Abstract
Taif rose (Rosa damascena trigintipetala Dieck) is a sort of damask rose, which is considered as one of the most important economic products of Taif. In this study, the authors investigated the possible cytotoxic, genotoxic, antimutagenic and anticancer effect of concrete and absolute rose oils. The results showed that both concrete and absolute rose oils were cytotoxically and genotoxically safe at a dose of 10 μg/ml when tested on cultures of normal human blood lymphocytes. Also, the results showed significant antimutagenic activity at p < 0.001 for absolute rose oil at the same dose level when tested on cultures of normal human blood lymphocytes supplemented with 300 ng/ml mitomycin C (MMC). On the other hand, concrete and absolute oils exerted a cytotoxic activity against two kinds of human cancer cell lines: HepG2 and MCF7. Concrete oil showed cytotoxic activity against HepG2 and MCF7 with a half maximal inhibitory concentration (IC50) of 16.28 and 18.09 μg/ml, respectively, whereas absolute rose oil showed its cytotoxic activity against HepG2 and MCF7 with an IC50 of 24.94 and 19.69, respectively. From this study, it is concluded that concrete and absolute rose oils are cytotoxically and genotoxically safe at a dose of 10 μg/ml when tested on cultures of normal human blood lymphocytes. In addition, absolute oil has an antimutagenic activity at the same dose. Further investigations are needed to study the activity of higher doses of both oils in vitro and in vivo in experimental animals in order to evaluate the capability of using these oils as therapeutic for treatment of some kinds of cancers.
Keywords: Mutagenic, Antimutagenic, HepG2, MCF7
Introduction
A carcinogen is a substance that is capable of causing cancer in human and animal. Carcinogenic substances can be classified into physical, biological and chemical ones. Chemical carcinogens are toxic substances, which are organ specific, target epithelial cells, and cause genetic damage (genotoxic) (Kumar et al. 2010). Today, several industrial chemicals were identified as risk factors for cancer e.g. arsenic, nickel, chromium, benzene, and polycyclic aromatic hydrocarbons, etc. In addition, the role of life style factors such as tobacco smoking and alcohol consumption in the development of cancer is evident from numerous epidemiological and experimental studies (Weinstein 1991).
To overcome the effect of these chemical carcinogens, it is recommended that our work should focus on two approaches. The first approach is to decrease the amount of these carcinogens. The second approach is the use of antimutagens and anticarcinogens in our daily life because it is the most effective procedure to prevent human cancer and genetic diseases. Dietary antioxidants are antimutagens and anticarcinogens which decrease the highly genotoxic and mutagenic effect of reactive oxygen species (ROS) and reactive nitrogen species in normal function in human (Mantena et al. 2008; Cornelli 2009). Hence, there is increasing demand for natural antioxidant molecules to replace the synthetic additives used in the food industry (Vatan et al. 2011). Therefore, a new approach is the detection of naturally occurring and biologically active substances with genoprotective and anticancer properties that can be used in pharmaceutical and food industries. In fact, plants and herbs are considered as the main sources of these genoprotective and anticancer substances. Antimutagenic activity was detected in a number of plant oils such as cape gooseberry, corn, sunflower, sea-buckthorn, rice, beech, olive, rose, wheat embryo, pumpkin seeds, etc. (Agabeyli and Mirzazadeh 2011).
Roses are important ornamental plants and have been referred to as the queen of flowers. Over 150 rose species and more than 2,000 cultivars have been registered and are spread in Europe, Asia, Middle East, and North America (Cai et al. 2005). Members of the Rosaceae family have long been used in perfumes, cosmetics, foods, and for medicinal purposes. The Taif rose (Rosa damascena trigintipetala Dieck) is a sort of damask rose which is considered as one of the most important economic products of Taif. The essential oil of Taif rose has an excellent reputation as perfume. In this study, the authors investigated the possible cytotoxic, genotoxic, antimutagenic and anticancer effect of concrete and absolute oils of Taif rose. In addition, major components of Taif concrete and absolute rose oils have been analyzed using GC–MS spectroscopy technique.
Materials and methods
Collection of roses and preparation of concrete and absolute rose oil
The fresh roses were collected from a rose farm (April 2011) in the Taif governorate. The green parts of roses were removed and the remaining was cut into small pieces. The fresh cut petals of roses (250 g) were defatted by soaking in 1,500 ml n-hexane (Sigma-Aldrich, St. Louis, MO, USA) for 1 week at room temperature with shaking from time to time followed by filtration. The n-hexane was removed under vacuum yielding rose concrete (soft as a wax material with rose-like odour). This extraction process was repeated four times. The absolute rose oil was prepared by stirring the concrete oil with 20-fold weight of ethyl alcohol (Sigma-Aldrich). The mixture was cooled at −20 °C for 6 h followed by filtration. The alcoholic solution was evaporated under vacuum resulting in absolute rose oil (semiliquid material also with rose-like odour). The process was repeated four times. The two oils were stored at −20 °C in brown glass vials.
