Abstract
Background:
Tissue reinforcement with allogeneic or xenogeneic acellular dermal matrices (ADMs) is increasingly used in single-stage (direct-to-implant) and 2-stage implant-based breast reconstruction following mastectomy. ADMs allow surgeons to control implant position and obviate the need for submuscular implant placement. Here, we review the benefits and risks of using ADMs in implant-based breast reconstruction based on available data.
Methods:
A comprehensive analysis of the literature with focus on recent publications was performed. Additional information regarding the proper use of ADMs was based on our institutional experience.
Results:
ADM use may improve definition of the lateral confines of the breast and lower pole projection. It may facilitate direct-to-implant procedures and improve aesthetic outcomes. The effect of ADMs on complication rates remains controversial. Known patient risk factors such as obesity, smoking, and radiotherapy should be considered during patient selection. For patients with healthy, well-vascularized skin envelopes, ADM-assisted direct-to- implant reconstruction is a safe and cost-effective alternative to 2-stage implant reconstruction, with low complication rates. ADMs may be used to treat capsular contracture, and limited available data further suggest the possibility that ADMs may reduce the risk of capsular contracture. Novel synthetic or biosynthetic tissue reinforcement devices with different physical and ease-of-use properties than ADMs are emerging options for reconstructive surgeons and patients who seek to avoid tissue products from human or mammalian cadavers.
Conclusions:
ADM-assisted implant-based breast reconstruction may improve aesthetic outcomes. However, appropriate patient selection, surgical technique, and postoperative management are critical for its success, including minimizing the risk of complications.
For women with breast cancer requiring mastectomy, breast reconstruction may improve body image and provide significant psychosocial benefits.1 Although autologous reconstruction traditionally has been associated with the highest patient satisfaction in terms of aesthetic outcomes,2–4 implant-based reconstruction is by far the most common approach to postmastectomy breast reconstruction in the United States.5 Of approximately 92,000 breast reconstructions performed in 2012, slightly more than 72,000 were implant based, and of those, the vast majority (64,575) were 2-stage tissue expander/implant reconstructions.5 Compared with autologous reconstructions, implant-based reconstructions are simpler, take less time to perform, are less invasive, support faster patient recovery,2,6 and avoid the need for donor site surgery, which may cause significant postoperative deterioration of physical well-being.7 In addition, they are believed to have more favorable third-party reimbursement limits.8
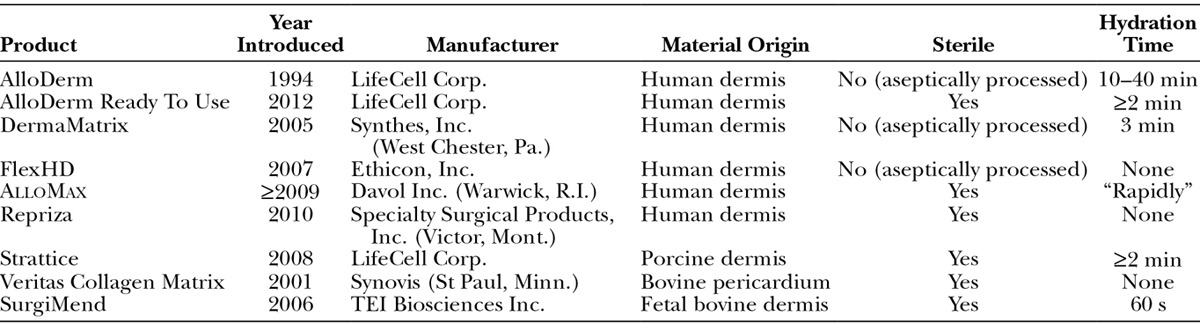
The increasing availability of acellular dermal matrices (ADMs) prepared from human or animal cadavers for tissue reinforcement provides plastic surgeons with a unique tool to improve aesthetic outcomes of implant-based reconstruction by expanding and reshaping the implant pocket, while further reducing the invasiveness of surgical intervention.9–15 The substantial pain caused by the serratus anterior elevation necessary for total or partial muscle coverage can be avoided, as lateral coverage is provided by the ADM. However, results of a prospective randomized study in 70 patients who underwent immediate ADM-assisted or conventional submuscular tissue expander/implant reconstruction after mastectomy revealed no significant differences between these patient groups in immediate postoperative pain or pain during the expansion phase.16 In addition to providing mechanical stability, ADMs facilitate cellular and vascular infiltration during wound healing and tissue regeneration through incorporation of the matrix. Since the first publication of ADM-assisted primary breast reconstruction in 2005,9 an increasing number of products have become available in the United States (Table 1). In Israel, AlloDerm (LifeCell Corp., Branchburg, N.J.), the prototypical ADM of human dermal origin, has been available since 2005 and SurgiMend (TEI Biosciences Inc., Boston, Mass.), a fenestrated ADM derived from fetal bovine dermis, has been available since 2008. Allogeneic ADMs are not extensively used in many European countries because of regulatory restrictions on human tissue products and cost considerations favoring alternative products.18,19
Table 1.
Allogeneic and Xenogeneic Soft Tissue Reinforcement Devices Available in the United States17
In this review, we describe the benefits of tissue reinforcement in implant-based breast reconstruction, such as improved aesthetic outcomes, provide cost considerations, and discuss the risk of postoperative complications. Moreover, we provide recommendations based on our own experience for how the risk of complications can be minimized by appropriate patient selection, surgical technique, and postoperative management.
IMMEDIATE IMPLANT-BASED RECONSTRUCTION WITH AND WITHOUT TISSUE REINFORCEMENT
Conventional Implant-based Reconstruction
Conventional implant-based reconstruction after mastectomy requires the creation of an implant pocket beneath the pectoralis major muscle for total or partial muscle implant coverage (Table 2). For total muscle coverage, elevation of the serratus anterior muscle is required for lateral coverage and support. Total muscle coverage limits the possible anterior and inferior projection and thus may lead to less than optimal aesthetic outcomes. Furthermore, because the size of the implant pocket that can initially be created with this approach is constrained by available skin and muscle tissue after the mastectomy, immediate reconstruction generally requires a 2-stage procedure, in which a tissue expander is implanted first, then gradually expanded over several months, and eventually replaced by a permanent implant.21
Table 2.
