Abstract
We investigated the efficacy and tolerability of various anticholinergics in Korean children with non-neurogenic overactive bladder (OAB). A total of 326 children (males:females= 157:169) aged under 18 yr (mean age 7.3±2.6 yr) who were diagnosed with OAB from 2008 to 2011 were retrospectively reviewed. The mean duration of OAB symptoms before anticholinergic treatment was 16.9±19.0 months. The mean duration of medication was 5.6±7.3 months. Urgency urinary incontinence episodes per week decreased from 1.9±3.1 to 0.4±1.5 times (P<0.001). The median voiding frequency during daytime was decreased from 9.2±5.4 to 6.3±4.2 times (P<0.001). According to 3-day voiding diaries, the maximum and average bladder capacity were increased from 145.5±66.9 to 196.8±80.3 mL and from 80.8±39.6 to 121.8±56.5 mL, respectively (P<0.001). On uroflowmetry, maximum flow rate was increased from 17.6±8.4 to 20.5±8.2 mL/sec (P<0.001). Adverse effects were reported in 14 (4.3%) children and six children (1.8%) discontinued medication due to adverse effects. Our results indicate that anticholinergics are effective to improve OAB symptoms and tolerability was acceptable without severe complications in children.
Keywords: Overactive Urinary Bladder, Urinary Incontinence, Anticholinergics, Efficacy, Tolerability
INTRODUCTION
Overactive bladder (OAB), or involuntary contraction of the detrusor muscle during the filling phase, is distressing for patients and physicians alike. Up to 20% of 4 to 6-yr-old children have daytime wetting, and 3% of them experience wetting episodes more than twice a week (1). The International Continence Society, in 2002, defined a symptomatic OAB as urinary urgency (a sudden and compelling desire to urinate), with or without urgency urinary incontinence (UUI), usually with frequency and nocturia, in the absence of pathologic or metabolic factors that would explain these symptoms (2). OAB is characterized by the presence of unstable bladder when urodynamic investigation shows involuntary detrusor contractions during the bladder filling phase, which may be spontaneous or provoked (3). Although the subjective hallmark of OAB is urgency (4), the objective diagnosis is based on the urodynamic findings revealing the involuntary bladder contractions caused by detrusor overactivity (DO) while patients are attempting to suppress voiding.
The first-line treatment of non-neurogenic OAB symptoms typically begins with conservative measures and urotherapy, including a bladder training regimen, treatment of constipation, timed voiding, behavioral modifications, pelvic floor physiotherapy, and electrical stimulation. For children whose symptoms persist, anticholinergic medications are often added to decrease DO via blockade of bladder M3 muscarinic receptors (5). The use of anticholinergic medications in treatment of OAB for children has not been studied extensively, however, 30%-40% of children were reported to have no response to the anticholinergics (6). Common adverse events reported in children were also typical of the antimuscarinic drug class, such as dry mouth and constipation (7, 8, 9, 10).
In Korea, currently oxybutynin chloride among various antimuscarinics is the only anticholinergic agent approved by the Korea Food and Drug Administration (FDA) for OAB in children. As there are limited data on the use of anticholinergics in pediatric patients with OAB, little is known about the benefits and tolerability of anticholinergics. Therefore, the aim of this study was to retrospectively investigate the efficacy and tolerability of anticholinergics used in Korean children with OAB.
MATERIALS AND METHODS
Study design and patients
A total of 347 patients diagnosed with lower urinary tract symptom (LUTS) between January 2008 and December 2011 was enrolled from 15 hospitals, and 326 patients who were suitable for inclusion criteria were retrospectively analyzed. The following clinical data were documented: age and gender of the patients, associated voiding symptoms, duration of symptoms, anticholinergics used, dosage of anticholinergics, duration of anticholinergics, baseline urgency, urgency incontinence, and frequency, the maximum and average bladder capacity, results of uroflowmetry, adverse effects, and treatment outcomes. A careful history focused on urinary symptoms, physical examination, and urinanalysis were obtained from all patients. Voiding diary and uroflowmetry with residual urine check were also performed for some selected patients.