Viability assay
Preheparinized syringe was used to collect 10 ml of venous blood from healthy, non-smoking and non-alcoholic persons who were tested to be free of viruses and common diseases. Lymphocytes were separated according to McFee et al. (1997) and counted. Cells were cultured at a concentration of one million/ml complete medium. Complete medium was set up by adding 4 ml of RPMI-1640 with l-glutamine (Biochrom AG, Berlin, Germany), 1 ml fetal calf serum (Biochrom AG), 100 μl penicillin/streptomycin (100×) (Biochrom AG), 100 μl antimycotic (100×) (Biochrom AG) and 25 μl preservative heparin (5,000 U/ml) (Biochrom AG) per culture tube. Culture tubes were divided into three groups. The first and second groups of culture tubes were treated with different doses of concrete and absolute oils, respectively (5, 10, 50, 75 and 100 μg/ml). The third group of culture tubes was used as untreated control. Lymphocytes were incubated at 37 °C for 72 h. Culture tubes were centrifuged at 115×g for 10 min (Mikro 22R, Andreas Hettich GmbH and Co. KG, Tuttlingen, Germany). The concentration of lymphocytes in the sample was adjusted to be less than 104 cells/μl (1010/l) using phosphate buffer saline (Biochrom AG). Twenty microliter of 7-amino-actinomycin D (7-AAD) dye solution (Immunotech S.A., Beckman Coulter Company, Marseille, France) and 100 μl of the test sample were added to each test tube and mixed well. Test tubes were incubated for 15–20 min at room temperature (18–25 °C), protected from light. Preparations were analyzed within 1 h on Beckman Coulter EPICS XL flow cytometer (Beckman Coulter Company, Miami, FL, USA) using 620 or 675 nm “band pass” filter (Schmid et al. 1992; Fetterhoff et al. 1993). The relative viability percent was calculated according to the following equation:
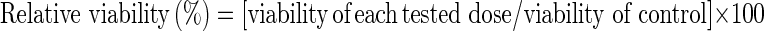 |
Genotoxicity/antimutagenicity assays
Heparinized peripheral blood obtained from ten healthy non-smoking and non-alcoholic donors (4 females and 6 males) were used in this experiment. Lymphocytes were prepared as mentioned in viability assay and cultured in the same complete medium supplemented with 96 μg/ml phytohemagglutinin (PHA-L) (Biochrom AG). Lymphocytes of each donor were divided into six culture tubes, the first was left as control, the second was treated with 10 μg/ml rose concrete oil, the third was treated with 10 μg/ml rose absolute oil, the fourth was treated with 300 ng/ml mitomycin C (MMC) (Sigma-Aldrich) as a mutagenic agent for induction of oxidative and chromosomal damage, the fifth was treated with 10 μg/ml rose concrete oil and 300 ng/ml MMC and finally the last tube was treated with 10 μg/ml rose absolute oil and 300 ng/ml MMC. All culture tubes were incubated at 37 °C for 72 h. Metaphases were obtained by adding colcemid (0.4 μg/ml) (Biochrom AG) 2 h prior to harvesting. Cells were centrifuged at 115×g for 10 min, then cells were suspended in 5 ml of pre-warmed (37 °C) 0.075 M KCl (hypotonic solution) (Merck Chemicals, Darmstadt, Germany) and incubated at 37 °C for 30 min. Cells were centrifuged at 115×g for 10 min, then suspended in 5 ml of fixative (methanol:glacial acetic acid; 3:1) (Merck) and were incubated at room temperature for 10 min. The supernatant was removed after centrifugation for 10 min at 115×g, the cells were suspended in another fixative, and the step was repeated three times. To prepare slides, 2–3 drops of fixed cell suspension were dropped on clean slides, stored in cold ethanol and then they were flame dried. Slides were stained in 4 % Giemsa solution (Sigma-Aldrich) in phosphate buffer (pH 6.8) for 7–10 min and microscopically screened (Savage 1975; Pereira et al. 1988).
For each culture, 25 metaphases were examined microscopically for chromosomal aberrations and the ratio of metaphase to interphase nuclei in 1,000 cells for determining the mitotic index. Statistical analysis was performed using Student t test.
Anticancer activity
The effect of concrete and absolute oils of the Taif rose were investigated in vitro on two kinds of human cancer cell lines: liver carcinoma cell line (HepG2) and breast carcinoma cell line (MCF7) [Obtained frozen in liquid nitrogen (−180 °C) from the American Type Culture Collection (Manassas, VA, USA) and were maintained in the National Cancer Institute, Cairo, Egypt, by serial sub-culturing] using Sulphorhodamine-B (SRB) assay as described by Skehan et al. (1990). The SRB assay is a colorimetric method and cell numbers are indirectly estimated by staining total cellular protein with the SRB dye (Sigma-Aldrich). This dye is a bright pink aminoxanthrene dye with two sulphonic groups. It is a protein stain that binds to the amino groups of intracellular proteins under moderate acidic conditions to provide a sensitive index of cellular protein content. Cells were seeded in 96-well microtiter plates at a concentration of 5 × 104–105 cell/well in a fresh medium and left to attach to the plates for 24 h. Wells were prepared for each individual sample and incubated for 48 h at 37 °C in 5 % CO2. After 24 h, cells were incubated with the appropriate different concentrations of the fraction under test (0, 5, 12.5, 25 and 50 μg/ml), completed to total of 200 μl volume/well using fresh medium and incubation was continued for 24, 48 and 72 h. Following 24, 48 and 72 h treatment, the cells were fixed with 50 μl cold 50 % trichloroacetic acid (Sigma-Aldrich) for 1 h at 4 °C. Wells were washed 5 times with distilled water and stained for 30 min at room temperature with 50 μl 0.4 % SRB dissolved in 1 % acetic acid (Sigma-Aldrich). The wells were then washed 4 times with 1 % acetic acid. The plates were air-dried and the dye was solubilized with 100 μl/well of 10 mM Tris base (pH 10.5) for 5 min on a shaker (Orbital shaker OS 20, Boeco, Hamburg, Germany) at 57×g. The optical density (OD) of each well was measured spectrophotometrically at 564 nm with an ELIZA microplate reader (Metertech Σ 960, Metertech Inc., Taipei, Taiwan). The mean background absorbances were automatically subtracted and mean values of each sample concentration were calculated.
The experiment was repeated 3 times for each cell line. The percentage of cell survival was calculated according to the following equation:
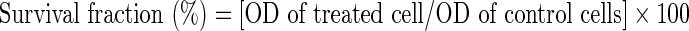 |
According to the National Cancer Institute guideline an extract and/or a compound with IC50 values <20 μg/ml is considered active (Boik 2001).
