Abstract
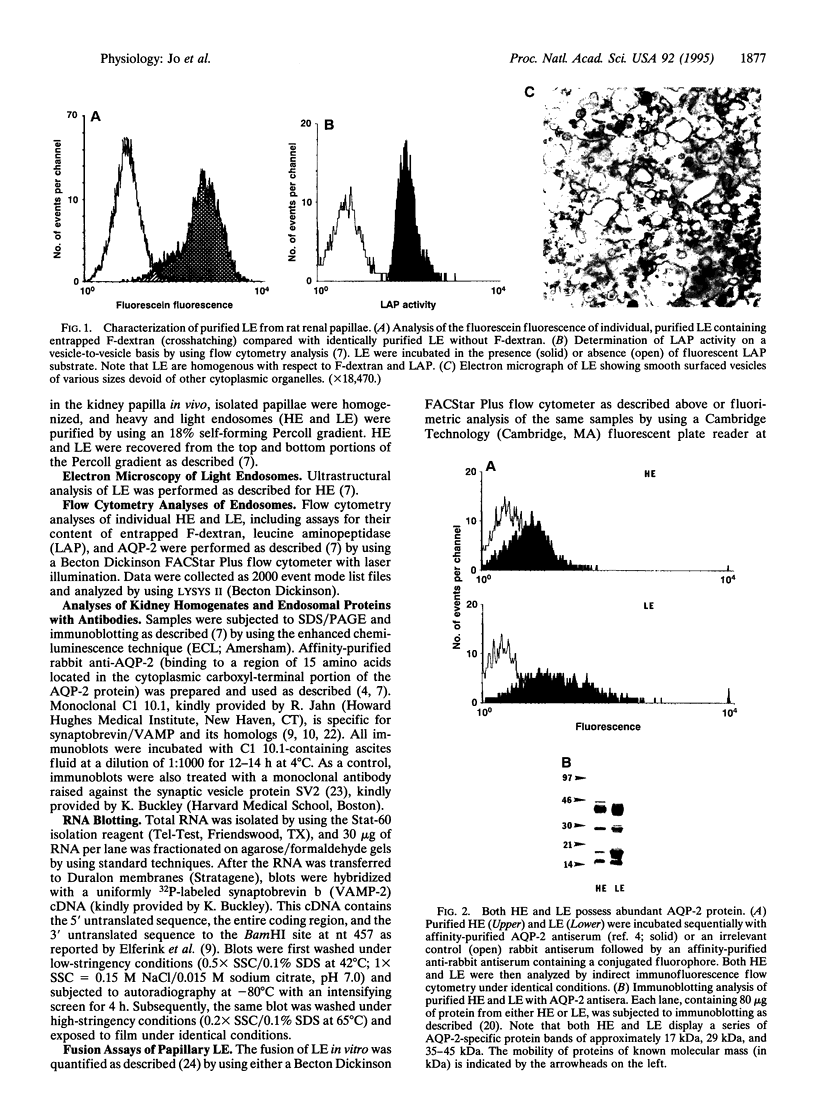
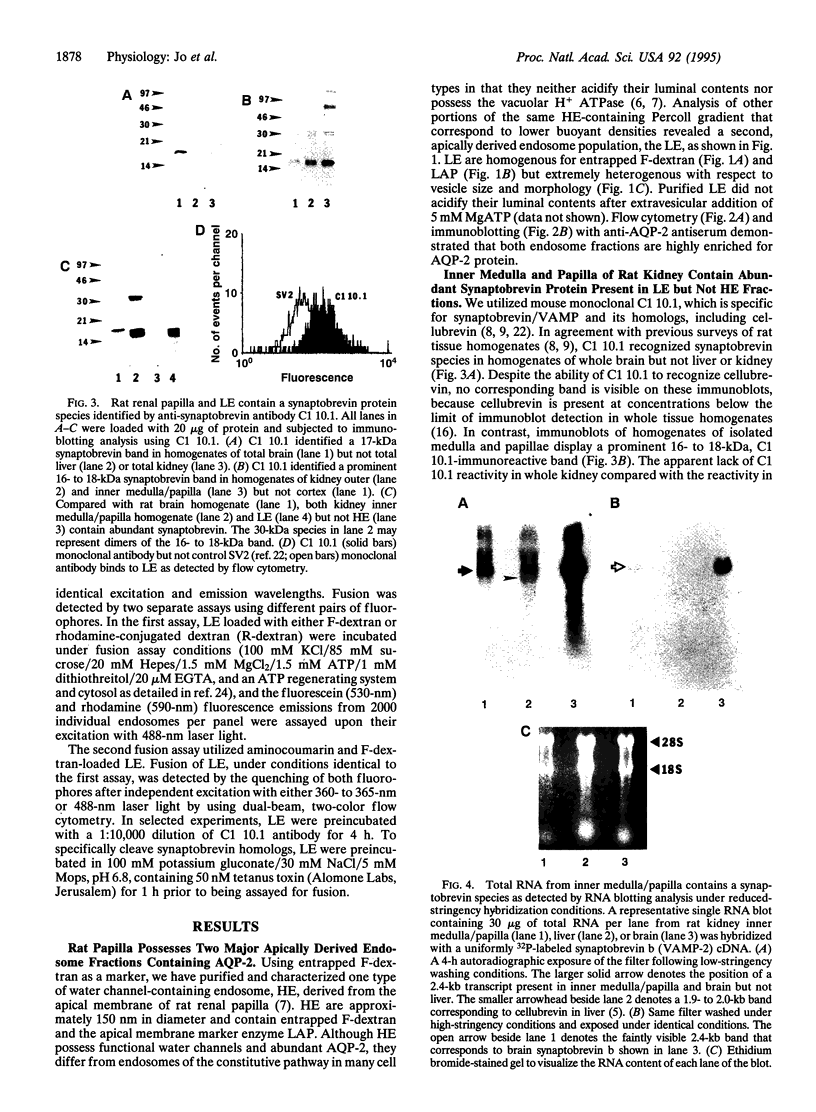
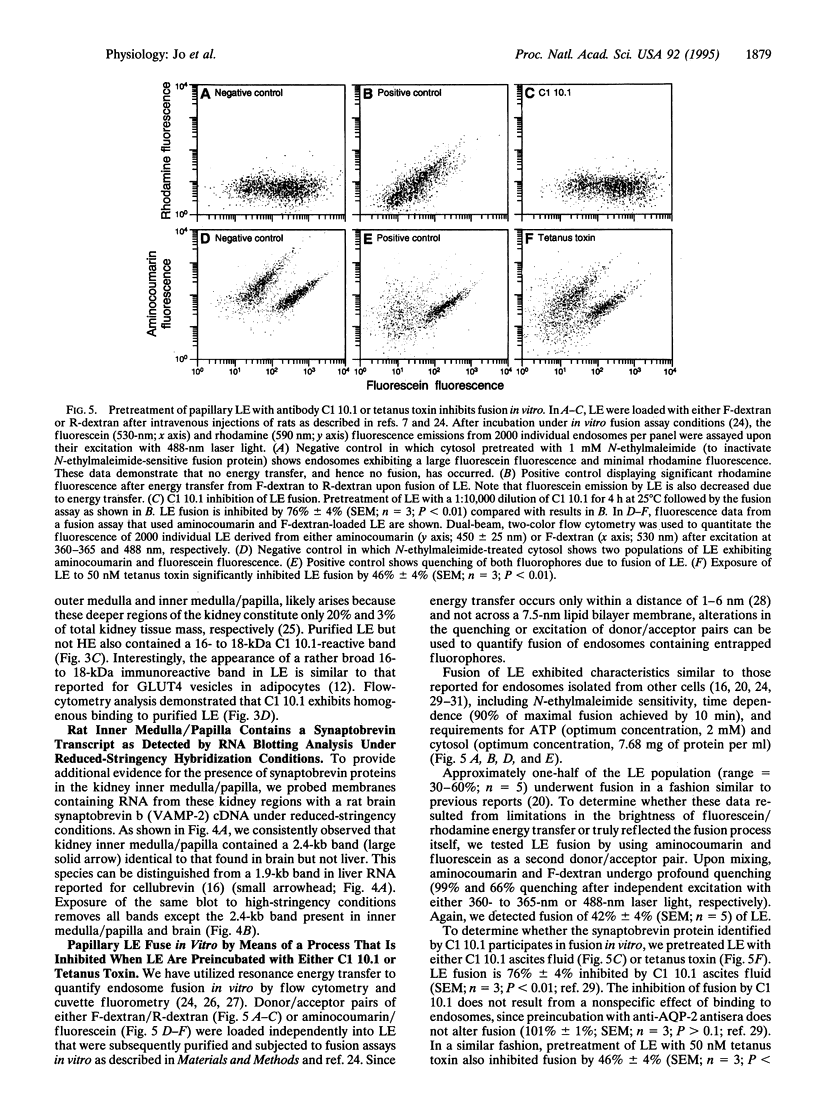
Antidiuretic hormone (ADH) regulates renal water excretion by altering the permeability of the collecting duct to water. ADH-responsive epithelial cells are the major cell type lining kidney tubules in the inner medulla and papilla. ADH modulates apical membrane water permeability by the insertion and removal of vesicles containing aquaporin collecting duct water channel protein (now termed AQP-2). To identify and characterize proteins responsible for trafficking of AQP-2-containing vesicles, we utilized antibody and cDNA probes to synaptobrevin b (also termed VAMP-2, for vesicle-associated membrane protein 2), a protein that mediates synaptic vesicle exocytosis in the brain and whose structural homologs are now considered to be components of a complex responsible for intracellular vesicle fusion in all cells. We now report that rat kidney inner medulla and papilla contain abundant synaptobrevin protein. Only light endosomes, one of two types of purified papillary AQP-2-containing endosomes, possess synaptobrevin. Light endosomes fuse in vitro by means of an ATP-dependent process that is significantly inhibited when endosomes are preincubated with either anti-synaptobrevin antibody or tetanus toxin. These data define a functional role for a synaptobrevin protein in the fusion of endosomes in vitro. The presence of abundant synaptobrevin proteins in endosomes containing AQP-2 water channels, as well as insulin-sensitive glucose transporters [Cain, C. C., Trimble, W. S. & Lienhard, G. E. (1992) J. Biol. Chem. 267, 11681-11684], and in cells of Malpighian tubules responsible for urine formation in insects [Chin, A. S., Burgess, R. W., Wong, B. R., Schwartz, T. L. & Scheller, R. H. (1993) Gene 131, 175-181] suggests a specialized role for synaptobrevin in vesicle-mediated membrane transport modulated by peptide hormones.
Full text
PDF