Surgical Techniques of Total and Partial Muscle Coverage and of ADM-assisted Direct-to-implant Reconstruction

Implant-based Reconstruction with ADMs
The use of ADMs enables the formation of larger implant pockets and optimal implant positioning without the need for serratus anterior muscle elevation. Provided the skin is sufficiently healthy, ADM use in 2-stage reconstruction allows for more predictable tissue expander position, larger intraoperative expander fill volumes, and fewer expansions compared with submuscular placement.13,22–25
An important aspect of ADM use is that it facilitates immediate direct-to-implant reconstruction (Table 2, Fig. 1) in appropriately selected patients by allowing precise positioning of a full-sized permanent implant, with favorable aesthetic outcomes and minimal risk of implant displacement, visibility, rippling, or extrusion.10–12,20,26–28 Moreover, ADM materials that are not completely resorbed help prevent pectoralis muscle retraction and offer additional soft tissue coverage in the lower pole of the breast. Experience from 331 consecutive immediate direct-to-implant reconstructions performed with AlloDerm at the Massachusetts General Hospital suggests that this approach is associated with favorable aesthetic outcomes and low complication rates in patients with thick, well-vascularized skin flaps after skin-sparing or nipple-sparing mastectomy.20 Similarly, the use of a SurgiMend in 341 consecutive immediate implant-based reconstructions after total skin-sparing mastectomy, including 270 direct-to-implant reconstructions, performed between 2001 and 2011 at the Assuta Medical Center in Tel Aviv, Israel, provided superior aesthetic results and similar rates of complications compared with traditional procedures.17
Fig. 1.
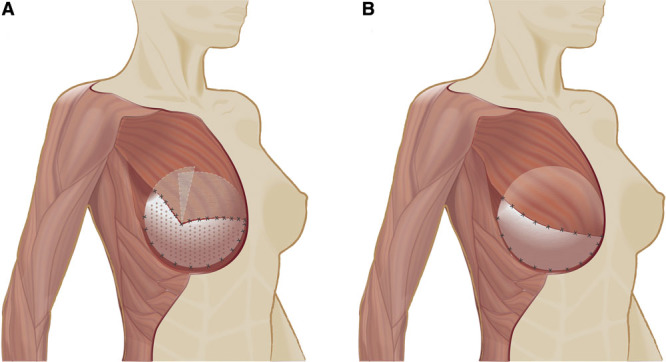
Use of ADM in implant-based breast reconstruction. Shown are the techniques practiced at the Assuta Medical Center (AMC), Tel Aviv (A)17 and the Massachusetts General Hospital (MGH), Boston (B).10,20 The inferior origin of the pectoralis major is released on the aponeurosis of the external oblique and on the inferior sternum, with pectoralis elevation up to the 3 and 9 o’clock position (AMC) or the 4 and 8 o’clock position (MGH). A tailored fenestrated semioval (AMC) or a standard rectangular (MGH) ADM sheet is sewn inferiorly to the thoracic fascia or inframammary fold (if intact) and laterally to the thoracic fascia to form the implant pocket. In the AMC technique, the semioval ADM is split, and the medial and lateral tails are sutured to the pectoralis major with deep overlap of the tails underneath the muscle (A). In the MGH technique, the rectangular ADM is sewn edge to edge to the pectoralis major without overlap (B).
In the past, patient satisfaction tended to be lower with implant-based rather than autologous reconstruction,3 which may be due in part to the limitations in shaping the implant pocket during conventional implant-based procedures. In our experience, the advent of ADM and nipple-sparing procedures, including ADM-assisted direct-to-implant reconstruction, has allowed for significant improvements in overall aesthetic results of implant-based reconstruction, with the potential to increase patient satisfaction scores in the future. Furthermore, evaluation of aesthetic outcomes of 183 tissue expander–based reconstructions performed at a single institution showed that ADM use (58 reconstructions) was associated with significantly higher aesthetic scores compared with total submuscular reconstruction (125 reconstructions).29
PATIENT RISK FACTORS FOR COMPLICATIONS AND RECONSTRUCTIVE FAILURE
A number of database analyses consistently identified high body mass index and smoking as independent risk factors for complications and/or implant loss.30–32 For 1170 two-stage breast reconstructions performed over a 2-year period, smoking, obesity, and hyper tension each increased the odds of reconstructive failure by factors of 5, 7, and 4, respectively.30 For more than 14,000 reconstructions with or without ADM captured in the Tracking Outcomes and Operations in Plastic Surgery (TOPS) database between 2008 and 2011, high body mass index, smoking, and diabetes were independent risk factors for expander/implant loss.31 ADM use seems to have no substantial influence on patient-related risk factors.31,33 A recent analysis of data from the American College of Surgeons National Surgical Quality Improvement Program, which identified smoking and body mass index as independent risk factors for short-term complications, found no statistically significant risk differences between immediate ADM-assisted (n = 1717) and submuscular (n = 7442) tissue expander reconstruction.32
ADM USE AND POSTOPERATIVE COMPLICATIONS
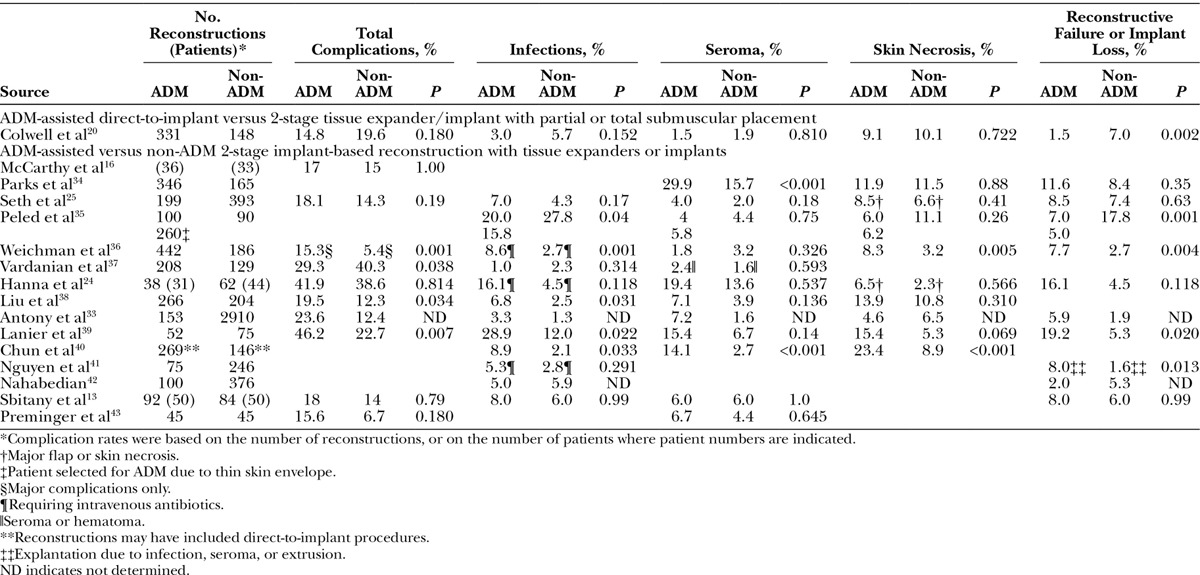
A persistent concern among plastic surgeons is whether ADMs increase the risk of short-term complications, given the conflicting findings from retrospective studies,13,16,20,24,25,33–43 systematic reviews,23,44 and meta-analyses.22,45,46 However, although meta-analyses (level III evidence) found increased risks of infection, seroma, and/or implant loss associated with ADM use,22,45,46 some recent large studies (level II or III evidence) not included in these analyses found similar or lower complication rates for ADM-assisted versus traditional 2-stage reconstruction.20,25,35,37 For example, a comparative study of 479 implant-based reconstructions found no difference in total complication rates or rates of infection and seroma between ADM-assisted direct-to-implant reconstruction compared with 2-stage tissue expander/implant reconstruction without ADM (Table 3).20 Similarly, in a recent prospective cohort study, ADM use was associated with significant reductions in expander/implant loss (Table 2) and unexpected returns to the operation room.35 These findings suggest that ADM use itself is not an independent risk factor for complications and that the large discrepancies in findings among different institutions may be attributable to other factors. This view is supported by the results of recent, large-scale National Surgical Quality Improvement Program and TOPS analyses.31,47 Although the TOPS analysis showed that ADM use (versus no ADM use) was associated with a statistically significant increase in the risk of expander/implant loss (odds ratio, 1.42; 95% confidence interval, 1.04–1.94; P = 0.026), the absolute risk increase was only 0.7%.31
Table 3.