GC–MS analysis of concrete and absolute rose oil
The analysis of the samples was performed using gas chromatograph (GC, Model CP 3800, Varian, Walnut Creek, CA, USA) coupled with a mass spectrometer (MS, Model Saturn 2200, Varian) and auto sampler (Model Combi Pal, Varian) system. The separation was done using a VF-5 fused silica capillary column (30 m × 0.25 i.d. mm film thickness 0.25 μm, Varian). For MS detector, electron impact (EI) ionization system with ionization energy of 70 eV was used. Helium gas was used as a carrier gas at a constant low rate of 1 ml/min. Injector and mass transfer line temperature were set at 250 and 300 °C, respectively. The oven temperature was programmed for 5 min at 60 °C, 60–290 °C at 6 °C/min and held for 5 min at 290 °C, solvent delay time 3 min. The injection of the samples was carried out with the auto-sampler for 1 μl with a split ratio 1/20. The conditions of analysis and specification of the instrument were optimized for a better separation and resolution. Identification of components of concrete and absolute oils was based on matching with Wiley and NIST electronic library.
Results
Viability assay
The results showed a decrease of the percent of relative viability with increasing the dose for both concrete and absolute oils (Table 1). Also, the results showed that concrete oil was less toxic than absolute oil. The doses of 5 and 10 μg/ml of both oils seemed to be non-toxic (depending closely on the same results between treated groups and normal control groups), so we chose 10 μg/ml as a highly safe dose for using in the antimutagenic test.
Table 1.
Relative viability percent of human peripheral blood lymphocytes in groups treated with different doses of concrete and absolute Taif rose oils
| Agent | Dose (μg/ml) | ||||
|---|---|---|---|---|---|
| 5 | 10 | 50 | 75 | 100 | |
| Concrete rose oil | 99.3 | 96.6 | 82.1 | 76.2 | 61.9 |
| Absolute rose oil | 100 | 98.8 | 77.2 | 59 | 42.4 |
Genotoxcicity/antimutagenicity assays
The mitotic index indicated proliferating activity of cultured cells. The mitotic index (mitotic index = number of metaphases per 1,000 observed cells) was determined in all groups. The exposure to 300 ng/ml MMC for 72 h showed high inhibition of mitotic activity at p < 0.001. Concrete and absolute rose oils (10 μg/ml) did not show differences in the mitotic activity in relation to the non-treated group. Adding 10 μg/ml of concrete and absolute oils to MMC culture showed no improvement in the mitotic activity in case of concrete and a small improvement in the mitotic activity in case of absolute oil (Table 2). Complete inhibition of mitotic activity appeared in culture tubes from three individuals treated with MMC, concrete rose oil + MMC and absolute rose oil + MMC, so the samples of the remaining seven individuals were only used for examining chromosomal aberrations as shown in Table 3.
Table 2.
The effect of concrete and absolute rose oils on mitotic indices of peripheral blood lymphocytes of both normal control and MMC treated cultures
| Group | No. of individuals | MI (X ± SE) |
|---|---|---|
| Normal control | 10 | 22.2 ± 4.5 |
| Concrete rose oil | 10 | 18.0 ± 3.7 |
| Absolute rose oil | 10 | 25.8 ± 4.19 |
| MMC | 10 | 2.9 ± 1.05*** |
| Concrete rose oil + MMC | 10 | 2.6 ± 1.02*** |
| Absolute rose oil + MMC | 10 | 6.1 ± 1.97** |
MI no. of metaphase/1,000 cell
MMC mitomycin C
** p < 0.01, *** p < 0.001 in relation to normal negative control group
Table 3.
The effect of concrete and absolute oils on total chromosomal aberrations in human peripheral blood lymphocytes of both normal control and MMC treated cultures
| Group | No. of individuals | No. of total chromosomal aberations/cell (X ± SE) with gaps | No. of total chromosomal aberations/cell (X ± SE) without gaps |
|---|---|---|---|
| Normal control | 10 | 0.11 ± 0.02 | 0.05 ± 0.01 |
| Concrete rose oil | 10 | 0.12 ± 0.03 | 0.04 ± 0.02 |
| Absolute rose oil | 10 | 0.05 ± 0.01 | 0.03 ± 0.01 |
| MMC | 7 | 3.49 ± 0.26*** | 2.98 ± 0.18*** |
| Concrete rose oil + MMC | 7 | 3.04 ± 0.12*** | 2.55 ± 0.11*** |
| Absolute rose oil + MMC | 7 | 1.67 ± 0.29*** ••• | 1.54 ± 0.25*** ••• |
MMC mitomycin C
*** p < 0.001 in relation to the normal negative control group
••• p < 0.001 in relation to the MMC positive control group
The number of chromosomal aberrations induced in all groups is shown in Table 3. The frequencies of different types of chromosomal aberrations were counted in 25 cells per individual for each group. In MMC treated groups, few individuals did not complete the 25 metaphases, and then we calculated the mean of aberrations per the number of counted cells. The results showed no increase in chromosomal aberrations in groups treated with concrete and absolute oils. All groups treated with MMC showed a significant increase in the frequencies of most types of chromosomal aberration and in the frequencies of total chromosomal aberrations with or without gaps. Adding absolute oil to MMC caused a significant decrease in the frequencies of most types of chromosomal aberrations and in the frequencies of total chromosomal aberrations with or without gaps. Each of the treated and control groups showed only one polyploid cell and one pulverized cell, so these two parameters were not suitable for statistical analysis and are not presented in Table 4. Gaps, breaks, deletions and radial forms were the most frequent types of chromosomal aberrations found in MMC treated groups. Several representative metaphase figures revealing different types of aberrations are shown in Fig. 1.
Table 4.
