Abstract
To clarify the recent trends in prostate-specific antigen (PSA) distribution in men in Japan, we analyzed the PSA distributions of men undergoing PSA-based population screening. We summarized the annual individual data of PSA-based population screening in Kanazawa, Japan, from 2000 to 2011, and analyzed baseline serum PSA values of the participants at the first population screening. Serum PSA distributions were estimated in all participants and those excluding prostate cancer patients according to age. From 2000 to 2011, 19 620 men participated aged 54–69 years old in this screening program. Mean baseline serum PSA level of all participants at the first screening was 2.64 ng ml−1 in 2000, and gradually decreased to approximately 1.30 ng ml−1 in 2006. That of participants excluding prostate cancer patients was 1.46 ng ml−1 in 2000, and there was no remarkable change during the study period. The 95th percentiles in the participants excluding prostate cancer patients detected at the first population screening of men aged 54–59, 60–64, and 65–69 years old were 2.90, 3.60, and 4.50 ng ml−1, respectively. After the commencement of population screening, the proportion of prostate cancer patients with high serum PSA levels decreased. However, there were no changes in serum PSA levels in men without prostate cancer. Age-specific PSA reference level of men without prostate cancer in Japan was similar to that in China and Korea.
Keywords: age-specific prostate-specific antigen reference range, prostate cancer, prostate-specific antigen-based population screening, serum prostate-specific antigen distribution
INTRODUCTION
Prostate-specific antigen (PSA)-based screening is widely used for the early detection of prostate cancer, and the high rate of PSA testing among middle-aged men may have partially contributed to the continuous decrease in prostate cancer mortality rate in several countries.1 In Japan, the rate of screening for prostate cancer is still very low compared to the USA and Western Europe, and it has been predicted that the incidence and mortality rates of prostate cancer will increase in future.2 Since the 1990s, screening systems have been administered by each municipal government in Japan, and decreases in prostate cancer mortality rate are expected in some regions where the exposure rate to PSA testing has increased markedly.2 PSA-based screening systems implemented by municipal governments are reasonable ways of increasing the exposure rate to PSA testing, and several studies suggested that these systems may be effective for the early detection of prostate cancer.3,4,5,6
On the other hand, changes in lifestyle may have affected the incidence of prostate cancer in recent decades in Japan. A previous study indicated that baseline PSA distribution of Japanese men did not change between 1988 and 2003;7 however, there have been no reports regarding recent trends of PSA distribution. It is necessary to elucidate the trends in PSA distribution in middle-aged men to promote PSA-based population screening.
In this study, we examined the individual results of annual PSA-based population screening between 2000 and 2011 in Kanazawa, Japan, and analyzed annual baseline PSA distributions of the participants at the first screening. The clinical characteristics of prostate cancer detected in the first screening according to time trends were also investigated.
MATERIALS AND METHODS
Study population
Kanazawa city government has entrusted the Kanazawa Medical Association, which is composed of general practitioners, to administer annual health check-ups for the population of the city. Annual health check-ups are performed as preventive measures for early detection of all types of illnesses and risk factors using comprehensive examinations, such as a blood test, chest X-ray, and electrocardiogram. The participants represent approximately 30% of the whole Japanese population of the city, and almost all participants are healthy, and undergo annual health check-ups to prevent illness. Since 2000, PSA-based population screening for prostate cancer has been added to health check-ups and performed in Japanese men aged 54–69 years old.6,8 Total PSA (tPSA) measurements were performed using a commercial kit (Tosoh, Tokyo, Japan) in primary medical facilities as the primary screen. The normal range of tPSA level was set as 0–2.0 ng ml−1, and for cases in the gray zone with tPSA ranging between 2.1 and 10.0 ng ml−1, free PSA (fPSA) level was measured with an Immulyze Free PSA kit (Nippon DPC Co., Ltd., Chiba, Japan). From 2000 to 2002, individuals with tPSA levels over 2.1 ng ml−1 were classified as requiring “secondary screening” and were advised to consult a specialist in urology at a primary medical facility. However, after 2003, individuals with tPSA between 2.1 and 10.0 ng ml−1 and fPSA/tPSA ratio higher than 0.22 were not referred for secondary screening.6,8
Digital rectal examination (DRE) and transrectal ultrasonography (TRUS) were performed by urologists in secondary screening in all cases. Systematic TRUS-guided prostate biopsy (6–12 cores) was performed at the discretion of each urologist, and individuals who did not undergo prostate biopsy were followed-up by PSA testing at the respective Urology Department or annual population screening. In patients diagnosed with prostate cancer, pathological tumor grading and clinical staging were determined in accordance with the unified tumor node metastasis criteria based on the results of DRE, TRUS, computed tomography, magnetic resonance imaging, and bone scan at each Urology Department.9 These results were reported to the office of the Kanazawa Medical Association.