Complication Rates with ADM-assisted and Conventional Implant-based Breast Reconstruction in Controlled Studies
In our experience, the vast majority of complications attributed to ADM-assisted reconstruction are avoidable by appropriate patient selection and surgical technique. An emerging consensus on the importance of these factors is reflected in the recently published joint guidelines from the Association of Breast Surgery and the British Association of Plastic, Reconstructive and Aesthetic Surgeons48 and recommendations by other experts in the field.49,50 Effective coordination of mastectomy and reconstructive surgery to ensure optimal viability of the skin envelope may further improve outcomes. Familiarity of the reconstructive surgeon with optimal device-specific techniques also is crucial for avoiding complications. In 331 consecutive ADM-assisted 1-stage implantations conducted between 2006 and 2009 at the Massachusetts General Hospital, increasing experience of the surgeons and better communication with the breast surgeon substantially reduced the incidence of skin necrosis, resulting in a significant 2-fold reduction in total complication rates from the surgeons’ first to subsequent years of performing the procedure (21.4%–10.9%; P < 0.02).20 However, if skin viability is questionable at the time of the mastectomy, total muscle coverage (rather than partial muscle release with or without ADM) should be considered even if this may negatively affect the final aesthetic outcome (Fig. 2).
Fig. 2.
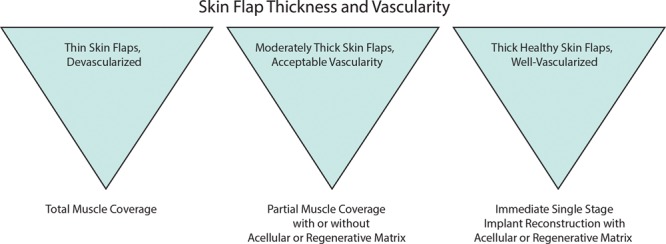
Algorithm for implant-based reconstruction.20 Reprinted with permission from Colwell AS, Damjanovic B, Zahedi B, et al. Retrospective review of 331 consecutive immediate single-stage implant reconstructions with acellular dermal matrix: indications, complications, trends, and cost. Plast Reconstr Surg. 2011;128:1170–1178.
THE RISK OF CAPSULAR CONTRACTURE
Capsular contracture is a common risk of implant-based reconstruction, particularly in the setting of radiotherapy.51–54 A variety of factors may contribute to development of capsular contracture, including but not limited to insufficient sterility during surgery, hematoma, mechanical strain on the inferior skin envelope, type and surface properties of the implant, and radiation therapy.55,56 It has been suggested that the use of ADMs may minimize capsular contracture by reducing pressure on the inferior breast skin envelope,20 which in turn may reduce fibroblast stimulation and inflammation.57,58 In a retrospective comparison of 2-stage reconstruction with and without ADM use during expander implantation in 203 patients, the capsular contracture rate was significantly lower for ADM-assisted reconstructions (3.8% versus 19.4%; P < 0.001) at a mean follow-up after implant exchange of 29 months.37 Moreover, a remarkably low capsular contracture rate of only 0.4% was observed in an 8-year study of 466 ADM-assisted direct-to-implant breast reconstructions with a mean follow-up of 29 months,27 and ADM use, including complete implant coverage, has been used successfully in the treatment of capsular contracture.55,59 How ever, given that the risk of implant-related capsular contracture increases over time,60 it remains to be demonstrated whether ADMs reduce the long-term risk of capsular contracture. Furthermore, it is presently unclear whether non–ADM-based tissue reinforcements can influence the incidence of capsular contracture.
EFFECT OF RADIOTHERAPY
A major concern with implant-based breast reconstruction is the effect of radiation therapy on complication rates.61 Radiation therapy given before or after mastectomy has been associated with significantly increased rates of major complications,62 including implant removal or replacement25,63 and capsular contracture.51,53,54 However, a recent systematic review found that ADM did not increase the complication rate in the setting of radiotherapy.64 Similarly, another recent literature review concluded that ADM use essentially had a neutral effect on postoperative complications among patients who received adjuvant radiation therapy after implant-based reconstruction,65 and results of a recent retrospective study even suggested that ADM use may significantly reduce the odds of complications (including explantation) in the setting of postmastectomy radiation therapy.25
Among patients who received a total of 479 ADM-assisted direct-to-implant or conventional 2-stage reconstructions at the Massachusetts General Hospital, radiotherapy was associated with an increased rate of early complications. Among patients who received radiation, the highest complication rate was seen in the setting of preoperative irradiation and conventional 2-stage reconstruction (41.1%), whereas the lowest rate was seen in direct-to-implant reconstructions with postoperative radiation (16.7%).20 The importance of timing of postmastectomy radiotherapy was also demonstrated in a prospective, controlled study of 257 patients undergoing subpectoral 2-stage breast reconstruction.66 Patients who received radiotherapy on the tissue expander had a significantly higher failure rate (40%) than those who received radiotherapy on the permanent implant (6.4%) or received no radiotherapy (2.3%; P < 0.0001).66
Radiation therapy also seems to affect capsular contracture rates after ADM-assisted implant-based breast reconstruction. The overall rate of clinically significant capsular contracture (grade III/IV) among 341 reconstructions at the Assuta Medical Center was only 2.0%. Remarkably, capsular contracture occurred exclusively in patients who previously received preoperative or postoperative radiation therapy at a rate of 12.3%.17 In a study of ADM-assisted 2-stage reconstruction in 289 women, radiation therapy before mastectomy and at expander stage resulted in dramatically increased rates of infection (53% and 73%, respectively, versus 1.4% without radiation) and grade III/IV capsular contractures (41% and 61%, respectively, versus 1.4% without radiation), although eventual explantation was avoided in most cases.54 Thus, although the use of ADM in implant-based reconstruction may reduce the risk of capsular contracture, it remains to be demonstrated whether this benefit extends to patients who receive pre- or postoperative radiation therapy.
