Abstract
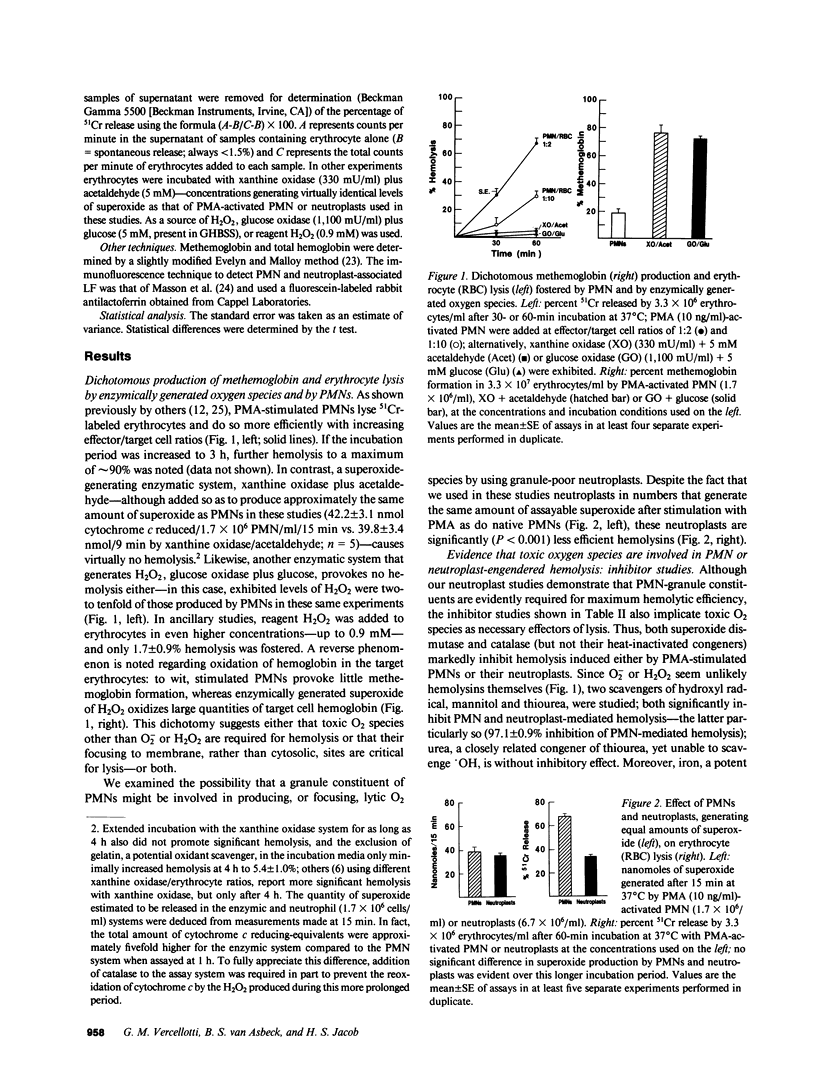
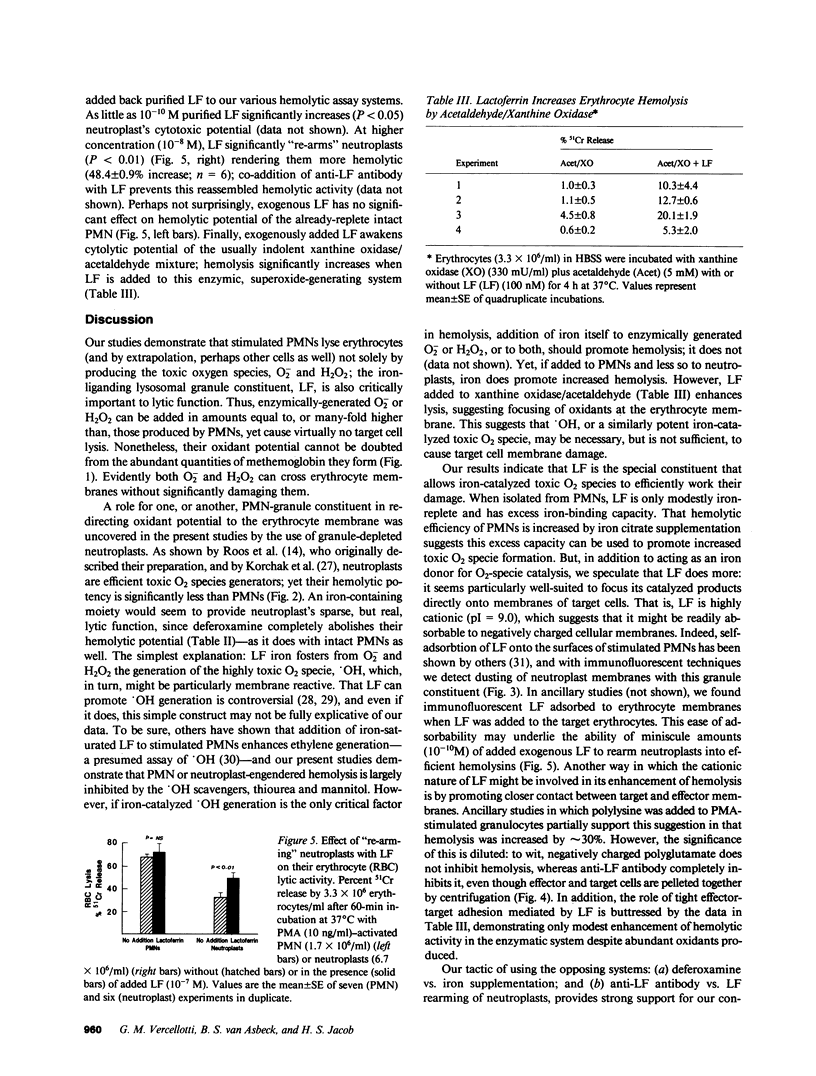
Human neutrophils (PMN), when stimulated with such chemotaxins as phorbol myristate acetate (PMA), destroy erythrocytes and other targets. Cytotoxicity depends on PMN-generated reactive oxygen metabolites, yet the exact toxic specie and its mode of production is a matter of some dispute. Using 51Cr-labeled erythrocytes as targets, we compared various reactive-O2 generating systems for their abilities to lyse erythrocytes as well as to oxidize hemoglobin to methemoglobin. PMA-activated PMNs or xanthine oxidase plus acetaldehyde were added to target erythrocytes in amounts that provided similar levels of superoxide. PMNs lysed 68.3 +/- 2.9% (SEM) of targets, whereas the xanthine oxidase system was virtually impotent (2.3 +/- 0.8%). In contrast, methemoglobin formation by xanthine oxidase plus acetaldehyde was significantly greater than that caused by stimulated PMNs (P less than 0.001). A similar dichotomy was noted with added reagent H2O2 or the H2O2-generating system, glucose plus glucose oxidase; neither of these caused 51Cr release, but induced 10-70% methemoglobin formation. Thus, although O2- and H2O2 can cross the erythrocyte membrane and rapidly oxidize hemoglobin, they do so evidently without damaging the cell membrane. That a granule constituent of PMNs is required to promote target cell lysis was suggested by the fact that agranular PMN cytoplasts (neutroplasts), although added to generate equal amounts of O2- as intact PMNs, were significantly less lytic to target erythrocytes (P less than 0.01). Iron was shown to be directly involved in lytic efficiency by supplementation studies with 2 microM iron citrate; such supplementation increased PMN cytotoxicity by approximately 30%, but had much less effect on erythrocyte lysis by neutroplasts (approximately 3% increase), and no effect on lysis in the enzymatic oxygen radical-generating systems. These results suggest a critical role for an iron-liganding moiety that is abundantly present in PMN, marginally so in neutroplasts, and not at all in purified enzymatic systems--a moiety that we presume catalyzes very toxic O2 specie generation in the vicinity of juxtaposed erythrocyte targets. The obvious candidate is lactoferrin (LF), and indeed, antilactoferrin IgG, but not nonspecific IgG, reduced PMN cytotoxicity by greater than 85%. Re-adding 10(-8) M pure LF to neutroplasts increased their ability to promote hemolysis by 48.4 +/- 0.9%--to a level near that of intact PMNs. We conclude that O-2 and H2O2 are not sufficient to mediate target cell lysis, but require iron bound to LF, which, in turn, probably generates and focuses toxic O2 radicals, such as OH, to target membrane sites.