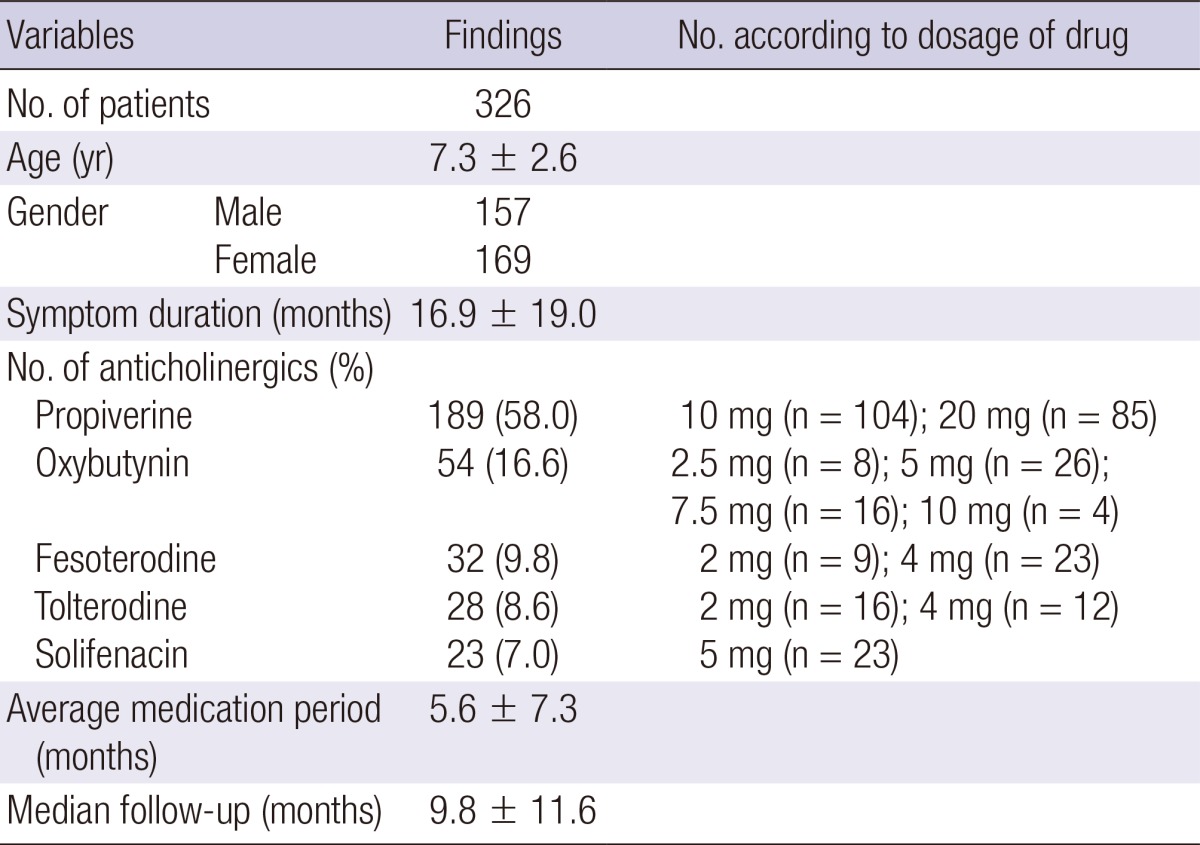
Among 326 children, there are 157 boys and 169 girls (mean age, 7.3±2.6 yr; range, 5-18 yr). The mean duration of OAB symptoms before anticholinergic treatment was 16.9±19 months (Table 1). There were no statistical differences in age or gender according to the types of anticholinergics. Propiverine was used for 189 patients (58%); oxybutynin was used for 54 patients (16.6%); fesoterodine was used for 32 patients (9.8%); tolterodine was used for 28 patients (8.6%); solifenacin was used for 23 patients (7%). The mean duration of medication was 5.6±7.3 months, and the mean duration of follow-up was 9.8±11.6 months. All the variables of the patients were compared between pretreatment and post-treatment with anticholinergics for OAB. The efficacy and tolerability were evaluated after anticholinergic treatment.
