Abstract
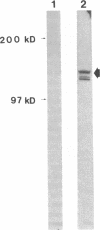
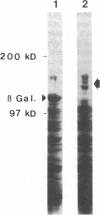
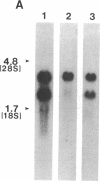
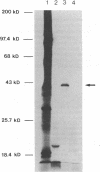
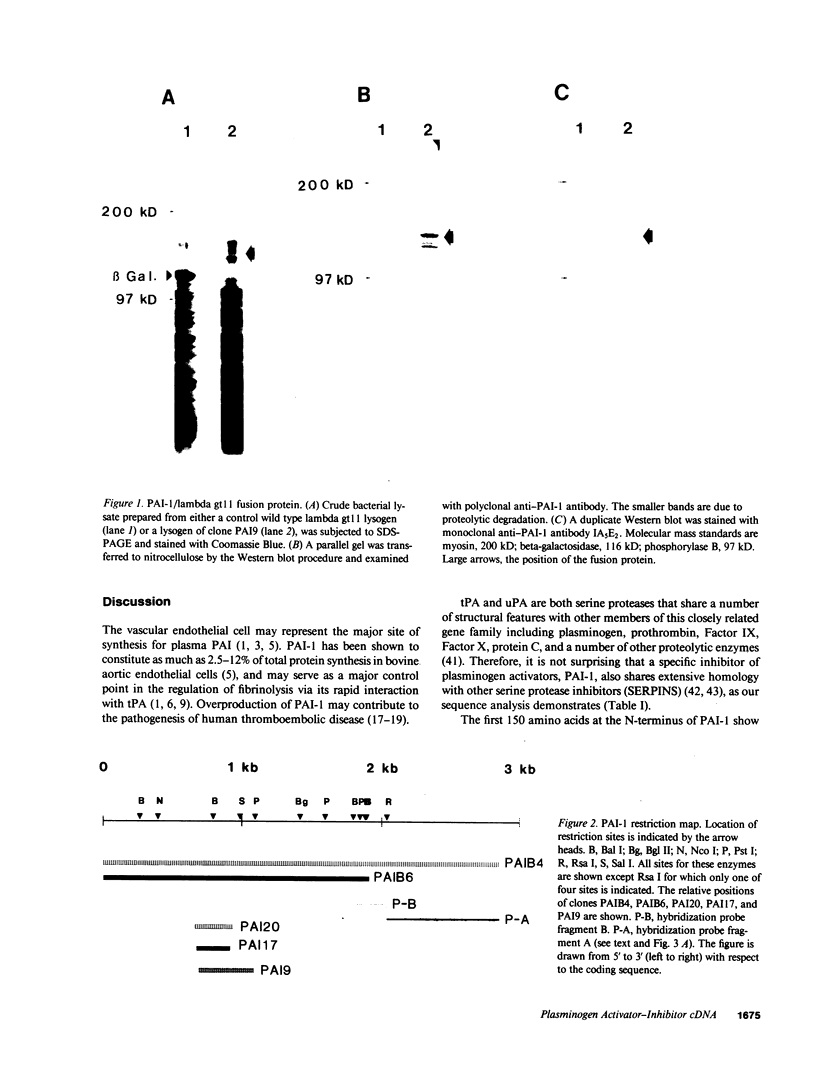
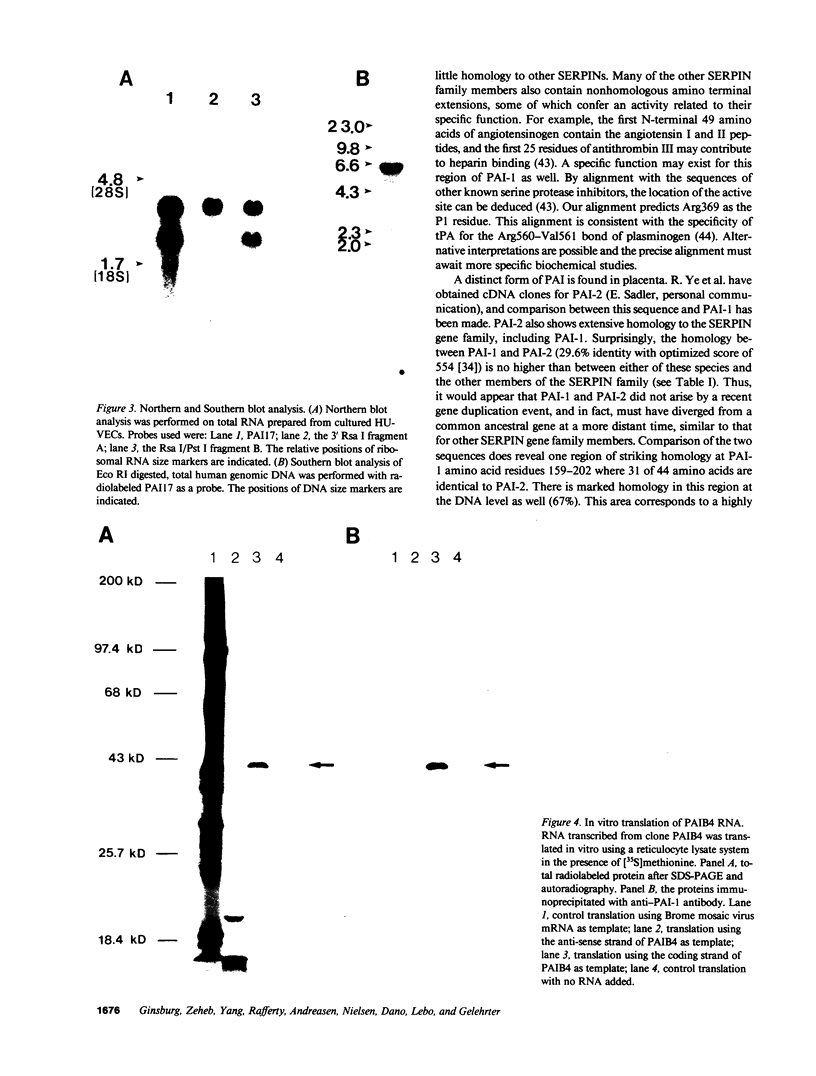
Full-length cDNA for plasminogen activator inhibitor (PAI-1) was isolated from a human umbilical vein endothelial cell (HUVEC) lambda gt11 cDNA library. Three overlapping clones were identified by immunologic screening of 10(6) recombinant phage using a rabbit anti-human fibrosarcoma PAI-1 antiserum. The fusion proteins encoded by these three clones also react strongly with a monoclonal mouse anti-human fibrosarcoma PAI-1 antibody. By nucleotide sequence analysis, PAI-1 cDNA encodes a protein containing 402 amino acids with a predicted, nonglycosylated molecular mass of 45 kD. Identity of this material as authentic PAI-1 was confirmed by the presence of high level homology with the primary amino acid sequence of an internal peptide prepared from purified rat hepatoma PAI-1. The predicted amino acid sequence also reveals extensive homology with other members of the serine protease inhibitor gene family. Cultured HUVECs contain two PAI-1 mRNA species, both encoded by a single gene, differing by 1 kb in the 3' untranslated region. The PAI-1 gene is located on human chromosome 7.
Full text
PDF

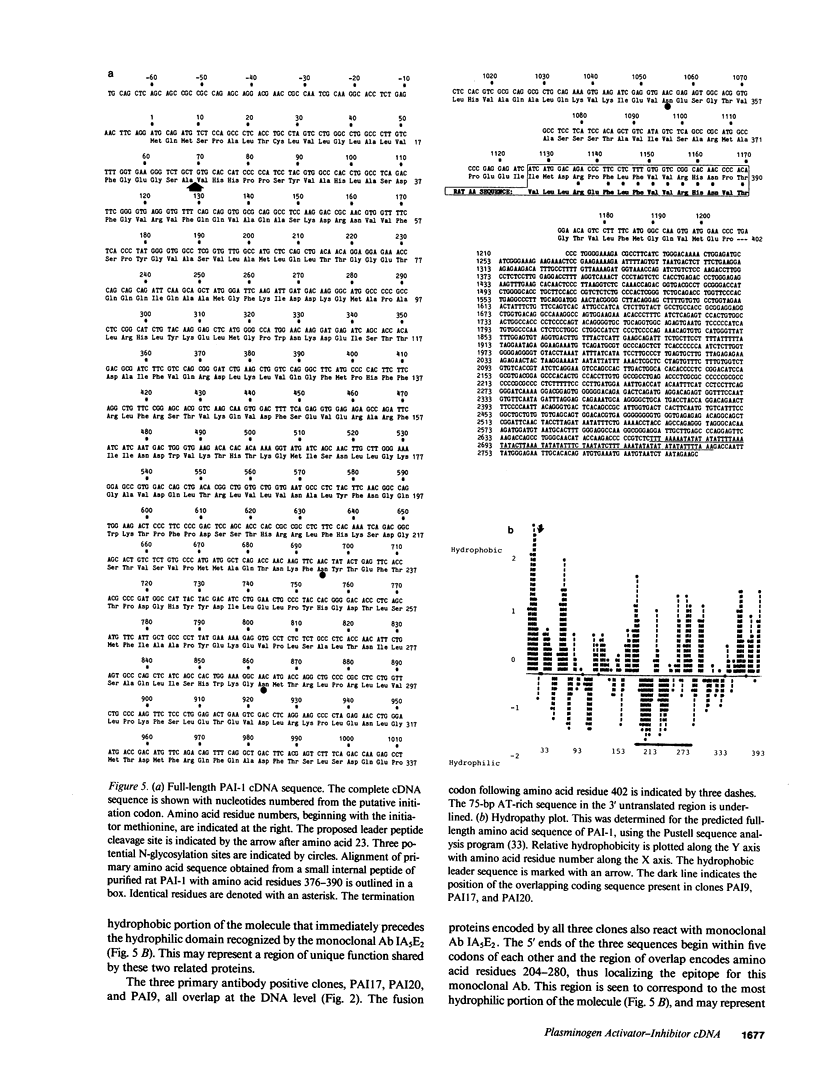



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Andreasen P. A., Christensen T. H., Huang J. Y., Nielsen L. S., Wilson E. L., Danø K. Hormonal regulation of extracellular plasminogen activators and Mr approximately 54,000 plasminogen activator inhibitor in human neoplastic cell lines, studied with monoclonal antibodies. Mol Cell Endocrinol. 1986 May;45(2-3):137–147. doi: 10.1016/0303-7207(86)90141-3. [DOI] [PubMed] [Google Scholar]
- Andreasen P. A., Nielsen L. S., Kristensen P., Grøndahl-Hansen J., Skriver L., Danø K. Plasminogen activator inhibitor from human fibrosarcoma cells binds urokinase-type plasminogen activator, but not its proenzyme. J Biol Chem. 1986 Jun 15;261(17):7644–7651. [PubMed] [Google Scholar]
- Astedt B., Lecander I., Brodin T., Lundblad A., Löw K. Purification of a specific placental plasminogen activator inhibitor by monoclonal antibody and its complex formation with plasminogen activator. Thromb Haemost. 1985 Feb 18;53(1):122–125. [PubMed] [Google Scholar]