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Ambruso D. R., Johnston R. B., Jr Lactoferrin enhances hydroxyl radical production by human neutrophils, neutrophil particulate fractions, and an enzymatic generating system. J Clin Invest. 1981 Feb;67(2):352–360. doi: 10.1172/JCI110042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Babior B. M., Kipnes R. S., Curnutte J. T. Biological defense mechanisms. The production by leukocytes of superoxide, a potential bactericidal agent. J Clin Invest. 1973 Mar;52(3):741–744. doi: 10.1172/JCI107236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baggiolini M., De Duve C., Masson P. L., Heremans J. F. Association of lactoferrin with specific granules in rabbit heterophil leukocytes. J Exp Med. 1970 Mar 1;131(3):559–570. doi: 10.1084/jem.131.3.559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boxer L. A., Haak R. A., Yang H. H., Wolach J. B., Whitcomb J. A., Butterick C. J., Baehner R. L. Membrane-bound lactoferrin alters the surface properties of polymorphonuclear leukocytes. J Clin Invest. 1982 Nov;70(5):1049–1057. doi: 10.1172/JCI110692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Breton-Gorius J., Mason D. Y., Buriot D., Vilde J. L., Griscelli C. Lactoferrin deficiency as a consequence of a lack of specific granules in neutrophils from a patient with recurrent infections. Detection by immunoperoxidase staining for lactoferrin and cytochemical electron microscopy. Am J Pathol. 1980 May;99(2):413–428. [PMC free article] [PubMed] [Google Scholar]
- Cohen G. The generation of hydroxyl radicals in biologic systems: toxicological aspects. Photochem Photobiol. 1978 Oct-Nov;28(4-5):669–675. doi: 10.1111/j.1751-1097.1978.tb06993.x. [DOI] [PubMed] [Google Scholar]
- Fehr J., Jacob H. S. In vitro granulocyte adherence and in vivo margination: two associated complement-dependent functions. Studies based on the acute neutropenia of filtration leukophoresis. J Exp Med. 1977 Sep 1;146(3):641–652. doi: 10.1084/jem.146.3.641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Graf E., Mahoney J. R., Bryant R. G., Eaton J. W. Iron-catalyzed hydroxyl radical formation. Stringent requirement for free iron coordination site. J Biol Chem. 1984 Mar 25;259(6):3620–3624. [PubMed] [Google Scholar]