Table 1.
Patient and clinical characteristic
Inclusion and exclusion criteria
All the patients were treated with an anticholinergic agent for more than 1 week. Patients younger than 5 or older than 19 yr were excluded from this study. Children with neuropathic bladder, spinal dysraphism, anatomical abnormalities (e.g., posterior urethral valves or vesicoureteral reflux), and bowel elimination problems (e.g., constipation or encopresis) were excluded. Patients who had had or were being treated for primary monosymptomatic nocturnal enuresis, patients with urinary tract infection, patients who were taking other anticholinergics or drug with anticholinergic-like effects, patients with the kidney, liver, intestine, cardiovascular system and psychological problems and patients with dysfunctional voiding in emptying phase were also excluded from this study. Patients who were allergic to or contraindicated to anticholinergic agents were also excluded.
Definitions in terminology
According to the International Continence Society and the International Children's Continence Society (2, 4), OAB was defined as urgency with or without urge incontinence, usually with increased daytime frequency and nocturia. Urinary incontinence was defined as the involuntary loss of urine, objectively demonstrable, and constituting a social or hygienic problem (11). UUI was defined as the complaint of involuntary leakage accompanied by or immediately preceded by urgeny (3). Clinical improvement of urgency and UUI was defined as the reply of "no urgency" and "90% or over improved" to the question about symptom severity after anticholinergic treatment from patients or their parents, respectively.
Statistical analyses
Statistical analyses were performed with SPSS for Windows version 18.0 (SPSS, Chicago, IL, USA). A paired t-test was used to compare the parameters in the pretreatment and posttreatment of anticholinergics. Descriptive statistics are expressed as mean±standard deviation. The chi-square test, Fisher exact test and ANOVA were used to complete the statistical determinations. All statistical analyses were 2-sided and P<0.05 was considered statistically significant.
Ethics statement
This study was approved by the institutional review board of our institution (UUH IRB no. 2012086). Clinical data were obtained by the retrospective review of the medical records of all of the patients. Therefore, the institutional review board permitted this study without requiring informed consent.