The effect of concrete and absolute oils on different types of structural chromosomal aberrations in peripheral blood lymphocytes of both normal control and MMC treated cultures
| Group | No. of individuals | Number of types of aberration/cell (X ± DS) | |||||
|---|---|---|---|---|---|---|---|
| Gap | Isochromatid gap | Break | Isochromatid break | Deletion | Fragment | ||
| Normal control | 10 | 0.056 ± 0.018 | 0.004 ± 0.004 | 0.04 ± 0.008 | 0.004 ± 0.004 | 0 ± 0 | 0 ± 0 |
| Concrete rose oil | 10 | 0.064 ± 0.023 | 0.008 ± 0.005 | 0.008 ± 0.005** | 0 ± 0 | 0.016 ± 0.012 | 0 ± 0 |
| Absolute rose oil | 10 | 0.012 ± 0.009* | 0.004 ± 0.004 | 0.008 ± 0.005** | 0.008 ± 0.005 | 0.004 ± 0.004 | 0 ± 0 |
| MMC | 7 | 0.469 ± 0.096*** | 0.034 ± 0.02 | 1.999 ± 0.108*** | 0.06 ± 0.036 | 0.29 ± 0.06*** | 0.12 ± 0.05* |
| Concrete rose oil + MMC | 7 | 0.441 ± 0.08*** | 0.044 ± 0.016* | 1.737 ± 0.08*** | 0.024 ± 0.012 | 0.041 ± 0.018* •• | 0.047 ± 0.016** |
| Absolute rose oil + MMC | 7 | 0.109 ± 0.04 •• | 0.039 ± 0.015* | 1.106 ± 0.199*** •• | 0.033 ± 0.021 | 0.05 ± 0.022* •• | 0.017 ± 0.012 |
| Group | Number of types of aberration/cell (X ± DS) | ||||||
|---|---|---|---|---|---|---|---|
| Minute | Acentric fragment | Double minute | Radial form | Complex rearrangement | Centromeric attenuation | ||
| Normal control | 0 ± 0 | 0 ± 0 | 0 ± 0 | 0 ± 0 | 0 ± 0 | 0.008 ± 0.008 | |
| Concrete rose oil | 0 ± 0 | 0.004 ± 0.004 | 0 ± 0 | 0 ± 0 | 0 ± 0 | 0.016 ± 0.009 | |
| Absolute rose oil | 0 ± 0 | 0.008 ± 0.005 | 0 ± 0 | 0 ± 0 | 0 ± 0 | 0.004 ± 0.004 | |
| MMC | 0.08 ± 0.03** | 0.117 ± 0.046** | 0.009 ± 0.009 | 0.266 ± 0.057*** | 0.04 ± 0.021* | 0 ± 0 | |
| Concrete rose oil + MMC | 0.047 ± 0.021* | 0.289 ± 0.021*** •• | 0.01 ± 0.01 | 0.273 ± 0.027*** | 0.06 ± 0.019** | 0.02 ± 0.02 | |
| Absolute rose oil + MMC | 0.016 ± 0.011 | 0.16 ± 0.06** | 0 ± 0 | 0.126 ± 0.038** | 0.031 ± 0.019 | 0 ± 0 | |
MMC mitomycin C
•• p < 0.01 in relation to the MMC positive control group
* p < 0.05, ** p < 0.01, *** p < 0.001 in relation to the normal negative control group
Fig. 1.
a Normal metaphase plate from human peripheral blood lymphocytes without any treatment. b and c are metaphase plates from treated human peripheral blood lymphocytes showing breaks (b), fragment (f), radial form (r), acentric fragment (af), minute (m), deletion (d) and complex rearrangement (cr). Human peripheral blood lymphocytes were separated and cultured in complete medium supplemented with phytohemagglutinin incubated at 37 °C for 72 h. Lymphocytes of each donor were divided into six culture tubes, the first one was left as control and while the other tubes were treated with 10 μg/ml rose concrete oil, 10 μg/ml rose absolute oil, 300 ng/ml mitomycin C (MMC), 10 μg/ml rose concrete oil + 300 ng/ml MMC and 10 μg/ml rose absolute oil + 300 ng/ml MMC, respectively. The cultures were treated with hypotonic solution at 37 °C for 30 min, and then they were centrifuged and the pellets were fixed with methanol glacial trice. The fixed cells were dropped on clean slides, dried, stained with 4 % Giemsa solution, and microscopically screened
Anticancer agents
Concrete and absolute oils were tested for cytotoxic activity against two kinds of human cancer cell lines; liver carcinoma cell line (HepG2) and breast carcinoma cell line (MCF7). Results showed that both oils were active toward both cell lines (Fig. 2).
Fig. 2.
Cytotoxicity of concrete (a) and absolute (b) rose oils against liver carcinoma cell line (HepG2) and breast carcinoma cell line (MCF7) at different concentrations using Sulphorhodamine-B (SRB) assay. The horizontal axis corresponds to different concentrations of oil and the vertical axis corresponds to percent of survival fraction. SRB is a colorimetric assay estimating cell numbers indirectly by staining total cellular proteins with the dye. The optical density (OD) of each well was measured spectrophotometrically at 564 nm with an ELIZA microplate reader. The percentage of survival fraction was calculated according to the following equation: Survival fraction (%) = [OD of treated cell/OD of control cells] × 100
GC–MS analysis
According to the extraction process of concrete rose oil, the compounds responsible for the distinguishable odor constitute 53.90 % of the oil and the remaining (46.10 %) is a waxy material as per the GC–MS chromatogram calculation. The GC–MS showed five major aroma compounds in the concrete oil: phenyl ethanol (37.83 %), β-citronellol (8.2 %), geraniol (4.04 %), eugenol (1.48 %) and methyl eugenol (0.72 %). The analysis of GC–MS chromatogram of absolute oil showed that aroma compounds constitute 56.89 % and the waxy material 43.11 % of the oil. The major aroma compounds in absolute oil are: phenyl ethanol (33.31 %), β-citronellol (12.45 %), geraniol (6.28 %), eugenol (2.03 %) and methyl eugenol (1.05 %).
Discussion
7-Amino-actinomycin D (7-AAD) is a cytochemical probe for DNA depending on the ability of 7-AAD to insert itself between the tops of successive cytosine/guanine bases of the DNA double strand, when the interior of the cell and the nuclear chromatin are accessible. Non-viable cells can be characterized by loss of membrane integrity, so they are permeable to 7-AAD and are stained whilst viable cells retaining their membranous integrity are impermeable to 7-AAD and are unstained (Gill et al. 1975). Our results showed a dose dependent decrease in the percentage of viable cells for both oils. The results revealed that concrete oil is less toxic for normal human peripheral blood lymphocytes than absolute oil. Also, the doses 5 and 10 μg/ml for both oils were considered safe due to the close reading values between normal control and treated groups (percent of relative viability ≥95 %), so the dose of 10 μg/ml was chosen for the antimutagenic assay.