COST CONSIDERATIONS
Recent cost analyses including the costs of probable complications in addition to physician and hospital fees estimate that ADM-assisted direct-to-implant reconstruction may result in moderate to substantial cost savings compared with traditional 2-stage implant reconstruction.67,68 At the Massachusetts General Hospital, overall hospital charges of the 2 procedures did not differ significantly (P = 0.8) because the substantially lower professional fees charged by anesthesiologists and surgeons for ADM-assisted direct-to-implant reconstruction were largely offset by higher hospital charges.20 An important cost factor when using tissue reinforcement can be the costs of the tissue support itself, particularly if AlloDerm is used. However, these costs may decrease with the increasing availability of lower cost xenogeneic ADMs18 and alternative tissue reinforcement devices.19,69 Furthermore, overall cost savings may potentially be larger than currently estimated if future research demonstrated that the use of ADM-assisted procedures substantially reduces the incidence of implant loss, capsular contracture, time off from work, and corrective surgery. The usage of ADM in 2-stage reconstruction increases the material costs for the procedure. For cost savings to be realized in this setting, a decreased need for revisions, shorter operative time, and/or lower complication rates would need to be demonstrated in a cost–benefit analysis.
NON-ADM OPTIONS FOR SOFT TISSUE REINFORCEMENT
ADMs may vary in their chemical composition and physical properties, with the potential to affect the quality and timing of tissue regeneration,70–72 and the risk of complications.18,73–80 Differences in material-associated risk of inflammation, thickness, requirements for hydration, and sterility may affect the handling facility of ADMs and their ability to simultaneously provide adequate structural support and sufficient pliability.81 Patients who do not accept cadaver material being part of their reconstructed breasts would benefit from alternative products with appropriate physical and ease-of-use properties that are either synthetic or made from biomaterials other than ADMs.
TiLOOP Bra (pfm medical titanium, Nuremberg, Germany), a nonabsorbable titanium-coated propylene mesh approved for breast reconstruction in Europe, was retrospectively evaluated in 231 breast reconstructions. Explantation (7.8%) was the most common major complication, with skin necrosis and capsule fibrosis identified as significant risk factors in multivariate analysis.19 TIGR Matrix (Novus Scientific, Uppsala, Sweden), a synthetic, long-term resorbable surgical mesh,82 may be beneficial as temporary tissue reinforcement, but clinical experience to date is very limited.83 A retrospective review of 76 direct-to-implant reconstructions with Vicryl mesh (Ethicon, Inc., Somerville, N.J.) in 50 consecutive patients was associated with complications in 5 breasts (6.6%) including 1 implant loss (1.3%) consequent to infection.69 Long-term capsular contracture rates were not reported.69
SERI (Allergan, Inc., Irvine, Calif.), a long-term bioresorbable, silk-derived surgical scaffold,84–86 received 510(k) clearance from the US Food and Drug Administration for use as soft tissue reinforcement in plastic and reconstructive surgery. In a single-arm prospective multicenter study of 2-stage implant-based breast reconstruction with SERI in the United States (NCT01256502), complications at 1-year follow-up (n = 105) included necrosis (6.7%), hematoma (4.8%), seroma (5.7%), implant loss (3.8%), cellulitis [minor breast infection requiring antibiotic treatment (2.9%)], breast infection [major, requiring surgical intervention (1.0%)], and capsular contracture (1.9%), but were considered unrelated to the use of SERI by the investigators.87
CONCLUSIONS
Tissue reinforcement in implant-based breast reconstruction may reduce the invasiveness of implant-based procedures and improve aesthetic outcomes. Appropriate patient selection, proper surgical technique, and adjusted postoperative management are critical for the success of ADM-assisted reconstruction, including minimizing the risk of complications. For qualifying patients with healthy, well-vascularized skin envelopes, ADM-assisted direct-to-implant reconstruction is a safe and cost-effective alternative to 2-stage implant reconstruction that can provide excellent aesthetic results. Known patient risk factors for implant-based reconstruction, such as obesity and smoking, also apply to ADM-assisted reconstruction and should be considered during patient selection.
The availability of novel synthetic or biosynthetic tissue reinforcement devices that have different physical and ease-of-use properties than ADMs may enhance the ability to refine surgical techniques to further optimize aesthetic outcomes and minimize complications.
ACKNOWLEDGMENT
Writing assistance was provided by Roland Tacke, PhD, of Evidence Scientific Solutions, Philadelphia, Pa., and funded by Allergan, Inc.
Footnotes
Disclosure: Medical writing support for this manuscript was funded by Allergan, Inc. Dr. Scheflan serves as a consultant for Allergan, Inc., and a clinical investigator for TEI Biosciences Inc. Dr. Colwell is a consultant for Allergan, Inc. and LifeCell Corp. The Article Processing Charge was paid for by Allergan, Inc.