For this study, we summarized the annual individual data from 2000 to 2011 and analyzed baseline serum PSA values of the participants at the first population screening. Baseline serum PSA distributions were estimated in all participants and those excluding prostate cancer patients according to age. In screening-detected cancer patients, we compared the clinical characteristics between the patients detected in the early period (2000–2005) and the late period (2006–2011). We also investigated clinicopathological features of screening-detected cancer stratified into favorable or unfavorable cancer according to the definition of active surveillance studies.10,11 We examined prostate cancer with clinical stage of T1cN0M0, PSA below 10 ng ml−1, Gleason score of 6 or less, and one or two positive cores within 6–12 systemic biopsy cores taken as favorable cancer.
Statistical analysis
The principles of the Declaration of Helsinki were followed in this retrospective analysis. PSA distributions in annual screening and age-specific trends were estimated by polynomial nonlinear curve fit analysis with 95% confidence interval (CI) using the commercially available software (Prism, GraphPad Software, San Diego, CA, USA). Statistical assessments were performed using the Mann–Whitney U-test and Fisher's exact test. In all analyses, P < 0.05 was taken to indicate statistical significance.
RESULTS
From 2000 to 2011, 19 620 men participated in this screening program. The 12 years trend in PSA-based population screening is shown in Table 1. In the 1st year (2000), the number of participants and the proportion of men 65–69 years old were high; however, the annual age distribution of the participant was not so different in other years. The number of prostate cancer patients detected in this screening cohort was 236, and the annual rates of cancer detection were 0.78%–1.61%.
Table 1.
Trends of the participants of first PSA-based population screening

Eligible subjects in this study were 19 620 baseline serum PSA values of the participants at the first population screening. Annual proportions of the participants with baseline serum PSA above the cut-off level categorized by age range are shown in Figure 1 and Table 2. There were varieties in the distribution, and no statistically significant differences in the proportion among calendar years; however, the proportion of participants with serum PSA levels above 4.0 and 10.0 ng ml−1 was high in the initial years, and it seemed to decrease, especially in men 60–69 years old.
Figure 1.
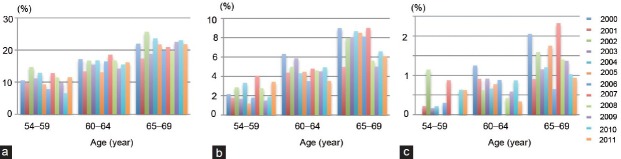
Percentages of men with prostate-specific antigen level greater than (a) 2.0 ng ml−1, (b) 4.0 ng ml−1, and (c) 10.0 ng ml−1, stratified by 5 years age range and calendar year.
Table 2.
Trends of the men with PSA level >4.0 ng ml−1 and 10.0 ng ml−1 stratified by 5 years age range and calendar year

Mean serum baseline PSA level of the participants was 2.64 ng ml−1 (median 1.0; 95% CI = 1.88–3.40) in 2000, and gradually decreased to approximately 1.30 ng ml−1 (median = 0.9) in 2006 (Figure 2a). That of the participants excluding prostate cancer patients detected at first population screening was 1.46 ng ml−1 (median 1.0; 95% CI = 1.40–1.51) in 2000, and there were no remarkable changes during the study period (Figure 2a). These tendencies were observed in participants aged 60–64 and 65–69 years old (Figure 2c and 2d). However, there was no difference in annual baseline PSA distribution between all participants aged 54–59 years old and the group excluding prostate cancer (Figure 2b).
Figure 2.