Chromosomal aberration assays are considered to be very sensitive end points for recognizing the genotoxic effects induced by chemicals (Sweify et al. 2005). The present data showed no change in the frequency of chromosomal aberrations in groups treated with concrete and absolute oils alone when compared with non-treated group. These results proved that concrete and absolute oils were not genotoxic at the tested dose (10 μg/ml).
Mitomycin C (MMC) is an antitumor antibiotic. The primary action of MMC has been considered to inhibit the deoxyribonucleic acid (DNA) synthesis by cross-linking of the complementary strand of DNA (Iyer and Szybalski 1964). This would inhibit DNA replication and probably might be the antitumor action of this drug. Despite the fact that MMC showed antitumor action, many reports proved that it may cause high frequency of chromosomal aberrations, sister chromatid exchanges and micronuclei formation (Latt 1974; Fauth et al. 2000; Yogesh and Fulzele 2009). In this study, we used this antitumor-antibiotic drug to test the possible antimutagenic activity of concrete and absolute oils. The study results of MMC in this experiment were coincided with previous reports (Fauth et al. 2000; Yogesh and Fulzele 2009; Unal et al. 2013). The group treated with MMC alone showed a significant elevation of chromosomal damage. In addition, chromatid gaps, chromatid breaks, deletions and radial forms were the most common aberrations found. Adding absolute oil to the culture caused a significant decrease in the frequency of chromosomal aberrations especially with respect to frequency of chromatid gap, chromatid break, deletions and the number of total aberrations. This indicates antimutagenic activity for rose absolute oil. In case of concrete oil, a slight improvement (decrease) in the frequency of chromosomal aberrations had been observed but did not reach significance, so we cannot consider it to have an antimutagenic activity.
Many reports proved that many essential oils have antimutagenic activity e.g. lavender, sage, Teucrium ramosissimum, origanum compactum and rose essential oils (Evandri et al. 2005; Knezevic et al. 2005; Mezzoug et al. 2007; Sghaier et al. 2010; Agabeyli and Mirzazadeh 2011). As mentioned before the addition of rose concrete oils showed some improvement in the frequency of chromosomal aberrations which is less than for rose absolute oil. This may be due to the higher percent of the compounds responsible for aroma in absolute than concrete rose oil.
Eugenol as one of rose concrete and absolute oils constituents was reported as antimutagenic agent against the mutagenicity caused by tobacco and benzo(a)pyrene (Bhattacharjee and Sengupta 2009; Yokota et al. 1986).
The results demonstrated that concrete and absolute oils were toxic against HepG2 and MCF7 cells. Concrete rose oil showed a more toxic effect in the HepG2 cell line (IC50 = 16.28 μg/ml) than in MCF7 (IC50 = 18.09 μg/ml). On the contrary absolute rose oil showed more toxic effects against the MCF7 cell line (IC50 = 19.69 μg/ml) than HepG2 (IC50 = 24.94 μg/ml). There are three mechanisms for the action of anticancer drugs; the first is to damage the DNA of the affected cancer cells; the second is to inhibit the synthesis of new DNA strands and preventing the cell from replicating which allows the tumor to grow; and the third is to stop mitosis or the actual splitting of the original cell into two cells that ultimately halt the progression of the cancer (Ophardt 2003). Rose oil is an essential oil and due to a number of chemical constitutes, essential oils apparently have no specific cellular targets (Hussain 2009). The cytoplasma membrane is passed through easily by essential oils that make it permeabilized and may cause its damage. Also, essential oils have the ability to coagulate the cytoplasm, hence damaging lipids and proteins (Ultee et al. 2000; Burt 2004; Hussain 2009). Injury to the cell wall and cell membrane can lead to lysis and the escape of macromolecules (Lambert et al. 2001; Oussalah et al. 2006). Essential oils are liable to stimulate depolarization in mitochondrial membranes of eukaryotic cells by decreasing the membrane potential, affect Ca2+ cycling and other ionic channels and reduce the pH gradient (Vercesi et al. 1997; Bakkali et al. 2005, 2008; Hussain 2009). The fluidity of membranes is disturbed by them, hence becoming noticeably permeable which results in leakage of radicals, cytochrome C, calcium ions and proteins as in case of oxidative stress. Permeabilization of the outer and inner mitochondrial membranes causes cell death by necrosis and apoptosis (Armstrong 2006; Hussain 2009). Although reduced forms of phytophenolics are powerful antioxidants, the phenoxyl radical produced through antioxidation reactions is a potential prooxidant which may exhibit cytotoxic activity when the life time of a radical is prolonged (Sakihama et al. 2002; Burt 2004; Barbehenn et al. 2005). In our study phenyl ethanol, one of the phenolic constitutes, comprised of about 33 and 37 % of total volume of the absolute and concrete oil, respectively. Phenyl ethanol, which has a rose-like aroma, is an important fragrance in the cosmetic industry and possesses organoleptic characteristics that contribute to the quality of beverage and foods. It has been estimated that approximately 700,000 kg of phenyl ethanol is consumed annually as a food component (Mo and Sung 2007). The antibacterial properties of some essential oils were attributed to their high phenyl ethanol content (Boskabady et al. 2011; Singh et al. 2012). To the best of our knowledge, there is no published report about the anticancer properties of phenyl ethanol. Then according to the high level of phenyl ethanol in the oil, it may be one of the components responsible for the anticancer properties of this oil.