- Grisham M. B., Jefferson M. M., Melton D. F., Thomas E. L. Chlorination of endogenous amines by isolated neutrophils. Ammonia-dependent bactericidal, cytotoxic, and cytolytic activities of the chloramines. J Biol Chem. 1984 Aug 25;259(16):10404–10413. [PubMed] [Google Scholar]
- Halliwell B., Gutteridge J. M. Oxygen toxicity, oxygen radicals, transition metals and disease. Biochem J. 1984 Apr 1;219(1):1–14. doi: 10.1042/bj2190001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harrison J. E., Schultz J. Studies on the chlorinating activity of myeloperoxidase. J Biol Chem. 1976 Mar 10;251(5):1371–1374. [PubMed] [Google Scholar]
- Hetherington S. V., Spitznagel J. K., Quie P. G. An enzyme-linked immunoassay (ELISA) for measurement of lactoferrin. J Immunol Methods. 1983 Dec 16;65(1-2):183–190. doi: 10.1016/0022-1759(83)90314-9. [DOI] [PubMed] [Google Scholar]
- Johnston R. B., Jr, Keele B. B., Jr, Misra H. P., Lehmeyer J. E., Webb L. S., Baehner R. L., RaJagopalan K. V. The role of superoxide anion generation in phagocytic bactericidal activity. Studies with normal and chronic granulomatous disease leukocytes. J Clin Invest. 1975 Jun;55(6):1357–1372. doi: 10.1172/JCI108055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klebanoff S. J., Clark R. A. Hemolysis and iodination of erythrocyte components by a myeloperoxidase-mediated system. Blood. 1975 May;45(5):699–707. [PubMed] [Google Scholar]
- Korchak H. M., Roos D., Giedd K. N., Wynkoop E. M., Vienne K., Rutherford L. E., Buyon J. P., Rich A. M., Weissmann G. Granulocytes without degranulation: neutrophil function in granule-depleted cytoplasts. Proc Natl Acad Sci U S A. 1983 Aug;80(16):4968–4972. doi: 10.1073/pnas.80.16.4968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Masson P. L., Heremans J. F., Schonne E. Lactoferrin, an iron-binding protein in neutrophilic leukocytes. J Exp Med. 1969 Sep 1;130(3):643–658. doi: 10.1084/jem.130.3.643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakajima K., Powers J. C., Ashe B. M., Zimmerman M. Mapping the extended substrate binding site of cathepsin G and human leukocyte elastase. Studies with peptide substrates related to the alpha 1-protease inhibitor reactive site. J Biol Chem. 1979 May 25;254(10):4027–4032. [PubMed] [Google Scholar]