RESULTS
Based on the 3-day bladder diaries, clinical efficacy were evaluated on an exploratory basis. Among 326 children with lower urinary tract symptoms, 243 children (74.5%) had urgency symptom and 158 children (48.5%) had UUI.
On global questionnaire, 190 (78.2%) of 243 children with urgency showed clinical improvement (no urgency) after anticholinergic treatment. UUI was improved (90% or over improved) in 125 (79.1%) of 158 children (Table 2). There were no significant differences in improvement of urgency or UUI according to the types of anticholinergics (P=0.139). UUI episodes per week had significantly decreased from 1.9±3.1 to 0.4±1.5 times (P<0.001).
Table 2.
Efficacy of anticholinergics
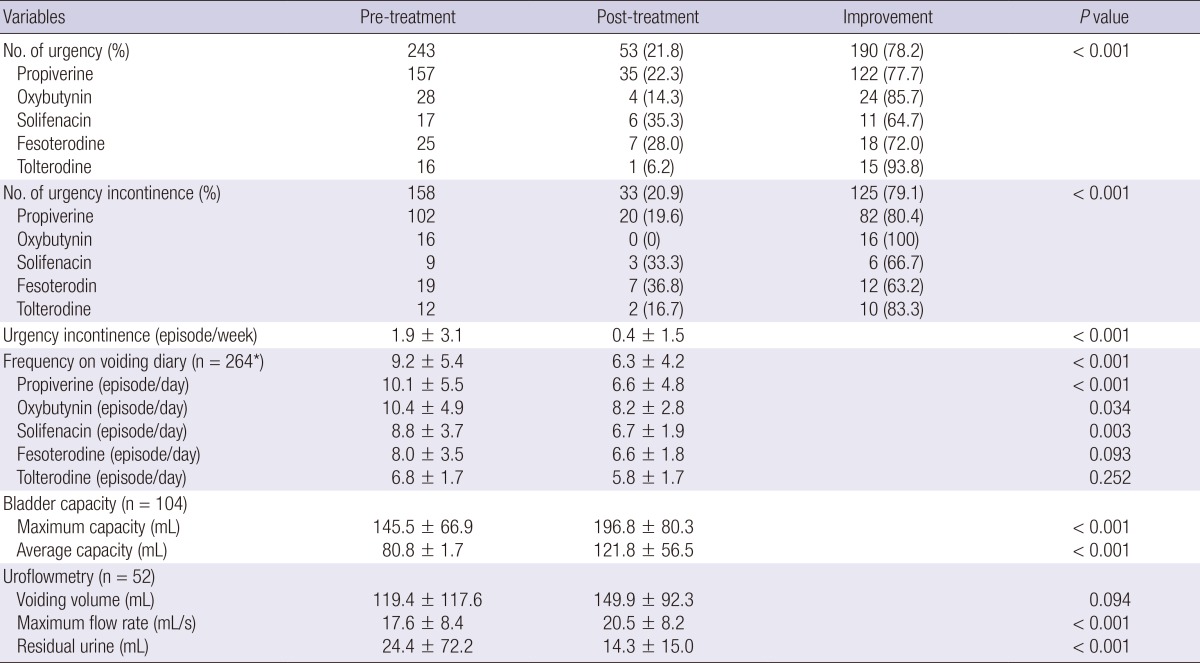
*One hundred and four children had frequency volume diaries before and after treatment. 160 children had only frequency check without volume information before or after treatment.
The mean voiding frequency during daytime was decreased from 9.2±5.4 to 6.3±4.2 times (P<0.001). The numbers of frequency episodes during daytime were all improved in 5 types of the drug. There were no significant differences in improvement of frequency according to the types of anticholinergics (P=0.207).
Both pretreatment and posttreatment 3-day voiding diaries were available in 104 children (mean age; 7.2±2.7 yr). The maximum and average bladder capacity were increased from 145.5±66.9 to 196.8±80.3 mL and from 80.8±39.6 to 121.8±56.5 mL, respectively (P<0.001).
Analysis of uroflowmetry was available in 52 children. The maximum flow rate was increased from 17.6±8.4 to 20.5±8.2 mL/sec (P<0.001), whereas post-voiding residual volume was decreased from 24.4 to 14.3 mL (P<0.001). The mean voiding volume was increased from 119.4 to 149.9 mL and it is statistically insignificant (P=0.094).
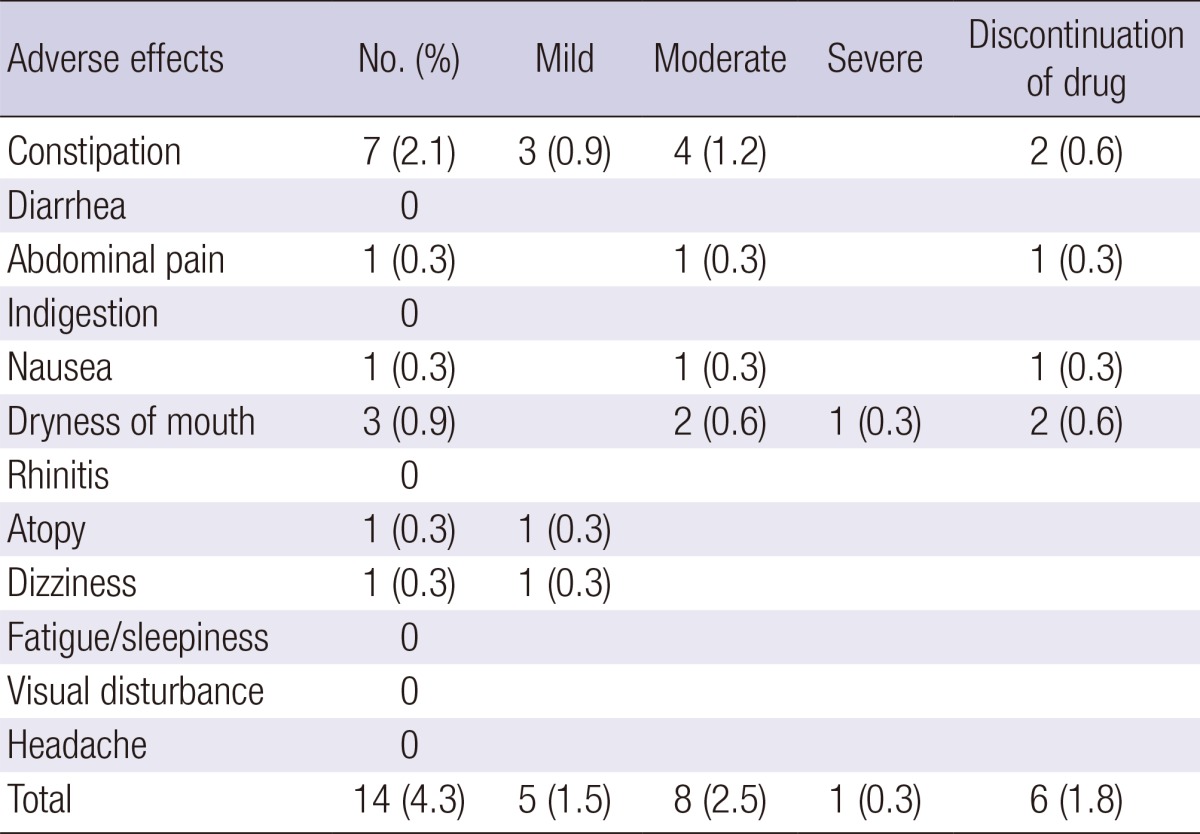
Adverse effects occurred in 14 (4.3%) children, including constipation (n=7), thirsty (n=3), abdominal pain (n=1), nausea (n=1), dizziness (n=1), and atopy (n=1). Six children (1.8%) stopped medication due to the adverse effects (Table 3).
Table 3.
Adverse effects
DISCUSSION