REFERENCES
- 1.Atisha D, Alderman AK, Lowery JC, et al. Prospective analysis of long-term psychosocial outcomes in breast reconstruction: two-year postoperative results from the Michigan Breast Reconstruction Outcomes Study. Ann Surg. 2008;247:1019–1028. doi: 10.1097/SLA.0b013e3181728a5c. [DOI] [PubMed] [Google Scholar]
- 2.Roostaeian J, Crisera C. Current options in breast reconstruction with or without radiotherapy. Curr Opin Obstet Gynecol. 2011;23:44–50. doi: 10.1097/GCO.0b013e328340e18a. [DOI] [PubMed] [Google Scholar]
- 3.Yueh JH, Slavin SA, Adesiyun T, et al. Patient satisfaction in postmastectomy breast reconstruction: a comparative evaluation of DIEP, TRAM, latissimus flap, and implant techniques. Plast Reconstr Surg. 2010;125:1585–1595. doi: 10.1097/PRS.0b013e3181cb6351. [DOI] [PubMed] [Google Scholar]
- 4.Christensen BO, Overgaard J, Kettner LO, et al. Long-term evaluation of postmastectomy breast reconstruction. Acta Oncol. 2011;50:1053–1061. doi: 10.3109/0284186X.2011.584554. [DOI] [PubMed] [Google Scholar]
- 5.American Society of Plastic Surgeons. 2012 Plastic Surgery Statistics Report. Available at: http://www.plasticsurgery.org/Documents/news-resources/statistics/2012-Plastic-Surgery-Statistics/full-plastic-surgery-statistics-report.pdf. Accessed October 31, 2013. [Google Scholar]
- 6.Sbitany H, Amalfi AN, Langstein HN. Preferences in choosing between breast reconstruction options: a survey of female plastic surgeons. Plast Reconstr Surg. 2009;124:1781–1789. doi: 10.1097/PRS.0b013e3181bf8056. [DOI] [PubMed] [Google Scholar]
- 7.Zhong T, McCarthy C, Min S, et al. Patient satisfaction and health-related quality of life after autologous tissue breast reconstruction: a prospective analysis of early postoperative outcomes. Cancer. 2012;118:1701–1709. doi: 10.1002/cncr.26417. [DOI] [PubMed] [Google Scholar]
- 8.Alderman AK, Atisha D, Streu R, et al. Patterns and correlates of postmastectomy breast reconstruction by U.S. plastic surgeons: results from a national survey. Plast Reconstr Surg. 2011;127:1796–1803. doi: 10.1097/PRS.0b013e31820cf183. [DOI] [PubMed] [Google Scholar]
- 9.Breuing KH, Warren SM. Immediate bilateral breast reconstruction with implants and inferolateral AlloDerm slings. Ann Plast Surg. 2005;55:232–239. doi: 10.1097/01.sap.0000168527.52472.3c. [DOI] [PubMed] [Google Scholar]
- 10.Breuing KH, Colwell AS. Inferolateral AlloDerm hammock for implant coverage in breast reconstruction. Ann Plast Surg. 2007;59:250–255. doi: 10.1097/SAP.0b013e31802f8426. [DOI] [PubMed] [Google Scholar]
- 11.Zienowicz RJ, Karacaoglu E. Implant-based breast reconstruction with allograft. Plast Reconstr Surg. 2007;120:373–381. doi: 10.1097/01.prs.0000267340.31742.1. [DOI] [PubMed] [Google Scholar]
- 12.Salzberg CA. Nonexpansive immediate breast reconstruction using human acellular tissue matrix graft (AlloDerm). Ann Plast Surg. 2006;57:1–5. doi: 10.1097/01.sap.0000214873.13102.9f. [DOI] [PubMed] [Google Scholar]
- 13.Sbitany H, Sandeen SN, Amalfi AN, et al. Acellular dermis-assisted prosthetic breast reconstruction versus complete submuscular coverage: a head-to-head comparison of outcomes. Plast Reconstr Surg. 2009;124:1735–1740. doi: 10.1097/PRS.0b013e3181bf803d. [DOI] [PubMed] [Google Scholar]
- 14.Gamboa-Bobadilla GM. Implant breast reconstruction using acellular dermal matrix. Ann Plast Surg. 2006;56:22–25. doi: 10.1097/01.sap.0000185460.31188.c1. [DOI] [PubMed] [Google Scholar]
- 15.Bindingnavele V, Gaon M, Ota KS, et al. Use of acellular cadaveric dermis and tissue expansion in postmastectomy breast reconstruction. J Plast Reconstr Aesthet Surg. 2007;60:1214–1218. doi: 10.1016/j.bjps.2007.03.015. [DOI] [PubMed] [Google Scholar]
- 16.McCarthy CM, Lee CN, Halvorson EG, et al. The use of acellular dermal matrices in two-stage expander/implant reconstruction: a multicenter, blinded, randomized controlled trial. Plast Reconstr Surg. 2012;130(5 Suppl 2):57S–66S. doi: 10.1097/PRS.0b013e31825f05b4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Scheflan M, Brown IM. Immediate implant-based breast reconstruction using variable lower pole support. In: Urban C, Rietjens M, editors. Oncoplastic and Reconstructive Breast Surgery. 1st ed. Milan, Italy: Springer-Verlag Italia; 2013. pp. 235–252. [Google Scholar]
- 18.Butterfield JL. 440 Consecutive immediate, implant-based, single-surgeon breast reconstructions in 281 patients: a comparison of early outcomes and costs between SurgiMend fetal bovine and AlloDerm human cadaveric acellular dermal matrices. Plast Reconstr Surg. 2013;131:940–951. doi: 10.1097/PRS.0b013e3182865ab3. [DOI] [PubMed] [Google Scholar]
- 19.Dieterich M, Paepke S, Zwiefel K, et al. Implant-based breast reconstruction using a titanium-coated polypropylene mesh (TiLOOP Bra): a multicenter study of 231 cases. Plast Reconstr Surg. 2013;132:8e–19e. doi: 10.1097/PRS.0b013e318290f8a0. [DOI] [PubMed] [Google Scholar]
- 20.Colwell AS, Damjanovic B, Zahedi B, et al. Retrospective review of 331 consecutive immediate single-stage implant reconstructions with acellular dermal matrix: indications, complications, trends, and costs. Plast Reconstr Surg. 2011;128:1170–1178. doi: 10.1097/PRS.0b013e318230c2f6. [DOI] [PubMed] [Google Scholar]
- 21.Spear SL, Spittler CJ. Breast reconstruction with implants and expanders. Plast Reconstr Surg. 2001;107:177–187; quiz 188. doi: 10.1097/00006534-200101000-00029. [DOI] [PubMed] [Google Scholar]
- 22.Hoppe IC, Yueh JH, Wei CH, et al. Complications following expander/implant breast reconstruction utilizing acellular dermal matrix: a systematic review and meta-analysis. Eplasty. 2011;11:e40. [PMC free article] [PubMed] [Google Scholar]