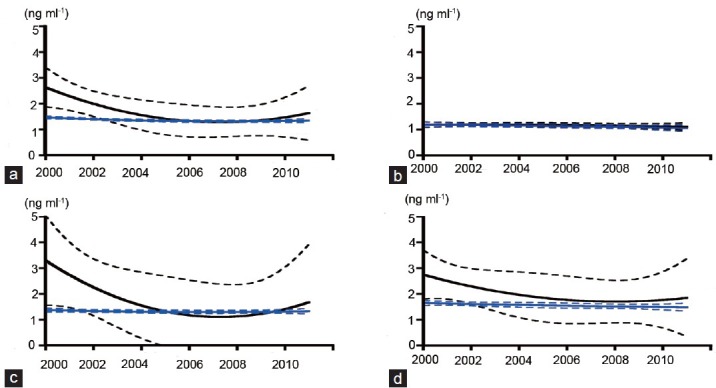
Mean serum prostate-specific antigen values in annual population screening by polynomial nonlinear curve fitting analysis (solid line) with 95% confidence interval (dotted line) in all participants (black) and in participants excluding those with screening-detected prostate cancer (blue). All participants (a), 54-59 years old (b), 60-64 years old (c), and participants 65-69 years old (d).
Age-specific baseline serum PSA levels are shown in Figure 3. In the participants excluding those detected at first population screening prostate cancer, mean serum PSA level in subjects 54 years old was 1.11 ng ml−1 (median 0.8; 95% CI = 1.03–1.18), and age-dependently increased to 1.76 ng ml−1 (median 1.1; 95% CI = 1.68–1.84) (Figure 3). The 95th percentiles in all participants aged 54–59, 60–64, and 65–69 years old were 2.90, 4.00, and 5.11 ng ml−1, respectively. Those in the participants excluding prostate cancer patients detected at first population screening aged 54–59, 60–64, and 65–69 years old were 2.90, 3.60, and 4.50 ng ml−1, respectively.
Figure 3.
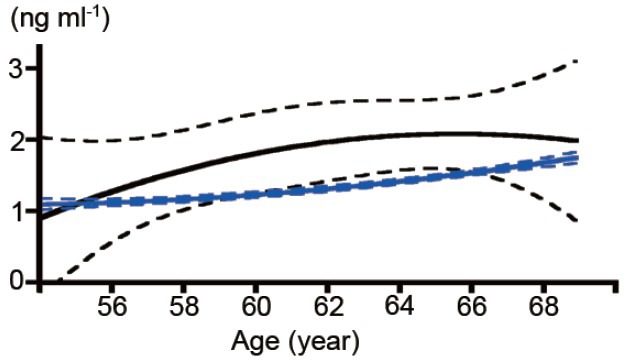
Mean serum prostate-specific antigen values according to age by polynomial nonlinear curve fitting analysis (solid line) with 95% confidence interval (dotted line) in all participants (black) and in participants excluding those with screening-detected prostate cancer (blue).
The clinical characteristics of the patients detected at first population screening were compared between those detected in the early period (2000–2005) and in the late period (2006–2011) (Table 3). There were significant differences in age and serum PSA level between these two groups (P = 0.0065, Mann–Whitney U-test). The proportions of stage T1c in patients detected in the late period were significantly higher than those in patients detected in the early period. Of all 236 screening-detected cancer patients, 36 (15.3%) and 186 (78.8%) were defined as favorable and unfavorable cancer patients, respectively. Statistical analysis could not be performed on Gleason score, the number of positive cancer cores, or the characteristics as active surveillance criteria between the two groups due to the relatively large number of cases without clinical data in the early period.
Table 3.
Trends of clinical characteristics of the patients detected at first populatuion screening

DISCUSSION
The incidence rate of prostate cancer has been increasing since the 1980s in the Asia-Pacific region, and this trend has been mainly attributed to the availability of cancer screening using PSA.12,13 On the other hand, the incidence of prostate cancer was higher in Asian Americans than in Asians, which suggests deep relationships between cancer incidence and environment factors, including diet and lifestyle.14 Environmental factors related to the increased incidence of prostate cancer may affect serum PSA in middle-aged men, and several previous studies demonstrated differences in age-specific PSA reference ranges among various countries.15,16,17,18 Moreover, a marked increase in mortality rate for prostate cancer in East Asia from the 1950s to the 2000s was reported, and these findings may be in part caused by the nutrition transition to “westernization.”19 Based on the hypothesis that recent changes in lifestyle in Asia, including Japan, have been toward “westernization,” which may result in increases in serum PSA levels and the incidence of prostate cancer, we investigated recent trends in changes in baseline serum PSA distribution in a PSA-based population screening cohort. However, there were no changes in annual baseline serum PSA distribution of men 54–69 years old without prostate cancer from the years 2000 to 2011 (blue line in Figure 2). These findings were similar to those in a previous Japanese study from 1988 to 2003.7 These findings regarding serum PSA distribution suggest that there are no changes in the serum PSA levels in 54–69 years old men without prostate cancer in Japan, at least over the last two decades, and further longitudinal studies will solve the problem regarding the relationship between serum PSA level and environment factors in Asia.