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Archer B. T., 3rd, Ozçelik T., Jahn R., Francke U., Südhof T. C. Structures and chromosomal localizations of two human genes encoding synaptobrevins 1 and 2. J Biol Chem. 1990 Oct 5;265(28):17267–17273. [PubMed] [Google Scholar]
- Baumert M., Maycox P. R., Navone F., De Camilli P., Jahn R. Synaptobrevin: an integral membrane protein of 18,000 daltons present in small synaptic vesicles of rat brain. EMBO J. 1989 Feb;8(2):379–384. doi: 10.1002/j.1460-2075.1989.tb03388.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bennett M. K., Scheller R. H. The molecular machinery for secretion is conserved from yeast to neurons. Proc Natl Acad Sci U S A. 1993 Apr 1;90(7):2559–2563. doi: 10.1073/pnas.90.7.2559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bennett M. K., Scheller R. H. The molecular machinery for secretion is conserved from yeast to neurons. Proc Natl Acad Sci U S A. 1993 Apr 1;90(7):2559–2563. doi: 10.1073/pnas.90.7.2559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bradley T. J. Membrane dynamics in insect malpighian tubules. Am J Physiol. 1989 Nov;257(5 Pt 2):R967–R972. doi: 10.1152/ajpregu.1989.257.5.R967. [DOI] [PubMed] [Google Scholar]
- Brown D. Membrane recycling and epithelial cell function. Am J Physiol. 1989 Jan;256(1 Pt 2):F1–12. doi: 10.1152/ajprenal.1989.256.1.F1. [DOI] [PubMed] [Google Scholar]
- Cain C. C., Trimble W. S., Lienhard G. E. Members of the VAMP family of synaptic vesicle proteins are components of glucose transporter-containing vesicles from rat adipocytes. J Biol Chem. 1992 Jun 15;267(17):11681–11684. [PubMed] [Google Scholar]
- Chin A. C., Burgess R. W., Wong B. R., Schwarz T. L., Scheller R. H. Differential expression of transcripts from syb, a Drosophila melanogaster gene encoding VAMP (synaptobrevin) that is abundant in non-neuronal cells. Gene. 1993 Sep 15;131(2):175–181. doi: 10.1016/0378-1119(93)90291-a. [DOI] [PubMed] [Google Scholar]