Geraniol—one of rose absolute and concrete oil constituents, a monoterpene alcohol—elicited a dramatic reduction in the amounts of thymidylate synthase (TS) and thymidine kinase (TK) expression in colon cancer cells (Carnesecchi et al. 2004). These two enzymes are involved in 5-fluorouracil (5-Fu) toxicity, in that a decrease in these enzymes is related to enhanced 5-Fu cytotoxicity (Mans et al. 1999). Geraniol lowered the resistance of cancer cells to 5-Fu, thus potentiate the inhibition of tumor growth by the drug, and increased the survival time of nude mice grafts with the human colorectal tumor cell TC118. Geraniol acts on at least two different targets involved in the resistance of cancer cells to 5-Fu: it increases cell membrane permeability leading to enhanced uptake of 5-Fu by colon cancer cells and causes a significant change in the resulting potential and cell membrane polarization, which may trigger modifications of membrane bound protein activity and alteration in intracellular signaling pathways (Carnesecchi et al. 2002a, b). Also, the growth inhibition effect of eugenol (another constituent of concrete and absolute rose oils) has been observed in the hepatoma cell line HepG2 and human promyelocytic leukemia cell HL-60. In leukemia cells, eugenol treatment induced cytotoxicity and internucleosomal DNA fragmentation (Okada et al. 2005). Eugenol transduced the apoptotic signal via reactive oxygen species generation, thereby inducing mitochondrial permeability transition, reducing the level of the anti-apoptotic protein bcl-2, and inducing cytochrome C release to the cytosol and subsequent apoptotic cell death (Yoo et al. 2005). Eugenol inhibited the proliferation of HT-29 cells and the mRNA expression of CoX-2 (Vidhya and Devaraj 2011). Also, the methylated form of eugenol (methyl eugenol) was found to inhibit melanoma cells growth by about 20–40 % (Pisano et al. 2007). Review of literature showed that essential oils including the previous compounds in other investigated plants showed antimutagenic and anticancer activities (Edris 2007; Hussain 2009; Adorjan and Buchbauer 2010).
Conclusion
In conclusion, the data presented here showed that the concrete and absolute oils have a cytotoxic effect toward normal human blood lymphocytes which is dose dependent. In addition, at a dose of 10 μg/ml both oils are cytotoxically safe and have no genotoxic effect. This study implies that pretreatment of rose absolute oil (10 μg/ml) has a strong inhibitory role against mutagenic action of MMC (300 ng/ml) which is an anticancer/antibiotic drug causing secondary genetic damage. On the other hand, rose concrete oil shows antimutagenic activity at the same dose level but without statistical significance. Both rose concrete and absolute oils showed anticancer activity against HepG2 and MCF7 cell with an IC50 nearly falling within the NCI criteria (IC50 < 20 μg/ml), thus these agents can be considered as having a promising anticancer potential. Further investigations are needed to study the activity of higher doses of both oils in vitro and in vivo in experimental animals to evaluating the capability to use these oils as therapeutic for the treatment of different kinds of cancers .
Acknowledgments
The authors are very grateful to Taif University and to the supporter of Chair of Research and Development Studied for Taif Rose, Taif University, Kingdom of Saudi Arabia for supporting this work. The authors wish to thank all members in Cytogenetic, Tissue Culture and Flow Cytometry Units, Main Laboratory, Cairo University hospitals for their cooperation and support.
Conflict of interest
There is no conflict of interest in this manuscript.
References
- Adorjan B, Buchbauer G. Biological properties of essential oils: an updated review. Flavour Fragr J. 2010;25:407–426. doi: 10.1002/ffj.2024. [DOI] [Google Scholar]
- Agabeyli RA, Mirzazadeh GG. Study of the genoprotective properties of oils from fruits and leaves of Fagus orientalis lipsky. Cytol Genet. 2011;45:220–224. doi: 10.3103/S0095452711040025. [DOI] [PubMed] [Google Scholar]
- Armstrong JS. Mitochondrial membrane permeabilization: the sine qua non for cell death. BioEssays. 2006;28:253–260. doi: 10.1002/bies.20370. [DOI] [PubMed] [Google Scholar]
- Bakkali F, Averbeck S, Averbeck D, Zhiri A, Idaomar M. Cytotoxicity and gene induction by some essential oils in the yeast Saccharomyces cerevisiae. Mutat Res. 2005;585:1–13. doi: 10.1016/j.mrgentox.2005.03.013. [DOI] [PubMed] [Google Scholar]
- Bakkali F, Averbeck S, Averbeck D, Idaomar M. Biological effects of essential oils: a review. Food Chem Toxicol. 2008;46:446–475. doi: 10.1016/j.fct.2007.09.106. [DOI] [PubMed] [Google Scholar]
- Barbehenn R, Cheek S, Gasperut A, Lister E, Maben R. Phenolic compounds in red oak and sugar maple leaves have prooxidant activities in the midgut fluids of Malacosoma disstria and Orgyia leucostigma caterpillars. J Chem Ecol. 2005;31:969–988. doi: 10.1007/s10886-005-4242-4. [DOI] [PubMed] [Google Scholar]
- Bhattacharjee S, Sengupta A. Spices in cancer prevention: an overview. Internet J Nutr Well. 2009;7:1–8. [Google Scholar]
- Boik J (2001) Natural compounds in cancer therapy. Oregon Medical Press, Princeton, MN, USA