- Bause E. Structural requirements of N-glycosylation of proteins. Studies with proline peptides as conformational probes. Biochem J. 1983 Feb 1;209(2):331–336. doi: 10.1042/bj2090331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burnette W. N. "Western blotting": electrophoretic transfer of proteins from sodium dodecyl sulfate--polyacrylamide gels to unmodified nitrocellulose and radiographic detection with antibody and radioiodinated protein A. Anal Biochem. 1981 Apr;112(2):195–203. doi: 10.1016/0003-2697(81)90281-5. [DOI] [PubMed] [Google Scholar]
- Caput D., Beutler B., Hartog K., Thayer R., Brown-Shimer S., Cerami A. Identification of a common nucleotide sequence in the 3'-untranslated region of mRNA molecules specifying inflammatory mediators. Proc Natl Acad Sci U S A. 1986 Mar;83(6):1670–1674. doi: 10.1073/pnas.83.6.1670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coleman P. L., Patel P. D., Cwikel B. J., Rafferty U. M., Sznycer-Laszuk R., Gelehrter T. D. Characterization of the dexamethasone-induced inhibitor of plasminogen activator in HTC hepatoma cells. J Biol Chem. 1986 Mar 25;261(9):4352–4357. [PubMed] [Google Scholar]
- Collins T., Ginsburg D., Boss J. M., Orkin S. H., Pober J. S. Cultured human endothelial cells express platelet-derived growth factor B chain: cDNA cloning and structural analysis. Nature. 1985 Aug 22;316(6030):748–750. doi: 10.1038/316748a0. [DOI] [PubMed] [Google Scholar]
- Colucci M., Paramo J. A., Collen D. Generation in plasma of a fast-acting inhibitor of plasminogen activator in response to endotoxin stimulation. J Clin Invest. 1985 Mar;75(3):818–824. doi: 10.1172/JCI111777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cwikel B. J., Barouski-Miller P. A., Coleman P. L., Gelehrter T. D. Dexamethasone induction of an inhibitor of plasminogen activator in HTC hepatoma cells. J Biol Chem. 1984 Jun 10;259(11):6847–6851. [PubMed] [Google Scholar]
- Dale R. M., McClure B. A., Houchins J. P. A rapid single-stranded cloning strategy for producing a sequential series of overlapping clones for use in DNA sequencing: application to sequencing the corn mitochondrial 18 S rDNA. Plasmid. 1985 Jan;13(1):31–40. doi: 10.1016/0147-619x(85)90053-8. [DOI] [PubMed] [Google Scholar]
- Deininger P. L., Jolly D. J., Rubin C. M., Friedmann T., Schmid C. W. Base sequence studies of 300 nucleotide renatured repeated human DNA clones. J Mol Biol. 1981 Sep 5;151(1):17–33. doi: 10.1016/0022-2836(81)90219-9. [DOI] [PubMed] [Google Scholar]