- De Camilli P., Jahn R. Pathways to regulated exocytosis in neurons. Annu Rev Physiol. 1990;52:625–645. doi: 10.1146/annurev.ph.52.030190.003205. [DOI] [PubMed] [Google Scholar]
- Deen P. M., Verdijk M. A., Knoers N. V., Wieringa B., Monnens L. A., van Os C. H., van Oost B. A. Requirement of human renal water channel aquaporin-2 for vasopressin-dependent concentration of urine. Science. 1994 Apr 1;264(5155):92–95. doi: 10.1126/science.8140421. [DOI] [PubMed] [Google Scholar]
- DiAntonio A., Burgess R. W., Chin A. C., Deitcher D. L., Scheller R. H., Schwarz T. L. Identification and characterization of Drosophila genes for synaptic vesicle proteins. J Neurosci. 1993 Nov;13(11):4924–4935. doi: 10.1523/JNEUROSCI.13-11-04924.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elferink L. A., Trimble W. S., Scheller R. H. Two vesicle-associated membrane protein genes are differentially expressed in the rat central nervous system. J Biol Chem. 1989 Jul 5;264(19):11061–11064. [PubMed] [Google Scholar]
- Feany M. B., Lee S., Edwards R. H., Buckley K. M. The synaptic vesicle protein SV2 is a novel type of transmembrane transporter. Cell. 1992 Sep 4;70(5):861–867. doi: 10.1016/0092-8674(92)90319-8. [DOI] [PubMed] [Google Scholar]
- Fushimi K., Uchida S., Hara Y., Hirata Y., Marumo F., Sasaki S. Cloning and expression of apical membrane water channel of rat kidney collecting tubule. Nature. 1993 Feb 11;361(6412):549–552. doi: 10.1038/361549a0. [DOI] [PubMed] [Google Scholar]
- Galli T., Chilcote T., Mundigl O., Binz T., Niemann H., De Camilli P. Tetanus toxin-mediated cleavage of cellubrevin impairs exocytosis of transferrin receptor-containing vesicles in CHO cells. J Cell Biol. 1994 Jun;125(5):1015–1024. doi: 10.1083/jcb.125.5.1015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goda Y., Pfeffer S. R. Cell-free systems to study vesicular transport along the secretory and endocytic pathways. FASEB J. 1989 Nov;3(13):2488–2495. doi: 10.1096/fasebj.3.13.2680705. [DOI] [PubMed] [Google Scholar]