OAB can have considerable impact on health-related quality of life (12). For adult OAB patients, various antimuscarinic drugs such as oxybutynin, propiverine, tolterodine, trospium chloride, and solifenacin have been introduced and are widely used with proven efficacy and stability. Pediatric OAB patients and their parents also experience difficulty in life. Not only can symptoms affect daily activities and productivity, but patients often have to deal with embarrassment and anxiety. The prevalence of OAB in Korean children between 5-13 yr old is reported to be 16.59% which is relatively high although it is lower than that of adults (13). While treatment is necessary in these patients, the treatment failure is substantial and anticholinergic treatment efficacy varies from 65 to 75% (14). Therefore, various anticholingergic drugs as the first or secondary treatments should also be needed for pediatric OAB patients like adult OAB patients. Through the current study, many urologic or pediatric doctors come out into the use various anticholinergics in practice. Propiverine (58%) was the most favorite anticholinergic agent in Korean pediatric doctors. Oxybutynin was still the second most used drug due to Korean national insurance. Half dose (10 mg) and old distribution in adult OAB patients may make propiverine the most favorite drug.
Currently, oxybutynin chloride is the only FDA-approved antimuscarinic drug licensed for use in pediatric patients in Korea. Moreover, oxybutynin extended release (ER) rather than oxybutynin immediate release (IR) is widely administered because of fewer adverse effects. Oxybutynin ER is only available in tablet form, which is difficult for infants and toddlers to swallow (15). Although several anticholinergics should be clinically used in the management of OAB for pediatric patients refractory to oxybutynin, use of antimuscarinic medications for treatment of OAB has not been studied as extensively in children as it has in adults. Therefore, it is very important that clinicians understand the efficacy and safety of various anticholinergics, which inevitably could be used in clinical practice. For this reason more studies about safety and tolerability of various anticholinergics for pediatric OAB patients are needed. Our study contains important information and clinical results in the current situations. In the current study, 78.2% of 243 children with urgency showed clinical improvement (no urgency) after anticholinergic treatment. UUI was improved (90% or over improved) in 79.1% of 158 children. These response rates are similar to those of previous studies (6, 15). There were no significant differences in improvement of urgency or UUI according to the types of anticholinergics. UUI episodes per week had significantly decreased from 1.9±3.1 to 0.4±1.5 times (P<0.001).