In this study, the decreasing trend of baseline serum PSA in all men, including prostate cancer patients, detected at first population screening was demonstrated (Figure 1, black line in Figure 2, Table 3). These findings may indicate a decrease in the proportion of prostate cancer with high serum PSA levels after starting population screening, especially in the initial few years. As participants in the first population screening were included in this study, this issue was somewhat curious. Repeat screening using serum PSA testing caused favorable shifts in cancer characteristics, including a decrease in serum PSA at diagnosis.20,21,22,23 However, there have been no reports of a decreasing trend of baseline PSA distribution in men undergoing first population screening. A previous epidemiological cohort study using data from Cancer of the Prostate Strategic Urologic Research Endeavor demonstrated that prostate cancer was increasingly diagnosed in younger men with lower risk, and at early disease stage in the PSA era.24,25 The database did not show the exposure rate of PSA testing in the subjects, but it was certain that the widespread use of PSA testing has led to these trends. PSA testing several times in middle-aged men after instigation of PSA screening may identify those men with high serum PSA levels.
Another hypothesis regarding the decreasing trend of prostate cancer with high serum PSA level was the possible existence of men who underwent PSA testing outside of the population screening program during the study period. It is possible that the widespread adoption of PSA-based population screening prevents awareness of PSA testing among general practitioners. The increase in PSA testing by general practitioners may lead to an increase in number of patients with high serum PSA levels detected outside of the population screening.
The widespread adoption of PSA testing may lead to over diagnosis and overtreatment for “insignificant” or “minimal” prostate cancer. In this study, it was demonstrated that the proportion of clinical T1c patients detected at population screening was higher in the late period than in the early period (Table 3), and this result may support previous reports indicating a favorable shift of the characteristics for prostate cancer in the PSA era.24,25 However, the proportion of prostate cancer with unfavorable cancerous features was over 80% in the late period (Table 3). Twelve years of PSA-based population screening was able to screen prostate cancer patients with high serum PSA levels, but not to change the majority of first screening-detected patients to those with favorable cancerous features, which means that the overdetection of insignificant prostate cancer did not occur in the study period. Although the mortality rate in screening-detected prostate cancer is needed to truly elucidate the relationship between the widespread adoption of PSA-based screening and overdetection of prostate cancer, these findings in this study may become one of the evidences that support the promotion of PSA-based population screening in countries in which PSA testing has not been widely adopted. A future trend of the clinical characteristics for first screening-detected prostate cancer is an interesting issue for further study.
In terms of age-specific serum PSA reference range, the 95th percentiles in the participants excluding prostate cancer detected at first population screening aged 54–59, 60–64, and 65–69 years old were 2.90, 3.60, and 4.50 ng ml−1, respectively. In a recent report, the 95th percentiles in healthy Irish men aged 55–59, 60–64, and 65–69 years old were 3.25, 4.02, and 4.96 ng ml−1, respectively,18 and the 95th percentile in health Jordanian men aged 55–59 years old was 3.76 ng ml−1.16 These results indicated regional differences in age-specific PSA reference range, and that serum PSA levels may be higher in European and Middle-Eastern than in Japanese men. On the other hand, the 95th percentiles in the participants excluding prostate cancer aged 60–69 years old were 4.10 ng ml−1 in this study, and the reference level was similar to recent studies in Korea (3.90 ng ml−1)15 and China (4.10 ng ml−1).17 The cut-off levels of PSA-based population screening should be set based on age-specific PSA reference range, and there should be regional differences. Although further examinations are needed to obtain data of serum PSA distribution in Asia, the present study clarified the trend of serum PSA distribution and age-specific PSA reference range in middle-aged men in Japan.
This study had several limitations based on the retrospective nature of the analysis in the screening cohort. First, the factors that independently affect serum PSA level, such as body mass index and waist circumference were not evaluated. Second, there was a possible bias in the results due to the different annual numbers of participants over the course of the program. Since annual health check-ups implemented by municipal governments in Japan are optional, not compulsory for the city population, the different annual numbers of participants is thought to be characteristic and unavoidable.4,23 Third, the mortality rate of the patients in this screening cohort is not available. These limitations and biases should be resolved by further prospective large-scale studies. However, we demonstrated the decreasing trend of patients with high serum PSA levels in our PSA-based population screening cohort, and this study provided evidence to promote population screening for prostate cancer in Asia.