- 23.Sbitany H, Serletti JM. Acellular dermis-assisted prosthetic breast reconstruction: a systematic and critical review of efficacy and associated morbidity. Plast Reconstr Surg. 2011;128:1162–1169. doi: 10.1097/PRS.0b013e318230c29e. [DOI] [PubMed] [Google Scholar]
- 24.Hanna KR, DeGeorge BR, Jr, Mericli AF, et al. Comparison study of two types of expander-based breast reconstruction: acellular dermal matrix-assisted versus total submuscular placement. Ann Plast Surg. 2013;70:10–15. doi: 10.1097/SAP.0b013e31822f6765. [DOI] [PubMed] [Google Scholar]
- 25.Seth AK, Hirsch EM, Fine NA, et al. Utility of acellular dermis-assisted breast reconstruction in the setting of radiation: a comparative analysis. Plast Reconstr Surg. 2012;130:750–758. doi: 10.1097/PRS.0b013e318262f009. [DOI] [PubMed] [Google Scholar]
- 26.Topol BM, Dalton EF, Ponn T, et al. Immediate single-stage breast reconstruction using implants and human acellular dermal tissue matrix with adjustment of the lower pole of the breast to reduce unwanted lift. Ann Plast Surg. 2008;61:494–499. doi: 10.1097/SAP.0b013e31816d82d9. [DOI] [PubMed] [Google Scholar]
- 27.Salzberg CA, Ashikari AY, Koch RM, et al. An 8-year experience of direct-to-implant immediate breast reconstruction using human acellular dermal matrix (AlloDerm). Plast Reconstr Surg. 2011;127:514–524. doi: 10.1097/PRS.0b013e318200a961. [DOI] [PubMed] [Google Scholar]
- 28.Cassileth L, Kohanzadeh S, Amersi F. One-stage immediate breast reconstruction with implants: a new option for immediate reconstruction. Ann Plast Surg. 2012;69:134–138. doi: 10.1097/SAP.0b013e3182250c60. [DOI] [PubMed] [Google Scholar]
- 29.Forsberg CG, Kelly DA, Wood BC, et al. Aesthetic outcomes of acellular dermal matrix in tissue expander/implant-based breast reconstruction. Ann Plast Surg. 2014;72(6 Suppl 2):S116–S120. doi: 10.1097/SAP.0000000000000098. [DOI] [PubMed] [Google Scholar]
- 30.McCarthy CM, Mehrara BJ, Riedel E, et al. Predicting complications following expander/implant breast reconstruction: an outcomes analysis based on preoperative clinical risk. Plast Reconstr Surg. 2008;121:1886–1892. doi: 10.1097/PRS.0b013e31817151c4. [DOI] [PubMed] [Google Scholar]
- 31.Pannucci CJ, Antony AK, Wilkins EG. The impact of acellular dermal matrix on tissue expander/implant loss in breast reconstruction: an analysis of the tracking outcomes and operations in plastic surgery database. Plast Reconstr Surg. 2013;132:1–10. doi: 10.1097/PRS.0b013e318290f917. [DOI] [PubMed] [Google Scholar]
- 32.Davila AA, Seth AK, Wang E, et al. Human acellular dermis versus submuscular tissue expander breast reconstruction: a multivariate analysis of short-term complications. Arch Plast Surg. 2013;40:19–27. doi: 10.5999/aps.2013.40.1.19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Antony AK, McCarthy CM, Cordeiro PG, et al. Acellular human dermis implantation in 153 immediate two-stage tissue expander breast reconstructions: determining the incidence and significant predictors of complications. Plast Reconstr Surg. 2010;125:1606–1614. doi: 10.1097/PRS.0b013e3181d4fb2a. [DOI] [PubMed] [Google Scholar]
- 34.Parks JW, Hammond SE, Walsh WA, et al. Human acellular dermis versus no acellular dermis in tissue expansion breast reconstruction. Plast Reconstr Surg. 2012;130:739–746. doi: 10.1097/PRS.0b013e318262f06e. [DOI] [PubMed] [Google Scholar]
- 35.Peled AW, Foster RD, Garwood ER, et al. The effects of acellular dermal matrix in expander-implant breast reconstruction after total skin-sparing mastectomy: results of a prospective practice improvement study. Plast Reconstr Surg. 2012;129:901e–908e. doi: 10.1097/PRS.0b013e31824ec447. [DOI] [PubMed] [Google Scholar]
- 36.Weichman KE, Wilson SC, Weinstein AL, et al. The use of acellular dermal matrix in immediate two-stage tissue expander breast reconstruction. Plast Reconstr Surg. 2012;129:1049–1058. doi: 10.1097/PRS.0b013e31824a2acb. [DOI] [PubMed] [Google Scholar]
- 37.Vardanian AJ, Clayton JL, Roostaeian J, et al. Comparison of implant-based immediate breast reconstruction with and without acellular dermal matrix. Plast Reconstr Surg. 2011;128:403e–410e. doi: 10.1097/PRS.0b013e31822b6637. [DOI] [PubMed] [Google Scholar]
- 38.Liu AS, Kao HK, Reish RG, et al. Postoperative complications in prosthesis-based breast reconstruction using acellular dermal matrix. Plast Reconstr Surg. 2011;127:1755–1762. doi: 10.1097/PRS.0b013e31820cf233. [DOI] [PubMed] [Google Scholar]
- 39.Lanier ST, Wang ED, Chen JJ, et al. The effect of acellular dermal matrix use on complication rates in tissue expander/implant breast reconstruction. Ann Plast Surg. 2010;64:674–678. doi: 10.1097/SAP.0b013e3181dba892. [DOI] [PubMed] [Google Scholar]
- 40.Chun YS, Verma K, Rosen H, et al. Implant-based breast reconstruction using acellular dermal matrix and the risk of postoperative complications. Plast Reconstr Surg. 2010;125:429–436. doi: 10.1097/PRS.0b013e3181c82d90. [DOI] [PubMed] [Google Scholar]
- 41.Nguyen MD, Chen C, Colakoğlu S, et al. Infectious complications leading to explantation in implant-based breast reconstruction with AlloDerm. Eplasty. 2010;10:e48. [PMC free article] [PubMed] [Google Scholar]
- 42.Nahabedian MY. AlloDerm performance in the setting of prosthetic breast surgery, infection, and irradiation. Plast Reconstr Surg. 2009;124:1743–1753. doi: 10.1097/PRS.0b013e3181bf8087. [DOI] [PubMed] [Google Scholar]
- 43.Preminger BA, McCarthy CM, Hu QY, et al. The influence of AlloDerm on expander dynamics and complications in the setting of immediate tissue expander/implant reconstruction: a matched-cohort study. Ann Plast Surg. 2008;60:510–513. doi: 10.1097/SAP.0b013e31816f2836. [DOI] [PubMed] [Google Scholar]
- 44.Jansen LA, Macadam SA. The use of AlloDerm in postmastectomy alloplastic breast reconstruction: part I. A systematic review. Plast Reconstr Surg. 2011;127:2232–2244. doi: 10.1097/PRS.0b013e3182131c56. [DOI] [PubMed] [Google Scholar]