- Harris H. W., Jr, Strange K., Zeidel M. L. Current understanding of the cellular biology and molecular structure of the antidiuretic hormone-stimulated water transport pathway. J Clin Invest. 1991 Jul;88(1):1–8. doi: 10.1172/JCI115263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harris H. W., Jr, Zeidel M. L., Jo I., Hammond T. G. Characterization of purified endosomes containing the antidiuretic hormone-sensitive water channel from rat renal papilla. J Biol Chem. 1994 Apr 22;269(16):11993–12000. [PubMed] [Google Scholar]
- Kelly R. B. Secretion. A question of endosomes. Nature. 1993 Aug 5;364(6437):487–488. doi: 10.1038/364487a0. [DOI] [PubMed] [Google Scholar]
- Lacy E. R. The mammalian renal pelvis: physiological implications from morphometric analyses. Anat Embryol (Berl) 1980;160(2):131–144. doi: 10.1007/BF00301856. [DOI] [PubMed] [Google Scholar]
- Link E., McMahon H., Fischer von Mollard G., Yamasaki S., Niemann H., Südhof T. C., Jahn R. Cleavage of cellubrevin by tetanus toxin does not affect fusion of early endosomes. J Biol Chem. 1993 Sep 5;268(25):18423–18426. [PubMed] [Google Scholar]
- McMahon H. T., Ushkaryov Y. A., Edelmann L., Link E., Binz T., Niemann H., Jahn R., Südhof T. C. Cellubrevin is a ubiquitous tetanus-toxin substrate homologous to a putative synaptic vesicle fusion protein. Nature. 1993 Jul 22;364(6435):346–349. doi: 10.1038/364346a0. [DOI] [PubMed] [Google Scholar]
- Nielsen S., DiGiovanni S. R., Christensen E. I., Knepper M. A., Harris H. W. Cellular and subcellular immunolocalization of vasopressin-regulated water channel in rat kidney. Proc Natl Acad Sci U S A. 1993 Dec 15;90(24):11663–11667. doi: 10.1073/pnas.90.24.11663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Protopopov V., Govindan B., Novick P., Gerst J. E. Homologs of the synaptobrevin/VAMP family of synaptic vesicle proteins function on the late secretory pathway in S. cerevisiae. Cell. 1993 Sep 10;74(5):855–861. doi: 10.1016/0092-8674(93)90465-3. [DOI] [PubMed] [Google Scholar]