- Boskabady MH, Shafei MN, Saberi Z, Amini S. Pharmacological effects of Rosa damascene. Iran J Basic Med Sci. 2011;14:295–307. [PMC free article] [PubMed] [Google Scholar]
- Burt S. Essential oils: their antimicrobial properties and potential application in foods—a review. Int J Food Microbiol. 2004;94:223–253. doi: 10.1016/j.ijfoodmicro.2004.03.022. [DOI] [PubMed] [Google Scholar]
- Cai YZ, Xing J, Sun M, Zhan ZQ, Corke H. Phenolic antioxidants (hydrolysable tannins, flavonols, and anthocyanins) identified by LC-ESI-MS and MALDI-QIT-TOF MS from Rosa chinensis flowers. J Agric Food Chem. 2005;53:9940–9948. doi: 10.1021/jf052137k. [DOI] [PubMed] [Google Scholar]
- Carnesecchi S, Langley K, Exinger F, Gosse F, Raul F. Geraniol, a component of plant essential oils, sensitizes human colonic cancer cell to 5-fluorouracil treatment. J Pharmacol Exp Ther. 2002;301:625–630. doi: 10.1124/jpet.301.2.625. [DOI] [PubMed] [Google Scholar]
- Carnesecchi S, Bradaia A, Fischer B, Coelho D, Scholler-Guinard M, Gosse F, Raul F. Perturbation by geraniol of cell membrane permeability and signal transduction pathways in human colon cancer cells. J Pharmacol Exp Ther. 2002;303:711–715. doi: 10.1124/jpet.102.039263. [DOI] [PubMed] [Google Scholar]
- Carnesecchi S, Bras-Gonçalves R, Bradaia A, Zeisel M, Gossé F, Poupon MF, Raul F. Geraniol, a component of plant essential oils, modulates DNA synthesis and potentiates 5-fluorouracil efficacy on human colon tumor xenografts. Cancer Lett. 2004;215:53–59. doi: 10.1016/j.canlet.2004.06.019. [DOI] [PubMed] [Google Scholar]
- Cornelli U. Antioxidant use in nutraceuticals. Clin Dermatol. 2009;27:175–194. doi: 10.1016/j.clindermatol.2008.01.010. [DOI] [PubMed] [Google Scholar]
- Edris AE. Pharmaceutical and therapeutic potentials of essential oils and their individual volatile constituents: a review. Phytother Res. 2007;21:308–323. doi: 10.1002/ptr.2072. [DOI] [PubMed] [Google Scholar]
- Evandri MG, Battinelli L, Daniele C, Mastrangelo S, Bolle P, Mazzanti G. The antimutagenic activity of Lavandula angustifolia (lavender) essential oil in the bacterial reverse mutation assay. Food Chem Toxicol. 2005;43:1381–1387. doi: 10.1016/j.fct.2005.03.013. [DOI] [PubMed] [Google Scholar]
- Fauth E, Scherthan H, Zankl H. Chromosome painting reveals specific patterns of chromosome occurrence in MMC and diethyl stilbostrol-induced micronuclei. Mutagen. 2000;15:459–467. doi: 10.1093/mutage/15.6.459. [DOI] [PubMed] [Google Scholar]
- Fetterhoff TJ, Holland SP, Wile KJ. Fluorescent detection of non-viable cells in fixed cell preparations. Cytometry. 1993;14:27–31. [Google Scholar]
- Gill JE, Jotz MM, Young SG, Modest EJ, Sengupta SK. 7-Amino-actinomycin D as a cytochemical probe I. Spectral properties. J Histochem Cytochem. 1975;23:793–799. doi: 10.1177/23.11.1194669. [DOI] [PubMed] [Google Scholar]
- Hussain AI (2009) Characterization and biological activities of essential oils of some species of Lamiaceae (Ph.D. Thesis) Department of Chemistry and Biochemistry, Faculty of Sciences, University of Agriculture, Faisalabad, Pakistan
- Iyer VN, Szybalski W. Mitomycin and porfiromycin: chemical mechanism of activation and cross-linking of DNA. Science. 1964;145:55–58. doi: 10.1126/science.145.3627.55. [DOI] [PubMed] [Google Scholar]
- Knezevic J, Vukovic-Gacic B, Stevic T, Stanojevic J, Nikolic B, Simic D. Antimutagenic effect of essential oil of sage (Salviaofficinalis L.) and its fractions against UV-induced mutations in bacterial and yeast cells. Arch Biol Sci. 2005;57:163–172. doi: 10.2298/ABS0503163K. [DOI] [Google Scholar]
- Kumar M, Meena P, Verma S, Kumar M, Kumar A. Anti-tumor, anti-mutagenic and chemomodulatory potential of Chlorophytum borivilianum. Asian Pac J Cancer Prev. 2010;11:327–3234. [PubMed] [Google Scholar]
- Lambert RJ, Skandamis P, Coote NP, Nychas GJ. A study of the minimum inhibitory concentration and mode of action of oregano essential oil, thymol and carvacrol. J Appl Microbiol. 2001;91:453–462. doi: 10.1046/j.1365-2672.2001.01428.x. [DOI] [PubMed] [Google Scholar]
- Latt SA. Sister chromatid exchanges, indices of human chromosome damage and repair: detection by fluorescence and induction by mitomycin C. Proc Natl Acad Sci USA. 1974;71:3162–3166. doi: 10.1073/pnas.71.8.3162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mans D, Grivicich I, Peters G, Schwartsmann G. Sequence dependent growth inhibition and DNA damage formation by the irinotecan-5-fluorouracil combination in human colon carcinoma cell lines. Eur J Cancer. 1999;35:1851–1861. doi: 10.1016/S0959-8049(99)00222-1. [DOI] [PubMed] [Google Scholar]
- Mantena RK, Wijburg OL, Vindurampulle C, Bennett VR, Walduck A, Drummond GR, Davies JK, Robins RM, Strugnell RA. Reactive oxygen species are the major antibacterial against Salmonella typhimurium purine auxotrophs in the phagosome of RAW2647.7 cells. Cell Microbiol. 2008;10:1058–1073. doi: 10.1111/j.1462-5822.2007.01105.x. [DOI] [PubMed] [Google Scholar]
- McFee AF, Sayer AM, Salomaa SI, Lindholm C, Littlefield LG. Methods for improving the yield and quality of metaphase preparations for FISH probing of human lymphocyte chromosomes. Environ Mol Mutagen. 1997;29:98–104. doi: 10.1002/(SICI)1098-2280(1997)29:1<98::AID-EM13>3.0.CO;2-C. [DOI] [PubMed] [Google Scholar]
