2006;114:1662–1669. doi: 10.1289/ehp.9150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu C, Xu X, Bai Y, Wang TY, Rao X, Wang A, Sun L, Ying Z, Gushchina L, Maiseyeu A, Morishita M, Sun Q, Harkema JR, Rajagopalan S. Air Pollution-Mediated Susceptibility to Inflammation and Insulin Resistance: Influence of CCR2 Pathways in Mice. Environ Health Perspect. 2013 doi: 10.1289/ehp.1306841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Madani KA, al-Amoudi NS, Kumosani TA. The state of nutrition in Saudi Arabia. Nutr Health. 2000;14:17–31. doi: 10.1177/026010600001400103. [DOI] [PubMed] [Google Scholar]
- Marcazzan GM, Ceriani M, Valli G, Vecchi R. Source apportionment of PM10 and PM2.5 in Milan (Italy) using receptor modelling. Sci Total Environ. 2003;317:137–147. doi: 10.1016/S0048-9697(03)00368-1. [DOI] [PubMed] [Google Scholar]
- Mazzoli-Rocha F, Fernandes S, Einicker-Lamas M, Zin WA. Roles of oxidative stress in signaling and inflammation induced by particulate matter. Cell Biol Toxicol. 2010;26:481–498. doi: 10.1007/s10565-010-9158-2. [DOI] [PubMed] [Google Scholar]
- Mehta JL, Chen J, Hermonat PL, Romeo F, Novelli G. Lectin-like, oxidized low-density lipoprotein receptor-1 (LOX-1): a critical player in the development of atherosclerosis and related disorders. Cardiovasc Res. 2006;69:36–45. doi: 10.1016/j.cardiores.2005.09.006. [DOI] [PubMed] [Google Scholar]
- Mehta JL, Sanada N, Hu CP, Chen J, Dandapat A, Sugawara F, Satoh H, Inoue K, Kawase Y, Jishage K, Suzuki H, Takeya M, Schnackenberg L, Beger R, Hermonat PL, Thomas M, Sawamura T. Deletion of LOX-1 reduces atherogenesis in LDLR knockout mice fed high cholesterol diet. Circ Res. 2007;100:1634–1642. doi: 10.1161/CIRCRESAHA.107.149724. [DOI] [PubMed] [Google Scholar]
- Menschikowski M, Hagelgans A, Fuessel S, Mareninova OA, Asatryan L, Wirth MP, Siegert G. Serum amyloid A, phospholipase A2-IIA and C-reactive protein as inflammatory biomarkers for prostate diseases. Inflamm Res. 2013;62:1063–1072. doi: 10.1007/s00011-013-0665-5. [DOI] [PubMed] [Google Scholar]
- Moldes M, Lasnier F, Gauthereau X, Klein C, Pairault J, Feve B, Chambaut-Guerin AM. Tumor necrosis factor-alpha-induced adipose-related protein (TIARP), a cell-surface protein that is highly induced by tumor necrosis factor-alpha and adipose conversion. J Biol Chem. 2001;276:33938–33946. doi: 10.1074/jbc.M105726200. [DOI] [PubMed] [Google Scholar]
- Motta V, Angelici L, Nordio F, Bollati V, Fossati S, Frascati F, Tinaglia V, Bertazzi PA, Battaglia C, Baccarelli AA. Integrative Analysis of miRNA and inflammatory gene expression after acute particulate matter exposure. Toxicol Sci. 2013;132:307–316. doi: 10.1093/toxsci/kft013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakashima Y, Raines EW, Plump AS, Breslow JL, Ross R. Upregulation of VCAM-1 and ICAM-1 at atherosclerosis-prone sites on the endothelium in the ApoE-deficient mouse. Arterioscler Thromb Vasc Biol. 1998;18:842–851. doi: 10.1161/01.atv.18.5.842. [DOI] [PubMed] [Google Scholar]
- Nanfang L, Yanying G, Hongmei W, Zhitao Y, Juhong Z, Ling Z, Wenli L. Variations of six transmembrane epithelial antigen of prostate 4 (STEAP4) gene are associated with metabolic syndrome in a female Uygur general population. Arch Med Res. 2010;41:449–456. doi: 10.1016/j.arcmed.2010.08.006. [DOI] [PubMed] [Google Scholar]
- Nascimento LF, Francisco JB. Particulate matter and hospital admission due to arterial hypertension in a medium-sized Brazilian city. Cad Saude Publica. 2013;29:1565–1571. doi: 10.1590/0102-311x00127612. [DOI] [PubMed] [Google Scholar]