- Rothman J. E., Orci L. Molecular dissection of the secretory pathway. Nature. 1992 Jan 30;355(6359):409–415. doi: 10.1038/355409a0. [DOI] [PubMed] [Google Scholar]
- Sabolić I., Wuarin F., Shi L. B., Verkman A. S., Ausiello D. A., Gluck S., Brown D. Apical endosomes isolated from kidney collecting duct principal cells lack subunits of the proton pumping ATPase. J Cell Biol. 1992 Oct;119(1):111–122. doi: 10.1083/jcb.119.1.111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schiavo G., Benfenati F., Poulain B., Rossetto O., Polverino de Laureto P., DasGupta B. R., Montecucco C. Tetanus and botulinum-B neurotoxins block neurotransmitter release by proteolytic cleavage of synaptobrevin. Nature. 1992 Oct 29;359(6398):832–835. doi: 10.1038/359832a0. [DOI] [PubMed] [Google Scholar]
- Szöllösi J., Trón L., Damjanovich S., Helliwell S. H., Arndt-Jovin D., Jovin T. M. Fluorescence energy transfer measurements on cell surfaces: a critical comparison of steady-state fluorimetric and flow cytometric methods. Cytometry. 1984 Mar;5(2):210–216. doi: 10.1002/cyto.990050216. [DOI] [PubMed] [Google Scholar]
- Söllner T., Whiteheart S. W., Brunner M., Erdjument-Bromage H., Geromanos S., Tempst P., Rothman J. E. SNAP receptors implicated in vesicle targeting and fusion. Nature. 1993 Mar 25;362(6418):318–324. doi: 10.1038/362318a0. [DOI] [PubMed] [Google Scholar]
- Südhof T. C., Baumert M., Perin M. S., Jahn R. A synaptic vesicle membrane protein is conserved from mammals to Drosophila. Neuron. 1989 May;2(5):1475–1481. doi: 10.1016/0896-6273(89)90193-1. [DOI] [PubMed] [Google Scholar]
- Trimble W. S., Cowan D. M., Scheller R. H. VAMP-1: a synaptic vesicle-associated integral membrane protein. Proc Natl Acad Sci U S A. 1988 Jun;85(12):4538–4542. doi: 10.1073/pnas.85.12.4538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Young I. T. Proof without prejudice: use of the Kolmogorov-Smirnov test for the analysis of histograms from flow systems and other sources. J Histochem Cytochem. 1977 Jul;25(7):935–941. doi: 10.1177/25.7.894009. [DOI] [PubMed] [Google Scholar]