AUTHOR CONTRIBUTIONS
YK designed the study, carried out analysis and interpretation of data, and drafted the manuscript. KM, HY, KN, MO, KI, SU, YK, and HK carried out the acquisition of data. AM and NM participated in its design and coordination and helped to draft the manuscript.
COMPETING INTERESTS
The authors declare no competing interests.
ACKNOWLEDGMENTS
We thank Dr. Yasuo Takeda and Dr. Atsushi Hashiba of Kanazawa Medical Association for providing data of the Kanazawa population-based screening cohort.
REFERENCES
- 1.Bouchardy C, Fioretta G, Rapiti E, Verkooijen HM, Rapin CH, et al. Recent trends in prostate cancer mortality show a continuous decrease in several countries. Int J Cancer. 2008;123:421–9. doi: 10.1002/ijc.23520. [DOI] [PubMed] [Google Scholar]
- 2.Ito K. Prostate-specific antigen-based screening for prostate cancer: evidence, controversies and future perspectives. Int J Urol. 2009;16:458–64. doi: 10.1111/j.1442-2042.2009.02293.x. [DOI] [PubMed] [Google Scholar]
- 3.Kubota Y, Ito K, Imai K, Yamanaka H. Effectiveness of mass screening for the prognosis of prostate cancer patients in Japanese communities. Prostate. 2002;50:262–9. doi: 10.1002/pros.10057. [DOI] [PubMed] [Google Scholar]
- 4.Kato T, Habuchi T, Tsuchiya N, Sato K, Kitajima S, et al. Mass screening of prostate cancer and its impact on inhabitants in Akita Prefecture, Japan. Aktuelle Urol. 2010;41(Suppl 1):S53–6. doi: 10.1055/s-0029-1224653. [DOI] [PubMed] [Google Scholar]
- 5.Okihara K, Kitamura K, Okada K, Mikami K, Ukimura O, et al. Ten year trend in prostate cancer screening with high prostate-specific antigen exposure rate in Japan. Int J Urol. 2008;15:156–60. doi: 10.1111/j.1442-2042.2007.01957.x. [DOI] [PubMed] [Google Scholar]
- 6.Kitagawa Y, Mizokami A, Nakashima K, Koshida K, Shimamura M, et al. Clinical outcomes of prostate cancer patients detected by prostate-specific antigen-based population screening in Kanazawa City, Japan. Int J Urol. 2011;18:592–6. doi: 10.1111/j.1442-2042.2011.02796.x. [DOI] [PubMed] [Google Scholar]
- 7.Ohi M, Ito K, Yamamoto T, Miyakubo M, Takechi H, et al. Changes in baseline PSA levels in Japanese men from 1988 to 2003. Urology. 2008;72:95–8. doi: 10.1016/j.urology.2008.03.014. [DOI] [PubMed] [Google Scholar]
- 8.Kobori Y, Kitagawa Y, Mizokami A, Komatsu K, Namiki M. Free-to-total prostate-specific antigen (PSA) ratio contributes to an increased rate of prostate cancer detection in a Japanese population screened using a PSA level of 2.1-10.0 ng/ml as a criterion. Int J Clin Oncol. 2008;13:229–32. doi: 10.1007/s10147-007-0742-2. [DOI] [PubMed] [Google Scholar]
- 9.Sobin LH, Wittekind CH, editors. TNM Classification of Malignant Tumours. 5th ed. New York: John Wiley and Sons; 1997. International Union against Cancer. Urologic tumors: prostate; pp. 170–3. [Google Scholar]
- 10.Kakehi Y, Kamoto T, Shiraishi T, Ogawa O, Suzukamo Y, et al. Prospective evaluation of selection criteria for active surveillance in Japanese patients with stage T1cN0M0 prostate cancer. Jpn J Clin Oncol. 2008;38:122–8. doi: 10.1093/jjco/hym161. [DOI] [PubMed] [Google Scholar]
- 11.Klotz L, Zhang L, Lam A, Nam R, Mamedov A, et al. Clinical results of long-term follow-up of a large, active surveillance cohort with localized prostate cancer. J Clin Oncol. 2010;28:126–31. doi: 10.1200/JCO.2009.24.2180. [DOI] [PubMed] [Google Scholar]