- Nathan C. F., Silverstein S. C., Brukner L. H., Cohn Z. A. Extracellular cytolysis by activated macrophages and granulocytes. II. Hydrogen peroxide as a mediator of cytotoxicity. J Exp Med. 1979 Jan 1;149(1):100–113. doi: 10.1084/jem.149.1.100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roos D., Voetman A. A., Meerhof L. J. Functional activity of enucleated human polymorphonuclear leukocytes. J Cell Biol. 1983 Aug;97(2):368–377. doi: 10.1083/jcb.97.2.368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Root R. K., Metcalf J., Oshino N., Chance B. H2O2 release from human granulocytes during phagocytosis. I. Documentation, quantitation, and some regulating factors. J Clin Invest. 1975 May;55(5):945–955. doi: 10.1172/JCI108024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sinaceur J., Ribière C., Nordmann J., Nordmann R. Desferrioxamine: a scavenger of superoxide radicals? Biochem Pharmacol. 1984 May 15;33(10):1693–1694. doi: 10.1016/0006-2952(84)90296-x. [DOI] [PubMed] [Google Scholar]
- Slivka A., LoBuglio A. F., Weiss S. J. A potential role for hypochlorous acid in granulocyte-mediated tumor cell cytotoxicity. Blood. 1980 Feb;55(2):347–350. [PubMed] [Google Scholar]
- Weiss S. J., Klein R., Slivka A., Wei M. Chlorination of taurine by human neutrophils. Evidence for hypochlorous acid generation. J Clin Invest. 1982 Sep;70(3):598–607. doi: 10.1172/JCI110652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weiss S. J., LoBuglio A. F. An oxygen-dependent mechanism of neutrophil-mediated cytotoxicity. Blood. 1980 Jun;55(6):1020–1024. [PubMed] [Google Scholar]
- Weiss S. J. Neutrophil-mediated methemoglobin formation in the erythrocyte. The role of superoxide and hydrogen peroxide. J Biol Chem. 1982 Mar 25;257(6):2947–2953. [PubMed] [Google Scholar]
- Weiss S. J., Slivka A. Monocyte and granulocyte-mediated tumor cell destruction. A role for the hydrogen peroxide-myeloperoxidase-chloride system. J Clin Invest. 1982 Feb;69(2):255–262. doi: 10.1172/JCI110447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weiss S. J. The role of superoxide in the destruction of erythrocyte targets by human neutrophils. J Biol Chem. 1980 Oct 25;255(20):9912–9917. [PubMed] [Google Scholar]
- Winterbourn C. C. Lactoferrin-catalysed hydroxyl radical production. Additional requirement for a chelating agent. Biochem J. 1983 Jan 15;210(1):15–19. doi: 10.1042/bj2100015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Asbeck B. S., Marx J. J., Struyvenberg A., van Kats J. H., Verhoef J. Deferoxamine enhances phagocytic function of human polymorphonuclear leukocytes. Blood. 1984 Mar;63(3):714–720. [PubMed] [Google Scholar]