- Mezzoug N, Elhadri A, Dallouh A, Amkiss S, Skali NS, Abrini J, Zhiri A, Baudoux D, Diallo B, Jaziri M, Idaomar M. Investigation of the mutagenic and antimutagenic effects of Origanum compactum essential oil and some of its constituents. Mutat Res. 2007;629:100–110. doi: 10.1016/j.mrgentox.2007.01.011. [DOI] [PubMed] [Google Scholar]
- Mo EK, Sung CK. Isolation and identification of phenyl ethyl alcohol from Pichia anomala SKM-T. J Appl Biol Chem. 2007;50:29–31. [Google Scholar]
- Okada N, Hirata A, Murakami Y, Shoji M, Sakagami H, Fujisawa S. Induction of cytotoxicity and apoptosis and inhibition of cyclooxygenase-2 gene expression by eugenol-related compounds. Anticancer Res. 2005;25:3263–3269. [PubMed] [Google Scholar]
- Ophardt CE (2003) Virtual chemistry book: anticancer drugs I. Elmhurst College. http://www.elmhurst.edu/chm/vchembook/655cancer.html
- Oussalah M, Caillet S, Lacroix M. Mechanism of action of Spanish oregano, Chinese cinnamon, and savory essential oils against cell membranes and walls of Escherichia coli O157:H7 and Listeria monocytogenes. J Food Prot. 2006;69:1046–1055. doi: 10.4315/0362-028x-69.5.1046. [DOI] [PubMed] [Google Scholar]
- Pereira CA, Bender MA, Awa AA, Brooks AL, Evans HJ, Groer PG, Littlefield LG, Preston RJ, Wachholz BW. Current status of cytogenetics procedures to detect and quantify previous exposures to radiation. Mutat Res. 1988;196:103–159. doi: 10.1016/0165-1110(88)90017-6. [DOI] [PubMed] [Google Scholar]
- Pisano M, Pagnan G, Loi M, Mura ME, Tilocca MG, Palmieri G, Fabbri D, Dettori MA, Delogu G, Ponzoni M, Rozzo C. Anti-proliferative and pro-apoptotic activity of eugenol-related biphenyls on malignant melanoma cells. Mol Cancer. 2007;6:1–12. doi: 10.1186/1476-4598-6-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sakihama Y, Cohen MF, Grace SC, Yamasaki H. Plant phenolic antioxidant and prooxidant activities: phenolics-induced oxidative damage mediated by metals in plants. Toxicology. 2002;177:67–80. doi: 10.1016/S0300-483X(02)00196-8. [DOI] [PubMed] [Google Scholar]
- Savage JRK. Classification and relationship of induced chromosomal structural changes. J Med Genet. 1975;12:103–122. doi: 10.1136/jmg.13.2.103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schmid I, Uittenbogaart CH, Krall WJ, Braun J, Giorgi JV. Dead cell discrimination with 7-amino-actinomycin D in combination with dual color immunofluorescence in single laser flow cytometry. Cytometry. 1992;13:204–208. doi: 10.1002/cyto.990130216. [DOI] [PubMed] [Google Scholar]
- Sghaier MB, Boubaker J, Neffati A, Limem I, Skandrani I, Bhouri W, Bouhlel I, Kilani S, Chekir-Ghedira L, Ghedira K. Antimutagenic and antioxidant potentials of Teucrium ramosissimum essential oil. Chem Biodivers. 2010;7:1754–1763. doi: 10.1002/cbdv.200900237. [DOI] [PubMed] [Google Scholar]
- Singh D, Kumar TR, Gupta VK, Chaturvedi P. Antimicrobial activity of some promising plant oils, molecules and formulations. Indian J Exp Biol. 2012;50:714–717. [PubMed] [Google Scholar]
- Skehan P, Storeng R, Scudiero D, Monks A, McMahon I, Vistica D. New colorimetric cytotoxicity assay for anticancer drug screening. J Nat Cancer Inst. 1990;82:1107–1112. doi: 10.1093/jnci/82.13.1107. [DOI] [PubMed] [Google Scholar]
- Sweify KM, Darwish IA, Mosallam SA. The cytogenetic effect of the new asthma and allergy drug montelukast on albino mice: chromosomal studies. Egypt J Biol. 2005;7:33–43. [Google Scholar]
- Ultee AE, Kets PW, Alberda M, Hoekstra FA, Smid EJ. Adaptation of the food-borne pathogen Bacillus cereus to carvacrol. Arch Microbiol. 2000;174:233–238. doi: 10.1007/s002030000199. [DOI] [PubMed] [Google Scholar]
- Unal F, Taner G, Yuzbasioglu D, Yilmaz S (2013) Antigenotoxic effect of lipoic acid against mitomycin-C in human lymphocyte cultures. Cytotechnology 65: 553–565 [DOI] [PMC free article] [PubMed]
- Vatan O, Celikler S, Yildiz G. In vitro antigenotoxic and anti-oxidative capacity of Hypnea musciformis (Wulfen) Lamouroux extract in human lymphocytes. Afr J Biotechnol. 2011;10:484–490. [Google Scholar]
- Vercesi AE, Kowaltowski AJ, Grijalba MT, Meinicke AR, Castilho RF. The role of reactive oxygen species in mitochondrial permeability transition. Biosci Rep. 1997;17:43–52. doi: 10.1023/A:1027335217774. [DOI] [PubMed] [Google Scholar]
- Vidhya N, Devaraj SN. Induction of apoptosis by eugenol in human breast cancer cells. Indian J Exp Biol. 2011;49:871–878. [PubMed] [Google Scholar]
- Weinstein IB. Cancer prevention: recent progress and future opportunities. Cancer Res. 1991;15:5080s–5085s. [PubMed] [Google Scholar]
- Yogesh AS, Fulzele RR. Cytogenetic study on genotoxicity of antitumor-antibiotic mitomycin C. Biomed Res. 2009;20:40–44. [Google Scholar]
- Yokota H, Hoshino J, Yuasa A. Suppressed mutagenicity of benzo(a)pyrene by the liver S9 fraction and microsomes from eugenol-treated rats. Mutat Res. 1986;172:231–236. doi: 10.1016/0165-1218(86)90060-1. [DOI] [PubMed] [Google Scholar]
- Yoo CB, Han KT, Cho KS, Ha J, Park HJ, Nam JH. Eugenol isolated from the essential oil of Eugeniacaryophyllata induces a reactive oxygen species mediated apoptosis in HL-60 human promyelocytic leukemia cells. Cancer Lett. 2005;225:41–52. doi: 10.1016/j.canlet.2004.11.018. [DOI] [PubMed] [Google Scholar]