- 45.Kim JY, Davila AA, Persing S, et al. A meta-analysis of human acellular dermis and submuscular tissue expander breast reconstruction. Plast Reconstr Surg. 2012;129:28–41. doi: 10.1097/PRS.0b013e3182361fd6. [DOI] [PubMed] [Google Scholar]
- 46.Ho G, Nguyen TJ, Shahabi A, et al. A systematic review and meta-analysis of complications associated with acellular dermal matrix-assisted breast reconstruction. Ann Plast Surg. 2012;68:346–356. doi: 10.1097/SAP.0b013e31823f3cd9. [DOI] [PubMed] [Google Scholar]
- 47.Ibrahim AM, Shuster M, Koolen PG, et al. Analysis of the National Surgical Quality Improvement Program database in 19,100 patients undergoing implant-based breast reconstruction: complication rates with acellular dermal matrix. Plast Reconstr Surg. 2013;132:1057–1066. doi: 10.1097/PRS.0b013e3182a3beec. [DOI] [PubMed] [Google Scholar]
- 48.Martin L, O’Donoghue JM, Horgan K, et al. Association of Breast Surgery and the British Association of Plastic, Reconstructive and Aesthetic Surgeons. Acellular dermal matrix (ADM) assisted breast reconstruction procedures: joint guidelines from the Association of Breast Surgery and the British Association of Plastic, Reconstructive and Aesthetic Surgeons. Eur J Surg Oncol. 2013;39:425–429. doi: 10.1016/j.ejso.2012.12.012. [DOI] [PubMed] [Google Scholar]
- 49.Salzberg CA. Focus on technique: one-stage implant-based breast reconstruction. Plast Reconstr Surg. 2012;130(5 Suppl 2):95S–103S. doi: 10.1097/PRS.0b013e318262e1a1. [DOI] [PubMed] [Google Scholar]
- 50.Nahabedian MY. Acellular dermal matrices in primary breast reconstruction: principles, concepts, and indications. Plast Reconstr Surg. 2012;130:44S–53S. doi: 10.1097/PRS.0b013e31825f2215. [DOI] [PubMed] [Google Scholar]
- 51.Whitfield GA, Horan G, Irwin MS, et al. Incidence of severe capsular contracture following implant-based immediate breast reconstruction with or without postoperative chest wall radiotherapy using 40 Gray in 15 fractions. Radiother Oncol. 2009;90:141–147. doi: 10.1016/j.radonc.2008.09.023. [DOI] [PubMed] [Google Scholar]
- 52.Benediktsson K, Perbeck L. Capsular contracture around saline-filled and textured subcutaneously-placed implants in irradiated and non-irradiated breast cancer patients: five years of monitoring of a prospective trial. J Plast Reconstr Aesthet Surg. 2006;59:27–34. doi: 10.1016/j.bjps.2005.08.005. [DOI] [PubMed] [Google Scholar]
- 53.Behranwala KA, Dua RS, Ross GM, et al. The influence of radiotherapy on capsule formation and aesthetic outcome after immediate breast reconstruction using biodimensional anatomical expander implants. J Plast Reconstr Aesthet Surg. 2006;59:1043–1051. doi: 10.1016/j.bjps.2006.01.051. [DOI] [PubMed] [Google Scholar]
- 54.Spear SL, Seruya M, Rao SS, et al. Two-stage prosthetic breast reconstruction using AlloDerm including outcomes of different timings of radiotherapy. Plast Reconstr Surg. 2012;130:1–9. doi: 10.1097/PRS.0b013e3182547a45. [DOI] [PubMed] [Google Scholar]
- 55.Namnoum JD, Moyer HR. The role of acellular dermal matrix in the treatment of capsular contracture. Clin Plast Surg. 2012;39:127–136. doi: 10.1016/j.cps.2012.02.005. [DOI] [PubMed] [Google Scholar]
- 56.Rieger UM, Mesina J, Kalbermatten DF, et al. Bacterial biofilms and capsular contracture in patients with breast implants. Br J Surg. 2013;100:768–774. doi: 10.1002/bjs.9084. [DOI] [PubMed] [Google Scholar]
- 57.Basu CB, Leong M, Hicks MJ. Acellular cadaveric dermis decreases the inflammatory response in capsule formation in reconstructive breast surgery. Plast Reconstr Surg. 2010;126:1842–1847. doi: 10.1097/PRS.0b013e3181f44674. [DOI] [PubMed] [Google Scholar]
- 58.Stump A, Holton LH, 3rd, Connor J, et al. The use of acellular dermal matrix to prevent capsule formation around implants in a primate model. Plast Reconstr Surg. 2009;124:82–91. doi: 10.1097/PRS.0b013e3181ab112d. [DOI] [PubMed] [Google Scholar]
- 59.Cheng A, Lakhiani C, Saint-Cyr M. Treatment of capsular contracture using complete implant coverage by acellular dermal matrix: a novel technique. Plast Reconstr Surg. 2013;132:519–529. doi: 10.1097/PRS.0b013e31829acc1e. [DOI] [PubMed] [Google Scholar]
- 60.Handel N, Cordray T, Gutierrez J, et al. A long-term study of outcomes, complications, and patient satisfaction with breast implants. Plast Reconstr Surg. 2006;117:757–767. doi: 10.1097/01.prs.0000201457.00772.1d. discussion 768–772. [DOI] [PubMed] [Google Scholar]
- 61.Kronowitz SJ, Robb GL. Radiation therapy and breast reconstruction: a critical review of the literature. Plast Reconstr Surg. 2009;124:395–408. doi: 10.1097/PRS.0b013e3181aee987. [DOI] [PubMed] [Google Scholar]
- 62.Brooks S, Djohan R, Tendulkar R, et al. Risk factors for complications of radiation therapy on tissue expander breast reconstructions. Breast J. 2012;18:28–34. doi: 10.1111/j.1524-4741.2011.01182.x. [DOI] [PubMed] [Google Scholar]
- 63.Ascherman JA, Hanasono MM, Newman MI, et al. Implant reconstruction in breast cancer patients treated with radiation therapy. Plast Reconstr Surg. 2006;117:359–365. doi: 10.1097/01.prs.0000201478.64877.87. [DOI] [PubMed] [Google Scholar]
- 64.Clemens MW, Kronowitz SJ. Acellular dermal matrix in irradiated tissue expander/implant-based breast reconstruction: evidence-based review. Plast Reconstr Surg. 2012;130(5 Suppl 2):27S–34S. doi: 10.1097/PRS.0b013e318265f690. [DOI] [PubMed] [Google Scholar]
- 65.Israeli R, Feingold RS. Acellular dermal matrix in breast reconstruction in the setting of radiotherapy. Aesthet Surg J. 2011;31(7 Suppl):51S–64S. doi: 10.1177/1090820X11418089. [DOI] [PubMed] [Google Scholar]