- Nasralla MM. Air pollution in the semitropical Saudi urban area. Environment International. 1983;9:225–264. [Google Scholar]
- Nishiwaki Y, Michikawa T, Takebayashi T, Nitta H, Iso H, Inoue M, Tsugane S. Long-term exposure to particulate matter in relation to mortality and incidence of cardiovascular disease: The JPHC Study. J Atheroscler Thromb. 2012;20(3):296–309. doi: 10.5551/jat.15347. 2013. [DOI] [PubMed] [Google Scholar]
- Palmieri VO, Coppola B, Grattagliano I, Casieri V, Cardinale G, Portincasa P, Palasciano G, Di Serio F. Oxidized LDL receptor 1 gene polymorphism in patients with metabolic syndrome. Eur J Clin Invest. 2013;43:41–48. doi: 10.1111/eci.12013. [DOI] [PubMed] [Google Scholar]
- Pedersen M, Giorgis-Allemand L, Bernard C, Aguilera I, Andersen AM, Ballester F, Beelen RM, Chatzi L, Cirach M, Danileviciute A, Dedele A, Eijsden M, Estarlich M, Fernandez-Somoano A, Fernandez MF, Forastiere F, Gehring U, Grazuleviciene R, Gruzieva O, Heude B, Hoek G, de Hoogh K, van den Hooven EH, Haberg SE, Jaddoe VW, Klumper C, Korek M, Kramer U, Lerchundi A, Lepeule J, Nafstad P, Nystad W, Patelarou E, Porta D, Postma D, Raaschou-Nielsen O, Rudnai P, Sunyer J, Stephanou E, Sorensen M, Thiering E, Tuffnell D, Varro MJ, Vrijkotte TG, Wijga A, Wilhelm M, Wright J, Nieuwenhuijsen MJ, Pershagen G, Brunekreef B, Kogevinas M, Slama R. Ambient air pollution and low birthweight: a European cohort study (ESCAPE) Lancet Respir Med. 2013;1:695–704. doi: 10.1016/S2213-2600(13)70192-9. [DOI] [PubMed] [Google Scholar]
- Rodriguez S, Querol X, Alastuey A, Viana MM, Alarcon M, Mantilla E, Ruiz CR. Comparative PM10-PM2.5 source contribution study at rural, urban and industrial sites during PM episodes in Eastern Spain. Sci Total Environ. 2004;328:95–113. doi: 10.1016/S0048-9697(03)00411-X. [DOI] [PubMed] [Google Scholar]
- Rosen MB, Thibodeaux JR, Wood CR, Zehr RD, Schmid JE, Lau C. Gene expression profiling in the lung and liver of PFOA-exposed mouse fetuses. Toxicology. 2007;239:15–33. doi: 10.1016/j.tox.2007.06.095. [DOI] [PubMed] [Google Scholar]
- Rosenblum MG, Donato NJ. Tumor necrosis factor alpha: A multifaceted peptide hormone. Crit Rev Immuno. 1989;l9:21–44. [PubMed] [Google Scholar]
- Samet J, Krewski D. Health effects associated with exposure to ambient air pollution. J Toxicol Environ Health A. 2007;70:227–242. doi: 10.1080/15287390600884644. [DOI] [PubMed] [Google Scholar]
- Scapini P, Lapinet-Vera JA, Gasperini S, Calzetti F, Bazzoni F, Cassatella MA. The neutrophil as a cellular source of chemokines. Immunol Rev. 2000;177:195–203. doi: 10.1034/j.1600-065x.2000.17706.x. [DOI] [PubMed] [Google Scholar]
- Schivo M AA, Yeates LC, Pasamontes A, Davis CE. Diabetes and the Metabolic Syndrome: Possibilities of a New Breath Test in a Dolphin Model. Front Endocrinol (Lausanne) 2013;4 doi: 10.3389/fendo.2013.00163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simkhovich BZ, Kleinman MT, Kloner RA. Air pollution and cardiovascular injury epidemiology, toxicology, and mechanisms. J Am Coll Cardiol. 2008;52:719–726. doi: 10.1016/j.jacc.2008.05.029. [DOI] [PubMed] [Google Scholar]
- Smythe GK, Gentleman S, Dudoit RI, Huber W. Limma: Linear models for microarray data. Bioinformatics and Computational Biology Solutions using R and Bioconductor. 2005:397–420. [Google Scholar]