The Korean Children's Continence and Enuresis Society reported maximum bladder capacity of healthy 219 children, aged 3 to 13 yr without history of voiding disorder was 232.7±84.6 mL (range, 161-301 mL) (16). In the current study, the maximum bladder capacity was significantly increased from 145.5±66.9 to 196.8±80.3 mL. Although symptom improved in current OAB patients and the age of healthy population was younger than that of current study, the maximum bladder capacity after anticholinergic treatment was still smaller than that of healthy population.
Adverse effects were reported in only 14 patients, and one patients experienced severe dry mouth and the medication was stopped. The medication was discontinued due to moderate symptoms in 5 patients. Most of adverse effects in the current study were less frequent than those in adult OAB patients (15, 17).
Our study has several limitations. First, it was an uncontrolled retrospective study. The potential for bias existed in the patient selection because children with a relatively good compliance with medication were included. Second, we included various anticholinergic drugs with uncontrolled drug dosage. Third, we do not know whether each patient underwent standard urotherapy or not. Standard urotherapy itself might have improved OAB symptoms. Finally, the reported complications may be relatively lower than actual occurrence because it is possible that all the complications might not have written on the medical records. In reality, it is unlikely for doctors to ask the patients and their parents about adverse effects in detail unless the study is conducted as a planned prospective study. Despite these limitations, our study results contain important and practical information for anticholinergic drug treatment in children with OAB. According to the current study results, anticholinergics have a good efficacy and are well tolerated for children suffering from OAB. On the basis of our study result, we hope that effectiveness and safety of antimuscarinics for pediatric OAB patients are clearly demonstrated by large-scale, prospective, and randomized studies in the future.
Footnotes
The authors have no conflicts of interest to disclose.
References
- 1.Sureshkumar P, Craig JC, Roy LP, Knight JF. Daytime urinary incontinence in primary school children: a population-based survey. J Pediatr. 2000;137:814–818. doi: 10.1067/mpd.2000.109196. [DOI] [PubMed] [Google Scholar]
- 2.Abrams P, Cardozo L, Fall M, Griffiths D, Rosier P, Ulmsten U, Van Kerrebroeck P, Victor A, Wein A Standardisation Sub-Committee of the International Continence Society. The standardisation of terminology of lower urinary tract function: report from the Standardisation Sub-committee of the International Continence Society. Neurourol Urodyn. 2002;21:167–178. doi: 10.1002/nau.10052. [DOI] [PubMed] [Google Scholar]
- 3.Abrams P, Andersson KE, Birder L, Brubaker L, Cardozo L, Chapple C, Cottenden A, Davila W, de Ridder D, Dmochowski R, et al. Fourth International Consultation on Incontinence Recommendations of the International Scientific Committee: Evaluation and treatment of urinary incontinence, pelvic organ prolapse, and fecal incontinence. Neurourol Urodyn. 2010;29:213–240. doi: 10.1002/nau.20870. [DOI] [PubMed] [Google Scholar]
- 4.Nevéus T, von Gontard A, Hoebeke P, Hjälmås K, Bauer S, Bower W, Jørgensen TM, Rittig S, Walle JV, Yeung CK, et al. The standardization of terminology of lower urinary tract function in children and adolescents: report from the Standardisation Committee of the International Children's Continence Society. J Urol. 2006;176:314–324. doi: 10.1016/S0022-5347(06)00305-3. [DOI] [PubMed] [Google Scholar]