- 12.Baade PD, Youlden DR, Cramb SM, Dunn J, Gardiner RA. Epidemiology of prostate cancer in the Asia-Pacific region. Prostate Int. 2013;1:47–58. doi: 10.12954/PI.12014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Zhang L, Yang BX, Zhang HT, Wang JG, Wang HL, et al. Prostate cancer: an emerging threat to the health of aging men in Asia. Asian J Androl. 2011;13:574–8. doi: 10.1038/aja.2010.126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.McCracken M, Olsen M, Chen MS, Jr, Jemal A, Thun M, et al. Cancer incidence, mortality, and associated risk factors among Asian Americans of Chinese, Filipino, Vietnamese, Korean, and Japanese ethnicities. CA Cancer J Clin. 2007;57:190–205. doi: 10.3322/canjclin.57.4.190. [DOI] [PubMed] [Google Scholar]
- 15.Lee SE, Kwak C, Park MS, Lee CH, Kang W, et al. Ethnic differences in the age-related distribution of serum prostate-specific antigen values: a study in a healthy Korean male population. Urology. 2000;56:1007–10. doi: 10.1016/s0090-4295(00)00837-2. [DOI] [PubMed] [Google Scholar]
- 16.Battikhi MN. Age-specific reference ranges for prostate-specific antigen (PSA) in Jordanian patients. Prostate Cancer Prostatic Dis. 2003;6:256–60. doi: 10.1038/sj.pcan.4500656. [DOI] [PubMed] [Google Scholar]
- 17.Liu ZY, Sun YH, Xu CL, Gao X, Zhang LM, et al. Age-specific PSA reference ranges in Chinese men without prostate cancer. Asian J Androl. 2009;11:100–3. doi: 10.1038/aja.2008.17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Casey RG, Hegarty PK, Conroy R, Rea D, Butler MR, et al. The distribution of PSA age-specific profiles in healthy Irish men between 20 and 70. ISRN Oncol 2012. 2012:832109. doi: 10.5402/2012/832109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Zhang J, Dhakal IB, Zhao Z, Li L. Trends in mortality from cancers of the breast, colon, prostate, esophagus, and stomach in East Asia: role of nutrition transition. Eur J Cancer Prev. 2012;21:480–9. doi: 10.1097/CEJ.0b013e328351c732. [DOI] [PubMed] [Google Scholar]
- 20.Mäkinen T, Tammela TL, Stenman UH, Määttänen L, Aro J, et al. Second round results of the Finnish population-based prostate cancer screening trial. Clin Cancer Res. 2004;10:2231–6. doi: 10.1158/1078-0432.ccr-03-0338. [DOI] [PubMed] [Google Scholar]
- 21.Hugosson J, Aus G, Lilja H, Lodding P, Pihl CG. Results of a randomized, population-based study of biennial screening using serum prostate-specific antigen measurement to detect prostate carcinoma. Cancer. 2004;100:1397–405. doi: 10.1002/cncr.20126. [DOI] [PubMed] [Google Scholar]
- 22.Postma R, Schröder FH, van Leenders GJ, Hoedemaeker RF, Vis AN, et al. Cancer detection and cancer characteristics in the European Randomized Study of Screening for Prostate Cancer (ERSPC) – Section Rotterdam. A comparison of two rounds of screening. Eur Urol. 2007;52:89–97. doi: 10.1016/j.eururo.2007.01.030. [DOI] [PubMed] [Google Scholar]
- 23.Kitagawa Y, Sawada K, Mizokami A, Nakashima K, Koshida K, et al. Clinical characteristics and prostate-specific antigen kinetics of prostate cancer detected in repeat annual population screening in Japan. Int J Urol. 2014;21:461–5. doi: 10.1111/iju.12304. [DOI] [PubMed] [Google Scholar]
- 24.Cooperberg MR, Broering JM, Kantoff PW, Carroll PR. Contemporary trends in low risk prostate cancer: risk assessment and treatment. J Urol. 2007;178:S14–9. doi: 10.1016/j.juro.2007.03.135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Glass AS, Cowan JE, Fuldeore MJ, Cooperberg MR, Carroll PR, et al. Patient demographics, quality of life, and disease features of men with newly diagnosed prostate cancer: trends in the PSA era. Urology. 2013;82:60–5. doi: 10.1016/j.urology.2013.01.072. [DOI] [PubMed] [Google Scholar]