- Doggett P. E., Blattner F. R. Personal access to sequence databases on personal computers. Nucleic Acids Res. 1986 Jan 10;14(1):611–619. doi: 10.1093/nar/14.1.611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Doolittle R. F. Angiotensinogen is related to the antitrypsin-antithrombin-ovalbumin family. Science. 1983 Oct 28;222(4622):417–419. doi: 10.1126/science.6604942. [DOI] [PubMed] [Google Scholar]
- Emeis J. J., Kooistra T. Interleukin 1 and lipopolysaccharide induce an inhibitor of tissue-type plasminogen activator in vivo and in cultured endothelial cells. J Exp Med. 1986 May 1;163(5):1260–1266. doi: 10.1084/jem.163.5.1260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Erickson L. A., Ginsberg M. H., Loskutoff D. J. Detection and partial characterization of an inhibitor of plasminogen activator in human platelets. J Clin Invest. 1984 Oct;74(4):1465–1472. doi: 10.1172/JCI111559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Erickson L. A., Hekman C. M., Loskutoff D. J. The primary plasminogen-activator inhibitors in endothelial cells, platelets, serum, and plasma are immunologically related. Proc Natl Acad Sci U S A. 1985 Dec;82(24):8710–8714. doi: 10.1073/pnas.82.24.8710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Erickson L. A., Schleef R. R., Ny T., Loskutoff D. J. The fibrinolytic system of the vascular wall. Clin Haematol. 1985 Jun;14(2):513–530. [PubMed] [Google Scholar]
- Gelehrter T. D., Barouski-Miller P. A., Coleman P. L., Cwikel B. J. Hormonal regulation of plasminogen activator in rat hepatoma cells. Mol Cell Biochem. 1983;53-54(1-2):11–21. doi: 10.1007/BF00225243. [DOI] [PubMed] [Google Scholar]
- Gelehrter T. D., Sznycer-Laszuk R. Thrombin induction of plasminogen activator-inhibitor in cultured human endothelial cells. J Clin Invest. 1986 Jan;77(1):165–169. doi: 10.1172/JCI112271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ginsburg D., Handin R. I., Bonthron D. T., Donlon T. A., Bruns G. A., Latt S. A., Orkin S. H. Human von Willebrand factor (vWF): isolation of complementary DNA (cDNA) clones and chromosomal localization. Science. 1985 Jun 21;228(4706):1401–1406. doi: 10.1126/science.3874428. [DOI] [PubMed] [Google Scholar]
- Hamsten A., Wiman B., de Faire U., Blombäck M. Increased plasma levels of a rapid inhibitor of tissue plasminogen activator in young survivors of myocardial infarction. N Engl J Med. 1985 Dec 19;313(25):1557–1563. doi: 10.1056/NEJM198512193132501. [DOI] [PubMed] [Google Scholar]