- Sun H, Shamy M, Kluz T, Munoz AB, Zhong M, Laulicht F, Alghamdi MA, Khoder MI, Chen LC, Costa M. Gene expression profiling and pathway analysis of human bronchial epithelial cells exposed to airborne particulate matter collected from Saudi Arabia. Toxicol Appl Pharmacol. 2012;265:147–157. doi: 10.1016/j.taap.2012.10.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sun Q, Yue P, Deiuliis JA, Lumeng CN, Kampfrath T, Mikolaj MB, Cai Y, Ostrowski MC, Lu B, Parthasarathy S, Brook RD, Moffatt-Bruce SD, Chen LC, Rajagopalan S. Ambient air pollution exaggerates adipose inflammation and insulin resistance in a mouse model of diet-induced obesity. Circulation. 2009;119:538–546. doi: 10.1161/CIRCULATIONAHA.108.799015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tagaris E, Liao KJ, DeLucia AJ, Deck L, Amar P, Russell AG. Sensitivity of air pollution-induced premature mortality to precursor emissions under the influence of climate change. Int J Environ Res Public Health. 2010;7:2222–2237. doi: 10.3390/ijerph7052222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taha Abdullah Kumosani M. N. A. a. A. I. Cardiovascular diseases in Saudi Arabia. Prime Research on Medicine. 2011;1:1–6. [Google Scholar]
- Thomson EM, Vladisavljevic D, Mohottalage S, Kumarathasan P, Vincent R. Mapping acute systemic effects of inhaled particulate matter and ozone: multiorgan gene expression and glucocorticoid activity. Toxicol Sci. 2013;135:169–181. doi: 10.1093/toxsci/kft137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Uhlar CM, Whitehead AS. Serum amyloid A, the major vertebrate acute-phase reactant. Eur J Biochem. 1999;265:501–523. doi: 10.1046/j.1432-1327.1999.00657.x. [DOI] [PubMed] [Google Scholar]
- Vigotti MA, Chiaverini F, Biagiola P, Rossi G. Urban air pollution and emergency visits for respiratory complaints in Pisa, Italy. J Toxicol Environ Health A. 2007;70:266–269. doi: 10.1080/15287390600884800. [DOI] [PubMed] [Google Scholar]
- Wente W, Brenner MB, Zitzer H, Gromada J, Efanov AM. Activation of liver X receptors and retinoid X receptors induces growth arrest and apoptosis in insulin-secreting cells. Endocrinology. 2007;148:1843–1849. doi: 10.1210/en.2006-1247. [DOI] [PubMed] [Google Scholar]
- Xu X, Liu C, Xu Z, Tzan K, Zhong M, Wang A, Lippmann M, Chen LC, Rajagopalan S, Sun Q. Long-term exposure to ambient fine particulate pollution induces insulin resistance and mitochondrial alteration in adipose tissue. Toxicol Sci. 2011;124:88–98. doi: 10.1093/toxsci/kfr211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yanagi Y, Assuncao JV, Barrozo LV. The impact of atmospheric particulate matter on cancer incidence and mortality in the city of Sao Paulo, Brazil. Cad Saude Publica. 2012;28:1737–1748. doi: 10.1590/s0102-311x2012000900012. [DOI] [PubMed] [Google Scholar]
- Yatera K, Hsieh J, Hogg JC, Tranfield E, Suzuki H, Shih CH, Behzad AR, Vincent R, van Eeden SF. Particulate matter air pollution exposure promotes recruitment of monocytes into atherosclerotic plaques. Am J Physiol Heart Circ Physiol. 2008;294:H944–H953. doi: 10.1152/ajpheart.00406.2007. [DOI] [PubMed] [Google Scholar]
- Yatkin S, Bayram A. Source apportionment of PM(10) and PM(2.5) using positive matrix factorization and chemical mass balance in Izmir, Turkey. Sci Total Environ. 2008;390:109–123. doi: 10.1016/j.scitotenv.2007.08.059. [DOI] [PubMed] [Google Scholar]
- Zhang HG, Zhou T, Yang P, Edwards CK, 3rd, Curiel DT, Mountz JD. Inhibition of tumor necrosis factor alpha decreases inflammation and prolongs adenovirus gene expression in lung and liver. Human Gene Ther. 1998;9:1875–1884. doi: 10.1089/hum.1998.9.13-1875. [DOI] [PubMed] [Google Scholar]
- Zhang Z, Chau PY, Lai HK, Wong CM. A review of effects of particulate matter-associated nickel and vanadium species on cardiovascular and respiratory systems. Int J Environ Health Res. 2009;19:175–185. doi: 10.1080/09603120802460392. [DOI] [PubMed] [Google Scholar]