- 66.Nava MB, Pennati AE, Lozza L, et al. Outcome of different timings of radiotherapy in implant-based breast reconstructions. Plast Reconstr Surg. 2011;128:353–359. doi: 10.1097/PRS.0b013e31821e6c10. [DOI] [PubMed] [Google Scholar]
- 67.de Blacam C, Momoh AO, Colakoglu S, et al. Cost analysis of implant-based breast reconstruction with acellular dermal matrix. Ann Plast Surg. 2012;69:516–520. doi: 10.1097/SAP.0b013e318217fb21. [DOI] [PubMed] [Google Scholar]
- 68.Jansen LA, Macadam SA. The use of AlloDerm in postmastectomy alloplastic breast reconstruction: part II. A cost analysis. Plast Reconstr Surg. 2011;127:2245–2254. doi: 10.1097/PRS.0b013e3182131c6b. [DOI] [PubMed] [Google Scholar]
- 69.Tessler O, Reish RG, Maman DY, et al. Beyond biologics: absorbable mesh as a low-cost, low-complication sling for implant-based breast reconstruction. Plast Reconstr Surg. 2014;133:90e–99e. doi: 10.1097/01.prs.0000437253.55457.63. [DOI] [PubMed] [Google Scholar]
- 70.Deeken CR, Melman L, Jenkins ED, et al. Histologic and biomechanical evaluation of crosslinked and non-crosslinked biologic meshes in a porcine model of ventral incisional hernia repair. J Am Coll Surg. 2011;212:880–888. doi: 10.1016/j.jamcollsurg.2011.01.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Cole PD, Stal D, Sharabi SE, et al. A comparative, long-term assessment of four soft tissue substitutes. Aesthet Surg J. 2011;31:674–681. doi: 10.1177/1090820X11415393. [DOI] [PubMed] [Google Scholar]
- 72.Melman L, Jenkins ED, Hamilton NA, et al. Early biocompatibility of crosslinked and non-crosslinked biologic meshes in a porcine model of ventral hernia repair. Hernia. 2011;15:157–164. doi: 10.1007/s10029-010-0770-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Glasberg SB, Light D. AlloDerm and Strattice in breast reconstruction: a comparison and techniques for optimizing outcomes. Plast Reconstr Surg. 2012;129:1223–1233. doi: 10.1097/PRS.0b013e31824ec429. [DOI] [PubMed] [Google Scholar]
- 74.Venturi ML, Mesbahi AN, Boehmler JH, 4th, et al. Evaluating sterile human acellular dermal matrix in immediate expander-based breast reconstruction: a multicenter, prospective, cohort study. Plast Reconstr Surg. 2013;131:9e–18e. doi: 10.1097/PRS.0b013e3182729d4f. [DOI] [PubMed] [Google Scholar]
- 75.Brooke S, Mesa J, Uluer M, et al. Complications in tissue expander breast reconstruction: a comparison of AlloDerm, DermaMatrix, and FlexHD acellular inferior pole dermal slings. Ann Plast Surg. 2012;69:347–349. doi: 10.1097/SAP.0b013e31824b3d97. [DOI] [PubMed] [Google Scholar]
- 76.Michelotti BF, Brooke S, Mesa J, et al. Analysis of clinically significant seroma formation in breast reconstruction using acellular dermal grafts. Ann Plast Surg. 2013;71:274–277. doi: 10.1097/SAP.0b013e3182923dc9. [DOI] [PubMed] [Google Scholar]
- 77.Liu DZ, Mathes DW, Neligan PC, et al. Comparison of outcomes using AlloDerm versus FlexHD for implant-based breast reconstruction. Ann Plast Surg. 2014;72:503–507. doi: 10.1097/SAP.0b013e318268a87c. [DOI] [PubMed] [Google Scholar]
- 78.Lee JH, Park KR, Kim TG, et al. A comparative study of CG CryoDerm and AlloDerm in direct-to-implant immediate breast reconstruction. Arch Plast Surg. 2013;40:374–379. doi: 10.5999/aps.2013.40.4.374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Buseman J, Wong L, Kemper P, et al. Comparison of sterile versus nonsterile acellular dermal matrices for breast reconstruction. Ann Plast Surg. 2013;70:497–499. doi: 10.1097/SAP.0b013e31827f52c8. [DOI] [PubMed] [Google Scholar]
- 80.Weichman KE, Wilson SC, Saadeh PB, et al. Sterile “ready-to-use” AlloDerm decreases postoperative infectious complications in patients undergoing immediate implant-based breast reconstruction with acellular dermal matrix. Plast Reconstr Surg. 2013;132:725–736. doi: 10.1097/PRS.0b013e31829fe35b. [DOI] [PubMed] [Google Scholar]
- 81.Cheng A, Saint-Cyr M. Comparison of different ADM materials in breast surgery. Clin Plast Surg. 2012;39:167–175. doi: 10.1016/j.cps.2012.02.004. [DOI] [PubMed] [Google Scholar]
- 82.Hjort H, Mathisen T, Alves A, et al. Three-year results from a preclinical implantation study of a long-term resorbable surgical mesh with time-dependent mechanical characteristics. Hernia. 2012;16:191–197. doi: 10.1007/s10029-011-0885-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Becker H, Lind JG., 2nd The use of synthetic mesh in reconstructive, revision, and cosmetic breast surgery. Aesthetic Plast Surg. 2013;37:914–921. doi: 10.1007/s00266-013-0171-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Horan RL, Toponarski I, Boepple HE, et al. Design and characterization of a scaffold for anterior cruciate ligament engineering. J Knee Surg. 2009;22:82–92. doi: 10.1055/s-0030-1247730. [DOI] [PubMed] [Google Scholar]
- 85.Horan RL, Bramono DS, Stanley JR, et al. Biological and biomechanical assessment of a long-term bioresorbable silk-derived surgical mesh in an abdominal body wall defect model. Hernia. 2009;13:189–199. doi: 10.1007/s10029-008-0459-9. [DOI] [PubMed] [Google Scholar]
- 86.Altman GH, McGill LD, Biber K, et al. 23rd Annual Meeting of the European Association of Plastic Surgeons. Munich, Germany:: European Association of Plastic Surgeons; 2012. May 24–26, In: Histology and histomorphology of the SeriScaffold™ device, a unique silk-derived, bioresorbable scaffold for soft tissue support, evaluated in a sheep model simulating human breast reconstruction. [Google Scholar]
- 87.Fine N, Lehfeldt M, Gross J, et al. Nottingham, UK: 2013. Sep 23–25, Clinical experience with SERI®, a silk-derived bioresorbable scaffold, in two-stage implant-based breast reconstruction: one year follow-up. Abstract presented at the 5th International Meeting of Oncoplastic and Reconstructive Breast Surgery; [Google Scholar]