- Hawkes R. Identification of concanavalin A-binding proteins after sodium dodecyl sulfate--gel electrophoresis and protein blotting. Anal Biochem. 1982 Jun;123(1):143–146. doi: 10.1016/0003-2697(82)90634-0. [DOI] [PubMed] [Google Scholar]
- Kluft C., Jie A. F., Sprengers E. D., Verheijen J. H. Identification of a reversible inhibitor of plasminogen activators in blood plasma. FEBS Lett. 1985 Oct 14;190(2):315–318. doi: 10.1016/0014-5793(85)81309-0. [DOI] [PubMed] [Google Scholar]
- Koide T., Ikenaka T. Studies on soybean trypsin inhibitors. 1. Fragmentation of soybean trypsin inhibitor (Kunitz) by limited proteolysis and by chemical cleavage. Eur J Biochem. 1973 Feb 1;32(3):401–407. doi: 10.1111/j.1432-1033.1973.tb02622.x. [DOI] [PubMed] [Google Scholar]
- Kozak M. Compilation and analysis of sequences upstream from the translational start site in eukaryotic mRNAs. Nucleic Acids Res. 1984 Jan 25;12(2):857–872. doi: 10.1093/nar/12.2.857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kruithof E. K., Tran-Thang C., Ransijn A., Bachmann F. Demonstration of a fast-acting inhibitor of plasminogen activators in human plasma. Blood. 1984 Oct;64(4):907–913. [PubMed] [Google Scholar]
- Lebo R. V., Gorin F., Fletterick R. J., Kao F. T., Cheung M. C., Bruce B. D., Kan Y. W. High-resolution chromosome sorting and DNA spot-blot analysis assign McArdle's syndrome to chromosome 11. Science. 1984 Jul 6;225(4657):57–59. doi: 10.1126/science.6587566. [DOI] [PubMed] [Google Scholar]
- Meijlink F., Curran T., Miller A. D., Verma I. M. Removal of a 67-base-pair sequence in the noncoding region of protooncogene fos converts it to a transforming gene. Proc Natl Acad Sci U S A. 1985 Aug;82(15):4987–4991. doi: 10.1073/pnas.82.15.4987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Messing J. New M13 vectors for cloning. Methods Enzymol. 1983;101:20–78. doi: 10.1016/0076-6879(83)01005-8. [DOI] [PubMed] [Google Scholar]
- Nielsen L. S., Andreasen P. A., Grøndahl-Hansen J., Huang J. Y., Kristensen P., Danø K. Monoclonal antibodies to human 54,000 molecular weight plasminogen activator inhibitor from fibrosarcoma cells--inhibitor neutralization and one-step affinity purification. Thromb Haemost. 1986 Apr 30;55(2):206–212. [PubMed] [Google Scholar]