- 5.Zinner N, Susset J, Gittelman M, Arguinzoniz M, Rekeda L, Haab F. Efficacy, tolerability and safety of darifenacin, an M(3) selective receptor antagonist: an investigation of warning time in patients with OAB. Int J Clin Pract. 2006;60:119–126. doi: 10.1111/j.1368-5031.2005.00770.x. [DOI] [PubMed] [Google Scholar]
- 6.MacDiarmid SA. How to choose the initial drug treatment for overactive bladder. Curr Urol Rep. 2007;8:364–369. doi: 10.1007/s11934-007-0032-6. [DOI] [PubMed] [Google Scholar]
- 7.Reddy PP, Borgstein NG, Nijman RJ, Ellsworth PI. Long-term efficacy and safety of tolterodine in children with neurogenic detrusor overactivity. J Pediatr Urol. 2008;4:428–433. doi: 10.1016/j.jpurol.2008.05.003. [DOI] [PubMed] [Google Scholar]
- 8.Alloussi S, Mürtz G, Braun R, Gerhardt U, Heinrich M, Hellmis E, Horn W, Marschall-Kehrel D, Niklas K, Raabe M, et al. Efficacy, tolerability and safety of propiverine hydrochloride in comparison to oxybutynin in children with urge incontinence due to overactive bladder: Results of a multicentre observational cohort study. BJU Int. 2010;106:550–556. doi: 10.1111/j.1464-410X.2009.09129.x. [DOI] [PubMed] [Google Scholar]
- 9.Bolduc S, Moore K, Nadeau G, Lebel S, Lamontagne P, Hamel M. Prospective open label study of solifenacin for overactive bladder in children. J Urol. 2010;184:1668–1673. doi: 10.1016/j.juro.2010.03.124. [DOI] [PubMed] [Google Scholar]
- 10.Schröder A, Thüroff JW. New strategies for medical management of overactive bladder in children. Curr Opin Urol. 2010;20:313–317. doi: 10.1097/MOU.0b013e32833aa185. [DOI] [PubMed] [Google Scholar]
- 11.Nørgaard JP, van Gool JD, Hjälmås K, Djurhuus JC, Hellström AL. Standardization and definitions in lower urinary tract dysfunction in children. International Children's Continence Society. Br J Urol. 1998;81:1–16. doi: 10.1046/j.1464-410x.1998.00025.x. [DOI] [PubMed] [Google Scholar]
- 12.Cardozo L, Thorpe A, Warner J, Sidhu M. The cost-effectiveness of solifenacin vs fesoterodine, oxybutynin immediate-release, propiverine, tolterodine extended-release and tolterodine immediate-release in the treatment of patients with overactive bladder in the UK National Health Service. BJU Int. 2010;106:506–514. doi: 10.1111/j.1464-410X.2009.09160.x. [DOI] [PubMed] [Google Scholar]
- 13.Chung JM, Lee SD, Kang DI, Kwon DD, Kim KS, Kim SY, Kim HG, Moon du G, Park KH, Park YH, et al. Prevalence and associated factors of overactive bladder in Korean children 5-13 years old: a nationwide multicenter study. Urology. 2009;73:63–67. doi: 10.1016/j.urology.2008.06.063. discussion 68-9. [DOI] [PubMed] [Google Scholar]
- 14.MacDiarmid SA. Maximizing anticholinergic therapy for overactive bladder: has the ceiling been reached? BJU Int. 2007;99:8–12. doi: 10.1111/j.1464-410X.2007.06881.x. [DOI] [PubMed] [Google Scholar]
- 15.Kim WJ, Lee DG, Lee SW, Lee YK, Lee JS, Park KH, Baek M. Efficacy and safety of propiverine in children with overactive bladder. Korean J Urol. 2012;53:275–279. doi: 10.4111/kju.2012.53.4.275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kim SO, Kim KD, Kim YS, Kim JM, Moon du G, Park S, Lee SD, Chung JM, Cho WY Korean Children's Continence and Enuresis Society. Evaluation of maximum voided volume in Korean children by use of a 48-h frequency volume chart. BJU Int. 2012;110:597–600. doi: 10.1111/j.1464-410X.2011.10799.x. [DOI] [PubMed] [Google Scholar]
- 17.Wada Y, Yoshida M, Kitani K, Kikukawa H, Ichinose A, Takahashi W, Gotoh S, Inadome A, Machida J, Ueda S. Comparison of the effects of various anticholinergic drugs on human isolated urinary bladder. Arch Int Pharmacodyn Ther. 1995;330:76–89. [PubMed] [Google Scholar]