- Patthy L. Evolution of the proteases of blood coagulation and fibrinolysis by assembly from modules. Cell. 1985 Jul;41(3):657–663. doi: 10.1016/s0092-8674(85)80046-5. [DOI] [PubMed] [Google Scholar]
- Perlman D., Halvorson H. O. A putative signal peptidase recognition site and sequence in eukaryotic and prokaryotic signal peptides. J Mol Biol. 1983 Jun 25;167(2):391–409. doi: 10.1016/s0022-2836(83)80341-6. [DOI] [PubMed] [Google Scholar]
- Prochownik E. V., Bock S. C., Orkin S. H. Intron structure of the human antithrombin III gene differs from that of other members of the serine protease inhibitor superfamily. J Biol Chem. 1985 Aug 15;260(17):9608–9612. [PubMed] [Google Scholar]
- Pustell J., Kafatos F. C. A convenient and adaptable microcomputer environment for DNA and protein sequence manipulation and analysis. Nucleic Acids Res. 1986 Jan 10;14(1):479–488. doi: 10.1093/nar/14.1.479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanger F., Nicklen S., Coulson A. R. DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5463–5467. doi: 10.1073/pnas.74.12.5463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sprengers E. D., Princen H. M., Kooistra T., van Hinsbergh V. W. Inhibition of plasminogen activators by conditioned medium of human hepatocytes and hepatoma cell line Hep G2. J Lab Clin Med. 1985 Jun;105(6):751–758. [PubMed] [Google Scholar]
- Vassalli J. D., Dayer J. M., Wohlwend A., Belin D. Concomitant secretion of prourokinase and of a plasminogen activator-specific inhibitor by cultured human monocytes-macrophages. J Exp Med. 1984 Jun 1;159(6):1653–1668. doi: 10.1084/jem.159.6.1653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Verstraete M., Collen D. Thrombolytic therapy in the eighties. Blood. 1986 Jun;67(6):1529–1541. [PubMed] [Google Scholar]
- Wiman B., Ljungberg B., Chmielewska J., Urdén G., Blombäck M., Johnsson H. The role of the fibrinolytic system in deep vein thrombosis. J Lab Clin Med. 1985 Feb;105(2):265–270. [PubMed] [Google Scholar]
- van Hinsbergh V. W., Bertina R. M., van Wijngaarden A., van Tilburg N. H., Emeis J. J., Haverkate F. Activated protein C decreases plasminogen activator-inhibitor activity in endothelial cell-conditioned medium. Blood. 1985 Feb;65(2):444–451. [PubMed] [Google Scholar]
- van Mourik J. A., Lawrence D. A., Loskutoff D. J. Purification of an inhibitor of plasminogen activator (antiactivator) synthesized by endothelial cells. J Biol Chem. 1984 Dec 10;259(23):14914–14921. [PubMed] [Google Scholar]